The Making of a CYP3A Biomarker Panel for Guiding Drug Therapy
Abstract
:1. Introduction
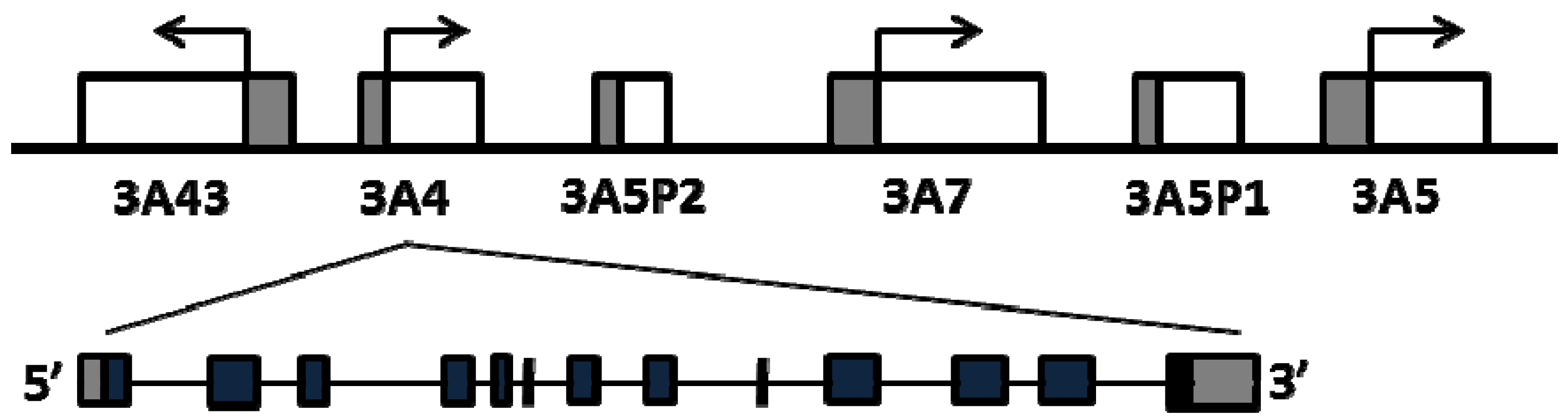
2. Common Regulatory Polymorphisms in CYP3A4
3. Coding Region Polymorphisms
| Allele | variant (cDNA) | Amino acid change | Key SNP rs# | Gene location | MAF | In vitro effect | In vivo effect | Ref | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Caucasian | African American | Asian | All (ESP) | ||||||||
| *2 | 664 T>C | S222P | rs55785340 | Exon 7 | 0.027 | 0 | 0 | Decreased activity | [44] | ||
| *3 | 1334 T>C | M445T | rs4986910 | Exon 12 | 0–0.04 | 0–0.033 | 0 | No change | [43,45,46,47] | ||
| *4 | 352 A>G | I118V | rs55951658 | Exon 5 | 0.015–0.033 | Decreased activity, associated with LDL | [50,51] | ||||
| *5 | 653 C>G | P218R | rs55901263 | Exon 7 | 0 | 0 | 0.006 | Decreased activity | [50] | ||
| *6 | 830_831 insA | 277 frameshift | rs4646438 | Exon 9 | 0.005 | No activity | [50] | ||||
| *7 | 167 G>A | G56D | rs56324128 | Exon 3 | 0.014 | Decreased activity | [43,44] | ||||
| *8 | 389 G>A | R130Q | rs72552799 | Exon 5 | 0.0033 | No protein | [43] | ||||
| *9 | 508 G>A | V170I | rs72552798 | Exon 6 | 0.0024 | No change | [43] | ||||
| *10 | 520 G>C or G>A | D174H or D174N | rs4986908 | Exon 6 | 0.003 for C | 0.012 for A | No change | [21,43] | |||
| *11 | 1088 C>T | T363M | rs67784355 | Exon 11 | 0.0034 | 0.002 | 0.001 | Decreased protein level and activity | [43,49] | ||
| *12 | I117 C>T | L373F | rs12721629 | Exon 11 | 0–0.004 | 0.004–0.007 | 0 | 0.001 | Decreased protein level and activity | [43] | |
| *13 | 1247 C>T | P416L | rs4986909 | Exon 11 | 0–0.004 | 0–0.021 | 0–0.012 | No protein | [43] | ||
| *14 | 44 T>C | L15P | rs12721634 | Exon 1 | 0–0.003 | [21] | |||||
| *15 | 485 G>A | R162Q | rs4986907 | Exon 6 | 0 | 0–0.042 | 0 | 0.014 | [21] | ||
| *16 | 554 C>G | T185S | rs12721627 | Exon 7 | 0.014 | 0.005 | Decreased protein level and activity | [44,48,49] | |||
| *17 | 566 T>C | F189S | rs4987161 | Exon 7 | 0–0.017 | 0 | 0 | Decreased activity | [45] | ||
| *18 | 878 T>C | L293P | rs28371759 | Exon 10 | 0.028 | 0.01 | No change or increased activity | Low midazolam clearance, associated with bone density | [44,45,48,49,52,53,54,55] | ||
| *19 | 1399 C>T | P467S | rs4986913 | Exon 12 | 0–0.022 | 0 | 0–0.012 | No change | [45] | ||
| *20 | 1461_1462 insA | 488 frameshift | rs7666821 | Exon 13 | <0.006 | No activity | Low midazolam clearance | [56] | |||
| *21 | 956 A>G | Y319C | Exon 10 | 0.005 | [21] | ||||||
| Gene | Allele | Location, variant | MAF | dbSNP number | CYP3A4 activity |
|---|---|---|---|---|---|
| CYP3A4 | *1 a | - | - | - | - |
