Phenotypic Diversity of Farmers’ Traditional Rice Varieties in the Philippines
Abstract
:1. Introduction
2. Results and Discussion
2.1. Germplasm Characterization
2.1.1. Diversity in Qualitative Traits
| Descriptor | Predominant State | % | States Observed | H′ Index |
|---|---|---|---|---|
| Invariant | ||||
| Ligule Shape | 2-ccenter | 100.00 | 1 | 0.00 |
| Culm Kneeing Ability | Absent | 100.00 | 1 | 0.00 |
| Low diversity | ||||
| Awn Color (Late) | Awnless | 95.11 | 4 | 0.12 |
| Panicle Type | Medium (~25 cm) | 91.86 | 3 | 0.12 |
| Awn Color (Early) | Awnless | 91.53 | 6 | 0.18 |
| Awn Distribution | Awnless | 91.53 | 5 | 0.20 |
| Culm Diameter Type | Thick | 96.42 | 2 | 0.22 |
| Awn Type | Awnless | 91.21 | 5 | 0.22 |
| Panicle Secondary Branching | Sparse | 89.58 | 3 | 0.25 |
| Culm Habit/Angle | Erect (<15°) | 85.67 | 4 | 0.26 |
| Sterile Lemma Type | Medium | 62.54 | 3 | 0.27 |
| Culm Anthocyanin Coloration on Nodes | Absent | 90.55 | 4 | 0.28 |
| Awn Presence | Absent | 91.53 | 3 | 0.29 |
| Leaf Blade Pubescence | Intermediate | 91.86 | 3 | 0.29 |
| Flag Leaf Attitude/Angle | Erect | 83.71 | 4 | 0.30 |
| Leaf Blade Attitude | Erect | 89.90 | 3 | 0.31 |
| Sterile Lemma Color | Straw | 88.27 | 4 | 0.32 |
| Panicle Attitude | Drooping | 84.36 | 5 | 0.37 |
| Stigma Color | White | 77.85 | 4 | 0.41 |
| Auricle Color | Whitish | 75.90 | 6 | 0.45 |
| Moderate diversity | ||||
| Lemma & Palea Color (Late Observation) | Straw | 66.45 | 8 | 0.46 |
| Panicle Attitude of Main Axis | Slightly drooping | 70.03 | 3 | 0.49 |
| Leaf Blade Length Type | Intermediate (~50 cm) | 60.59 | 3 | 0.54 |
| Culm Underlying Node Color | Green | 73.29 | 3 | 0.56 |
| Leaf Blade Width Type | Intermediate | 65.15 | 2 | 0.59 |
| Flag Leaf Attitude (Late Measurement) | Descending | 71.01 | 4 | 0.63 |
| Culm Length Type | Intermediate to long | 36.81 | 6 | 0.63 |
| Culm Number Type | Intermediate (~15 culms) | 75.24 | 3 | 0.65 |
| Late Lemma Apiculus Color | Straw | 47.56 | 9 | 0.65 |
| Lemma & Palea Pubescence | Short hairs | 60.91 | 5 | 0.66 |
| Panicle Exsertion | Well exerted | 55.37 | 5 | 0.67 |
| Caryopsis Pericarp Color (Seed Coat Color) | White | 50.49 | 7 | 0.68 |
| Caryopsis Shape | Long spindle-shaped | 43.97 | 5 | 0.71 |
| Productivity | Intermediate | 70.03 | 3 | 0.71 |
| Apiculus Shape | Curved | 78.83 | 2 | 0.74 |
| High diversity | ||||
