1. Introduction
The genus
Leptothrix, a type of Fe/Mn-oxidizing bacteria belonging to the aquatic β-proteobacteria [
1,
2,
3], is characterized by its formation of an extracellular, tubular, Fe- or Mn-encrusted sheath, enabled by the Fe/Mn-oxidizing capacities of this genus [
4,
5,
6]. The formation of a sheath that contains bacterial cells and extracellular metal-oxidizing activities are the two major phenotypic criteria that distinguish
Leptothrix species from closely related bacteria [
1,
7]. Although the presence of a sheath is thought to confer nutritional and ecological advantages to species of
Sphaerotilus and
Leptothrix and enables the bacterial cells to attach themselves to solid surfaces and protects them against parasites and predators [
1,
3], sufficient evidence to explain the significance of the sheath in these bacteria is still lacking.
Recently, Takeda
et al. [
8] studied sheath elongation in
L. cholodnii in detail using fluorescence and phase-contrast microscopy and discovered that the bacterial cells continuously elongated and divided regardless of their position in the sheath and that the sheath itself was open-ended and elongated at its terminal regions. This observation is consistent with the earlier description by Ghiorse [
9] that sheath formation of
S. natans and
Leptothrix spp. takes place during elongation of bacterial cells at the ends of cell chains. Their results indicate that the bacterial cells maintain their ability to proliferate and continuously excrete polymers, which leads to the formation of the initial frame of sheath [
3,
4,
5]. When isolated strains are cultured in artificial media, most sheaths contain bacterial cells at an early stage of incubation [
2,
4,
5,
8]. However, the majority of sheaths, especially those that are thick and yellow-brownish, become at least partially empty within a few days after the start of the incubation, resembling most of the sheaths of
L. ochracea when harvested from natural aquatic environments [
10,
11]. According to Emerson
et al. [
12], when the sheaths are actively growing, only approximately 10% of the sheaths contain filaments of rod-shaped bacterial cells. Ghiorse noted in his review [
9] that in low-nutrient, Fe-containing enrichment cultures,
L. ochracea synthesized and vacated iron-encrusted sheaths very rapidly, leaving behind a large mass of empty sheaths. However, the manner in which the sheaths were vacated was not explained. Are the bacterial cells released one by one from the open ends of the sheaths? Or degraded by cell autolysis or some other process? Evidence to answer these queries still remains insufficient.
As illustrated in
Figure 1A, almost all sheaths of
L. ochracea harvested from the pilot groundwater tank at the farm of Okayama University had clear thick walls. Although most of these sheaths looked empty, a few chained bacterial cells were occasionally seen within some sheaths. Treatment of such sheaths with a protein-specific fluorescent dye, Ruby, a protein detector frequently used in molecular biology, clearly marked the location of active bacterial cells (
Figure 1B, solid arrow) and occasional intercellular spaces running lengthwise within the sheaths. In addition, small fluorescent aggregates of various sizes were occasionally seen in these intercellular spaces (
Figure 1B, dotted arrows). These observations led us to assume that such spaces and aggregates might be associated with the hollowing mechanism in the sheaths. To verify this assumption, we studied the behavior of cells of
Leptothrix sp. strain OUMS1 (NITE BP-860) (hereafter referred to as OUMS1) cultivated in an artificial medium and in natural groundwater using light and electron microscopy.
Figure 1.
Sheaths produced by Leptothrix ochracea harvested from natural aquatic environment. (A) Thick, yellowish-brown, empty sheaths and (B) entire bacterial cell (solid arrow) and proteinic remnants (dotted arrows) within a sheath detected by protein-specific reagent, Ruby. Scale bar = 10 μm.
Figure 1.
Sheaths produced by Leptothrix ochracea harvested from natural aquatic environment. (A) Thick, yellowish-brown, empty sheaths and (B) entire bacterial cell (solid arrow) and proteinic remnants (dotted arrows) within a sheath detected by protein-specific reagent, Ruby. Scale bar = 10 μm.
