Assessing the Consequences of Microbial Infection in Field Trials: Seen, Unseen, Beneficial, Parasitic and Pathogenic
Abstract
:1. Introduction
2. Visual Disease Assessment
2.1. Field Scoring Visible Disease Symptoms
2.2. Physiological and Environmental Interactions with Symptom Expression
3. Molecular Detection and Quantification of Crop Pathogens
4. Measurements of Host Response to Stress
5. “Non-Pathogenic” Microbe Interactions, with Pathogens, Each Other, and with Their Host
6. End-User Consequences of Microbial Infection
7. Yield Loss Implications of Different Microbial Infections
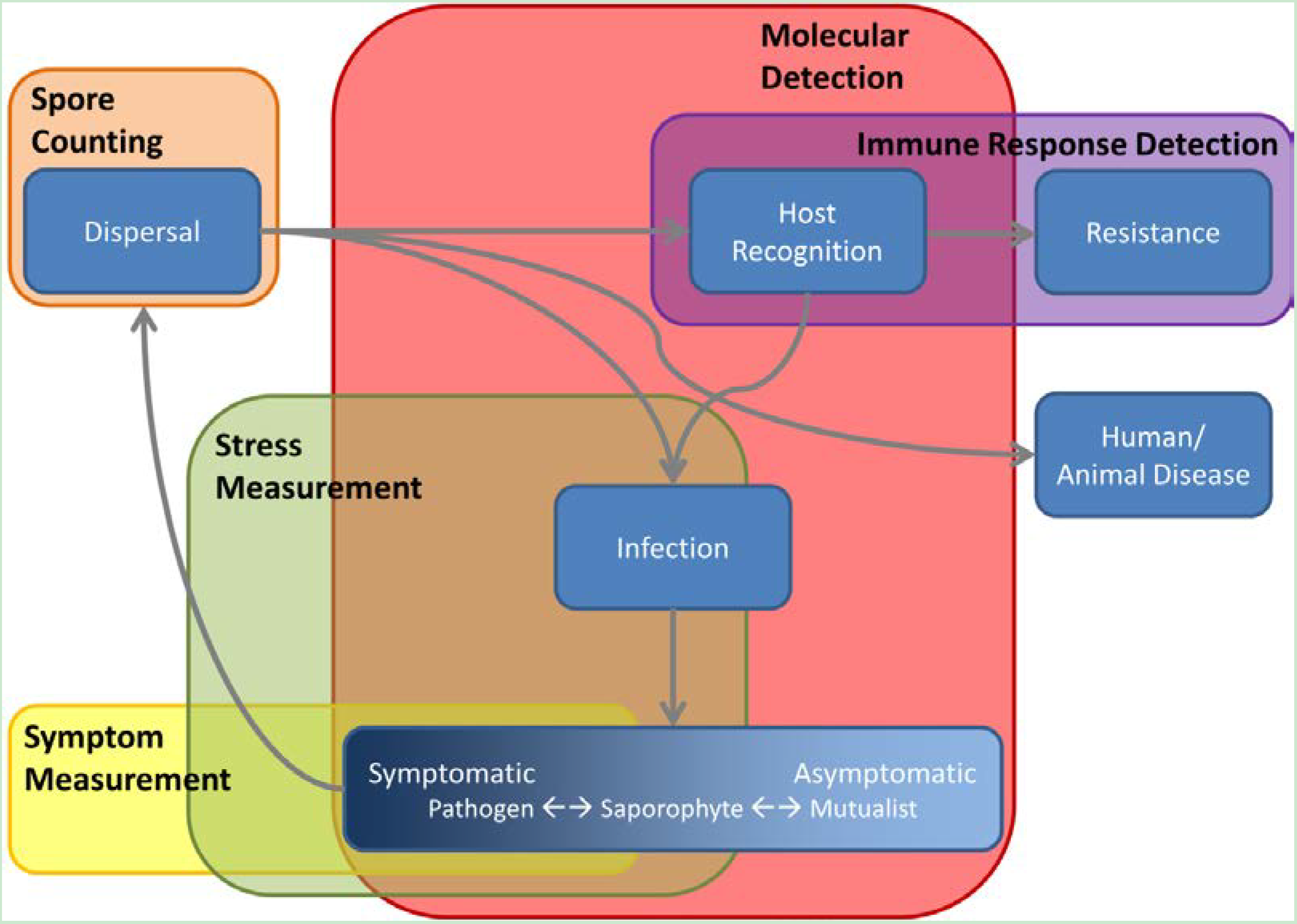
8. The Ecology of Crop Phenotyping
9. Nutrient-Agronomy-Disease: Infection Interactions
10. Yield Effects of Crop Diseases
11. Conclusions
Author Contributions
Conflicts of Interest
References
- Cooper, H.D.; Spillane, C.; Hodgkin, T. Broadening the Genetic Base of Crop Production; CABI: Wallingford, Oxfordshire, UK, 2001. [Google Scholar]
- Chakraborty, S.; Newton, A.C. Climate change, plant diseases and food security: An overview. Plant Pathol. 2011, 60, 2–14. [Google Scholar] [CrossRef]
- Esquinas-Alcazar, J. Protecting crop genetic diversity for food security: Political, ethical and technical challenges. Nat. Rev. Genet. 2005, 6, 946–953. [Google Scholar] [CrossRef]
- Zhao, D.L.; Glynn, N.C.; Glaz, B.; Comstock, J.C.; Sood, S. Orange Rust Effects on Leaf Photosynthesis and Related Characters of Sugarcane. Plant Dis. 2011, 95, 640–647. [Google Scholar] [CrossRef]
- Berger, S.; Sinha, A.K.; Roitsch, T. Plant physiology meets phytopathology: Plant primary metabolism and plant-pathogen interactions. J. Exp. Bot. 2007, 58, 4019–4026. [Google Scholar] [CrossRef]
- Gruber, B.R.; Kruger, E.L.; McManus, P.S. Effects of Cherry Leaf Spot on Photosynthesis in Tart Cherry “Montmorency” Foliage. Phytopathology 2012, 102, 656–661. [Google Scholar] [CrossRef]
- Bassanezi, R.B.; Amorim, L.; Bergamin, A.; Hau, B.; Berger, R.D. Accounting for photosynthetic efficiency of bean leaves with rust, angular leaf spot and anthracnose to assess crop damage. Plant Pathol. 2001, 50, 443–452. [Google Scholar] [CrossRef]
- Bowden, R.L.; Rouse, D.I.; Sharkey, T.D. Mechanism of Photosynthesis Decrease by Verticillium dahliae in Potato. Plant Physiol. 1990, 94, 1048–1055. [Google Scholar] [CrossRef]
- Newton, A.C.; Fitt, B.D.L.; Atkins, S.D.; Walters, D.R.; Daniell, T.J. Pathogenesis, parasitism and mutualism in the trophic space of microbe-plant interactions. Trends Microbiol. 2010, 18, 365–373. [Google Scholar] [CrossRef]
- Waller, F.; Achatz, B.; Baltruschat, H.; Fodor, J.; Becker, K.; Fischer, M.; Heier, T.; Hückelhoven, R.; Neumann, C.; von Wettstein, D.; et al. The endophytic fungus Piriformospora indica reprograms barley to salt-stress tolerance, disease resistance, and higher yield. Proc. Natl. Acad. Sci. USA 2005, 102, 13386–13391. [Google Scholar]
- Holden, N.; Pritchard, L.; Toth, I. Colonization outwith the colon: Plants as an alternative environmental reservoir for human pathogenic enterobacteria. FEMS Microbiol. Rev. 2009, 33, 689–703. [Google Scholar] [CrossRef]
