Ecology and Physiology of the Pathogenic Cyanobacterium Roseofilum reptotaenium
Abstract
:1. Introduction
1.1. Cyanobacteria and Black Band Disease of Corals


1.2. The Ecology of Caribbean Black Band Disease
2. Methods
2.1. Isolation of Roseofilum reptotaenium Strains
2.2. Infection of Coral Fragments
2.3. Scanning Electron Microscopy
2.4. Motility Patterns
2.5. Growth and Clumping as a Factor of pH
2.6. Microcystin Exposure and Metabolite Production
2.7. Metabolite Extraction and Metabolomic Analysis Using UPLC-HRMS
2.8. Metabolomic Data Processing and Analysis
3. Results
3.1. Laboratory Infection Using R. reptotaenium

3.2. Microscopic Analysis

3.3. Chemotactic Responses to Sulfide and Microcystin
| Test Substance and Light Source | Distance from Sulfide or Microcystin Source (cm) | p Value |
|---|---|---|
| Directional light | ||
| Sulfide | 3.1 ± 1.7 | |
| Control | 1.8 ± 3.1 | <0.05 |
| MC-LR (1 µg/L) | 1.2 ± 1.1 | |
| MC-LR (50 µg/L) | 1.1 ± 1.1 | |
| MC-LR (100 µg/L) | 1.2 ± 1.7 | |
| Control (MC-LR) | 1.3 ± 1.3 | 0.99 |
| Diffuse light | ||
| MC-LR (1 µg/L) | 3.5 ± 1.0 | |
| MC-LR (50 µg/L) | 3.5 ± 0.5 | |
| MC-LR (100 µg/L) | 3.6 ± 0.3 | |
| Control | 3.6 ± 0.5 | 0.75 |
3.4. Clumping Behavior



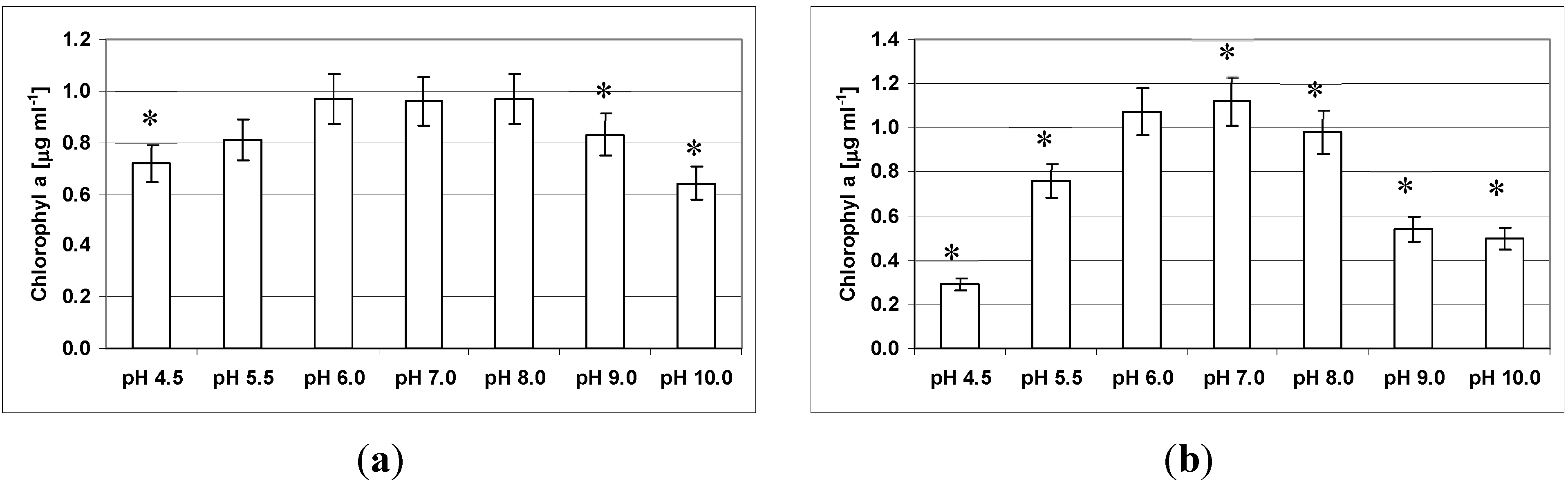
3.5. Effect of pH on Growth and Clumping
3.6. Effect of Microcystin on Metabolite Production in the BBD Community
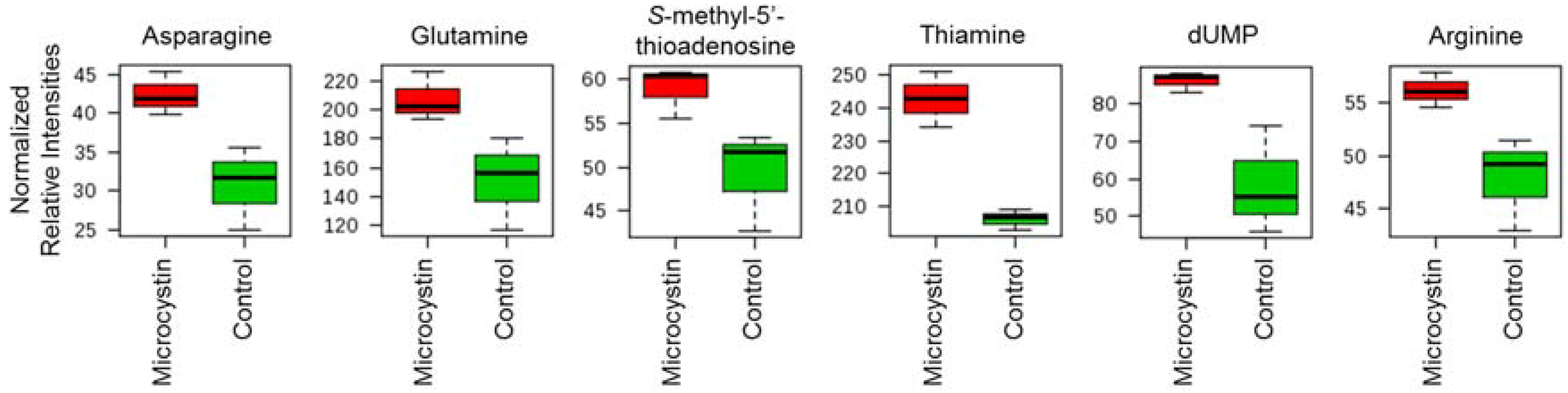
| Metabolite | Fold Change | p Value |