2. Results and Discussion
When OUMS1 was cultured in silicon–glucose–peptone broth (SGP broth [
13]) with a small iron plate (hereafter referred to as SGPI broth) for 3 days, a great number of the initial sheaths surrounding chained cells were seen in addition to abundant free or aggregated cells, as reported previously [
6]. The adjacent, aligned cells (
Figure 2A, solid arrow) in the initial sheath comprised a faint, thin film but were not always tightly connected, and thus intercellular spaces were often observed with DIC microscopy (
Figure 2A, dotted arrow). Most of the cells enclosed in sheaths proved to be alive by fluorescing green after the bacterial viability kit staining (LIVE/DEAD
® BacLight™, L7007, Molecular Probes, Eugene, OR, USA) (hereafter referred to as L/D staining) [
4] (
Figure 2B). The characteristic blue fluorescence of 4′,6-diamidino-2-phenylindole dihydrochloride
n-hydrate (DAPI, WAKO Chemicals, Osaka, Japan) [
4] showed nucleic acids in the chained cells (
Figure 2C). Probably because of its intensity, the blue fluorescence seemed to be emitted from the entirety of each cell, and the precise location of nucleic acids within the cells could not be determined. Although the body marked by a solid arrow in
Figure 2A looks like one independent long cell with DIC imagery, the L/D and DAPI double-stained image (
Figure 2B,C) revealed a slight constriction at a center of the body, and the fluorescing nucleic acid regions were “split” on each side of the constriction. Such images may represent mid-stream cell division as described by Takeda
et al. [
2], indicating that cell division and sheath elongation take place successively in the medium. Van Veen
et al. [
3] noted in their review that the cells of
L. ochracea moved out of the sheaths at a rate of 1 to 2 μm/min, and subsequently produce new sheaths, suggesting that the formation of empty sheaths might ascribe to cellular motility. If the cell division occurs always at the end of sheath, the release of cells from open ends of sheath may be persuasive. However, the present observations (
Figure 2B,C) showing the continuous occurrence of cell division at the central part of sheaths raised a question how these cells move out of the sheaths. Although most sheaths contained single and chained live cells, sheaths having fully shaped cells and lengthwise empty-looking intercellular spaces were found intermittently. In the DIC image
Figure 2D, solid and dotted arrows indicate bacterial cells and empty-looking spaces, respectively. In the same view area, L/D detected live cells with greenish fluorescence (
Figure 2E, solid arrows) and dead cells with reddish fluorescence (
Figure 2E, dotted arrow). DAPI detected fluorescent nucleic acid in the live cells (
Figure 2F, solid arrows) and atypical pinkish fluorescence from dead cells with damaged membranes (
Figure 2F, dotted arrows). These observations warned us not to judge the emptiness of sheaths only by DIC imaging.
It is noteworthy that the sheaths in
Figure 2D were coated with fibrous materials (
Figure 2D, fm). Furutani
et al. [
4] reported that cultivation of OUMS1 in silicon-iron-glucose-peptone (SIGP) medium [
6] with three iron plates led to the formation of sheaths with extremely thick walls, with an outer coat covered with thin, loosely woven fibrils and thick assembled fibrils. Suzuki
et al. [
14] observed that when OUMS1 was cultured in natural groundwater by the dialysis tube method, the sheath wall became much thinner; thus artificial cultivation was considered to promote the excretion of polymers from the cells, resulting in wall thickening. The fibrous materials on the sheath surfaces in
Figure 2D most likely correspond to the fibrous assembly of extracellular polymers from the bacterial cells, as Furutani
et al. [
4] reported.
As illustrated in
Figure 2G–I, the continuous cultivation of OUMS1 until day 7 resulted in more sheaths with dead cells. Most sheaths had sharply defined walls coated with fibrous materials (
Figure 2G). Bacterial cells were barely distinguishable in the sheaths when viewed with DIC (
Figure 2G, solid arrows) and were interspersed with granular zones (
Figure 2G, dotted arrows) in the intercellular spaces. These barely distinguishable cells fluoresced when stained with L/D or DAPI, showing them to be live cells with nucleic acids (
Figure 2H,I). By contrast, dead cells were detected in the indistinct, granular, intercellular zones (
Figure 2G, dotted arrows) (
Figure 2H,I). There was no regularity in the appearance of dead cells in the cell chains, but fewer cells tended to die at the ends of the sheath. The clearly delineated sheath zone (marked with an arrowhead in
Figure 2G) never responded to L/D or DAPI (
Figure 2H,I), suggesting that the bacterial cells and their remnants completely degenerated there to leave an empty sheath. All these observations led us to consider that formation of empty sheaths was at least partially due to autolysis of the bacterial cells in addition to cell release from the sheath ends as suggested by van Veen [
3]. However, we cannot rule out the possibility that such autolysis could be caused by artificial cultivation.
