Continuous Hydrogen Production via Hydrothermal Gasification of Biodiesel Industry Wastewater: Experimental Optimization and Energy Integration Simulation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Influent Sample
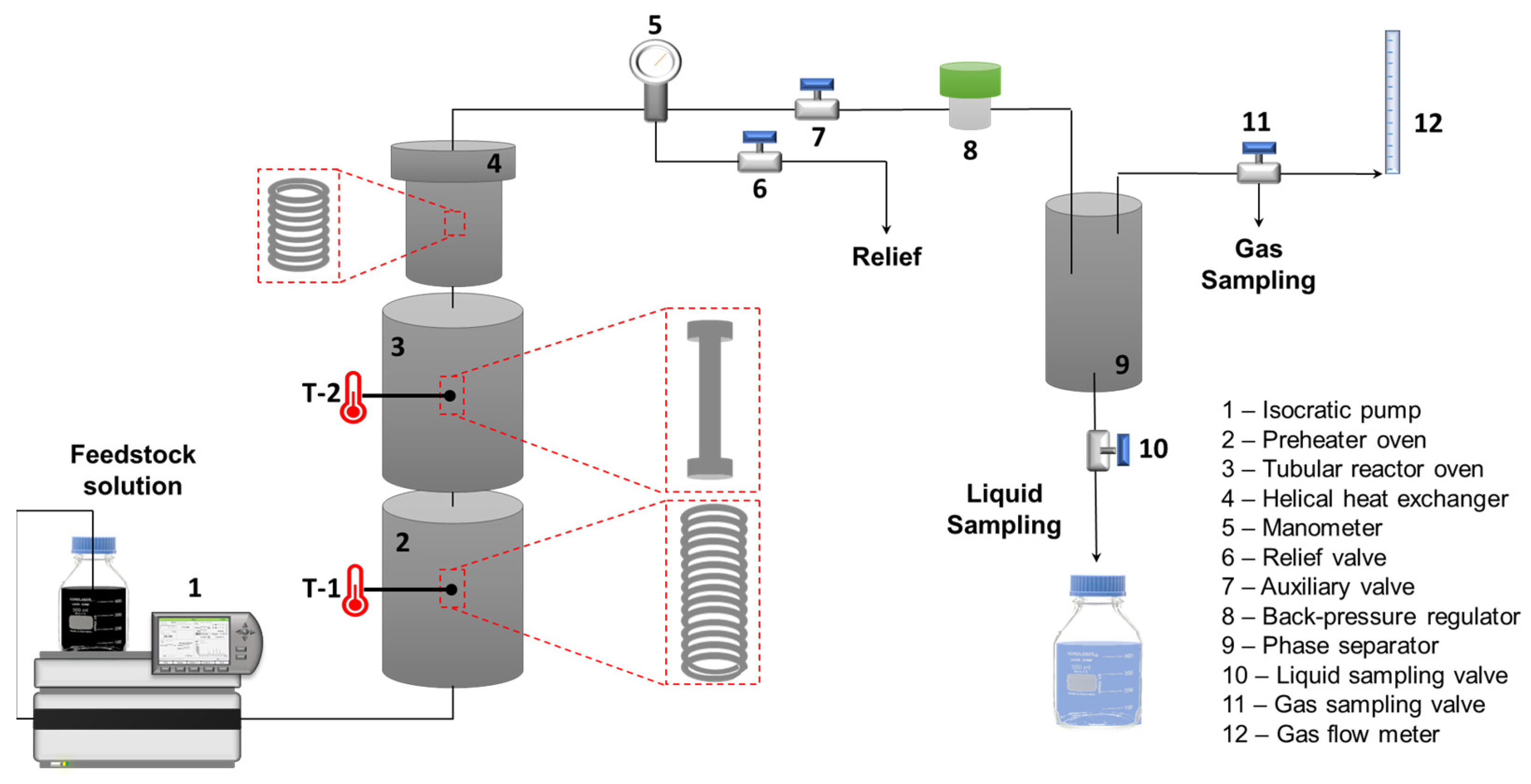
2.2. Reaction System
2.3. Chemical Characterization
2.4. Experimental Conditions and Central Composite Design
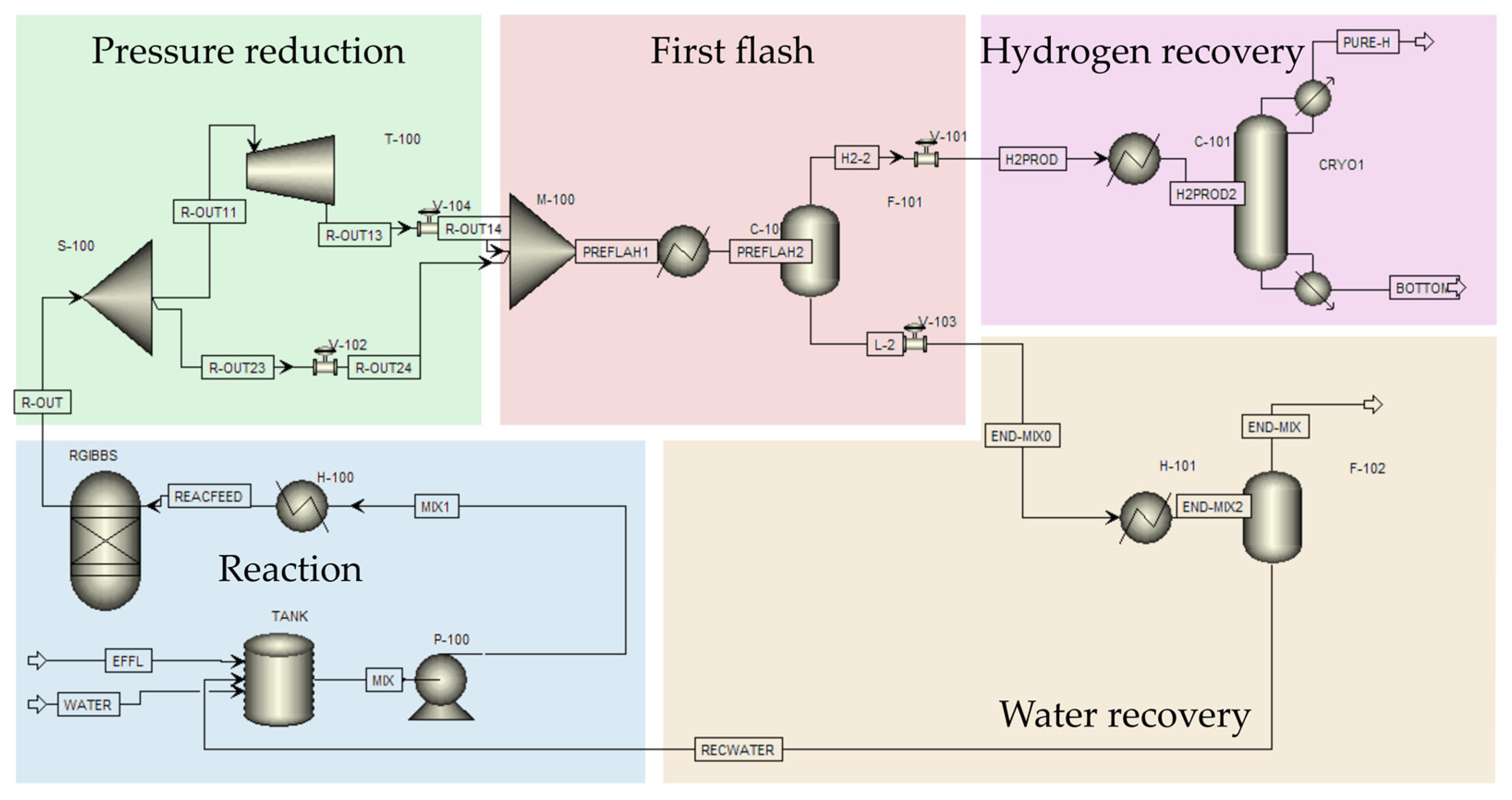
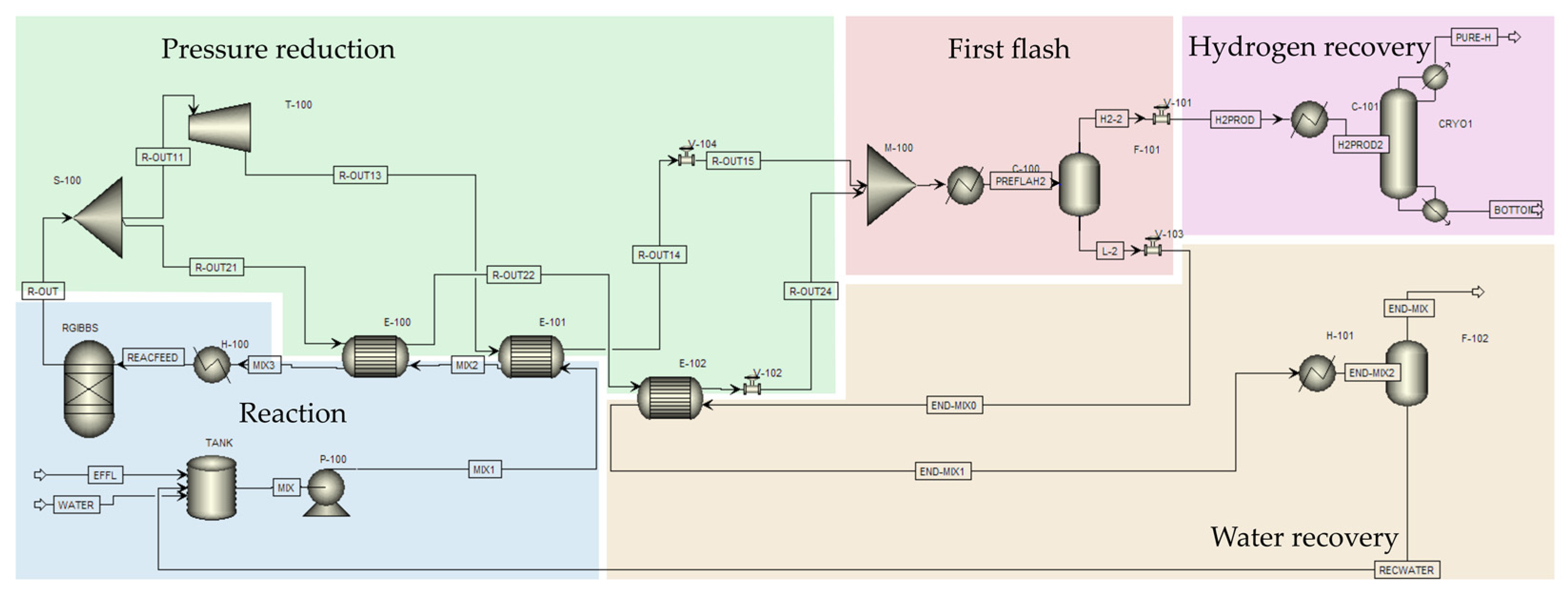
2.5. Scale-Up Process Simulation and Energy Efficiency Analysis
3. Results and Discussion
3.1. SCWG of Biodiesel Industry Wastewater
3.2. SCW Process Optimization: Effect of the Operational Parameters
3.3. Validation of the Mathematical Regression Model
3.4. Results of the Effluent Treatment
