Environmental Risk of Metal Mining Contaminated River Bank Sediment at Redox-Transitional Zones
Abstract
:1. Introduction
2. Biogeochemical Processes
2.1. Changes in Mineral Form and Mobility of Trace Metal Contaminants on Submergence of Contaminated Sediment
| Redox Half Reaction | Eo/mV | pEo | pEo (w)c | References |
|---|---|---|---|---|
| O2(g) + 4H+ + 4e− — 2H2O | +122.9 | +20.8 | +13.8 | [50] |
| 2NO3− + 12H+ + 10e− — N2(g) + 6H2O | +125 | +21.1 | +12.7 | [50] |
| MnO2(s) + 4H+ + 2e− — Mn2+ + 2H2O | +8.9 * | [51] | ||
| FeOOH(s) + 3H+ + 2e− — Fe2+ + 2H2O | −2.7 * | [51] | ||
| SO42− + 9H+ + 8e− — HS− + 4H2O | −3.4 * | [51] | ||
| CO2 + 8H+ + 8e− — CH4 + 2H2O | +17 | +2.87 | −4.13 | [50] |
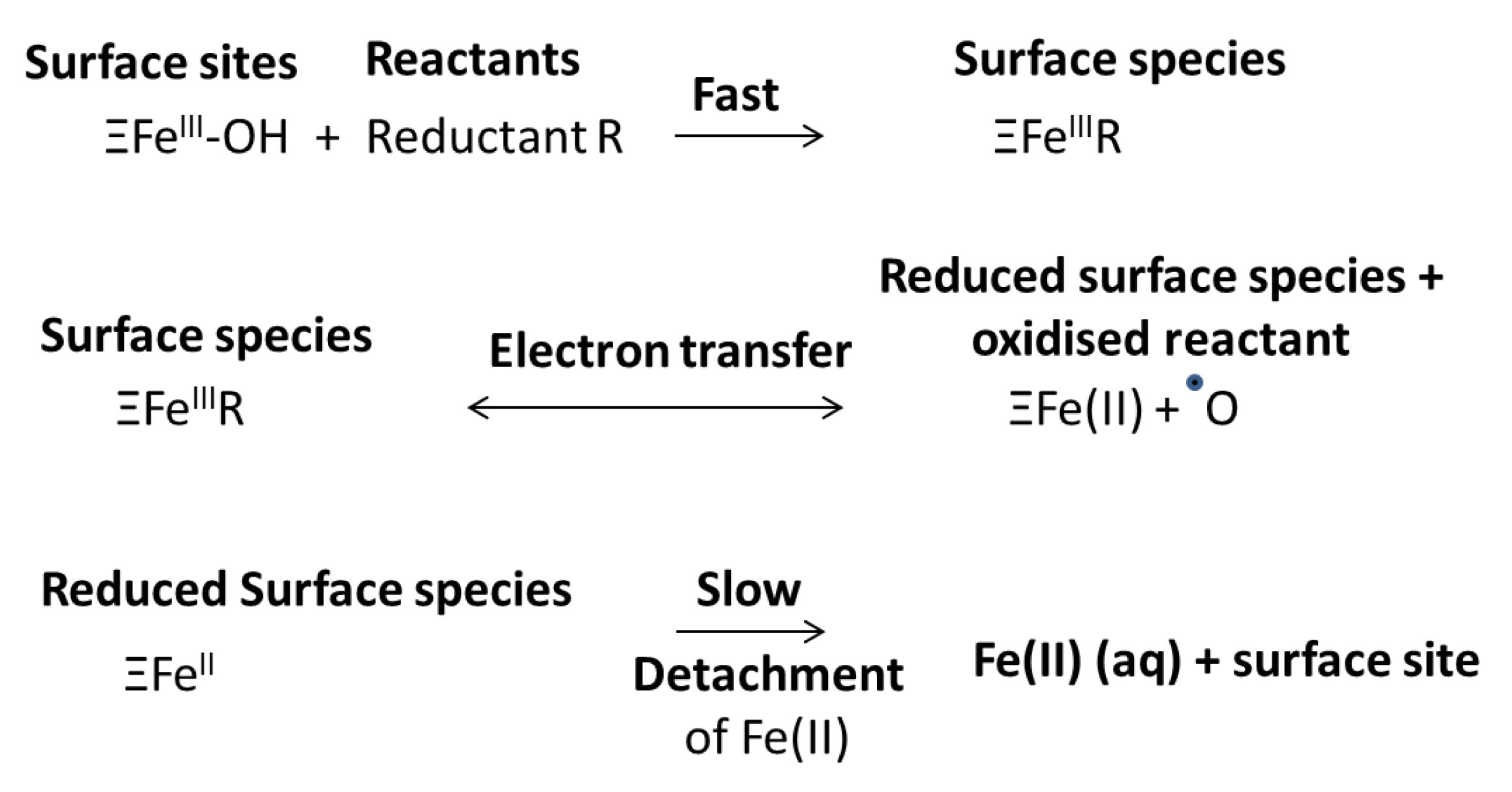
2.2. Changes in Mineral Form during Prolonged Flooding: Sulphate Reduction and Metal Sulphide Precipitation





2.3. Changes in Mineral Form and the Release of Trace Metal Contaminants during Oxidation of Reduced Sediments: Sulphide Oxidation and Iron and Manganese Mineral Precipitation
2.4. Sorption of Trace Metals onto Iron (Hydr)Oxides
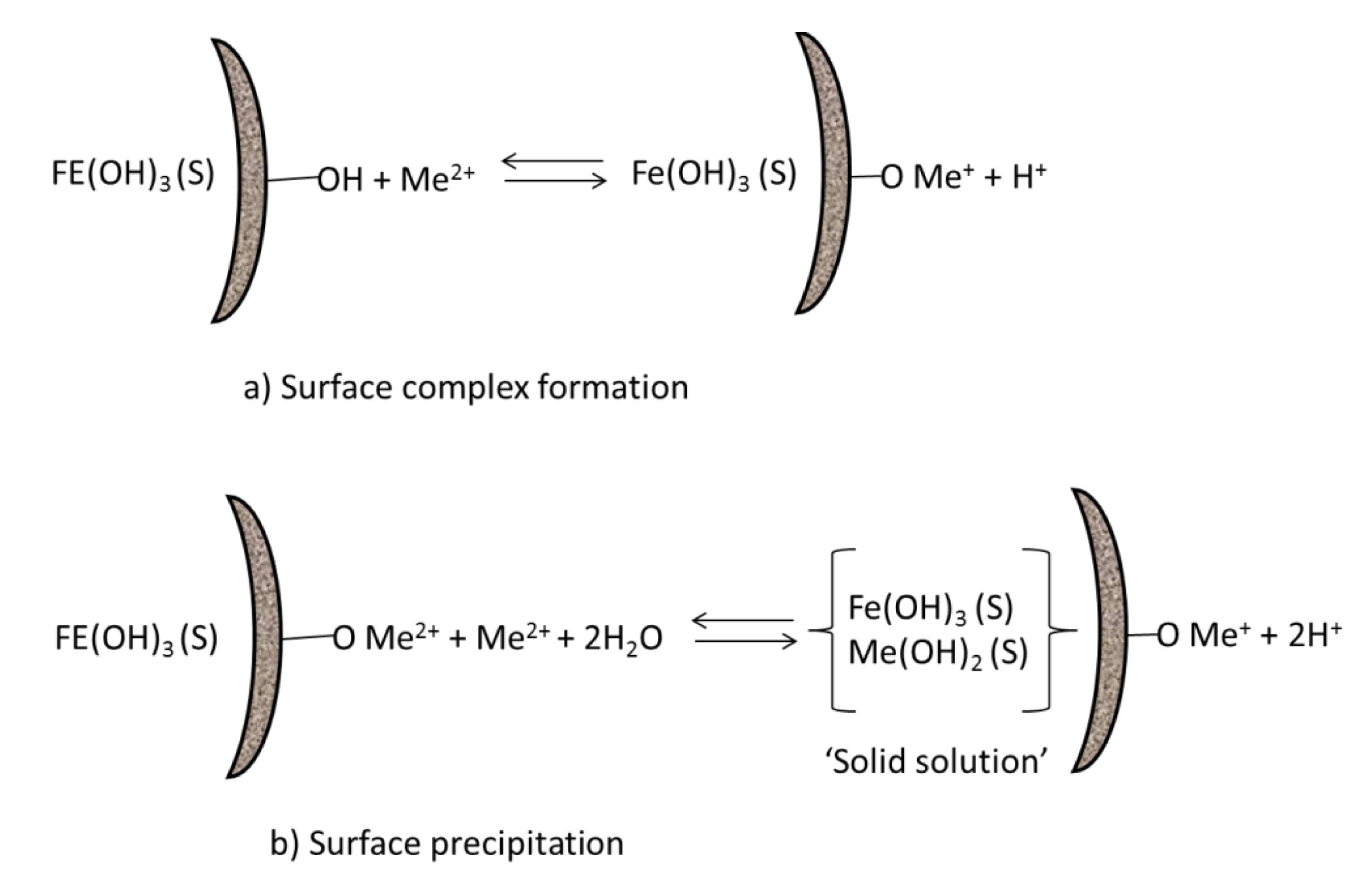
2.5. Effects of Long Dry Episodes on Mineral Form and Reactivity
2.6. Simplified Conceptual Model

3. Conclusions
- Mesocosm experiments are undertaken that simulate river bank environment in a controlled environment under different frequency and duration of wet and dry cycles.
- Characterization of the sediment under these conditions: sequential extractions that target reactive iron minerals (ferrihydrite), in addition to more crystalline forms (goethite and hematite) and scanning electron microscopy/energy dispersive X-ray spectroscopy (SEM/EDS) analysis to identify mineralogical changes.