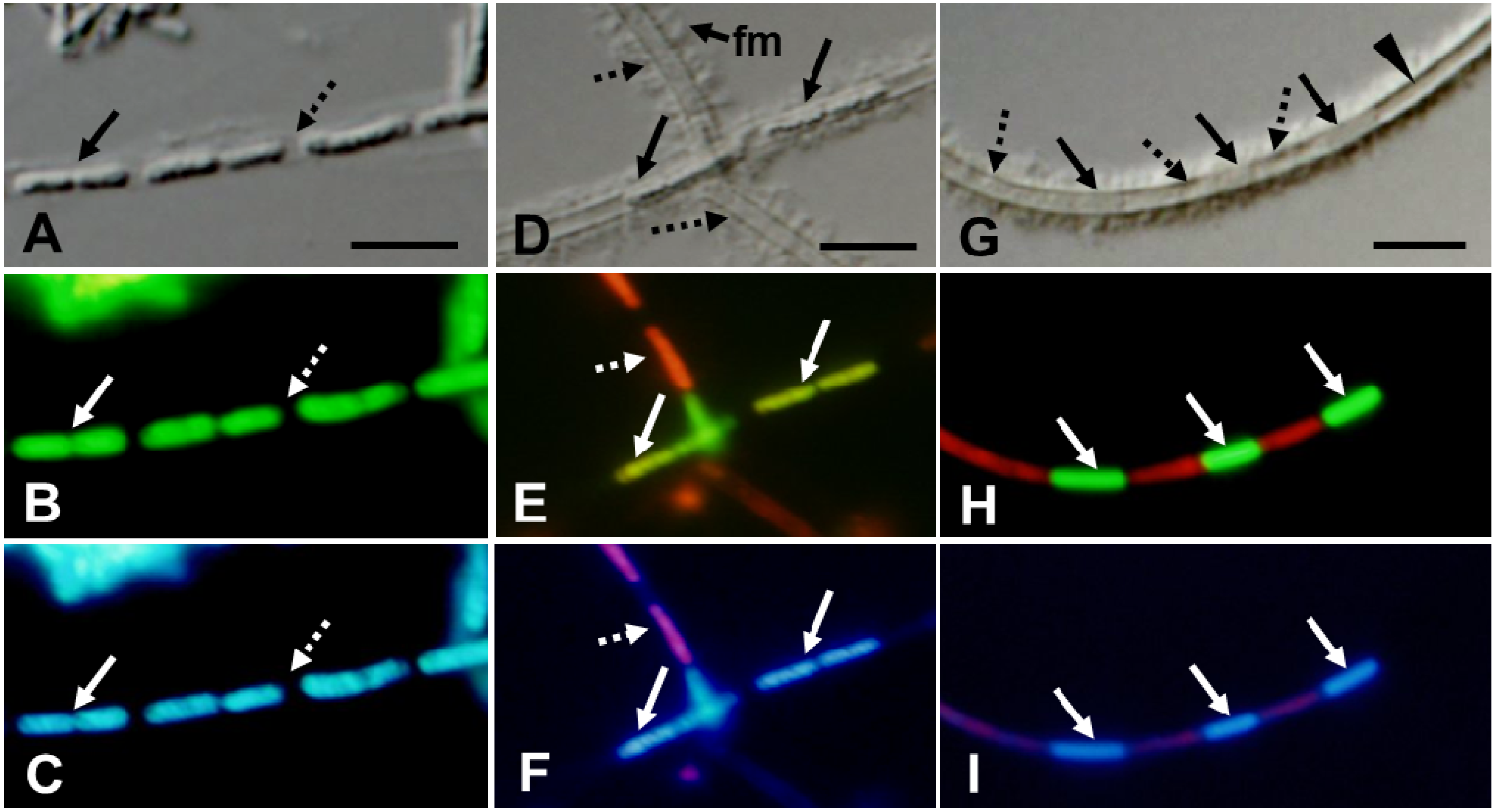
Figure 2.
