Reducing Friction and Wear of Tribological Systems through Hybrid Tribofilm Consisting of Coating and Lubricants
Abstract
:1. Introduction

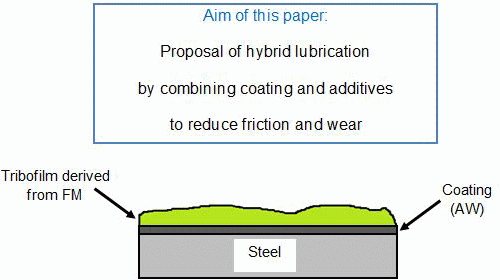
2. Hybrid Lubrication Concept
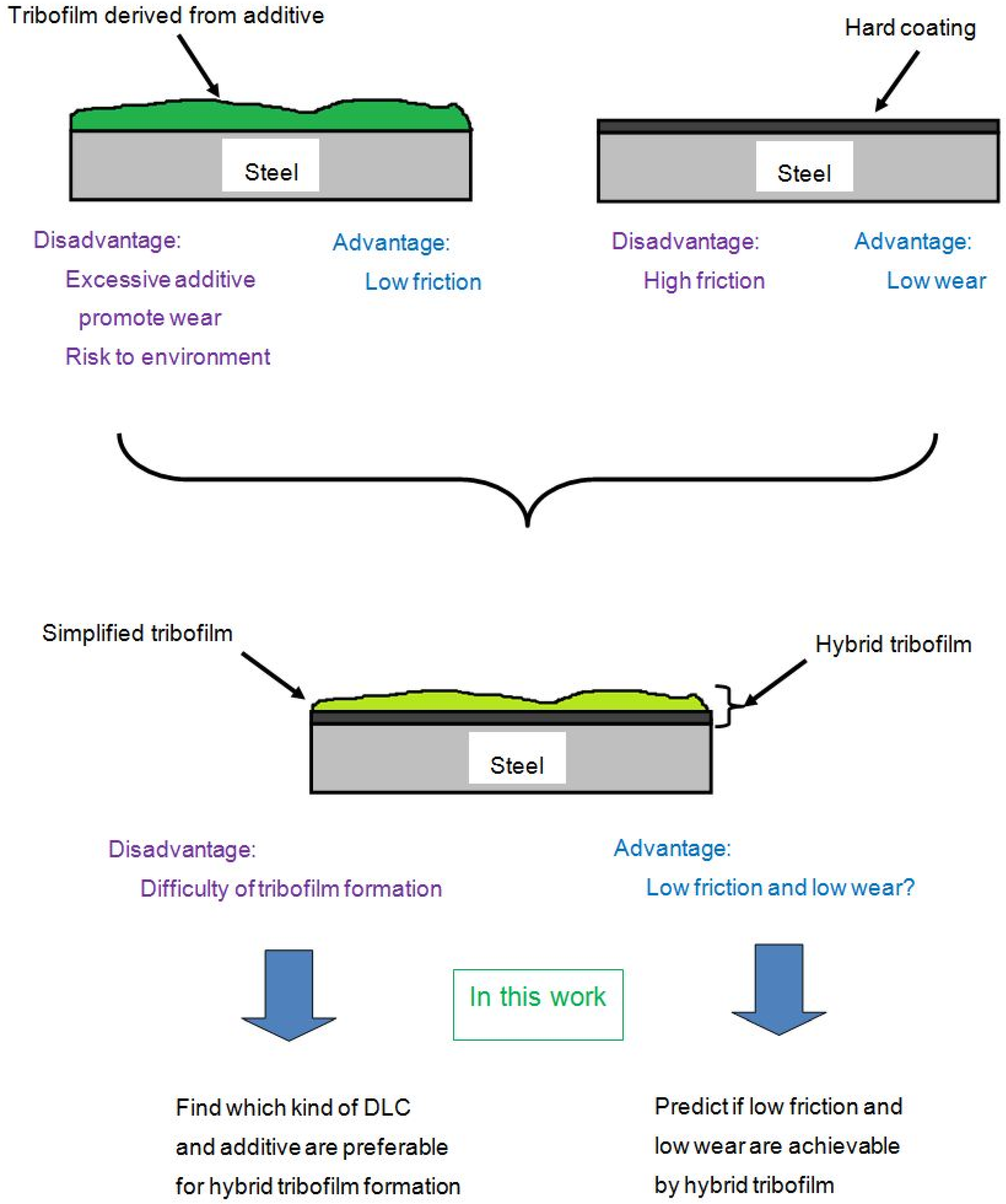
3. Reference Survey
3.1. Effect of DLC Coatings and Lubricants on Tribofilm Formation
| DLC coatings (See Nomenclature) | Lubricants (See Nomenclature) | Remarks about tribofilm | Analysis | Reference |
|---|---|---|---|---|
| a-C, a-C:H, a-C:H:W | API (American Petroleum Institute) Group III oil, ZnDTP | Formation of tribofilm = detected: The formation of ZnDTP-derived tribofilms on all DLC surfaces is clearly evident and has been demonstrated by means of SEM, EDX and AFM. However, ZnDTP pads form only on the 18% W-containing a-C:H:W coating, whereas for higher W% a-C:H:W coatings, graphitization appears to disrupt pad formation. | SEM, EDX, AFM | [20] |
| a-C, a-C:H, ta-C, DLC:W, DLC:WC, DLC:Si | API Group III oil, ZnDTP | Formation of tribofilm = detected: The formation of ZnDTP-derived tribofilms on all DLC surfaces is clearly evident and has been demonstrated by means of SEM, EDX, ToF-SIMS (Secondary Ion Mass Spectrometry) and AFM. However, ZnDTP pads form only on DLC:W. | SEM, EDX, ToF-SIMS, AFM | [21] |
| a-C, a-C:H, C:H:Ti | PAO, ZnDTP, MoDTC | Formation of tribofilm = detected: MoDTC and ZnDTP additives react directly on amorphous carbon surfaces. They seem to be more active with selected hydrogenated amorphous carbon surfaces. Performance of tribofilm = positive: The friction and wear performances are improved by coating both counter faces. Morphology of tribofilm: XPS analysis shows the formation of MoS2 in the contact area only. Additive interactions: On amorphous carbon surfaces, the role of the antiwear ZnDTP agent seems to be principally to enhance the formation of MoS2 sheets. Chemical property of tribofilm: The presence of zinc phosphate has been detected by EDX in the wear debris collected inside the tribofilm. The composition of tribofilm appears similar to that of tribofilm obtained on steel surfaces in the same lubrication conditions. Tribochemical reactions can occur without the presence of an iron catalyst element in the tribo-system. | XPS, EDX | [22] |
| ta-C | PAO, ZnDTP, GMO | Formation of tribofilm = detected: A white distributed ZnDTP-derived tribofilm was formed on ta-C surfaces in the PAO + ZnDTP lubricant. Performance of tribofilm = negative: Tribofilm does not provide wear protection. Additive interactions: FESEM and AFM analyses showed that ZnDTP-derived pad-like tribofilm did not form on the steel surface when lubricated with PAO + GMO + ZnDTP oil. It is believed that GMO may suppress the ZnDTP and, thus, prohibit the formation of ZnDTP tribofilm. | FESEM, AFM | [23] |
| a-C:H | PAO, ZnDTP | Formation of tribofilm = detected: The formation of a tribofilm by ZnDTP in PAO on both DLC and steel (self-mated) surfaces was demonstrated by means of SEM, EDX and ToF-SIMS. This showed that the film could be formed under very low wear conditions in the absence of iron. Morphology of tribofilm: It was seen from the AFM images that the tribofilm on the DLC surfaces had a different structure from that found on steel surfaces. Performance of tribofilm = negative: After washing the sample in an ultrasonic bath with cyclohexane, the tribofilm on DLC could no longer be observed. This indicates that the adhesion of the tribofilm to the DLC surface is much weaker than that of the tribofilm to steel. | SEM, EDX, ToF-SIMS, AFM | [24] |
| ta-C | PAO, ZnDTP, GMO | Formation of tribofilm = detected: Performance of tribofilm = negative: Relatively high friction is attributed to the formation of pad-like tribofilm, both on ta-C and steel surfaces for DLC/steel combination. No additional friction reduction or no ZnDTP-derived wear protective tribofilm formations are recorded under PAO + GMO + ZnDTP lubricated conditions for DLC/steel and DLC/DLC combinations. Performance of tribofilm = positive: On the other hand, formation of ZnDTP-derived thin white layer on ta-C DLC generates the lowest friction for DLC/DLC contact. | Fe-SEM, AFM | [25] |