- Techniques such as Diffusive Gradients in Thin Film (DGT) and Diffusive Equilibrium in thin films (DET) are applied to understand the interactions between solutes. Measurements made passively in the sediment at high spatial resolution could prevent mixing of larger pore water samples and avoid averaging of concentrations and chemical reactions during sampling that could confound results. These techniques could promote a greater understanding of the mechanisms that control trace metal mobility in mining contaminated sediment under dynamic redox potential conditions.
Acknowledgments
Conflicts of Interest
References
- Mayes, W.M.; Potter, H.A.B.; Jarvis, A.P. Riverine flux of metals from historically mined orefields in England and Wales. Water Air Soil Pollut. 2013, 224, 1425. [Google Scholar] [CrossRef]
- Gozzard, E.; Mayes, W.M.; Potter, H.A.B.; Jarvis, A.P. Seasonal and spatial variation of diffuse (non-point) source zinc pollution in a historically metal mined river catchment, UK. Environ. Pollut. 2011, 159, 3113–3122. [Google Scholar] [CrossRef]
- Caruso, B.S.; Bishop, M. Seasonal and spatial variation of metal loads from natural flows in the Upper Tenmile Creek watershed, Montana. Mine Water Environ. 2009, 28, 166–181. [Google Scholar] [CrossRef]
- Younger, P.L. Adit Hydrology in the Long-Term: Observations from the Pb-Zn Mines of Northern England. In Proceedings of the International Mine Water Association (IMWA) Symposium, Johannesburg, South Africa, 7–13 September 1998; pp. 347–356.