| Early Lemma Apiculus Color | Straw | 26.71 | 8 | 0.79 |
| Culm Lodging Resistance | Intermediate | 42.02 | 5 | 0.84 |
| Culm Strength | Intermediate | 41.04 | 5 | 0.84 |
| Endosperm Type | Non-glutinous | 41.37 | 3 | 0.99 |
| Average diversity | 0.45 | |||

2.1.2. Diversity in Quantitative Traits
| Descriptors | H′ | Min Trait Value | Variety | Max Trait Value | Variety | Mean Trait Value (± Standard Deviation) |
|---|---|---|---|---|---|---|
| Low Diversity | ||||||
| Awn Length (mm) | 0.02 | 1.98 | Puchagwan (11241) | 69.64 | Burdagol (11083) | 0.80 (6.01) |
| Moderate Diversity | ||||||
| Caryopsis Length (mm) | 0.64 | 4.37 | Milagrosa (11102) | 13.72 | Binaka (10838) | 6.49 (1.04) |
| Caryopsis Width (mm) | 0.70 | 4.15 | Kaimpas (11315) | 1.28 | Doriat Pula (10934) | 2.23 (0.37) |
| Culm Diameter at Basal Internode (mm) | 0.72 | 0.58 | Fancy-1 (11105) | 12.40 | Speaker (11080) | 8.06 (1.92) |
| Culm Number | 0.73 | 5.20 | Lubang (11341) | 34.80 | Rc 18 (Pula) (11312) | 16.30 (4.48) |
| Caryopsis Length/Width Ratio Score | 0.74 | 1.42 | Kaimpas (11315) | 5.12 | Binaka (10838) | 2.98 (0.60) |
| High Diversity | ||||||
| Panicle Length (cm) | 0.77 | 20.90 | Pinarompong (10932) | 38.80 | Unoy (Umangan) (11245) | 27.27 (2.67) |
| Maturity (day) | 0.77 | 71.00 | Inuway (10869) | 154.00 | 18-Pula (11061); Galo (11205) | 106.84 (35.69) |
| Grain Width (mm) | 0.78 | 1.51 | Duriat (10943) | 6.56 | Kipil (10837) | 2.58 (0.53) |
| Panicle Number Per Plant | 0.78 | 5.20 | Lubang (11341) | 34.80 | Rc 18 (Pula) (11312) | 16.26 (4.46) |
| Flag Leaf Length (cm) | 0.79 | 23.80 | Bolinao (11256) | 72.80 | Kinakaw (10857) | 38.57 (7.56) |
| Leaf Blade Length (cm) | 0.80 | 34.80 | C-4 Dinorado (11317) | 88.40 | Lubang (11356) | 58.36 (9.13) |
| Sterile Lemma Length (mm) | 0.81 | 1.56 | M10-2 (11086) | 3.86 | Minindoro (Pula) (10844) | 2.47 (0.39) |
| 100 Grain Weight (g) | 0.81 | 1.10 | Kinakaw (10857) | 5.50 | Binaka (10810) | 2.45 (0.54) |
| Culm Length (cm) | 0.82 | 67.80 | Kinakaw (10857) | 160.60 | Dinorado (11049) | 116.03 (15.67) |
| Grain Length (mm) | 0.84 | 5.29 | Dinorado (10860) | 10.76 | Binaka (10810) | 8.48 (1.05) |
| Flag Leaf Width (cm) | 0.85 | 1.10 | Elon-Peta (11346) | 2.60 | Binisaya (10826) | 1.95 (0.32) |
| Leaf Blade Width (cm) | 0.85 | 0.96 | Rc 18 (Pula) (11312) | 2.48 | Binisaya (10826) | 1.64 (0.30) |
| Average Diversity | 0.73 | |||||