| CYP3A4 | *22 | Intron 6, C>T | 3%–6% | rs35599367 | Decreased protein |
| CYP3A4 | *6 b | Exon 9, insA | rare | rs4646438 | Frameshift |
| CYP3A4 | *20 b | Exon 13, insA | rare | rs7666821 | Frameshift |
| CYP3A5 | *1 a | - | - | - | - |
| CYP3A5 | *3 | Intron3, G>A | 15%–88% | rs776746 | No protein |
| CYP3A5 | *7 | Exon 11, delT | 4%–21% | rs41303343 | Frameshift |
4. Interaction between CYP3A4 and CYP3A5
5. Polymorphisms in Transcription Factors
6. microRNA and Epigenetics Regulation
7. Clinical Implications
Acknowledgement
Conflict of Interest
References and Notes
- Garcia, Y.; Muquillaza, P.; Valdebenito, S. Individualized neoral doses in pediatric renal transplantation. Transplant. Proc. 2010, 42, 357–360. [Google Scholar] [CrossRef]
- Wakahashi, K.; Yamamori, M.; Minagawa, K.; Ishii, S.; Nishikawa, S.; Shimoyama, M.; Kawano, H.; Kawano, Y.; Kawamori, Y.; Sada, A.; et al. Pharmacokinetics-based optimal dose prediction of donor source-dependent response to mycophenolate mofetil in unrelated hematopoietic cell transplantation. Int. J. Hematol. 2011, 94, 193–202. [Google Scholar] [CrossRef]
- Bartelink, I.H.; Boelens, J.J.; Bredius, R.G.; Egberts, A.C.; Wang, C.; Bierings, M.B.; Shaw, P.J.; Nath, C.E.; Hempel, G.; Zwaveling, J.; et al. Body weight-dependent pharmacokinetics of busulfan in paediatric haematopoietic stem cell transplantation patients: Towards individualized dosing. Clin. Pharmacokinet. 2012, 51, 331–345. [Google Scholar] [CrossRef]
- Xiang, Y.; Remily-Wood, E.R.; Oliveira, V.; Yarde, D.; He, L.; Cheng, J.Q.; Mathews, L.; Boucher, K.; Cubitt, C.; Perez, L.; et al. Monitoring a nuclear factor-kappab signature of drug resistance in multiple myeloma. Mol. Cell. Proteomics 2011. [Google Scholar] [CrossRef]
- Frandsen, T.L.; Abrahamsson, J.; Lausen, B.; Vettenranta, K.; Heyman, M.; Behrentz, M.; Castor, A.; Wehner, P.S.; Frost, B.M.; Andersen, E.W.; et al. Individualized toxicity-titrated 6-mercaptopurine increments during high-dose methotrexate consolidation treatment of lower risk childhood acute lymphoblastic leukaemia. A nordic society of paediatric haematology and oncology (nopho) pilot study. Br. J. Haematol. 2011, 155, 244–247. [Google Scholar] [CrossRef]
- Johnson, J.A.; Cavallari, L.H.; Beitelshees, A.L.; Lewis, J.P.; Shuldiner, A.R.; Roden, D.M. Pharmacogenomics: Application to the management of cardiovascular disease. Clin. Pharmacol. Ther. 2011, 90, 519–531. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Table of Pharmacogenomic Biomarkers in Drug Labels. Available online: http://www.fda.gov/Drugs/ScienceResearch/ReaserchAreas/Pharmacogenetics/ucm083378.htm (accessed on 1 October 2012).
