A Review of Ionic Liquid Lubricants
Abstract
:1. Introduction
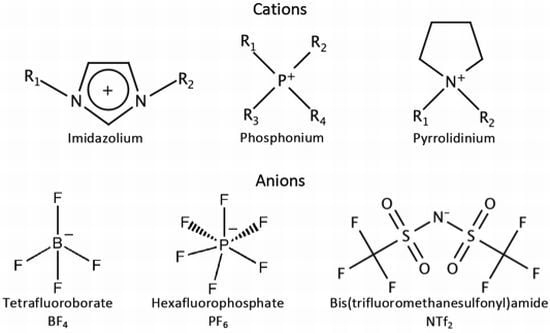
2. Ionic Liquids

2.1. Anions
2.2. Cations
3. Overview of the Application of ILs in Different Material Combinations
| Facility | State Key Laboratory, Lanzhou, China [35] | Polytechnic University of Cartagena, Spain [31] | Oak Ridge National Laboratory, TN, USA [15] |
|---|---|---|---|
| Wear Test Type | Optimol SRV Pin-on-flat Reciprocating | Microtest Pin-on-disk Rotating | Phoenix Ltd. Pin-on-disk Reciprocating and rotating |
| Ball Diameter and Type | 10 mm ASTM 52100 | 0.8 mm ASTM 52100 | 9.525 mm ASTM 52100 |
| Load | 50–300 N | 2.45–4.45 N | 38.3 and 100 N |
| Velocity | 1 mm stroke, 25 Hz, 0.1 m/s | 0.06–0.20 m/s | 0.02–1.0 m/s |
| Distance | 180 m | 850–2500 m | 400–500 m |
| Aluminium alloy | 2024 | 2011 | 6061, 1100, 319 |
| Lubricant amount | 2 drops | 2 mL | 5 drops |
| Standard | Phosphazene (X1-P) | Mineral Oil Propylene glycol dioleate (PGDO) | Mineral oil 15W40 diesel engine oil |
| Ionic Liquid | Viscosity 100 °C (mm2s−1) | Room T Wear Test | Ionic Liquid | Viscosity 100 °C (mm2s−1) | Room T Wear Test | ||
|---|---|---|---|---|---|---|---|
| Friction | Wear (mm3m−1) | Friction | Wear (mm3m−1) | ||||
| Ye et al., 2001. Reciprocating 10 mm ball 50 N [6]. | Zhang et al., 2009. Reciprocating 10 mm ball 100 N [23]. | ||||||
| X-1P | n/m | 0.065 | 12 × 10−7 | X-1P | 11.2 | 0.118 | 0.29 |
| Perfluoropolyether (PFPE) | n/m | 0.110 | 11 × 10−7 | C2mIm DEP | 16.6 | 0.076 | 0.21 |
| C6C2Im BF4 | n/m | 0.055 | 1.2 × 10−7 | C4mIm DEP | 19.3 | 0.074 | 0.17 |
| C8mIm DEP | 22.9 | 0.072 | 0.07 | ||||
| Liu et al., 2002. Four ball test [18]. | C6mIm BF4 | 10.9 | 0.096 | 0.24 | |||
| Mineral oil + 1% ZDDP | n/m | 0.085 | 0.51 mm | ||||
| C6C2Im BF4 | 11.8 | 0.045 | 0.54 mm | Minami et al., 2010. Reciprocating 6.35 mm ball 20 N [22]. | |||
| C8C2Im BF4 | 15.5 | 0.043 | 0.47 mm | C4mIm NTf2 | 38.5 a | 0.112 | 0.265 mm |
| P2,2,2,5 NTf2 | 66.7 a | 0.089 | 0.254 mm | ||||
| Wang et al., 2004. Reciprocating 10 mm ball 1000 N [19]. | P2,2,2,8 NTf2 | 102.4 a | 0.096 | 0.188 mm | |||
| C6mIm PF6 | 15.3 | 0.062 | 9.2 × 10−7 | P2,2,2,12 NTf2 | 148.8 a | 0.089 | 0.215 mm |
| C8C2Im PF6 | 17.7 | 0.065 | 5.3 × 10−7 | P2,2,2,(C2H5C6H5) NTf2 | 173.4 a | 0.091 | 0.192 mm |
| C8C3Im PF6 | 19.8 | 0.065 | 4.4 × 10−7 | P2,2,2,(C2H4OCH3) NTf2 | 24.6 a | 0.119 | 0.262 mm |
| P1,4,4,4 NTf2 | 161.7 a | 0.079 | 0.215 mm | ||||
| Lu et al., 2004. Reciprocating 10 mm ball 800 N [20]. | P4,4,4,8 NTf2 | 211.9 a | 0.092 | 0.185 mm | |||
| C6C2Im NTf2 | 5.5 | 0.075 | 3.6 × 10−7 | P4,4,4,12 NTf2 | 268.1 a | 0.096 | 0.215 mm |
| C6C2Im BF4 | 11.8 | 0.045 | 4.3 × 10−7 | P1,4,4,4 DMP | 570.5 a | 0.084 | 0.162 mm |
| P1,4,4,4 DEPD | 1176.5 a | 0.076 | 0.154 mm | ||||
| Weng et al., 2007. Reciprocating 10 mm ball 800 N [21]. | |||||||
| C6C2Im PF6 | n/m | 0.058 | 2.2 × 10−6 | Jiang et al., 2011. Reciprocating 10 mm ball 500 N [25]. | |||
| P4,4,4,6 BF4 | n/m | 0.050 | 1.3 × 10−6 | PFPE | 23.9 | 0.130 | 10.4 × 10−6 |