|---|---|---|
| N,N-Dimethylglycine | 1.299 | 0.012 |
| Betaine | 1.281 | 0.051 |
| Thymine | 1.703 | 0.082 |
| l-Asparagine | 2.486 | 0.020 |
| l-Glutamine | 2.392 | 0.044 |
| l-Glutamate | 1.378 | 0.005 |
| Xanthine | 1.388 | 0.103 |
| l-Arginine | 1.588 | 0.039 |
| N-Carbamoyl-l-Aspartate | 1.349 | 0.086 |
| Deoxyuridine | 2.015 | 0.056 |
| Thiamine | 1.630 | 0.018 |
| S-Methyl-5’-Thioadenosine | 1.666 | 0.048 |
| N-Acetyl-Glucosamine-1/6-Phosphate | 1.423 | 0.019 |
| dUMP | 2.840 | 0.037 |
| Aminoimidazole Carboxamide Ribonucleotide | 1.471 | 0.001 |
| CDP-Choline | 1.426 | 0.078 |
| UDP-N-Acetyl-glucosamine | 1.376 | 0.034 |
| NAD+ | 1.435 | 0.008 |
| Cyclic bis-3,5-dimeric GMP | 1.802 | 0.065 |
| FAD | 1.367 | 0.042 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Supplementary Materials
Conflicts of Interest
References
- Dow, C.; Swoboda, U. Cyanotoxins. In The Ecology of Cyanobacteria; Whitton, B., Potts, M., Eds.; Kluwer: Dordrecht, The Netherlands, 2000; pp. 613–632. [Google Scholar]
- Codd, G.A.; Lindsay, J.; Young, F.M.; Morrison, L.F.; Metcalf, J.S. Cyanobacterial toxins. In Harmful Cyanobacteria; Huisman, J., Matthijs, H.C.P., Visser, P.M., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 1–23. [Google Scholar]
- Antonius, A. Coral mortality in reefs: A problem for science and management. In Proceedings of the 3rd International Coral Reef Symposium, Miami, FL, USA, May 1977; Volume 2, pp. 618–623.