- Newton, A.C.; Hackett, C.A. Subjective components of mildew assessment on spring barley. Eur. J. Plant Pathol. 1994, 100, 395–412. [Google Scholar] [CrossRef]
- Seifers, D.L.; Martin, T.J.; Harvey, T.L.; Fellers, J.P.; Stack, J.P.; Ryba-White, M.; Haber, S.; Krokhin, O.; Spicer, V.; Lovat, N.; et al. Triticum mosaic virus: A New Virus Isolated from Wheat in Kansas. Plant Dis. 2008, 92, 808–817. [Google Scholar] [CrossRef]
- Qu, X.S.; Wanner, L.A.; Christ, B.J. Multiplex real-time PCR (TaqMan) assay for the simultaneous detection and discrimination of potato powdery and common scab diseases and pathogens. J. Appl. Microbiol. 2011, 110, 769–777. [Google Scholar] [CrossRef]
- Dumalasova, V.; Bartos, P. Resistance of winter wheat cultivars to common bunt, Tilletia tritici (Bjerk.) Wint. and T. laevis Kuhn. J. Plant Dis. Prot. 2006, 113, 159–163. [Google Scholar]
- Eifler, J.; Martinelli, E.; Santonico, M.; Capuano, R.; Schild, D.; Di Natale, C. Differential detection of potentially hazardous Fusarium species in wheat grains by an electronic nose. PLoS One 2011, 6, e21026. [Google Scholar]
- Christensen, N.W.; Hayes, P.M. Genetics of Chloride Deficiency Expression in Barley. Commun. Soil Sci. Plan. Anal. 2009, 40, 407–418. [Google Scholar] [CrossRef]
- Sundaraj, S.; Srinivasan, R.; Culbreath, A.K.; Riley, D.G.; Pappu, H.R. Host Plant Resistance Against Tomato spotted wilt virus in Peanut (Arachis hypogaea) and Its Impact on Susceptibility to the Virus, Virus Population Genetics, and Vector Feeding Behavior and Survival. Phytopathology 2014, 104, 202–210. [Google Scholar] [CrossRef]
- Bock, C.H.; Parker, P.E.; Cook, A.Z.; Gottwald, T.R. Characteristics of the perception of different severity measures of citrus canker and the relationships between the various symptom types. Plant Dis. 2008, 92, 927–939. [Google Scholar] [CrossRef]
- Postman, J.; Volk, G.; Aldwinckle, H. Standardized Plant Disease Evaluations Will Enhance Resistance Gene Discovery. Hortscience 2010, 45, 1317–1320. [Google Scholar]
- Bock, C.H.; Cook, A.Z.; Parker, P.E.; Gottwald, T.R. Automated Image Analysis of the Severity of Foliar Citrus Canker Symptoms. Plant Dis. 2009, 93, 660–665. [Google Scholar] [CrossRef]
- Poland, J.A.; Nelson, R.J. In the eye of the beholder: The effect of rater variability and different rating scales on QTL mapping. Phytopathology 2011, 101, 290–298. [Google Scholar] [CrossRef]
- Arnal Barbedo, J.G. Digital image processing techniques for detecting, quantifying and classifying plant diseases. SpringerPlus 2013, 2. [Google Scholar] [CrossRef]
- Macedo-Cruz, A.; Pajares, G.; Santos, M.; Villegas-Romero, I. Digital image sensor-based assessment of the status of oat (Avena sativa L.) crops after frost damage. Sensors 2011, 11, 6015–6036. [Google Scholar] [CrossRef]
- Lloret, J.; Bosch, I.; Sendra, S.; Serrano, A. A wireless sensor network for vineyard monitoring that uses image processing. Sensors 2011, 11, 6165–6196. [Google Scholar] [CrossRef]