| P4,4,4,10 BF4 | n/m | 0.048 | 1.1 × 10−6 | Diimidazolium1,1 BEHP | 100.3 | 0.102 | 3.6 × 10−6 |
| Diimidazolium3,3 BEHP | 38.2 | 0.102 | 3.0 × 10−6 | ||||
| Minami et al., 2008. Reciprocating 6.35 mm ball 60 N [24] | Zhu et al., 2012. Reciprocating 10 mm ball 400 N [26]. | ||||||
| C2mIm FAP | 4.7 | 0.089 | 0.31 mm | PFPE | n/m | 0.122 | 27.7 × 10−7 |
| C6mIm FAP | 5.4 | 0.098 | 0.21 mm | C4mIm BF4 | 7.25 | 0.077 | 6.9 × 10−7 |
| C18mIm FAP | n/m | 0.079 | 0.21 mm | EstermIm BF4 | 26.98 | 0.076 | 4.1 × 10−7 |
| C2mIm NTf2 | 3.8 | 0.260 | 0.32 mm | EstermIm NTf2 | 8.13 | 0.082 | 3.0 × 10−7 |
| EsterC4Im NTf2 | 11.09 | 0.077 | 2.7 × 10−7 | ||||
| Ionic Liquid | Viscosity (mm2s−1) | Room T Wear Test | Ionic Liquid | Viscosity (mm2s−1) | Room T Wear Test | ||
|---|---|---|---|---|---|---|---|
| Friction | Wear (mm3m−1) | Friction | Wear (mm3m−1) | ||||
| Chen et al., 2003. Reciprocating ball on disc 10 mm ball 80 N [28]. | Mu et al., 2008. Reciprocating ball on disc 10 mm ball 400 N [35]. | ||||||
| C6C2Im BF4 | 11.8 a | 0.05 | 6.2 × 10−5 | X-1P | n/m | 0.160 | 9.4 × 10−5 |
| Phosphonylimidazolium3,4 BF4 | 540.1 b | 0.058 | 7.2 × 10−5 | ||||
| Mu et al., 2005. Reciprocating ball on disc 10 mm ball 100 N [32]. | Phosphonylimidazolium3,6 BF4 | 571.9 b | 0.060 | 5.5 × 10−5 | |||
| X-1P | n/m | 0.129 | 8.2 × 10−5 | Phosphonylimidazolium3,8 BF4 | 447.3 b | 0.062 | 4.4 × 10−5 |
| Phosphonylimidazolium3,6 BF4 | 571.9 b | 0.063 | 1.3 × 10−5 | C6C2Im BF4 | 88.0 b | 0.104 | 14.4 × 10−5 |
| Phosphonylimidazolium3,8 BF4 | 447.3 b | 0.056 | 0.8 × 10−5 | ||||
| Phosphonylimidazolium3,6 PF6 | 759.7 b | 0.070 | 1.3 × 10−5 | Somers et al., 2010. Rotating ball on disc 6 mm ball 30 N [36]. | |||
| Phosphonylimidazolium3,8 PF6 | 690.9 b | 0.070 | 0.2 × 10−5 | 15W-50 Oil | 130 b,d | 0.11 | 37.5 × 10−5 |
| C6C2Im BF4 | 88.0 b | 0.067 | 6.2 × 10−5 | P6,6,6,14 DBP | 130 b,d | 0.17 | 201 × 10−5 |
| C8C3Im PF6 | 258.0 b | 0.053 | 1.5 × 10−5 | P6,6,6,14 (iC8)2PO2 | 120 b,d | 0.085 | 21.0 × 10−5 |
| P6,6,6,14 BEHP | 260 b,d | 0.085 | 13.5 × 10−5 | ||||
| Liu et al., 2006. Reciprocating ball on disc 10 mm ball 300 N [33]. | P6,6,6,14 DPP | 210 b,d | 0.080 | 1.2 × 10−5 | |||
| P4,4,4,8 BF4 | 33.35 a | 0.046 | 0.8 × 10−4 | P6,6,6,14 NTf2 | 140 b,d | 0.070 | 4.2 × 10−5 |
| P4,4,4,10 BF4 | n/m | 0.045 | 1.0 × 10−4 | P6,6,6,14 Br | 610 b,d | 0.065 | 3.0 × 10−5 |
| P4,4,4,12 BF4 | n/m | 0.045 | 0.7 × 10−4 | ||||
| C6C2Im PF6 | 14.35 a | 0.060 | 2.4 × 10−4 | Shah et al., 2011. Rotating ball on disc 6 mm ball 40 N [39]. | |||
| 15W-50 Oil | n/m | 0.102 | 16.6 × 10−5 | ||||
| Jiminez et al., 2006. Rotating ball on disc 0.8 mm ball 2.45 N [29]. | P6,6,6,14 BMB | 940 b | 0.067 | 1.8 × 10−5 | |||
| C2mIm BF4 | 22.1 a,d | 0.284 | 662 × 10−5 | P6,6,6,14 BScB | 350 b | 0.085 | 7.8 × 10−5 |
| C6mIm BF4 | 19.8 a,d | 0.115 | 1.54 × 10−5 | P6,6,6,14 BOB | 1130 b | 0.063 | 5.4 × 10−5 |
| C8mIm BF4 | 18.7 a,d | 0.021 | 1.09 × 10−5 | P6,6,6,14 BMLB | 280 b | 0.068 | 2.1 × 10−5 |
| C2mIm CF3SO3 | 15.6 a,d | 0.103 | 169 × 10−5 | ||||