3.5. Insights on Energy-Efficient Scale-Up
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Adamu, A.; Russo-Abegão, F.; Boodhoo, K. Process Intensification Technologies for CO2 Capture and Conversion—A Review. BMC Chem. Eng. 2020, 2, 2. [Google Scholar] [CrossRef]
- Abed, K.A.; Gad, M.S.; El Morsi, A.K.; Sayed, M.M.; Elyazeed, S.A. Effect of Biodiesel Fuels on Diesel Engine Emissions. Egypt. J. Pet. 2019, 28, 183–188. [Google Scholar] [CrossRef]
- Hoang, A.T.; Foley, A.M.; Nižetić, S.; Huang, Z.; Ong, H.C.; Ölçer, A.I.; Pham, V.V.; Nguyen, X.P. Energy-Related Approach for Reduction of CO2 Emissions: A Critical Strategy on the Port-to-Ship Pathway. J. Clean. Prod. 2022, 355, 131772. [Google Scholar] [CrossRef]
- Dong, K.; Dong, X.; Jiang, Q.; Zhao, J. Assessing Energy Resilience and Its Greenhouse Effect: A Global Perspective. Energy Econ. 2021, 104, 105659. [Google Scholar] [CrossRef]
- Zhai, P.; Zhou, B.; Chen, Y. A Review of Climate Change Attribution Studies. J. Meteorol. Res. 2018, 32, 671–692. [Google Scholar] [CrossRef]
- Du, Z.-H.; Wang, L.; Wei, Z.-Q.; Liu, J.-F.; Lin, P.-L.; Lin, J.-H.; Li, Y.-Z.; Jin, Z.-Z.; Chen, J.-Z.; Wang, X.-X.; et al. CH4 and CO2 Observations from a Melting High Mountain Glacier, Laohugou Glacier No. 12. Adv. Clim. Chang. Res. 2022, 13, 146–155. [Google Scholar] [CrossRef]
- International Energy Agency. Renewables 2022; IEA: Paris, France, 2022; Available online: https://www.iea.org/reports/renewables-2022 (accessed on 19 November 2023).