Strain OUMS1 and associated structures cultured in SGPI broth for 3 (A–F) and 7 days (G–I); DIC images (A,D,G) and fluorescent images from L/D stain (B,E,H) and from DAPI stain (C,F,I); images within a column are of the same cells. (A–C) Line of bacterial cells (solid arrow) and lengthwise intercellular spaces (dotted arrow) are encompassed by a faint, thin sheath. The cells (solid arrow) fluoresced a typical green from the L/D stain (vital sign), their nucleic acids fluoresced blue from DAPI, but the intercellular spaces (dotted arrow) did not fluoresce. Note the constriction at the center of the bacterial body [solid arrow in (B) and (C)], signifying active cell division. (D–F) DIC-fluorescing cells (solid arrows) and barely fluorescing cells (dotted arrows) are enclosed with clearly defined walls covered with fibrous materials (fm). L/D and DAPI detected live cells (solid arrows) with intact nucleic acids and dead cells (dotted arrow) with atypical nucleic acids fluorescing pinkish. And (G–I) Barely distinguishable cells (solid arrows) enclosed with a thick-walled sheath, empty-looking (dotted arrows) and transparent zones with no detectable cells or cell remnants (arrowhead) in (G). Solid arrows: live cells with intact nucleic acid response; dotted arrows: dead cells with degenerated nucleic acids. Scale bar = 5 μm.
Figure 2.
Strain OUMS1 and associated structures cultured in SGPI broth for 3 (A–F) and 7 days (G–I); DIC images (A,D,G) and fluorescent images from L/D stain (B,E,H) and from DAPI stain (C,F,I); images within a column are of the same cells. (A–C) Line of bacterial cells (solid arrow) and lengthwise intercellular spaces (dotted arrow) are encompassed by a faint, thin sheath. The cells (solid arrow) fluoresced a typical green from the L/D stain (vital sign), their nucleic acids fluoresced blue from DAPI, but the intercellular spaces (dotted arrow) did not fluoresce. Note the constriction at the center of the bacterial body [solid arrow in (B) and (C)], signifying active cell division. (D–F) DIC-fluorescing cells (solid arrows) and barely fluorescing cells (dotted arrows) are enclosed with clearly defined walls covered with fibrous materials (fm). L/D and DAPI detected live cells (solid arrows) with intact nucleic acids and dead cells (dotted arrow) with atypical nucleic acids fluorescing pinkish. And (G–I) Barely distinguishable cells (solid arrows) enclosed with a thick-walled sheath, empty-looking (dotted arrows) and transparent zones with no detectable cells or cell remnants (arrowhead) in (G). Solid arrows: live cells with intact nucleic acid response; dotted arrows: dead cells with degenerated nucleic acids. Scale bar = 5 μm.
![Minerals 03 00247 g002]()
To verify this possibility, OUMS1 was cultivated in natural groundwater using the dialysis tube method [
14]. As illustrated in
Figure 3A, a line of fully shaped bacterial cells (solid arrows) and small aggregates (dotted arrow) were occasionally seen in a clearly delineated sheath after 3 days in groundwater. As expected, the fully shaped cells showed positive L/D and DAPI responses (
Figure 3B,C, arrows), while the small aggregates did not fluoresce from L/D (
Figure 3B, dotted arrow) and gave only a slightly blue DAPI response (
Figure 3C, dotted arrow), suggesting that the nucleic acids had probably degenerated. After 7 days, more dead cells appeared in sheaths (
Figure 3D–F, dotted arrow). Again, in cells that looked fully shaped in the DIC image (
Figure 3D, solid arrows) actually had a slight constriction in the center of the body in the L/D and DAPI images (
Figure 3E,F, solid arrows). These observations demonstrated that bacterial cells were successively dividing within the sheath and that a number of cells had autolysed within the same sheath, regardless of their position, similar to cultivation in broth. In groundwater cultivation, abundant particle assemblies always gathered at discrete locations along the sheaths (
Figure 3A,D, ap). Van Veen
et al. [
3] reported a similar phenomenon when
L. cholodnii was grown in the presence of Mn
2+; the sheaths became irregularly covered with granular manganese oxide. In a separate SEM-EDX observation, these particles were shown to contain Fe, O, P and Si as major elements, suggesting that they could aggregate via autooxidation of aquatic-phase elements and/or unidentified chemical bonds (Appendix
Figure A1).
