Ion Channels and Transporters in Inflammation: Special Focus on TRP Channels and TRPC6
Abstract
:1. Introduction
2. Elementary Immunology: An Expanding Landscape
3. Multiple Roles for Members of the TRP Channel Family in Inflammation
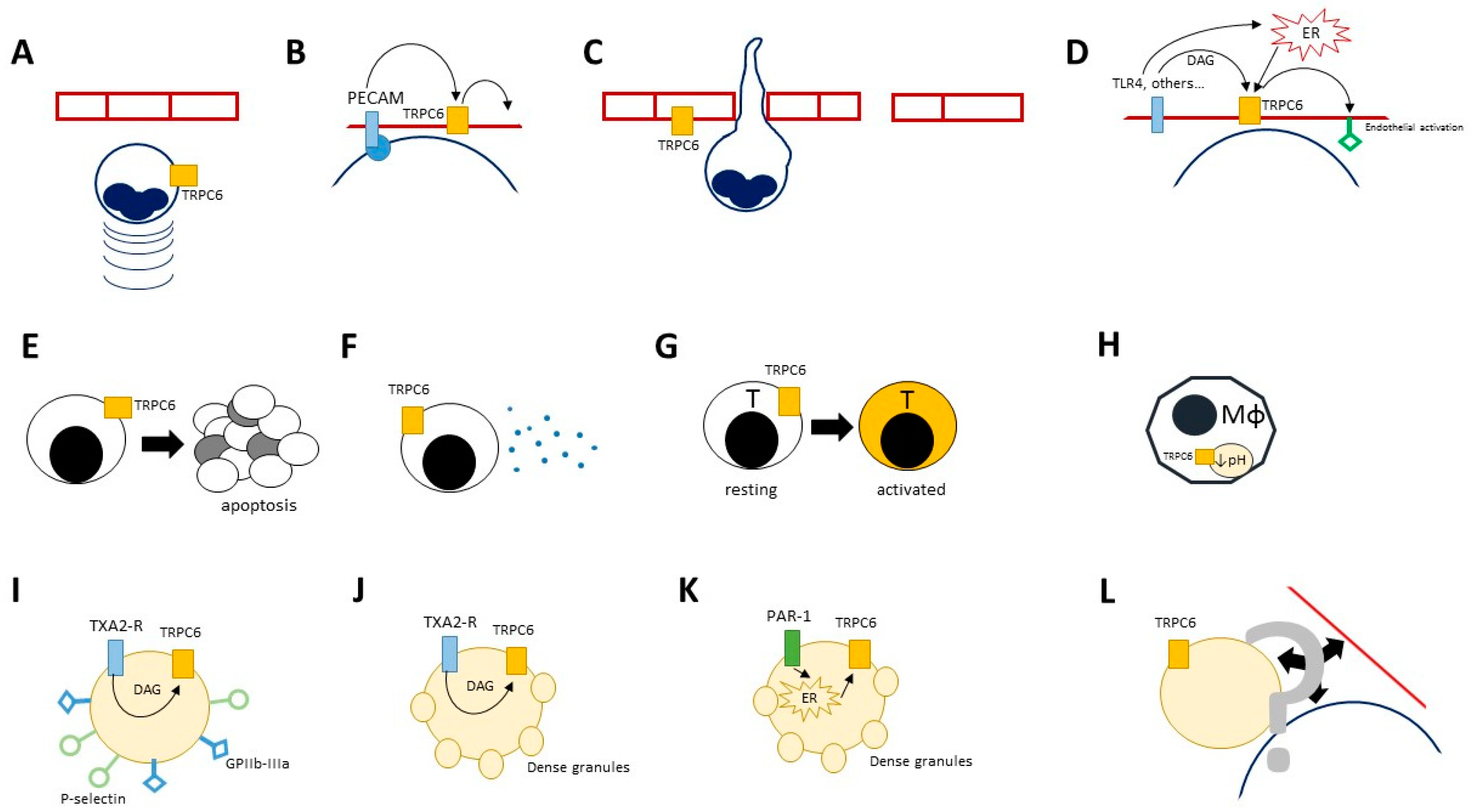
4. TRPC6 and Immune Responses
5. Effects of TRPC6 Activation and Function on Inflamed Tissues
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Venter, J.C.; Adams, M.D.; Myers, E.W.; Li, P.W.; Mural, R.J.; Sutton, G.G.; Smith, H.O.; Yandell, M.; Evans, C.A.; Holt, R.A.; et al. The sequence of the human genome. Science 2001, 291, 1304–1351. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; He, F.F.; Wang, H.; Fang, Z.; Shao, N.; Tian, X.J.; Liu, J.S.; Zhu, Z.H.; Wang, Y.M.; Wang, S.; et al. Calcium entry via TRPC6 mediates albumin overload-induced endoplasmic reticulum stress and apoptosis in podocytes. Cell Calcium 2011, 50, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Schatz, V.; Neubert, P.; Schroder, A.; Binger, K.; Gebhard, M.; Muller, D.N.; Luft, F.C.; Titze, J.; Jantsch, J. Elementary immunology: Na(+) as a regulator of immunity. Pediatr. Nephrol. 2017, 32, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Parenti, A.; De Logu, F.; Geppetti, P.; Benemei, S. What is the evidence for the role of TRP channels in inflammatory and immune cells? Br. J. Pharmacol. 2016, 173, 953–969. [Google Scholar] [CrossRef] [PubMed]
- European Bioinformatics Institute (EMBL-EBI); SIB Swiss Institute of Bioinformatics; (PIR), P.I.R. Universal Protein Resource (Uniprot). Available online: http://www.uniprot.org/ (accessed on 5 June 2018).
- Sumoza-Toledo, A.; Lange, I.; Cortado, H.; Bhagat, H.; Mori, Y.; Fleig, A.; Penner, R.; Partida-Sanchez, S. Dendritic cell maturation and chemotaxis is regulated by TRPM2-mediated lysosomal Ca2+ release. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2011, 25, 3529–3542. [Google Scholar] [CrossRef] [PubMed]
- Maxson, M.E.; Grinstein, S. The Vacuolar-Type H+-ATPase at a Glance—More Than a Proton Pump; The Company of Biologists Ltd.: Cambridge, UK, 2014. [Google Scholar]
- Riazanski, V.; Gabdoulkhakova, A.G.; Boynton, L.S.; Eguchi, R.R.; Deriy, L.V.; Hogarth, D.K.; Loaec, N.; Oumata, N.; Galons, H.; Brown, M.E.; et al. TRPC6 channel translocation into phagosomal membrane augments phagosomal function. Proc. Natl. Acad. Sci. USA 2015, 112, E6486–E6495. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salao, K.; Jiang, L.; Li, H.; Tsai, V.W.; Husaini, Y.; Curmi, P.M.; Brown, L.J.; Brown, D.A.; Breit, S.N. Clic1 regulates dendritic cell antigen processing and presentation by modulating phagosome acidification and proteolysis. Biol. Open 2016, 5, 620–630. [Google Scholar] [CrossRef] [PubMed]
- Clemens, R.A.; Lowell, C.A. Store-operated calcium signaling in neutrophils. J. Leukoc. Biol. 2015, 98, 497–502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tintinger, G.R.; Steel, H.C.; Theron, A.J.; Anderson, R. Pharmacological control of neutrophil-mediated inflammation: Strategies targeting calcium handling by activated polymorphonuclear leukocytes. Drug Des. Dev. Ther. 2009, 2, 95–104. [Google Scholar]
- Vaeth, M.; Maus, M.; Klein-Hessling, S.; Freinkman, E.; Yang, J.; Eckstein, M.; Cameron, S.; Turvey, S.E.; Serfling, E.; Berberich-Siebelt, F. Store-operated Ca2+ entry controls clonal expansion of T cells through metabolic reprogramming. Immunity 2017, 47, 664–679. [Google Scholar] [CrossRef] [PubMed]
- Clemens, R.A.; Chong, J.; Grimes, D.; Hu, Y.; Lowell, C.A. STIM1 and STIM2 cooperatively regulate mouse neutrophil store-operated calcium entry and cytokine production. Blood 2017, 130, 1565–1577. [Google Scholar] [CrossRef] [PubMed]
- Vaeth, M.; Eckstein, M.; Shaw, P.J.; Kozhaya, L.; Yang, J.; Berberich-Siebelt, F.; Clancy, R.; Unutmaz, D.; Feske, S. Store-operated Ca(2+) entry in follicular T cells controls humoral immune responses and autoimmunity. Immunity 2016, 44, 1350–1364. [Google Scholar] [CrossRef] [PubMed]
- Vaeth, M.; Feske, S. Ion channelopathies of the immune system. Curr. Opin. Immunol. 2018, 52, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Harr, M.W.; Rong, Y.; Bootman, M.D.; Roderick, H.L.; Distelhorst, C.W. Glucocorticoid-mediated inhibition of LCK modulates the pattern of T cell receptor-induced calcium signals by down-regulating inositol 1,4,5-trisphosphate receptors. J. Biol. Chem. 2009, 284, 31860–31871. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Wang, H.; Lin, Q.; Fan, F.; Zhang, F.; Peng, X.; Fang, X.; Liu, J.; Ouyang, K. Loss of IP3 receptor-mediated Ca(2+) release in mouse B cells results in abnormal B cell development and function. J. Immunol. 2017, 199, 570–580. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, K.; Leandro Gomez-Amaro, R.; Stachura, D.L.; Tang, H.; Peng, X.; Fang, X.; Traver, D.; Evans, S.M.; Chen, J. Loss of IP3R-dependent Ca2+ signalling in thymocytes leads to aberrant development and acute lymphoblastic leukemia. Nat. Commun. 2014, 5, 4814. [Google Scholar] [CrossRef] [PubMed]
- Lichtenegger, M.; Tiapko, O.; Svobodova, B.; Stockner, T.; Glasnov, T.N.; Schreibmayer, W.; Platzer, D.; Cruz, G.G.; Krenn, S.; Schober, R. An optically controlled probe identifies lipid-gating fenestrations within the TRPC3 channel. Nat. Chem. Biol. 2018, 14, 396. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, A.; Gudermann, T. TRPC6: Physiological function and pathophysiological relevance. Handb. Exp. Pharmacol. 2014, 222, 157–188. [Google Scholar] [PubMed]
- Li, F.-Y.; Chaigne-Delalande, B.; Kanellopoulou, C.; Davis, J.C.; Matthews, H.F.; Douek, D.C.; Cohen, J.I.; Uzel, G.; Su, H.C.; Lenardo, M.J. Signaling role for Mg2+ revealed by immunodeficiency due to loss of MAGT1. Nature 2011, 475, 471. [Google Scholar] [CrossRef] [PubMed]
- Deason-Towne, F.; Perraud, A.-L.; Schmitz, C. Identification of SER/THR phosphorylation sites in the C2-domain of phospholipase c γ2 (plcγ2) using TRPM7-kinase. Cell. Signal. 2012, 24, 2070–2075. [Google Scholar] [CrossRef] [PubMed]
- Cahalan, M.D.; Chandy, K.G. The functional network of ion channels in T lymphocytes. Immunol. Rev. 2009, 231, 59–87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmitz, C.; Perraud, A.-L.; Johnson, C.O.; Inabe, K.; Smith, M.K.; Penner, R.; Kurosaki, T.; Fleig, A.; Scharenberg, A.M. Regulation of vertebrate cellular Mg2+ homeostasis by TRPM7. Cell 2003, 114, 191–200. [Google Scholar] [CrossRef]
- Gotru, S.K.; Gil-Pulido, J.; Beyersdorf, N.; Diefenbach, A.; Becker, I.C.; Vögtle, T.; Remer, K.; Chubanov, V.; Gudermann, T.; Hermanns, H.M. Cutting edge: Imbalanced cation homeostasis in MAGT1-deficient B cells dysregulates B cell development and signaling in mice. J. Immunol. 2018, 200, 2529–2534. [Google Scholar] [CrossRef] [PubMed]
- Hojyo, S.; Fukada, T. Roles of zinc signaling in the immune system. J. Immunol. Res. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Feske, S.; Wulff, H.; Skolnik, E.Y. Ion channels in innate and adaptive immunity. Annu. Rev. Immunol. 2015, 33, 291–353. [Google Scholar] [CrossRef] [PubMed]
- Mizoguchi, Y.; Monji, A. TRPC channels and brain inflammation. In Transient Receptor Potential Canonical Channels and Brain Diseases; Springer: Berlin/Heidelberg, Germany, 2017; pp. 111–121. [Google Scholar]
- Di Virgilio, F.; Dal Ben, D.; Sarti, A.C.; Giuliani, A.L.; Falzoni, S. The P2X7 receptor in infection and inflammation. Immunity 2017, 47, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Hamanaka, K.; Jian, M.Y.; Townsley, M.I.; King, J.A.; Liedtke, W.; Weber, D.S.; Eyal, F.G.; Clapp, M.M.; Parker, J.C. TRPV4 channels augment macrophage activation and ventilator-induced lung injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 2010, 299, L353–L362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davenport, B.; Li, Y.; Heizer, J.W.; Schmitz, C.; Perraud, A.L. Signature channels of excitability no more: L-type channels in immune cells. Front. Immunol. 2015, 6, 375. [Google Scholar] [CrossRef] [PubMed]