| C2mIm CH3C6H4SO3 | 14.6 a,d | 0.101 | 77.3 × 10−5 | Somers et al., 2012. Rotating ball on disc 6 mm ball 40N [38]. | |||
| C4mIm4 NTf2 | 14.5 a,d | 0.217 | 283 × 10−5 | Mineral Oil | 80 c,d | 0.110 | 43.2 × 10−5 |
| C6mIm PF6 | 18.7 a,d | 0.163 | 147 × 10−5 | P6,6,6,14 NTf2 | 315 c,d | 0.060 | 2.2 × 10−5 |
| P6,6,6,14 FAP | 360 c,d | 0.075 | 2.1 × 10−5 | ||||
| Qu et al., 2006. Reciprocating ball on disc 9.525 mm ball 38.3 N [7]. | P6,6,6,14 DPP | 785 c,d | 0.070 | 2.1 × 10−5 | |||
| Mineral Oil | 7.3 a | 0.10 | 16.0 × 10−3 | P1,4,4,4 DPP | 340 c,d | 0.079 | 1.7 × 10−5 |
| 15W-40 Oil | 13.1 a | 0.09 | 12.5 × 10−3 | C4mPyr NTf2 | 100 c,d | 0.107 | 49.5 × 10−5 |
| N8,8,8H NTf2 | 11.0 a | 0.06 | 18.0 × 10−3 | C4mPyr FAP | 220 c,d | 0.101 | 2.2 × 10−5 |
| C3mPyr NTf2 | 80 c,d | 0.108 | 99.6 × 10−5 | ||||
| C2mIm FAP | 80 c,d | 0.137 | 798 × 10−5 | ||||
| C8mIm BF4 | 345 b,c,d | 0.063 | 3.3 × 10−5 | ||||
Thin Film Lubrication on Silicon Substrates
4. Surface Characterisation
5. Fundamental Studies of Confined IL

6. Ionic Liquids as Additives
| Ionic Liquid | Soluble | Viscosity | Room Temp Wear Test | |
|---|---|---|---|---|
| 100 °C (mm2s−1) | Friction | Wear (mm3m−1) | ||
| Jiminez et al., 2006. 1 wt% IL in mineral oil. Steel on aluminium [30]. | ||||
| 1% C2mIm BF4 | - | 22.1 a | 0.08 | 8.5 × 10−5 |
| 1% C6mIm BF4 | - | 19.8 a | 0.13 | 4.3 × 10−5 |
| 1% C8mIm BF4 | - | 18.7 a | 0.11 | 20 × 10−5 |
| 1% C6mIm PF6 | - | 18.7 a | 0.10 | 13 × 10−5 |
| 1% C2mIm Triflate | - | 15.6 a | 0.13 | 17 × 10−5 |
| 1% C2mIm Tosylate | - | 14.6 a | 0.10 | 2.8 × 10−5 |
| 1% C4mPyr NTf2 | - | 14.5 a | 0.13 | 18 × 10−5 |
| Qu et al., 2006. 10 wt% IL in mineral oil. Steel on aluminium [7]. | ||||
| Mineral Oil | n/a | 7.3 | 0.10 | 3.2 × 10−5 |
| 15W40 oil | n/a | 13.1 | 0.09 | 2.5 × 10−5 |
| N6,6,6,H NTf2 | No | 10.4 | 0.05 | 3.7 × 10−5 |
| 10% N6,6,6,H NTf2 | No | n/m | 0.10 | 2.8 × 10−5 |
| Jiminez et al.,2008. 1 wt% IL in propylene glycol dioleate (PGDO). Steel on aluminium [31]. | ||||
| PGDO | n/a | 9.3 a | 0.05 | 8.2 × 10−5 |
| 1% C2mIm BF4 | No | 8.8 a | 0.08 | 5.6 × 10−5 |
| 1% C6mIm BF4 | No | 8.2 a | 0.08 | 13.0 × 10−5 |
| 1% C2mIm Triflate | No | 9.2 a | 0.07 | 5.4 × 10−5 |
| 1% C6mIm PF6 | No | 8.5 a | 0.03 | 9.8 × 10−5 |
| 1% P6,6,6,14 NTf2 | Yes | n/m | 0.14 | 107.6 × 10−5 |
| Mistry et al., 2008. 1 wt% IL in group III mineral oil. Cr coated steel on Ni–SiC coated Al [43]. | ||||
| Mineral oil | n/a | n/m | 0.078 | 1.9 × 10−7 b |
| 1% C6mIm BF4 | Yes | n/m | 0.028 | 2.9 × 10−7 b |
| 1% Tricresylphosphate(TCP) | Yes | n/m | 0.030 | 0.5 × 10−7 b |
| 1% IL + 1% TCP | Yes | n/m | 0.015 | 1.8 × 10−7 b |
| Cai et al., 2010. 1 wt% IL in poly (ethylene glycol) (PEG). Steel on steel [17]. | ||||
| PEG | n/a | 4.20 | 0.16 | 280 × 10−7 |
| 1% BHTmIm PF6 | Yes | 4.35 | 0.11 | 2.5 × 10−7 |
| 1% BHTmIm BF4 | Yes | 4.33 | 0.14 | 37 × 10−7 |
| 1% BHTmIm NTf2 | Yes | 4.25 | 0.16 | 4.7 × 10−7 |
| Kronberger et al., 2012. 2.5 wt% IL in glycerol. Steel on steel. 100 °C [68]. | ||||