2.2. Correlation among Traits

2.3. Cluster and Principal Component Analyses of Rice Germplasm
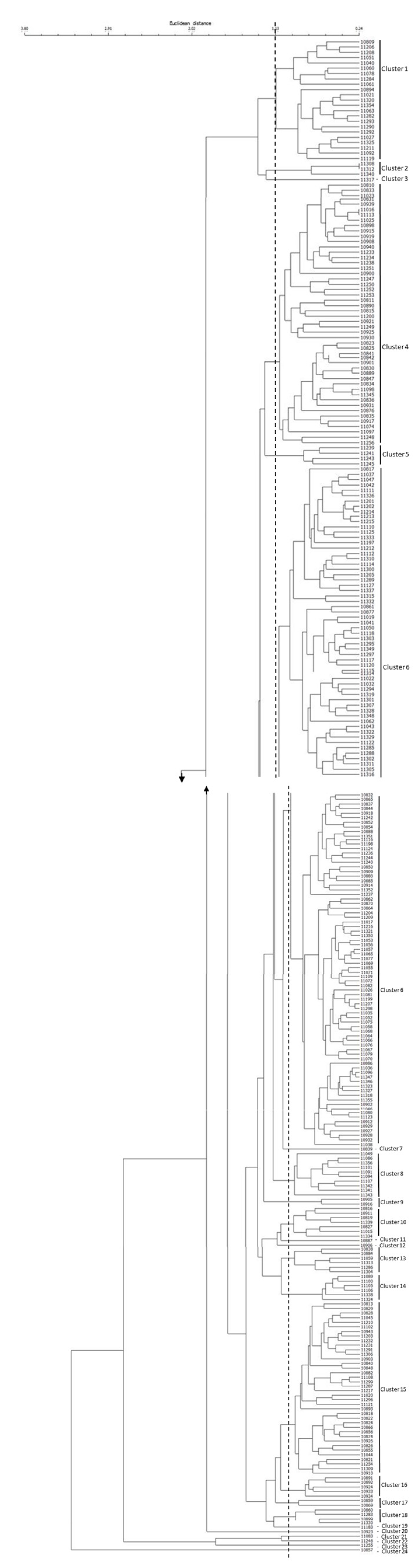
| Components | Eigenvalue | Explained Variance | |
|---|---|---|---|
| Percent | Cumulative | ||
| 1 | 4.04 | 22.46 | 22.46 |
| 2 | 2.61 | 14.52 | 36.98 |
| 3 | 1.91 | 10.61 | 47.59 |
| 4 | 1.61 | 8.97 | 56.56 |
| 5 | 1.26 | 7.01 | 63.58 |
| 6 | 1.05 | 5.84 | 69.41 |
| 7 | 1.00 | 5.53 | 74.95 |
| 8 | 0.82 | 4.57 | 79.52 |
| 9 | 0.71 | 3.96 | 83.48 |
| 10 | 0.64 | 3.53 | 87.01 |
| 11 | 0.57 | 3.19 | 90.21 |
| 12 | 0.49 | 2.75 | 92.95 |
| 13 | 0.41 | 2.30 | 95.26 |
| 14 | 0.33 | 1.85 | 97.11 |
| 15 | 0.29 | 1.61 | 98.72 |
| 16 | 0.21 | 1.16 | 99.87 |
| 17 | 0.02 | 0.12 | 99.99 |
| 18 | 0.00 | 0.01 | >100% |
| Descriptors | Principal Components | ||||||
|---|---|---|---|---|---|---|---|
| PC 1 | PC 2 | PC 3 | PC 4 | PC 5 | PC 6 | PC 7 | |
| Maturity (day) | 0.33 | 0.29 | 0.34 | 0.10 | 0.40 | 0.23 | 0.23 |
| Leaf Blade Length (mm) | −0.11 | 0.48 | 0.53 | 0.14 | 0.12 | −0.08 | −0.31 |
| Leaf Blade Width (mm) | −0.67 | −0.21 | −0.04 | −0.26 | −0.25 | 0.25 | 0.04 |
| Flag Leaf Length (mm) | −0.17 | 0.21 | 0.43 | 0.46 | −0.11 | 0.13 | −0.26 |
| Flag Leaf Width (mm) | −0.73 | −0.12 | −0.01 | 0.00 | −0.14 | 0.21 | 0.21 |
| Culm Number | 0.65 | −0.28 | −0.36 | 0.52 | −0.08 | 0.17 | −0.09 |
| Culm Length (cm) | −0.34 | 0.37 | 0.45 | 0.11 | −0.14 | 0.33 | 0.02 |
| Basal Culm Diameter (mm) | −0.33 | −0.33 | −0.01 | 0.15 | −0.32 | 0.54 | 0.17 |
| Awn Length (mm) | −0.11 | −0.01 | 0.06 | 0.22 | −0.54 | −0.57 | 0.41 |
| Panicle Number/plant | 0.65 | −0.28 | −0.36 | 0.51 | −0.08 | 0.18 | −0.10 |
| Panicle Length (cm) | −0.27 | 0.01 | 0.33 | 0.59 | −0.27 | −0.20 | 0.01 |
| Sterile Lemma Length (mm) | −0.51 | −0.67 | 0.10 | −0.12 | 0.18 | −0.03 | −0.08 |
| Grain Length (mm) | 0.06 | −0.47 | 0.37 | 0.26 | 0.46 | −0.06 | 0.46 |
| Grain Width (mm) | −0.66 | 0.13 | −0.29 | 0.34 | 0.39 | −0.09 | 0.10 |
| 100-Grain Weight (g) | −0.51 | −0.46 | −0.03 | 0.28 | 0.29 | −0.06 | −0.02 |
| Caryopsis Length (mm) | −0.10 | −0.73 | 0.31 | −0.08 | −0.05 | −0.14 | −0.43 |
| Caryopsis Width (mm) | −0.72 | 0.08 | −0.38 | 0.18 | 0.10 | −0.13 | −0.33 |
| Caryopsis Length/Width Ratio Score | 0.53 | −0.60 | 0.52 | −0.18 | −0.10 | −0.02 | −0.05 |
3. Experimental Section
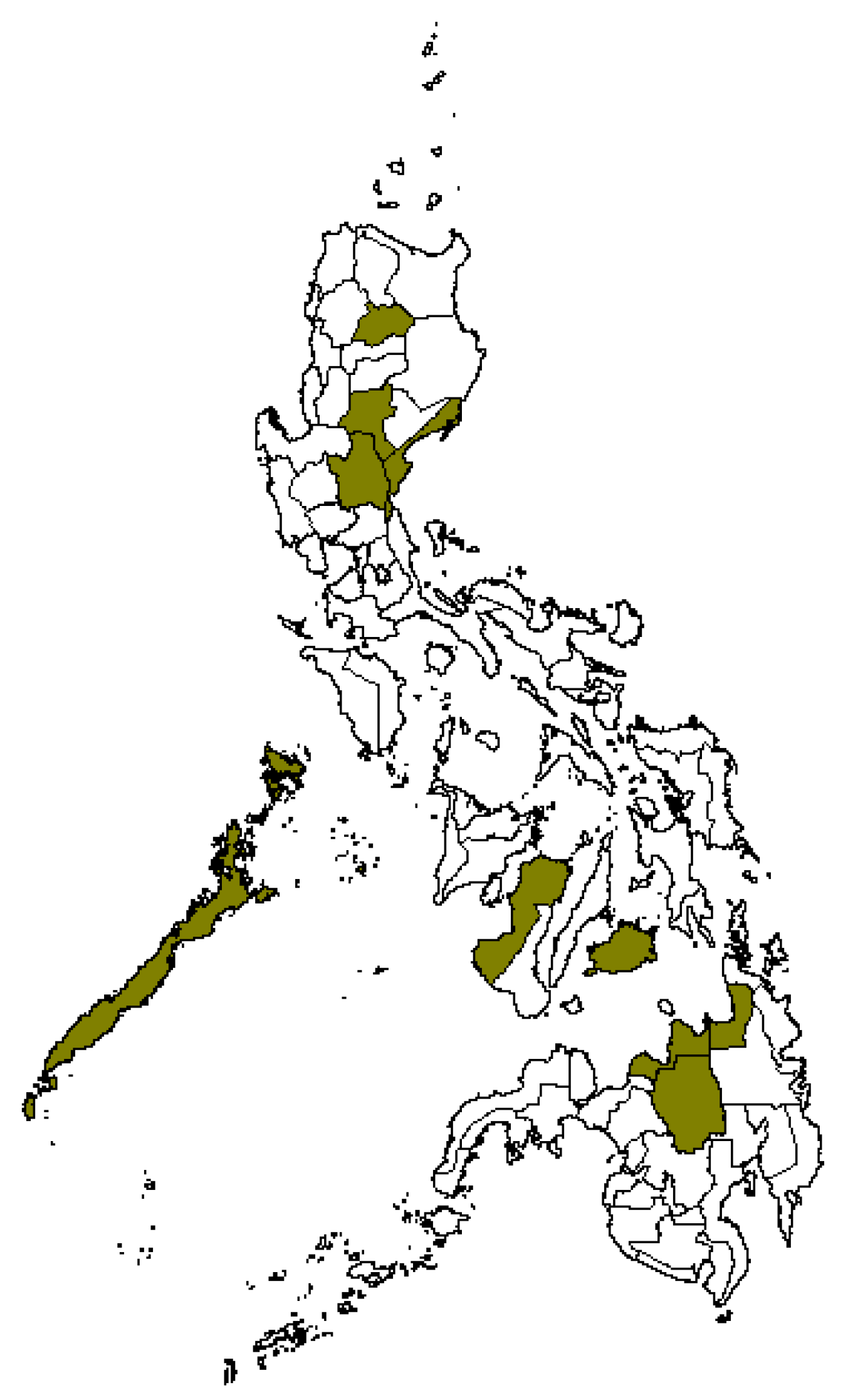
3.1. Germplasm Characterization
3.2. Data Analyses
