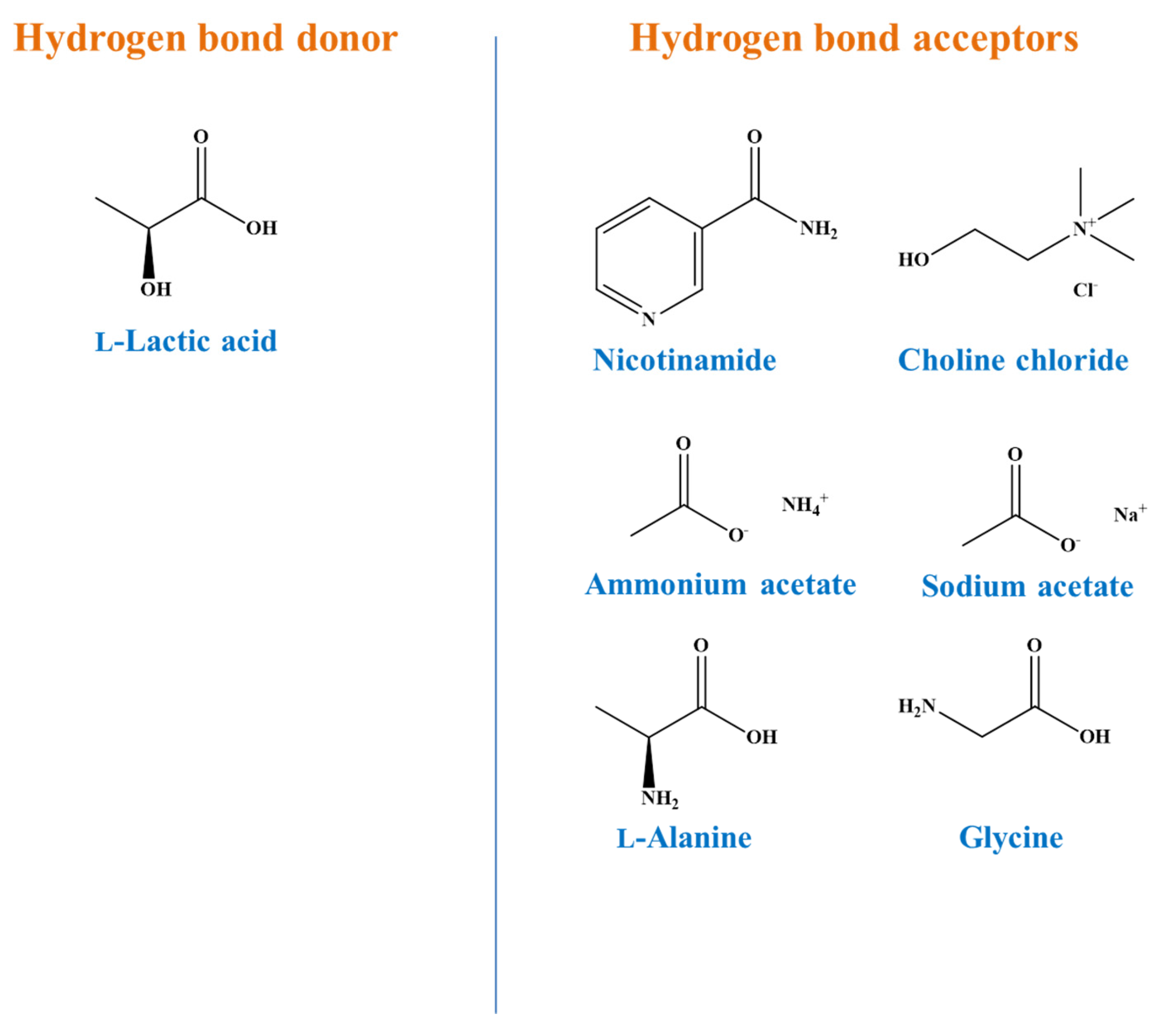
3.1. DES Synthesis
The evidence accumulated so far has shown that extraction of polyphenolic compounds from various plant sources using DES may depend significantly on the nature of DES constituents. However, recent studies on polyphenol recovery from a red grape pomace suggested
l-lactic acid-based DES to be far more efficient than those containing glycerol or 2,3-butanediol as the HBD [
17]. On the other hand, an extensive screening of
l-lactic acid-based DES with different amino-bearing HBAs indicated that the nature of HBA also plays a prominent role in the polyphenol extraction efficiency [
18]. This being the case, the investigation undertaken aimed at synthesising
l-lactic acid-based DES with highly efficient HBAs, including two bases (choline chloride and nicotinamide), two salts (sodium and ammonium acetate) and two amino acids (
l-alanine and glycine) (
Figure 1).