- Lamba, J.K.; Lin, Y.S.; Schuetz, E.G.; Thummel, K.E. Genetic contribution to variable human CYP3A-mediated metabolism. Adv. Drug Deliv. Rev. 2002, 54, 1271–1294. [Google Scholar] [CrossRef]
- Finta, C.; Zaphiropoulos, P.G. The human cytochrome P450 3A locus. Gene evolution by capture of downstream exons. Gene 2000, 260, 13–23. [Google Scholar] [CrossRef]
- Gellner, K.; Eiselt, R.; Hustert, E.; Arnold, H.; Koch, I.; Haberl, M.; Deglmann, C.J.; Burk, O.; Buntefuss, D.; Escher, S.; et al. Genomic organization of the human CYP3A locus: Identification of a new, inducible CYP3A gene. Pharmacogenetics 2001, 11, 111–121. [Google Scholar] [CrossRef]
- Kuehl, P.; Zhang, J.; Lin, Y.; Lamba, J.; Assem, M.; Schuetz, J.; Watkins, P.B.; Daly, A.; Wrighton, S.A.; Hall, S.D.; et al. Sequence diversity in CYP3A promoters and characterization of the genetic basis of polymorphic CYP3A5 expression. Nat. Genet. 2001, 27, 383–391. [Google Scholar] [CrossRef]
- Shimada, T.; Yamazaki, H.; Mimura, M.; Wakamiya, N.; Ueng, Y.F.; Guengerich, F.P.; Inui, Y. Characterization of microsomal cytochrome P450 enzymes involved in the oxidation of xenobiotic chemicals in human fetal liver and adult lungs. Drug Metab. Dispos. 1996, 24, 515–522. [Google Scholar]
- Burk, O.; Tegude, H.; Koch, I.; Hustert, E.; Wolbold, R.; Glaeser, H.; Klein, K.; Fromm, M.F.; Nuessler, A.K.; Neuhaus, P.; et al. Molecular mechanisms of polymorphic CYP3A7 expression in adult human liver and intestine. J. Biol. Chem. 2002, 277, 24280–24288. [Google Scholar]
- Westlind, A.; Malmebo, S.; Johansson, I.; Otter, C.; Andersson, T.B.; Ingelman-Sundberg, M.; Oscarson, M. Cloning and tissue distribution of a novel human cytochrome P450 of the CYP3A subfamily, CYP3A43. Biochem. Biophys. Res. Commun. 2001, 281, 1349–1355. [Google Scholar] [CrossRef]
- Thummel, K.E.; Wilkinson, G.R. In vitro and in vivo drug interactions involving human CYP3A. Annu. Rev. Pharmacol. Toxicol. 1998, 38, 389–430. [Google Scholar] [CrossRef]
- Williams, J.A.; Ring, B.J.; Cantrell, V.E.; Jones, D.R.; Eckstein, J.; Ruterbories, K.; Hamman, M.A.; Hall, S.D.; Wrighton, S.A. Comparative metabolic capabilities of CYP3A4, CYP3A5, and CYP3A7. Drug Metab. Dispos. 2002, 30, 883–891. [Google Scholar] [CrossRef]
- Klees, T.M.; Sheffels, P.; Dale, O.; Kharasch, E.D. Metabolism of alfentanil by cytochrome P4503A (CYP3A) enzymes. Drug Metab. Dispos. 2005, 33, 303–311. [Google Scholar]
- Shimada, T.; Yamazaki, H.; Mimura, M.; Inui, Y.; Guengerich, F.P. Interindividual variations in human liver cytochrome P-450 enzymes involved in the oxidation of drugs, carcinogens and toxic chemicals: Studies with liver microsomes of 30 Japanese and 30 Caucasians. J. Pharmacol. Exp. Ther. 1994, 270, 414–423. [Google Scholar]
- Westlind, A.; Lofberg, L.; Tindberg, N.; Andersson, T.B.; Ingelman-Sundberg, M. Interindividual differences in hepatic expression of CYP3A4: Relationship to genetic polymorphism in the 5'-upstream regulatory region. Biochem. Biophys. Res. Commun. 1999, 259, 201–205. [Google Scholar] [CrossRef]
- Schellens, J.H.; Soons, P.A.; Breimer, D.D. Lack of bimodality in nifedipine plasma kinetics in a large population of healthy subjects. Biochem. Pharmacol. 1988, 37, 2507–2510. [Google Scholar] [CrossRef]
- Hellriegel, E.T.; Bjornsson, T.D.; Hauck, W.W. Interpatient variability in bioavailability is related to the extent of absorption: Implications for bioavailability and bioequivalence studies. Clin. Pharmacol. Ther. 1996, 60, 601–607. [Google Scholar] [CrossRef]
- Williams, J.A.; Cook, J.; Hurst, S.I. A significant drug-metabolizing role for CYP3A5? Drug Metab. Dispos. 2003, 31, 1526–1530. [Google Scholar] [CrossRef]
- Wojnowski, L.; Kamdem, L.K. Clinical implications of CYP3A polymorphisms. Expert Opin. Drug Metab. Toxicol. 2006, 2, 171–182. [Google Scholar] [CrossRef]