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- FAOSTAT, Classic Version; Food and Agriculture Organization of the United Nations: Rome, Italy, 2013. Available online: http://faostat.fao.org/site/339/default.aspx (accessed on 5 August 2013).
- Sebastian, L.S.; Alviola, P.A.; Francisco, S.R. Bridging the rice yield gap in the Philippines. In Bridging the Rice Yield Gap in the Asia-Pacific Region; Papademetriou, M.K., Dent, F.J., Herath, E.M., Eds.; FAO Regional Office for Asia and the Pacific: Bangkok, Thailand, 2000; p. 13. [Google Scholar]
- Cororaton, C.B. Philippine Rice and Rural Poverty: An Impact Analysis of Market Reform Using CGE; International Food Policy Research Institute (IFPRI): Washington, DC, USA, 2006. [Google Scholar]
- Reyes, L.C. Banking seeds. Rice Today 2012, 11, 16–19. [Google Scholar]
- Aguilar, F.V. Rice in the Filipino Diet and Culture; Philippine Institute for Development Studies: Makati, Philippines, 2005. [Google Scholar]
- Establishing the Philippine Rice Research Institute (PRRI). Executive Order 1061; PRRI: Manila, Philippines, 1985. [Google Scholar]
- Donald, C.M. The breeding of crop ideotypes. Euphytica 1968, 17, 385–403. [Google Scholar] [CrossRef]
- Peng, S.; Khush, G.; Cassman, K. Evolution of the new plant ideotype for increased yield potential. In Breaking the Yield Barrier Proceedings of a Workshop on Rice Yield Potential in Favourable Environments; International Rice Research Institute: Manila, Philippines, 1994. [Google Scholar]
- Yoshida, S. Physiological aspects of grain yield. Annu. Rev. Plant Physiol. 1972, 23, 437–464. [Google Scholar] [CrossRef]
- Tonini, A.; Cabrera, E. Opportunities for Global Rice Research in a Changing World; International Rice Research Institute: Los Baños, Philippines, 2011. [Google Scholar]
- Peng, S.; Khush, G.S.; Virk, P.; Tang, Q.; Zou, Y. Progress in ideotype breeding to increase rice yield potential. Field Crops Res. 2008, 108, 32–38. [Google Scholar] [CrossRef]
- Bellon, M.; Pham, J.; Jackson, M. Genetic conservation: A role for rice farmers. In Plant Genetic Conservation: The in situ Approach; Maxted, N., Ford-Lloyd, B., Hawkes, J., Eds.; Chapman and Hall: London, UK, 1997; pp. 263–289. [Google Scholar]
- Caldo, R.; Sebastian, L.; Hernandez, J. Morphology-based genetic diversity analysis of ancestral lines of Philippine rice cultivars. Philipp. J. Crop Sci. 1996, 21, 86–92. [Google Scholar]