- Department for Environment, Food and Rural Affairs. Catchment Based Approach: Improving the Quality of Our Water Environment; Department for Environment, Food and Rural Affairs: London, UK, 2013.
- Gandy, C.J.; Smith, J.W.N.; Jarvis, A.P. Attenuation of mining-derived pollutants in the hyporheic zone: A review. Sci. Total Environ. 2007, 373, 435–446. [Google Scholar] [CrossRef]
- Mayes, W.M.; Gozzard, E.; Potter, H.A.B.; Jarvis, A.P. Quantifying the importance of diffuse minewater pollution in a historically heavily coal mined catchment. Environ. Pollut. 2008, 151, 165–175. [Google Scholar] [CrossRef]
- Jarvis, A.P. Prioritisation of Abandoned Non-Coal Mine Impacts on the Environment Project Summary sc030136/s14; Environment Agency: Bristol, UK, 2010. [Google Scholar]
- Hudson-Edwards, K.A.; Macklin, M.G.; Brewer, P.A.; Dennis, I.A. Assessment of Metal Mining Contaminated River Sediments in England and Wales; Environment Agency: Bristol, UK, 2008. [Google Scholar]
- Lu, X.; Wang, H. Microbial oxidation of sulfide tailings and the environmental consequences. Elements 2012, 8, 119–124. [Google Scholar] [CrossRef]
- Hofmann, T.; Schuwirth, N. Zn and Pb release of sphalerite (ZnS)-bearing mine waste tailings. J. Soils Sediments 2008, 8, 433–441. [Google Scholar] [CrossRef]
- Merrington, G.; Alloway, B.J. The transfer and fate of Cd, Cu, Pb, and Zn from two historic metalliferous mine sites in the U.K. Appl. Geochem. 1994, 9, 677–687. [Google Scholar] [CrossRef]
- Palumbo-Roe, B.; Wragg, J.; Banks, V.J. Lead mobilisation in the hyporheic zone and river bank sediments of a contaminated stream: Contribution to diffuse pollution. J. Soils Sediments 2012, 12, 1633–1640. [Google Scholar] [CrossRef] [Green Version]
- Byrne, P.; Reid, I.; Wood, P.J. Short-Term Fluctuations in Heavy Metal Concentrations during Flood Events through Abandoned Metal Mines, with Implications for Aquatic Ecology and Mine Water Treament. In Proceedings of the International Mine Water Conference (IMWC), Pretoria, South Africa, 19–23 October 2009.
- Dennis, I.A.; Coulthard, T.J.; Brewer, P.; Macklin, M.G. The role of floodplains in attenuating contaminated sediment fluxes in formerly mined drainage basins. Earth Surf. Process. Landf. 2009, 34, 453–466. [Google Scholar] [CrossRef]
- Hudson-Edwards, K.A.; Macklin, M.G.; Taylor, M.P. 2000 years of sediment-borne heavy metal storage in the Yorkshire Ouse Basin, NE England, UK. Hydrol. Process. 1999, 13, 1087–1102. [Google Scholar] [CrossRef]
- Coulthard, T.J.; Macklin, M.G. Modeling long-term contamination in river systems from historical metal mining. Geology 2003, 31, 451–454. [Google Scholar] [CrossRef]
- Caetano, M.; Madureira, M.J.; Vale, C. Metal remobilisation during resuspension of anoxic contaminated sediment: Short-term laboratory study. Water Air Soil Pollut. 2003, 143, 23–40. [Google Scholar] [CrossRef]
- Eggleton, J.; Thomas, K.V. A review of factors affecting the release and bioavailability of contaminants during sediment distrubance events. Environ. Int. 2004, 30, 973–980. [Google Scholar] [CrossRef]
- Linge, K.L. Methods for investigating trace element binding in sediments. Crit. Rev. Environ. Sci. Technol. 2008, 38, 165–196. [Google Scholar] [CrossRef]
- Krause, S.; Hannah, D.M.; Fleckenstein, J.H.; Heppell, C.M.; Kaeser, D.; Pickup, R.; Pinay, G.; Robertson, A.L.; Wood, P.J. Inter-disciplinary perspectives on processes in the hyporheic zone. Ecohydrology 2011, 4, 481–499. [Google Scholar] [CrossRef]
- Du Laing, G.; Rinklebe, J.; Vandecasteele, B.; Meers, E.; Tack, F.M.G. Trace metal behaviour in estuarine and riverine floodplain soils and sediments: A review. Sci. Total Environ. 2009, 407, 3972–3985. [Google Scholar] [CrossRef]
- Wragg, J.; Palumbo-Roe, B.; British Geological Survey. Contaminant Mobility as a Result of Sediment Inundation: Literature Review and Laboratory Scale Pilot Study on Mining Contaminated Sediments: British Geological Survey report OR/11/051; Natural Environment Research Council: Swindon, UK, 2011; p. 101. [Google Scholar]
- Nordstrom, D.K. Acid rock drainage and climate change. J. Geochem. Explor. 2009, 100, 97–104. [Google Scholar] [CrossRef]