- Huang, Y.J.; Pirie, E.J.; Evans, N.; Delourme, R.; King, G.J.; Fitt, B.D.L. Quantitative resistance to symptomless growth of Leptosphaeria maculans (phoma stem canker) in Brassica napus (oilseed rape). Plant Pathol. 2009, 58, 314–323. [Google Scholar] [CrossRef]
- Looseley, M.E.; Newton, A.C.; Atkins, S.D.; Fitt, B.D.L.; Fraaije, B.A.; Thomas, W.T.B.; Keith, R.; Macaulay, M.; Lynott, J.; Harrap, D. Genetic basis of control of Rhynchosporium secalis infection and symptom expression in barley. Euphytica 2011, 184, 47–56. [Google Scholar]
- Bhathal, J.S.; Loughman, R.; Speijers, J. Yield reduction in wheat in relation to leaf disease from yellow (tan) spot and Septoria nodorum blotch. Eur. J. Plant Pathol. 2003, 109, 435–443. [Google Scholar] [CrossRef]
- Araus, J.L.; Tapia, L. Photosynthetic Gas-Exchange Characteristics of Wheat Flag Leaf Blades and Sheaths during Grain Filling—The Case of a Spring Crop Grown under Mediterranean Climate Conditions. Plant Physiol. 1987, 85, 667–673. [Google Scholar] [CrossRef]
- Willey, R.W.; Holliday, R. Plant population and shading studies in barley. J. Agric. Sci. 1971, 77, 445–452. [Google Scholar] [CrossRef]
- Brown, J.K.M.; Rant, J.C. Fitness costs and trade-offs of disease resistance and their consequences for breeding arable crops. Plant Pathol. 2013, 62, 83–95. [Google Scholar] [CrossRef]
- Newton, A.; Guy, D. Exploration and Exploitation Strategies of Powdery Mildew on Barley Cultivars with Different Levels of Nutrients. Eur. J. Plant Pathol. 1998, 104, 829–833. [Google Scholar] [CrossRef]
- Rasmussen, O.F.; Wulff, B.S. Detection of P.s. pv. pisi using PCR. In Proceedings of the 4th International Working Group on Pseudomonas syringae Pathovars, Florence, Italy, 10–13 June 1991; Durbin, R.D., Surico, G., Mugnai, L., Eds.; Stamperia Granducale: Florence, Italy, 1991; pp. 369–376. [Google Scholar]
- Wessling, R.; Panstruga, R. Rapid quantification of plant-powdery mildew interactions by qPCR and conidiospore counts. Plant Methods 2012, 8. [Google Scholar] [CrossRef] [Green Version]
- Silvar, C.; Diaz, J.; Merino, F. Real-Time Polymerase Chain Reaction Quantification of Phytophthora capsici in Different Pepper Genotypes. Phytopathology 2005, 95, 1423–1429. [Google Scholar] [CrossRef]
- Fountaine, J.M.; Shaw, M.W.; Napier, B.; Ward, E.; Fraaije, B.A. Application of real-time and multiplex polymerase chain reaction assays to study leaf blotch epidemics in barley. Phytopathology 2007, 97, 297–303. [Google Scholar] [CrossRef]
- Brouwer, M.; Lievens, B.; van Hemelrijck, W.; van den Ackerveken, G.; Cammue, B.P.A.; Thomma, B.P.H.J. Quantification of disease progression of several microbial pathogens on Arabidopsis thaliana using real-time fluorescence PCR. FEMS Microbiol. Lett. 2003, 228, 241–248. [Google Scholar] [CrossRef]
