Short-Term Effects of Drying-Rewetting and Long-Term Effects of Nutrient Loading on Periphyton N:P Stoichiometry
Abstract
:1. Introduction
2. Materials and Methods
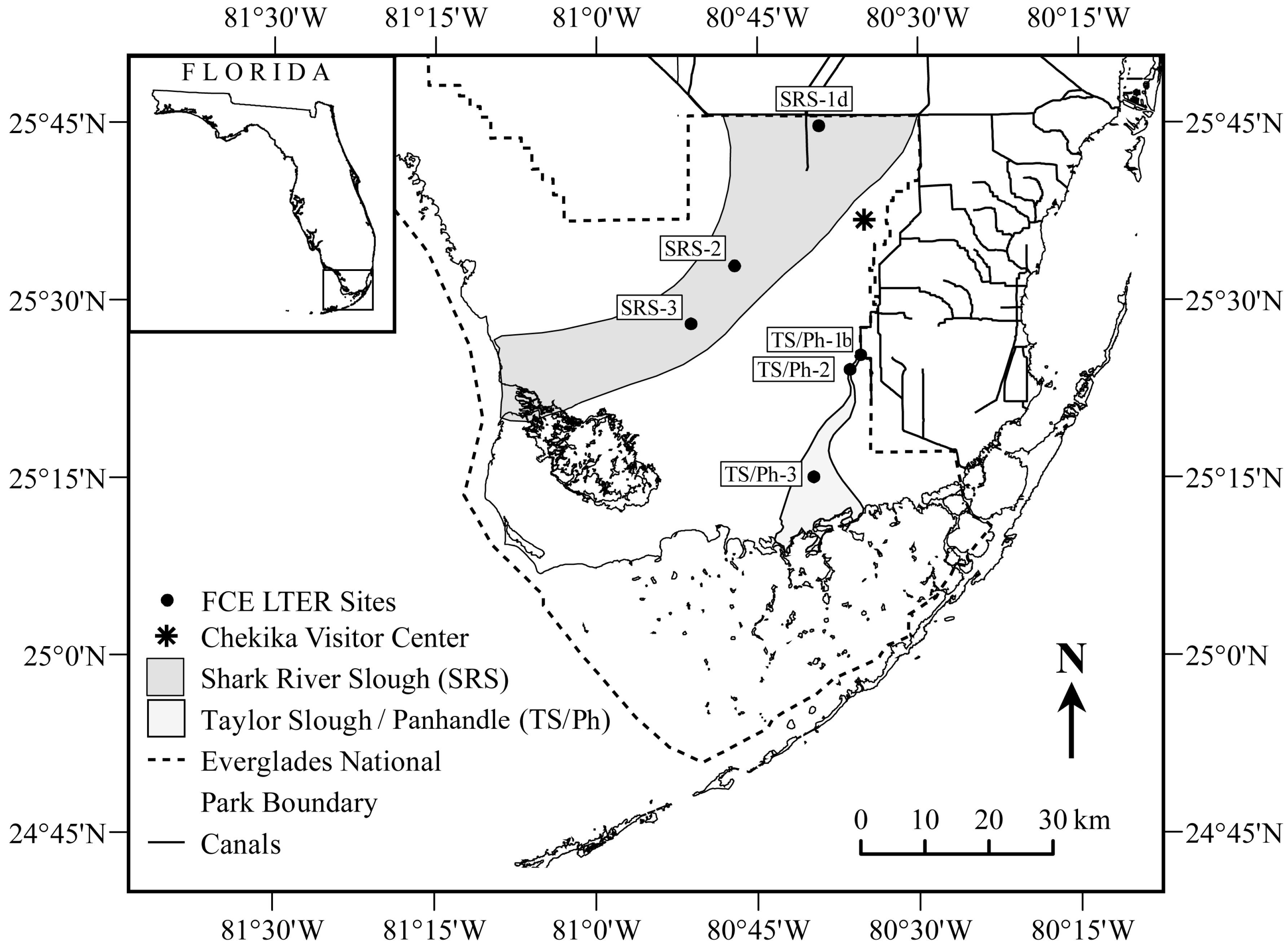
2.1. Study Region
2.2. Experiment on the Effects of Dry-Wet Cycles on Mat Nutrient Standing Stocks
2.2.1. Periphyton Mat Collection
2.2.2. Drying/Rewetting Manipulations
2.2.3. Periphyton Mat Sampling and Processing
2.2.4. Periphyton Mat Nutrient Standing Stocks
2.3. Long-Term Nutrient Standing Stock Dynamics
2.4. Data Analyses
3. Results
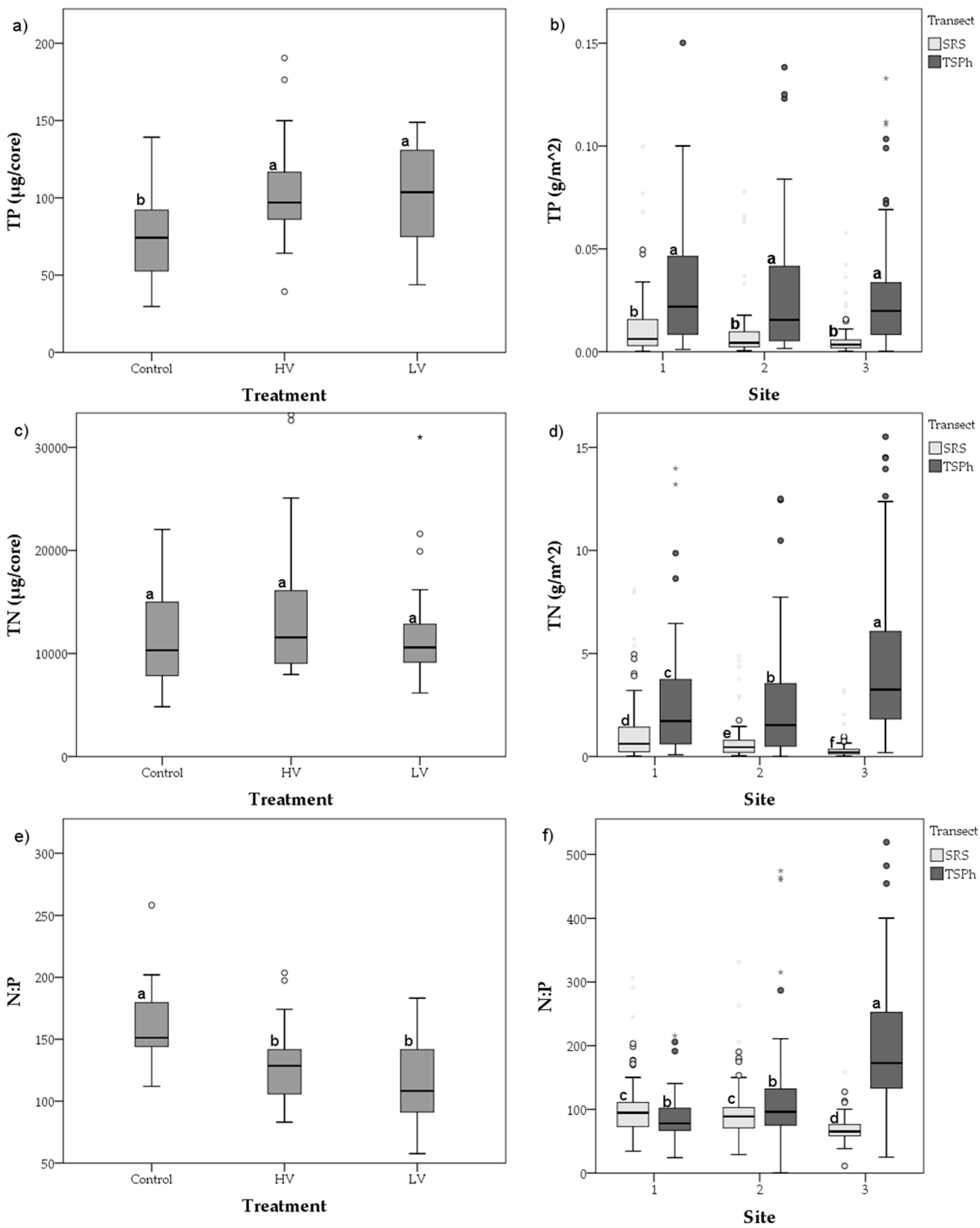
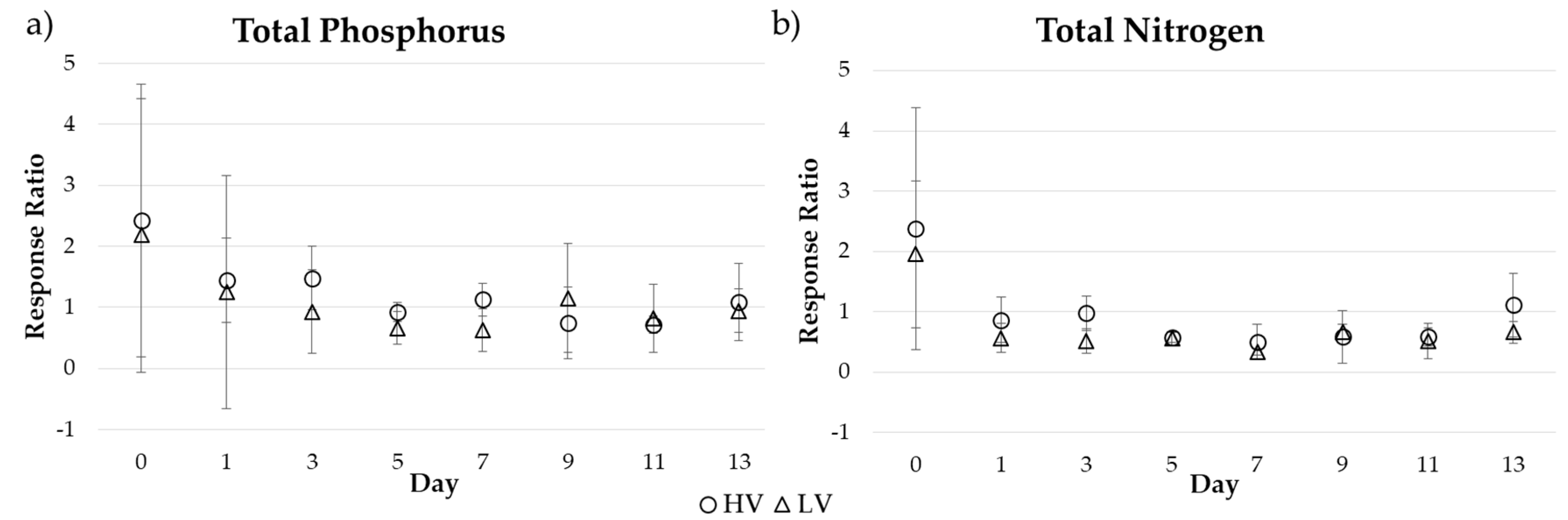
3.1. Short-Term Drying-Rewetting Microcosm
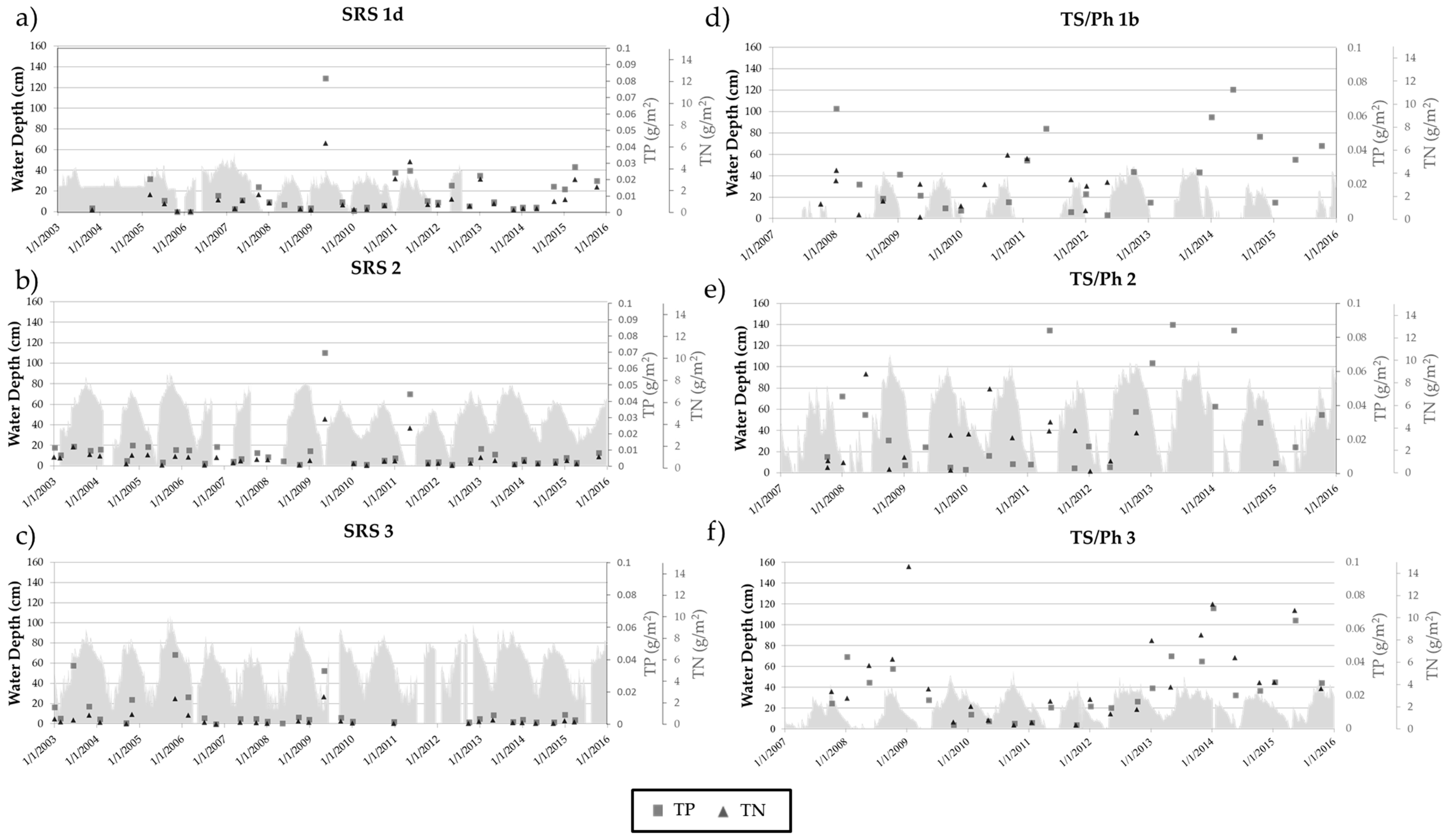
3.2. Long-Term Nutrient Standing Stock Dynamics
4. Discussion
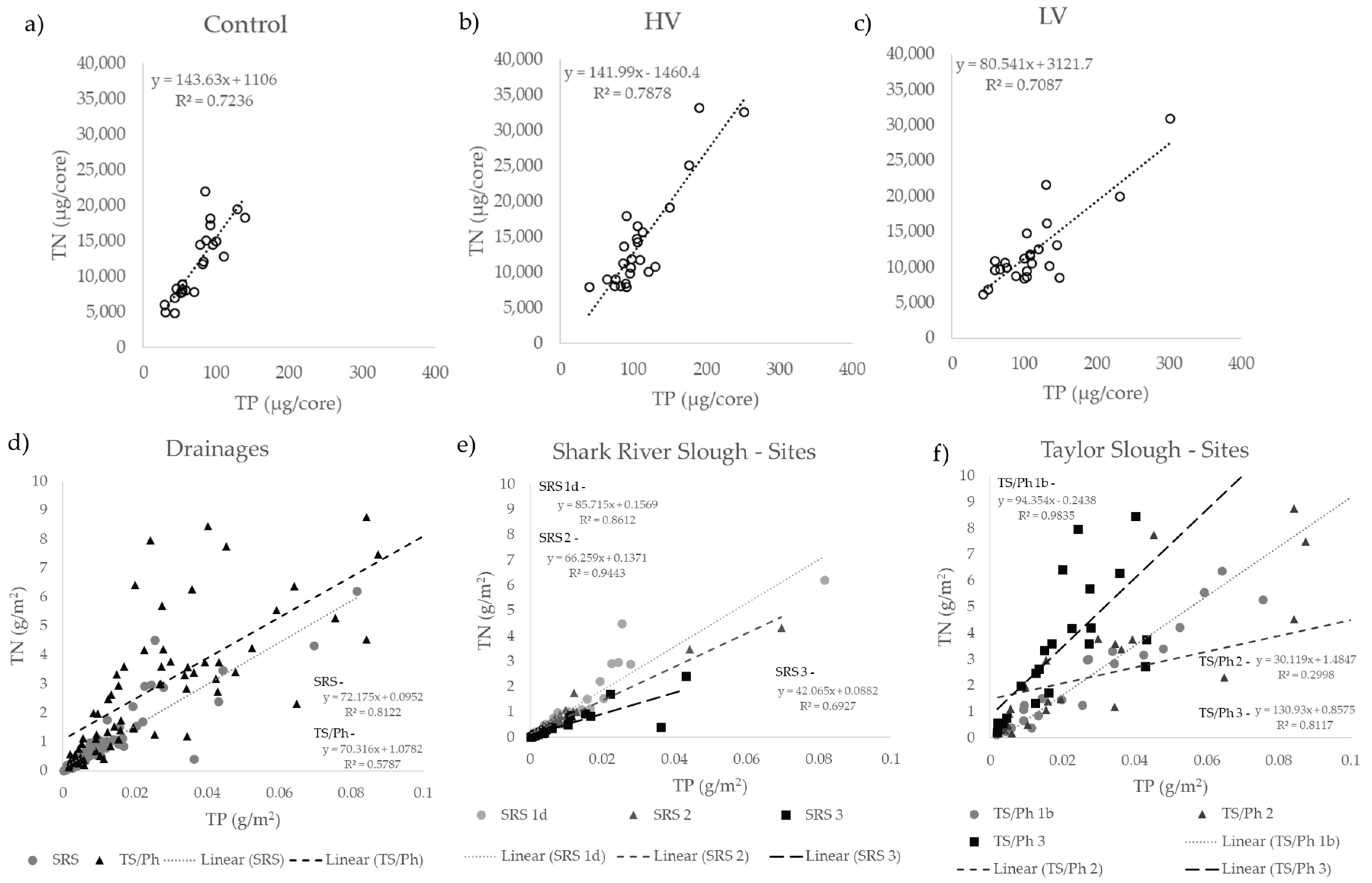
4.1. Short-Term Drying-Rewetting Effects on Periphyton Nutrient Dynamics
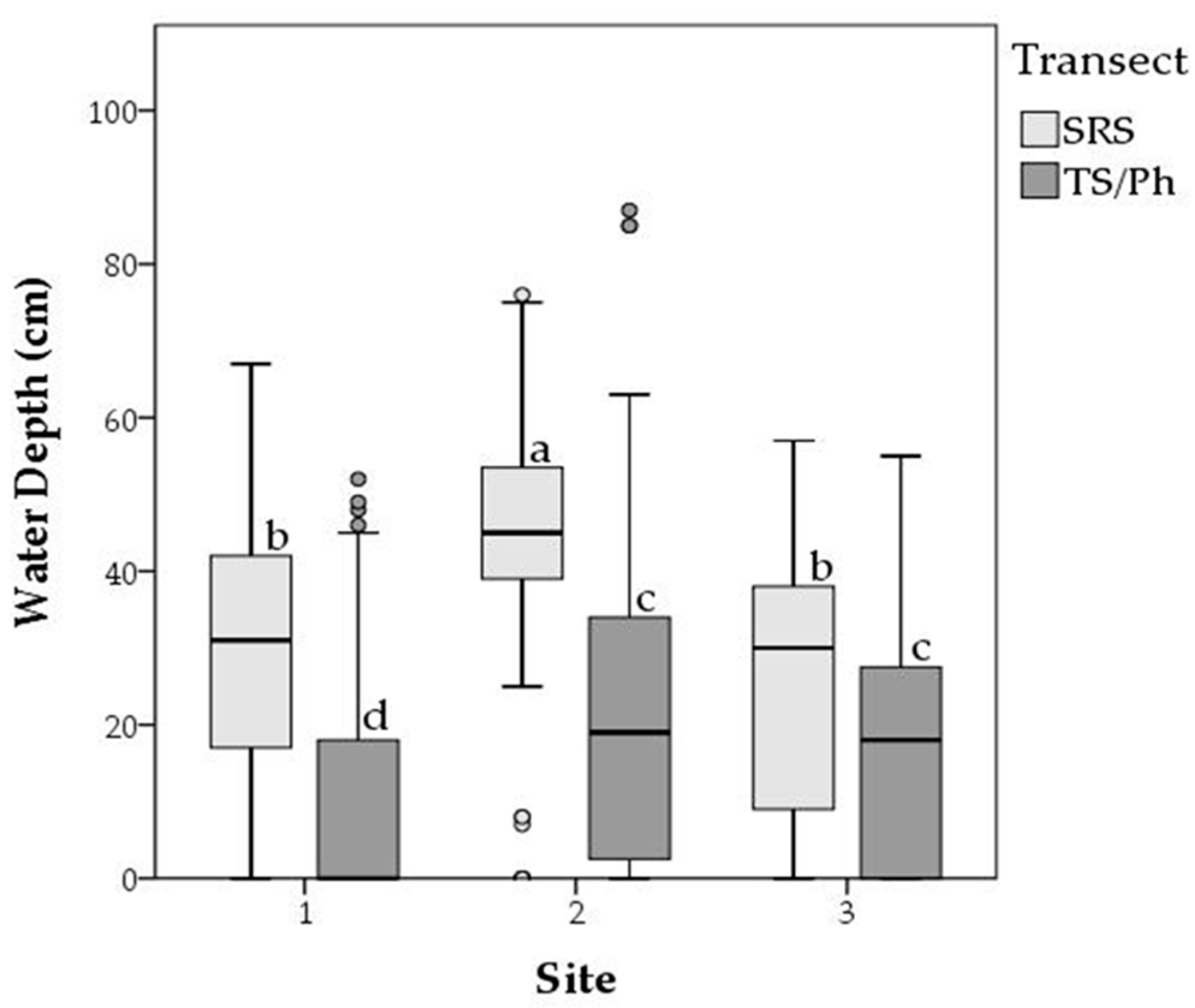
4.2. Long-Term Periphyton Nutrient Dynamics across Space
4.3. Long-Term Periphyton Nutrient Dynamics across Time
4.4. Short- and Long-Term Drivers of Periphyton Nutrient Retention
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
Appendix B
References
- Sterner, R.W.; Andersen, T.; Elser, J.J.; Hessen, D.O.; Hood, J.M.; McCauley, E.; Urabe, J. Scale-dependent carbon: Nitrogen: Phosphorus seston stoichiometry in marine and freshwaters. Limnol. Oceanogr. 2008, 53, 1169–1180. [Google Scholar] [CrossRef]
- Howarth, R.; Paerl, H.W. Coastal marine eutrophication: Control of both nitrogen and phosphorus is necessary. Proc. Natl. Acad. Sci. USA 2008, 105, E103. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.S.; Driscoll, C.T.; Jordan, T.E.; Reay, W.G.; Boynton, W.R. Sources of nitrogen to estuaries in the United States. Estuaries 2003, 26, 803–814. [Google Scholar] [CrossRef]
- Daniel, T.C.; Sharpley, A.N.; Lemunyon, J.L. Agricultural phosphorus and eutrophication: A symposium overview. J. Environ. Qual. 1998, 27, 251–257. [Google Scholar] [CrossRef]
- Davis, S.M. Phosphorus inputs and vegetation sensitivity in the Everglades. In Everglades: The Ecosystem and Its Restoration; CRC Press: Boca Raton, FL, USA, 1994; pp. 357–378. [Google Scholar]