- Sutherland, K.; Porter, J.; Torres, C. Disease and immunity in Caribbean and Indo-Pacific zooxanthellate corals. Mar. Ecol. Prog. Ser. 2004, 266, 273–302. [Google Scholar] [CrossRef]
- Weil, E. Coral reef diseases in the wider Caribbean. In Coral Health and Disease; Loya, Y., Rosenberg, E., Eds.; Springer: Berlin, Germany, 2004; pp. 35–68. [Google Scholar]
- Voss, J.; Richardson, L.L. Coral diseases near Lee Stocking Island, Bahamas: Patterns and potential drivers. Dis. Aquat. Org. 2006, 69, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Antonius, A. Black band disease infection experiments on hexacorals and octocorals. In Proceedings of the 5th International Coral Reef Symposium, Tahiti, French Polynesia, 27 May–1 June 1985; Volume 6, pp. 155–160.
- Aeby, G.S.; Santavy, D.L. Factors affecting susceptibility of the coral Montastrea faveolata to black-band disease. Mar. Ecol. Prog. Ser. 2006, 318, 103–110. [Google Scholar] [CrossRef]
- Viehman, T.S.; Richardson, L.L. Motility patterns of Beggiatoa and Phormidium corallyticum in black band disease. In Proceedings of the 9th International Coral Reef Symposium, Bali, Indonesia, 23–27 October 2000; Volume 2, pp. 1251–1256.
- Rützler, K.; Santavy, D. The black band disease of Atlantic reef corals. I. Description of the cyanophyte pathogen. Mar. Ecol. 1983, 4, 301–319. [Google Scholar] [CrossRef]
- Antonius, A. The “band” diseases in coral reefs. In Proceedings of the Fourth International Coral Reef Symposium, Manila, Philippines, 18–22 May 1981; Volume 2, pp. 7–14.
- Cooney, R.; Pantos, O.; le Tissier, M.; Barer, M.; O’Donnell, A.; Bythell, J. Characterization of the bacterial consortium associated with black band disease in coral using molecular microbiological techniques. Environ. Microbiol. 2002, 4, 401–413. [Google Scholar] [CrossRef] [PubMed]
- Frias-Lopez, J.; Bonheyo, G.; Qusheng, J.; Fouke, B. Cyanobacteria associated with coral black band disease in caribbean and indo-pacific reefs. Appl. Environ. Microbiol. 2003, 69, 2409–2413. [Google Scholar] [CrossRef] [PubMed]
- Sussman, M.; Bourne, D.; Willis, B. A single cyanobacterial ribotype is associated with both red and black bands on diseased corals from Palau. Dis. Aquat. Org. 2006, 69, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Sekar, R.; Mills, D.; Remily, E.; Voss, J.; Richardson, L.L. Microbial communities in the surface mucopolysaccharide layer and the black band microbial mat of black band diseased Siderastrea siderea. Appl. Environ. Microbiol. 2006, 72, 5963–5973. [Google Scholar] [CrossRef] [PubMed]
- Sekar, R.; Kaczmarsky, L.T.; Richardson, L.L. Microbial community composition of black band disease on the coral host Siderastrea siderea from three regions of the wider Caribbean. Mar. Ecol. Prog. Ser. 2008, 362, 85–98. [Google Scholar] [CrossRef]
- Rasoulouniriana, D.; Siboni, N.; Ben-Dov, E.; Kramarsky-Winter, E.; Loya, Y.; Kushmaro, A. Pseudoscillatoria coralii gen. nov., sp. nov., a cyanobacterium associated with coral black band disease (BBD). Dis. Aquat. Org. 2009, 87, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Casamatta, D.; Staniç, D.; Gantar, M.; Richardson, L.L. Characterization of Roseofilum reptotaenium (Cyanobacteria, Oscillatoriales) gen. et sp. nov. isolated from Caribbean black band disease. Phycologia 2012, 51, 489–499. [Google Scholar] [CrossRef]