- Badou, A.; Jha, M.K.; Matza, D.; Flavell, R.A. Emerging roles of L-type voltage-gated and other calcium channels in T lymphocytes. Front. Immunol. 2013, 4, 243. [Google Scholar] [CrossRef] [PubMed]
- Blaustein, M.P.; Zhang, J.; Chen, L.; Song, H.; Raina, H.; Kinsey, S.P.; Izuka, M.; Iwamoto, T.; Kotlikoff, M.I.; Lingrel, J.B.; et al. The pump, the exchanger, and endogenous Ouabain: Signaling mechanisms that link salt retention to hypertension. Hypertension 2009, 53, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Boscia, F.; D’Avanzo, C.; Pannaccione, A.; Secondo, A.; Casamassa, A.; Formisano, L.; Guida, N.; Scorziello, A.; Di Renzo, G.; Annunziato, L. New roles of NCX in glial cells: Activation of microglia in ischemia and differentiation of oligodendrocytes. Adv. Exp. Med. Biol. 2013, 961, 307–316. [Google Scholar] [PubMed]
- Iwamoto, T.; Kita, S.; Zhang, J.; Blaustein, M.P.; Arai, Y.; Yoshida, S.; Wakimoto, K.; Komuro, I.; Katsuragi, T. Salt-sensitive hypertension is triggered by Ca2+ entry via Na+/Ca2+ exchanger type-1 in vascular smooth muscle. Nat. Med. 2004, 10, 1193. [Google Scholar] [CrossRef] [PubMed]
- Citterio, L.; Simonini, M.; Zagato, L.; Salvi, E.; Delli Carpini, S.; Lanzani, C.; Messaggio, E.; Casamassima, N.; Frau, F.; D’Avila, F.; et al. Genes involved in vasoconstriction and vasodilation system affect salt-sensitive hypertension. PLoS ONE 2011, 6, e19620. [Google Scholar] [CrossRef] [PubMed]
- Kleinewietfeld, M.; Manzel, A.; Titze, J.; Kvakan, H.; Yosef, N.; Linker, R.A.; Muller, D.N.; Hafler, D.A. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature 2013, 496, 518–522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hernandez, A.L.; Kitz, A.; Wu, C.; Lowther, D.E.; Rodriguez, D.M.; Vudattu, N.; Deng, S.; Herold, K.C.; Kuchroo, V.K.; Kleinewietfeld, M.; et al. Sodium chloride inhibits the suppressive function of FOXP3+ regulatory T cells. J. Clin. Investig. 2015, 125, 4212–4222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Junger, W.G.; Liu, F.C.; Loomis, W.H.; Hoyt, D.B. Hypertonic saline enhances cellular immune function. Circ. Shock 1994, 42, 190–196. [Google Scholar] [PubMed]
- Binger, K.J.; Gebhardt, M.; Heinig, M.; Rintisch, C.; Schroeder, A.; Neuhofer, W.; Hilgers, K.; Manzel, A.; Schwartz, C.; Kleinewietfeld, M.; et al. High salt reduces the activation of IL-4- and IL-13-stimulated macrophages. J. Clin. Investig. 2015, 125, 4223–4238. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, L.; Dinarello, C.A. Osmotic regulation of cytokine synthesis in vitro. Proc. Natl. Acad. Sci. USA 1995, 92, 12230–12234. [Google Scholar] [CrossRef] [PubMed]
- Barbaro, N.R.; Foss, J.D.; Kryshtal, D.O.; Tsyba, N.; Kumaresan, S.; Xiao, L.; Mernaugh, R.L.; Itani, H.A.; Loperena, R.; Chen, W.; et al. Dendritic cell amiloride-sensitive channels mediate sodium-induced inflammation and hypertension. Cell Rep. 2017, 21, 1009–1020. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, G.A.; Lanzani, C.; Bozzolo, E.P.; Zagato, L.; Citterio, L.; Casamassima, N.; Canti, V.; Sabbadini, M.G.; Rovere-Querini, P.; Manunta, P.; et al. Beta-adducin and sodium-calcium exchanger 1 gene variants are associated with systemic lupus erythematosus and lupus nephritis. Rheumatol. Int. 2015, 35, 1975–1983. [Google Scholar] [CrossRef] [PubMed]
- Staiano, R.I.; Granata, F.; Secondo, A.; Petraroli, A.; Loffredo, S.; Frattini, A.; Annunziato, L.; Marone, G.; Triggiani, M. Expression and function of Na+/Ca2+ exchangers 1 and 3 in human macrophages and monocytes. Eur. J. Immunol. 2009, 39, 1405–1418. [Google Scholar] [CrossRef] [PubMed]
- Carrithers, L.M.; Hulseberg, P.; Sandor, M.; Carrithers, M.D. The human macrophage sodium channel NAV1.5 regulates mycobacteria processing through organelle polarization and localized calcium oscillations. FEMS Immunol. Med. Microbiol. 2011, 63, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Lo, W.L.; Donermeyer, D.L.; Allen, P.M. A voltage-gated sodium channel is essential for the positive selection of Cd4(+) T cells. Nat. Immunol. 2012, 13, 880–887. [Google Scholar] [CrossRef] [PubMed]
- Di, L.; Srivastava, S.; Zhdanova, O.; Ding, Y.; Li, Z.; Wulff, H.; Lafaille, M.; Skolnik, E.Y. Inhibition of the K+ channel KCa3.1 ameliorates T cell-mediated colitis. Proc. Natl. Acad. Sci. USA 2010, 107, 1541–1546. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, A.; D’Alessandro, G.; Golia, M.T.; Grossinger, E.M.; Di Angelantonio, S.; Ragozzino, D.; Santoro, A.; Esposito, V.; Wulff, H.; Catalano, M.; et al. KCa3.1 inhibition switches the phenotype of glioma-infiltrating microglia/macrophages. Cell Death Dis. 2016, 7, e2174. [Google Scholar] [CrossRef] [PubMed]
- Prud’homme, G.J.; Glinka, Y.; Wang, Q. Immunological gabaergic interactions and therapeutic applications in autoimmune diseases. Autoimmun. Rev. 2015, 14, 1048–1056. [Google Scholar] [CrossRef] [PubMed]
- Simonin-Le Jeune, K.; Le Jeune, A.; Jouneau, S.; Belleguic, C.; Roux, P.F.; Jaguin, M.; Dimanche-Boitre, M.T.; Lecureur, V.; Leclercq, C.; Desrues, B.; et al. Impaired functions of macrophage from cystic fibrosis patients: Cd11b, TLR-5 decrease and SCD14, inflammatory cytokines increase. PLoS ONE 2013, 8, e75667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leveque, M.; Penna, A.; Le Trionnaire, S.; Belleguic, C.; Desrues, B.; Brinchault, G.; Jouneau, S.; Lagadic-Gossmann, D.; Martin-Chouly, C. Phagocytosis depends on TRPV2-mediated calcium influx and requires TRPV2 in lipids rafts: Alteration in macrophages from patients with cystic fibrosis. Sci. Rep. 2018, 8, 4310. [Google Scholar] [CrossRef] [PubMed]
- Lindemann, O.; Strodthoff, C.; Horstmann, M.; Nielsen, N.; Jung, F.; Schimmelpfennig, S.; Heitzmann, M.; Schwab, A. TRPC1 regulates FMLP-stimulated migration and chemotaxis of neutrophil granulocytes. Biochim. Biophys. Acta 2015, 1853, 2122–2130. [Google Scholar] [CrossRef] [PubMed]
- Capasso, M.; Bhamrah, M.K.; Henley, T.; Boyd, R.S.; Langlais, C.; Cain, K.; Dinsdale, D.; Pulford, K.; Khan, M.; Musset, B. HVCN1 modulates BCR signal strength via regulation of BCR-dependent generation of reactive oxygen species. Nat. Immunol. 2010, 11, 265. [Google Scholar] [CrossRef] [PubMed]
- Di, A.; Gao, X.P.; Qian, F.; Kawamura, T.; Han, J.; Hecquet, C.; Ye, R.D.; Vogel, S.M.; Malik, A.B. The redox-sensitive cation channel TRPM2 modulates phagocyte ROS production and inflammation. Nat. Immunol. 2011, 13, 29–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsai, L.J.; Hsiao, S.H.; Tsai, L.M.; Lin, C.Y.; Tsai, J.J.; Liou, D.M.; Lan, J.L. The sodium-dependent glucose cotransporter SLC5A11 as an autoimmune modifier gene in SLE. Tissue Antigens 2008, 71, 114–126. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Castejon, G.; Brough, D. Understanding the mechanism of IL-1β secretion. Cytokine Growth Factor Rev. 2011, 22, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Py, B.F.; Jin, M.; Desai, B.N.; Penumaka, A.; Zhu, H.; Kober, M.; Dietrich, A.; Lipinski, M.M.; Henry, T.; Clapham, D.E. Caspase-11 controls interleukin-1β release through degradation of TRPC1. Cell Rep. 2014, 6, 1122–1128. [Google Scholar] [CrossRef] [PubMed]
- Malik, M.; Jividen, K.; Padmakumar, V.; Cataisson, C.; Li, L.; Lee, J.; Howard, O.Z.; Yuspa, S.H. Inducible NOS-induced chloride intracellular channel 4 (CLIC4) nuclear translocation regulates macrophage deactivation. Proc. Natl. Acad. Sci. USA 2012, 109, 6130–6135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Domingo-Fernández, R.; Coll, R.C.; Kearney, J.; Breit, S.; O’Neill, L.A. The intracellular chloride channel proteins CLIC1 and CLIC4 induce IL-1β transcription and activate the NLRP3 inflammasome. J. Biol. Chem. 2017, 292, 12077–12087. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hogan, P.G.; Lewis, R.S.; Rao, A. Molecular basis of calcium signaling in lymphocytes: STIM and ORAI. Annu. Rev. Immunol. 2010, 28, 491–533. [Google Scholar] [CrossRef] [PubMed]