- Demeke, T.; Graafenhan, T.; Clear, R.M.; Phan, A.; Ratnayaka, I.; Chapados, J.; Patrick, S.K.; Gaba, D.; Levesque, C.A.; Seifert, K.A. Development of a specific TaqMan (R) real-time PCR assay for quantification of Fusarium graminearum clade 7 and comparison of fungal biomass determined by PCR vvith deoxynivalenol content in wheat and barley. Int. J. Food Microbiol. 2010, 141, 45–50. [Google Scholar] [CrossRef]
- Llorente, B.; Bravo-Almonacid, F.; Cvitanich, C.; Orlowska, E.; Torres, H.N.; Flawia, M.M.; Alonso, G.D. A quantitative real-time PCR method for in planta monitoring of Phytophthora infestans growth. Lett. Appl. Microbiol. 2010, 51, 603–610. [Google Scholar] [CrossRef]
- Zhu, Y.; Lim, S.S.; Schenck, S.; Arcinas, A.; Komor, E. RT-PCR and quantitative real-time RT-PCR detection of Sugarcane Yellow Leaf Virus (SCYLV) in symptomatic and asymptomatic plants of Hawaiian sugarcane cultivars and the correlation of SCYLV titre to yield. Eur. J. Plant Pathol. 2010, 127, 263–273. [Google Scholar] [CrossRef]
- Taylor, J.M.G.; Paterson, L.J.; Havis, N.D. A quantitative real-time PCR assay for the detection of Ramularia collo-cygni from barley (Hordeum vulgare). Lett. Appl. Microbiol. 2010, 50, 493–499. [Google Scholar] [CrossRef]
- Saville, R.J.; Gosman, N.; Burt, C.J.; Makepeace, J.; Steed, A.; Corbitt, M.; Chandler, E.; Brown, J.K.M.; Boulton, M.I.; Nicholson, P. The “Green Revolution” dwarfing genes play a role in disease resistance in Triticum aestivum and Hordeum vulgare. J. Exp. Bot. 2011. [CrossRef]
- Baker, S.J.; Newton, A.C.; Gurr, S.J. Cellular characteristics of temporary partial breakdown of mlo-resistance in barley to powdery mildew. Physiol. Mol. Plant Pathol. 2000, 56, 1–11. [Google Scholar] [CrossRef]
- Tellenbach, C.; Grunig, C.R.; Sieber, T.N. Suitability of quantitative real-time PCR to estimate the biomass of fungal root endophytes. Appl. Environ. Microbiol. 2010, 76, 5764–5772. [Google Scholar] [CrossRef]
- Gamper, H.A.; Young, J.P.W.; Jones, D.L.; Hodge, A. Real-time PCR and microscopy: Are the two methods measuring the same unit of arbuscular mycorrhizal fungal abundance? Fungal Genet. Biol. 2008, 45, 581–596. [Google Scholar] [CrossRef]
- Skelsey, P.; Newton, A. Scale-dependent assessment of relative disease resistance to plant pathogens. Agronomy 2014, 4, 178–190. [Google Scholar] [CrossRef]
- Cooke, B.M. Disease assessment and yield loss. In The Epidemiology of Plant Diseases; Cooke, B.M., Jones, D.G., Kaye, B., Eds.; Springer: Dordrecht, The Netherlands, 2006; pp. 43–80. [Google Scholar]
- Garces, F.F.; Gutierrez, A.; Hoy, J.W. Detection and Quantification of Xanthomonas albilineans by qPCR and Potential Characterization of Sugarcane Resistance to Leaf Scald. Plant Dis. 2013, 98, 121–126. [Google Scholar] [CrossRef]
- Abd-Elsalam, K.; Bahkali, A.H.; Moslem, M.; de Wit, P.J.G.M.; Verreet, J.-A. Detection of Mycosphaerella graminicola in Wheat Leaves by a Microsatellite Dinucleotide Specific-Primer. Int. J. Mol. Sci. 2011, 12, 682–693. [Google Scholar] [CrossRef]
- Trivedi, P.; Sagaram, U.S.; Kim, J.S.; Brlansky, R.H.; Rogers, M.E.; Stelinski, L.L.; Oswalt, C.; Wang, N. Quantification of viable Candidatus Liberibacter asiaticus in hosts using quantitative PCR with the aid of ethidium monoazide (EMA). Eur. J. Plant Pathol. 2009, 124, 553–563. [Google Scholar]