- Myers, J.L.; Sekar, R.; Richardson, L. Molecular detection and ecological significance of the cyanobacteria Geitlerinema and Leptolyngbya in black band disease of corals. Appl. Environ. Microbiol. 2007, 73, 5173–5182. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Willis, B.L.; Bourne, D.G. Successional changes in bacterial communities during the development of black band disease on the reef coral, Montipora hispida. ISME J. 2010, 4, 203–214. [Google Scholar] [CrossRef] [PubMed]
- Richardson, L.L.; Sekar, R.; Myers, J.L.; Gantar, M.; Remily, E.R.; Kaczmarsky, L.T.; Voss, J.D.; Boyer, G.L.; Zimba, P.V. The presence of the cyanobacterial toxin microcystin in black band disease of corals. FEMS Microbiol. Ecol. 2007, 272, 182–187. [Google Scholar] [CrossRef]
- Stanić, D.; Oehrle, S.; Gantar, M.; Richardson, L.L. Microcystin production and ecological physiology of Caribbean black band disease cyanobacteria. Environ. Microbiol. 2011, 14, 900–910. [Google Scholar] [CrossRef]
- Glas, M.S.; Motti, C.A.; Negri, A.P.; Sato, Y.; Froscio, S.; Humpage, A.R.; Krock, B.; Cembella, A.; Bourne, D.G. Cyanotoxins are not implicated in the etiology of coral black band disease outbreaks on Pelorus Island, Great Barrier Reef. FEMS Microbiol. Ecol. 2010, 73, 43–54. [Google Scholar] [PubMed]
- Kramarsky-Winter, E.; Arotsker, L.; Rasoulouniriana, D.; Siboni, N.; Loya, Y.; Kushmaro, A. The possible role of cyanobacterial filaments in coral black band disease pathology. Microb. Ecol. 2014, 67, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Moreno, C.; Romero, J.; Espejo, R.T. Polymorphism in repeated 16S rRNA genes is a common property of type strains and environmental isolates of the genus vibrio. Microbiology 2002, 148, 1233–1239. [Google Scholar] [PubMed]
- Nilsson, W.B.; Paranjype, R.N.; DePaola, A.; Strom, M.S. Sequence polymorphism of the 16S rRNA gene of Vibrio vulnificus is a possible indicator of strain virulence. J. Clin. Microbiol. 2003, 41, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Carlton, R.G.; Richardson, L.L. Oxygen and sulfide dynamics in a horizontally migrating cyanobacterial mat: Black band disease of corals. FEMS Microbiol. Ecol. 1995, 18, 155–162. [Google Scholar] [CrossRef]
- Viehman, T.S.; Mills, D.K.; Meichel, G.W.; Richardson, L.L. Culture and identification of Desulfovibrio spp. from black band disease of corals on reefs of the Florida Keys and Dominica. Dis. Aquat. Org. 2006, 69, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Frias-Lopez, J.; Klaus, J.S.; Bonheyo, G.T.; Fouke, B.W. Bacterial community associated with black band disease in corals. Appl. Environ. Microbiol. 2004, 70, 5955–5962. [Google Scholar] [CrossRef] [PubMed]
- Richardson, L.L.; Kuta, K.G. Ecological physiology of the black band disease cyanobacterium Phormidium corallyticum. FEMS Microbiol. Ecol. 2003, 43, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Richardson, L.; Kuta, K.; Schnell, S.; Carlton, R. Ecology of the black band disease microbial consortium. In Proceedings of the 8th International Coral Reef Symposium, Panama city, Panama, 24–29 June 1996; Volume 1, pp. 597–600.