- Naylor, R.L.; Higgins, M.M. The Political Economy of Biodiesel in an Era of Low Oil Prices. Renew. Sustain. Energy Rev. 2017, 77, 695–705. [Google Scholar] [CrossRef]
- Gupta, P.K.; Kumar, V.; Maity, S. Renewable Fuels from Different Carbonaceous Feedstocks: A Sustainable Route through Fischer–Tropsch Synthesis. J. Chem. Technol. Biotechnol. 2021, 96, 853–868. [Google Scholar] [CrossRef]
- Specchia, S. Fuel Processing Activities at European Level: A Panoramic Overview. Int. J. Hydrogen Energy 2014, 39, 17953–17968. [Google Scholar] [CrossRef]
- Martinez-Villarreal, S.; Kammoun, M.; Richel, A. The Critical Role of Hydrogen in the Development of New Biofuels. Curr. Opin. Green Sustain. Chem. 2023, 39, 100716. [Google Scholar] [CrossRef]
- Abdin, Z.; Zafaranloo, A.; Rafiee, A.; Mérida, W.; Lipiński, W.; Khalilpour, K.R. Hydrogen as an Energy Vector. Renew. Sustain. Energy Rev. 2020, 120, 109620. [Google Scholar] [CrossRef]
- Rodriguez Correa, C.; Kruse, A. Supercritical Water Gasification of Biomass for Hydrogen Production—Review. J. Supercrit. Fluids 2018, 133, 573–590. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, X.; Ding, L. Partial Oxidation of Phenol in Supercritical Water with NaOH and H2O2: Hydrogen Production and Polymer Formation. Sci. Total Environ. 2020, 722, 137985. [Google Scholar] [CrossRef]
- Pipitone, G.; Zoppi, G.; Pirone, R.; Bensaid, S. Sustainable Aviation Fuel Production Using In-Situ Hydrogen Supply via Aqueous Phase Reforming: A Techno-Economic and Life-Cycle Greenhouse Gas Emissions Assessment. J. Clean. Prod. 2023, 418, 138141. [Google Scholar] [CrossRef]
- Mishra, K.; Singh Siwal, S.; Kumar Saini, A.; Thakur, V.K. Recent Update on Gasification and Pyrolysis Processes of Lignocellulosic and Algal Biomass for Hydrogen Production. Fuel 2023, 332, 126169. [Google Scholar] [CrossRef]
- Lepage, T.; Kammoun, M.; Schmetz, Q.; Richel, A. Biomass-to-Hydrogen: A Review of Main Routes Production, Processes Evaluation and Techno-Economical Assessment. Biomass Bioenergy 2021, 144, 105920. [Google Scholar] [CrossRef]
- Ferreira-pinto, L.; Prado, M.; Parizi, S.; Fatima, A.; Cardozo, P.; De Arau, C.; Cardozo-Filho, L. ScienceDirect Experimental Basic Factors in the Production of H2 via Supercritical Water Gasification. Int. J. Hydrogen Energy 2019, 44, 25365–25383. [Google Scholar] [CrossRef]
- Reddy, S.N.; Nanda, S.; Dalai, A.K.; Kozinski, J.A. Supercritical Water Gasification of Biomass for Hydrogen Production. Int. J. Hydrogen Energy 2014, 39, 6912–6926. [Google Scholar] [CrossRef]
- Karimi, A.; Kazemi, N.; Tavakoli, O.; Pirbazari, A.E. Catalytic Supercritical Water Gasification of Black Liquor along with Lignocellulosic Biomass. Int. J. Hydrogen Energy 2022, 47, 16729–16740. [Google Scholar] [CrossRef]
- de Souza, G.B.M.; Pereira, M.B.; Mourão, L.C.; dos Santos, M.P.; de Oliveira, J.A.; Garde, I.A.A.; Alonso, C.G.; Jegatheesan, V.; Cardozo-Filho, L. Supercritical Water Technology: An Emerging Treatment Process for Contaminated Wastewaters and Sludge. Rev. Environ. Sci. Biotechnol. 2022, 21, 75–104. [Google Scholar] [CrossRef]
- Mascarell, J.J.; Abelleira-Pereira, J.M.; García-Jarana, B.; Portela, J.R. Hydrothermal Techniques for Biofuels Production. In Advances in Biofuels Production, Optimization and Applications; Elsevier: Amsterdam, The Netherlands, 2024; pp. 127–144. [Google Scholar]
- García-Jarana, M.B.; Portela, J.R.; Sánchez-Oneto, J.; Martinez de la Ossa, E.J.; Al-Duri, B. Analysis of the Supercritical Water Gasification of Cellulose in a Continuous System Using Short Residence Times. Appl. Sci. 2020, 10, 5185. [Google Scholar] [CrossRef]