- Rebbeck, T.R.; Jaffe, J.M.; Walker, A.H.; Wein, A.J.; Malkowicz, S.B. Modification of clinical presentation of prostate tumors by a novel genetic variant in CYP3A4. J. Natl. Cancer Inst. 1998, 90, 1225–1229. [Google Scholar] [CrossRef]
- Lamba, J.K.; Lin, Y.S.; Thummel, K.; Daly, A.; Watkins, P.B.; Strom, S.; Zhang, J.; Schuetz, E.G. Common allelic variants of cytochrome P4503A4 and their prevalence in different populations. Pharmacogenetics 2002, 12, 121–132. [Google Scholar] [CrossRef]
- Garcia-Martin, E.; Martinez, C.; Pizarro, R.M.; Garcia-Gamito, F.J.; Gullsten, H.; Raunio, H.; Agundez, J.A. CYP3A4 variant alleles in white individuals with low CYP3A4 enzyme activity. Clin. Pharmacol. Ther. 2002, 71, 196–204. [Google Scholar] [CrossRef]
- Amirimani, B.; Walker, A.H.; Weber, B.L.; Rebbeck, T.R. Response: Re: Modification of clinical presentation of prostate tumors by a novel genetic variant in CYP3A4. J. Natl. Cancer Inst. 1999, 91, 1588–1590. [Google Scholar] [CrossRef]
- Spurdle, A.B.; Goodwin, B.; Hodgson, E.; Hopper, J.L.; Chen, X.; Purdie, D.M.; McCredie, M.R.; Giles, G.G.; Chenevix-Trench, G.; Liddle, C. The CYP3A4*1b polymorphism has no functional significance and is not associated with risk of breast or ovarian cancer. Pharmacogenetics 2002, 12, 355–366. [Google Scholar] [CrossRef]
- Ball, S.E.; Scatina, J.; Kao, J.; Ferron, G.M.; Fruncillo, R.; Mayer, P.; Weinryb, I.; Guida, M.; Hopkins, P.J.; Warner, N.; et al. Population distribution and effects on drug metabolism of a genetic variant in the 5' promoter region of CYP3A4. Clin. Pharmacol. Ther. 1999, 66, 288–294. [Google Scholar] [CrossRef]
- Felix, C.A.; Walker, A.H.; Lange, B.J.; Williams, T.M.; Winick, N.J.; Cheung, N.K.; Lovett, B.D.; Nowell, P.C.; Blair, I.A.; Rebbeck, T.R. Association of CYP3A4 genotype with treatment-related leukemia. Proc. Natl. Acad. Sci. USA 1998, 95, 13176–13181. [Google Scholar]
- Zeigler-Johnson, C.; Friebel, T.; Walker, A.H.; Wang, Y.; Spangler, E.; Panossian, S.; Patacsil, M.; Aplenc, R.; Wein, A.J.; Malkowicz, S.B.; et al. CYP3A4, CYP3A5, and CYP3A43 genotypes and haplotypes in the etiology and severity of prostate cancer. Cancer Res. 2004, 64, 8461–8467. [Google Scholar] [CrossRef]
- Miao, J.; Jin, Y.; Marunde, R.L.; Gorski, C.J.; Kim, S.; Quinney, S.; Radovich, M.; Li, L.; Hall, S.D. Association of genotypes of the CYP3A cluster with midazolam disposition in vivo. Pharmacogenomics J. 2009, 9, 319–326. [Google Scholar] [CrossRef]
- Schirmer, M.; Rosenberger, A.; Klein, K.; Kulle, B.; Toliat, M.R.; Nurnberg, P.; Zanger, U.M.; Wojnowski, L. Sex-dependent genetic markers of CYP3A4 expression and activity in human liver microsomes. Pharmacogenomics 2007, 8, 443–453. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, L.R.; Fu, Q. CYP3A4*1g polymorphism is associated with lipid-lowering efficacy of atorvastatin but not of simvastatin. Eur. J. Clin. Pharmacol. 2008, 64, 877–882. [Google Scholar] [CrossRef]
- Miura, M.; Satoh, S.; Kagaya, H.; Saito, M.; Numakura, K.; Tsuchiya, N.; Habuchi, T. Impact of the CYP3A4*1g polymorphism and its combination with CYP3A5 genotypes on tacrolimus pharmacokinetics in renal transplant patients. Pharmacogenomics 2011, 12, 977–984. [Google Scholar] [CrossRef]
- He, B.X.; Shi, L.; Qiu, J.; Tao, L.; Li, R.; Yang, L.; Zhao, S.J. A functional polymorphism in the CYP3A4 gene is associated with increased risk of coronary heart disease in the Chinese han population. Basic Clin. Pharmacol. Toxicol. 2011, 108, 208–213. [Google Scholar] [CrossRef]
- Chen, C.H.; Wang, S.C.; Tsou, H.H.; Ho, I.K.; Tian, J.N.; Yu, C.J.; Hsiao, C.F.; Chou, S.Y.; Lin, Y.F.; Fang, K.C.; et al. Genetic polymorphisms in CYP3A4 are associated with withdrawal symptoms and adverse reactions in methadone maintenance patients. Pharmacogenomics 2011, 12, 1397–1406. [Google Scholar] [CrossRef]