- Miller, A.W.; Richardson, L.L. Fine structure analysis of black band disease (BBD) infected coral and coral exposed to the BBD toxins microcystin and sulfide. J. Invertebr. Pathol. 2011, 109, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Richardson, L.L.; Miller, A.W.; Broderick, E.; Kaczmarsky, L.; Gantar, M.; Stanić, D.; Sekar, R. Sulfide, microcystin, and the etiology of black band disease. Dis. Aquat. Org. 2009, 87, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Frias-Lopez, J.; Zerkle, A.; Bonheyo, G.; Fouke, B. Partitioning of bacterial communities between seawater and healthy, black band diseased, and dead coral surfaces. Appl. Environ. Microbiol. 2002, 68, 2214–2228. [Google Scholar] [CrossRef] [PubMed]
- Gantar, M.; Kaczmarsky, L.; Stanić, D.; Miller, A.W.; Richardson, L.L. Antibacterial activity of marine and black band disease cyanobacteria against coral-associated bacteria. Mar. Drugs 2011, 9, 2089–2105. [Google Scholar] [CrossRef] [PubMed]
- Rützler, K.; Santavy, D.; Antonius, A. The black band disease of Atlantic reef corals. III. Distribution, ecology and development. Mar. Ecol. 1983, 4, 329–358. [Google Scholar] [CrossRef]
- Brownell, A.; Richardson, L.L. Sulfate reducing bacteria as secondary and necessary pathogens in black band black band disease of coral. Rev. Biol. Trop. 2014, 62, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Richardson, L.L.; Castenholz, R.W. Diel vertical movements of the cyanobacterium Oscillatoria terebriformis in a sulfide-rich hot spring microbial mat. Appl. Environ. Microbiol. 1987, 53, 2142–2150. [Google Scholar] [PubMed]
- Dere, S.; Gunes, T.; Sivaci, R. Spectrophotometric determination of chlorophyll-a, b and total carotenoid contents of some algae species using different solvents. Turk. J. Bot. 1998, 22, 13–17. [Google Scholar]
- Rabinowitz, J.D.; Kimball, E. Acidic acetonitrile for cellular metabolome extraction from Escherichia coli. Anal. Chem. 2007, 79, 6167–6173. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Clasquin, M.F.; Melamud, E.; Amador-Noguez, D.; Caudy, A.A.; Rabinowitz, J.D. Metabolomic analysis via reversed-phase ion-pairing liquid chromatography coupled to a stand alone orbitrap mass spectrometer. Anal. Chem. 2010, 82, 3212–3221. [Google Scholar] [CrossRef] [PubMed]
- Martens, L.; Chambers, M.; Sturm, M.; Kessner, D.; Levander, F.; Shofstahl, J.; Tang, W.H.; Rompp, A.; Neumann, S.; Pizarro, A.D.; et al. mzML—A community standard for mass spectrometry data. Mol. Cell. Proteomics 2011, 10. [Google Scholar] [CrossRef] [PubMed]
- Chambers, M.C.; Maclean, B.; Burke, R.; Amodei, D.; Ruderman, D.L.; Neumann, S.; Gatto, L.; Fischer, B.; Pratt, B.; Egertson, J.; et al. A cross-platform toolkit for mass spectrometry and proteomics. Nat. Biotechnol. 2012, 30, 918–920. [Google Scholar] [CrossRef] [PubMed]
- Clasquin, M.F.; Melamud, E.; Rabinowitz, J.D. LC-MS data processing with MAVEN: A metabolomic analysis and visualization engine. Curr. Protoc. Bioinform. 2012, 37. [Google Scholar] [CrossRef]