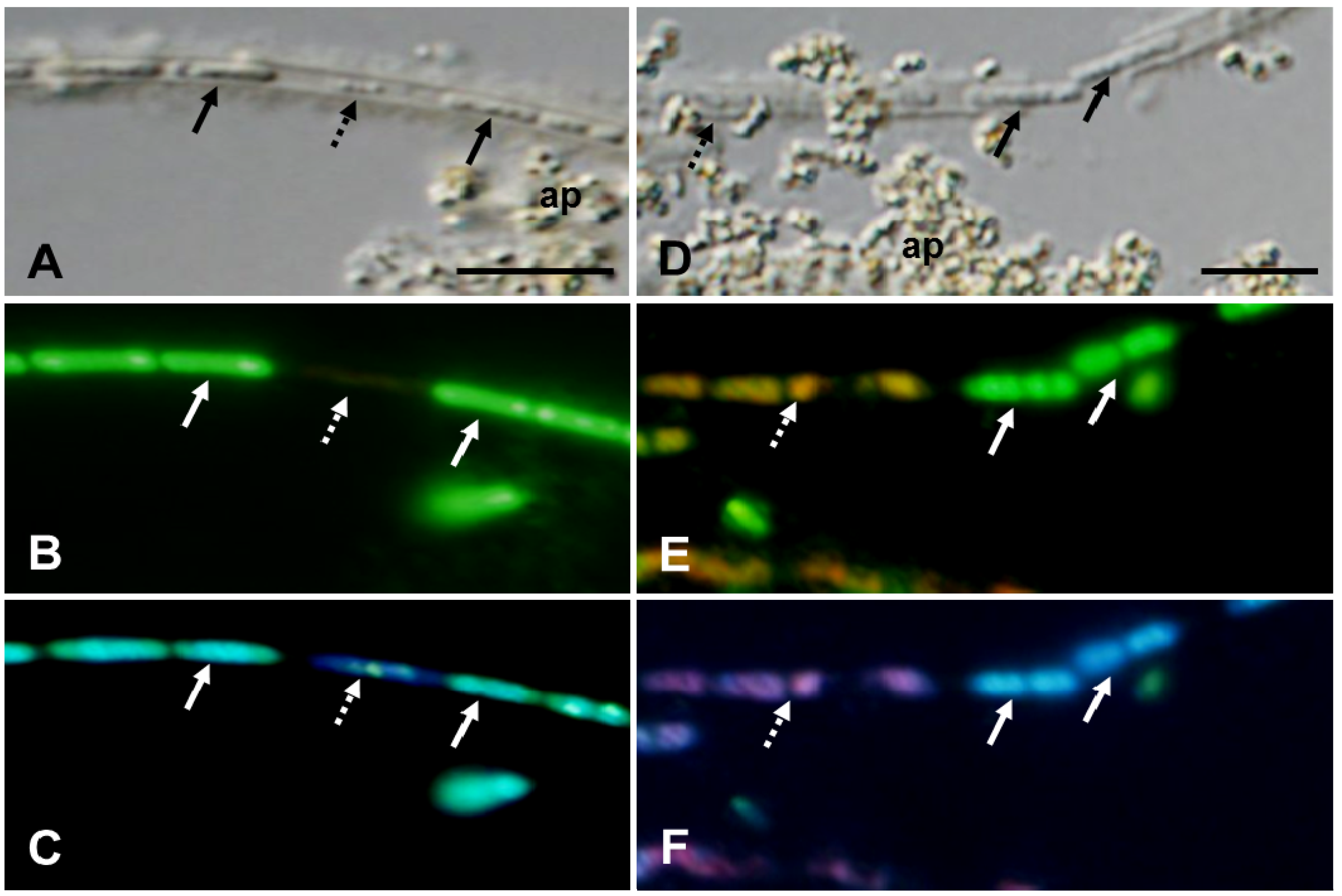
Figure 3.
OUMS1 and associated structures cultivated in groundwater for 3 (A–C) and 7 days (D–F). DIC images (A,D) and L/D- (B,E) and DAPI- (C,F) fluorescent images. (A–C) Line of cells (solid arrows) and cell remnants (dotted arrow) enclosed in a thick-walled sheath. Fully shaped cells (solid arrows) with a live response and sign of nucleic acids and cell remnants (dotted arrow) lacking a vital response and nucleic acids. Assembled particles (ap) are seen at discrete locations along the sheaths. And (D–F) fully shaped live cells with fluorescing nucleic acids (solid arrows) and deformed, and dead cells with damaged DNA (dotted arrow) in a thick-walled sheath. Scale bar = 5 μm.
Figure 3.