- Brown, P.; Wright, A.L. The Role of Periphyton in the Everglades; SL 3101-3; Soil and Water Science Department, University of Florida: Gainesville, FL, USA, 2009. [Google Scholar]
- Schade-Poole, K.; Möller, G. Impact and mitigation of nutrient pollution and overland water flow change on the Florida Everglades, USA. Sustainability 2016, 8, 940. [Google Scholar] [CrossRef]
- Hessen, D.O.; Elser, J.J.; Sterner, R.W.; Urabe, J. Ecological stoichiometry: An elementary approach using basic principles. Limnol. Oceanogr. 2013, 58, 2219–2236. [Google Scholar] [CrossRef]
- Elser, J.J.; Bracken, M.E.; Cleland, E.E.; Gruner, D.S.; Harpole, W.S.; Hillebrand, H.; Ngai, J.T.; Seabloom, E.W.; Shurin, J.B.; Smith, J.E. Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol. Lett. 2007, 10, 1135–1142. [Google Scholar] [CrossRef] [PubMed]
- Drake, W.M.; Scott, J.T.; Evans-White, M.; Haggard, B.; Sharpley, A.; Rogers, C.W.; Grantz, E.M. The effect of periphyton stoichiometry and light on biological phosphorus immobilization and release in streams. Limnology 2012, 13, 97–106. [Google Scholar] [CrossRef]
- McCormick, P.V.; Laing, J.A. Effects of increased phosphorus loading on dissolved oxygen in a subtropical wetland, the Florida Everglades. Wetl. Ecol. Manag. 2003, 11, 199–216. [Google Scholar] [CrossRef]
- Schindler, D.W.; Hecky, R.E.; Findlay, D.L.; Stainton, M.P.; Parker, B.R.; Paterson, M.J.; Beaty, K.G.; Lyng, M.; Kasian, S.E.M. Eutrophication of lakes cannot be controlled by reducing nitrogen input: Results of a 37-year whole-ecosystem experiment. Proc. Natl. Acad. Sci. USA 2008, 105, 11254–11258. [Google Scholar] [CrossRef] [PubMed]
- Dodds, W.K.; Smith, V.H. Nitrogen, phosphorus, and eutrophication in streams. Inland Waters 2016, 6, 155–164. [Google Scholar] [CrossRef]
- McCormick, P.V.; Cairns, J. Algae as indicators of environmental change. J. Appl. Phycol. 1994, 6, 509–526. [Google Scholar] [CrossRef]
- McCormick, P.V.; Stevenson, R.J. Periphyton as a tool for ecological assessment and management in the Florida Everglades. J. Phycol. 1998, 34, 726–733. [Google Scholar] [CrossRef]
- Hagerthey, S.E.; Bellinger, B.J.; Wheeler, K.; Gantar, M.; Gaiser, E. Everglades periphyton: A biogeochemical perspective. Crit. Rev. Environ. Sci. Technol. 2011, 41, 309–343. [Google Scholar] [CrossRef]