| a-C:H | PAO, ZnDTP, b-TPPT, AP | Formation of tribofilm = detected: ZnDTP and b-TPPT showed a similar tribological behavior for steel/steel and DLC/DLC contacts. Both built up similar pad-like structures on steel and a tribofilm was also formed on DLC for the two cases, while no AP film was observed on DLC with AFM, although ToF-SIMS confirmed the presence of the physisorbed and chemisorbed additive on the surface. Performance of tribofilm = negative: The weaker adhesion of the ZnDTP tribofilm built up on DLC compared with that built up on steel was confirmed with scratch tests and can be explained by the inertness of the DLC coating preventing the reaction with the DLC surface. | AFM, ToF-SIMS | [26] |
| a-C:H, a-C:H:W, a-C:H:WC, ta-C, DLC:Si | API Group III oil, ZnDTP | Formation of tribofilm = detected: Chemical property of tribofilm: The use of ToF-SIMS to study pre-formed ZnDTP tribofilms before and after rubbing in base oil indicates that rubbing in base oil results in almost complete loss of sulfur species, both sulfide and thiophosphate from the film. Performance of tribofilm = negative: This suggests both that these materials are normally present in the outermost part of ZnDTP films and that they are relatively weakly bound and lost during rubbing. | AFM, ToF-SIMS | [27] |
| DLC | PAO, ZnDTP, MoDTC | Formation of tribofilm = detected: Chemical property of tribofilm: Zn phosphate was absorbed on DLC surface. MoO3 and MoS2 were not absorbed on the DLC surface. | XANES, XPS | [28] |
| a-C | PAO, ZnDTP, MoDTC, MT | Formation of tribofilm = detected: Additive interactions: The antiwear tribofilm composed of zinc phosphate was detected at the a-C/CI interface when ZnDTP was used alone. The performance of ZnDTP had to be compromised when it was used with MoDTC or Moly Trimer. | SEM, AFM, XPS | [29] |
| a-C:H | PAO, ZnDTP, MoDTC | Formation of tribofilm = detected: Chemical property of tribofilm: ZnDTP decomposed under boundary lubrication condition and formed Zn-phosphate/ZnS/ZnO anti-wear species in the tribofilms formed on a-C:H. | SEM, XPS | [30] |
| DLC:WC | PAO, ZnDTP, Alkyl thiadiazole | Formation of tribofilm = not detected: In the current study, ZnDTP did not form P-containing tribofilms on any of the fully-coated tested systems, indicating that the antiwear mechanism of this additive cannot be attributed to the formation of phosphate glass tribofilm. | XPS | [31] |
| DLC:H | PAO, ZnDTP, MoDTC | Formation of tribofilm = not detected: The appearance of C-1’s peak after 240 s indicated that no tribofilm was left, and the X-ray beam reached the surface of the pure DLC:H coating. | XPS | [32] |
| a-C:H | SAE 5W-30, 5W-20, ZnDTP, MoDTC | Formation of tribofilm = not detected: A tribofilm did not form on the DLC-coated disc due to the inactive chemical properties of DLC. | XPS | [33] |
| DLC | PAO, ZnDTP, MoDTC | Formation of tribofilm = not detected: The presence of an additive-derived tribofilm on the DLC surface, as evidenced by the XPS study, confirmed that the boundary lubrication between the cam and follower resulted in the decomposition of additives. | XPS | [34] |
| DLC coatings (See Nomenclature) | Lubricants (See Nomenclature) | Remarks about tribofilm formation | Analysis | Reference |
|---|---|---|---|---|