- Zoppi, G.; Pipitone, G.; Pirone, R.; Bensaid, S. Aqueous Phase Reforming Process for the Valorization of Wastewater Streams: Application to Different Industrial Scenarios. Catal. Today 2022, 387, 224–236. [Google Scholar] [CrossRef]
- Rocha, D.H.D.; de Souza, T.A.Z.; Coronado, C.J.R.; Silveira, J.L.; Silva, R.J. Exergoenvironmental Analysis of Hydrogen Production through Glycerol Steam Reforming. Int. J. Hydrogen Energy 2021, 46, 1385–1402. [Google Scholar] [CrossRef]
- Li, S.; Savage, P.E.; Guo, L. Stability and Activity Maintenance of Al2O3-and Carbon Nanotube-Supported Ni Catalysts during Continuous Gasification of Glycerol in Supercritical Water. J. Supercrit. Fluids 2018, 135, 188–197. [Google Scholar] [CrossRef]
- Zhu, B.; Li, S.; Wang, W.; Zhang, H. Supercritical Water Synthesized Ni/ZrO2 Catalyst for Hydrogen Production from Supercritical Water Gasification of Glycerol. Int. J. Hydrogen Energy 2019, 44, 30917–30926. [Google Scholar] [CrossRef]
- Bac, S.; Keskin, S.; Avci, A.K. Recent Advances in Sustainable Syngas Production by Catalytic CO2 Reforming of Ethanol and Glycerol. Sustain. Energy Fuels 2020, 4, 1029–1047. [Google Scholar] [CrossRef]
- Mourão, L.C.; Meireles de Souza, G.B.; Dias, I.M.; Oliveira, S.B.; Leandro de Souza, T.; Alonso, C.G. Hydrogen Production via Supercritical Water Gasification of Glycerol Enhanced by Simple Structured Catalysts. Int. J. Hydrogen Energy 2023, 48, 27929–27943. [Google Scholar] [CrossRef]
- Ribeiro, T.S.S.; Mourão, L.C.; Souza, G.B.M.; Dias, I.M.; Andrade, L.A.; Souza, P.L.M.; Cardozo-Filho, L.; Oliveira, G.R.; Oliveira, S.B.; Alonso, C.G. Treatment of Hormones in Wastewater from the Pharmaceutical Industry by Continuous Flow Supercritical Water Technology. J. Environ. Chem. Eng. 2021, 9, 106095. [Google Scholar] [CrossRef]
- Dias, I.M.; Mourão, L.C.; Andrade, L.A.; Souza, G.B.M.; Viana, J.C.V.; Oliveira, S.B.; Alonso, C.G. Degradation of Antibiotic Amoxicillin from Pharmaceutical Industry Wastewater into a Continuous Flow Reactor Using Supercritical Water Gasification. Water Res. 2023, 234, 119826. [Google Scholar] [CrossRef] [PubMed]
- Baird, R.B.; Eaton, A.D.; Rice, E.W. Standard Methods for the Examination of Water and Wasterwater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2017; ISBN 087553287X. [Google Scholar]
- Khuri, A.I.; Mukhopadhyay, S. Response Surface Methodology. WIREs Comput. Stat. 2010, 2, 128–149. [Google Scholar] [CrossRef]
- Yu, S.; Bai, J.; Xie, Q.; Liang, X.; Nie, Y. Efficient Treatment of Biodiesel Wastewater by Catalytic Combustion. J. Water Process Eng. 2021, 43, 102207. [Google Scholar] [CrossRef]
- Mainil, R.I.; Matsumura, Y. New Application of Supercritical Water Gasification to Palm Oil Mill Effluent: Gasification and Phosphorus Recovery. Energy Fuels 2019, 33, 11145–11152. [Google Scholar] [CrossRef]
- Guo, S.; Guo, L.; Cao, C.; Yin, J.; Lu, Y.; Zhang, X. Hydrogen Production from Glycerol by Supercritical Water Gasification in a Continuous Flow Tubular Reactor. Int. J. Hydrogen Energy 2012, 37, 5559–5568. [Google Scholar] [CrossRef]
- CONAMA Conselho Nacional Do Meio Ambiente-Resolução No 436, de 13 de Maio de 2011. Available online: https://www.suape.pe.gov.br/images/publicacoes/CONAMA_n.430.2011.pdf (accessed on 7 March 2022).
- CODEGO Companhia de Desenvolvimento Econômico de Goiás-Regulamento Dos Serviços de Água e Esgoto. Available online: http://www.codego.com.br/docs/REGULAMENTO_AGUA_ESGOTO_2016.pdf (accessed on 7 March 2022).
- USEPA United States Environmental Protection Agency: The Centralized Waste Treatment Effluent Guidelines and Standards-Part 437. Available online: https://www.epa.gov/eg/centralized-waste-treatment-effluent-guidelines (accessed on 7 March 2022).