- Bogeski, I.; Kummerow, C.; Al-Ansary, D.; Schwarz, E.C.; Koehler, R.; Kozai, D.; Takahashi, N.; Peinelt, C.; Griesemer, D.; Bozem, M. Differential redox regulation of ORAI ion channels: A mechanism to tune cellular calcium signaling. Sci. Signal. 2010, 3, ra24. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Zhuang, Y.; Zhou, Z.; Zhao, J.; Chen, Q.; Zheng, J. Nf-kappab dependent up-regulation of TRPC6 by abeta in bv-2 microglia cells increases Cox-2 expression and contributes to hippocampus neuron damage. Neurosci. Lett. 2017, 651, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Desvignes, L.; Weidinger, C.; Shaw, P.; Vaeth, M.; Ribierre, T.; Liu, M.; Fergus, T.; Kozhaya, L.; McVoy, L.; Unutmaz, D.; et al. Stim1 controls T cell-mediated immune regulation and inflammation in chronic infection. J. Clin. Investig. 2015, 125, 2347–2362. [Google Scholar] [CrossRef] [PubMed]
- Nunes-Hasler, P.; Maschalidi, S.; Lippens, C.; Castelbou, C.; Bouvet, S.; Guido, D.; Bermont, F.; Bassoy, E.Y.; Page, N.; Merkler, D.; et al. STIM1 promotes migration, phagosomal maturation and antigen cross-presentation in dendritic cells. Nat. Commun. 2017, 8, 1852. [Google Scholar] [CrossRef] [PubMed]
- Paria, B.C.; Vogel, S.M.; Ahmmed, G.U.; Alamgir, S.; Shroff, J.; Malik, A.B.; Tiruppathi, C. Tumor necrosis factor-alpha-induced TRPC1 expression amplifies store-operated Ca2+ influx and endothelial permeability. Am. J. Physiol. Lung Cell. Mol. Physiol. 2004, 287, L1303–L1313. [Google Scholar] [CrossRef] [PubMed]
- Tiruppathi, C.; Ahmmed, G.U.; Vogel, S.M.; Malik, A.B. Ca2+ signaling, TRP channels, and endothelial permeability. Microcirculation 2006, 13, 693–708. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.Y.; Wang, L.M.; Zhong, H.; Liu, Y.M.; Tang, N.; Zhu, L.P.; He, F.; Hu, Q.H. TRPC1 stimulates calciumsensing receptorinduced storeoperated Ca2+ entry and nitric oxide production in endothelial cells. Mol. Med. Rep. 2017, 16, 4613–4619. [Google Scholar] [CrossRef] [PubMed]
- Lopez, E.; Bermejo, N.; Berna-Erro, A.; Alonso, N.; Salido, G.M.; Redondo, P.C.; Rosado, J.A. Relationship between calcium mobilization and platelet alpha- and delta-granule secretion. A role for TRPC6 in thrombin-evoked delta-granule exocytosis. Arch. Biochem. Biophys. 2015, 585, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Melzer, N.; Hicking, G.; Gobel, K.; Wiendl, H. TRPM2 cation channels modulate T cell effector functions and contribute to autoimmune CNS inflammation. PLoS ONE 2012, 7, e47617. [Google Scholar] [CrossRef] [PubMed]
- Wehrhahn, J.; Kraft, R.; Harteneck, C.; Hauschildt, S. Transient receptor potential melastatin 2 is required for lipopolysaccharide-induced cytokine production in human monocytes. J. Immunol. 2010, 184, 2386–2393. [Google Scholar] [CrossRef] [PubMed]
- Syed Mortadza, S.A.; Wang, L.; Li, D.; Jiang, L.H. TRPM2 channel-mediated ROS-sensitive Ca(2+) signaling mechanisms in immune cells. Front. Immunol. 2015, 6, 407. [Google Scholar] [CrossRef] [PubMed]
- Hiroi, T.; Wajima, T.; Negoro, T.; Ishii, M.; Nakano, Y.; Kiuchi, Y.; Mori, Y.; Shimizu, S. Neutrophil TRPM2 channels are implicated in the exacerbation of myocardial ischaemia/reperfusion injury. Cardiovasc. Res. 2013, 97, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Mittal, M.; Nepal, S.; Tsukasaki, Y.; Hecquet, C.M.; Soni, D.; Rehman, J.; Tiruppathi, C.; Malik, A.B. Neutrophil activation of endothelial cell-expressed TRPM2 mediates transendothelial neutrophil migration and vascular injury. Circ. Res. 2017, 121, 1081–1091. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, S.; Shimizu, S.; Kiyonaka, S.; Takahashi, N.; Wajima, T.; Hara, Y.; Negoro, T.; Hiroi, T.; Kiuchi, Y.; Okada, T. TRPM2-mediated Ca2+ influx induces chemokine production in monocytes that aggravates inflammatory neutrophil infiltration. Nat. Med. 2008, 14, 738. [Google Scholar] [CrossRef] [PubMed]
- Di, A.; Kiya, T.; Gong, H.; Gao, X.; Malik, A.B. Role of the phagosomal redox-sensitive TRP channel TRPM2 in regulating bactericidal activity of macrophages. J. Cell Sci. 2017, 130, 735–744. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Philipp, S.; Strauss, B.; Hirnet, D.; Wissenbach, U.; Mery, L.; Flockerzi, V.; Hoth, M. TRPC3 mediates T-cell receptor-dependent calcium entry in human T-lymphocytes. J. Biol. Chem. 2003, 278, 26629–26638. [Google Scholar] [CrossRef] [PubMed]
- Solanki, S.; Dube, P.R.; Birnbaumer, L.; Vazquez, G. Reduced necrosis and content of apoptotic m1 macrophages in advanced atherosclerotic plaques of mice with macrophage-specific loss of TRPC3. Sci. Rep. 2017, 7, 42526. [Google Scholar] [CrossRef] [PubMed]
- Feng, M.; Xu, D.; Wang, L. miR-26a inhibits atherosclerosis progression by targeting TRPC3. Cell Biosci. 2018, 8, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Damann, N.; Owsianik, G.; Li, S.; Poll, C.; Nilius, B. The calcium-conducting ion channel transient receptor potential canonical 6 is involved in macrophage inflammatory protein-2-induced migration of mouse neutrophils. Acta Physiol. 2009, 195, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Lindemann, O.; Umlauf, D.; Frank, S.; Schimmelpfennig, S.; Bertrand, J.; Pap, T.; Hanley, P.J.; Fabian, A.; Dietrich, A.; Schwab, A. TRPC6 regulates CXCR2-mediated chemotaxis of murine neutrophils. J. Immunol. 2013, 190, 5496–5505. [Google Scholar] [CrossRef] [PubMed]
- Weber, E.W.; Han, F.; Tauseef, M.; Birnbaumer, L.; Mehta, D.; Muller, W.A. TRPC6 is the endothelial calcium channel that regulates leukocyte transendothelial migration during the inflammatory response. J. Exp. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Singh, I.; Knezevic, N.; Ahmmed, G.U.; Kini, V.; Malik, A.B.; Mehta, D. Gαq-TRPC6-mediated Ca2+ entry induces RHOA activation and resultant endothelial cell shape change in response to thrombin. J. Biol. Chem. 2007, 282, 7833–7843. [Google Scholar] [CrossRef] [PubMed]
- Kini, V.; Chavez, A.; Mehta, D. A new role for pten in regulating transient receptor potential canonical channel 6-mediated Ca2+ entry, endothelial permeability, and angiogenesis. J. Biol. Chem. 2010, 285, 33082–33091. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, G.; Sciorati, C.; Bozzolo, E.; Zagato, L.; Citterio, L.; Coletto, L.; Lanzani, C.; Rovere-Querini, P.; Sabbadini, M.; Manunta, P. TRPC6 and Neuropsychiatric SLE: From bedside to bench. In Clinical and Experimental Rheumatology; Clinical & Exper Rheumatology: Pisa, Italy, 2016; p. S67. [Google Scholar]
- Ramirez, G.A.; Lanzani, C.; Bozzolo, E.P.; Citterio, L.; Zagato, L.; Casamassima, N.; Canti, V.; Sabbadini, M.G.; Rovere-Querini, P.; Manunta, P.; et al. TRPC6 gene variants and neuropsychiatric lupus. J. Neuroimmunol. 2015, 288, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Ulmann, L.; Hirbec, H.; Rassendren, F. P2X4 receptors mediate PGE2 release by tissue-resident macrophages and initiate inflammatory pain. EMBO J. 2010, 29, 2290–2300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burnstock, G. P2X ion channel receptors and inflammation. Purinergic Signal. 2016, 12, 59–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Savio, L.E.B.; de Andrade Mello, P.; da Silva, C.G.; Coutinho-Silva, R. The P2X7 receptor in inflammatory diseases: Angel or demon? Front. Pharmacol. 2018, 9, 52. [Google Scholar] [CrossRef] [PubMed]
- Muller, T.; Vieira, R.P.; Grimm, M.; Durk, T.; Cicko, S.; Zeiser, R.; Jakob, T.; Martin, S.F.; Blumenthal, B.; Sorichter, S.; et al. A potential role for P2X7R in allergic airway inflammation in mice and humans. Am. J. Respir. Cell Mol. Biol. 2011, 44, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Labasi, J.M.; Petrushova, N.; Donovan, C.; McCurdy, S.; Lira, P.; Payette, M.M.; Brissette, W.; Wicks, J.R.; Audoly, L.; Gabel, C.A. Absence of the P2X7 receptor alters leukocyte function and attenuates an inflammatory response. J. Immunol. 2002, 168, 6436–6445. [Google Scholar] [CrossRef] [PubMed]