- Wang, Z.C.; Yu, D.D.; Li, X.Y.; Zeng, M.J.; Chen, Z.; Bi, L.; Liu, J.J.; Jin, L.H.; Hu, D.Y.; Yang, S.; et al. The Development and Application of a Dot-ELISA Assay for Diagnosis of Southern Rice Black-Streaked Dwarf Disease in the Field. Viruses 2012, 4, 167–183. [Google Scholar] [CrossRef]
- Newton, A.C. Detection of components of partial resistance to mildew (Erysiphe graminis f. sp. hordei) incorporated into advanced breeding lines of barley using measurement of fungal cell wall sterol. Plant Pathol. 1990, 39, 598–602. [Google Scholar] [CrossRef]
- Newton, A.C. Measuring the sterol content of barley leaves infected with powdery mildew as a means of assessing partial resistance to Erysiphe graminis f. sp. hordei. Plant Pathol. 1989, 38, 534–540. [Google Scholar] [CrossRef]
- Fitt, B.D.L.; McCartney, H.A.; West, J.S. Dispersal of foliar plant pathogens: Mechanisms, gradients and spatial patterns. In The Epidemiology of Plant Diseases; Cooke, B.M., Jones, D.G., Kaye, B., Eds.; Springer: Dordrecht, The Netherlands, 2006; pp. 159–192. [Google Scholar]
- West, J.; Bravo, C.; Oberti, R.; Moshou, D.; Ramon, H.; McCartney, H.A. Detection of Fungal Diseases Optically and Pathogen Inoculum by Air Sampling. In Precision Crop Protection—The Challenge and Use of Heterogeneity; Oerke, E.-C., Gerhards, R., Menz, G., Sikora, R.A., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 135–149. [Google Scholar]
- Martinelli, F.; Reagan, R.L.; Uratsu, S.L.; Phu, M.L.; Albrecht, U.; Zhao, W.; Davis, C.E.; Bowman, K.D.; Dandekar, A.M. Gene regulatory networks elucidating huanglongbing disease mechanisms. PLoS One 2013, 8, e74256. [Google Scholar] [CrossRef] [Green Version]
- Wagner, A.M.W.; Jamiokowska, A. Chlorophyll fluorescence measurements as indicators of fusariosis severity in tomato plants. Agron. Res. 2006, 4, 461–464. [Google Scholar]
- Chaerle, L.; Hagenbeek, D.; Bruyne, E.; Straeten, D. Chlorophyll fluorescence imaging for disease-resistance screening of sugar beet. Plant Cell Tissue Org. Cult. 2007, 91, 97–106. [Google Scholar] [CrossRef]
- Pineda, M.; Olejnickova, J.; Csefalvay, L.; Baron, M. Tracking viral movement in plants by means of chlorophyll fluorescence imaging. J. Plant Physiol. 2011, 168, 2035–2040. [Google Scholar] [CrossRef]
- Newton, A.C.; Hackett, C.A.; Lowe, R.; Wale, S.J. Relationship between canopy reflectance and yield loss due to disease in barley. Ann. Appl. Biol. 2004, 145, 95–106. [Google Scholar] [CrossRef]
- Newton, A.C.; Toth, I.K. Helper bacteria and pathogenicity assessments. New Phytol. 1999, 144, 385–386. [Google Scholar] [CrossRef]
- Newton, A.C.; Toth, I.K.; Neave, P.; Hyman, L.J. Bacterial inoculum from a previous crop affects fungal disease development on subsequent nonhost crops. New Phytol. 2004, 163, 133–138. [Google Scholar] [CrossRef]