- Bermejo, M.D.; Cocero, M.J. Supercritical Water Oxidation: A Technical Review. AIChE J. 2006, 52, 3933–3951. [Google Scholar] [CrossRef]


| Independent Variables | Levels | |||||
|---|---|---|---|---|---|---|
| −1.41 | −1.00 | 0 | 1.00 | 1.41 | ||
| Feed flow rate (mL/min), X1 | 10 | 12.2 | 17.5 | 28.8 | 25 | |
| Temperature (°C), X2 | 500 | 529 | 600 | 670 | 700 | |
| Run | Feed flow rate (mL/min) | Temperature (°C) | X1 | X2 | ||
| 1 | 12.2 | 529 | −1.00 | −1.00 | ||
| 2 | 12.2 | 670 | −1.00 | 1.00 | ||
| 3 | 22.9 | 529 | 1.00 | −1.00 | ||
| 4 | 22.9 | 670 | 1.00 | 1.00 | ||
| 5 | 10.0 | 600 | −1.41 | 0.00 | ||
| 6 | 25.0 | 600 | 1.41 | 0.00 | ||
| 7 | 17.5 | 500 | 0.00 | −1.41 | ||
| 8 | 17.5 | 700 | 0.00 | 1.41 | ||
| 9 | 17.5 | 600 | 0.00 | 0.00 | ||
| 10 | 17.5 | 600 | 0.00 | 0.00 | ||
| 11 | 17.5 | 600 | 0.00 | 0.00 | ||
| 12 | 17.5 | 600 | 0.00 | 0.00 | ||
| Run | T (°C) | F (mL/min) | Molar Fraction (% mol/mol) | Total Gaseous Flow Rate (mL/min) | |||||
|---|---|---|---|---|---|---|---|---|---|
| H2 | CH4 | CO | CO2 | C2H4 | C2H6 | ||||
| 1 | 529 | 12.3 | 65.89 | 0.88 | 10.76 | 22.24 | 0.13 | 0.11 | 818 |
| 2 | 670 | 12.3 | 71.29 | 1.83 | 4.33 | 22.09 | 0.03 | 0.44 | 3332 |
| 3 | 529 | 22.9 | 62.71 | 0.90 | 13.77 | 22.39 | 0.13 | 0.08 | 282 |
| 4 | 670 | 22.9 | 71.95 | 1.48 | 4.74 | 21.47 | 0.08 | 0.28 | 4927 |
| 5 | 600 | 10.0 | 68.59 | 2.09 | 7.42 | 21.32 | 0.15 | 0.42 | 1271 |
| 6 | 600 | 25.0 | 69.60 | 1.42 | 9.16 | 19.47 | 0.21 | 0.15 | 1576 |
| 7 | 500 | 17.5 | 66.46 | 0.61 | 12.36 | 20.45 | 0.08 | 0.04 | 182 |
| 8 | 700 | 17.5 | 73.86 | 1.82 | 4.54 | 19.33 | 0.06 | 0.39 | 5541 |
| 9 | 600 | 17.5 | 68.80 | 1.81 | 9.94 | 18.96 | 0.22 | 0.26 | 1260 |
| 10 | 600 | 17.5 | 72.84 | 1.42 | 4.92 | 20.47 | 0.17 | 0.19 | 1373 |
| 11 | 600 | 17.5 | 66.28 | 1.62 | 9.60 | 22.06 | 0.21 | 0.23 | 1514 |
| 12 | 600 | 17.5 | 71.44 | 1.33 | 4.87 | 22.04 | 0.15 | 0.16 | 1639 |
| Run | Feed Flow Rate (mL/min) | Temperature (°C) | Volumetric Flow Rate (mL/min) | Molar Flow Rate (mol/min) | Mass Flow Rate (g/min) |
|---|---|---|---|---|---|
| 1 | 12.3 | 529 | 538.9 | 22.0 | 0.044 |
| 2 | 12.3 | 671 | 2375.6 | 97.1 | 0.194 |
| 3 | 22.9 | 529 | 176.9 | 7.2 | 0.014 |
| 4 | 22.9 | 671 | 3544.5 | 144.8 | 0.290 |
| 5 | 10.0 | 600 | 871.7 | 35.6 | 0.071 |
| 6 | 25.0 | 600 | 1097.0 | 44.8 | 0.090 |
| 7 | 17.5 | 500 | 121.1 | 4.9 | 0.010 |
| 8 | 17.5 | 700 | 4092.6 | 167.2 | 0.335 |
| 9 | 17.5 | 600 | 866.6 | 35.4 | 0.071 |
| 10 | 17.5 | 600 | 1000.2 | 40.8 | 0.082 |
| 11 | 17.5 | 600 | 1003.5 | 41.0 | 0.082 |
| 12 | 17.5 | 600 | 1171.0 | 47.8 | 0.096 |
| Factor | Df | Sum Sq | Mean Sq | F Valor | p-Value |
|---|---|---|---|---|---|
| Feed flow rate (L) | 1 | 159,565 | 159,565 | 10.2675 | 0.049170 |
| Temperature (L) | 1 | 14,708,961 | 14,708,961 | 946.4733 | 0.000075 |
| Temperature (Q) | 1 | 2,191,096 | 2,191,096 | 140.9898 | 0.001284 |
| Two-way interaction | 1 | 582,685 | 582,685 | 37.4938 | 0.008756 |
| Lack-of-fit | 4 | 80,307 | 20,077 | 1.2919 | 0.433878 |