| Glycerol | n/a | n/m | 0.15 | 0.16 × 10−3 c |
| Choline SO4 | Yes | n/m | 0.11 | 0.05 × 10−3 c |
| Methoxycholine SO4 | Yes | n/m | 0.12 | 0.12 × 10−3 c |
| N1,2,2,2 SO4 | Yes | n/m | 0.11 | 0.07 × 10−3 c |
| C4mPyr SO4 | Yes | n/m | 0.11 | 0.07 × 10−3 c |
| Choline SO3 | Yes | n/m | 0.13 | 0.13 × 10−3 c |
| Methoxycholine SO3 | Yes | n/m | 0.13 | 0.13 × 10−3 c |
| N1,1,1,4 SO3 | Yes | n/m | 0.13 | 0.13 × 10−3 c |
| Choline NTf2 | No | n/m | 0.14 | 0.10 × 10−3 c |
| Methoxycholine NTf2 | No | n/m | 0.13 | 0.06 × 10−3 c |
| N1,1,1,4 NTf2 | No | n/m | 0.14 | 0.08 × 10−3 c |
| Qu et al., 2012. 5 wt% IL in poly alpha-olefin (PAO) and 5W-30 oil. Steel on cast iron [45]. | ||||
| PAO | n/a | 3.7 | 0.22 | 9400 × 10−5 |
| 5W-30 | n/a | 10.5 | 0.094 | 7.5 × 10−5 |
| 5% P6,6,6,14 BEHP in PAO | Yes | 3.8 | 0.098 | 9.0 × 10−5 |
| 5% P6,6,6,14 BEHP in 5W-30 | Yes | 10.5 | 0.090 | 2.1 × 10−5 |
| Yu et al., 2012. 5 wt% IL in 10 W base oil and 10W-30 oil. Steel on cast iron [46]. | ||||
| 10 W base oil | n/a | 5.6 | 0.22 | 784 × 10−5 |
| 10W-30 | n/a | 10.1 | 0.10 | 14.4 × 10−5 |
| 5% P6,6,6,14 (iC8)2PO2 in 10 W oil | Yes | 5.7 | 0.09 | 7.5 × 10−5 |
| 5% P6,6,6,14 (iC8)2PO2 in 10W-30 | Yes | n/m | 0.11 | 4.0 × 10−5 |
7. Conclusion

Acknowledgments
References
- Street, K.W.; Morales, W.; Koch, V.R.; Valco, D.J.; Richard, R.M.; Hanks, N. Evaluation of vapor pressure and ultra-high vacuum tribological properties of ionic liquids. Tribol. Trans. 2011, 54, 911–919. [Google Scholar] [CrossRef]
- Palacio, M.; Bhushan, B. A review of ionic liquids for green molecular lubrication in nanotechnology. Tribol. Lett. 2010, 40, 247–268. [Google Scholar] [CrossRef]
- Bermúdez, M.D.; Jiménez, A.E.; Sanes, J.; Carrión, F.J. Ionic liquids as advanced lubricant fluids. Molecules 2009, 14, 2888–2908. [Google Scholar] [CrossRef]
- Minami, I. Ionic liquids in tribology. Molecules 2009, 14, 2286–2305. [Google Scholar] [CrossRef]
- Zhou, F.; Liang, Y.; Liu, W. Ionic liquid lubricants: Designed chemistry for engineering applications. Chem. Soc. Rev. 2009, 38, 2590–2599. [Google Scholar] [CrossRef]
- Ye, C.; Liu, W.; Chen, Y.; Yu, L. Room-Temperature ionic liquids: A novel versatile lubricant. Chem. Commun. 2001, 21, 2244–2245. [Google Scholar]
- Qu, J.; Truhan, J.; Dai, S.; Luo, H.; Blau, P. Ionic liquids with ammonium cations as lubricants or additives. Tribol. Lett. 2006, 22, 207–214. [Google Scholar] [CrossRef]
- Zhang, Q.B.; Hua, Y.X. Corrosion inhibition of mild steel by alkylimidazolium ionic liquids in hydrochloric acid. Electrochim. Acta 2009, 54, 1881–1887. [Google Scholar] [CrossRef]
- Howlett, P.C.; Zhang, S.; MacFarlane, D.R.; Forsyth, M. An investigation of a phosphinate-based ionic liquid for corrosion protection of magnesium alloy AZ31. Aust. J. Chem. 2007, 60, 43–46. [Google Scholar] [CrossRef]
- Special Issue: Ionic Liquids in Tribology; Springer: New York, NY, USA, 2010; 40, pp. 213–284. Available online: http://link.springer.com/journal/11249/40/2/page/1 (accessed on 17 January 2013).