Judgement upon the potency of the HBAs selected was based on evidence provided by the polyphenol extraction from a few agri-food wastes [
19], botanicals [
13] and industrial cereal solid wastes [
18]. Thus, efforts were made to compose stable DES, by combining the HBD and HBAs at appropriate molar ratios (
. Although HBAs such as sodium acetate, ammonium acetate and choline chloride may readily produce stable DES at
of 1:1,
l-alanine and glycine interacted with the HBD at
higher than 6:1. To maintain identical composition, all the DES were produced using
of 7:1 (
Table 2). It is to be emphasised that the DES coded as LA-Nic, to the best of the authors’ knowledge, is reported for the first time.
3.2. Extraction Efficiency
The screening process was designed to select highly effective DES and for this purpose a simple and efficient extraction methodology was implemented, to enable simultaneous extraction of a large number of samples. The liquid-to-solid ratio (R
L/S) was set at 35 mL g
−1, which was an average level within the optimum range of 29.5 to 36.2 mL g
−1 found for polyphenol extraction from
Satureja thymbra with DES [
20]. Extraction temperature was maintained at 55 °C, because higher temperatures may not favour increased polyphenol yield and extraction time was 60 min, which according to kinetic investigations, appeared sufficiently long to prevent variances that could stem from incomplete extraction [
21]. The extraction was assisted by ultrasonication, which has been shown to promote higher extraction yields when viscous solvents are employed at moderate temperatures [
22].
Two eco-friendly solvents conventionally used for polyphenol extraction were tested as reference solvents, including water and 60% (
v/
v) aqueous ethanol. All DES were tested as 75% (
w/
v) aqueous solutions, using an average water content of those previously reported for polyphenol extraction with DES [
21,
23]. Appropriate amounts of water are considered critical for DES extraction efficiency, given the usually high DES viscosity. Water regulates effectively DES viscosity downwards, thus facilitating diffusion-controlled extraction. Moreover, water also modifies DES polarity, which in several instances has been shown to play a key role in solute (polyphenol) solubility, entailing higher extraction yield [
20].
The results of the performance screening with regard to total polyphenol yield are given in
Table 3. It was observed that in several cases DES were far more effective than water and significantly so than aqueous ethanol (
p < 0.05). Although no particular pattern was recorded regarding composition-efficiency relationships, it was noticed that LA-Alan gave statistically higher values for
Coridothymus capitatus (CC) and
Thymus vulgaris (TV) extraction, LA-Niam for CC,
Salvia fruticosa (SF) and
Salvia officinalis (SO), while LA-AmAc and LA-ChCl performed exceptionally only for
Origanum vulgare. In no case LA-Glyc and LA-SoAc gave statistically higher Y
TP. The richest extract was obtained from OV using LA-ChCl (140.72 mg CAE g
−1 dw), while the lowest performance was seen for the extraction of CC with water, which yielded 33.91 mg CAE g
−1 dw. The OV yield is similar to 151 mg CAE g
−1 dw previously reported [
24], yet as high as 288 mg sinapic acid equivalents per g dw have been found [
25].
When Y
TFn was considered as extraction performance indicator, the overall picture was profoundly different (
Table 4). In all cases examined aqueous extracts, as well as extracts obtained with aqueous ethanol were shown to give statistically higher values (
p < 0.05). LA-SoAc was the most efficient solvent, providing significantly higher Y
TFn for CC, SF and TV, while LA-Niam performed likewise for OV. By contrast, LA-Alan, LA-AmAc, LA-ChCl and LA-Glyc did not show increased efficacy for any of the specimens tested. The highest Y
TFn was found for the SF extract obtained with aqueous ethanol, amounting 30.89 mg RtE g
−1 dw, but the aqueous ethanolic extract from SO had comparable Y
TFn of 28.93 mg RtE g
−1 dw. Total flavonoid content in SO extracts was determined to be 16.89 mg quercetin equivalents per g dw [
26], but levels as low as 3.50 mg RtE g
−1 dw have also been found [
27].