| Pure Error | 3 | 46,627 | 15,541 | ||
| Total SS | 11 | 17,698,445 |
| Run | Feed Flow Rate (mL/min) | Temperature (°C) | Carbon Mass Flow Rate (g/min) | ||||
|---|---|---|---|---|---|---|---|
| Inlet total (Liquid Phase) | Outlet (Liquid Phase) | Outlet (Gaseous Phase) | Outlet Total | Error (%) | |||
| 1 | 12.3 | 529 | 0.592 | 0.479 | 0.129 | 0.608 | 2.7 |
| 2 | 12.3 | 671 | 0.592 | 0.103 | 0.477 | 0.580 | −2.0 |
| 3 | 22.9 | 529 | 1.104 | 1.011 | 0.099 | 1.109 | 0.5 |
| 4 | 22.9 | 671 | 1.104 | 0.471 | 0.687 | 1.158 | 4.9 |
| 5 | 10.0 | 600 | 0.485 | 0.252 | 0.209 | 0.460 | −5.1 |
| 6 | 25.0 | 600 | 1.209 | 0.997 | 0.238 | 1.215 | 0.5 |
| 7 | 17.5 | 500 | 0.848 | 0.809 | 0.162 | 0.970 | 14.5 |
| 8 | 17.5 | 700 | 0.848 | 0.220 | 0.723 | 0.942 | 11.2 |
| 9 | 17.5 | 600 | 0.848 | 0.617 | 0.161 | 0.778 | −8.2 |
| 10 | 17.5 | 600 | 0.848 | 0.611 | 0.241 | 0.852 | 0.5 |
| 11 | 17.5 | 600 | 0.848 | 0.644 | 0.171 | 0.815 | −3.8 |
| 12 | 17.5 | 600 | 0.848 | 0.630 | 0.168 | 0.798 | −5.8 |
| Parameter | Sample (mg/L) | (2) Uncertainty | Legislations | ||||
|---|---|---|---|---|---|---|---|
| Untreated | (1) Treated | (3) CONAMA [37] | (4) CODEGO [38] | (5) USEPA [39] | |||
| pH | 5.37 | 3.56 | 0.011 | 5–9 | 6–9 | 6–9 | ✗ |
| Nitrate | 1.10 | 0.20 | 0.030 | - | - | - | ✓ |
| Nitrite | 0.41 | 0.33 | 0.004 | - | - | - | ✓ |
| TOC | 48,250.00 | 20,615.00 | - | Rem. > 60% | Rem. > 60% | - | ✓ |
| COD | 339,708.00 | 89,919.00 | 0.060 | - | 1000.0 | - | ✗ |
| BOD | 141,724.00 | 30,959.50 | 0.145 | Rem. > 60% | 500.0 | 53.0 | ✗ |
| Oils and greases | 97.63 | 0.30 | 0.030 | 20.0 | 20.0 | 38.0 | ✓ |
| Sulfate | 4.4 | < | 0.010 | - | 250.0 | - | ✓ |
| Aluminum (Al) | 0.1079 | 0.1459 | 0.0023 | - | - | ✓ | |
| Barium (Ba) | 0.03666 | 0.0161 | 0.002 | 0.5 | 0.5 | 0.1040 | ✓ |
| Beryllium (Be) | 0.00555 | < | 0.001 | - | - | ✓ | |
| Calcium (Ca) | 3.5385 | 1.8652 | 0.003 | - | - | ✓ | |
| Cobalt (Co) | 0.0048 | 0.6962 | 0.004 | ✓ | |||
| Copper (Cu) | 0.0499 | 0.1213 | 0.0007 | 1.0 | 1.0 | 0.2420 | ✓ |
| Iron (Fe) | 0.5264 | 0.3501 | 0.003 | 15.0 | 15.0 | ✓ | |
| Potassium (K) | 3.2208 | 4.3826 | 0.004 | - | - | ✓ | |
| Magnesium (Mg) | 0.8501 | 0.2616 | 0.001 | - | - | ✓ | |
| Manganese (Mn) | 0.0528 | 0.9177 | 0.003 | 1.0 | 1.0 | ✓ | |
| Molybdenum (Mo) | 0.0082 | 0.1172 | 0.003 | - | - | ✓ | |
| Sodium (Na) | 11.0503 | 10.1663 | 0.005 | - | - | ✓ | |
| Nickel (Ni) | 0.0026 | 0.2877 | 0.004 | 2.0 | 2.0 | 1.4500 | ✓ |
| Tin (Sn) | 0.5883 | 0.0064 | 0.004 | ✓ | |||
| Zinc (Zn) | 0.0010 | 0.0290 | 0.006 | 5.0 | 5.0 | 0.4200 | ✓ |
| Case Study | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|---|---|---|---|---|---|---|---|---|
| Dilution | 10:1 | 10:1 | 10:1 | 10:1 | 1:1 | 1:1 | 1:1 | 1:1 |
| Splitting ratio to turbine | 0.0 | 0.0 | 0.5 | 0.5 | 0.0 | 0.0 | 0.5 | 0.5 |
| E-100 duty (kW) | 0.0 | 8100.0 | 0.0 | 4100.0 | 0.0 | 1100.0 | 0.0 | 500.0 |
| E-101 duty (kW) | 0.0 | 0.0 | 0.0 | 1500.0 | 0.0 | 0.0 | 0.0 | 300.0 |