- Gaiser, E.E.; McCormick, P.V.; Hagerthey, S.E.; Gottlieb, A.D. Landscape patterns of periphyton in the Florida Everglades. Crit. Rev. Environ. Sci. Technol. 2011, 41, 92–120. [Google Scholar] [CrossRef]
- La Hée, J.M.; Gaiser, E.E. Benthic diatom assemblages as indicators of water quality in the Everglades and three tropical karstic wetlands. Freshw. Sci. 2012, 31, 205–221. [Google Scholar] [CrossRef]
- Cattaneo, A. Grazing on epiphytes. Limnol. Oceanogr. 1983, 28, 124–132. [Google Scholar] [CrossRef]
- Campeau, S.; Murkin, H.R.; Titman, R.D. Relative importance of algae and emergent plant litter to freshwater marsh. Can. J. Fish. Aquat. Sci. 1994, 51, 681–692. [Google Scholar] [CrossRef]
- Gaiser, E. Periphyton as an indicator of restoration in the Florida Everglades. Ecol. Indic. 2009, 9, S37–S45. [Google Scholar] [CrossRef]
- Wu, Y.; Liu, J.; Rene, E.R. Periphytic biofilms: A promising nutrient utilization regulator in wetlands. Bioresour. Technol. 2018, 248, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, D.L.; Craggs, R.J. Utilising periphytic algae as nutrient removal systems for the treatment of diffuse nutrient pollution in waterways. Algal Res. 2017, 25, 496–506. [Google Scholar] [CrossRef]
- Gottlieb, A.; Richards, J.; Gaiser, E. Comparative study of periphyton community structure in long and short-hydroperiod Everglades marshes. Hydrobiologia 2006, 569, 195–207. [Google Scholar] [CrossRef]
- Iwaniec, D.; Childers, D.; Rondeau, D.; Madden, C.; Saunders, C. Effects of hydrologic and water quality drivers on periphyton dynamics in the southern Everglades. Hydrobiologia 2006, 569, 223–235. [Google Scholar] [CrossRef]
- Hilton, J.; O’Hare, M.; Bowes, M.J.; Jones, J.I. How green is my river? A new paradigm of eutrophication in rivers. Sci. Total Environ. 2006, 365, 66–83. [Google Scholar] [CrossRef] [PubMed]
- McCormick, P.V.; Rawlik, P.S.; Lurding, K.; Smith, E.P.; Sklar, F.H. Periphyton-water quality relationships along a nutrient gradient in the northern Florida Everglades. J. N. Am. Benthol. Soc. 1996, 15, 433–449. [Google Scholar] [CrossRef]