| a-C, a-C:H, C:H:Ti | PAO, ZnDTP, MoDTC | Formation of tribofilm = detected: MoDTC and ZnDTP additives react directly on amorphous carbon surfaces. They seem to be more active with selected hydrogenated amorphous carbon surfaces. Performance of tribofilm = positive: The friction and wear performances are improved by coating both counter faces. Morphology of tribofilm: XPS analysis shows the formation of MoS2 in the contact area only. Additive interactions: On amorphous carbon surfaces, the role of the antiwear ZnDTP agent seems to be principally to enhance the formation of MoS2 sheets. Chemical property of tribofilm: The presence of zinc phosphate has been detected by EDX in the wear debris embedded in the tribofilm. The composition of tribofilm appears similar to that of tribofilm obtained on steel surfaces in the same lubrication conditions. Tribochemical reactions can occur without the presence of an iron catalyst element in the tribo-system. | XPS, EDX | [22] |
| a-C:H, g-DLC | API Group II oil, ZnDTP, MoDTC, GMO, PFA, Amine | Formation of tribofilm = detected: MoDTC clearly forms a friction-reducing film on a-C:H. Morphology of tribofilm: This film takes the form of rows of tiny pads, a few nanometers high, each pad showing low lateral force. These pads are not oriented along the sliding direction, but follow the topographic morphology features of the coating, which are oriented perpendicular to the sliding direction. Chemical property of tribofilm: These low friction regions are characteristic of the friction response of MoDTC and are almost certainly nanocrystallites of MoS2 formed from MoDTC by a tribochemical reaction. Mechanism of formation of tribofilms Such pads are normally found to form only on asperity peaks, where their generation is believed to be stimulated by solid-solid rubbing. | AFM | [35] |
| a-C, a-C:H, a-C:H:W, a-C:H:WC, ta-C, ta-C:H, DLC:Si | API Group III oil, MoDTC | Formation of tribofilm = detected: The formation of MoDTC-derived tribofilms on all DLC surfaces was clearly evident and was demonstrated by means of SEM, EDX, ToF-SIMS and AFM. Performance of tribofilm = positive: With MoDTC, all DLCs showed reduced boundary friction, and in particular, W-containing DLCs showed low boundary friction, because of the formation of WS2 and Mo-derived tribolayers on the surface. | SEM, EDX, ToF-SIMS, AFM | [36] |
| a-C:H:W | PAO, GMO, ZnDTP, MoDTC | Formation of tribofilm = detected: Chemical property of tribofilm: Friction reduction is contributed by both formation of MoS2 and surface graphitization. The formation of MoS2 by chemical decomposition from MoDTC is dominant rather than the possible formation of WS2. | AFM, optical microscope | [37] |
| DLC | PAO, ZnDTP, MoDTC, T307 | Formation of tribofilm = detected: Performance of tribofilm = positive: The introduction of MoDTC into PAO-4 is beneficial for reducing the friction coefficients of DLC coatings; however, it simultaneously decreases the wear resistance of the coatings. The friction coefficients and wear rates of DLC coatings lubricated with PAO-4 can be reduced by the addition of ZnDTP in PAO-4. The addition of T307 in PAO-4 improves the wear resistance of DLC coatings; however, it has a negligible effect on the friction coefficients of the coatings. | SEM | [38] |
| DLC:W | PAO, MoDTC | Formation of tribofilm = detected: Chemical property of tribofilm: The improved tribological properties of the sulfurized DLC:W coating lubricated with MoDTC are mainly due to the formation of WSx and the higher ratio of Mo sulfide/Mo oxide and sp2/sp3 in the tribofilm. | SEM, XPS | [39] |
| a-C:H:CrC | MoDTC | Formation of tribofilm = detected: A tribolayer formed from the additives is only deposited in the loaded areas of the friction track and not in the unloaded environment of the coating. | SEM, EDX | [40] |
| DLC:H | PAO, ZnDTP, MoDTC | Formation of tribofilm = not detected: The appearance of C-1’s peak after 240 s indicated that no tribofilm was left and the X-ray beam reached the surface of the pure DLC:H coating. | XPS | [32] |
| a-C:H | SAE 5W-30, SAE 5W-20, ZnDTP, MoDTC | Formation of tribofilm = not detected: A tribofilm did not form on the DLC-coated disc, due to the inactive chemical properties of DLC. | XPS | [33] |