- Ronald, P. A case study of rice from traditional breeding to genomics. In The Role of Biotechnology in a Sustainable Food Supply; Popp, J.S., Jahn, M.M., Matlock, M.D., Kemper, N.P., Eds.; Cambridge University Press: New York, NY, USA, 2012; p. 10. [Google Scholar]
- Hargrove, T.; Coffman, W.R. Breeding history. Rice Today 2006, 5, 34–38. [Google Scholar]
- Bailey-Serres, J.; Fukao, T.; Ronald, P.; Ismail, A.; Heuer, S.; Mackill, D. Submergence tolerant rice: SUB1’s journey from landrace to modern cultivar. Rice 2010, 3, 138–147. [Google Scholar] [CrossRef]
- Fujita, D.; Trijatmiko, K.R.; Tagle, A.G.; Sapasap, M.V.; Koide, Y.; Sasaki, K.; Tsakirpaloglou, N.; Gannaban, R.B.; Nishimura, T.; Yanagihara, S.; et al. NAL1 allele from a rice landrace greatly increases yield in modern Indica cultivars. Proc. Natl. Acad. Sci. USA 2013, 110, 20431–20436. [Google Scholar] [CrossRef]
- Bajracharya, J.; Steele, K.A.; Jarvis, D.I.; Sthapit, B.R.; Witcombe, J.R. Rice landrace diversity in Nepal: Variability of agro-morphological traits and SSR markers in landraces from a high-altitude site. Field Crops Res. 2006, 95, 327–335. [Google Scholar] [CrossRef]
- Yawen, Z.; Shiquan, S.; Zichao, L.; Zhongyi, Y.; Xiangkun, W.; Hongliang, Z.; Guosong, W. Ecogeographic and genetic diversity based on morphological characters of indigenous rice (Oryza sativa L.) in Yunnan, China. Genet. Resour. Crop Evol. 2003, 50, 567–577. [Google Scholar] [CrossRef]
- Wu, L.-L.; Liu, Z.-L.; Wang, J.-M.; Zhou, C.-Y.; Chen, K.-M. Morphological, anatomical, and physiological characteristics involved in development of the large culm trait in rice. Aust. J. Crop Sci. 2011, 5, 1356–1363. [Google Scholar]
- Bullard, R.; York, J. Breeding for bird resistance in sorghum and maize. In Progress in Plant Breeding-1; Russell, G., Ed.; Butterworths: London, UK, 1985; pp. 193–222. [Google Scholar]
- Bullard, R.W. Characteristics of Bird-resistance in Agricultural Crops; Denver Wildlife Research Center: Denver, CO, USA, 1988. [Google Scholar]
- Jowett, D. Breeding bird-resistant sorghum in East Africa. Plant Breed. Abstr. 1967, 37, 85. [Google Scholar]
- Vaughan, D.A.; Lu, B.-R.; Tomooka, N. The evolving story of rice evolution. Plant Sci. 2008, 174, 394–408. [Google Scholar] [CrossRef]