- Childers, D.L.; Boyer, J.N.; Davis, S.E.; Madden, C.J.; Rudnick, D.T.; Sklar, F.H. Relating precipitation and water management to nutrient concentrations in the oligotrophic “upside-down” estuaries of the Florida Everglades. Limnol. Oceanogr. 2006, 51, 602–616. [Google Scholar] [CrossRef]
- Gaiser, E.E.; Childers, D.L.; Jones, R.D.; Richards, J.H.; Scinto, L.J.; Trexler, J.C. Periphyton responses to eutrophication in the Florida Everglades: Cross-system patterns of structural and compositional change. Limnol. Oceanogr. 2006, 51, 617–630. [Google Scholar] [CrossRef]
- Gottlieb, A.; Richards, J.; Gaiser, E. Effects of desiccation duration on the community structure and nutrient retention of short and long-hydroperiod Everglades periphyton mats. Aquat. Bot. 2005, 82, 99–112. [Google Scholar] [CrossRef]
- Marazzi, L.; Gaiser, E.E.; Tobias, F.A.C. Phosphorus scarcity and desiccation stress increase the occurrence of dominant taxa in wetland benthic primary producer communities. Aquat. Ecol. 2017, 51, 571–589. [Google Scholar] [CrossRef]
- Gaiser, E.E.; Trexler, J.C.; Richards, J.H.; Childers, D.L.; Lee, D.; Edwards, A.L.; Scinto, L.J.; Jayachandran, K.; Noe, G.B.; Jones, R.D. Cascading ecological effects of low-level phosphorus enrichment in the Florida Everglades. J. Environ. Qual. 2005, 34, 717–723. [Google Scholar] [CrossRef] [PubMed]
- Sand-Jensen, K.; Borum, J. Interactions among phytoplankton, periphyton, and macrophytes in temperate freshwaters and estuaries. Aquat. Bot. 1991, 41, 137–175. [Google Scholar] [CrossRef]
- Zhang, X.; Mei, X. Periphyton response to nitrogen and phosphorus enrichment in a eutrophic shallow aquatic ecosystem. Chin. J. Oceanol. Limnol. 2013, 31, 59–64. [Google Scholar] [CrossRef]
- Obeysekera, J.; Barnes, J.; Nungesser, M. Climate sensitivity runs and regional hydrologic modeling for predicting the response of the greater Florida Everglades ecosystem to climate change. Environ. Manag. 2015, 55, 749–762. [Google Scholar] [CrossRef] [PubMed]
- Bondar-Kunze, E.; Tritthart, M.; Hein, T. The influence of short term water level fluctuations and desiccation stress on periphyton development at a riparian zone of a large regulated river. Fundam. Appl. Limnol. 2015, 186, 283–296. [Google Scholar] [CrossRef]
- Scinto, L.J. Phosphorus Cycling in a Periphyton-Dominated Freshwater Wetland. Ph.D. Thesis, University of Florida, Gainesville, FL, USA, 1997. [Google Scholar]