- Special Issue: Ionic Liquids as Lubricants. SAGE Publications: London, UK, 2012; 226, pp. 889–1006. Available online: http://pij.sagepub.com/content/226/11.toc (accessed on 17 January 2013).
- Minami, I.; Kamimuram, H.; Mori, S. Thermo-Oxidative stability of ionic liquids as lubricating fluids. J. Synth. Lubr. 2007, 24, 135–147. [Google Scholar] [CrossRef]
- Canter, N. Evaluating ionic liquids as potential lubricants. Tribol. Lubr. Technol. 2005, 61, 15–17. [Google Scholar]
- Silvester, D.S.; Compton, R.G. Electrochemistry in room temperature ionic liquids: A review and some possible applications. Z. Phys. Chem. 2006, 220, 1247–1274. [Google Scholar] [CrossRef]
- Qu, J.; Blau, P.J.; Dai, S.; Luo, H.; Meyer, H.M., III; Truhan, J.J. Tribological characteristics of aluminum alloys sliding against steel lubricated by ammonium and imidazolium ionic liquids. Wear 2009, 267, 1226–1231. [Google Scholar] [CrossRef]
- Schneider, A.; Brenner, J.; Tomastik, C.; Franek, F. Capacity of selected ionic liquids as alternative ep/aw additive. Lubr. Sci. 2010, 22, 215–223. [Google Scholar] [CrossRef]
- Cai, M.; Liang, Y.; Yao, M.; Xia, Y.; Zhou, F.; Liu, W. Imidazolium ionic liquids as antiwear and antioxidant additive in poly(ethylene glycol) for steel/steel contacts. ACS Appl. Mater. Interfaces 2010, 2, 870–876. [Google Scholar] [CrossRef]
- Liu, W.; Ye, C.; Gong, Q.; Wang, H.; Wang, P. Tribological performance of room-temperature ionic liquids as lubricant. Tribol. Lett. 2002, 13, 81–85. [Google Scholar] [CrossRef]
- Wang, H.; Lu, Q.; Ye, C.; Liu, W.; Cui, Z. Friction and wear behaviors of ionic liquid of alkylimidazolium hexafluorophosphates as lubricants for steel/steel contact. Wear 2004, 256, 44–48. [Google Scholar] [CrossRef]
- Lu, Q.; Wang, H.; Ye, C.; Liu, W.; Xue, Q. Room temperature ionic liquid 1-ethyl-3-hexylimidazolium-bis(trifluoromethylsulfonyl)-imide as lubricant for steel-steel contact. Tribol. Int. 2004, 37, 547–552. [Google Scholar] [CrossRef]
- Weng, L.J.; Liu, X.Q.; Liang, Y.M.; Xue, Q.J. Effect of tetraalkylphosphonium based ionic liquids as lubricants on the tribological performance of a steel-on-steel system. Tribol. Lett. 2007, 26, 11–17. [Google Scholar] [CrossRef]
- Minami, I.; Inada, T.; Sasaki, R.; Nanao, H. Tribo-Chemistry of phosphonium-derived ionic liquids. Tribol. Lett. 2010, 40, 225–235. [Google Scholar] [CrossRef]
- Zhang, L.; Feng, D.; Xu, B. Tribological characteristics of alkylimidazolium diethyl phosphates ionic liquids as lubricants for steel-steel contact. Tribol. Lett. 2009, 34, 95–101. [Google Scholar] [CrossRef]
- Minami, I.; Kita, M.; Kubo, T.; Nanao, H.; Mori, S. The tribological properties of ionic liquids composed of trifluorotris(pentafluoroethyl) phosphate as a hydrophobic anion. Tribol. Lett. 2008, 30, 215–223. [Google Scholar] [CrossRef]
- Jiang, D.; Hu, L.; Feng, D. Crown-Type ionic liquids as lubricants for steel-on-steel system. Tribol. Lett. 2011, 41, 417–424. [Google Scholar] [CrossRef]