OUMS1 and associated structures cultivated in groundwater for 3 (A–C) and 7 days (D–F). DIC images (A,D) and L/D- (B,E) and DAPI- (C,F) fluorescent images. (A–C) Line of cells (solid arrows) and cell remnants (dotted arrow) enclosed in a thick-walled sheath. Fully shaped cells (solid arrows) with a live response and sign of nucleic acids and cell remnants (dotted arrow) lacking a vital response and nucleic acids. Assembled particles (ap) are seen at discrete locations along the sheaths. And (D–F) fully shaped live cells with fluorescing nucleic acids (solid arrows) and deformed, and dead cells with damaged DNA (dotted arrow) in a thick-walled sheath. Scale bar = 5 μm.
When specimens cultured in SGPI broth for 3 days were observed with TEM, some sheaths, characterized by a thin or electron-dense thick wall, enclosed bacterial cells (
Figure 4A, arrows), while other sheaths contained degenerated and deformed cell remnants (
Figure 4A, dotted arrows), as reported for cultured
L. discophora SP-6 by Emerson and Ghiorse [
7]. As illustrated in a longitudinal section of a sheath (
Figure 4B), a body that maintained the shape of a cell (arrow) occasionally coexisted side-by-side with cell remnants (dotted arrow) within a sheath. The extremely electron-dense fibrous assembly in the sheath zone corresponds to saccharic bacterial fibrils incorporated with aquatic inorganic elements [
5].
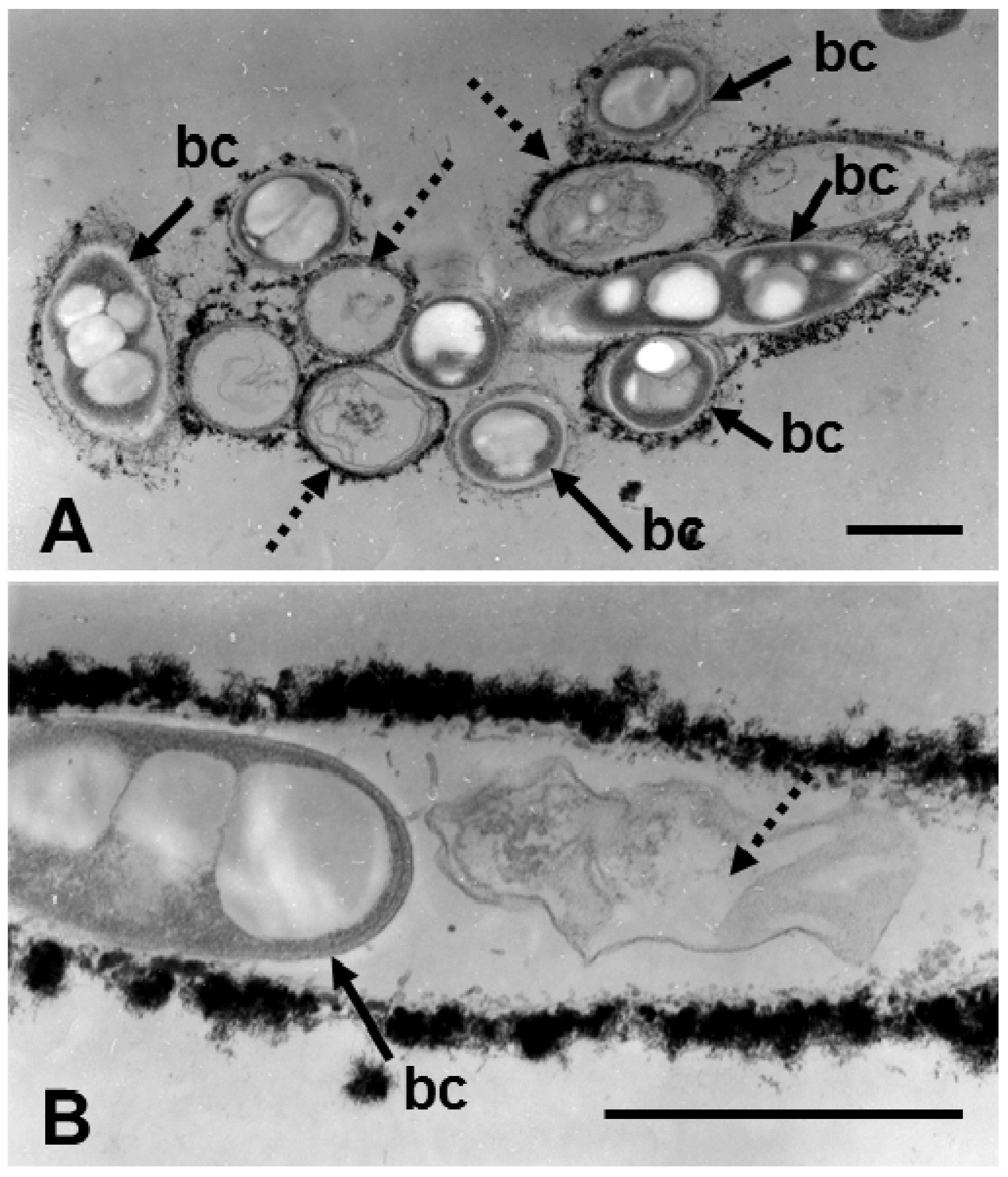
Figure 4.
