Selective Liberation of High-Phosphorous Oolitic Hematite Assisted by Microwave Processing and Acid Leaching
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Basic Test Flowchart
2.2.2. Iron ore Pretreatment
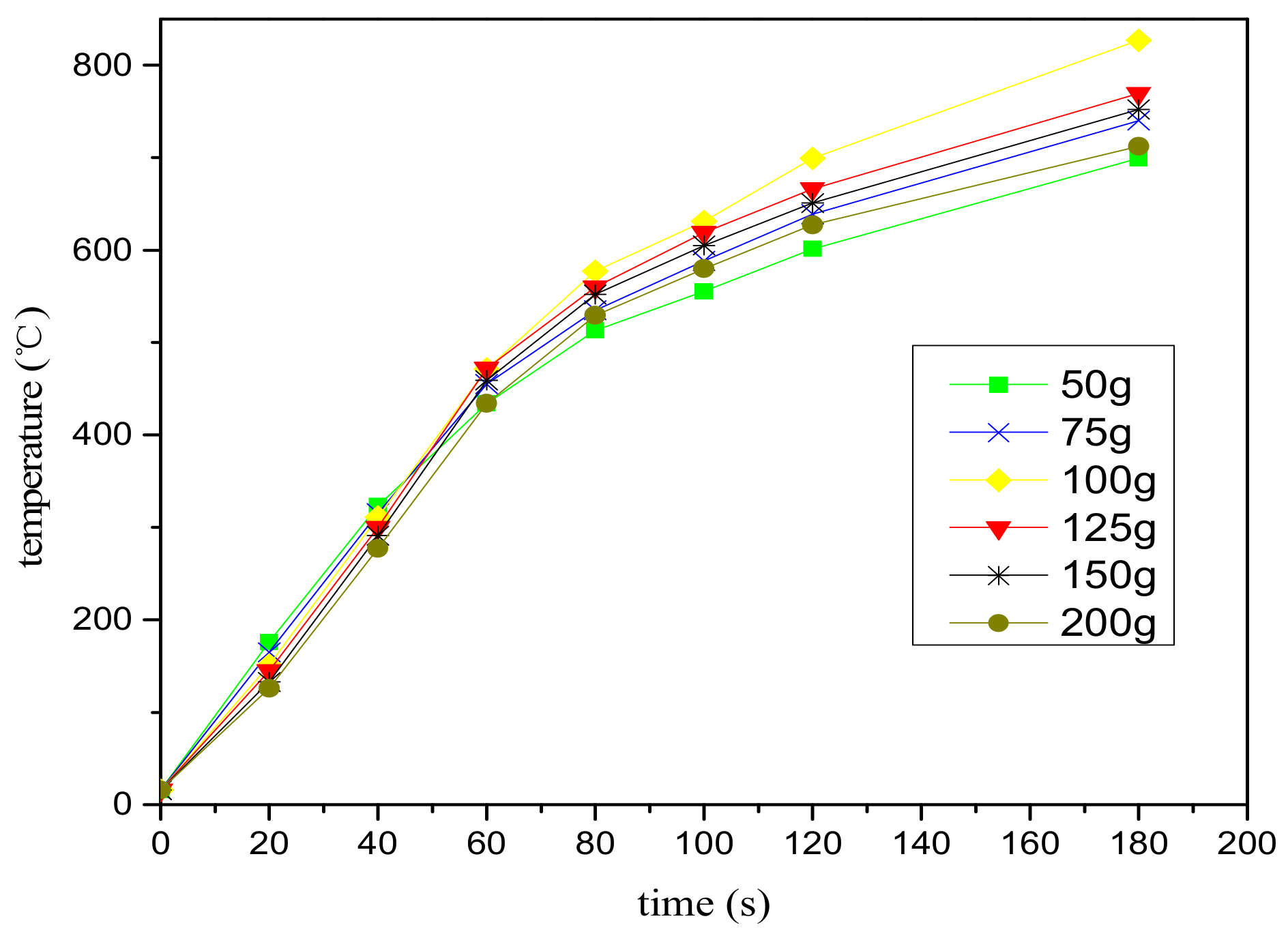
2.2.3. Microwave Processing and Grinding Experiment