- Zhu, L.Y.; Chen, L.G.; Yang, X.; Song, H.B. Functionalized ionic liquids as lubricants for steel-steel contact. Appl. Mech. Mat. 2012, 138-139, 630–634. [Google Scholar]
- Iglesias, P.; Bermúdez, M.D.; Carrión, F.J.; Martínez-Nicolás, G. Friction and wear of aluminium-steel contacts lubricated with ordered fluids-neutral and ionic liquid crystals as oil additives. Wear 2004, 256, 386–392. [Google Scholar] [CrossRef]
- Chen, Y.; Ye, C.; Wang, H.; Liu, W. Tribological performance of an ionic liquid as a lubricant for steel/aluminium contacts. J. Synth. Lubr. 2003, 20, 217–225. [Google Scholar] [CrossRef]
- Jiménez, A.E.; Bermúdez, M.D.; Iglesias, P.; Carrión, F.J.; Martínez-Nicolás, G. 1-N-alkyl-3-methylimidazolium ionic liquids as neat lubricants and lubricant additives in steel-aluminium contacts. Wear 2006, 260, 766–782. [Google Scholar] [CrossRef]
- Jiménez, A.E.; Bermúdez, M.D.; Carrión, F.J.; Martínez-Nicolás, G. Room temperature ionic liquids as lubricant additives in steel-aluminium contacts: Influence of sliding velocity, normal load and temperature. Wear 2006, 261, 347–359. [Google Scholar] [CrossRef]
- Jiménez, A.E.; Bermúdez, M.D. Imidazolium ionic liquids as additives of the synthetic ester propylene glycol dioleate in aluminium-steel lubrication. Wear 2008, 265, 787–798. [Google Scholar] [CrossRef]
- Mu, Z.; Zhou, F.; Zhang, S.; Liang, Y.; Liu, W. Effect of the functional groups in ionic liquid molecules on the friction and wear behavior of aluminum alloy in lubricated aluminum-on-steel contact. Tribol. Int. 2005, 38, 725–731. [Google Scholar] [CrossRef]
- Liu, X.; Zhou, F.; Liang, Y.; Liu, W. Tribological performance of phosphonium based ionic liquids for an aluminum-on-steel system and opinions on lubrication mechanism. Wear 2006, 261, 1174–1179. [Google Scholar] [CrossRef]
- Jiménez, A.E.; Bermúdez, M.D. Ionic liquids as lubricants for steel-aluminum contacts at low and elevated temperatures. Tribol. Lett. 2007, 26, 53–60. [Google Scholar] [CrossRef]
- Mu, Z.; Wang, X.; Zhang, S.; Liang, Y.; Bao, M.; Liu, W. Investigation of tribological behavior of Al-Si alloy against steel lubricated with ionic liquids of 1-diethylphosphonyl-N-propyl-3-alkylimidazolium tetrafluoroborate. J. Tribol. 2008, 130. Available online: http://cat.inist.fr/?aModele=afficheN&cpsidt=20551426 (accessed on 16 January 2013).
- Somers, A.E.; Howlett, P.C.; Sun, J.; MacFarlane, D.R.; Forsyth, M. Phosphonium Ionic Liquids as Lubricants for Aluminium-Steel, Proceedings of the 3rd International Conference on Tribology and Design, Algarve, Portugal, 11-13 May 2010; pp. 273–283.
- Somers, A.E.; Howlett, P.C.; Sun, J.; MacFarlane, D.R.; Forsyth, M. Transition in wear performance for ionic liquid lubricants under increasing load. Tribol. Lett. 2010, 40, 279–284. [Google Scholar] [CrossRef]
- Somers, A.E.; Biddulph, S.M.; Howlett, P.C.; Sun, J.; MacFarlane, D.R.; Forsyth, M. A comparison of phosphorus and fluorine containing IL lubricants for steel on aluminium. Phys. Chem. Chem. Phys. 2012.