- Darling, D.; Harling, R.; Simpson, R.A.; McRoberts, N.; Hunter, E.A. Susceptibility of Broccoli Cultivars to Bacterial Head Rot: In Vitro Screening and the Role of Head Morphology in Resistance. Eur. J. Plant Pathol. 2000, 106, 11–17. [Google Scholar] [CrossRef]
- Dewey, F.M.; Wong, Y.L.; Seery, R.; Hollins, T.W.; Gurr, S.J. Bacteria associated with Stagonospora (Septoria) nodorum increase pathogenicity of the fungus. New Phytol. 1999, 144, 489–497. [Google Scholar] [CrossRef]
- Goswami, R.S.; Kistler, H.C. Heading for disaster: Fusarium graminearum on cereal crops. Mol. Plant Pathol. 2004, 5, 515–525. [Google Scholar] [CrossRef]
- Brooks, D.H.; Dawson, M.G. Influence of direct-drilling of winter wheat on incidence of take-all and eyespot. Ann. Appl. Biol. 1968, 61, 57–64. [Google Scholar] [CrossRef]
- Yarham, D.J.; Norton, J. Effects of cultivation methods on disease. In Strategies for the Control of Cereal Disease; Jenkyn, J.F., Plumb, R.T., Eds.; Blackwell Scientific Publications: Oxford, UK, 1981. [Google Scholar]
- Burnett, F.J.; Hughes, G. The Development of a Risk Assessment Method to Identify Wheat Crops at Risk from Eyespot; Home-Grown Cereals Authority: London, UK, 2004. [Google Scholar]
- Jalaluddin, M.; Jenkyn, J.F. Effects of wheat crop debris on the sporulation and survival of Pseudocercosporella herpotrichoides. Plant Pathol. 1996, 45, 1052–1064. [Google Scholar]
- Kuntzsch, E. Yield, yield components and infection with Pseudocercosporella herpotrichoides during long-term winter wheat monoculture in the Etzdorf teaching and research station 1972–1987. Wiss. Z. Martin-Luther-Univ. Halle-Wittenb. Math.-Naturwiss. Reihe 1990, 39, 107–113. [Google Scholar]
- Newton, A.C.; Guy, D.C.; Bengough, A.G.; Gordon, D.C.; McKenzie, B.M.; Sun, B.; Valentine, T.A.; Hallett, P.D. Soil tillage effects on the efficacy of cultivars and their mixtures in winter barley. Field Crops Res. 2012, 128, 91–100. [Google Scholar] [CrossRef]
- Turkington, T.K.; Clayton, G.W.; Klein-Gebbinck, H.W.; Lupwayi, N.Z.; Harker, K.N.; O’Donovan, J.T.; Burnett, P.A.; Xi, K. Impact of crop management on leaf diseases in Alberta barley fields, 1995–1997. Can. J. Plant Pathol. 2006, 28, 441–449. [Google Scholar] [CrossRef]
- Fountaine, J.M.; Shaw, M.W.; Ward, E.; Fraaije, B.A. The role of seeds and airborne inoculum in the initiation of leaf blotch (Rhynchosporium secalis) epidemics in winter barley. Plant Pathol. 2010, 59, 330–337. [Google Scholar] [CrossRef]
- Oxley, S.J.P.; Havis, N.D.; Burnett, F.J.; Roberts, A.M.I. Spread and early control of Rhynchosporium secalis. In Proceedings of the Dundee Conference—Crop Protection in Northern Britain 2008, Dundee, UK, 26–27 February 2008; pp. 133–138.
- Havis, N.D.; Nyman, M.; Oxley, S.J.P. Evidence for seed transmission and symptomless growth of Ramularia collo-cygni in barley (Hordeum vulgare). Plant Pathol. 2013. [Google Scholar] [CrossRef]
- Havis, N.D.; Nyman, M.; Oxley, S.J.P. Potential of seed treatment to control Ramularia collo-cygni in barley. In Proceedings of the Dundee Conference—Crop Protection in Northern Britain 2010, Dundee, UK, 23–24 February 2010.