2.2.4. Acid Leaching Experiment
2.2.5. Magnetic Separation Experiment
2.2.6. Analytical Techniques
- Wi, ball mill work index, kW·h/t;
- Pi, screen size, μm;
- Gbg, net weight of the ball mill product passing sieve size Pi produced per mill revolution, g/r;
- P80, sieve opening through which 80% of the product passes, μm;
- F80, sieve opening through which 80% of the product passes, μm;
- µ, the dephosphorization ratio, %;
- p0, the content of phosphorus before grinding, %;
- p1, the content of phosphorus after grinding, %.
3. Results and Discussion
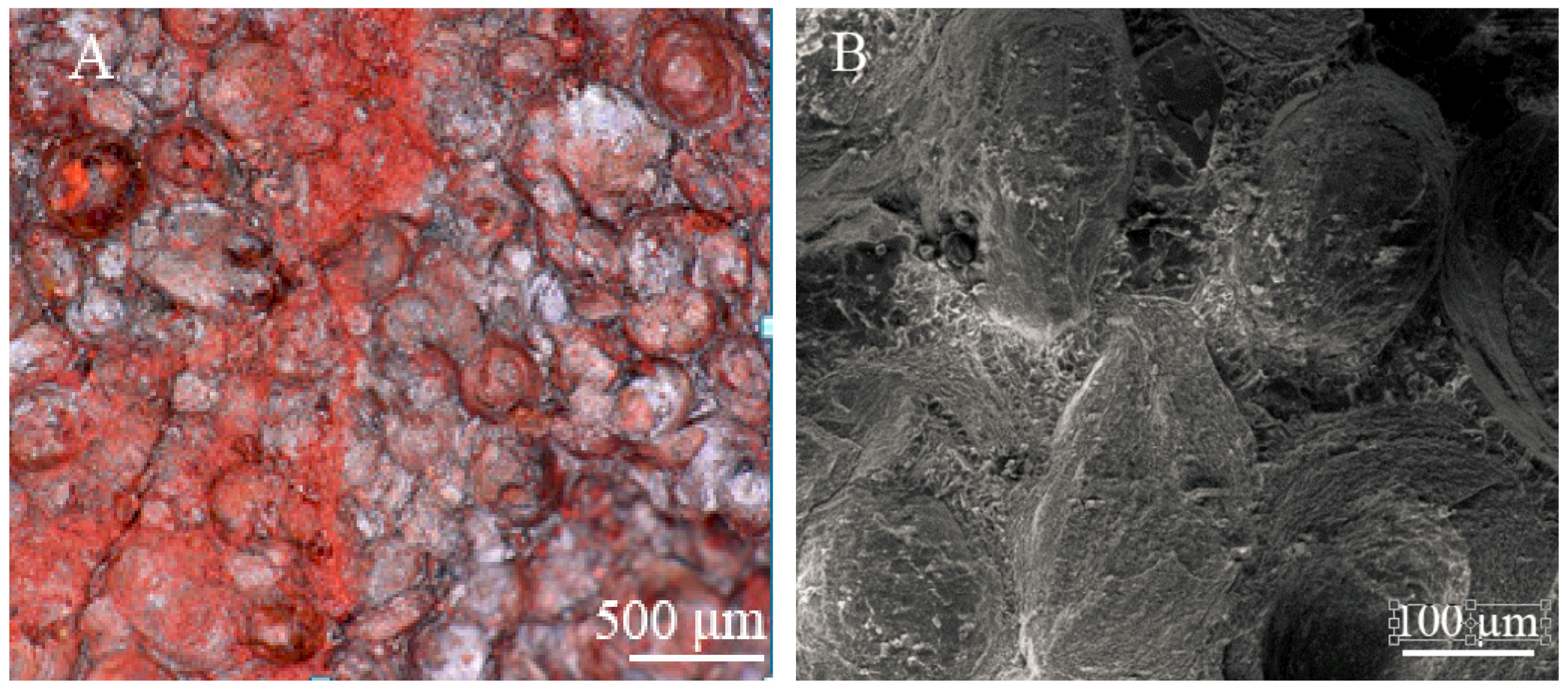