- Shah, F.U.; Glavatskih, S.; MacFarlane, D.R.; Somers, A.; Forsyth, M.; Antzutkin, O.N. Novel halogen-free chelated orthoborate-phosphonium ionic liquids: Synthesis and tribophysical properties. Phys. Chem. Chem. Phys. 2011, 13, 12865–12873. [Google Scholar]
- Jiménez, A.E.; Bermúdez, M.D. Ionic liquids as lubricants of titanium-steel contact. Tribol. Lett. 2009, 33, 111–126. [Google Scholar] [CrossRef]
- Jiménez, A.E.; Bermúdez, M.D. Ionic liquids as lubricants of titanium-steel contact. Part 3. Ti6Al4V lubricated with imidazolium ionic liquids with different alkyl chain lengths. Tribol. Lett. 2010, 40, 237–246. [Google Scholar] [CrossRef]
- Jiménez, A.E.; Bermúdez, M.D. Ionic liquids as lubricants of titanium-steel contact. Part 2: Friction, wear and surface interactions at high temperature. Tribol. Lett. 2010, 37, 431–443. [Google Scholar] [CrossRef]
- Mistry, K.; Fox, M.F.; Priest, M. Lubrication of an electroplated nickel matrix silicon carbide coated eutectic aluminium-silicon alloy automotive cylinder bore with an ionic liquid as a lubricant additive. Proc. Inst. Mech. Eng. J: J. Eng. Tribol. 2009, 223, 563–569. [Google Scholar]
- Qu, J.; Blau, P.J.; Dai, S.; Luo, H.; Meyer, H.M., III. Ionic liquids as novel lubricants and additives for diesel engine applications. Tribol. Lett. 2009, 35, 181–189. [Google Scholar] [CrossRef]
- Qu, J.; Bansal, D.G.; Yu, B.; Howe, J.Y.; Luo, H.; Dai, S.; Li, H.; Blau, P.J.; Bunting, B.G.; Mordukhovich, G.; Smolenski, D.J. Antiwear performance and mechanism of an oil-miscible ionic liquid as a lubricant additive. ACS Appl. Mater. Interfaces 2012, 4, 997–1002. [Google Scholar] [CrossRef]
- Yu, B.; Bansal, D.G.; Qu, J.; Sun, X.; Luo, H.; Dai, S.; Blau, P.J.; Bunting, B.G.; Mordukhovich, G.; Smolenski, D.J. Oil-Miscible and non-corrosive phosphonium-based ionic liquids as candidate lubricant additives. Wear 2012, 289, 58–64. [Google Scholar] [CrossRef]
- Pu, J.; Wang, L.; Mo, Y.; Xue, Q. Preparation and characterization of ultrathin dual-layer ionic liquid lubrication film assembled on silica surfaces. J. Colloid Interface Sci. 2011, 354, 858–865. [Google Scholar] [CrossRef]
- Zhao, W.; Wang, Y.; Wang, L.; Bai, M.; Xue, Q. Influence of heat treatment on the micro/nano-tribological properties of ultra-thin ionic liquid films on silicon. Colloids Surf. A: Physicochem. Eng. Asp. 2010, 361, 118–125. [Google Scholar] [CrossRef]
- Yu, B.; Zhou, F.; Mu, Z.; Liang, Y.; Liu, W. Tribological properties of ultra-thin ionic liquid films on single-crystal silicon wafers with functionalized surfaces. Tribol. Int. 2006, 39, 879–887. [Google Scholar] [CrossRef]
- Palacio, M.; Bhushan, B. Ultrathin wear-resistant ionic liquid films for novel mems/nems applications. Adv. Mater. 2008, 20, 1194–1198. [Google Scholar] [CrossRef]
- Qu, J.; Chi, M.; Meyer, H.M., III; Blau, P.J.; Dai, S.; Luo, H. Nanostructure and composition of tribo-boundary films formed in ionic liquid lubrication. Tribol. Lett. 2011, 43, 205–211. [Google Scholar] [CrossRef]
- Battez, A.H.; González, R.; Viesca, J.L.; Blanco, D.; Asedegbega, E.; Osorio, A. Tribological behaviour of two imidazolium ionic liquids as lubricant additives for steel/steel contacts. Wear 2009, 266, 1224–1228. [Google Scholar] [CrossRef]
- Blanco, D.; Battez, A.H.; Viesca, J.L.; González, R.; Fernández-González, A. Lubrication of CrN coating with ethyl-dimethyl-2-methoxy ethyl ammonium tris(pentafluoroethyl) trifluorophosphate ionic liquid as additive to PAO 6. Tribol. Lett. 2011, 41, 295–302. [Google Scholar] [CrossRef]
- Yagi, T.; Sasaki, S.; Mano, H.; Miyake, K.; Nakano, M.; Ishida, T. Lubricity and chemical reactivity of ionic liquid used for sliding metals under high-vacuum conditions. Proc. Inst. Mech. Eng. J: J. Eng. Tribol. 2009, 223, 1083–1090. [Google Scholar]
- Bermúdez, M.D.; Jiménez, A.E. Surface interactions and tribochemical processes in ionic liquid lubrication of aluminium-steel contacts. Int. J. Surf. Sci. Eng. 2007, 1, 100–110. [Google Scholar] [CrossRef]