| E-102 duty (kW) | 0.0 | 364.7 | 0.0 | 364.7 | 0.0 | 49.5 | 0.0 | 49.5 |
| H2 in reactor outlet (kmol/h) | 31.32 | 31.32 | 31.32 | 31.32 | 4.856 | 4.856 | 4.856 | 4.856 |
| H2 mole fraction in reactor outlet | 0.0507 | 0.0507 | 0.0507 | 0.0507 | 0.0490 | 0.0490 | 0.0490 | 0.0490 |
| H2 in F-101 vapor (kmol/h) | 30.98 | 30.98 | 30.98 | 30.98 | 4.849 | 4.849 | 4.849 | 4.849 |
| H2 mole fraction in F-101 vapor | 0.6475 | 0.6475 | 0.6475 | 0.6475 | 0.1232 | 0.1232 | 0.1232 | 0.1232 |
| H-100 duty (kW) | 10,733.2 | 2633.2 | 10,733.2 | 5133.2 | 1623.3 | 523.3 | 1623.3 | 823.3 |
| H-101 duty (kW) | 364.7 | 0.0 | 364.7 | 0.0 | 49.5 | 0.0 | 48.5 | 0.0 |
| C-100 duty (kW) | −9749.2 | −1649.24 | −8834.3 | −3234.3 | −1241.6 | −141.6 | −1084.3 | −234.0 |
| T-100 work (kW) | 0.0 | 0.0 | −914.9 | −914.9 | 0.0 | 0.0 | −157.3 | −157.3 |
| Case Study | 4.1 | 4.2 | 4.3 |
|---|---|---|---|
| F-101 pressure (MPa) | 1.0 | 3.0 | 5.0 |
| Distillate rate (kmol/h) | 31.01 | 30.20 | 29.31 |
| Feed H2 mole flow (kmol/h) | 30.98 | 30.17 | 29.29 |
| Feed H2 composition | 0.6475 | 0.8273 | 0.8805 |
| Dist. H2 mole flow (kmol/h) | 30.98 | 30.17 | 29.29 |
| Dist. H2 composition | 0.999 | 0.999 | 0.999 |
| T-100 work (kW) | −914.9 | −738.2 | −590.2 |
| C-101 duty (kW) | −188.3 | −110.6 | −91.8 |
| Condenser duty (kW) | −110.1 | −93.6 | −96.2 |
| Reboiler duty (kW) | 136.0 | 105.3 | 96.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teixeira, I.R.; Dias, I.M.; Mourão, L.C.; Andrade, L.A.; Pavão, L.V.; Abelleira-Pereira, J.M.; Souza, G.B.M.; Cardozo-Filho, L.; Alonso, C.G.; Guirardello, R. Continuous Hydrogen Production via Hydrothermal Gasification of Biodiesel Industry Wastewater: Experimental Optimization and Energy Integration Simulation. Water 2023, 15, 4062. https://doi.org/10.3390/w15234062
Teixeira IR, Dias IM, Mourão LC, Andrade LA, Pavão LV, Abelleira-Pereira JM, Souza GBM, Cardozo-Filho L, Alonso CG, Guirardello R. Continuous Hydrogen Production via Hydrothermal Gasification of Biodiesel Industry Wastewater: Experimental Optimization and Energy Integration Simulation. Water. 2023; 15(23):4062. https://doi.org/10.3390/w15234062
Chicago/Turabian StyleTeixeira, Isabela R., Isabela M. Dias, Lucas C. Mourão, Laiane A. Andrade, Leandro V. Pavão, Jose M. Abelleira-Pereira, Guilherme B. M. Souza, Lucio Cardozo-Filho, Christian G. Alonso, and Reginaldo Guirardello. 2023. "Continuous Hydrogen Production via Hydrothermal Gasification of Biodiesel Industry Wastewater: Experimental Optimization and Energy Integration Simulation" Water 15, no. 23: 4062. https://doi.org/10.3390/w15234062
APA StyleTeixeira, I. R., Dias, I. M., Mourão, L. C., Andrade, L. A., Pavão, L. V., Abelleira-Pereira, J. M., Souza, G. B. M., Cardozo-Filho, L., Alonso, C. G., & Guirardello, R. (2023). Continuous Hydrogen Production via Hydrothermal Gasification of Biodiesel Industry Wastewater: Experimental Optimization and Energy Integration Simulation. Water, 15(23), 4062. https://doi.org/10.3390/w15234062








