A Novel Point-of-Care Biomarker Recognition Method: Validation by Detecting Marker for Diabetic Nephropathy
Abstract
:1. Introduction
1.1. Methodology Development and Design of the Biomarker Detection Kit
- (1)
- A nitrocellulose or PVDF strip that is coated with a capture antibody (AB1) that can bind to a specific biomarker in the body fluid to be tested.
- (2)
- A tube containing a wash solution to remove other proteins non-specifically binding to the strip.
- (3)
- A tube containing solution of gold nano beads or latex beads adsorbed with detection antibody (AB2) directed against the same biomarker protein.
1.2. Procedure
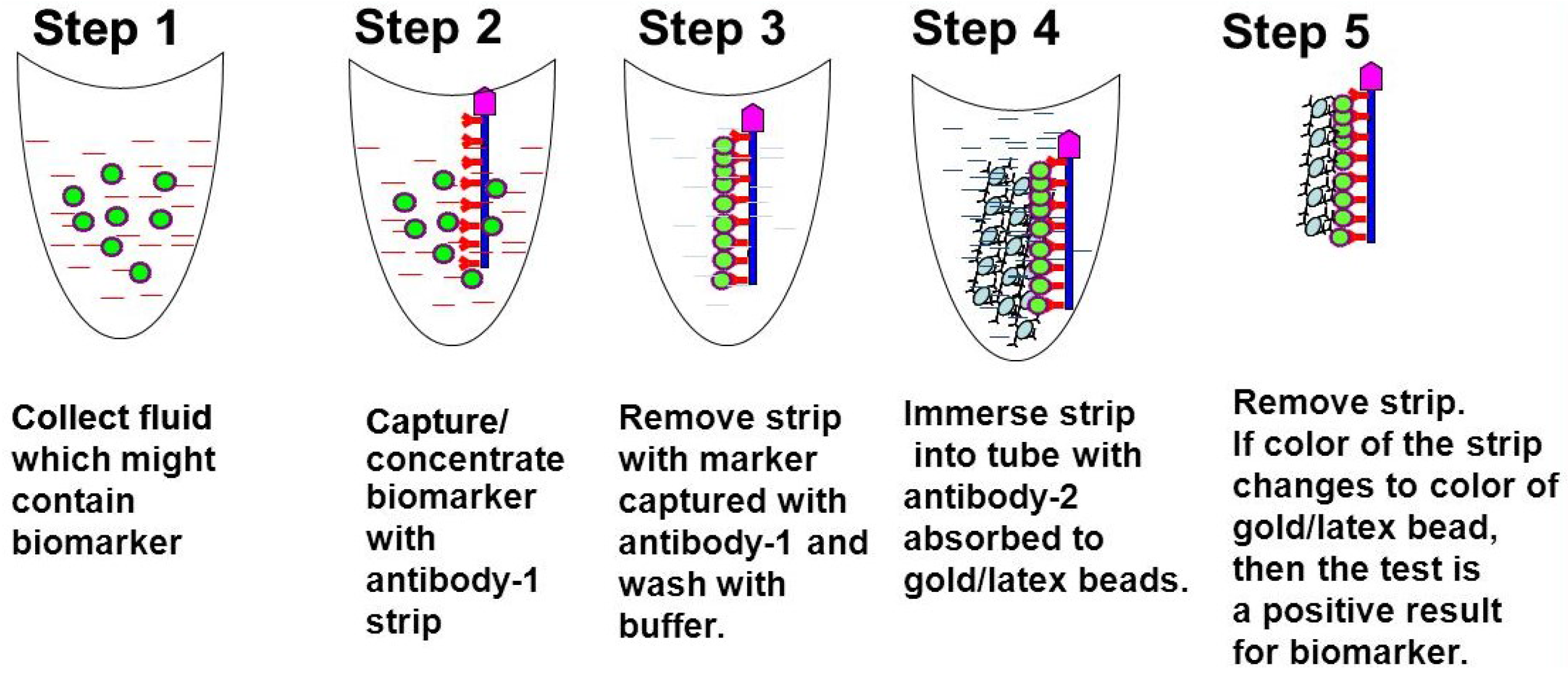
1.3. Diabetic Nephropathy and Lipocalin Type Prostaglandin D2-synthase (LPGDS)
Validation of the Biomarker Detection Methodology by Detecting a Specific Biomarker for Diabetic Nephropathy
2. Materials and Methods
- Hybridoma screening;
- Specificity of AB1 and AB2;
- Adsorption of capture antibody (AB1) to nanogold beads; and
- Detection of LPGDS marker in mock urine.