- Hang, P.; Zhao, J.; Cai, B.; Tian, S.; Huang, W.; Guo, J.; Sun, C.; Li, Y.; Du, Z. Brain-derived neurotrophic factor regulates TRPC3/6 channels and protects against myocardial infarction in rodents. Int. J. Biol. Sci. 2015, 11, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Mizoguchi, Y.; Kato, T.A.; Seki, Y.; Ohgidani, M.; Sagata, N.; Horikawa, H.; Yamauchi, Y.; Sato-Kasai, M.; Hayakawa, K.; Inoue, R.; et al. Brain-derived neurotrophic factor (BDNF) induces sustained intracellular Ca2+ elevation through the up-regulation of surface transient receptor potential 3 (TRPC3) channels in rodent microglia. J. Biol. Chem. 2014, 289, 18549–18555. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Lu, Z.H.; Gabius, H.J.; Rohowsky-Kochan, C.; Ledeen, R.W.; Wu, G. Cross-linking of GM1 ganglioside by galectin-1 mediates regulatory T cell activity involving TRPC5 channel activation: Possible role in suppressing experimental autoimmune encephalomyelitis. J. Immunol. 2009, 182, 4036–4045. [Google Scholar] [CrossRef] [PubMed]
- Alawi, K.M.; Russell, F.A.; Aubdool, A.A.; Srivastava, S.; Riffo-Vasquez, Y.; Baldissera, L., Jr.; Thakore, P.; Saleque, N.; Fernandes, E.S.; Walsh, D.A.; et al. Transient receptor potential canonical 5 (TRPC5) protects against pain and vascular inflammation in arthritis and joint inflammation. Ann. Rheum. Dis. 2017, 76, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Bertin, S.; Aoki-Nonaka, Y.; de Jong, P.R.; Nohara, L.L.; Xu, H.; Stanwood, S.R.; Srikanth, S.; Lee, J.; To, K.; Abramson, L.; et al. The ion channel TRPV1 regulates the activation and proinflammatory properties of Cd4(+) T cells. Nat. Immunol. 2014, 15, 1055–1063. [Google Scholar] [CrossRef] [PubMed]
- Link, T.M.; Park, U.; Vonakis, B.M.; Raben, D.M.; Soloski, M.J.; Caterina, M.J. TRPV2 has a pivotal role in macrophage particle binding and phagocytosis. Nat. Immunol. 2010, 11, 232–239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomilin, V.N.; Cherezova, A.L.; Negulyaev, Y.A.; Semenova, S.B. TRPV5/V6 channels mediate Ca(2+) influx in jurkat T cells under the control of extracellular pH. J. Cell. Biochem. 2016, 117, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Ko, J.H.; Nam, J.H.; Woo, J.E.; Min, K.M.; Earm, Y.E.; Kim, S.J. Higher expression of TRPM7 channels in murine mature B lymphocytes than immature cells. Korean J. Physiol. Pharmacol. 2005, 9, 69–75. [Google Scholar]
- Jin, J.; Desai, B.N.; Navarro, B.; Donovan, A.; Andrews, N.C.; Clapham, D.E. Deletion of TRPM7 disrupts embryonic development and thymopoiesis without altering Mg2+ homeostasis. Science 2008, 322, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Wykes, R.C.; Lee, M.; Duffy, S.M.; Yang, W.; Seward, E.P.; Bradding, P. Functional transient receptor potential melastatin 7 channels are critical for human mast cell survival. J. Immunol. 2007, 179, 4045–4052. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Ng, N.M.; Chen, M.; Lin, X.; Tang, T.; Cheng, H.; Yang, C.; Jiang, S. Inhibition of TRPM7 channels reduces degranulation and release of cytokines in rat bone marrow-derived mast cells. Int. J. Mol. Sci. 2014, 15, 11817–11831. [Google Scholar] [CrossRef] [PubMed]
- Schilling, T.; Miralles, F.; Eder, C. TRPM7 regulates proliferation and polarisation of macrophages. J. Cell Sci. 2014, 127, 4561–4566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ryazanova, L.V.; Hu, Z.; Suzuki, S.; Chubanov, V.; Fleig, A.; Ryazanov, A.G. Elucidating the role of the TRPM7 alpha-kinase: TRPM7 kinase inactivation leads to magnesium deprivation resistance phenotype in mice. Sci. Rep. 2014, 4, 7599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ravell, J.; Chaigne-Delalande, B.; Lenardo, M. X-linked immunodeficiency with magnesium defect, epstein-barr virus infection, and neoplasia disease: A combined immune deficiency with magnesium defect. Curr. Opin. Pediatr. 2014, 26, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Serafini, N.; Dahdah, A.; Barbet, G.; Demion, M.; Attout, T.; Gautier, G.; Arcos-Fajardo, M.; Souchet, H.; Jouvin, M.H.; Vrtovsnik, F.; et al. The TRPM4 channel controls monocyte and macrophage, but not neutrophil, function for survival in sepsis. J. Immunol. 2012, 189, 3689–3699. [Google Scholar] [CrossRef] [PubMed]
- Vennekens, R.; Olausson, J.; Meissner, M.; Bloch, W.; Mathar, I.; Philipp, S.E.; Schmitz, F.; Weissgerber, P.; Nilius, B.; Flockerzi, V.; et al. Increased IGE-dependent mast cell activation and anaphylactic responses in mice lacking the calcium-activated nonselective cation channel TRPM4. Nat. Immunol. 2007, 8, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Weber, K.S.; Hildner, K.; Murphy, K.M.; Allen, P.M. TRPM4 differentially regulates th1 and TH2 function by altering calcium signaling and NFAT localization. J. Immunol. 2010, 185, 2836–2846. [Google Scholar] [CrossRef] [PubMed]
- Barbet, G.; Demion, M.; Moura, I.C.; Serafini, N.; Leger, T.; Vrtovsnik, F.; Monteiro, R.C.; Guinamard, R.; Kinet, J.P.; Launay, P. The calcium-activated nonselective cation channel TRPM4 is essential for the migration but not the maturation of dendritic cells. Nat. Immunol. 2008, 9, 1148–1156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moss, R.B.; Bocian, R.C.; Hsu, Y.P.; Dong, Y.J.; Kemna, M.; Wei, T.; Gardner, P. Reduced IL-10 secretion by Cd4+ T lymphocytes expressing mutant cystic fibrosis transmembrane conductance regulator (CFTR). Clin. Exp. Immunol. 1996, 106, 374–388. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Yang, Y.; Hu, S.G.; Zhang, Q.B.; Yu, J.; Zhang, Y.M. T-lymphocyte KV1.3 channel activation triggers the NLRP3 inflammasome signaling pathway in hypertensive patients. Exp. Ther. Med. 2017, 14, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Stevens, A.; Yuasa, M.; Peckham, D.; Olsen, C.; Iadonato, S.; Probst, P. Thu0285 Dalazatide, an Inhibitor of the KV1.3 Channel on Activated Effector Memory T Cells, Has Immunotherapy Potential in Systemic Lupus Erythematosus; BMJ Publishing Group Ltd.: London, UK, 2016. [Google Scholar]
- Tarcha, E.J.; Olsen, C.M.; Probst, P.; Peckham, D.; Munoz-Elias, E.J.; Kruger, J.G.; Iadonato, S.P. Safety and pharmacodynamics of dalazatide, a KV1.3 channel inhibitor, in the treatment of plaque psoriasis: A randomized phase 1b trial. PLoS ONE 2017, 12, e0180762. [Google Scholar] [CrossRef] [PubMed]
- Grgic, I.; Wulff, H.; Eichler, I.; Flothmann, C.; Kohler, R.; Hoyer, J. Blockade of T-lymphocyte KCa3.1 and KV1.3 channels as novel immunosuppression strategy to prevent kidney allograft rejection. Transplant. Proc. 2009, 41, 2601–2606. [Google Scholar] [CrossRef] [PubMed]
- Grgic, I.; Eichler, I.; Heinau, P.; Si, H.; Brakemeier, S.; Hoyer, J.; Kohler, R. Selective blockade of the intermediate-conductance Ca2+-activated K+ channel suppresses proliferation of microvascular and macrovascular endothelial cells and angiogenesis in vivo. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 704–709. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.C.; Lunn, C.A.; Murgolo, N.J. KCa3.1: Target and marker for cancer, autoimmune disorder and vascular inflammation? Expert Rev. Mol. Diagn. 2008, 8, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Toyama, K.; Wulff, H.; Chandy, K.G.; Azam, P.; Raman, G.; Saito, T.; Fujiwara, Y.; Mattson, D.L.; Das, S.; Melvin, J.E.; et al. The intermediate-conductance calcium-activated potassium channel KCa3.1 contributes to atherogenesis in mice and humans. J. Clin. Investig. 2008, 118, 3025–3037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buisman, H.P.; Steinberg, T.H.; Fischbarg, J.; Silverstein, S.C.; Vogelzang, S.A.; Ince, C.; Ypey, D.L.; Leijh, P.C. Extracellular ATP induces a large nonselective conductance in macrophage plasma membranes. Proc. Natl. Acad. Sci. USA 1988, 85, 7988–7992. [Google Scholar] [CrossRef] [PubMed]