- Hornby, D.; Bateman, G.L. Take-All Disease of Cereals: A Regional Perspective; CAB International: Wallingford, Oxfordshire, UK, 1998. [Google Scholar]
- Edwards, S. Improving Risk Assessment to Minimise Fusarium Mycotoxins in Harvested Wheat Grain; Home Grown Cereals Authority: London, UK, 2011. [Google Scholar]
- Edwards, S. Investigation of Fusarium Mycotoxins in UK Barley and Oat Production; Home Grown Cereals Authority: Caledonia House, London, UK, 2007. [Google Scholar]
- Kriss, A.B.; Paul, P.A.; Xu, X.; Nicholson, P.; Doohan, F.M.; Hornok, L.; Rietini, A.; Edwards, S.G.; Madden, L.V. Quantification of the relationship between the environment and Fusarium head blight, Fusarium pathogen density, and mycotoxins in winter wheat in Europe. Eur. J. Plant Pathol. 2012, 133, 975–993. [Google Scholar] [CrossRef]
- Landschoot, S.; Waegeman, W.; Audenaert, K.; Vandepitte, J.; Haesaert, G.; de Baets, B. Toward a Reliable Evaluation of Forecasting Systems for Plant Diseases: A Case Study Using Fusarium Head Blight of Wheat. Plant Dis. 2012, 96, 889–896. [Google Scholar] [CrossRef]
- Bennett, A.E.; Daniell, T.J.; White, P.J. Benefits of Breeding Crops for Yield Response to Soil Organisms. In Molecular Microbial Ecology of the Rhizosphere; de Bruijn, F.J., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013; pp. 17–27. [Google Scholar]
- Savary, S.; Teng, P.S.; Willocquet, L.; Nutter, F.W., Jr. Quantification and modeling of crop losses: A review of purposes. Ann. Rev. Phytopathol. 2006, 44, 89–112. [Google Scholar] [CrossRef]
- Newton, A.C.; Guy, D.C.; Nadziak, J.; Gacek, E.S. The Effect of Inoculum Pressure, Germplasm Selection and Environment on Spring Barley Cultivar Mixtures Efficacy. Euphytica 2002, 125, 325–335. [Google Scholar] [CrossRef]
- Ruske, R.E.; Gooding, M.J.; Jones, S.A. The effects of adding picoxystrobin, azoxystrobin and nitrogen to a triazole programme on disease control, flag leaf senescence, yield and grain quality of winter wheat. Crop Prot. 2003, 22, 975–987. [Google Scholar] [CrossRef]
- Beck, C.; Oerke, E.C.; Dehne, H.W. Impact of strobilurins on physiology and yield formation of wheat. Meded. Rijksuniv. Gent Fak. Landbouwkd. Toegep. Biol. Wet. 2002, 67, 181–187. [Google Scholar]
- Gu, L.K.; Bai, Z.H.; Jin, B.; Hu, Q.; Wang, H.L.; Zhuang, G.Q.; Zhang, H.X. Assessing the impact of fungicide enostroburin application on bacterial community in wheat phyllosphere. J. Environ. Sci. China 2010, 22, 134–141. [Google Scholar] [CrossRef]
- Fountaine, J.M.; Gravouil, C.; Daniell, T.; Harling, R.; Shepherd, T.; Taylor, J.; Dickinson, M.; Newton, A.C. Leaf wax and cultivar effects on phylloplane organisms and disease in barley. Aspects Appl. Biol. 2009, 98, 207–212. [Google Scholar]
- Hughes, G.; Burnett, F.J.; Havis, N.D. Disease risk curves. Phytopathology 2013, 103, 1108–1114. [Google Scholar] [CrossRef]
- Walters, D.R.; McRoberts, N.; Fitt, B.D.L. Are green islands red herrings? Significance of green islands in plant interactions with pathogens and pests. Biol. Rev. 2008, 83, 79–102. [Google Scholar] [CrossRef]
- Walters, D.; Walsh, D.; Newton, A.; Lyon, G. Induced resistance for plant disease control: Maximizing the efficacy of resistance elicitors. Phytopathology 2005, 95, 1368–1373. [Google Scholar] [CrossRef]
- Newton, A.; Guy, D.; Gaunt, R.; Thomas, W. The effect of powdery mildew inoculum pressure and fertilizer levels on disease tolerance in spring barley. Z. für Pflanzenkrankheit. und Pflanzenschutz 2000, 107, 67–73. [Google Scholar]
- Bingham, I.; Newton, A. Crop Tolerance of Foliar Pathogens: Possible Mechanisms and Potential for Exploitation. In Disease Control in Crops: Biological and Environmentally-Friendly Approaches; Walters, D., Ed.; Wiley-Blackwell: Chichester, UK, 2009; pp. 142–161. [Google Scholar]