- Butler, B.A.; Ranville, J.F.; Ross, P.E. Observed and modeled seasonal trends in dissolved and particulate Cu, Fe, Mn, and Zn in a mining-impacted stream. Water Res. 2008, 42, 3135–3145. [Google Scholar] [CrossRef]
- Charlatchka, R.; Cambier, P. Influence of reducing conditions on solubility of trace metals in contaminated soils. Water Air Soil Pollut. 2000, 118, 143–167. [Google Scholar] [CrossRef]
- Wahid, P.A.; Kamalam, N.V. Reductive dissolution of crystalline and amorphous Fe(III) oxides by microorganisms in submerged soil. Biol. Fertil. Soils 1993, 15, 144–148. [Google Scholar] [CrossRef]
- Buckby, T.; Black, S.; Coleman, M.L.; Hodson, M.E. Fe-sulphate-rich evaporative mineral precipitates from the Rio Tinto, southwest Spain. Mineral. Mag. 2003, 67, 263–278. [Google Scholar] [CrossRef]
- Alastuey, A.; Garcia-Sanchez, A.; Lopez, F.; Querol, X. Evolution of pyrite mud weathering and mobility of heavy metals in the Guadiamar valley after the Aznalcollar spill, south-west Spain. Sci. Total Environ. 1999, 242, 41–55. [Google Scholar] [CrossRef]
- Zakir, H.M.; Shikazono, N. Environmental mobility and geochemical partitioning of Fe, Mn, Co, Ni, and Mo in sediments of an urban river. J. Environ. Chem. Ecotoxicol. 2011, 3, 116–126. [Google Scholar]
- Buekers, J.; Amery, F.; Maes, A.; Smolders, E. Long-term reactions of Ni, Zn and Cd with iron oxyhydroxides depend on crystallinity and structure and on metal concentrations. Eur. J. Soil Sci. 2008, 59, 706–715. [Google Scholar] [CrossRef]
- Bradley, S.B.; Lewin, J. Transport of heavy-metals on suspended sediments under high flow conditions in a mineralized region of Wales. Environ. Pollut. Ser. B Chem. Phys. 1982, 4, 257–267. [Google Scholar] [CrossRef]
- Tripole, S.; Gonzalez, P.; Vallania, A.; Garbagnati, M.; Mallea, M. Evaluation of the impact of acid mine drainage on the chemistry and the macrobenthos in the Carolina stream (San Luis-Argentina). Environ. Monit. Assess. 2006, 114, 377–389. [Google Scholar] [CrossRef]
- Fuge, R.; Pearce, F.M.; Pearce, N.J.G.; Perkins, W.T. Acid-Mine Drainage in Wales and Influence of Ochre Precipitation on Water Chemistry. In Environmental Geochemistry of Sulfide Oxidation; Alpers, C.L., Blowes, D.W., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 1994; Volume 550, pp. 261–274. [Google Scholar]
- Lee, G.; Bigham, J.M.; Faure, G. Removal of trace metals by coprecipitation with Fe, Al and Mn from natural waters contaminated with acid mine drainage in the Ducktown Mining District, Tennessee. Appl. Geochem. 2002, 17, 569–581. [Google Scholar] [CrossRef]
- Wen, X.H.; Allen, H.E. Mobilization of heavy metals from Le An river sediment. Sci. Total Environ. 1999, 227, 101–108. [Google Scholar] [CrossRef]
- Gambrell, R.P. Trace and toxic metals in wetlands—A review. J. Environ. Qual. 1994, 23, 883–891. [Google Scholar]
- Ford, R.G.; Bertsch, P.M.; Farley, K.J. Changes in transition and heavy metal partitioning during hydrous iron oxide aging. Environ. Sci. Technol. 1997, 31, 2028–2033. [Google Scholar] [CrossRef]
- Shuman, L.M. Adsorption of Zn by Fe and Al hydrous oxides as influenced by aging and pH. Soil Sci. Soc. Am. J. 1977, 41, 703–706. [Google Scholar] [CrossRef]
- Rance, J.; Wade, S.D.; Hurford, A.P.; Bottins, E.; Reynard, N.S. Climate Change Risk Assessment for the Water Sector; Department for Environment, Food and Rural Affairs: London, UK, 2012. [Google Scholar]
- Ramsbottom, D.; Sayers, P.; Panzeri, M. Climate Change Risk Assessment for the Floods and Coastal Erosion Sector; Department for Environment, Food and Rural Affairs: London, UK, 2012. [Google Scholar]
- Carroll, S.A.; O’Day, P.A.; Piechowski, M. Rock-water interactions controlling zinc, cadmium, and lead concentrations in surface waters and sediments, U.S. Tri-State Mining District. 2. Geochemical interpretation. Environ. Sci. Technol. 1998, 32, 956–965. [Google Scholar] [CrossRef]
- Nordstrom, D.K.; Alpers, C.N. Geochemistry of Acid Mine Waters. In The Environmental Geochemistry of Mineral Deposits. Part A: Processes, Techniques, and Health Issues; Plumlee, G.S., Logsdon, M.J., Filipek, L.H., Eds.; Society of Economic Geologists: Littleton, CO, USA, 1999; Volume 6, pp. 133–160. [Google Scholar]
- Byrne, P.; Reid, I.; Wood, P.J. Stormflow hydrochemistry of a river draining an abandoned metal mine: The Afon Twymyn, Central Wales. Environ. Monit. Assess. 2013, 185, 2817–2832. [Google Scholar] [CrossRef]