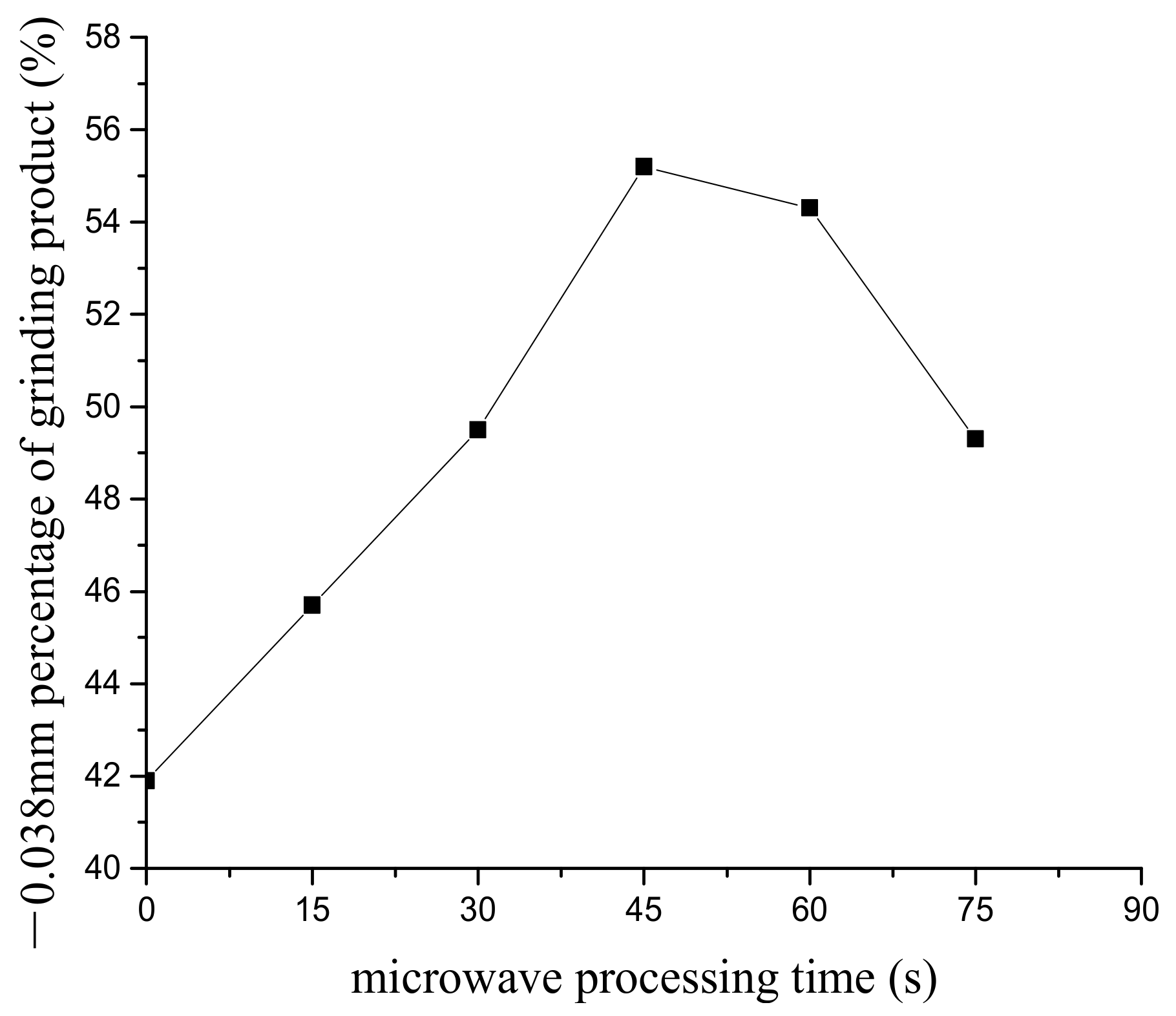
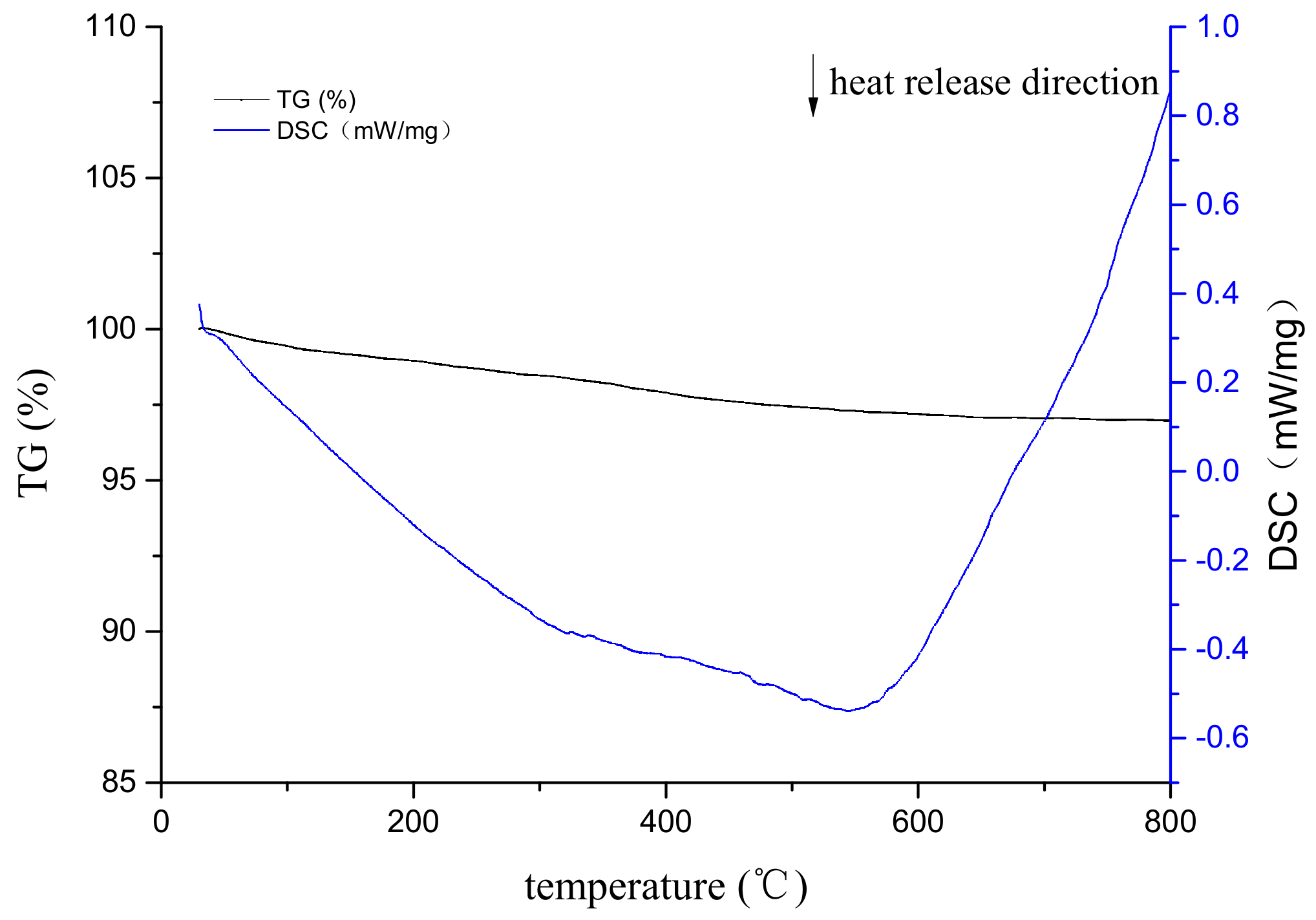
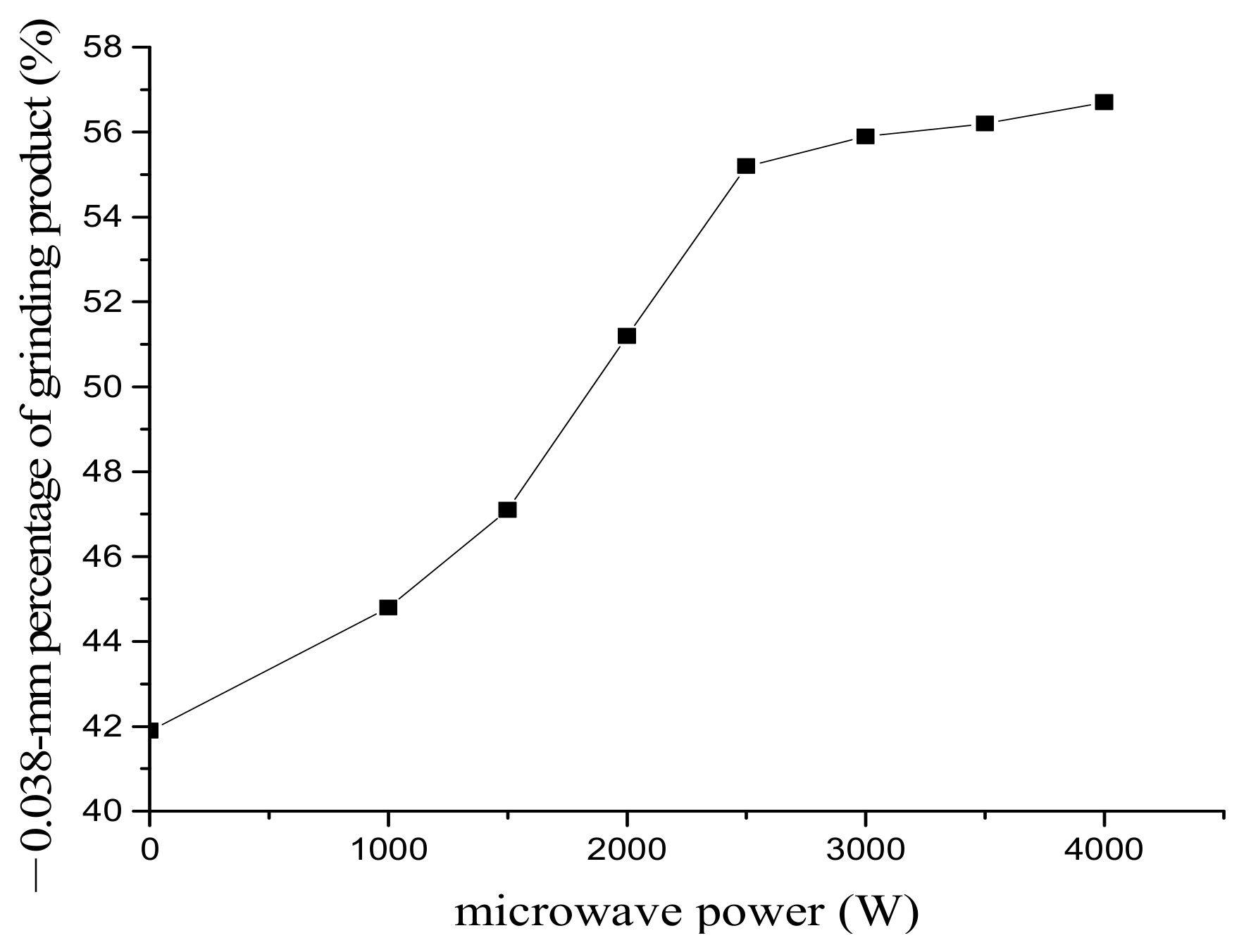
3.1. The Effect of Microwave Processing on the Ore and Grinding
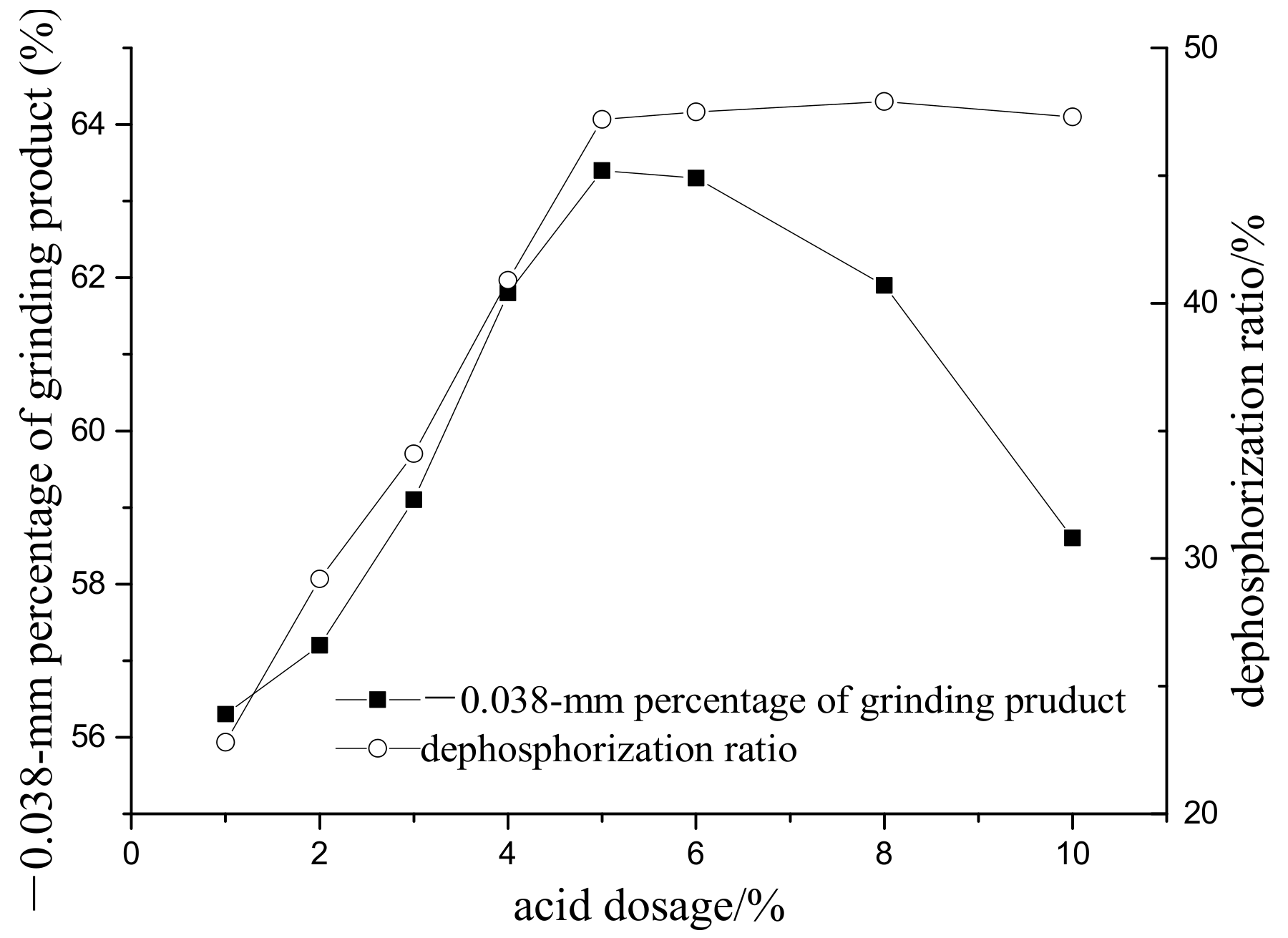
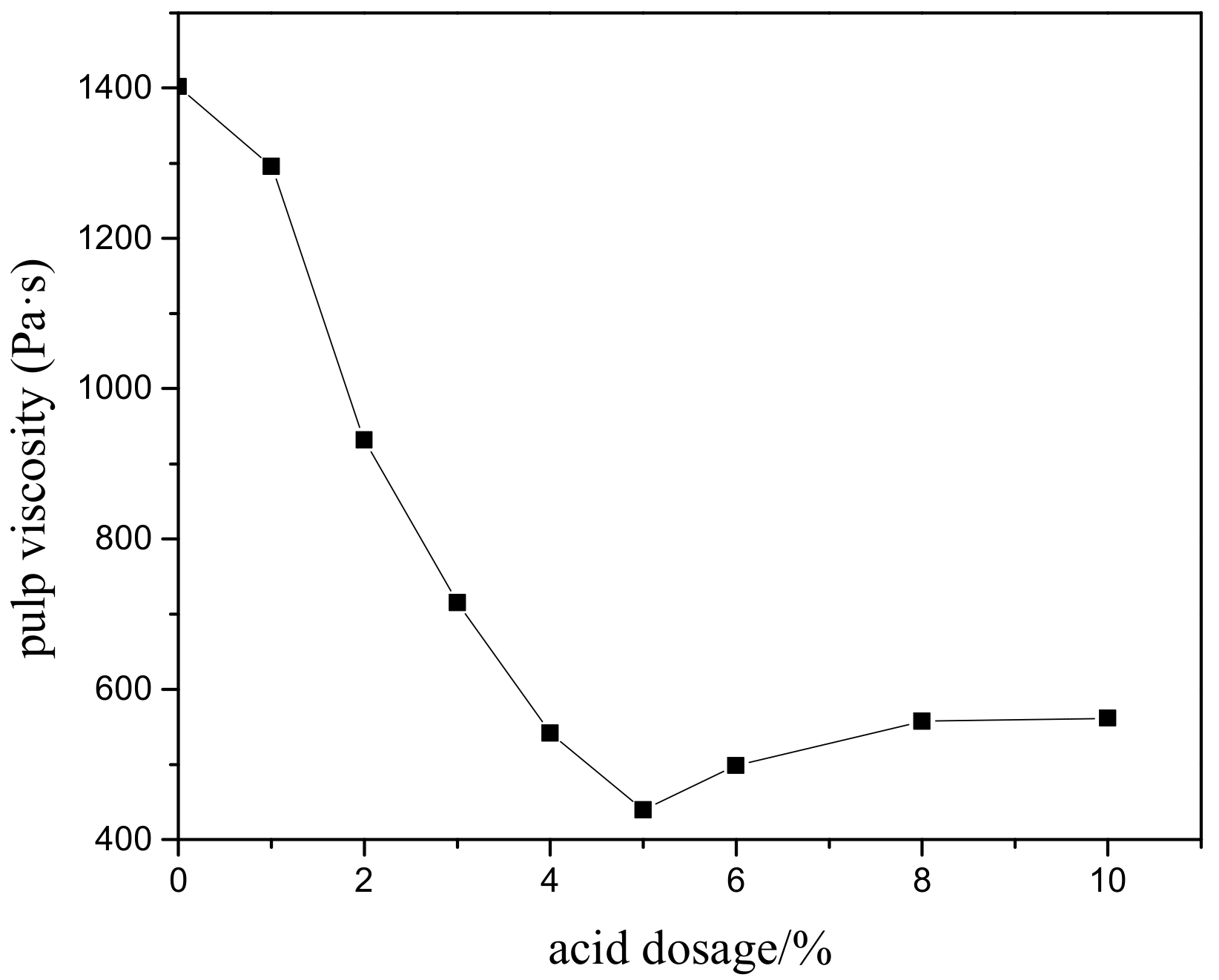
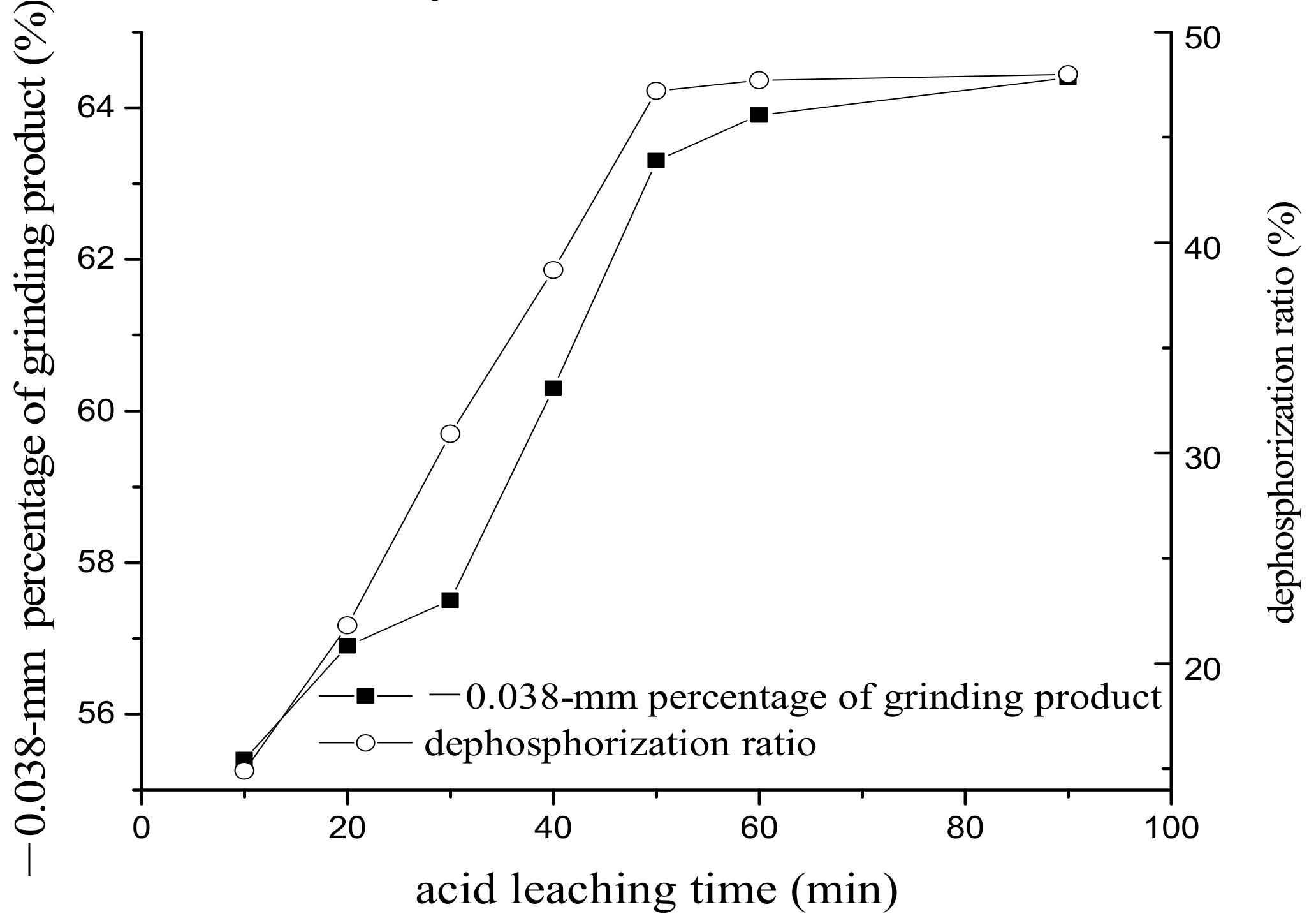