- Fukushima-Uesaka, H.; Saito, Y.; Watanabe, H.; Shiseki, K.; Saeki, M.; Nakamura, T.; Kurose, K.; Sai, K.; Komamura, K.; Ueno, K.; et al. Haplotypes of CYP3A4 and their close linkage with CYP3A5 haplotypes in a Japanese population. Hum. Mutat. 2004, 23. [Google Scholar] [CrossRef]
- Perera, M.A.; Thirumaran, R.K.; Cox, N.J.; Hanauer, S.; Das, S.; Brimer-Cline, C.; Lamba, V.; Schuetz, E.G.; Ratain, M.J.; di Rienzo, A. Prediction of CYP3A4 enzyme activity using haplotype tag SNPs in African Americans. Pharmacogenomics J. 2009, 9, 49–60. [Google Scholar] [CrossRef]
- Matsumura, K.; Saito, T.; Takahashi, Y.; Ozeki, T.; Kiyotani, K.; Fujieda, M.; Yamazaki, H.; Kunitoh, H.; Kamataki, T. Identification of a novel polymorphic enhancer of the human CYP3A4 gene. Mol. Pharmacol. 2004, 65, 326–334. [Google Scholar] [CrossRef]
- Wang, D.; Guo, Y.; Wrighton, S.A.; Cooke, G.E.; Sadee, W. Intronic polymorphism in CYP3A4 affects hepatic expression and response to statin drugs. Pharmacogenomics J. 2011, 11, 274–286. [Google Scholar] [CrossRef]
- Birdwell, K.A.; Grady, B.; Choi, L.; Xu, H.; Bian, A.; Denny, J.C.; Jiang, M.; Vranic, G.; Basford, M.; Cowan, J.D.; et al. The use of a DNA biobank linked to electronic medical records to characterize pharmacogenomic predictors of tacrolimus dose requirement in kidney transplant recipients. Pharmacogenet. Genomics 2012, 22, 32–42. [Google Scholar] [CrossRef]
- Elens, L.; Bouamar, R.; Hesselink, D.A.; Haufroid, V.; van der Heiden, I.P.; van Gelder, T.; van Schaik, R.H. A new functional CYP3A4 intron 6 polymorphism significantly affects tacrolimus pharmacokinetics in kidney transplant recipients. Clin. Chem. 2011, 57, 1574–1583. [Google Scholar] [CrossRef]
- Elens, L.; van Schaik, R.H.; Panin, N.; de Meyer, M.; Wallemacq, P.; Lison, D.; Mourad, M.; Haufroid, V. Effect of a new functional CYP3A4 polymorphism on calcineurin inhibitors’ dose requirements and trough blood levels in stable renal transplant patients. Pharmacogenomics 2011, 12, 1383–1396. [Google Scholar] [CrossRef]
- Elens, L.; Becker, M.L.; Haufroid, V.; Hofman, A.; Visser, L.E.; Uitterlinden, A.G.; Stricker, B.; van Schaik, R.H. Novel CYP3A4 intron 6 single nucleotide polymorphism is associated with simvastatin-mediated cholesterol reduction in the rotterdam study. Pharmacogenet. Genomics 2011, 21, 861–866. [Google Scholar] [CrossRef]
- Elens, L.; Bouamar, R.; Hesselink, D.A.; Haufroid, V.; van Gelder, T.; van Schaik, R.H. The new CYP3A4 intron 6 C>T polymorphism (CYP3A4*22) is associated with an increased risk of delayed graft function and worse renal function in cyclosporine-treated kidney transplant patients. Pharmacogenet. Genomics 2012, 22, 373–380. [Google Scholar]
- Eiselt, R.; Domanski, T.L.; Zibat, A.; Mueller, R.; Presecan-Siedel, E.; Hustert, E.; Zanger, U.M.; Brockmoller, J.; Klenk, H.P.; Meyer, U.A.; et al. Identification and functional characterization of eight CYP3A4 protein variants. Pharmacogenetics 2001, 11, 447–458. [Google Scholar] [CrossRef]
- Miyazaki, M.; Nakamura, K.; Fujita, Y.; Guengerich, F.P.; Horiuchi, R.; Yamamoto, K. Defective activity of recombinant cytochromes P450 3A4.2 and 3A4.16 in oxidation of midazolam, nifedipine, and testosterone. Drug Metab. Dispos. 2008, 36, 2287–2291. [Google Scholar] [CrossRef]
- Dai, D.; Tang, J.; Rose, R.; Hodgson, E.; Bienstock, R.J.; Mohrenweiser, H.W.; Goldstein, J.A. Identification of variants of CYP3A4 and characterization of their abilities to metabolize testosterone and chlorpyrifos. J. Pharmacol. Exp. Ther. 2001, 299, 825–831. [Google Scholar]