3. Results
3.1. Hybridoma Screening

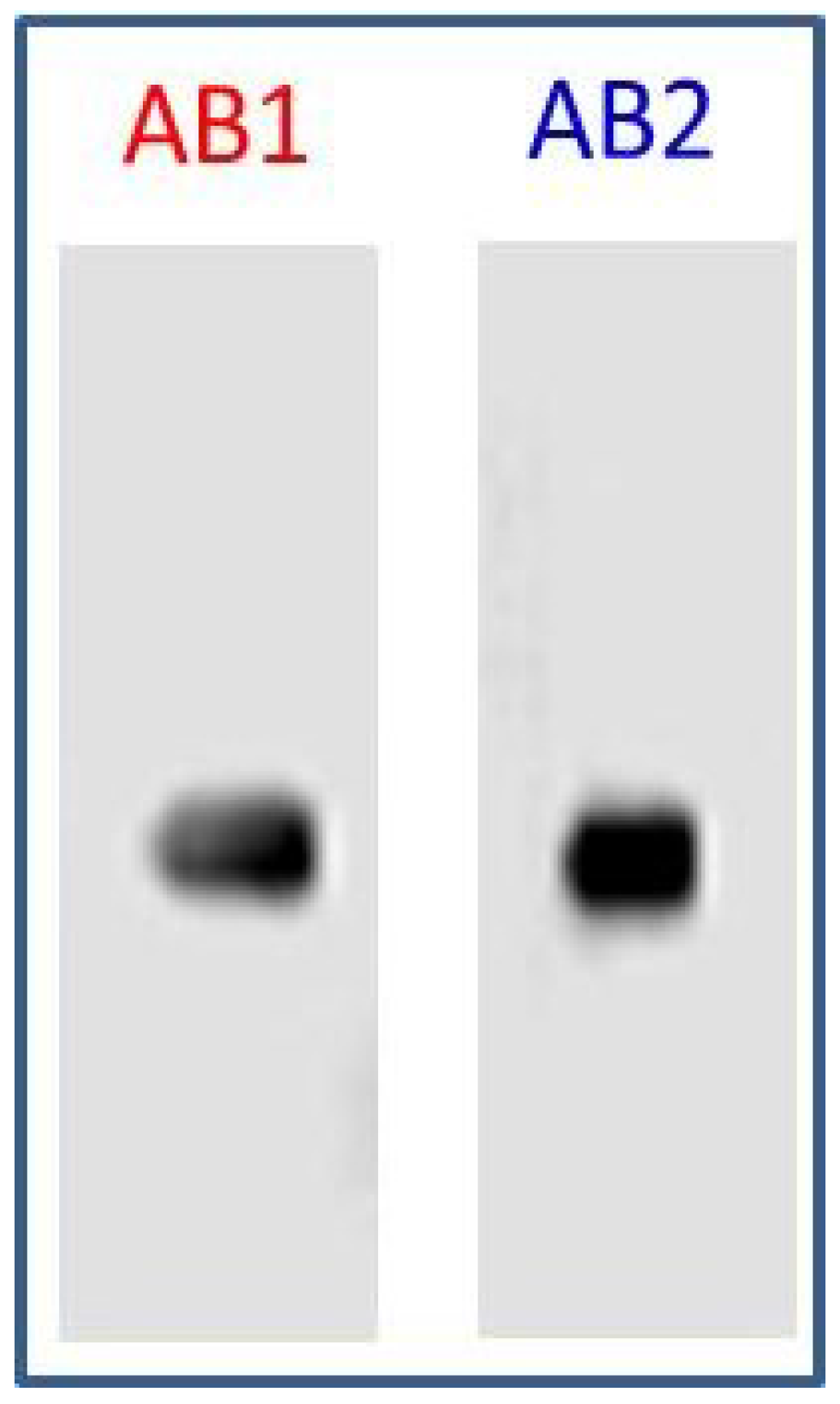
3.2. Specificity of AB1 and AB2
3.3. Adsorption of Detection Antibody (AB2) to Nanogold Beads

3.4. Detection of LPGDS (Diabetic Nephropathy Marker) in Mock Urine
3.4.1. Preparation of Test Strips with LPGDS Antibody
3.4.2. Detection of Disease Marker (LPGDS) in Urine Samples


4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Lathrop, J.T.; Hayes, T.K.; Carrick, K.; Hammond, D.J. Rarity gives a charm: Evaluation of trace proteins in plasma and serum. Expert Rev. Proteomics 2005, 2, 393–406. [Google Scholar] [PubMed]
- Worsley, G.J.; Attree, S.L.; Noble, J.E.; Horgan, A.M. Rapid duplex immunoassay for wound biomarkers at the point-of-care. Biosens. Bioelectron. 2012, 34, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Merenbloom, B.K.; Oberhardt, B.J. Homogeneous immunoassay of whole-blood samples. Clin. Chem. 1995, 41, 1385–1390. [Google Scholar] [PubMed]
- Bokenkamp, A.; Franke, I.; Schlieber, M.; Duker, G.; Schmitt, J.; Buderus, S.; Lentze, M.J.; Stoffel-Wagner, B. Beta-trace protein—A marker of kidney function in children: “Original research communication-clinical investigation”. Clin. Biochem. 2007, 40, 969–975. [Google Scholar] [PubMed]
- Kristensen, K.; Wide-Swensson, D.; Schmidt, C.; Blirup-Jensen, S.; Lindstrom, V.; Strevens, H.; Grubb, A. Cystatin C, beta-2-microglobulin and beta-trace protein in pre-eclampsia. Acta Obstet. Gynecol. Scand. 2007, 86, 921–926. [Google Scholar] [CrossRef] [PubMed]
- Hansmeier, N.; Chao, T.C.; Goldman, L.R.; Witter, F.R.; Halden, R.U. Prioritization of biomarker targets in human umbilical cord blood: Identification of proteins in infant blood serving as validated biomarkers in adults. Environ. Health Perspect. 2012, 120, 764–769. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Wang, K.; Zhang, L.; Li, C.; Yang, Y. The discovery of putative urine markers for the specific detection of prostate tumor by integrative mining of public genomic profiles. PLoS ONE 2011, 6, e28552. [Google Scholar] [CrossRef] [PubMed]
- Ghasemzadeh, N.; Wilhelmsen, T.W.; Nyberg, F.; Hjerten, S. Precautions to improve the accuracy of quantitative determinations of biomarkers in clinical diagnostics. Electrophoresis 2010, 31, 2722–2729. [Google Scholar] [CrossRef] [PubMed]