- Singh, C.R.; Moulton, R.A.; Armitige, L.Y.; Bidani, A.; Snuggs, M.; Dhandayuthapani, S.; Hunter, R.L.; Jagannath, C. Processing and presentation of a mycobacterial antigen 85b epitope by murine macrophages is dependent on the phagosomal acquisition of vacuolar proton ATPase and in situ activation of cathepsin D. J. Immunol. 2006, 177, 3250–3259. [Google Scholar] [CrossRef] [PubMed]
- Rahgozar, K.; Wright, E.; Carrithers, L.M.; Carrithers, M.D. Mediation of protection and recovery from experimental autoimmune encephalomyelitis by macrophages expressing the human voltage-gated sodium channel nav1.5. J. Neuropathol. Exp. Neurol. 2013, 72, 489–504. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Salao, K.; Li, H.; Rybicka, J.M.; Yates, R.M.; Luo, X.W.; Shi, X.X.; Kuffner, T.; Tsai, V.W.; Husaini, Y.; et al. Intracellular chloride channel protein clic1 regulates macrophage function through modulation of phagosomal acidification. J. Cell Sci. 2012, 125, 5479–5488. [Google Scholar] [CrossRef] [PubMed]
- Medic, N.; Desai, A.; Olivera, A.; Abramowitz, J.; Birnbaumer, L.; Beaven, M.A.; Gilfillan, A.M.; Metcalfe, D.D. Knockout of the TRPC1 gene reveals that TRPC1 can promote recovery from anaphylaxis by negatively regulating mast cell TNF-alpha production. Cell Calcium 2013, 53, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.A.; Brown, E.E.; Williams, A.H.; Ramos, P.S.; Berthier, C.C.; Bhangale, T.; Alarcon-Riquelme, M.E.; Behrens, T.W.; Criswell, L.A.; Graham, D.C.; et al. Lupus nephritis susceptibility loci in women with systemic lupus erythematosus. J. Am. Soc. Nephrol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Nilius, B.; Owsianik, G. The transient receptor potential family of ion channels. Genome Biol. 2011, 12, 218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Freichel, M.; Almering, J.; Tsvilovskyy, V. The role of TRP proteins in mast cells. Front. Immunol. 2012, 3, 150. [Google Scholar] [CrossRef] [PubMed]
- Montell, C.; Rubin, G.M. Molecular characterization of the drosophila TRP locus: A putative integral membrane protein required for phototransduction. Neuron 1989, 2, 1313–1323. [Google Scholar] [CrossRef]
- Dietrich, A.; Chubanov, V.; Kalwa, H.; Rost, B.R.; Gudermann, T. Cation channels of the transient receptor potential superfamily: Their role in physiological and pathophysiological processes of smooth muscle cells. Pharmacol. Ther. 2006, 112, 744–760. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, A.; Fahlbusch, M.; Gudermann, T. Classical transient receptor potential 1 (TRPC1): Channel or channel regulator? Cells 2014, 3, 939–962. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Zeng, W.; Yuan, J.P.; Shin, D.M.; Worley, P.F.; Muallem, S. Native store-operated Ca2+ influx requires the channel function of orai1 and TRPC1. J. Biol. Chem. 2009, 284, 9733–9741. [Google Scholar] [CrossRef] [PubMed]
- Ambudkar, I.S.; Ong, H.L.; Liu, X.; Bandyopadhyay, B.; Cheng, K.T. TRPC1: The link between functionally distinct store-operated calcium channels. Cell Calcium 2007, 42, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Storch, U.; Forst, A.L.; Philipp, M.; Gudermann, T.; Mederos y Schnitzler, M. Transient receptor potential channel 1 (TRPC1) reduces calcium permeability in heteromeric channel complexes. J. Biol. Chem. 2012, 287, 3530–3540. [Google Scholar] [CrossRef] [PubMed]
- Vassilieva, I.O.; Tomilin, V.N.; Marakhova, I.I.; Shatrova, A.N.; Negulyaev, Y.A.; Semenova, S.B. Expression of transient receptor potential vanilloid channels TRPV5 and TRPV6 in human blood lymphocytes and Jurkat leukemia T cells. J. Membr. Biol. 2013, 246, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.T.; Peng, Z.; Hiragun, T.; Iwaki, S.; Gilfillan, A.M.; Beaven, M.A. Canonical transient receptor potential 5 channel in conjunction with orai1 and stim1 allows Sr2+ entry, optimal influx of Ca2+, and degranulation in a rat mast cell line. J. Immunol. 2008, 180, 2233–2239. [Google Scholar] [CrossRef] [PubMed]
- Van Abel, M.; Hoenderop, J.G.; Bindels, R.J. The epithelial calcium channels TRPV5 and TRPV6: Regulation and implications for disease. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2005, 371, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Soboloff, J.; Spassova, M.; Xu, W.; He, L.P.; Cuesta, N.; Gill, D.L. Role of endogenous TRPC6 channels in Ca2+ signal generation in A7R5 smooth muscle cells. J. Biol. Chem. 2005, 280, 39786–39794. [Google Scholar] [CrossRef] [PubMed]
- Guilbert, A.; Dhennin-Duthille, I.; Hiani, Y.E.; Haren, N.; Khorsi, H.; Sevestre, H.; Ahidouch, A.; Ouadid-Ahidouch, H. Expression of TRPC6 channels in human epithelial breast cancer cells. BMC Cancer 2008, 8, 125. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.L.; Liu, B.C.; Lu, X.Y.; Yan, Y.; Zhai, Y.J.; Bao, Q.; Doetsch, P.W.; Deng, X.; Thai, T.L.; Alli, A.A.; et al. Inhibition of TRPC6 reduces non-small cell lung cancer cell proliferation and invasion. Oncotarget 2017, 8, 5123–5134. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Liang, C.; Chen, E.; Chen, W.; Liang, F.; Zhi, X.; Wei, T.; Xue, F.; Li, G.; Yang, Q.; et al. Regulation of multi-drug resistance in hepatocellular carcinoma cells is TRPC6/calcium dependent. Sci. Rep. 2016, 6, 23269. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Li, X.; Liu, J.; Li, J.; Li, L.J.; Qiu, M.X. Effects of TRPC6 on invasibility of low-differentiated prostate cancer cells. Asian Pac. J. Trop. Med. 2014, 7, 44–47. [Google Scholar] [CrossRef]
- Zhang, S.S.; Wen, J.; Yang, F.; Cai, X.L.; Yang, H.; Luo, K.J.; Liu, Q.W.; Hu, R.G.; Xie, X.; Huang, Q.Y.; et al. High expression of transient potential receptor C6 correlated with poor prognosis in patients with esophageal squamous cell carcinoma. Med. Oncol. 2013, 30, 607. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Wang, Y.; Li, X.; Shen, Y.; Yin, M.; Guo, Y.; Diao, L.; Liu, Y.; Yue, D. Critical role of TRPC6 channels in the development of human renal cell carcinoma. Mol. Biol. Rep. 2013, 40, 5115–5122. [Google Scholar] [CrossRef] [PubMed]
- Amin, M.R.; Piplani, H.; Sharma, T.; Mehta, D. Switching off TRPC6 signaling: A new anti-edemagenic strategy. FASEB J. 2017, 31, 676.4. [Google Scholar]
- Chen, W.; Oberwinkler, H.; Werner, F.; Gassner, B.; Nakagawa, H.; Feil, R.; Hofmann, F.; Schlossmann, J.; Dietrich, A.; Gudermann, T.; et al. Atrial natriuretic peptide-mediated inhibition of microcirculatory endothelial Ca2+ and permeability response to histamine involves CGMP-dependent protein kinase I and TRPC6 channels. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 2121–2129. [Google Scholar] [CrossRef] [PubMed]
- Tauseef, M.; Knezevic, N.; Chava, K.R.; Smith, M.; Sukriti, S.; Gianaris, N.; Obukhov, A.G.; Vogel, S.M.; Schraufnagel, D.E.; Dietrich, A.; et al. TLR4 activation of TRPC6-dependent calcium signaling mediates endotoxin-induced lung vascular permeability and inflammation. J. Exp. Med. 2012, 209, 1953–1968. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finney-Hayward, T.K.; Popa, M.O.; Bahra, P.; Li, S.; Poll, C.T.; Gosling, M.; Nicholson, A.G.; Russell, R.E.; Kon, O.M.; Jarai, G.; et al. Expression of transient receptor potential c6 channels in human lung macrophages. Am. J. Respir. Cell Mol. Biol. 2010, 43, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, C.; Hichami, A.; Andreoletti, P.; Cherkaoui-Malki, M.; del Mar Cavia, M.; Abdoul-Azize, S.; Alonso-Torre, S.R.; Khan, N.A. Diacylglycerol-containing oleic acid induces increases in [Ca(2+)](i) via TRPC3/6 channels in human T-cells. Biochim. Biophys. Acta 2012, 1821, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.-Y.; Sun, M.-R.; Wu, C.-L.; Li, Y.; Du, J.-J.; Zeng, J.-Y.; Bi, H.-L.; Sun, Y.-H. Activation of calcium-sensing receptor increases TRPC3/6 expression in T lymphocyte in sepsis. Mol. Immunol. 2015, 64, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Sel, S.; Rost, B.R.; Yildirim, A.O.; Sel, B.; Kalwa, H.; Fehrenbach, H.; Renz, H.; Gudermann, T.; Dietrich, A. Loss of classical transient receptor potential 6 channel reduces allergic airway response. Clin. Exp. Allergy 2008, 38, 1548–1558. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Qin, W.; Zhang, L.; Wu, X.; Du, N.; Hu, Y.; Li, X.; Shen, N.; Xiao, D.; Zhang, H.; et al. Microrna-26a prevents endothelial cell apoptosis by directly targeting TRPC6 in the setting of atherosclerosis. Sci. Rep. 2015, 5, 9401. [Google Scholar] [CrossRef] [PubMed]