- Hu, G.; Zhang, D.; Pan, H.; Li, B.; Wu, J.; Zhou, X.; Zhang, Q.; Zhou, L.; Yao, G.; Li, J.; et al. Fine mapping of the awn gene on chromosome 4 in rice by association and linkage analyses. Chin. Sci. Bull. 2011, 56, 835–839. [Google Scholar] [CrossRef]
- Bertuso, A. Farmers’ Management of Rice Genetic Diversity: A Study on Enhancing Red Rices in Bohol, Philippines, in Department of Crop Science; Wageningen University: Wageningen, The Netherlands, 2000; p. 144. [Google Scholar]
- Hitaka, H. Studies on the lodging of rice plants. Jpn. Agric. Res. Q. 1969, 4, 1–6. [Google Scholar]
- Ookawa, T.; Hobo, T.; Yano, M.; Murata, K.; Ando, T.; Miura, H.; Asano, K.; Ochiai, Y.; Ikeda, M.; Nishitani, R.; et al. New approach for rice improvement using a pleiotropic QTL gene for lodging resistance and yield. Nat. Commun. 2010, 1, 1–11. [Google Scholar]
- Taylor, R. Interpretation of the correlation coefficient: A basic review. J. Diagn. Med. Sonogr. 1990, 6, 35–39. [Google Scholar] [CrossRef]
- Gladun, I.; Karpov, E. Production and partitioning of assimilates between the panicle and vegetative organs of rice after flowering. Russ. J. Plant Physiol. 1993, 40, 629–633. [Google Scholar]
- Yue, B.; Xue, W.Y.; Luo, L.J.; Xing, Y.Z. QTL analysis for flag leaf characteristics and their relationships with yield and yield traits in rice. Acta Genet. Sin. 2006, 33, 824–832. [Google Scholar] [CrossRef]
- Narayanan, N.N.; Vasconcelos, M.W.; Grusak, M.A. Expression profiling of Oryza sativa metal homeostasis genes in different rice cultivars using a cDNA macroarray. Plant Physiol. Biochem. 2007, 45, 277–286. [Google Scholar] [CrossRef]
- Abou-khalifa, A.A.B.; Misra, A.; Salem, A.E.-A.K. Effect of leaf cutting on physiological traits and yield of two rice cultivars. Afr. J. Plant Sci. 2008, 2, 147–150. [Google Scholar]
- Biswal, A.; Kohli, A. Cereal flag leaf adaptations for grain yield under drought: Knowledge status and gaps. Mol. Breed. 2013, 31, 749–766. [Google Scholar]
- Garcia, A.; Dorado, M.; Perez, I.; Montilla, E. Effect of water deficit on the distribution of photoassimilates in rice plants (Oryza sativa L.). Interciencia 2010, 35, 46–54. [Google Scholar]
- Chen, Y.; Lubberstedt, T. Molecular basis of trait correlations. Trends Plant Sci. 2010, 15, 454–461. [Google Scholar] [CrossRef]
- Ringnér, M. What is principal component analysis? Nat. Biotechnol. 2008, 26, 303–304. [Google Scholar]
