Protocells Models in Origin of Life and Synthetic Biology
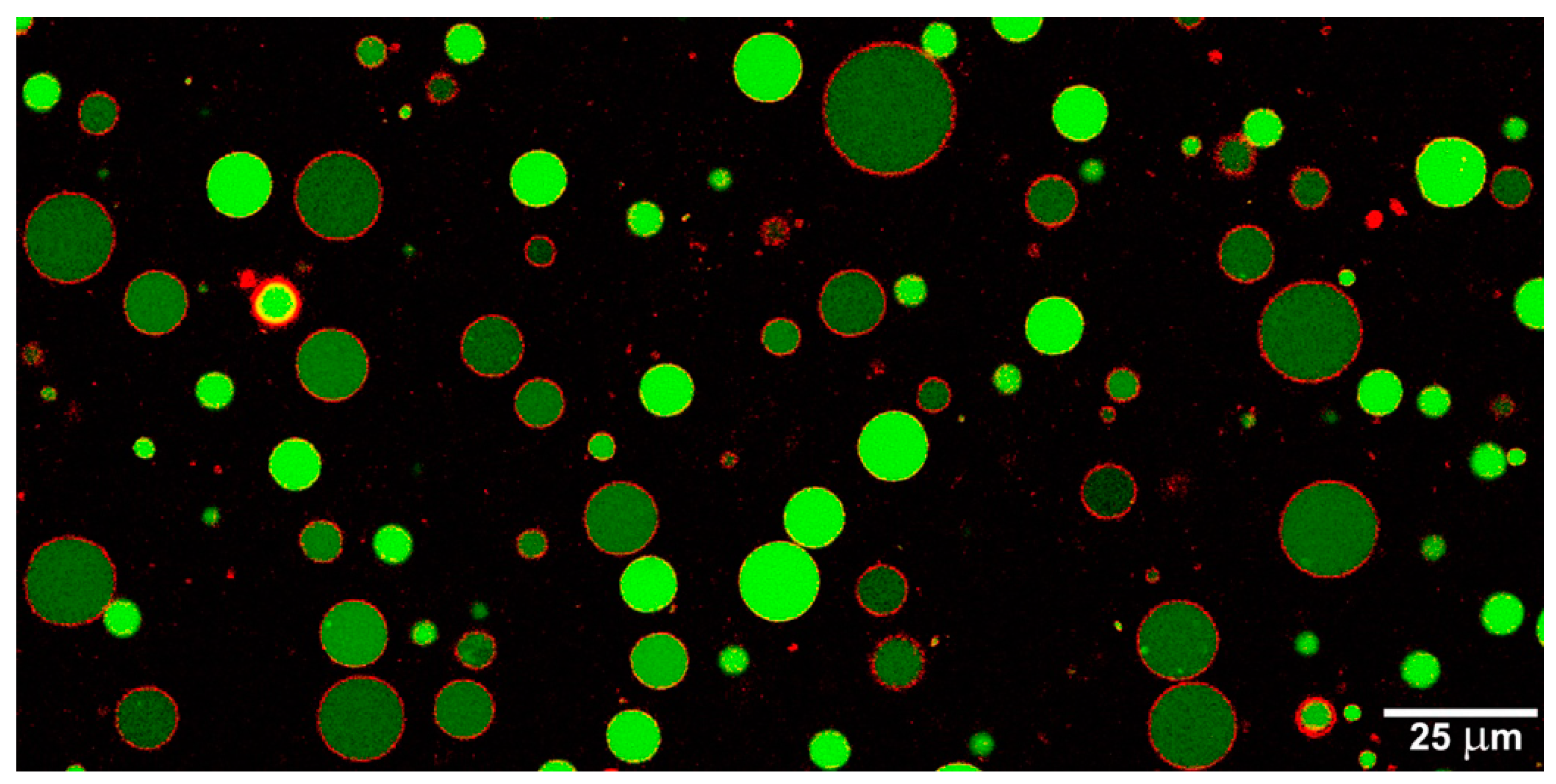
Acknowledgments
Conflicts of Interest
References
- Monnard, P.-A.; Deamer, D.W. Membrane self-assembly processes: Steps toward the first cellular life. Anat. Rec. 2002, 268, 196–207. [Google Scholar] [CrossRef] [PubMed]
- Luisi, P.L.; Ferri, F.; Stano, P. Approaches to semi-synthetic minimal cells: A review. Naturwissenschaften 2006, 93, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Mansy, S.S.; Szostak, J.W. Reconstructing the emergence of cellular life through the synthesis of model protocells. Cold Spring Harb. Symp. Quant. Biol. 2009, 74, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, K.; Okura, Y.; Matsuo, M.; Toyota, T.; Suzuki, K.; Sugawara, T. A recursive vesicle-based model protocell with a primitive model cell cycle. Nat. Commun. 2015, 6, 8352. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Sato, K.; Wakabayashi, M.; Nakaishi, T.; Ko-Mitamura, E.P.; Shima, Y.; Urabe, I.; Yomo, T. Synthesis of functional protein in liposome. J. Biosci. Bioeng. 2001, 92, 590–593. [Google Scholar] [CrossRef]
- Noireaux, V.; Libchaber, A. A vesicle bioreactor as a step toward an artificial cell assembly. Proc. Natl. Acad. Sci. USA 2004, 101, 17669–17674. [Google Scholar] [CrossRef] [PubMed]
- Stano, P.; Carrara, P.; Kuruma, Y.; Souza, T.P.; Luisi, P.L. Compartmentalized reactions as a case of soft-matter biotechnology: Synthesis of proteins and nucleic acids inside lipid vesicles. J. Mater. Chem. 2011, 21, 18887–18902. [Google Scholar] [CrossRef]
- Nourian, Z.; Danelon, C. Linking genotype and phenotype in protein synthesizing liposomes with external supply of resources. ACS Synth. Biol. 2013, 2, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Stano, P.; D’Aguanno, E.; Bolz, J.; Fahr, A.; Luisi, P.L. A Remarkable Self-Organization Process as the Origin of Primitive Functional Cells. Angew. Chem. Int. Ed. 2013, 52, 13397–13400. [Google Scholar] [CrossRef] [PubMed]
- Lentini, R.; Santero, S.P.; Chizzolini, F.; Cecchi, D.; Fontana, J.; Marchioretto, M.; del Bianco, C.; Terrell, J.L.; Spencer, A.C.; Martini, L.; et al. Integrating artificial with natural cells to translate chemical messages that direct E. coli behaviour. Nat. Commun. 2014, 5, 4012. [Google Scholar] [CrossRef] [PubMed]
- Gánti, T. Chemoton Theory: Theory of Living Systems; Kluver Academic/Plenum Publishers: New York, NY, USA, 2003. [Google Scholar]
- Varela, F.G.; Maturana, H.R.; Uribe, R. Autopoiesis: The organization of living systems, its characterization and a model. Biosystems 1974, 5, 187–196. [Google Scholar] [CrossRef]
- Martino, C.; Kim, S.-H.; Horsfall, L.; Abbaspourrad, A.; Rosser, S.J.; Cooper, J.; Weitz, D.A. Protein Expression, Aggregation, and Triggered Release from Polymersomes as Artificial Cell-like Structures. Angew. Chem. Int. Ed. 2012, 51, 6416–6420. [Google Scholar] [CrossRef] [PubMed]