| DLC | PAO, ZnDTP, MoDTC | Formation of tribofilm = not detected: The presence of an additive-derived tribofilm on the DLC surface, as evidenced by the XPS study, confirmed that the boundary lubrication between cam and follower resulted in the decomposition of additives. | XPS | [34] |
| DLC:H | Two synthesized engine oil, MoDTC | Formation of tribofilm = not detected: It was remarkable that the molybdenum compounds were not observed on the DLC surface. | SEM, EDX | [41] |
| a-C:H | Fully formulated oil, ZnDTP, MoDTC | Formation of tribofilm = not detected: Formation of MoS2, which is responsible for providing lower friction, was mainly dominant in the tribofilm formed on the counterpart (cast iron pin) from high concentration Mo-FM-containing fully-formulated oils, while the amount of Mo formed on the DLC wear scar was negligible. | SEM, XPS | [42] |
| DLC coatings (See Nomenclature) | Lubricants (See Nomenclature) | Remarks about tribofilm formation | Analysis | Reference |
|---|---|---|---|---|
| a-C:H, ta-C | PAO, GMO | Formation of tribofilm = detected: The thickness of the tribofilm is lower than 2 nm, as indicated by SIMS profiling. | SIMS | [13] |
| DLC | PAO, GMO, BO, ODA | Formation of tribofilm = detected: The boundary film composed of GMO and PAO was detected by TOF-SIMS analysis. | TOF-SIMS | [14] |
| a-C:H, ta-C | PAO, GMO | Formation of tribofilm = not detected: The antiwear tribofilm grown on steel is very easy to observe, whereas on DLC coatings, such a film cannot be evidenced by optical microscopy. This means that either there is no growth of tribofilm on DLC or it is too thin to observe. | Optical microscopy | [43] |
| DLC coatings (See Nomenclature) | Lubricants (See Nomenclature) | Remarks about tribofilm formation | Analysis | Reference |
|---|---|---|---|---|
| DLC | PAO, calcium sulfonate | Formation of tribofilm = detected: A remarkable running-in process was observed at the steel-DLC contact, and it was not observed in the friction test with the “fresh steel ball/rubbed DLC disk”. It is concluded from the results that the film formation on the DLC disk occurred during the running-in process. | ToF-SIMS | [44] |
| a-C:H:WC | PAO, sulfurized olefin polysulfide | Formation of tribofilm = detected: In the presence of the S-based EP (extream pressure) additive, tungsten in the transferred layer reacts with sulfur in the additive, forming lamellar WS2 tribofilms on steel counter-face or the revealed steel substrate. Chemical property of tribofilm: These tribofilms have tribological properties similar to MoS2. Performance of tribofilm = positive: Depending on the additive concentration, the tribofilms may increase in thickness and density, thus providing conditions for low-friction behavior. | SEM | [45] |
| DLC | PAO, [BMP][FAP] | Formation of tribofilm = detected: During the friction tests, the high pressure and temperature conditions in the contact caused the reaction of the FAP-anion with the DLC surface to a high extent (up to 80%), forming a tribofilm. [BMP][FAP] also reacted with the surface outside the wear scar, forming a tribofilm. Performance of tribofilm = positive: This tribofilm significantly improved the antifriction behavior, even under severe loads. Chemical property of tribofilm: This tribofilm contained between 20% and 40% of the total fluorine atoms. | XPS | [46] |
| DLC:Cr | PAO, ZnDTP, [BMP][FAP], [(NEMM)MOE] [FAP] | Formation of tribofilm = detected: SEM images and XPS analysis indicated that the tribological improvement was due to additive-surface interactions, which promoted the formation of a tribofilm. | SEM, XPS | [47] |
| C:H:WC | PAO, Nitrogen sulfur, Amine phosphate, Phosphate ester, Sulfurized isobutylene | Formation of tribofilm = detected: Additive-WC-DLC interactions formed tribofilms composed of carbon- and sulfur-rich species and a lower concentration of oxygen, nitrogen and phosphorous compared to additive-steel interactions. | XPS | [48] |