- Kajinami, K.; Brousseau, M.E.; Ordovas, J.M.; Schaefer, E.J. CYP3A4 genotypes and plasma lipoprotein levels before and after treatment with atorvastatin in primary hypercholesterolemia. Am. J. Cardiol. 2004, 93, 104–107. [Google Scholar] [CrossRef]
- Thompson, J.F.; Man, M.; Johnson, K.J.; Wood, L.S.; Lira, M.E.; Lloyd, D.B.; Banerjee, P.; Milos, P.M.; Myrand, S.P.; Paulauskis, J.; et al. An association study of 43 SNPs in 16 candidate genes with atorvastatin response. Pharmacogenomics J. 2005, 5, 352–358. [Google Scholar] [CrossRef]
- Nakajima, Y.; Yoshitani, T.; Fukushima-Uesaka, H.; Saito, Y.; Kaniwa, N.; Kurose, K.; Ozawa, S.; Aoyagi, N.; Kamatani, N.; Yamamoto, N.; et al. Impact of the haplotype CYP3A4*16b harboring the Thr185Ser substitution on paclitaxel metabolism in japanese patients with cancer. Clin. Pharmacol. Ther. 2006, 80, 179–191. [Google Scholar] [CrossRef]
- Murayama, N.; Nakamura, T.; Saeki, M.; Soyama, A.; Saito, Y.; Sai, K.; Ishida, S.; Nakajima, O.; Itoda, M.; Ohno, Y.; et al. CYP3A4 gene polymorphisms influence testosterone 6beta-hydroxylation. Drug Metab. Pharmacokinet. 2002, 17, 150–156. [Google Scholar] [CrossRef]
- Hsieh, K.P.; Lin, Y.Y.; Cheng, C.L.; Lai, M.L.; Lin, M.S.; Siest, J.P.; Huang, J.D. Novel mutations of CYP3A4 in Chinese. Drug Metab. Dispos. 2001, 29, 268–273. [Google Scholar]
- Wang, A.; Yu, B.N.; Luo, C.H.; Tan, Z.R.; Zhou, G.; Wang, L.S.; Zhang, W.; Li, Z.; Liu, J.; Zhou, H.H. Ile118Val genetic polymorphism of CYP3A4 and its effects on lipid-lowering efficacy of simvastatin in chinese hyperlipidemic patients. Eur. J. Clin. Pharmacol. 2005, 60, 843–848. [Google Scholar] [CrossRef]
- Kang, Y.S.; Park, S.Y.; Yim, C.H.; Kwak, H.S.; Gajendrarao, P.; Krishnamoorthy, N.; Yun, S.C.; Lee, K.W.; Han, K.O. The CYP3A4*18 genotype in the cytochrome P450 3A4 gene, a rapid metabolizer of sex steroids, is associated with low bone mineral density. Clin. Pharmacol. Ther. 2009, 85, 312–318. [Google Scholar] [CrossRef]
- Hu, Y.F.; Tu, J.H.; Tan, Z.R.; Liu, Z.Q.; Zhou, G.; He, J.; Wang, D.; Zhou, H.H. Association of CYP3A4*18B polymorphisms with the pharmacokinetics of cyclosporine in healthy subjects. Xenobiotica 2007, 37, 315–327. [Google Scholar] [CrossRef]
- Shi, X.J.; Geng, F.; Jiao, Z.; Cui, X.Y.; Qiu, X.Y.; Zhong, M.K. Association of ABCB1, CYP3A4*18B and CYP3A5*3 genotypes with the pharmacokinetics of tacrolimus in healthy Chinese subjects: A population pharmacokinetic analysis. J. Clin. Pharm. Ther. 2011, 36, 614–624. [Google Scholar] [CrossRef]
- Qiu, X.Y.; Jiao, Z.; Zhang, M.; Zhong, L.J.; Liang, H.Q.; Ma, C.L.; Zhang, L.; Zhong, M.K. Association of MDR1, CYP3A4*18B, and CYP3A5*3 polymorphisms with cyclosporine pharmacokinetics in Chinese renal transplant recipients. Eur. J. Clin. Pharmacol. 2008, 64, 1069–1084. [Google Scholar] [CrossRef]
- Westlind-Johnsson, A.; Hermann, R.; Huennemeyer, A.; Hauns, B.; Lahu, G.; Nassr, N.; Zech, K.; Ingelman-Sundberg, M.; von Richter, O. Identification and characterization of CYP3A4*20, a novel rare CYP3A4 allele without functional activity. Clin. Pharmacol. Ther. 2006, 79, 339–349. [Google Scholar] [CrossRef]
- Busi, F.; Cresteil, T. CYP3A5 mRNA degradation by nonsense-mediated mRNA decay. Mol. Pharmacol. 2005, 68, 808–815. [Google Scholar]
- Anglicheau, D.; Legendre, C.; Beaune, P.; Thervet, E. Cytochrome P450 3A polymorphisms and immunosuppressive drugs: An update. Pharmacogenomics 2007, 8, 835–849. [Google Scholar] [CrossRef]
- Martinez-Jimenez, C.P.; Jover, R.; Donato, M.T.; Castell, J.V.; Gomez-Lechon, M.J. Transcriptional regulation and expression of CYP3A4 in hepatocytes. Curr. Drug Metab. 2007, 8, 185–194. [Google Scholar] [CrossRef]
- Tirona, R.G.; Lee, W.; Leake, B.F.; Lan, L.B.; Cline, C.B.; Lamba, V.; Parviz, F.; Duncan, S.A.; Inoue, Y.; Gonzalez, F.J.; et al. The orphan nuclear receptor HNF4alpha determines PXR- and CAR-mediated xenobiotic induction of CYP3A4. Nat. Med. 2003, 9, 220–224. [Google Scholar] [CrossRef]