- Inglett, P.W.; Reddy, K.R.; McCormick, P.V. Periphyton chemistry and nitrogenase activity in a northern Everglades ecosystem. Biogeochemistry 2004, 67, 213–233. [Google Scholar] [CrossRef]
- Liao, X.; Inglett, P.W. Dynamics of periphyton nitrogen fixation in short-hydroperiod wetlands revealed by high-resolution seasonal sampling. Hydrobiologia 2014, 722, 263–277. [Google Scholar] [CrossRef]
- Jöbgen, A.M.; Palm, A.; Melkonian, M. Phosphorus removal from eutrophic lakes using periphyton on submerged artificial substrata. Hydrobiologia 2004, 528, 123–142. [Google Scholar] [CrossRef]
- Thomas, S.; Gaiser, E.E.; Gantar, M.; Scinto, L.J. Quantifying the responses of calcareous periphyton crusts to rehydration: A microcosm study (Florida Everglades). Aquat. Bot. 2006, 84, 317–323. [Google Scholar] [CrossRef]
- Gottlieb, A.D. Short and Long Hydroperiod Everglades Periphyton Mats: Community Characterization and Experimental Hydroperiod Manipulation. Ph.D. Thesis, Florida International University, Miami, FL, USA, 2003. [Google Scholar]
- Davis, S.M.; Gaiser, E.E.; Loftus, W.F.; Huffman, A.E. Southern marl prairies conceptual ecological model. Wetlands 2005, 25, 821–831. [Google Scholar] [CrossRef]
- Craft, C.B.; Vymazal, J.; Richardson, C.J. Response of Everglades plant communities to nitrogen and phosphorus additions. Wetlands 1995, 15, 258–271. [Google Scholar] [CrossRef]
- Havens, K.E.; Hauxwell, J.; Tyler, A.C.; Thomas, S.; McGlathery, K.J.; Cebrian, J.; Valiela, I.; Steinman, A.D.; Hwang, S.J. Complex interactions between autotrophs in shallow marine and freshwater ecosystems: Implications for community responses to nutrient stress. Environ. Pollut. 2001, 113, 95–107. [Google Scholar] [CrossRef]
- Strickland, J.D.H.; Parsons, T.R. A Practical Handbook of Seawater Analysis, 2nd ed.; Supply and Services Canada: Ottawa, ON, Canada, 1972; pp. 57–62. [Google Scholar]
- Bremner, J.M. Nitrogen-total. In Methods of Soil Analysis Part 3—Chemical Methods; Soil Science Society of America, American Society of Agronomy: Madison, WI, USA, 1996; pp. 1085–1121. [Google Scholar]