Transmission electron micrographs of cross (A) and longitudinal sections (B) of sheaths harvested on day 3 of culture in SGPI broth. (A) Some sheaths enclosed fully shaped cells (bc), but others contained deformed cells or their remnants (dotted arrows). And (B) thick, electron-dense sheath walls enclose a fully shaped cell and cell remnants (dotted arrow) side by side. Scale bar = 1 μm.
Figure 4.
Transmission electron micrographs of cross (A) and longitudinal sections (B) of sheaths harvested on day 3 of culture in SGPI broth. (A) Some sheaths enclosed fully shaped cells (bc), but others contained deformed cells or their remnants (dotted arrows). And (B) thick, electron-dense sheath walls enclose a fully shaped cell and cell remnants (dotted arrow) side by side. Scale bar = 1 μm.
In a similar TEM study of specimens cultivated in groundwater for 3 and 7 days, some of the 3-day sheaths were comprised of a fibrous assembly, and cells were in a line but separated by the intercellular spaces (
Figure 5A). As Takeda
et al. [
8] reported, at the end of the initial sheath comprised of fine fibrils, two cells were occasionally seen to be connected by a thin constriction (
Figure 5B, arrow), most likely reflecting the phase right before completion of cell division. Such a cell may readily move out of the end of sheaths, as noted by van Veen [
3]. In most of the 7-day sheaths, aligned cells were separated by broader intercellular spaces, and their cytoplasm was electron-lucent, suggesting initiation of autolysis (
Figure 5C). Some cells in the sheaths had lost organelles, and the cytoplasm had become granular, signs of autolysis (
Figure 5D). The presence of intercellular spaces likely reflects that free, independent cells could move after cell division within fluid-phased sheaths. Although OUMS1 cells have been reported to have a monotrichous, polar flagellum [
4], it is unknown whether the movement of cells is due to active flagellar locomotion and/or passive fluid flow within the sheaths. The possibility remains that such solo travel of free cells within sheaths may trigger their early autolysis after cell division, because it is generally accepted that formation of a biofilm on solid surfaces and/or colony through the attachment of cells are essential for survival [
3,
15].
All these observations demonstrated that (i) bacterial cells maintain the ability to proliferate within sheaths; (ii) cells separate from adjacent cells soon after cell division; (iii) cells seem to autolyse probably within a short time after cell division (the precise time is unknown at present, because the life span of bacterial cells is largely influenced by the species, environmental and nutritional factors as noted by Madigan
et al. [
15]); and (iv) their autolysis leads to a hollowing of the
Leptothrix sheaths. Since the presence of the phage-related intergrases and DNA polymerase-related components is shown in genomic DNA of
L. cholodnii SP-6 (DNA Data Bank of Japan), possible involvement of the template phage in autolysis of the cells can not be ruled out. Based on the present microscopic observation and the literature information, we are continuing to elucidate why and when the cells autolyse within the sheaths.