| a-C:H:Ti | PFPE, 1-alkyl-3-octylimidazolium hexafluorophosphate (L-P801) | Formation of tribofilm = detected: XPS analyses indicated that tribochemical films and chemical adsorption films were formed on the worn surface of Ti-DLC coatings under L-P801 lubrication conditions. | SEM, XPS | [49] |
| DLC | PAO, [(NEMM)MOE] [FAP] | Formation of tribofilm = detected: Performance of tribofilm = negative: The ionic liquid does not react with the DLC surface unless there is an increase in the loading conditions. | SEM, XPS | [50] |
3.2. Formation of Tribofilm and Properties
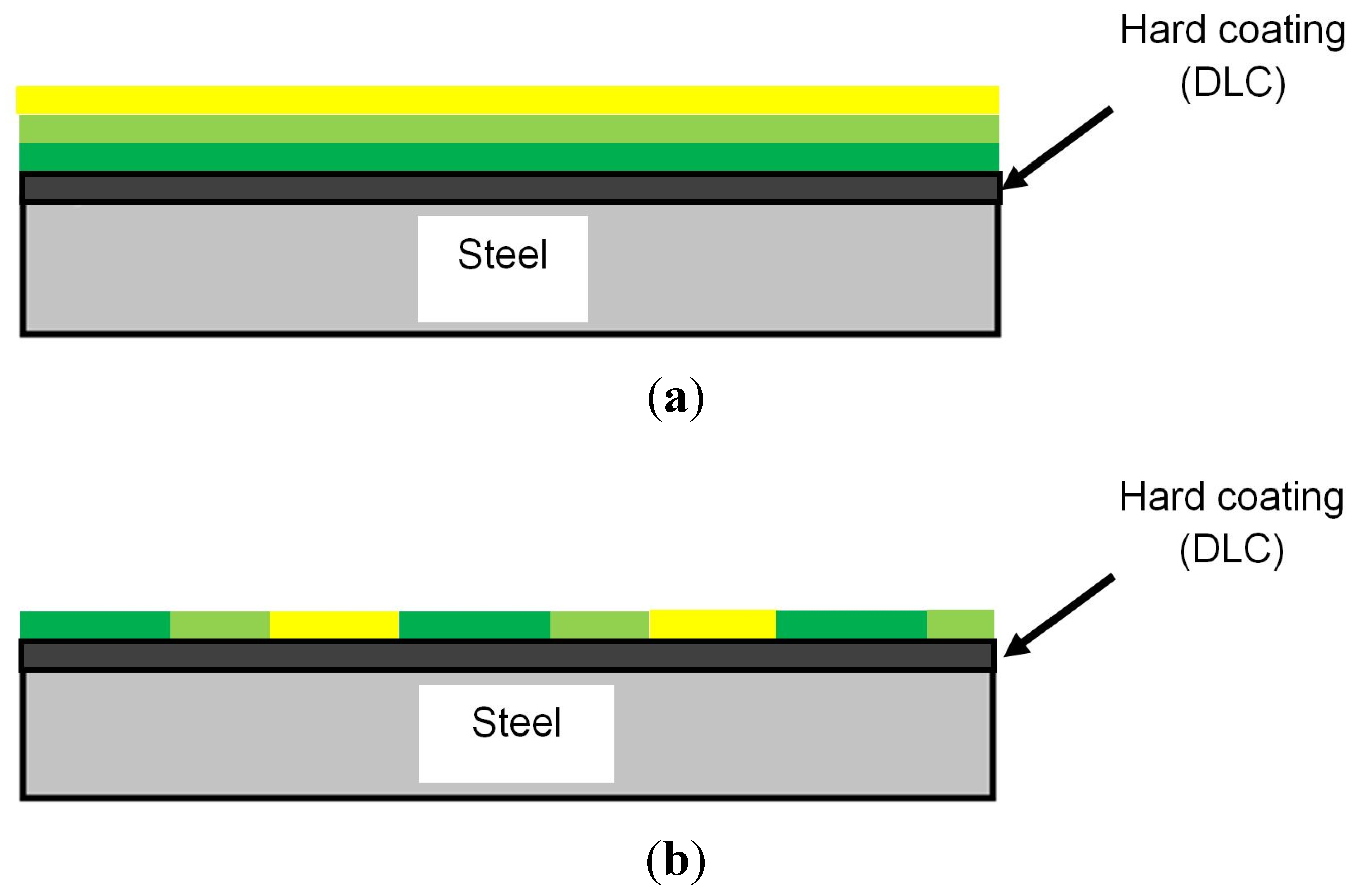
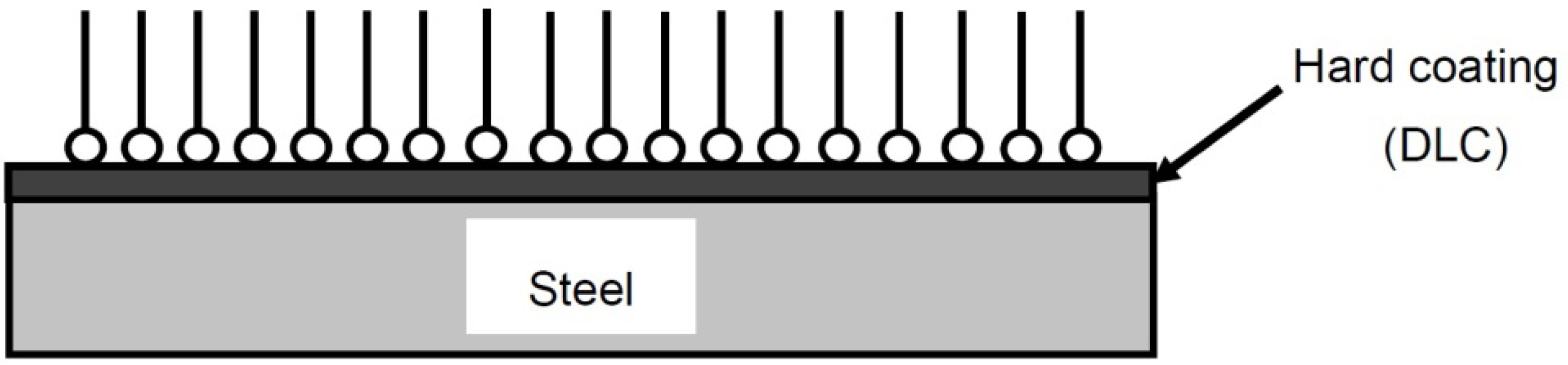
3.3. The Effect of Tribofilm Formation on the Friction Coefficient and Wear Rate
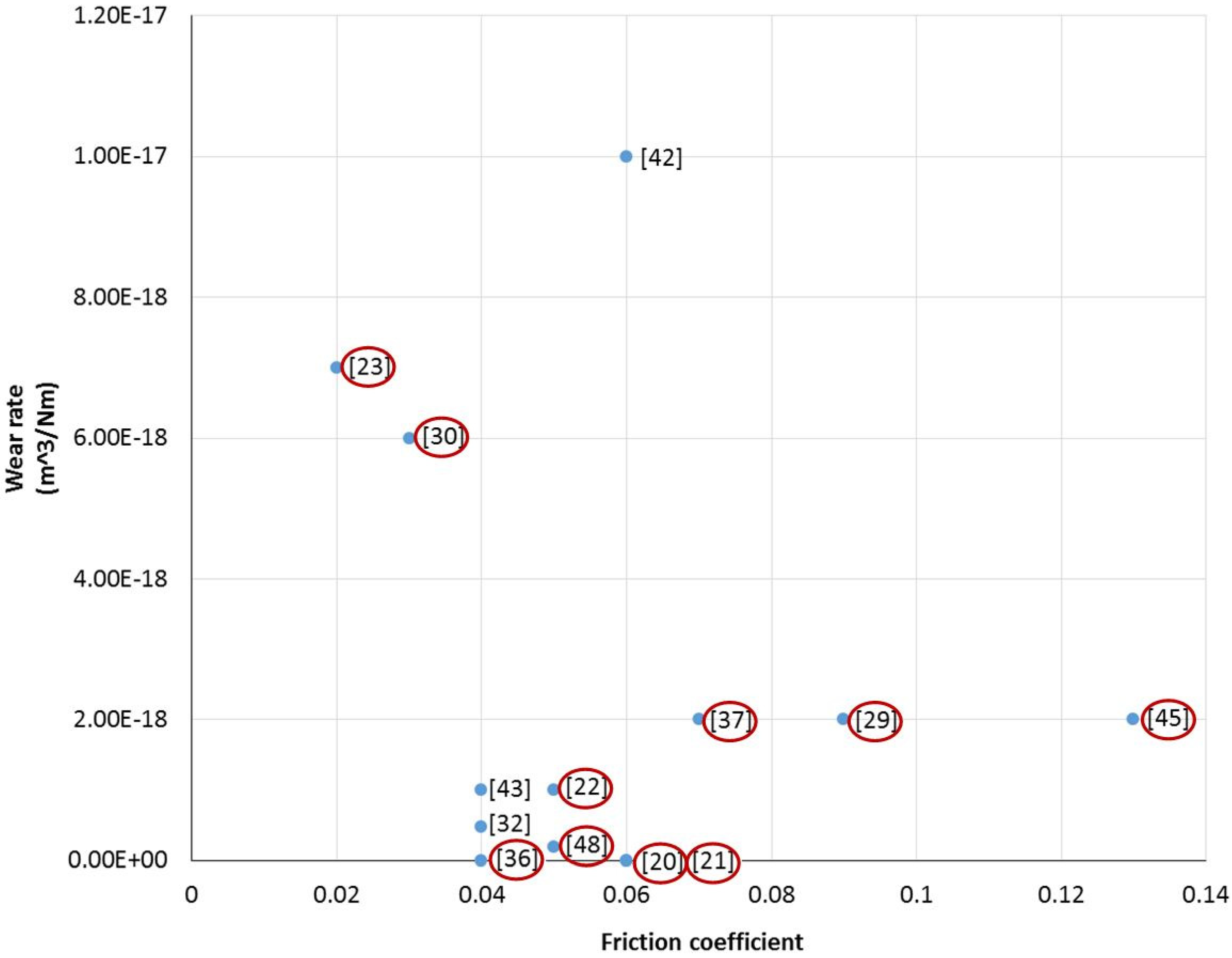
| DLC coatings (See Nomenclature) | Lubricants (See Nomenclature) | Tribo tester | Test conditions | Tribofilm formation | Reference |
|---|---|---|---|---|---|
| a-C, a-C:H, a-C:H:W | API Group III oil, ZnDTP | Friction tests: rotating, ball on disk | 1 GPa, 0.1 m/s, 100 °C | Detected | [20] |
| Wear tests: reciprocating, ball on disk | 1 GPa, 0.08 m/s, 100 °C | ||||
| a-C, a-C:H, ta-C, DLC:W, DLC:WC, DLC:Si | API Group III oil, ZnDTP | Friction tests: rotating, ball on disk | 0.95 GPa, 0.1 m/s, 100 °C | Detected | [21] |
| Wear tests: reciprocating, ball on disk | 0.95 GPa, 0.08 m/s, 100 °C | ||||
| a-C, a-C:H, C:H:Ti | PAO, ZnDTP, MoDTC | reciprocating, cylinder on disk | 0.6 GPa, 0.2 m/s, 100 °C | Detected | [22] |
| ta-C | PAO, ZnDTP | rotating, cylinder on disk | 0.15 GPa, 0.1 m/s, 80 °C | Detected | [23] |
| a-C | PAO, ZnDTP, MoDTC, MT | reciprocating, pin on disk | 0.7 GPa, 0.03 m/s, 100 °C | Detected | [29] |
| a-C:H | PAO, ZnDTP, MoDTC | reciprocating, pin on disk | 0.7 GPa, 0.02 m/s, 100 °C | Detected | [30] |
| DLC:H | PAO, ZnDTP, MoDTC | reciprocating, pin on disk | 0.6 GPa, 0.015 m/s, 100 °C | Not detected | [32] |
| a-C, a-C:H, a-C:H:W, a-C:H:WC, ta-C, ta-C:H, DLC:Si | API Group III oil, MoDTC | Friction tests: rotating, ball on disk | 1 GPa, 0.1 m/s, 100 °C | Detected | [36] |
| Wear tests: reciprocating, ball on disk | 1 GPa, 0.08 m/s, 100 °C | ||||
| a-C:H:W | PAO, GMO, ZnDTP, MoDTC | reciprocating, pin on disk | 0.7 GPa, 0.25 m/s, 100 °C | Detected | [37] |
| a-C:H | fully formulated oil, ZnDTP, MoDTC | reciprocating, pin on disk | 0.7 GPa, 0.02 m/s, 100 °C | Not detected | [42] |
| a-C:H, ta-C | PAO, GMO | reciprocating, ball on disk | 0.54 GPa, 0.035 m/s, 110 °C | Not detected | [43] |
| a-C:H:WC | PAO, sulfurized olefin polysulfide | reciprocating, ball on disk | 1.5 GPa, 0.02 m/s, 50 °C | Detected | [45] |
| H:WC | PAO, nitrogen sulfur, amine phosphate, phosphate ester, sulfurized isobutylene | rotating, ball on disk | 1.5 GPa, 1.02 m/s, 80 °C | Detected | [48] |
4. Conclusions
Nomenclature
| Abbreviation | Details |
| a-C | Hydrogen-free amorphous carbon DLC |
| a-C:H | Hydrogenated amorphous carbon DLC |
| a-C:H:CrC | Chromium carbide-doped hydrogenated amorphous carbon DLC |
| a-C:H:Ti | Titanium-doped hydrogenated amorphous carbon DLC |
| a-C:H:W | Tungsten-doped hydrogenated amorphous carbon DLC |
| a-C:H:WC | Tungsten carbide-doped hydrogenated amorphous carbon DLC |
| AP | Amine phosphate |
| API | American Petroleum Institute |
| AW | Anti-wear |
| [BMP][FAP] | 1-Butyl-1-methylpyrrolidinium tris(pentafluoroethyl)trifluorophosphate |
| BO | Butyl oleate |
| b-TPPT | Butylated triphenyl phosphorothionate |
| C:H:Ti | Titanium-doped DLC |
| C:H:WC | Tungsten carbide-doped hydrogenated DLC |
| DLC | Diamond-like carbon |
| DLC:Cr | Chrome-doped DLC |
| DLC:H | Hydrogenated DLC |
| DLC:Si | Silicon-doped DLC |
| DLC:W | Tungsten-doped DLC |
| DLC:WC | Tungsten carbide-doped DLC |
| EP | Extream pressure |
| FM | Friction modifier |
| g-DLC | Primarily sp2 and hydrogen-free DLC |
| GMO | Glycerol mono-oleate |
| MoDTC | Molybdenum dithio-carbonate |
| MT | Trinuclear molybdenum complex |