- Gonzalez, C.A.; Choquette, S.J. Certificate of Analysis Standard Reference Material 1515 Apple Leaves; National Institute of Standards & Technology: Gaithersburg, MD, USA, 2017.
- Gaiser, E. Periphyton Biomass Accumulation from the Shark River and Taylor Sloughs, Everglades National Park (FCE), from January 2003 to Present. Long Term Ecological Research Network, 22 February 2017. Available online: http://dx.doi.org/10.6073/pasta/d773a9573ce77c9b8548d921ef98d864 (accessed on 30 March 2017).
- Environmental Protection Agency. Methods for Chemical Analysis of Water and Wastes (EPA-600/4-79-020); Environmental Protection Agency: Cincinnati, OH, USA, 1983.
- Castaneda, E.; Rivera-Monroy, V. Water Levels from the Shark River Slough and Taylor Slough, Everglades National Park (FCE), South Florida from May 2001 to Present. Long Term Ecological Research Network, 30 September 2014. Available online: http://dx.doi.org/10.6073/pasta/96730deb08e9a150633771afc965f106 (accessed on 30 March 2017).
- Troxler, T.; Childers, D. Water Levels from the Taylor Slough, Everglades National Park (FCE), South Florida from August 1999 to Present. Long Term Ecological Research Network, 6 March 2017. Available online: http://dx.doi.org/10.6073/pasta/c66a3111977828c21b6fa31cc9d25bcf (accessed on 30 March 2017).
- Cotner, J.B.; Hall, E.K.; Scott, J.T.; Heldal, M. Freshwater bacteria are stoichiometrically flexible with a nutrient composition similar to seston. Front. Microbiol. 2010, 1, 132. [Google Scholar] [CrossRef] [PubMed]
- Weemaes, C.A.; Ooms, V.; Van Loey, A.M.; Hendrickx, M.E. Kinetics of chlorophyll degradation and color loss in heated broccoli juice. J. Agric. Food Chem. 1999, 47, 2404–2409. [Google Scholar] [CrossRef] [PubMed]
- Koca, N.; Karadeniz, F.; Burdurlu, H.S. Effect of pH on chlorophyll degradation and colour loss in blanched green peas. Food Chem. 2007, 100, 609–615. [Google Scholar] [CrossRef]
- Rosen, B.H.; Mareš, J. Catalog of Microscopic Organisms of the Everglades, Part 1—The Cyanobacteria; U.S. Geological Survey Open-File Report 2016-1114; USGS Publications Warehouse: Gainesville, FL, USA, 2016; pp. 1–108. [CrossRef]
- Garcia-Pichel, F.; Castenholz, R.W. Characterization and biological implications of scytonemin, a cyanobacterial sheath pigment. J. Phycol. 1991, 27, 395–409. [Google Scholar] [CrossRef]