- O’Rourke, N.; Hatcher, L. A Step-by-Step Approach to Using SAS for Factor Analysis and Structural Equation Modeling, 2nd ed.; SAS Institute: Cary, NC, USA, 2013. [Google Scholar]
- Bioversity International, International Rice Research Institute and WARDA Africa Rice Center. Descriptors for Wild and Cultivated Rice (Oryza spp.); Bioversity International; International Rice Research Institute: Rome, Italy, 2007. [Google Scholar]
- Hijmans, R.; Guarino, L.; Cruz, M.; Rojas, E. Computer tools for spatial analysis of plant genetic resources data: 1. DIVA-GIS. Plant Genet. Resour. Newsl. 2001, 127, 15–19. [Google Scholar]
- SAS Institute. I. What’s New in SAS 9.3; SAS Institute: Cary, NC, USA, 2012. [Google Scholar]
- Sotto, R.; Rabara, R.C. Morphological diversity of Musa balbisiana in the Philippines. J. Nat. Stud. 2007, 6, 37–46. [Google Scholar]
- Jamago, J.M.; Cortes, R.V. Seed diversity and utilization of the upland rice landraces and traditional varieties from selected areas in Bukidnon, Philippines. IAMURE Int. J. Ecol. Conserv. 2012, 4, 112–130. [Google Scholar]
- Pielou, E.C. An Introduction to Mathematical Ecology; John Wiley & Sons Inc.: Hoboken, NJ, USA, 1969. [Google Scholar]
- Lexerød, N.L.; Eid, T. An evaluation of different diameter diversity indices based on criteria related to forest management planning. For. Ecol. Manag. 2006, 222, 17–28. [Google Scholar] [CrossRef]
- Rohlf, F. NTSYSpc:Numerical Taxonomy System; Version 2.1; Exeter Publishing, Ltd.: Setauket, New York, NY, USA, 2002. [Google Scholar]
- Akita, S. Improving yield potential in tropical rice. In Progressin Irrigated Rice Research; International Rice Research Institute: Manila, Philippines, 1989; pp. 41–73. [Google Scholar]
Appendix
| Collection Number | Cultivar Name | Province | Eco-geographic Race |
|---|---|---|---|
| 10809 | Galo | Aurora | Indica |
| 10810 | Binaka (Malagkit) | Aurora | Indica |
| 10811 | Palawan | Aurora | Indica |
| 10813 | Galo (Malagkit) | Aurora | Indica |
| 10815 | Galo | Aurora | Indica |
| 10816 | Galo (Malagkit) | Aurora | Indica |
| 10817 | Brilyante | Aurora | Indica |
| 10818 | Galo | Aurora | Indica |
| 10819 | Malagkit | Aurora | Indica |
| 10821 | Inuway | Aurora | Indica |
| 10822 | Lubag/Galo | Aurora | Indica |
| 10823 | Palawan | Aurora | Javanica |
| 10824 | Lubag (With Awn) | Aurora | Indica |
| 10825 | Palawan | Aurora | Javanica |
| 10826 | Binisaya | Aurora | Indica |