| [(NEMM)MOE][FAP] | Ethyl-dimethyl-2-methoxyethylammonium tris(pentafluoroethyl)trifluorophosphate |
| ODA | Octadecanoic acid |
| PAO | Poly alpha-olefin |
| PFA | Polycondensed fatty acid/ester |
| PFPE | Perfluoropolyethers |
| SIMS | Secondary Ion Mass Spectrometry |
| SAE | Society of Automotive Engineers |
| ta-C | Hydrogen-free tetrahedral amorphous carbon DLC |
| ta-C:H | Hydrogenated tetrahedral amorphous carbon DLC |
| ZnDTP | Zinc dithiophosphate |
Acknowledgments
Conflicts of Interest
References
- Jost, H.P. Tribology: How a word was coined 40 years ago. Tribol. Lubr. Technol. 2006, 3, 24–28. [Google Scholar]
- Jost, H.P. Tribology—Origin and future. Wear 1990, 136, 1–17. [Google Scholar]
- Czichos, H. Tribology: A Systems Approach to the Science and Technology of Friction, Lubrication and Wear; Elsevier Scientific Publishing Company: Amsterdam, The Netherlands, 1978; pp. 130–131. [Google Scholar]
- Roegiers, M.; Zhmud, B. Tribological performance of ionised vegetable oils as lubricity and fatty oiliness additives in lubricants and fuels. Lubr. Sci. 2009, 21, 169–182. [Google Scholar] [CrossRef]
- Zhmud, B. Developing energy-efficient lubricants and coatings for automotive applications. Tribol. Lubr. Technol. 2011, 9, 42–49. [Google Scholar]
- Spikes, H. The history and mechanisms of ZDDP. Tribol. Lett. 2004, 17, 469–489. [Google Scholar] [CrossRef]
- Nicholls, M.A.; Do, T.; Norton, P.R.; Kasrai, M.; Bancroft, G.M. Review of the lubrication of metallic surfaces by zinc dialkyl-dithiophosphates. Tribol. Int. 2005, 38, 15–39. [Google Scholar] [CrossRef]
- Minami, I.; Mori, H.; Katoh, A.; Marukane, Y.; Kohzu, Y. Proposal of Lubricant Maintenance by Monitoring Peroxide Value. J. Jpn. Pet. Inst. 2009, 52, 351–356. [Google Scholar] [CrossRef]
- Whitby, R.D. Competition for metal surfaces. Tribol. Lubr. Technol. 2013, 9, 88. [Google Scholar]
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford University Press: New York, NY, USA, 1998; p. 30. [Google Scholar]
- Bartz, W.J. Lubricants and the environment. Tribol. Int. 1998, 31, 35–47. [Google Scholar] [CrossRef]
- Spikes, H. Low- and zero-sulphated ash, phosphorus and sulphur anti-wear additives for engine oils. Lubr. Sci. 2008, 20, 103–136. [Google Scholar] [CrossRef]
- Kano, M.; Yasuda, Y.; Okamoto, Y.; Mabuchi, Y.; Hamada, T.; Ueno, T.; Ye, J.; Konishi, S.; Takeshima, S.; Martine, J.M.; et al. Ultralow friction of DLC in presence of glycerol mono-oleate (GMO). Tribol. Lett. 2005, 18, 245–251. [Google Scholar] [CrossRef]
- Minami, I.; Kubo, T.; Nanao, H.; Mori, S. Investigation of tribo-chemistry by means of stable isotopic tracers, Part 2: Lubrication mechanism of friction modifiers on diamond-like carbon. Tribol. Trans. 2007, 50, 477–487. [Google Scholar]
- Erdemir, A.; Donnet, C. Tribology of diamond-like carbon films: Recent progress and future prospects. J. Phys. D 2006, 39, 311–327. [Google Scholar] [CrossRef]
- Charitidis, C.A.; Koumoulos, E.P.; Dragatogiannis, D.A. Nanotribological Behavior of Carbon Based Thin Films: Friction and Lubricity Mechanisms at the Nanoscale. Lubricants 2013, 1, 22–47. [Google Scholar]
- Erdemir, A. Friction and wear of diamond and diamond-like carbon films. J. Eng. Tribol. 2002, 216, 387–400. [Google Scholar]
- Erdemir, A. Genesis of superlow friction and wear in diamondlike carbon films. Tribol. Int. 2004, 37, 1005–1012. [Google Scholar]
- Minami, I.; He, X.; Prakash, B. Coating-lubricant combination for improving tribo-system performance. Lubr. Sci. in press.
- Vengudusamy, B.; Green, J.H.; Lamb, G.D.; Spikes, H.A. Influence of hydrogen and tungsten concentration on the tribological properties of DLC/DLC contacts with ZDDP. Wear 2013, 298–299, 109–119. [Google Scholar] [CrossRef]
- Vengudusamy, B.; Green, J.H.; Lamb, G.D.; Spikes, H.A. Tribological properties of tribofilms formed from ZDDP in DLC/DLC and DLC/steel contacts. Tribol. Int. 2011, 44, 165–174. [Google Scholar] [CrossRef]