- Rudnick, D.T.; Chen, Z.; Childers, D.L.; Fontaine, T.D. Phosphorus and nitrogen inputs to Florida Bay: The importance of the Everglades watershed. Estuaries 1999, 22, 398–416. [Google Scholar] [CrossRef]
- Wozniak, J.R.; Anderson, W.T.; Childers, D.L.; Gaiser, E.E.; Madden, C.J.; Rudnick, D.T. Potential N processing by southern Everglades freshwater marshes: Are Everglades marshes passive conduits for nitrogen? Estuar. Coast. Shelf Sci. 2012, 96, 60–68. [Google Scholar] [CrossRef]
- Kominoski, J.S.; Gaiser, E.E.; Castañeda-Moya, E.; Davis, S.E.; Dessu, S.; Lee, D.Y.; Marazzi, L.; Rivera-Monroy, V.H.; Sola, A.; Surratt, D.; et al. Enhanced marine connectivity and freshwater restoration increase phosphorus in wetland ecosystems of the Florida Coastal Everglades. Ecosystems 2018. submitted for publication. [Google Scholar]
- Flower, H.; Rains, M.; Lewis, D.; Zhang, J.Z.; Price, R. Control of phosphorus concentration through adsorption and desorption in shallow groundwater of subtropical carbonate estuary. Estuar. Coast. Shelf Sci. 2016, 169, 238–247. [Google Scholar] [CrossRef]

| Treatment | Conditioning Phase | Final Rewetting | ||||||
|---|---|---|---|---|---|---|---|---|
| 26-Sept | 3-Oct | 9-Oct | 14-Oct | 19-Oct | 25-Oct | 31-Oct | 1–25-Nov | |
| Control | ||||||||
| High Variance | ||||||||
| Low Variance | ||||||||
| Dry | Wet | |||||||
| Treatment | TP (μg/Core) | TN (μg/Core) | N:P | N:P Correlation | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Min | Max | Mean | SD | Min | Max | Mean | r | p | |
| Pre-treatment | 29.3 | 8.3 | 19.3 | 42.2 | 4345 | 1025 | 3289 | 6538 | 155 | 0.58 | 0.10 |
| Control | 73.4 | 29.4 | 29.7 | 139.2 | 11,642 | 4966 | 4824 | 22,034 | 160 | 0.85 | 1.40 × 10−7 |
| High Variance | 109.4 | 44.8 | 39.3 | 251.5 | 14,073 | 7173 | 7958 | 33,177 | 129 | 0.89 | 7.37 × 10−9 |
| Low Variance | 112.5 | 56.7 | 43.7 | 301.8 | 12,183 | 5428 | 6167 | 30,983 | 116 | 0.84 | 2.52 × 10−7 |
| Drainage/Site | TP (g/m2) | TN (g/m2) | N:P | N:P Correlation | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Range | Mean | SD | Range | Mean | SD | Range | r | p | |
| SRS | 0.009 | 0.014 | 0.000–0.100 | 0.78 | 1.18 | 0.01–8.08 | 90.7 | 42.0 | 11.1–331.7 | 0.901 | 1.23 × 10−36 |