- Melamud, E.; Vastag, L.; Rabinowitz, J.D. Metabolomic analysis and visualization engine for LC-MS data. Anal. Chem. 2010, 82, 9818–9826. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.G.; Psychogios, N.; Young, N.; Wishart, D.S. MetaboAnalyst: A web server for metabolomic data analysis and interpretation. Nucleic Acids Res. 2009, 37, W652–W660. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.G.; Mandal, R.; Sinelnikov, I.V.; Broadhurst, D.; Wishart, D.S. MetaboAnalyst 2.0-a comprehensive server for metabolomic data analysis. Nucleic Acids Res. 2012, 40, W127–W133. [Google Scholar] [CrossRef] [PubMed]
- Meikle, P.; Richards, G.; Yellowlees, D. Structural investigations on the mucus from 6 species of coral. Mar. Biol. 1988, 99, 187–193. [Google Scholar] [CrossRef]
- Tamulonis, C.; Kaandorp, J. A model of filamentous cyanobacteria leading to reticulate pattern formation. Life 2014, 4, 433–456. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.W.; Blackwelder, P.; Al-Sayegh, H.; Richardson, L.L. Insights into migration and development of coral black band disease based on fine structure analysis. Rev. Biol. Trop. 2012, 60, 21–27. [Google Scholar]
- Miller, A.W.; Blackwelder, P.; Al-Sayegh, H.; Richardson, L.L. Fine-structural analysis of black band disease-infected coral reveals boring cyanobacteria and novel bacteria. Dis. Aquat. Org. 2011, 93, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Bourne, D.G.; Muirhead, A.; Sato, Y. Changes in sulfate-reducing bacterial populations during the onset of black band disease. ISME J. 2010, 5, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Bourne, D.G.; Willis, B.L. Dynamics of seasonal outbreaks of black band disease in an assemblage of Montipora species at Pelorus Island (Great Barrier Reef, Australia). Proc. R. Soc. B 2009, 276, 2795–2803. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.W.; Richardson, L.L. A meta-analysis of 16S rRNA gene clone libraries from the polymicrobial black band disease of corals. FEMS Microbiol. Ecol. 2011, 75, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, B.L.; May, A.L.; Bhedi, C.D.; Dearth, S.P.; Prevatte, C.W.; Pratte, Z.; Campagna, S.R.; Richardson, L.L. Quorum sensing signal production and microbial interactions in a polymicrobial disease of corals and the coral surface mucopolysaccharide layer. PLoS One 2014. [Google Scholar] [CrossRef]
- He, J.; Chen, J.; Wu, L.; Li, G.; Xie, P. Metabolic response to oral microcystin-LR exposure in the rat by NMR-based metabonomic study. J. Proteome Res. 2012, 11, 5934–5946. [Google Scholar] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richardson, L.L.; Stanić, D.; May, A.; Brownell, A.; Gantar, M.; Campagna, S.R. Ecology and Physiology of the Pathogenic Cyanobacterium Roseofilum reptotaenium. Life 2014, 4, 968-987. https://doi.org/10.3390/life4040968
Richardson LL, Stanić D, May A, Brownell A, Gantar M, Campagna SR. Ecology and Physiology of the Pathogenic Cyanobacterium Roseofilum reptotaenium. Life. 2014; 4(4):968-987. https://doi.org/10.3390/life4040968
Chicago/Turabian StyleRichardson, Laurie L., Dina Stanić, Amanda May, Abigael Brownell, Miroslav Gantar, and Shawn R. Campagna. 2014. "Ecology and Physiology of the Pathogenic Cyanobacterium Roseofilum reptotaenium" Life 4, no. 4: 968-987. https://doi.org/10.3390/life4040968