- Ponnamperuma, F.N.; Estrella, M.T.; Teresita, L. Redox equilibria in flooded soils: I. The iron hydroxide systems. Soil Sci. 1967, 103, 374–382. [Google Scholar] [CrossRef]
- Van der Geest, H.G.; Paumen, M.L. Dynamics of metal availability and toxicity in historically polluted floodplain sediments. Sci. Total Environ. 2008, 406, 419–425. [Google Scholar]
- Gambrell, R.P.; Delaune, R.D.; Patrick, W.H., Jr. Redox Processes in Soils Following Oxygen Depletion. In Plant Life Under Oxygen Depriviation: Ecology, Physiology and Biochemistry; Jackson, M.B., Davies, D.D., Lambers, H., Eds.; SPB Academic Publishing: The Hague, The Netherlands, 1991; pp. 101–117. [Google Scholar]
- Patrick, W.H.; Delaune, R.D. Characterization of oxidized and reduced zones in flooded soil. Soil Sci. Soc. Am. J. 1972, 36, 573–576. [Google Scholar] [CrossRef]
- Younger, P.L.; Banwart, S.A.; Hedin, R.S. Mine Water: Hydrology, Pollution, Remediation; Kluwer Academic Publishers: London, UK, 2002. [Google Scholar]
- VanLoon, G.W.; Duffy, S.J. Environmental Chemistry, 3rd ed.; Oxford University Press: Oxford, UK, 2011. [Google Scholar]
- Emerson, S.; Cranston, R.E.; Liss, P.S. Redox species in a reducing fjord: Equilibrium and kinetic considerations. Deep Sea Res. Part A Oceanogr. Res. Pap. 1979, 26, 859–878. [Google Scholar] [CrossRef]
- Thamdrup, B.; Fossing, H.; Jorgensen, B.B. Manganese, iron and sulphur cycling in a coastal marine sediment, Aarhus Bay, Denmark. Geochim. Cosmochim. Acta 1994, 58, 5115–5129. [Google Scholar] [CrossRef]
- Rickard, D.; Luther, G.W. Chemistry of iron sulfides. Chem. Rev. 2007, 107, 514–562. [Google Scholar] [CrossRef]
- Gotoh, S.; Patrick, W.H. Transformation of iron in a waterlogged soil as influenced by redox potential and pH. Soil Sci. Soc. Am. J. 1974, 38, 66–71. [Google Scholar] [CrossRef]
- Patrick, W.H.; Henderson, R.E. Reduction and reoxidation cycles of manganese and iron in flooded soil and in water solution. Soil Sci. Soc. Am. J. 1981, 45, 855–859. [Google Scholar] [CrossRef]
- Lovley, D.R.; Phillips, E.J.P. Competitive mechanisms for inhibition of sulfate reduction and methane production in the zone of ferric iron reduction in sediments. Appl. Environ. Microbiol. 1987, 53, 2636–2641. [Google Scholar]
- Lovley, D.R.; Phillips, E.J.P. Organic matter mineralization with reduction of ferric iron in anaerobic sediments. Appl. Environ. Microbiol. 1986, 51, 683–689. [Google Scholar]
- Brennan, E.W.; Lindsay, W.L. Reduction and oxidation effect on the solubility and transformation of iron oxides. Soil Sci. Soc. Am. J. 1998, 62, 930–937. [Google Scholar] [CrossRef]
- Zhao, J.; Huggins, F.E.; Feng, Z.; Huffman, G.P. Ferrihydrite: Surface structure and its effects on phase transformation. Clays Clay Miner. 1994, 42, 737–746. [Google Scholar]
- Cravotta, C.A.; Bilger, M.D. Water-quality trends for a stream draining the Southern Anthracite Field, Pennsylvania. Geochem. Explor. Environ. Anal. 2001, 1, 33–50. [Google Scholar] [CrossRef]
- Stumm, W.; Sulzberger, B. The cycling of iron in natural environments: Considerations based on laboratory studies of heterogeneous redox processes. Geochim. Cosmochim. Acta 1992, 56, 3233–3257. [Google Scholar] [CrossRef]
- Langmuir, D.; Whittemore, D.O. Variations in the Stability of Precipitated Ferric Oxyhydroxides. In Non-Equilibrium Systems in Natural Water Chemistry, Advances in Chemistry Series 106; Gould, R.F., Ed.; American Chemical Society: Washington, DC, USA, 1971; pp. 209–234. [Google Scholar]
- Stumm, W. Chemistry of the Solid-Water Interface, Processes at the Mineral-Water and Particle-Water Interface in Natural Systems; John Wiley & Sons: Hoboken, NJ, USA, 1992. [Google Scholar]
- Lesven, L.; Lourino-Cabana, B.; Billon, G.; Recourt, P.; Ouddane, B.; Mikkelsen, O.; Boughriet, A. On metal diagenesis in contaminated sediments of the Deule River (Northern France). Appl. Geochem. 2010, 25, 1361–1373. [Google Scholar] [CrossRef]
- Davis, J.A.; Kent, D.B. Surface complexation modeling in aqueous geochemistry. Rev. Mineral. Geochem. 1990, 23, 177–260. [Google Scholar]
- Du Laing, G.; Vanthuyne, D.R.; Vandecasteele, B.; Tack, F.M.; Verloo, M.G. Influence of hydrological regime on pore water metal concentrations in a contaminated sediment-derived soil. Environ. Pollut. 2007, 147, 615–625. [Google Scholar] [CrossRef]