3.3. The Effect of β-Cyclodextrin (β-CD) Addition
Polyphenol extraction with the aid of β-CD has been shown to affect both extraction yield and extract composition, due to selectivity of β-CD towards complexation of certain polyphenols [
28,
29]. Aqueous solutions of β-CD have been successfully used to assist polyphenol extraction from various matrices, including pomegranate [
30], tea leaves [
31], vine shoots [
32] and
Polygonum cuspidatum [
33], but combination of β-CD with DES has never been reported. In the view of clarifying whether such a combination could boost polyphenol extraction yield, extractions with the DES tested were also performed in the presence of 1.5% (
w/
v) β-CD. The amount of β-CD was carefully chosen, in the light of previous data [
30,
33].
The addition of β-CD changed drastically the overall picture of total polyphenol yield obtained from the extractions carried out only with DES (
Table 3). For CC, β-CD increased the extraction capacity of LA-AmAc by approximately 21%, which gave significantly higher Y
TP (
p < 0.05). On the contrary, LA-Alan and LA-Niam, which exhibited high performance in the absence of β-CD, showed lower efficacy. Similarly, β-CD boosted total polyphenol recovery from OV with LA-Niam, but depressed the efficiency of LA-AmAc and LA-ChCl. For the SF extraction, β-CD incorporation improved the capacity of LA-Alan and for SO improvement was seen with LA-Alan and LA-AmAc. For TV extraction, LA-Alan efficiency dropped significantly upon addition of β-CD (
p < 0.05), but LA-AmAc displayed over than 32% higher Y
TP. Regarding Y
TFn, β-CD enhanced LA-Niam performance for CC and SO, LA-AmAc for TV and LA-SoAc for OV, but depressed extraction of SF with LA-SoAc (
Table 4). Overall, TFn recovery was impacted to a rather lesser extent compared with TP.
Encapsulation of a ligand in the cavity of CD obeys a certain stoichiometry, which is usually 1:1 [
34]. In aqueous media the displacement of water outside the β-CD cavity and retention of the polyphenol within through hydrophobic association is the driving force for polyphenol-β-CD complex formation [
35]. However, polyphenols may behave as HBDs and interact with the HBA of the DES, which may give rise to some sort of antagonism, because β-CD might weaken polyphenol-HBA interactions since polyphenol/β-CD complex formation involves strong hydrogen bond formation [