- Fenton, E.M.; Mascarenas, M.R.; Lopez, G.P.; Sibbett, S.S. Multiplex lateral-flow test strips fabricated by two-dimensional shaping. ACS Appl. Mater. Interfaces 2009, 1, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Dixit, C.K.; Kaushik, A. Nano-structured arrays for multiplex analyses and Lab-on-a-Chip applications. Biochem. Biophys. Res. Commun. 2012, 419, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Yu, X.; Sun, M.; Wang, H.; Xu, S.; Dixon, J.D.; Wang, Y.A.; Li, Y.; Yang, Q.; Xu, X. Preparation of quantum dots encoded microspheres by electrospray for the detection of biomolecules. J. Colloid Interface Sci. 2011, 358, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Jun, B.H.; Kang, H.; Lee, Y.S.; Jeong, D.H. Fluorescence-based multiplex protein detection using optically encoded microbeads. Molecules 2012, 17, 2474–2490. [Google Scholar] [CrossRef] [PubMed]
- Ito, H.; Yan, X.; Nagata, N.; Aritake, K.; Katsumata, Y.; Matsuhashi, T.; Nakamura, M.; Hirai, H.; Urade, Y.; Asano, K.; et al. PGD2-CRTH2 pathway promotes tubulointerstitial fibrosis. J. Am. Soc. Nephrol. 2012, 23, 1797–1809. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, H.; Echizen, H.; Gomi, T.; Shibuya, Y.; Nakamura, Y.; Nakano, K.; Arashi, H.; Itai, T.; Ohnishi, S.; Tanaka, M.; et al. Urinary lipocalin-type prostaglandin D synthase: A potential marker for early gentamicin-induced renal damage? Ther. Drug Monit. 2009, 31, 126–130. [Google Scholar] [CrossRef] [PubMed]
- Uehara, Y.; Makino, H.; Seiki, K.; Urade, Y. L-PGDS Clinical Research Group of Kidney. Urinary excretions of lipocalin-type prostaglandin D synthase predict renal injury in type-2 diabetes: A cross-sectional and prospective multicentre study. Nephrol. Dial. Transplant. 2009, 24, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Oda, H.; Shiina, Y.; Seiki, K.; Sato, N.; Eguchi, N.; Urade, Y. Development and evaluation of a practical ELISA for human urinary lipocalin-type prostaglandin D synthase. Clin. Chem. 2002, 48, 1445–1453. [Google Scholar] [PubMed]
- Pentyala, S. Method and Kits for Detecting Cerebrospinal Fluid in a Sample. US Patent 7794953, 14 September 2010. [Google Scholar]
- Pentyala, S. Methods, Antibodies and Kits for Detecting Cerebrospinal Fluid in a Sample. US Patent 8003765, 23 August 2011. [Google Scholar]
- Ankarberg-Lindgren, C.; Norjavaara, E. Sensitive RIA measures testosterone concentrations in prepubertal and pubertal children comparable to tandem mass spectrometry. Scand. J. Clin. Lab. Invest. 2015. [Google Scholar] [CrossRef]
- Chan, P.C.; Chen, J. Value of reflex testing based on hypogammaglobulinemia as demonstrated in serum protein electrophoresis. Clin. Biochem. 2015. [Google Scholar] [CrossRef]