- Tang, T.-Y.D.; van Swaay, D.; deMello, A.; Anderson, J.L.R.; Mann, S. In vitro gene expression within membrane-free coacervate protocells. Chem. Commun. 2015, 51, 11429–11432. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, S.; Chen, L.; Deamer, D.; Krakauer, D.C.; Packard, N.H.; Stadler, P.F.; Bedau, M.A. Evolution. Transitions from nonliving to living matter. Science 2004, 303, 963–965. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, K. Life: An Introduction to Complex Systems Biology; Understanding Complex Systems; Springer: Berlin, Germany; Heidelberg, Germany, 2006. [Google Scholar]
- Damiano, L.; Cañamero, L. The Frontier of Synthetic Knowledge: Toward a Constructivist Science. World Futures 2012, 68, 171–177. [Google Scholar] [CrossRef]
- De Lorenzo, V.; Danchin, A. Synthetic biology: Discovering new worlds and new words. EMBO Rep. 2008, 9, 822–827. [Google Scholar] [CrossRef] [PubMed]
- Mavelli, F.; Ruiz-Mirazo, K. Stochastic simulations of minimal self-reproducing cellular systems. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2007, 362, 1789–1802. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.; Noireaux, V. An E. coli Cell-Free Expression Toolbox: Application to Synthetic Gene Circuits and Artificial Cells. ACS Synth. Biol. 2012, 1, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Stögbauer, T.; Windhager, L.; Zimmer, R.; Rädler, J.O. Experiment and mathematical modeling of gene expression dynamics in a cell-free system. Integr. Biol. 2012, 4, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Calviello, L.; Stano, P.; Mavelli, F.; Luisi, P.L.; Marangoni, R. Quasi-cellular systems: Stochastic simulation analysis at nanoscale range. BMC Bioinform. 2013, 14, S7. [Google Scholar] [CrossRef]
- Shirt-Ediss, B.; Ruiz-Mirazo, K.; Mavelli, F.; Solé, R.V. Modelling lipid competition dynamics in heterogeneous protocell populations. Sci. Rep. 2014, 4, 5675. [Google Scholar] [CrossRef] [PubMed]
- Siegal-Gaskins, D.; Tuza, Z.A.; Kim, J.; Noireaux, V.; Murray, R.M. Gene circuit performance characterization and resource usage in a cell-free “breadboard”. ACS Synth. Biol. 2014, 3, 416–425. [Google Scholar] [CrossRef] [PubMed]
- Walde, P.; Cosentino, K.; Engel, H.; Stano, P. Giant vesicles: Preparations and applications. ChemBioChem 2010, 11, 848–865. [Google Scholar] [CrossRef] [PubMed]
- Von Kiedrowski, G.; Otto, S.; Herdewijn, P. Welcome Home, Systems Chemists! J. Syst. Chem. 2010, 1, 1–6. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stano, P.; Mavelli, F. Protocells Models in Origin of Life and Synthetic Biology. Life 2015, 5, 1700-1702. https://doi.org/10.3390/life5041700
Stano P, Mavelli F. Protocells Models in Origin of Life and Synthetic Biology. Life. 2015; 5(4):1700-1702. https://doi.org/10.3390/life5041700
Chicago/Turabian StyleStano, Pasquale, and Fabio Mavelli. 2015. "Protocells Models in Origin of Life and Synthetic Biology" Life 5, no. 4: 1700-1702. https://doi.org/10.3390/life5041700
APA StyleStano, P., & Mavelli, F. (2015). Protocells Models in Origin of Life and Synthetic Biology. Life, 5(4), 1700-1702. https://doi.org/10.3390/life5041700