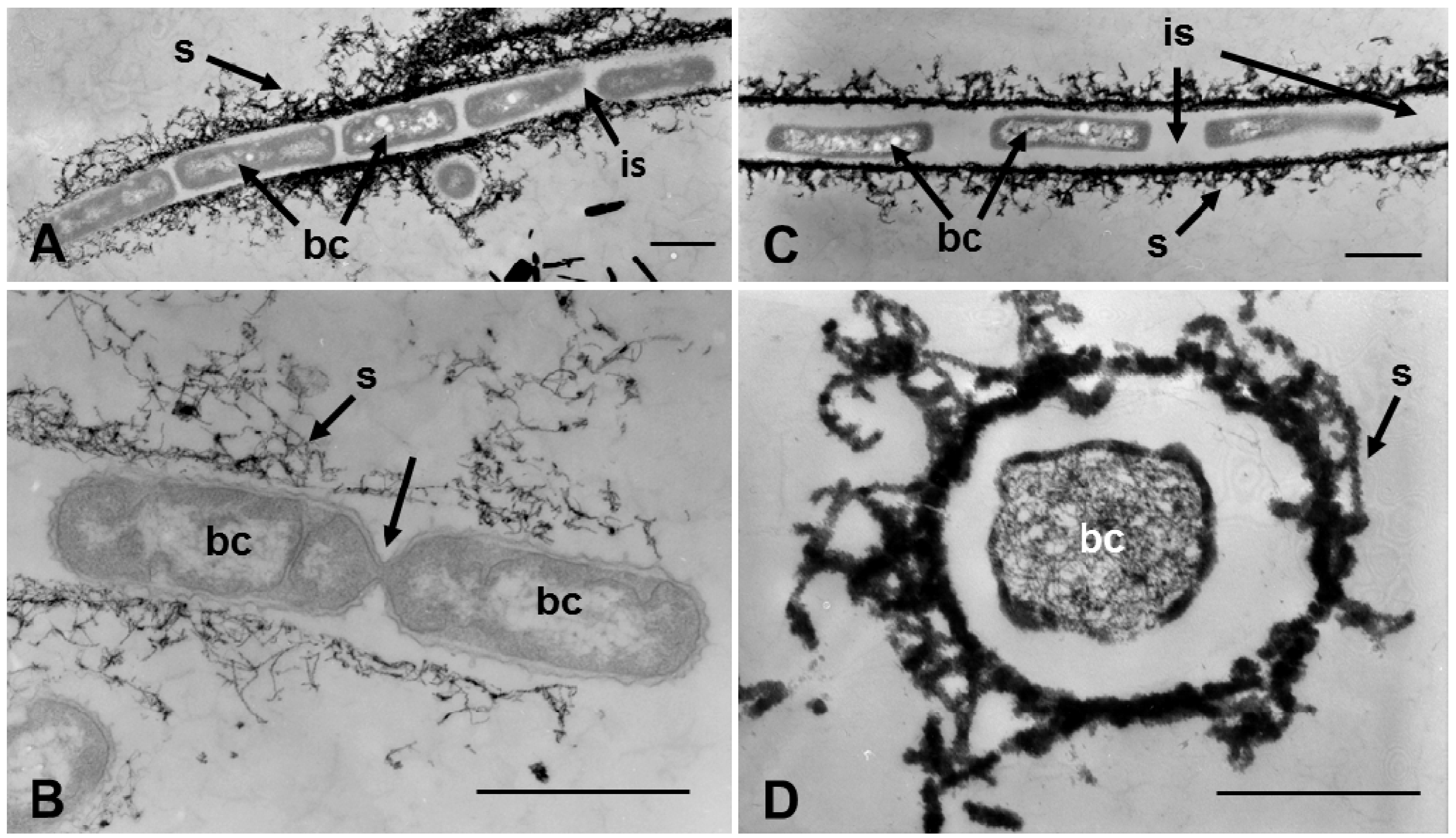
Figure 5.
Transmission electron micrographs of sheaths harvested at day 3 (A,B) and 7 (C,D) of culture in groundwater. (A) Cells (bc) aligned in a sheath (s) composed of fibrous materials. Note that adjacent cells in the sheath are separated by intercellular spaces (is). (B) Two dividing cells connected to each other by a thin connection (arrow). The sheath comprises an assembly of thin fibrils, suggesting that this part of sheath could be the terminal end. (C) Cells separated by broad intercellular spaces in a sheath harvested from 7-day culture. Their cytoplasm has begun to degenerate. And (D) Cell with granular cytoplasm (a sign of degeneration). (A–C) Scale bar = 1 μm; (D) Scale bar = 500 nm.
Figure 5.
Transmission electron micrographs of sheaths harvested at day 3 (A,B) and 7 (C,D) of culture in groundwater. (A) Cells (bc) aligned in a sheath (s) composed of fibrous materials. Note that adjacent cells in the sheath are separated by intercellular spaces (is). (B) Two dividing cells connected to each other by a thin connection (arrow). The sheath comprises an assembly of thin fibrils, suggesting that this part of sheath could be the terminal end. (C) Cells separated by broad intercellular spaces in a sheath harvested from 7-day culture. Their cytoplasm has begun to degenerate. And (D) Cell with granular cytoplasm (a sign of degeneration). (A–C) Scale bar = 1 μm; (D) Scale bar = 500 nm.