- Bloch, E.M.; Sakac, D.; Branch, H.A.; Cserti-Gazdewich, C.; Pendergrast, J.; Pavenski, K.; Branch, D.R. Western immunoblotting as a new tool for investigating direct antiglobulin test-negative autoimmune hemolytic anemias. Transfusion 2015. [Google Scholar] [CrossRef]
- Polanski, M.; Anderson, N.L. A list of candidate cancer biomarkers for targeted proteomics. Biomark Insights 2007, 1, 1–48. [Google Scholar] [PubMed]
- Hudecova, I. Digital PCR analysis of circulating nucleic acids. Clin. Biochem. 2015. [Google Scholar] [CrossRef]
- Wilson, A.D. Advances in electronic-nose technologies for the detection of volatile biomarker metabolites in the human breath. Metabolites 2015, 5, 140–163. [Google Scholar] [CrossRef] [PubMed]
- Guo, A.; Li, Y.; Cao, W.; Meng, X.; Wu, D.; Wei, Q.; Du, B. An electrochemical immunosensor for ultrasensitive detection of carbohydrate antigen 199 based on Au@CuxOS yolk–shell nanostructures with porous shells as labels. Biosens. Bioelectron. 2015, 63, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Vashist, S.K.; Mudanyali, O.; Schneider, E.M.; Zengerle, R.; Ozcan, A. Cellphone-based devices for bioanalytical sciences. Anal. Bioanal. Chem. 2014, 406, 3263–3277. [Google Scholar] [CrossRef] [PubMed]
- Vashist, S.K.; van Oordt, T.; Schneider, E.M.; Zengerle, R.; von Stetten, F.; Luong, J.H. A smartphone-based colorimetric reader for bioanalytical applications using the screen-based bottom illumination provided by gadgets. Biosens. Bioelectron. 2015, 67, 248–255. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pentyala, S.; Muller, J.; Tumillo, T.; Roy, A.; Mysore, P.; Pentyala, S. A Novel Point-of-Care Biomarker Recognition Method: Validation by Detecting Marker for Diabetic Nephropathy. Diagnostics 2015, 5, 177-188. https://doi.org/10.3390/diagnostics5020177
Pentyala S, Muller J, Tumillo T, Roy A, Mysore P, Pentyala S. A Novel Point-of-Care Biomarker Recognition Method: Validation by Detecting Marker for Diabetic Nephropathy. Diagnostics. 2015; 5(2):177-188. https://doi.org/10.3390/diagnostics5020177
Chicago/Turabian StylePentyala, Sahana, John Muller, Thomas Tumillo, Avijit Roy, Pooja Mysore, and Srinivas Pentyala. 2015. "A Novel Point-of-Care Biomarker Recognition Method: Validation by Detecting Marker for Diabetic Nephropathy" Diagnostics 5, no. 2: 177-188. https://doi.org/10.3390/diagnostics5020177
APA StylePentyala, S., Muller, J., Tumillo, T., Roy, A., Mysore, P., & Pentyala, S. (2015). A Novel Point-of-Care Biomarker Recognition Method: Validation by Detecting Marker for Diabetic Nephropathy. Diagnostics, 5(2), 177-188. https://doi.org/10.3390/diagnostics5020177