- Rodriguez-Antona, C.; Bort, R.; Jover, R.; Tindberg, N.; Ingelman-Sundberg, M.; Gomez-Lechon, M.J.; Castell, J.V. Transcriptional regulation of human CYP3A4 basal expression by CCAAT enhancer-binding protein alpha and hepatocyte nuclear factor-3 gamma. Mol. Pharmacol. 2003, 63, 1180–1189. [Google Scholar] [CrossRef]
- Lim, Y.P.; Huang, J.D. Interplay of pregnane X receptor with other nuclear receptors on gene regulation. Drug Metab. Pharmacokinet. 2008, 23, 14–21. [Google Scholar] [CrossRef]
- Lamba, V.; Panetta, J.C.; Strom, S.; Schuetz, E.G. Genetic predictors of interindividual variability in hepatic CYP3A4 expression. J. Pharmacol. Exp. Ther. 2010, 332, 1088–1099. [Google Scholar] [CrossRef]
- Lamba, V.; Yasuda, K.; Lamba, J.K.; Assem, M.; Davila, J.; Strom, S.; Schuetz, E.G. PXR (NR1I2): Splice variants in human tissues, including brain, and identification of neurosteroids and nicotine as PXR activators. Toxicol. Appl. Pharmacol. 2004, 199, 251–265. [Google Scholar] [CrossRef]
- Lamba, J.; Lamba, V.; Schuetz, E. Genetic variants of PXR (NR1I2) and CAR (NR1I3) and their implications in drug metabolism and pharmacogenetics. Curr. Drug Metab. 2005, 6, 369–383. [Google Scholar] [CrossRef]
- He, P.; Court, M.H.; Greenblatt, D.J.; von Moltke, L.L. Human pregnane X receptor: Genetic polymorphisms, alternative mRNA splice variants, and cytochrome P450 3A metabolic activity. J. Clin. Pharmacol. 2006, 46, 1356–1369. [Google Scholar] [CrossRef]
- Lamba, J.; Lamba, V.; Strom, S.; Venkataramanan, R.; Schuetz, E. Novel single nucleotide polymorphisms in the promoter and intron 1 of human pregnane X receptor/NR1I2 and their association with CYP3A4 expression. Drug Metab. Dispos. 2008, 36, 169–181. [Google Scholar]
- Schipani, A.; Siccardi, M.; D'Avolio, A.; Baietto, L.; Simiele, M.; Bonora, S.; Rodriguez Novoa, S.; Cuenca, L.; Soriano, V.; Chierakul, N.; et al. Population pharmacokinetic modeling of the association between 63396C→T pregnane X receptor polymorphism and unboosted atazanavir clearance. Antimicrob. Agents Chemother. 2010, 54, 5242–5250. [Google Scholar] [CrossRef]
- Sandanaraj, E.; Lal, S.; Selvarajan, V.; Ooi, L.L.; Wong, Z.W.; Wong, N.S.; Ang, P.C.; Lee, E.J.; Chowbay, B. PXR pharmacogenetics: Association of haplotypes with hepatic CYP3A4 and ABCB1 messenger RNA expression and doxorubicin clearance in Asian breast cancer patients. Clin. Cancer Res. 2008, 14, 7116–7126. [Google Scholar] [CrossRef]
- Hor, S.Y.; Lee, S.C.; Wong, C.I.; Lim, Y.W.; Lim, R.C.; Wang, L.Z.; Fan, L.; Guo, J.Y.; Lee, H.S.; Goh, B.C.; et al. PXR, CAR and HNF4alpha genotypes and their association with pharmacokinetics and pharmacodynamics of docetaxel and doxorubicin in Asian patients. Pharmacogenomics J. 2008, 8, 139–146. [Google Scholar] [CrossRef]
- Tham, L.S.; Holford, N.H.; Hor, S.Y.; Tan, T.; Wang, L.; Lim, R.C.; Lee, H.S.; Lee, S.C.; Goh, B.C. Lack of association of single-nucleotide polymorphisms in pregnane X receptor, hepatic nuclear factor 4alpha, and constitutive androstane receptor with docetaxel pharmacokinetics. Clin. Cancer Res. 2007, 13, 7126–7132. [Google Scholar] [CrossRef]
- DeKeyser, J.G.; Stagliano, M.C.; Auerbach, S.S.; Prabhu, K.S.; Jones, A.D.; Omiecinski, C.J. Di(2-ethylhexyl) phthalate is a highly potent agonist for the human constitutive androstane receptor splice variant CAR2. Mol. Pharmacol. 2009, 75, 1005–1013. [Google Scholar] [CrossRef]
- Lin, Y.S.; Yasuda, K.; Assem, M.; Cline, C.; Barber, J.; Li, C.W.; Kholodovych, V.; Ai, N.; Chen, J.D.; Welsh, W.J.; et al. The major human pregnane X receptor (PXR) splice variant, PXR.2, exhibits significantly diminished ligand-activated transcriptional regulation. Drug Metab. Dispos. 2009, 37, 1295–1304. [Google Scholar] [CrossRef]