- Bouts, Y.M.; Wolthuis, D.F.; Dirkx, M.F.; Pieterse, E.; Simons, E.M.; van Boekel, A.M.; Dieker, J.W.; van der Vlag, J. Apoptosis and net formation in the pathogenesis of SLE. Autoimmunity 2012, 45, 597–601. [Google Scholar] [CrossRef] [PubMed]
- Dieker, J.; Tel, J.; Pieterse, E.; Thielen, A.; Rother, N.; Bakker, M.; Fransen, J.; Dijkman, H.B.; Berden, J.H.; de Vries, J.M.; et al. Circulating apoptotic microparticles in systemic lupus erythematosus patients drive the activation of dendritic cell subsets and prime neutrophils for netosis. Arthritis Rheumatol. 2016, 68, 462–472. [Google Scholar] [CrossRef] [PubMed]
- Souliotis, V.L.; Sfikakis, P.P. Increased DNA double-strand breaks and enhanced apoptosis in patients with lupus nephritis. Lupus 2015, 24, 804–815. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.C.; Lai, N.S.; Yu, H.C.; Hsieh, S.C.; Tung, C.H.; Yu, C.L. Nifedipine suppresses TH1/TH2 cytokine production and increased apoptosis of anti-Cd3 + anti-Cd28-activated mononuclear cells from patients with systemic lupus erythematosus via calcineurin pathway. Clin. Immunol. 2008, 129, 462–470. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, G.; Mannhalter, C. Increased expression of transient receptor potential canonical 6 (TRPC6) in differentiating human megakaryocytes. Cell Biol. Int. 2016, 40, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Vemana, H.P.; Karim, Z.A.; Conlon, C.; Khasawneh, F.T. A critical role for the transient receptor potential channel type 6 in human platelet activation. PLoS ONE 2015, 10, e0125764. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, E.V.P.; Murad, J.P.; Ting, H.J.; Khasawneh, F.T. Mouse transient receptor potential channel 6: Role in hemostasis and thrombogenesis. Biochem. Biophys. Res. Commun. 2012, 417, 853–856. [Google Scholar] [CrossRef] [PubMed]
- Semple, J.W.; Italiano, J.E., Jr.; Freedman, J. Platelets and the immune continuum. Nat. Rev. Immunol. 2011, 11, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Nurden, A.T. Platelets, inflammation and tissue regeneration. Thromb. Haemost. 2011, 105, S13–S33. [Google Scholar] [CrossRef] [PubMed]
- Maugeri, N.; Baldini, M.; Rovere-Querini, P.; Maseri, A.; Sabbadini, M.G.; Manfredi, A.A. Leukocyte and platelet activation in patients with giant cell arteritis and polymyalgia rheumatica: A clue to thromboembolic risks? Autoimmunity 2009, 42, 386–388. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, G.A.; Rovere-Querini, P.; Sabbadini, M.G.; Manfredi, A.A. Parietal and intravascular innate mechanisms of vascular inflammation. Arthritis Res. Ther. 2015, 17, 16. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Cassatella, M.A.; Costantini, C.; Jaillon, S. Neutrophils in the activation and regulation of innate and adaptive immunity. Nat. Rev. Immunol. 2011, 11, 519–531. [Google Scholar] [CrossRef] [PubMed]
- Scherlinger, M.; Guillotin, V.; Truchetet, M.E.; Contin-Bordes, C.; Sisirak, V.; Duffau, P.; Lazaro, E.; Richez, C.; Blanco, P. Systemic lupus erythematosus and systemic sclerosis: All roads lead to platelets. Autoimmun. Rev. 2018, 17, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Weissmann, N.; Sydykov, A.; Kalwa, H.; Storch, U.; Fuchs, B.; Mederos y Schnitzler, M.; Brandes, R.P.; Grimminger, F.; Meissner, M.; Freichel, M.; et al. Activation of TRPC6 channels is essential for lung ischaemia-reperfusion induced oedema in mice. Nat. Commun. 2012, 3, 649. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malczyk, M.; Erb, A.; Veith, C.; Ghofrani, H.A.; Schermuly, R.T.; Gudermann, T.; Dietrich, A.; Weissmann, N.; Sydykov, A. The role of transient receptor potential channel 6 channels in the pulmonary vasculature. Front. Immunol. 2017, 8, 707. [Google Scholar] [CrossRef] [PubMed]
- Urban, N.; Hill, K.; Wang, L.; Kuebler, W.M.; Schaefer, M. Novel pharmacological TRPC inhibitors block hypoxia-induced vasoconstriction. Cell Calcium 2012, 51, 194–206. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Wang, J.; Shimoda, L.A.; Sylvester, J.T. Differences in STIM1 and TRPC expression in proximal and distal pulmonary arterial smooth muscle are associated with differences in Ca2+ responses to hypoxia. Am. J. Physiol. Lung Cell. Mol. Physiol. 2008, 295, L104–L113. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Shimoda, L.A.; Weigand, L.; Wang, W.; Sun, D.; Sylvester, J.T. Acute hypoxia increases intracellular [Ca2+] in pulmonary arterial smooth muscle by enhancing capacitative Ca2+ entry. Am. J. Physiol. Lung Cell. Mol. Physiol. 2005, 288, L1059–L1069. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Strotmann, R.; Schultz, G.; Plant, T.D. TRPC6 is a candidate channel involved in receptor-stimulated cation currents in A7R5 smooth muscle cells. Am. J. Physiol. Cell Physiol. 2002, 282, C347–C359. [Google Scholar] [CrossRef] [PubMed]
- Erac, Y.; Selli, C.; Kosova, B.; Akcali, K.C.; Tosun, M. Expression levels of TRPC1 and TRPC6 ion channels are reciprocally altered in aging rat aorta: Implications for age-related vasospastic disorders. Age 2010, 32, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Chen, X.; Riley, A.M.; Hiett, S.C.; Temm, C.J.; Beli, E.; Long, X.; Chakraborty, S.; Alloosh, M.; White, F.A.; et al. Long-term spironolactone treatment reduces coronary TRPC expression, vasoconstriction, and atherosclerosis in metabolic syndrome pigs. Basic Res. Cardiol. 2017, 112, 54. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Li, S.; Liu, B.; Susperreguy, S.; Formoso, K.; Yao, J.; Kang, J.; Shi, A.; Birnbaumer, L.; Liao, Y. Major contribution of the 3/6/7 class of TRPC channels to myocardial ischemia/reperfusion and cellular hypoxia/reoxygenation injuries. Proc. Natl. Acad. Sci. USA 2017, 114, E4582–E4591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, R.; Hang, P.; Zhu, W.; Su, Z.; Liang, H.; Du, Z. Whole genome network analysis of ion channels and connexins in myocardial infarction. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2011, 27, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Kuwahara, K.; Wang, Y.; McAnally, J.; Richardson, J.A.; Bassel-Duby, R.; Hill, J.A.; Olson, E.N. TRPC6 fulfills a calcineurin signaling circuit during pathologic cardiac remodeling. J. Clin. Investig. 2006, 116, 3114–3126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eder, P. Cardiac remodeling and disease: SOCE and TRPC signaling in cardiac pathology. Adv. Exp. Med. Biol. 2017, 993, 505–521. [Google Scholar] [PubMed]
- Shen, B.; He, Y.; Zhou, S.; Zhao, H.; Mei, M.; Wu, X. TRPC6 may protect renal ischemia-reperfusion injury through inhibiting necroptosis of renal tubular epithelial cells. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2016, 22, 633. [Google Scholar] [CrossRef]
- Li, W.; Ding, Y.; Smedley, C.; Wang, Y.; Chaudhari, S.; Birnbaumer, L.; Ma, R. Increased glomerular filtration rate and impaired contractile function of mesangial cells in TRPC6 knockout mice. Sci. Rep. 2017, 7, 4145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Graham, S.; Gorin, Y.; Abboud, H.E.; Ding, M.; Lee, D.Y.; Shi, H.; Ding, Y.; Ma, R. Abundance of TRPC6 protein in glomerular mesangial cells is decreased by ROS and PKC in diabetes. Am. J. Physiol. Cell Physiol. 2011, 301, C304–C315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santin, S.; Ars, E.; Rossetti, S.; Salido, E.; Silva, I.; Garcia-Maset, R.; Gimenez, I.; Ruiz, P.; Mendizabal, S.; Luciano Nieto, J.; et al. TRPC6 mutational analysis in a large cohort of patients with focal segmental glomerulosclerosis. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2009, 24, 3089–3096. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Winn, M.P.; Conlon, P.J.; Lynn, K.L.; Farrington, M.K.; Creazzo, T.; Hawkins, A.F.; Daskalakis, N.; Kwan, S.Y.; Ebersviller, S.; Burchette, J.L.; et al. A mutation in the TRPC6 cation channel causes familial focal segmental glomerulosclerosis. Science 2005, 308, 1801–1804. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.Y.; Yazdizadeh Shotorbani, P.; Dryer, S.E. TRPC6 inactivation confers protection in a model of severe nephrosis in rats. J. Mol. Med. 2018, 96, 631–644. [Google Scholar] [CrossRef] [PubMed]