3.2. The Effect of Acid Leaching on Grinding
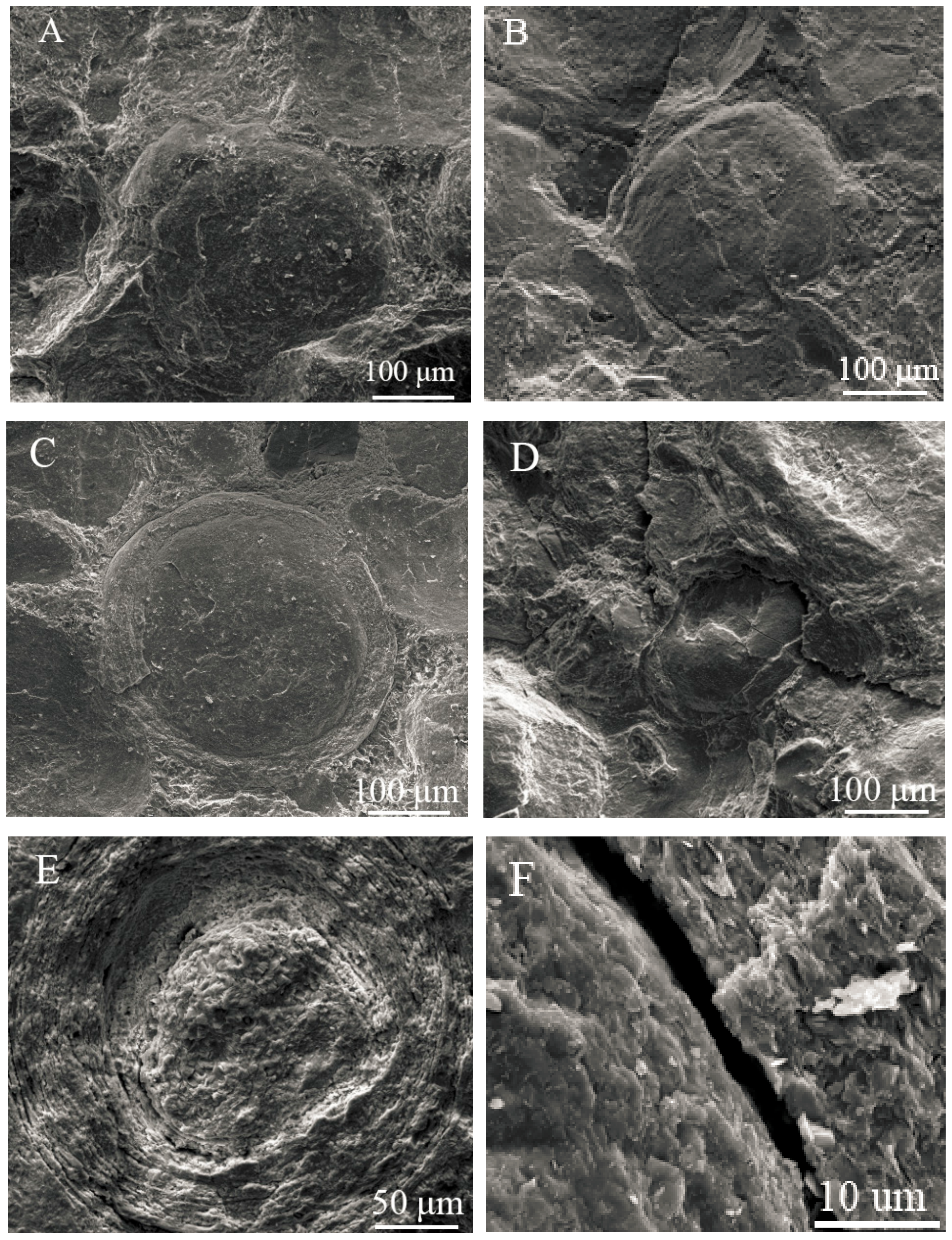
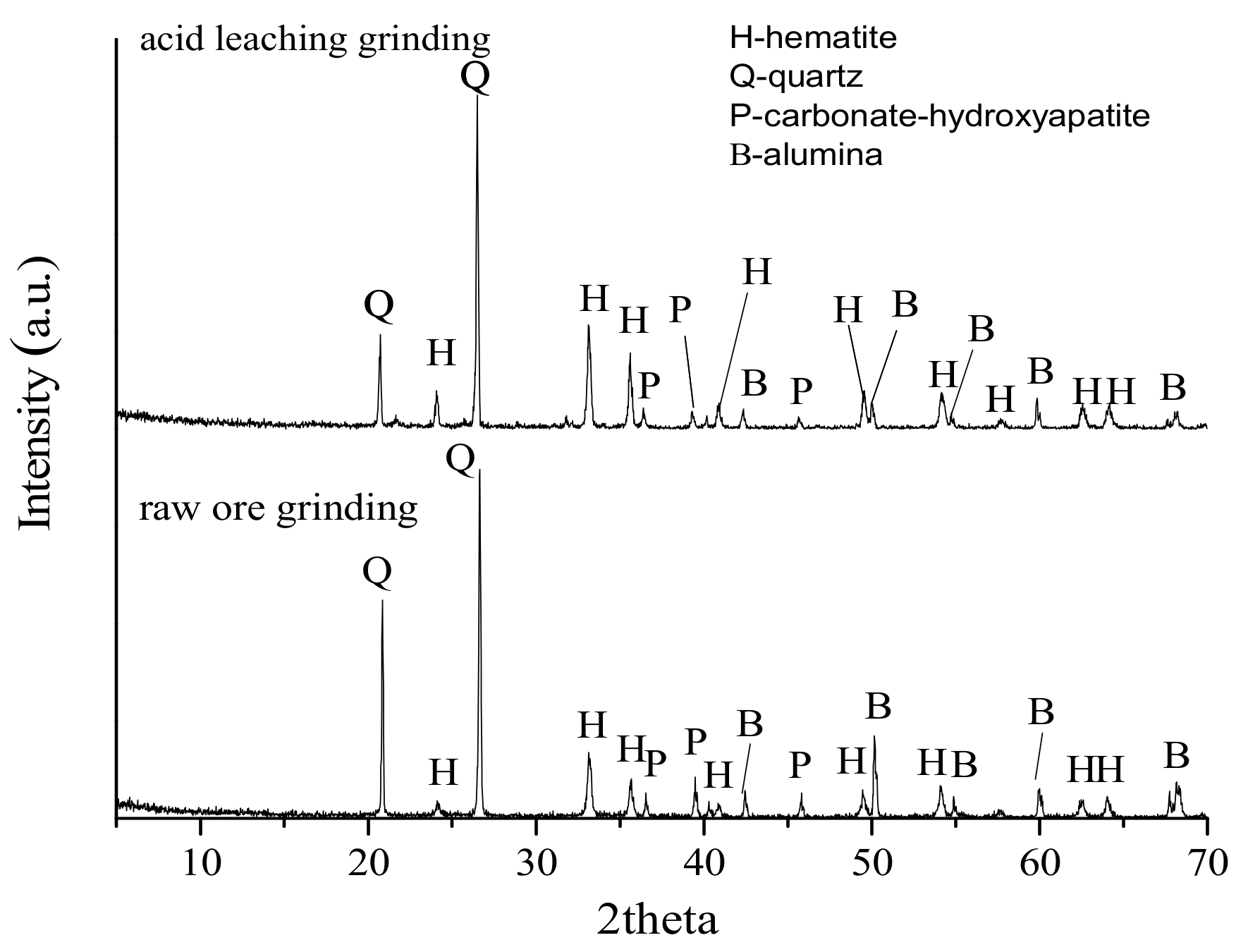
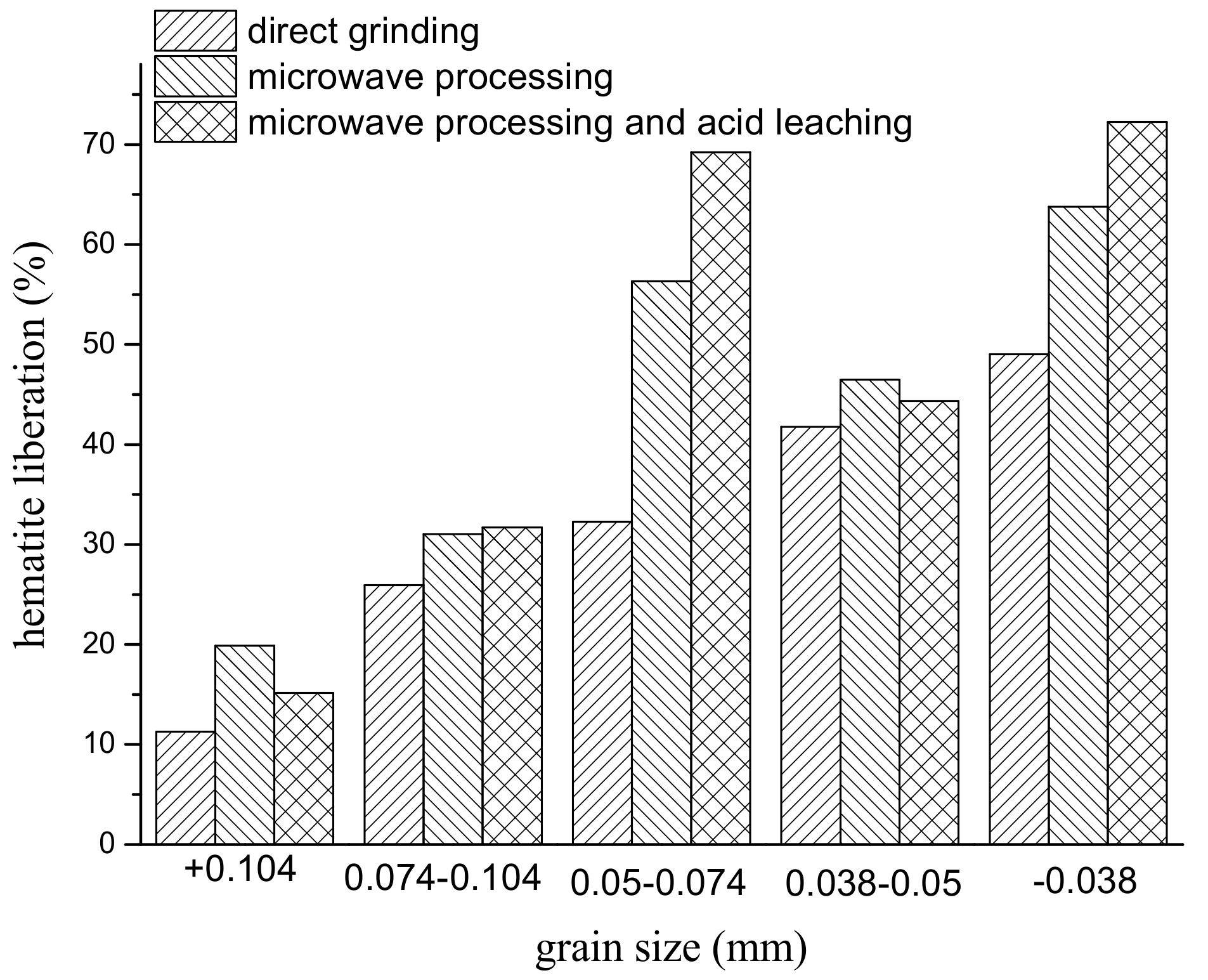
3.3. Liberation Analysis
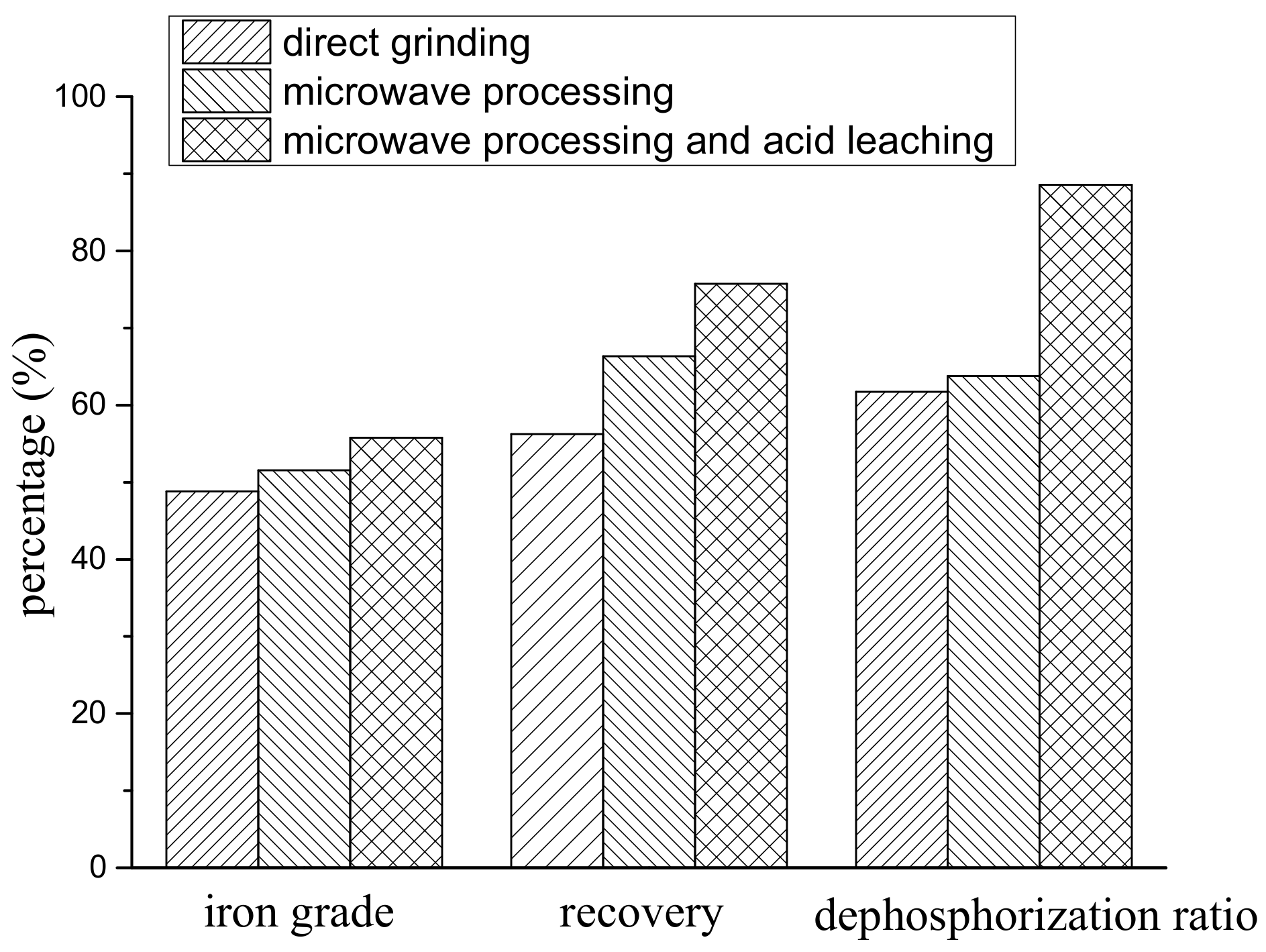
3.4. Magnetic Separation Results Analysis
3.5. Error Analysis
4. Conclusions
- (1)
- Microwave processing is able to make this ore be selectively liberated in the grinding process, and acid leaching further improves the selective liberation and reduces the content of phosphorus in the grinding product.
- (2)
- Excessive microwave processing treatment has a negative effect on the liberation of ores. Excessive acid dosage is not beneficial to the improvement of selective liberation and the dephosphorization ratio.
- (3)
- Microwave processing and acid leaching comprise an efficient and feasible method to liberate high-phosphorus oolitic hematite.