- Dordas, C. Role of nutrients in controlling plant diseases in sustainable agriculture: A review. Agron. Sustain. Dev. 2008, 28, 33–46. [Google Scholar] [CrossRef]
- De la Fuente, L.; Parker, J.K.; Oliver, J.E.; Granger, S.; Brannen, P.M.; van Santen, E.; Cobine, P.A. The Bacterial Pathogen Xylella fastidiosa Affects the Leaf Ionome of Plant Hosts during Infection. PLoS One 2013, 8, e62945. [Google Scholar] [CrossRef]
- Walters, D.R.; Bingham, I.J. Influence of nutrition on disease development caused by fungal pathogens: Implications for plant disease control. Ann. Appl. Biol. 2007, 151, 307–324. [Google Scholar] [CrossRef]
- Silvia, H.; Elke, B.; Ewald, S. Plant disease control by nutrient management: sulphur. In Disease Control in Crops: Biological and Environmentally-Friendly Approaches; Walters, D., Ed.; Wiley-Blackwell: Chichester, UK, 2009; pp. 221–236. [Google Scholar]
- Lemmens, M.; Buerstmayr, H.; Krska, R.; Schuhmacher, R.; Grausgruber, H.; Ruckenbauer, P. The effect of inoculation treatment and long-term application of moisture on Fusarium head blight symptoms and deoxynivalenol contamination in wheat grains. Eur. J. Plant Pathol. 2004, 110, 299–308. [Google Scholar] [CrossRef]
- Newton, A.; Thomas, W.T.B. Resistance to spots and blotches in spring barley. In Proceedings of the Dundee Conference—Crop Protection in Northern Britain 2006, Dundee, UK, 28 February–1 March 2006; pp. 191–194.
- Thomas, W.T.B.; Baird, E.; Fuller, J.D.; Lawrence, P.; Young, G.R.; Russell, J.; Ramsay, L.; Waugh, R.; Powell, W. Identification of a QTL decreasing yield in barley linked to Mlo powdery mildew resistance. Mol. Breed. 1998, 4, 381–393. [Google Scholar] [CrossRef]
- McGrann, G.R.D.; Stavrinides, A.; Russell, J.; Corbitt, M.M.; Booth, A.; Chartrain, L.; Thomas, W.T.B.; Brown, J.K.M. A trade off between mlo resistance to powdery mildew and increased susceptibility of barley to a newly important disease, Ramularia leaf spot. J. Exp. Bot. 2014, 65, 1025–1037. [Google Scholar] [CrossRef]
- Bastiaans, L. Ratio between Virtual and Visual Lesion Size as a Measure to Describe Reduction in Leaf Photosynthesis of Rice Due to Leaf Blast. Phytopathology 1991, 81, 611–615. [Google Scholar] [CrossRef]
- Roloff, I.; Scherm, H.; van Iersel, M.W. Photosynthesis of blueberry leaves as affected by septoria leaf spot and abiotic leaf damage. Plant Dis. 2004, 88, 397–401. [Google Scholar] [CrossRef]
- Elings, A.; Rossing, W.A.; van der Werf, W. Virtual Lesion Extension: A Measure to Quantify the Effects of Bacterial Blight on Rice Leaf CO2 Exchange. Phytopathology 1999, 89, 789–795. [Google Scholar] [CrossRef]
- Erickson, J.E.; Stanosz, G.R.; Kruger, E.L. Photosynthetic consequences of Marssonina leaf spot differ between two poplar hybrids. New Phytol. 2004, 161, 577–583. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Looseley, M.E.; Newton, A.C. Assessing the Consequences of Microbial Infection in Field Trials: Seen, Unseen, Beneficial, Parasitic and Pathogenic. Agronomy 2014, 4, 302-321. https://doi.org/10.3390/agronomy4020302
Looseley ME, Newton AC. Assessing the Consequences of Microbial Infection in Field Trials: Seen, Unseen, Beneficial, Parasitic and Pathogenic. Agronomy. 2014; 4(2):302-321. https://doi.org/10.3390/agronomy4020302
Chicago/Turabian StyleLooseley, Mark E., and Adrian C. Newton. 2014. "Assessing the Consequences of Microbial Infection in Field Trials: Seen, Unseen, Beneficial, Parasitic and Pathogenic" Agronomy 4, no. 2: 302-321. https://doi.org/10.3390/agronomy4020302