36]. On the other hand, β-CD would increase solubility of the less polar polyphenols through entrapment. Therefore, the eventual outcome could represent the integration of such an effect.
3.4. Antioxidant Activity
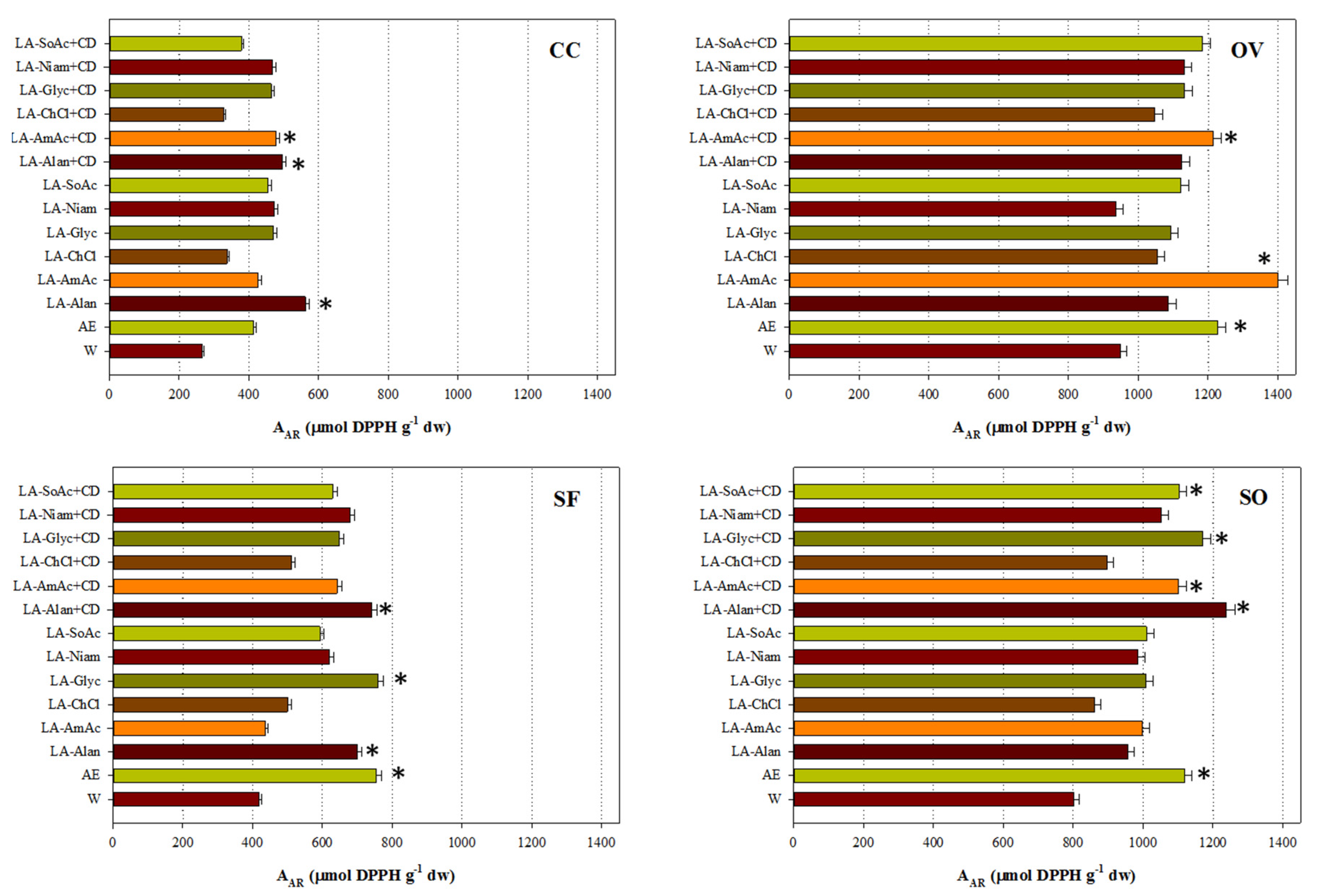
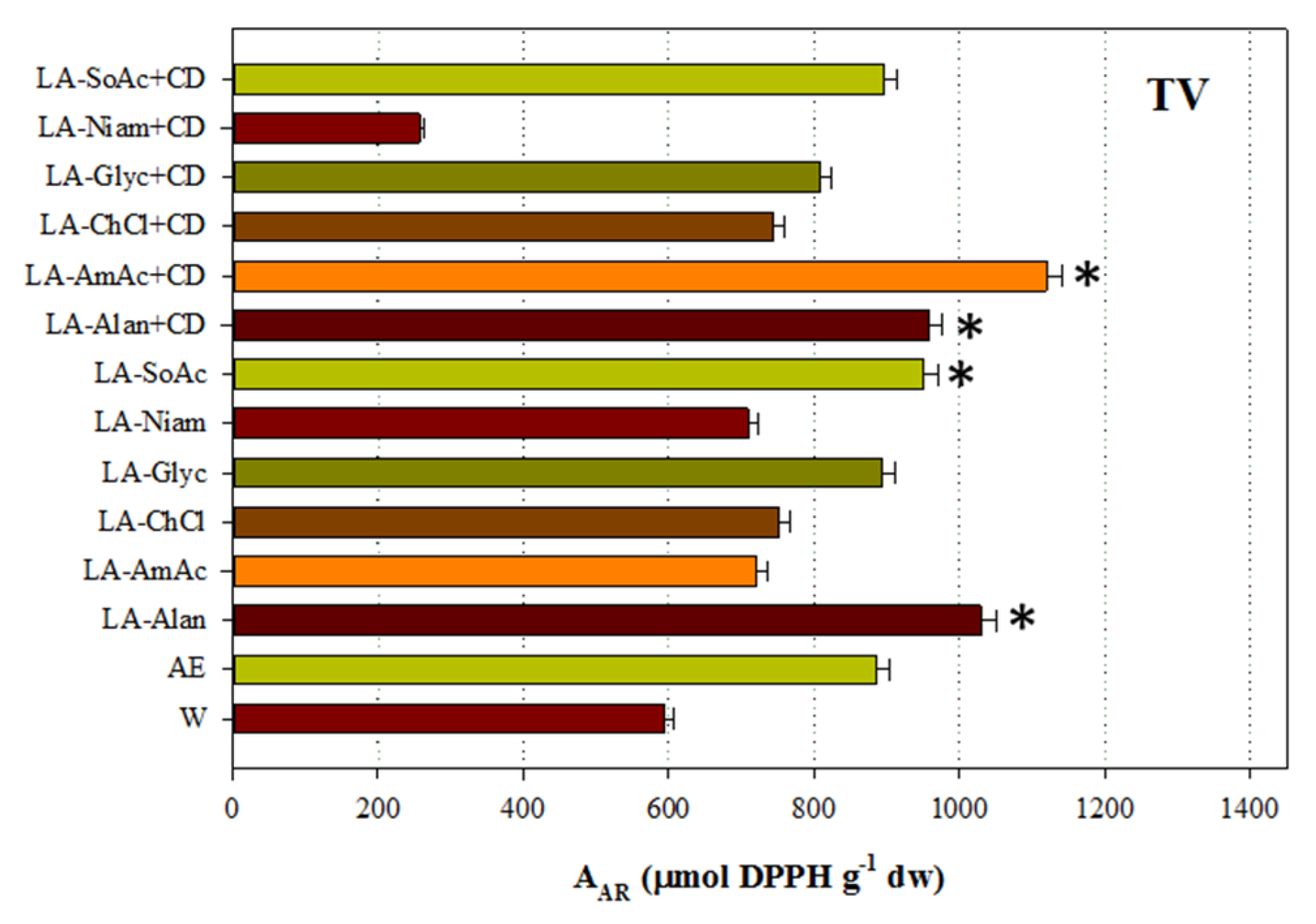
Two representative and complementary tests were carried out to assess the antioxidant activity of all the extracts generated. With the exception of SF extracts, significantly higher A
AR levels were determined for those obtained with LA-AmAc/β-CD. The same phenomenon was observed for LA-Alan/β-CD, with the exception of OV (
Figure 2). This finding highlighted the importance of these extraction media in obtaining extracts with strong antiradical effects. It should also be stressed that in the cases of CC, SF and TV, LA-Alan was also very effective in providing extracts with significantly higher A
AR (
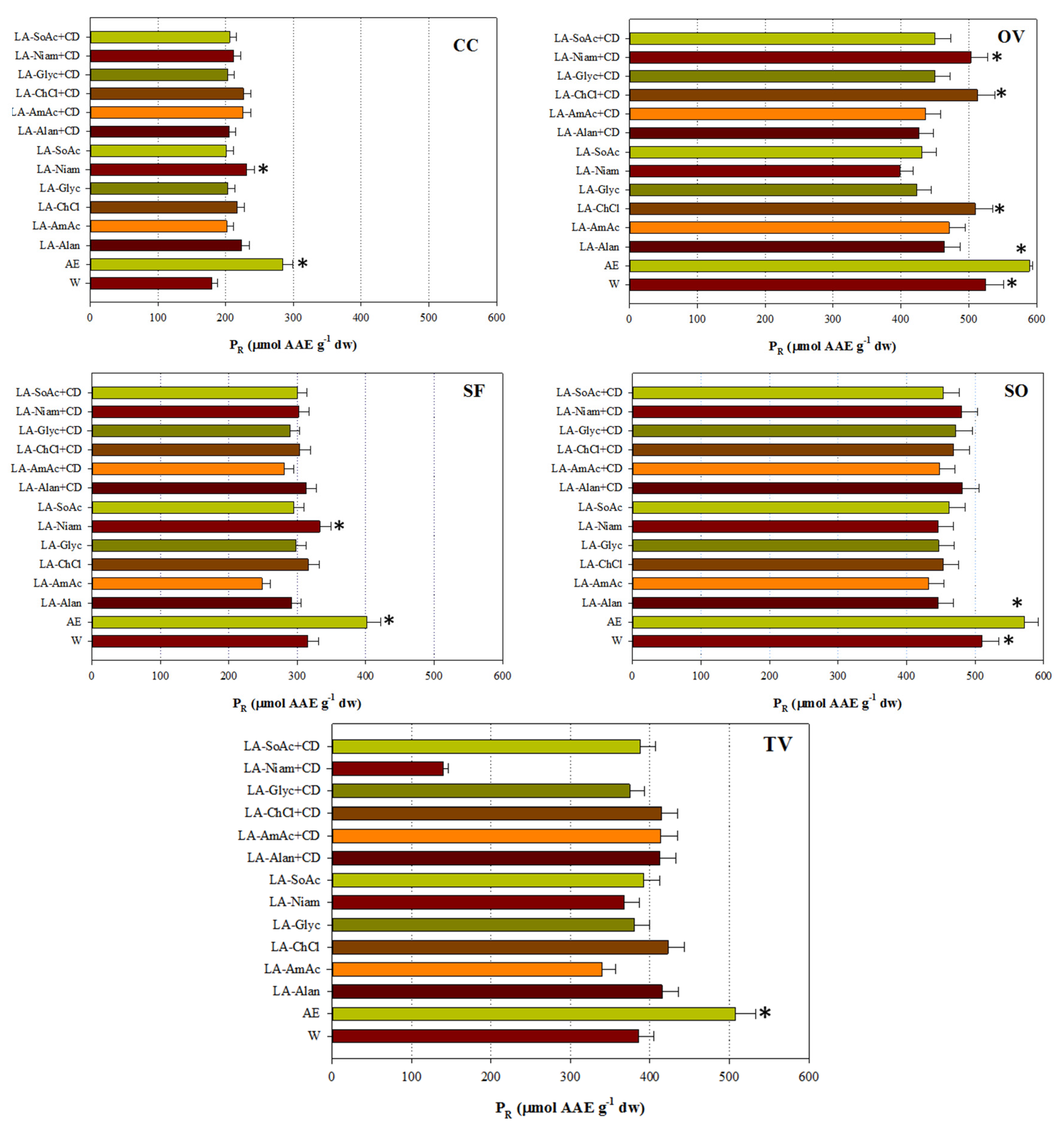
p < 0.05). By contrast, exceptionally high P
R was displayed mainly by the extracts obtained with aqueous ethanol (
Figure 3), and no consistency with A
AR was found.
The role of β-CD in the expression of antioxidant effects should not be overlooked, because it has been demonstrated that β-CD complexes with rosmarinic acid [
37], quercetin and rutin [
38], but also hydroxypropyl β-CD complexes with polyphenols such as chlorogenic acid [
39] and quercetin [
36], showed significantly higher antioxidant activity compared with the non-encapsulated polyphenols. This phenomenon has also been demonstrated for chlorogenate-rich coffee extracts [
40]. The orientation of the encapsulated molecule in the β-CD cavity is probably critical in this respect. It has been shown that complex of β-CD with oleuropein from olive leaf extracts involves deep insertion of the dihydroxyphenethyl moiety inside the cavity from its secondary side [
41]. This could affect oxidant/antioxidant interactions and therefore, the antioxidant effects observed in DES/β-CD extracts may not be simply attributed to higher polyphenol concentration, but to interactions associated with polyphenol/β-CD complexes as well.