- Krall, P.; Canales, C.P.; Kairath, P.; Carmona-Mora, P.; Molina, J.; Carpio, J.D.; Ruiz, P.; Mezzano, S.A.; Li, J.; Wei, C.; et al. Podocyte-specific overexpression of wild type or mutant TRPC6 in mice is sufficient to cause glomerular disease. PLoS ONE 2010, 5, e12859. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, M.; Bringhenti, R.N.; Rodrigues, P.G.; do Nascimento, J.F.; Pereira, S.V.; Zancan, R.; Monticielo, O.A.; Gasparin, A.A.; de Castro, W.P.; Veronese, F.V. Podocyte-associated mRNA profiles in kidney tissue and in urine of patients with active lupus nephritis. Int. J. Clin. Exp. Pathol. 2015, 8, 4600–4613. [Google Scholar] [PubMed]
- Wu, Y.L.; Xie, J.; An, S.W.; Oliver, N.; Barrezueta, N.X.; Lin, M.H.; Birnbaumer, L.; Huang, C.L. Inhibition of TRPC6 channels ameliorates renal fibrosis and contributes to renal protection by soluble klotho. Kidney Int. 2017, 91, 830–841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davis, J.; Burr, A.R.; Davis, G.F.; Birnbaumer, L.; Molkentin, J.D. A TRPC6-dependent pathway for myofibroblast transdifferentiation and wound healing in vivo. Dev. Cell 2012, 23, 705–715. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, K.; Fiedler, S.; Vierkotten, S.; Weber, J.; Klee, S.; Jia, J.; Zwickenpflug, W.; Flockerzi, V.; Storch, U.; Yildirim, A.O.; et al. Classical transient receptor potential 6 (TRPC6) channels support myofibroblast differentiation and development of experimental pulmonary fibrosis. Biochim. Biophys. Acta 2017, 1863, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Kurahara, L.H.; Sumiyoshi, M.; Aoyagi, K.; Hiraishi, K.; Nakajima, K.; Nakagawa, M.; Hu, Y.; Inoue, R. Intestinal myofibroblast TRPC6 channel may contribute to stenotic fibrosis in crohn’s disease. Inflamm. Bowel Dis. 2015, 21, 496–506. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Huang, J.; Yao, H.; Zhou, K.; Duan, B.; Wang, Y. Inhibition of TRPC6 degradation suppresses ischemic brain damage in rats. J. Clin. Investig. 2010, 120, 3480–3492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.; Huang, J.; Du, W.; Jia, C.; Yao, H.; Wang, Y. TRPC6 inhibited NMDA receptor activities and protected neurons from ischemic excitotoxicity. J. Neurochem. 2012, 123, 1010–1018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Mao, X.; Zhou, T.; Cheng, X.; Lin, Y. IL-17a contributes to brain ischemia reperfusion injury through calpain-TRPC6 pathway in mice. Neuroscience 2014, 274, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Griesi-Oliveira, K.; Acab, A.; Gupta, A.R.; Sunaga, D.Y.; Chailangkarn, T.; Nicol, X.; Nunez, Y.; Walker, M.F.; Murdoch, J.D.; Sanders, S.J.; et al. Modeling non-syndromic autism and the impact of TRPC6 disruption in human neurons. Mol. Psychiatry 2015, 20, 1350–1365. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Sun, S.; Wu, L.; Pchitskaya, E.; Zakharova, O.; Fon Tacer, K.; Bezprozvanny, I. Store-operated calcium channel complex in postsynaptic spines: A new therapeutic target for Alzheimer’s disease treatment. J. Neurosci. Off. J. Soc. Neurosci. 2016, 36, 11837–11850. [Google Scholar] [CrossRef] [PubMed]

| 1. Modulation of Calcium Currents | |||||
| 1.1 Through Direct Involvement in Calcium Influx/Efflux | |||||
| 1.1.1 SOCE | |||||
| Channel | Permeability | Expression (immune cells) | Biological effects | Clinical correlates | Ref. |
| ORAI1 | Ca2+ | Neutrophils, Lymphocytes | Neutrophils: proliferation, degranulation, cytokines production, cell polarization, migrational guidance with LFA1. Lymphocytes: B, T and NK cell proliferation, cytokine production and/or cytotoxicity in vitro; immunity to infection, T cell-mediated autoimmunity and inflammation, and allogeneic T cell responses in vivo; Treg cell development | CRAC channelopathywith immunodeficiency, autoimmunity, lymphoproliferation, muscular hypotonia and ectodermal dysplasia caused by mutations in STIM1 and ORAI1 | [10,27,60,61] |
| ORAI2/3 | Ca2+ | Neutrophils, Lymphocytes | Cell proliferation, Cytokines production | ND | |
| STIM1 | NA | Neutrophils, Lymphocytes, DC, mast cells | Neutrophils: phagocytosis and ROS production Lymphocytes: cytokine production in T and B cells, Treg functionality Mast cells: FcεR-triggered SOCE | ND | [13,14,27,62,63,64] |
| STIM2 | NA | Mice deficient of STIM1/2 develop a lymphoproliferative disorder because of dysfunction of Treg cells. | |||
| IP3Rs | Ca2+ | All cells | Physiological development of B and T cells | ND | [16,17,18,19] |
| TRPC1 | Ca2+, Na+ | Endothelium | Enhanced vascular permeability after TNF/thrombin stimulation | ND | [65,66,67] |
| TRPC6 | Ca2+, Na+ | Platelets | Dense granules secretion after thrombin stimulation | ND | [68] |
| 1.1.2 ROCE | |||||
| Channel | Permeability | Expression (immune cells) | Biological effects | Clinical correlates | Ref. |
| TRPM2 | Ca2+, Na+ | Neutrophils, lymphocytes, macrophages and DC | Neutrophils: increased activation and endothelial adhesion Lymphocytes: T cell proliferation and cytokine secretion Macrophages and dendritic cells: regulation of ROS formation | Mice lacking TRPM2 have milder ischaemia-reperfusion injury after myocardial infarction and attenuated experimental brain inflammation | [54,69,70,71,72,73,74,75] |
| TRPC3 | Ca2+, Na+ | Lymphocytes, macrophages | Lymphocytes: T cell activation downstream the TCR Macrophages: enhanced pro-inflammatory activation | Mice: accelerated atherosclerosis | [76,77,78] |
| TRPC6 | Ca2+, Na+ | Lymphocytes, neutrophils, endothelium, platelets | Lymphocytes: T cell activation Neutrophils: chemotaxis, Endothelium: enhanced endothelial permeability and activation Platelets: TXA2-dependent expression of glycoproteins IIb-IIIa and P-selectin, release of platelet dense granules | Mice: TRPC6 ko associates with milder airway hypersensitivity in asthma models Humans: single study suggesting an association between a TRPC6 polymorphism and neuropsychiatric SLE | [79,80,81,82,83,84,85] |
| TRPV4 | Ca2+, Na+ | Macrophages | Cell activation after lung barotrauma. | Mice: exacerbated lung inflammation in acute lung injury and increased inflammatory hyperalgesia | [30] |
| P2X1R, P2X4R | Ca2+, Na+ | Lymphocytes, neutrophils, eosinophils, monocytes/macrophages, mast cells, and DC | Lymphocytes: T cell proliferation; cytokine production; thymocyte apoptosis Macrophages: PGE2 release, inflammasome activation | ND | [86,87] |
| P2X7R | Ca2+, Na+, other cations | Lymphocytes: T cell survival and cytokine production (downstream the TCR); T cell differentiation into Th17 vs. Treg Macrophages: activation of the NLRP3 inflammasome Mast cells, eosinophils, DC: inflammatory activation | Mice lacking P2X7R have attenuated allergic airway response, graft vs. host disease, allograft rejection | [88,89,90] | |
| 1.1.3 VOCE | |||||
| Channel | Permeability | Expression (immune cells) | Biological effects | Clinical correlates | Ref. |
| Cav1.1-4 | Ca2+ | Lymphocytes | T cell survival, differentiation and progression to effector function | ND | [31,32] |
| 1.1.4 Direct calcium entry following upregulation | |||||
| Channel | Permeability | Expression (immune cells) | Biological effects | Clinical correlates | Ref. |