- Pan, Y.Z.; Gao, W.; Yu, A.M. Micrornas regulate CYP3A4 expression via direct and indirect targeting. Drug Metab. Dispos. 2009, 37, 2112–2117. [Google Scholar] [CrossRef]
- Takagi, S.; Nakajima, M.; Mohri, T.; Yokoi, T. Post-transcriptional regulation of human pregnane X receptor by micro-RNA affects the expression of cytochrome P450 3A4. J. Biol. Chem. 2008, 283, 9674–9680. [Google Scholar] [CrossRef]
- Xie, Y.; Ke, S.; Ouyang, N.; He, J.; Xie, W.; Bedford, M.T.; Tian, Y. Epigenetic regulation of transcriptional activity of pregnane X receptor by protein arginine methyltransferase 1. J. Biol. Chem. 2009, 284, 9199–9205. [Google Scholar]
- Dannenberg, L.O.; Edenberg, H.J. Epigenetics of gene expression in human hepatoma cells: Expression profiling the response to inhibition of DNA methylation and histone deacetylation. BMC Genomics 2006, 7. [Google Scholar] [CrossRef]
- Thervet, E.; Loriot, M.A.; Barbier, S.; Buchler, M.; Ficheux, M.; Choukroun, G.; Toupance, O.; Touchard, G.; Alberti, C.; Le Pogamp, P.; et al. Optimization of initial tacrolimus dose using pharmacogenetic testing. Clin. Pharmacol. Ther. 2010, 87, 721–726. [Google Scholar]
- Wang, P.; Mao, Y.; Razo, J.; Zhou, X.; Wong, S.T.; Patel, S.; Elliott, E.; Shea, E.; Wu, A.H.; Gaber, A.O. Using genetic and clinical factors to predict tacrolimus dose in renal transplant recipients. Pharmacogenomics 2010, 11, 1389–1402. [Google Scholar] [CrossRef]
- Haufroid, V.; Mourad, M.; van Kerckhove, V.; Wawrzyniak, J.; de Meyer, M.; Eddour, D.C.; Malaise, J.; Lison, D.; Squifflet, J.P.; Wallemacq, P. The effect of CYP3A5 and MDR1 (ABCB1) polymorphisms on cyclosporine and tacrolimus dose requirements and trough blood levels in stable renal transplant patients. Pharmacogenetics 2004, 14, 147–154. [Google Scholar] [CrossRef]
- Press, R.R.; Ploeger, B.A.; den Hartigh, J.; van der Straaten, T.; van Pelt, J.; Danhof, M.; de Fijter, J.W.; Guchelaar, H.J. Explaining variability in tacrolimus pharmacokinetics to optimize early exposure in adult kidney transplant recipients. Ther. Drug Monit. 2009, 31, 187–197. [Google Scholar] [CrossRef]
- Jun, K.R.; Lee, W.; Jang, M.S.; Chun, S.; Song, G.W.; Park, K.T.; Lee, S.G.; Han, D.J.; Kang, C.; Cho, D.Y.; et al. Tacrolimus concentrations in relation to CYP3A and ABCB1 polymorphisms among solid organ transplant recipients in Korea. Transplantation 2009, 87, 1225–1231. [Google Scholar] [CrossRef]
- Hesselink, D.A.; van Schaik, R.H.; van der Heiden, I.P.; van der Werf, M.; Gregoor, P.J.; Lindemans, J.; Weimar, W.; van Gelder, T. Genetic polymorphisms of the CYP3A4, CYP3A5, and MDR-1 genes and pharmacokinetics of the calcineurin inhibitors cyclosporine and tacrolimus. Clin. Pharmacol. Ther. 2003, 74, 245–254. [Google Scholar] [CrossRef]
- Op den Buijsch, R.A.; Christiaans, M.H.; Stolk, L.M.; de Vries, J.E.; Cheung, C.Y.; Undre, N.A.; van Hooff, J.P.; van Dieijen-Visser, M.P.; Bekers, O. Tacrolimus pharmacokinetics and pharmacogenetics: Influence of adenosine triphosphate-binding cassette B1 (ABCB1) and cytochrome (CYP) 3A polymorphisms. Fundam. Clin. Pharmacol. 2007, 21, 427–435. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, D.; Sadee, W. The Making of a CYP3A Biomarker Panel for Guiding Drug Therapy. J. Pers. Med. 2012, 2, 175-191. https://doi.org/10.3390/jpm2040175
Wang D, Sadee W. The Making of a CYP3A Biomarker Panel for Guiding Drug Therapy. Journal of Personalized Medicine. 2012; 2(4):175-191. https://doi.org/10.3390/jpm2040175
Chicago/Turabian StyleWang, Danxin, and Wolfgang Sadee. 2012. "The Making of a CYP3A Biomarker Panel for Guiding Drug Therapy" Journal of Personalized Medicine 2, no. 4: 175-191. https://doi.org/10.3390/jpm2040175