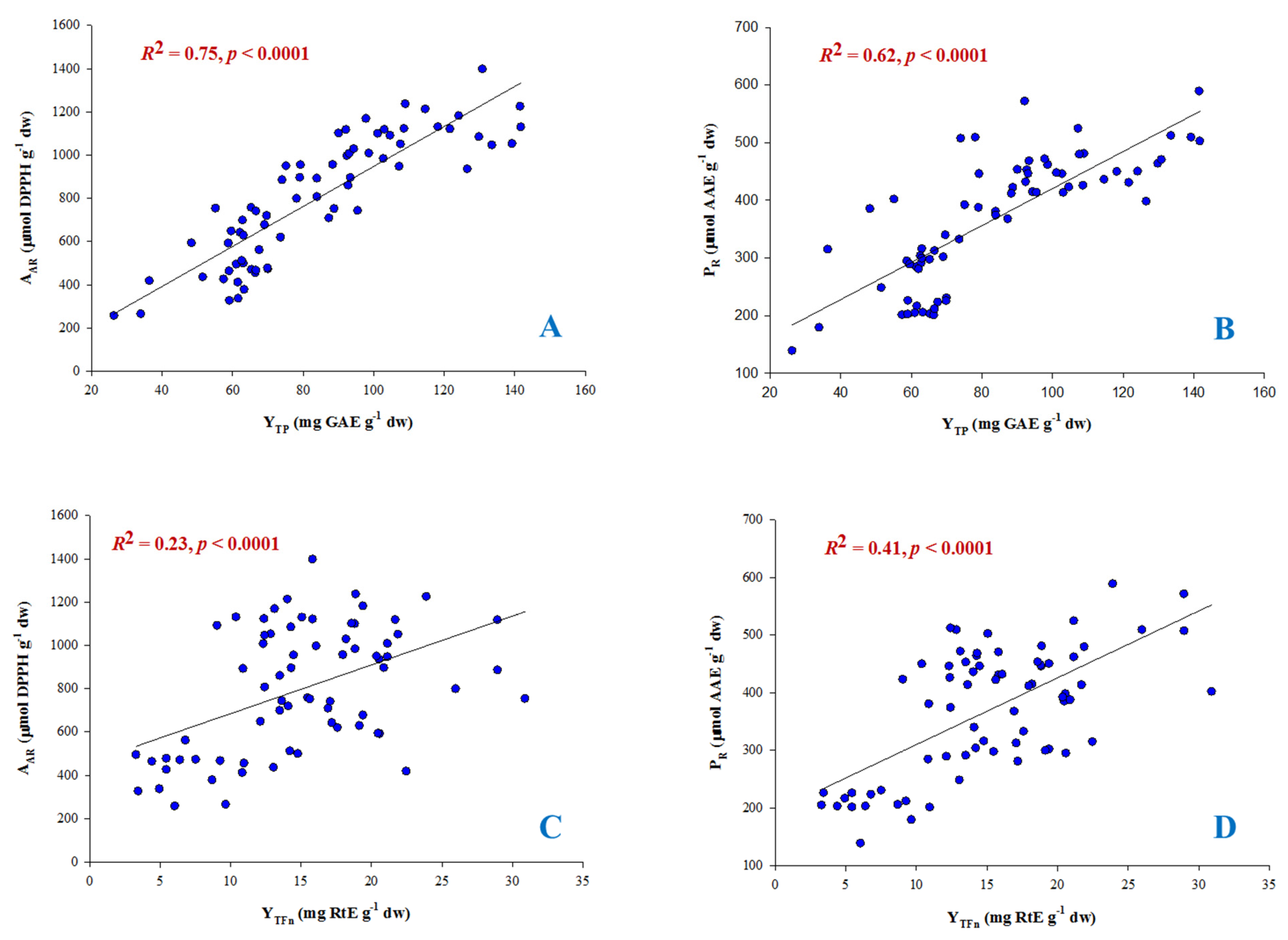
In
Figure 4 it can be seen that the correlation of Y
TP with both P
R and A
AR was highly significant (
p < 0.0001) and the same held true for Y
TFn (
p < 0.0001). However,
R2 was notably higher for correlations with Y
TP. This outcome highlighted the importance of total polyphenol concentration for the antioxidant activity of the extracts. Similar outcome was seen for various agri-food waste extracts [
19], but also botanicals [
13]. Nevertheless, mixture effects that can be manifested either as synergism or antagonism among the various polyphenols occurring in the extracts should also be considered. Such phenomena were demonstrated for A
AR in mixtures of antioxidants [
42] and mixtures of antioxidants with plant extracts [
43]. Similar mixture effects for P
R have also been found [
44].