| TRPC3 | Ca2+, Na+ | Macrophages/microglia | Regulation of cellular activation | Mice: reduced brain inflammation and post-ischaemic myocardial damage | [28,91,92] |
| TRPC5 | Ca2+, Na+ | Lymphocytes | Inhibition of Teff activation by Treg | Mice: protection from experimental arthritis | [93,94] |
| TRPV1 | Ca2+, Na+ | T lymphocytes | Cell activation (by associating to TCR) | ND | [95] |
| TRPV2 | Ca2+, Na+ | Macrophages | Phagocytosis, chemotaxis, following FCγR activation | Mice: TRPV2 deletion prompts accelerated mortality in bacterial infections Humans: cystic Fibrosis macrophages exhibit a defect in TRPV2-mediated calcium influx | [51,96] |
| TRPV5,6 | Ca2+, Na+ | Lymphocytes | Cell activation and proliferation (the channels are constitutively active and regulated by endocytosis or at gene expression level). | ND | [97] |
| 1.2 Through intracellular second messengers | |||||
| Channel | Permeability | Expression (immune cells) | Biological effects | Clinical correlates | Ref. |
| TRPM7 | Mg2+, Ca2+ | Lymphocytes, macrophages, mast cells | Lymphocytes: activation downstream BCR and TCR; thymocyte development; production of thymocyte growth factor Macrophages: survival and M2 polarisation Mast cells: survival and activation | ND | [98,99,100,101,102,103] |
| MAGT1 | Mg2+ | Lymphocytes | CD4+ T cell development and activation; immunity to EBV | XMEN syndrome (X-linked mutations in MAGT1) | [104] |
| ZIP6 | Zn2+ | T cells, DC | T cells: sustained calcium currents enhancing TCR-related pathways and promoting T cell activation DC: inhibition of maturation for antigen presentation | Genetically determined zinc deficit (mutated ZIP4 in the intestinal mucosa) causes acrodermatitis enteropathica with immunodeficiency | [26] |
| ZIP8 | T cells | Sustained calcium currents enhancing TCR-related pathways and promoting T cell activation | |||
| 1.3 Through alterations of cell polarisation | |||||
| Channel | Permeability | Expression (immune cells) | Biological effects | Clinical correlates | Ref. |
| NCX1 | Ca2+, Na+ | NeutrophilsMacrophages | Neutrophils: recovery from activation Macrophages: activation, cytokine (TNF) secretion | A single association study suggests potential links among NCX polymorphisms and SLE phenotypes (including severe nephritis) | [11,43,44] |
| NKCC2 | Na+, K+, 2Cl− | Lymphocytes | Adaptation to extracellular hypertonicity, which eventually leads to the activation of the p38/MAPK → NFAT5 → SGK pathway, which favours Th17 differentiation | ND | [37] |
| ENaC | Na+ | ||||
| NHE1 | Na+, H+ | ||||
| TRPM4 | Na+, Ca2+ | Lymphocytes, macrophages and DC, mast cells | Lymphocytes: T helper motility and cytokine production (IL2, IL4, and IFNγ). Macrophages: phagocytosis and cytokine release DC: motility Mast cells: regulation of cell activation | Mice: lack of TRPM4 associates with reduced survival in sepsis and more intense anaphylaxis | [105,106,107,108] |
| GABAA-R | Cl− | Lymphocytes, macrophages and DC, neutrophils | Inhibition of cell activation | In preclinical models GABAergic drugs, protects against type 1 diabetes (T1D), experimental autoimmune encephalomyelitis (EAE), collagen-induced arthritis (CIA), contact dermatitis and allergic asthma. Treatment with gabapentin and pregabalin improved psoriasis (case report). | [49] |
| CFTR | Cl− | Lymphocytes, macrophages | Lymphocytes: modulation of cytokine secretory profile (IL5, IL10) in T cells Macrophages: cytokine release, phagocytosis | Cystic fibrosis | [51,109] |
| Kv1.3 | K+ | Lymphocytes | Enhanced activation of the NLRP3 inflammasome and of IL1β production. Enhanced cell survival and prolonged activation. | A single phase Ib study on dalazatide (a specific Kv1.3 inhibitor) shows promise. Applications in SLE have been proposed. | [110,111,112] |
| KCa3.1 | K+ | Lymphocytes, macrophages, endothelium | Lymphocytes: sustained TCR-induced calcium currents to support long-lasting effector functions. Macrophages: activation, chemotaxis, infiltration of atherosclerotic plaques Endothelium: proliferation | Encouraging evidence of efficacy of KCa3.1blockers in several models of inflammatory vasculopathy and autoimmunity. | [47,113,114,115,116] |
| Nav1.5 (SCN5A) | Na+ | T cells | Positive selection of thymocytes | ND | [46] |
| P2X7R | Ca2+, Na+ and other cations | Macrophages | Cell death for prolonged depolarisation in case of sustained receptor ligation. | ND | [117] |
| 1.4 Through alterations in the geographical distribution of intracellular calcium | |||||
| Channel | Permeability | Expression (immune cells) | Biological effects | Clinical correlates | Ref. |
| TRPC1 | Ca2+, Na+ | Neutrophils | Cell polarisation for chemotaxis | ND | [65,66,67] |
| 2. Modulation of intracellular pH and production of reactive oxygen species | |||||
| Channel | Permeability | Expression (immune cells) | Biological effects | Clinical correlates | Ref. |
| TRPM2 | Ca2+, Na+ | Macrophages and DC | Macrophages and DC: regulation of ROS formation, phagocytosis and bacterial killing | ND | [71,75] |
| Hv1/VSOP | H+ | lymphocytes, granulocytes, macrophages and DC | All cells: phagocytosis and ROS production B cells: BCR signalling | Mice: loss of the receptor prompts impaired killing of phagocytosed bacteria, ROS production and migration by leukocytes and impaired antibody responses. | [15,53] |
| NCX | Ca2+, Na+ | DC | Activation of NADPH oxidase and polarisation towards pro-inflammatory DC. | [42] | |
| ENac | Na+ | ||||
| NHE | Na+, H+ | ||||
| 3. Modulation of endosomal pH | |||||
| Channel | Permeability | Expression (immune cells) | Biological effects | Clinical correlates | Ref. |
| TRPC6 | Ca2+, Na+ | Macrophages | Phagocytosis and bacterial killing | ND | [8] |
| TRPM2 | Ca2+, Na+ | Macrophages and DC | Phagocytosis and bacterial killing | ND | [71,75] |
| Proton ATPases | H+ | Macrophages | Phagocytosis and bacterial killing | ND | [118] |
| Nav1.5 (SCN5A) | Na+ | Macrophages | endosomal acidification and phagocytosis. Possible polarisation towards an antinflammatory phenotype | Mice: enhanced recovery from EAE. | [45,119] |
| CLIC 1 | Cl− | Macrophages and DC | Phagocytosis, antigen processing and presentation. | ND | [9,120] |
| 4. Modulation of other intracellular signalling pathways | |||||
| Channel | Permeability | Expression (immune cells) | Biological effects | Clinical correlates | Ref. |
| TRPC1 | Ca2+, Na+ | Macrophages, Mast cells | Macrophages: inhibition of IL1β through other ion channels and transporters Mast-cells: inhibition of calcium-dependent release of TNF in the late phase of cell activation | Mice: delayed recovery from anaphylaxis | [77,121] |
| 5. Other effects | |||||
| Channel | Permeability | Expression (immune cells) | Biological effects | Clinical correlates | Ref. |
| SLC5A11 | Na+, glucose | Leukocytes (low) | Leukocytes: control of cell osmolarity under hypernatriemic environment, energy uptake, TNF-dependent apoptosis | Polymorphisms associated with susceptibility to SLE | [55,122] |
| CLIC 1 | Cl− | Macrophages | Modulation of cytokine gene expression and processing (conflicting results) | ND | [9,58,59] |
| CLIC 4 | Cl− | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramirez, G.A.; Coletto, L.A.; Sciorati, C.; Bozzolo, E.P.; Manunta, P.; Rovere-Querini, P.; Manfredi, A.A. Ion Channels and Transporters in Inflammation: Special Focus on TRP Channels and TRPC6. Cells 2018, 7, 70. https://doi.org/10.3390/cells7070070
Ramirez GA, Coletto LA, Sciorati C, Bozzolo EP, Manunta P, Rovere-Querini P, Manfredi AA. Ion Channels and Transporters in Inflammation: Special Focus on TRP Channels and TRPC6. Cells. 2018; 7(7):70. https://doi.org/10.3390/cells7070070
Chicago/Turabian StyleRamirez, Giuseppe A., Lavinia A. Coletto, Clara Sciorati, Enrica P. Bozzolo, Paolo Manunta, Patrizia Rovere-Querini, and Angelo A. Manfredi. 2018. "Ion Channels and Transporters in Inflammation: Special Focus on TRP Channels and TRPC6" Cells 7, no. 7: 70. https://doi.org/10.3390/cells7070070