- De Barros’Bouchet, M.I.; Martin, J.M.; Le-Mogne, T.; Vacher, B. Boundary lubrication mechanisms of carbon coatings by MoDTC and ZDDP additives. Tribol. Int. 2005, 38, 257–264. [Google Scholar] [CrossRef]
- Tasdemir, H.A.; Wakayama, M.; Tokoroyama, T.; Kousaka, H.; Umehara, N.; Mabuchi, Y.; Higuchi, T. Wear behavior of tetrahedral amorphous diamond-like carbon (ta-C DLC) in additive containing lubricants. Wear 2013, 307, 1–9. [Google Scholar] [CrossRef]
- Equey, S.; Roos, S.; Mueller, U.; Hauert, R.; Spencer, N.D.; Crockett, R. Tribofilm formation from ZnDTP on diamond-like carbon. Wear 2008, 264, 316–321. [Google Scholar] [CrossRef]
- Tasdemir, H.A.; Wakayama, M.; Tokoroyama, T.; Kousaka, H.; Umehara, N.; Mabuchi, Y.; Higuchi, T. Ultra-low friction of tetrahedral amorphous diamond-like carbon (ta-C DLC) under boundary lubrication in polyalpha-olefin (PAO) with additives. Tribol. Int. 2013, 65, 286–294. [Google Scholar]
- Equey, S.; Roos, S.; Mueller, U.; Hauert, R.; Spencer, N.D.; Crockett, R. Reactions of zinc-free anti-wear additives in DLC/DLC and steel/steel contacts. Tribol. Int. 2008, 41, 1090–1096. [Google Scholar]
- Vengudusamy, B.; Green, J.H.; Lamb, G.D.; Spikes, H.A. Durability of ZDDP Tribofilms Formed in DLC/DLC Contacts. Tribol. Lett. 2013, 51, 469–478. [Google Scholar] [CrossRef]
- Morina, A.; Zhao, H.; Mosselmans, J.F.W. In-situ reflection-XANES study of ZDDP and MoDTC lubricant films formed on steel and diamond like carbon (DLC) surfaces. Appl. Surf. Sci. 2014, 297, 167–175. [Google Scholar] [CrossRef]
- Haque, T.; Morina, A.; Neville, A. Influence of friction modifier and antiwear additives on the tribological performance of a non-hydrogenated DLC coating. Surf. Coat. Technol. 2010, 204, 4001–4011. [Google Scholar] [CrossRef]
- Kosarieh, S.; Morina, A.; Lainé, E.; Flemming, J.; Neville, A. Tribological performance and tribochemical processes in a DLC/steel system when lubricated in a fully formulated oil and base oil. Surf. Coat. Technol. 2013, 217, 1–12. [Google Scholar] [CrossRef]
- Mistry, K.K.; Morina, A.; Neville, A. A tribochemical evaluation of a WC-DLC coating in EP lubrication conditions. Wear 2011, 271, 1739–1744. [Google Scholar] [CrossRef]
- Haque, T.; Morina, A.; Neville, A.; Kapadia, R.; Arrowsmith, S. Effect of oil additives on the durability of hydrogenated DLC coating under boundary lubrication conditions. Wear 2009, 266, 147–157. [Google Scholar] [CrossRef]
- Kano, M.; Yasuda, Y. The Effect of ZDDP and MoDTC additives in engine oil on the friction properties of DLC-coated and steel cam followers. Lubr. Sci. 2004, 17, 95–103. [Google Scholar] [CrossRef]
- Haque, T.; Morina, A.; Neville, A. Tribological performance evaluation of a hydrogenated diamond-like carbon coating in sliding/rolling contact—Effect of lubricant additives. J. Eng. Tribol. 2011, 225, 393–405. [Google Scholar]
- Topolovec-Miklozic, K.; Lockwood, F.; Spikes, H. Behaviour of boundary lubricating additives on DLC coatings. Wear 2008, 265, 1893–1901. [Google Scholar] [CrossRef]
- Vengudusamy, B.; Green, J.H.; Lamb, G.D.; Spikes, H.A. Behaviour of MoDTC in DLC/DLC and DLC/steel contacts. Tribol. Int. 2012, 54, 68–76. [Google Scholar] [CrossRef]
- Yang, L.; Neville, A.; Brown, A.; Ransom, P.; Morina, A. Friction reduction mechanisms in boundary lubricated W-doped DLC coatings. Tribol. Int. 2014, 70, 26–33. [Google Scholar] [CrossRef]
- Zhiqiang, F.; Jian, S.; Chengbiao, W.; Wei, Z.; Wen, Y.; Zhijian, P.; Xiang, Y.; Songsheng, L.; Mingjiang, D. Tribological performance of DLC coatings deposited by ion beam deposition under dry friction and oil lubricated conditions. Vacuum 2013, 94, 14–18. [Google Scholar] [CrossRef]
- Yue, W.; Liu, C.; Fu, Z.; Wang, C.; Huang, H.; Liu, J. Synergistic effects between sulfurized W-DLC coating and MoDTC lubricating additive for improvement of tribological performance. Tribol. Int. 2013, 62, 117–123. [Google Scholar] [CrossRef]