- Billon, G.; Ouddane, B.; Laureyns, J.; Boughriet, A. Chemistry of metal sulfides in anoxic sediments. Phys. Chem. 2001, 3, 3586–3592. [Google Scholar]
- Whiteley, J.D.; Pearce, N.J.G. Metal distribution during diagenesis in the contaminated sediments of Dulas Bay, Anglesey, N. Wales, UK. Appl. Geochem. 2003, 18, 901–913. [Google Scholar] [CrossRef]
- Bartlett, R.J. Characterizing Soil Redox Behaviour. In Soil Physical Chemistry, 2nd ed.; Sparks, D.L., Ed.; CRC Press: Boca Raton, FL, USA, 1999; pp. 371–397. [Google Scholar]
- Ross, S. Soil Processes; Routledge: New York, NY, USA, 1989. [Google Scholar]
- Ku, T.C.W.; Kay, J.; Browne, E.; Martini, A.M.; Peters, S.C.; Chen, M.D. Pyritization of iron in tropical coastal sediments: Implications for the development of iron, sulfur, and carbon diagenetic properties, Saint Lucia, Lesser Antilles. Mar. Geol. 2008, 249, 184–205. [Google Scholar] [CrossRef]
- Morse, J.W.; Gledhill, D.K.; Sell, K.S.; Arvidson, R.S. Pyritization of iron in sediments from the continental slope of the northern Gulf of Mexico. Aquat. Geochem. 2002, 8, 3–13. [Google Scholar] [CrossRef]
- Lovley, D.R.; Klug, M.J. Model for the distribution of sulfate reduction and methanogenesis in freshwater sediments. Geochim. Cosmochim. Acta 1986, 50, 11–18. [Google Scholar] [CrossRef]
- Younger, P.L. Coalfield Abandonment: Geochemical Processes and Hydrochemical Products. In Energy and the Environment: Geochemistry of Fossil, Nuclear and Renewable Resources; Nicholson, K., Ed.; Thistle Press: Bellevue, WA, USA, 1998; pp. 1–29. [Google Scholar]
- Andrews, J.E.; Brimblecombe, P.; Jickells, T.D.; Liss, P.S.; Reid, B.J. An Introduction to Environmental Chemistry, 2nd ed.; Blackwell Publishing: Oxford, UK, 2008. [Google Scholar]
- Cranfield, D.E.; Raiswell, R.; Bottrell, S. The reactivity of sedimentary iron minerals toward sulphide. Am. J. Sci. 1992, 292, 659–683. [Google Scholar] [CrossRef]
- Poulton, S.W.; Krom, M.D.; Raiswell, R. A revised scheme for the reactivity of iron (oxyhydr)oxide minerals towards dissolved sulfide. Geochim. Cosmochim. Acta 2004, 68, 3703–3715. [Google Scholar] [CrossRef]
- Raiswell, R.; Cranfield, D.E. Sources of iron for pyrite formation in marine sediments. Am. J. Sci. 1998, 298, 219–245. [Google Scholar] [CrossRef]
- Lord, C.J.L.I.; Church, T.M. The geochemistry of salt marshes: Sedimentary ion diffusion, sulfate reduction and pyritization. Geochim. Cosmochim. Acta 1983, 47, 1381–1391. [Google Scholar] [CrossRef]
- Wilkin, R.T.; Barnes, H.L. Formation processes of framboidal pyrite. Geochim. Cosmochim. Acta 1997, 61, 323–339. [Google Scholar] [CrossRef]
- Starkey, R.L. Oxidation and reduction of sulfur compounds in soils. Soil Sci. 1966, 101, 297–306. [Google Scholar] [CrossRef]
- Lesven, L.; Gao, Y.; Billon, G.; Leermakers, M.; Ouddane, B.; Fischer, J.-C. Early diagenesis processes aspects controlling the mobility of dissolved trace metals in three riverine sediment columns. Sci. Total Environ. 2008, 407, 447–459. [Google Scholar] [CrossRef]
- Schoonen, M.A.A.; Barnes, H.L. Reactions formation pyrite and marcasite from solution: I. Nucleation of FeS2 below 100 °C. Geochim. Cosmochim. Acta 1991, 55, 1495–1504. [Google Scholar] [CrossRef]
- Giblin, A.E. Pyrite formation in marshes during early diagenesis. Geomicrobiol. J. 1988, 6, 77–97. [Google Scholar] [CrossRef]
- Gandy, J.; Jarvis, A.P. The influence of engineering scale and environmental conditions on the performance of compost bioreactors for the remediation of Zinc in mine water discharges. Mine Water Environ. 2012, 31, 82–91. [Google Scholar] [CrossRef]
- Larsen, D.; Mann, R. Origin of high manganese concentrations in coal mine drainage, eastern Tennessee. J. Geochem. Explor. 2005, 86, 143–163. [Google Scholar] [CrossRef]
- Cravotta, C.A. Dissolved metals and associated constituents in abandoned coal-mine discharges, Pennsylvania, USA. Part 2: Geochemical controls on constituent concentrations. Appl. Geochem. 2008, 23, 203–226. [Google Scholar] [CrossRef]
- Caille, N.; Tiffreau, C.; Leyval, C.; Morel, J.L. Solubility of metals in an anoxic sediment during prolonged aeration. Sci. Total Environ. 2003, 301, 239–250. [Google Scholar] [CrossRef]