- (4)
- Microwave processing and acid leaching can improve magnetic separation products’ indexes significantly.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sun, B. Progress in China’s Beneficiation Technology for Complex Refractory Iron Ore. Met. Mine 2006, 3, 11–13. [Google Scholar]
- Wu, J.; Wen, Z.; Cen, M. Development of Technologies for High Phosphorus Oolitic Hematite Utilization. Steel Res. Int. 2011, 82, 494–500. [Google Scholar] [CrossRef]
- Zhang, J.; Hu, L.; Liang, Y.; Yang, H.; Sun, D. The research on refractory oolitic hematite and expectation. China Min. Mag. 2007, 16, 74–76. [Google Scholar]
- Xu, C.; Sun, T.; Kou, J.; Li, Y.; Mo, X.; Tang, L. Mechanism of phosphorus removal in beneficiation of high phosphorous oolitic hematite by direct reduction roasting with dephosphorization agent. Trans. Nonferr. Met. Soc. China 2012, 22, 2806–2812. [Google Scholar] [CrossRef]
- Dai, H.; Zhao, W.; Gao, L.; Song, B. Process Mineralogy of an Oolitic Hematite Ore and its Implications for Mineral Processing. Adv. Mater. Res. 2012, 567, 131–134. [Google Scholar] [CrossRef]
- Ueda, T.; Oki, T.; Koyanaka, S. A general quantification method for addressing stereological bias in mineral liberation assessment in terms of volume fraction and size of mineral phase. Miner. Eng. 2018, 119, 156–165. [Google Scholar] [CrossRef]
- Mariano, R.A.; Evans, E.L.; Manlapig, E. Definition of random and non-random breakage in mineral liberation—A review. Miner. Eng. 2016, 94, 51–60. [Google Scholar] [CrossRef]
- Veasey, T.J.; Wills, B.A. Review of methods of improving mineral liberation. Miner. Eng. 1991, 4, 747–752. [Google Scholar] [CrossRef]
- Little, L.; Mainza, A.N.; Becker, M.; Wiese, J.G. Using mineralogical and particle shape analysis to investigate enhanced mineral liberation through phase boundary fracture. Powder Technol. 2016, 301, 794–804. [Google Scholar] [CrossRef]
- Fandrich, R.G.; Bearman, R.A.; Boland, J.; Lim, W. Mineral liberation by particle bed breakage. Miner. Eng. 1997, 10, 175–187. [Google Scholar] [CrossRef]
- Wang, E.; Shi, F.; Manlapig, E. Mineral liberation by high voltage pulses and conventional comminution with same specific energy levels. Miner. Eng. 2012, 27–28, 28–36. [Google Scholar] [CrossRef]
- Jones, D.A.; Lelyveld, T.D.; Mavrofidis, S.D.; Kingman, S.W.; Miles, N.J. Microwave heating applications in environmental engineering—A review. Resour. Conserv. Recycl. 2002, 34, 75–90. [Google Scholar] [CrossRef]
- Chen, G.; Chen, J.; Guo, S.; Li, J.; Srinivasakannan, C.; Peng, J. Dissociation behavior and structural of ilmenite ore by microwave irradiation. Appl. Surf. Sci. 1990, 258, 4826–4829. [Google Scholar] [CrossRef]
- Sahoo, B.K.; De, S.; Carsky, M.; Meikap, B.C. Rheological characteristics of coal–water slurry using microwave pretreatment—A statistical approach. J. Ind. Eng. Chem. 2011, 17, 62–70. [Google Scholar] [CrossRef]
- Scott, G.; Bradshaw, S.M.; Eksteen, J.J. The effect of microwave pretreatment on the liberation of a copper carbonatite ore after milling. Int. J. Miner. Process. 2008, 85, 121–128. [Google Scholar] [CrossRef]
- Amankwah, R.K.; Khan, A.U.; Pickles, C.A.; Yen, W.T. Improved grindability and gold liberation by microwave pretreatment of a free-milling gold ore. Miner. Process. Extr. Metall. 2005, 114, 30–36. [Google Scholar] [CrossRef]
- Song, S.; Campos-Toro, E.F.; Valdivieso, A.L. Formation of microfractures on an oolitic iron ore under microwave treatment and its effect on selective fragmentation. Powder Technol. 2013, 243, 155–160. [Google Scholar] [CrossRef]
- Fitzgibbon, K.E.; Veasey, T.J. Thermally assisted liberation―A review. Miner. Eng. 1990, 3, 181–185. [Google Scholar] [CrossRef]
- Jones, D.A.; Kingman, S.W.; Whittles, D.N.; Lowndes, I.S. Understanding microwave assisted breakage. Miner. Eng. 2005, 18, 659–669. [Google Scholar] [CrossRef]
- Gu, S.; Chen, G.; Peng, J.; Chen, J.; Li, D.; Liu, L. Microwave assisted grinding of ilmenite ore. Trans. Nonferr. Met. Soc. China 2011, 21, 2122–2126. [Google Scholar] [CrossRef]
- Walkiewicz, J.W.; Clark, A.E.; McGill, S.L. Microwave-assisted grinding. IEEE Trans. Ind. Appl. 1991, 27, 239–243. [Google Scholar] [CrossRef]
- Omran, M.; Fabritius, T.; Mattila, R. Thermally assisted liberation of high phosphorus oolitic iron ore: A comparison between microwave and conventional furnaces. Powder Technol. 2015, 269, 7–14. [Google Scholar] [CrossRef]
- He, Z.; Zhang, H.; Jin, Y. Mechanism Study on the High-phosphorus Hematite Carbothermal Reduction in Microwave Field. Adv. Mater. Res. 2011, 291–294, 1358–1361. [Google Scholar] [CrossRef]
- Omran, M.; Fabritius, T.; Mattila, R.; Abdel-Khalek, N.; Gornostayev, S. Improvement of phosphorus removal from iron ore using combined microwave pretreatment and ultrasonic treatment. Sep. Purif. Technol. 2015, 156, 724–737. [Google Scholar] [CrossRef]
- Zhong, L.; Gong, W.; Li, Y.; Xin, Z.; Lu, Y.; Liu, S. Experimental study on acid leaching intensified by ultrasonic wave to remove phosphorus from high-phosphorus iron ore. J. Wuhan Univ. Technol. 2012, 34, 107–110. [Google Scholar]
- Deniz, V.; Ozdag, H. A new approach to Bond grindability and work index: Dynamic elastic parameters. Miner. Eng. 2003, 16, 211–217. [Google Scholar] [CrossRef]

| Formula | TFe | SiO2 | Al2O3 | CaO | MgO | MnO | K2O | P2O5 | S |
|---|---|---|---|---|---|---|---|---|---|
| Weight % | 43.31 | 20.44 | 6.89 | 3.91 | 1.28 | 0.11 | 0.39 | 3.41 | 0.03 |
| Element | Phase | wt % | Distribution Rate (%) |
|---|---|---|---|
| Fe | Magnetite | 0.31 | 0.72 |
| Hematite | 41.54 | 95.91 | |
| Iron carbonate | 0.13 | 0.30 | |
| Iron sulfide | 0.06 | 0.14 | |
| Ferric silicate | 1.27 | 2.39 | |
| P | Carbonate-hydroxyapatite | 0.31 | 87.74 |
| Hematite/goethite | 41.54 | 7.11 | |
| Ferric silicate | 0.13 | 2.30 | |
| Other mineral | 0.06 | 1.86 |
| Microwave Processing Condition | Specific Surface Area(m2/g) | Compressive Strength (N/cm2) | Porosity (%) | Ball Mill Work Index (kW·h/t) |
|---|---|---|---|---|
| raw ore | 8.74 | 1.93 | 4.14 | 15.25 |
| 2.5 kW, 45 s | 13.28 | 0.39 | 5.79 | 10.11 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, X.; Qian, G.; Zhang, B.; Chen, Q.; Hu, C. Selective Liberation of High-Phosphorous Oolitic Hematite Assisted by Microwave Processing and Acid Leaching. Minerals 2018, 8, 245. https://doi.org/10.3390/min8060245
Cai X, Qian G, Zhang B, Chen Q, Hu C. Selective Liberation of High-Phosphorous Oolitic Hematite Assisted by Microwave Processing and Acid Leaching. Minerals. 2018; 8(6):245. https://doi.org/10.3390/min8060245
Chicago/Turabian StyleCai, Xianyan, Gongming Qian, Bo Zhang, Qiushi Chen, and Chenqiang Hu. 2018. "Selective Liberation of High-Phosphorous Oolitic Hematite Assisted by Microwave Processing and Acid Leaching" Minerals 8, no. 6: 245. https://doi.org/10.3390/min8060245