- Lu, R.; Mori, S.; Kobayashi, K.; Nanao, H. Study of tribochemical decomposition of ionic liquids on a nascent steel surface. Appl. Surf. Sci. 2009, 255, 8965–8971. [Google Scholar] [CrossRef]
- Atkin, R.; El Abedin, S.Z.; Hayes, R.; Gasparotto, L.H.S.; Borisenko, N.; Endres, F. AFM and STM studies on the surface interaction of [BMP]TFSA and [EMIm]TFSA ionic liquids with au(111). J. Phys. Chem. C 2009, 113, 13266–13272. [Google Scholar] [CrossRef]
- Perkin, S.; Albrecht, T.; Klein, J. Layering and shear properties of an ionic liquid, 1-ethyl-3-methylimidazolium ethylsulfate, confined to nano-films between mica surfaces. Phys. Chem. Chem. Phys. 2010, 12, 1243–1247. [Google Scholar] [CrossRef]
- Perkin, S. Ionic liquids in confined geometries. Phys. Chem. Chem. Phys. 2012, 14, 5052–5062. [Google Scholar] [CrossRef]
- Ueno, K.; Kasuya, M.; Watanabe, M.; Mizukami, M.; Kurihara, K. Resonance shear measurement of nanoconfined ionic liquids. Phys. Chem. Chem. Phys. 2010, 12, 4066–4071. [Google Scholar]
- Klein, J.; Kumacheva, E. Simple liquids confined to molecularly thin layers. I. Confinement-Induced liquid-to-solid phase transitions. J. Chem. Phys. 1998, 108, 6996–7009. [Google Scholar] [CrossRef]
- Kumacheva, E.; Klein, J. Simple liquids confined to molecularly thin layers. II. Shear and frictional behavior of solidified films. J. Chem. Phys. 1998, 108, 7010–7022. [Google Scholar] [CrossRef]
- Xiao, H.; Guo, D.; Liu, S.; Pan, G.; Lu, X. Film thickness of ionic liquids under high contact pressures as a function of alkyl chain length. Tribol. Lett. 2011, 41, 471–477. [Google Scholar] [CrossRef]
- Hamrock, B.J.; Dowson, D. Isothermal elastohydrodynamic lubrication of point contacts. J. Lubr. Technol. Trans. ASME 1977, 99, 264–276. [Google Scholar] [CrossRef]
- Pensado, A.S.; Comuñas, M.J.P.; Fernández, J. The pressure-viscosity coefficient of several ionic liquids. Tribol. Lett. 2008, 31, 107–118. [Google Scholar] [CrossRef]
- Paredes, X.; Fandiño, O.; Pensado, A.S.; Comuñas, M.J.P.; Fernández, J. Pressure-Viscosity coefficients for polyalkylene glycol oils and other ester or ionic lubricants. Tribol. Lett. 2012, 45, 89–100. [Google Scholar] [CrossRef]
- Gaciño, F.M.; Paredes, X.; Comuñas, M.J.P.; Fernández, J. Effect of the pressure on the viscosities of ionic liquids: Experimental values for 1-ethyl-3-methylimidazolium ethylsulfate and two bis(trifluoromethyl- sulfonyl)imide salts. J. Chem. Thermodyn. 2012, 54, 302–309. [Google Scholar] [CrossRef]
- Kronberger, M.; Pejaković, V.; Gabler, C.; Kalin, M. How anion and cation species influence the tribology of a green lubricant based on ionic liquids. Proc. Inst. Mech. Eng. J: J. Eng. Tribol. 2012, 226, 933–951. [Google Scholar] [CrossRef]
- Blanco, D.; González, R.; Hernández Battez, A.; Viesca, J.L.; Fernández-González, A. Use of ethyl-dimethyl-2-methoxyethylammonium tris(pentafluoroethyl) trifluorophosphate as base oil additive in the lubrication of tin pvd coating. Tribol. Int. 2011, 44, 645–650. [Google Scholar] [CrossRef]
- Pejaković, V.; Kronberger, M.; Mahrova, M.; Vilas, M.; Tojo, E.; Kalin, M. Pyrrolidinium sulfate and ammonium sulfate ionic liquids as lubricant additives for steel/steel contact lubrication. Proc. Inst. Mech. Eng. J: J. Eng. Tribol. 2012, 226, 923–932. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Somers, A.E.; Howlett, P.C.; MacFarlane, D.R.; Forsyth, M. A Review of Ionic Liquid Lubricants. Lubricants 2013, 1, 3-21. https://doi.org/10.3390/lubricants1010003
Somers AE, Howlett PC, MacFarlane DR, Forsyth M. A Review of Ionic Liquid Lubricants. Lubricants. 2013; 1(1):3-21. https://doi.org/10.3390/lubricants1010003
Chicago/Turabian StyleSomers, Anthony E., Patrick C. Howlett, Douglas R. MacFarlane, and Maria Forsyth. 2013. "A Review of Ionic Liquid Lubricants" Lubricants 1, no. 1: 3-21. https://doi.org/10.3390/lubricants1010003
APA StyleSomers, A. E., Howlett, P. C., MacFarlane, D. R., & Forsyth, M. (2013). A Review of Ionic Liquid Lubricants. Lubricants, 1(1), 3-21. https://doi.org/10.3390/lubricants1010003