| 1d | 0.012 | 0.016 | 0.000–0.100 | 1.20 | 1.61 | 0.01–8.08 | 103.1 | 46.9 | 34.3–306.9 | 0.928 | 5.79 × 10−14 |
| 2 | 0.008 | 0.013 | 0.000–0.078 | 0.68 | 0.89 | 0.04–4.88 | 94.4 | 42.8 | 28.9–331.7 | 0.972 | 3.66 × 10−24 |
| 3 | 0.006 | 0.010 | 0.000–0.058 | 0.40 | 0.59 | 0.02–3.24 | 68.9 | 20.6 | 11.2–158.7 | 0.832 | 2.17 × 10−08 |
| TS/Ph | 0.033 | 0.050 | 0.000–0.496 | 3.39 | 4.14 | 0.00–40.07 | 145.3 | 147.0 | 0.0–1731.7 | 0.761 | 5.78 × 10−15 |
| 1b | 0.036 | 0.051 | 0.001–0.343 | 3.14 | 5.32 | 0.08–40.07 | 87.5 | 34.7 | 24.1–215.7 | 0.992 | 3.31 × 10−20 |
| 2 | 0.035 | 0.063 | 0.002–0.496 | 2.56 | 2.74 | 0.00–12.50 | 118.2 | 89.7 | 0.0–474.2 | 0.548 | 4.61 × 10−3 |
| 3 | 0.027 | 0.029 | 0.000–0.133 | 4.44 | 3.89 | 0.19–15.52 | 226.2 | 210.4 | 24.9–1731.7 | 0.901 | 8.30 × 10−10 |
| Drainage/Site | Season | Hydrological Season | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Autumn | Winter | Spring | Dry | Wet | ||||||
| r | p | r | p | r | p | r | p | r | p | |
| SRS | ||||||||||
| 1d | 0.13 | −0.30 | 0.47 | −0.71 | 0.07 | −0.32 | 0.36 | −0.17 | ||
| 2 | 0.25 | −0.60 | 0.12 | −0.71 | 0.05 | −0.35 | 0.29 | −0.35 | 0.50 | |
| 3 | 0.19 | 0.38 | 0.45 | N/A | 0.03 | 0.03 | ||||
| TS/Ph | ||||||||||
| 1b | −0.29 | N/A | N/A | N/A | −0.16 | |||||
| 2 | −0.20 | −0.69 | 0.09 | N/A | −0.69 | 0.09 | −0.69 | 0.53 | ||
| 3 | 0.49 | 0.18 | −0.07 | N/A | −0.07 | −0.07 | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sola, A.D.; Marazzi, L.; Flores, M.M.; Kominoski, J.S.; Gaiser, E.E. Short-Term Effects of Drying-Rewetting and Long-Term Effects of Nutrient Loading on Periphyton N:P Stoichiometry. Water 2018, 10, 105. https://doi.org/10.3390/w10020105
Sola AD, Marazzi L, Flores MM, Kominoski JS, Gaiser EE. Short-Term Effects of Drying-Rewetting and Long-Term Effects of Nutrient Loading on Periphyton N:P Stoichiometry. Water. 2018; 10(2):105. https://doi.org/10.3390/w10020105
Chicago/Turabian StyleSola, Andres D., Luca Marazzi, Monica M. Flores, John S. Kominoski, and Evelyn E. Gaiser. 2018. "Short-Term Effects of Drying-Rewetting and Long-Term Effects of Nutrient Loading on Periphyton N:P Stoichiometry" Water 10, no. 2: 105. https://doi.org/10.3390/w10020105