- Keunecke, M.; Bewilogua, K.; Becker, J.; Gies, A.; Grischke, M. CrC/a-C:H coatings for highly loaded, low friction applications under formulated oil lubrication. Surf. Coat. Technol. 2012, 207, 270–278. [Google Scholar] [CrossRef]
- Sugimoto, I.; Honda, F.; Inoue, K. Analysis of wear behavior and graphitization of hydrogenated DLC under boundary lubricant with MoDTC. Wear 2013, 305, 124–128. [Google Scholar] [CrossRef]
- Kosarieh, S.; Morina, A.; Laine, E.; Flemming, J.; Neville, A. The effect of MoDTC-type friction modifier on the wear performance of a hydrogenated DLC coating. Wear 2013, 302, 890–898. [Google Scholar] [CrossRef]
- Héau, C.; Ould, C.; Maurin-Perrier, P. Tribological behaviour analysis of hydrogenated and nonhydrogenated DLC lubricated by oils with and without additives. Lubr. Sci. 2013, 25, 275–285. [Google Scholar] [CrossRef]
- Kubo, T.; Fujiwara, S.; Nanao, H.; Minami, I.; Mori, S. Boundary film formation from overbased calcium sulfonate additives during running-in process of steel-DLC contact. Wear 2008, 265, 461–467. [Google Scholar] [CrossRef]
- Podgornik, B.; Hren, D.; Vizintin, J.; Jacobson, S.; Stavlid, N.; Hogmark, S. Combination of DLC coatings and EP additives for improved tribological behaviour of boundary lubricated surfaces. Wear 2006, 261, 32–40. [Google Scholar] [CrossRef]
- González, R.; Battez, A.H.; Blanco, D.; Viesca, J.L.; Fernández-González, A. Lubrication of TiN, CrN and DLC PVD Coatings with 1-butyl-1-methylpyrrolidinium tris(pentafluoroethyl)trifluorophosphate. Tribol. Lett. 2010, 40, 269–277. [Google Scholar] [CrossRef]
- González, R.; Battez, A.H.; Viesca, J.L.; Higuera-Garrido, A.; Fernández-gonzá lez, A. Lubrication of DLC Coatings with Two Tris(pentafluoroethyl)trifluorophosphate Anion-Based Ionic Liquids. Tribol. Trans. 2013, 56, 887–895. [Google Scholar] [CrossRef]
- Mistry, K.K.; Morina, A.; Erdemir, A.; Neville, A. Extreme pressure lubricant additives interacting on the surface of steel- and tungsten carbide-doped diamond-like carbon. Tribol. Trans. 2013, 56, 623–629. [Google Scholar] [CrossRef]
- Feng, X.; Xia, Y. Tribological properties of Ti-doped DLC coatings under ionic liquids lubricated conditions. Appl. Surf. Sci. 2012, 258, 2433–2438. [Google Scholar] [CrossRef]
- Battez, A.H.; González, R.; Viesca, J.L.; Fernández-González, A.; Hadfield, M. Lubrication of PVD coatings with ethyl-dimethyl-2-methoxyethylammonium tris(pentafluoroethyl)trifluorophosphate. Tribol. Int. 2013, 58, 71–78. [Google Scholar] [CrossRef]
- Minami, I. Ionic liquids in tribology. Molecules 2009, 14, 2286–2305. [Google Scholar] [CrossRef]
- Bermúdez, M.; Jiménez, A.; Sanes, J.; Carrión, F. Ionic Liquids as Advanced Lubricant Fluids. Molecules 2009, 14, 2888–2908. [Google Scholar] [CrossRef]
- Zhou, F.; Liang, Y.; Liu, W. Ionic liquid lubricants: Designed chemistry for engineering applications. Chem. Soc. Rev. 2009, 38, 2590–2599. [Google Scholar] [CrossRef]
- Palacio, M.; Bhushan, B. A Review of Ionic Liquids for Green Molecular Lubrication in Nanotechnology. Tribol. Lett. 2010, 40, 247–268. [Google Scholar] [CrossRef]
- Somers, A.E.; Howlett, P.C.; MacFarlane, D.R.; Forsyth, M. A Review of Ionic Liquid Lubricants. Lubricants 2013, 1, 3–21. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yazawa, S.; Minami, I.; Prakash, B. Reducing Friction and Wear of Tribological Systems through Hybrid Tribofilm Consisting of Coating and Lubricants. Lubricants 2014, 2, 90-112. https://doi.org/10.3390/lubricants2020090
Yazawa S, Minami I, Prakash B. Reducing Friction and Wear of Tribological Systems through Hybrid Tribofilm Consisting of Coating and Lubricants. Lubricants. 2014; 2(2):90-112. https://doi.org/10.3390/lubricants2020090
Chicago/Turabian StyleYazawa, Shuichiro, Ichiro Minami, and Braham Prakash. 2014. "Reducing Friction and Wear of Tribological Systems through Hybrid Tribofilm Consisting of Coating and Lubricants" Lubricants 2, no. 2: 90-112. https://doi.org/10.3390/lubricants2020090