- Moses, C.O.; Nordstrom, D.K.; Herman, J.S.; Mills, A.L. Aqueous pyrite oxidation by dissolved oxygen and ferric iron. Geochim. Cosmochim. Acta 1987, 51, 1561–1571. [Google Scholar] [CrossRef]
- Merinero, R.; Lunar, R.; Somoza, L.; Diaz-del-Rio, V.; Martinez-Frias, J. Nucleation, growth and oxidation of framboidal pyrite associated with hydrocarbon-derived submarine chimneys: Lessons learned from the gulf of cadiz. Eur. J. Mineral. 2009, 21, 947–961. [Google Scholar] [CrossRef]
- Domenech, C.; Pablo, J.; Ayora, C. Oxidative dissolution of pyritic sludge from the Azanlcollar mine (SW Spain). Chem. Geol. 2002, 190, 339–353. [Google Scholar] [CrossRef]
- Palumbo-Roe, B.; Wragg, J.; Cave, M.; Wagner, D. Effect of weathering product assemblages on Pb bioaccessibility in mine waste: Implications for risk management. Environ. Sci. Pollut. Res. 2013, 20, 7699–7710. [Google Scholar] [CrossRef] [Green Version]
- Hudson-Edwards, K.A. Sources, mineralogy, chemistry and fate of heavy metal-bearing particles in mining-affected river systems. Mineral. Mag. 2003, 67, 205–217. [Google Scholar] [CrossRef]
- Baskerville, S.; Evans, W. Acid Mine Drainage—A Legacy of Industrial Past. Available online: http://www.rsc.org/Education/EiC/Restricted/2006/Mar/AcidMineDrainage.asp (accessed on 29 January 2014).
- Johnson, D.J.; Potter, H.A.B.; Jones, C.; Rolley, S.; Watson, I.; Pritchard, J. Abandoned Mines and the Water Environment; Environment Agency: Bristol, UK, 2008. [Google Scholar]
- Komárek, M.; Zeman, J. Dynamics of Cu, Zn, Cd, and Hg release from sediments at surface conditions. Bull. Geosci. 2004, 79, 99–106. [Google Scholar]
- Palumbo-Roe, B.; Klinck, B.; Banks, V.; Quigley, S. Prediction of the long-term performance of abandoned lead zinc mine tailings in a Welsh catchment. J. Geochem. Explor. 2009, 100, 169–181. [Google Scholar] [CrossRef] [Green Version]
- Byrne, P.; Wood, P.J.; Reid, I. The impairment of river systems by metal mine contamination: A review including remediation options. Crit. Rev. Environ. Sci. Technol. 2012, 42, 2017–2077. [Google Scholar] [CrossRef]
- Grundl, T.; Delwiche, J. Kinetics of ferric oxyhydrixide precipitation. J. Contam. Hydrol. 1993, 14, 71–87. [Google Scholar] [CrossRef]
- Lovley, D.R.; Phillips, E.J.P. Manganese inhibition of microbial iron reduction in anaerobic sediments. Geomicrobiol. J. 1989, 6, 145–155. [Google Scholar] [CrossRef]
- Hem, J.D. Reactions of metal-ions at surfaces of hydrous iron-oxide. Geochim. Cosmochim. Acta 1977, 41, 527–538. [Google Scholar] [CrossRef]
- Whitney, P.R. Relationship of manganese-iron oxides and associated heavy metals to grain size in stream sediments. J. Geochem. Explor. 1975, 4, 251–263. [Google Scholar] [CrossRef]
- Baltpurvins, K.A.; Burns, R.C.; Lawrance, G.A.; Stuart, A.D. Effect of pH and anion type on the aging of freshly precipitated iron(III) hydroxide sludges. Environ. Sci. Technol. 1996, 30, 939–944. [Google Scholar] [CrossRef]
- Kodama, H.; Schnitzer, M. Effect of fulvic acid on the crystallization of Fe(III) oxides. Geoderma 1977, 19, 279–291. [Google Scholar] [CrossRef]
- Anderson, P.R.; Benjamin, M.M. Effects of silicon on the crystallization and adsorption properties of ferric oxides. Environ. Sci. Technol. 1985, 19, 1048–1053. [Google Scholar] [CrossRef]
- Axe, L.; Trivedi, P. Intraparticle surface diffusion of metal contaminants and their attenuation in microporous amorphous Al, Fe, and Mn oxides. J. Colloid Interface Sci. 2002, 247, 259–265. [Google Scholar] [CrossRef]
- Webster, J.G.; Swedlund, P.J.; Webster, K.S. Trace metal adsorption onto an acid mine drainage iron(iii) oxyhydroxy sulfate. Environ. Sci. Technol. 1998, 32, 1361–1368. [Google Scholar] [CrossRef]
- Burton, G.A. Metal bioavailability and toxicity in sediments. Crit. Rev. Environ. Sci. Technol. 2010, 40, 852–907. [Google Scholar] [CrossRef]
- Desbarats, A.J.; Dirom, G.C. Temporal variationsin the chemistry of circum-neutral drainage from the 10-level portal, Myra Mine, Vancouver Island, British Columbia. Appl. Geochem. 2007, 22, 415–435. [Google Scholar] [CrossRef]
- Evans, D. Chemical and physical partitioning in contaminated stream sediments in the River Ystwyth, mid-Wales. Environ. Geochem. Health 1991, 13, 84–92. [Google Scholar] [CrossRef]
- Stone, M.; Droppo, I.G. Distribution of lead, copper and zinc in size-fractionated river bed sediment in two agricultural catchments of southern Ontario, Canada. Environ. Pollut. 1996, 93, 353–362. [Google Scholar] [CrossRef]
- Wang, L.; Yu, R.; Hu, G.; Tu, X. Speciation and assessment of heavy metals in surface sediments of Jinjiang River tidal reach, southeast of China. Environ. Monit. Assess. 2010, 165, 491–499. [Google Scholar] [CrossRef]
- Coston, J.A.; Fuller, C.C.; Davis, J.A. Pb2+ and Zn2+ adsorption by a natural aluminium- and iron-bearing surface coating on an aquifer sand. Geochim. Cosmochim. Acta 1995, 59, 3535–3547. [Google Scholar] [CrossRef]
- Desbarats, A.J.; Dirom, G.C. Temporal variation in discharge chemistry and portal flow from the 8-level adit, Lynx Mine, Myra Falls operations, Vancouver Island, British Colombia. Environ. Geol. 2005, 47, 445–456. [Google Scholar] [CrossRef]
- Burton, E.D.; Phillips, I.R.; Hawker, D.W. Geochemical partitioning of copper, lead and zinc in benthic, estuarine sediment profiles. J. Environ. Qual. 2005, 34, 263–273. [Google Scholar] [CrossRef]
- Jean, G.E.; Bancroft, G.M. Heavy metal adsorption by sulphide mineral surfaces. Geochim. Cosmochim. Acta 1986, 50, 1455–1463. [Google Scholar] [CrossRef]
- Galan, E.; Gomex-Ariza, J.L.; Gonzalez, I.; Gernandez-Caliani, J.C.; Morales, E.; Giraldez, I. Heavy metal partitioning in river sediments severely polluted by acid mine drainage in the Iberian pyrite belt. Appl. Geochem. 2003, 18, 409–421. [Google Scholar] [CrossRef]
- Hamilton-Taylor, J.; Giusti, L.; Davison, W.; Tych, W.; Hewitt, C.N. Sorption of trace metals (Cu, Pb, Zn) by suspended lake particles in artificial (0.005 m NaNO3) and natural (Esthwaite water) freshwaters. Colloids Surf. A Physicochem. Eng. Asp. 1997, 120, 205–219. [Google Scholar] [CrossRef]
- Ghanem, S.A.; Mikkelsen, D.S. Sorption of zinc on iron hydrous oxide. Soil Sci. 1988, 146, 15–21. [Google Scholar] [CrossRef]
- Torres, E.; Ayora, C.; Canovas, C.R.; Garcia-Robledo, E.; Glalvan, L.; Sarmiento, A.M. Metal cycling during sediment early diagenesis in a water reservoir affected by acid mine drainage. Sci. Total Environ. 2013, 416–429. [Google Scholar]
- Ford, R.G.; Kemner, K.M.; Bertsch, P.M. Influence of sorbate-sorbent interactions on the crystallization kinetics of nickel- and lead-ferrihydrite coprecipitates. Geochim. Cosmochim. Acta 1999, 63, 39–48. [Google Scholar] [CrossRef]
- Majzlan, J.; Lalinska, B.; Chovan, M.; Jurkovic, L.; Milovska, S.; Gottlicher, J. The formation, structure, and ageing of As-rich hydrous ferric oxide at the abandoned Sb deposit Pezinok (Slovakia). Geochim. Cosmochim. Acta 2007, 71, 4206–4220. [Google Scholar] [CrossRef]
- Hudson-Edwards, K.A.; Jamieson, H.E.; Charnock, J.M.; Macklin, M.G. Arsenic speciation in waters and sediment of ephemeral floodplain pools, Rios Agrio-Guadiamar, Aznalcollar, Spain. Chem. Geol. 2005, 219, 175–192. [Google Scholar] [CrossRef]
- Alpers, C.N.; Nordstrom, D.K.; Thompson, J.M. Seasonal variations of Zn/Cu ratios in acid mine water from iron mountain, California. Am. Chem. Soc. 1993, 550, 324–344. [Google Scholar]
- Hudson-Edwards, K.A.; Schell, C.; Macklin, M.G. Mineralogy and geochemistry of alluvium contaminated by metal mining in the Rio Tinto area, Southwest Spain. Appl. Geochem. 1999, 14, 1015–1030. [Google Scholar] [CrossRef]
- Taylor, M.P.; Hudson-Edwards, K.A. The dispersal and storage of sediment-associated metals in an arid river system: The Leichhardt River, Mount Isa, Queensland, Australia. Environ. Pollut. 2008, 152, 193–204. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lynch, S.F.L.; Batty, L.C.; Byrne, P. Environmental Risk of Metal Mining Contaminated River Bank Sediment at Redox-Transitional Zones. Minerals 2014, 4, 52-73. https://doi.org/10.3390/min4010052
Lynch SFL, Batty LC, Byrne P. Environmental Risk of Metal Mining Contaminated River Bank Sediment at Redox-Transitional Zones. Minerals. 2014; 4(1):52-73. https://doi.org/10.3390/min4010052
Chicago/Turabian StyleLynch, Sarah F. L., Lesley C. Batty, and Patrick Byrne. 2014. "Environmental Risk of Metal Mining Contaminated River Bank Sediment at Redox-Transitional Zones" Minerals 4, no. 1: 52-73. https://doi.org/10.3390/min4010052