| 10827 | Galo (Malagkit) | Aurora | Indica |
| 10828 | Gayanggang | Aurora | Indica |
| 10829 | Lubag | Aurora | Indica |
| 10830 | Malagkit (Puti) | Aurora | Javanica |
| 10831 | Inuhay | Aurora | Indica |
| 10832 | Palawan | Aurora | Javanica |
| 10833 | Inuhay | Aurora | Indica |
| 10834 | Galo | Aurora | Javanica |
| 10835 | Galo (Malagkit) | Aurora | Javanica |
| 10836 | Galo (Haba) | Aurora | Indica |
| 10837 | Kipil | Aurora | Japonica |
| 10838 | Binaka (Malagkit) | Aurora | Javanica |
| 10839 | Gayanggang | Aurora | Indica |
| 10840 | Palawan | Aurora | Javanica |
| 10841 | Sinagat | Aurora | Javanica |
| 10842 | Minindoro (Puti) | Aurora | Indica |
| 10844 | Minindoro (Pula) | Aurora | Indica |
| 10847 | Malagkit-Itim | Aurora | Javanica |
| 10848 | Palawan | Aurora | Javanica |
| 10850 | Lubag (Malagkit) | Aurora | Javanica |
| 10852 | Binernal | Aurora | Javanica |
| 10854 | Francis Rice | Aurora | Javanica |
| 10855 | Inuway | Aurora | Indica |
| 10856 | Galo | Aurora | Indica |
| 10857 | Kinakaw | Aurora | Indica |
| 10859 | Binisaya | Aurora | Javanica |
| 10860 | Dinorado | Aurora | Javanica |
| 10861 | Sinabado | Aurora | Indica |
| 10862 | Galo | Aurora | Indica |
| 10864 | Binisaya | Aurora | Indica |
| 10865 | Kinamalig | Aurora | Javanica |
| 10866 | Galo | Aurora | Indica |
| 10869 | Inuway | Aurora | Indica |
| 10870 | Galo | Aurora | Indica |
| 10874 | Azucena | Aurora | Indica |
| 10876 | Pirurutong (Malagkit) | Aurora | Javanica |
| 10877 | Kapirit | Aurora | Indica |
| 10880 | Milagrosa (Pilit) | Palawan | Indica |
| 10882 | Minantika | Palawan | Indica |
| 10884 | Pinutyukan | Palawan | Indica |
| 10885 | Milagrosa (Pula) | Palawan | Indica |
| 10886 | Doryat | Palawan | Indica |
| 10887 | Blandi | Palawan | Indica |
| 10888 | Doryat | Palawan | Indica |
| 10889 | Tipak | Palawan | Indica |
| 10890 | Rambo | Palawan | Indica |
| 10891 | Kinadoy | Palawan | Indica |
| 10892 | Kinadoy | Palawan | Indica |
| 10893 | Malagkit | Palawan | Indica |
| 10894 | Unknown | Palawan | Indica |
| 10898 | Tipak | Palawan | Javanica |
| 10899 | Dinorado | Palawan | Indica |
| 10900 | Malagkit (Pula) | Palawan | Javanica |
| 10901 | Dinorado | Palawan | Indica |
| 10902 | Dinorado | Palawan | Indica |
| 10903 | Buringan | Palawan | Indica |
| 10905 | Mating | Palawan | Indica |
| 10906 | Lawang | Palawan | Indica |
| 10908 | Muring-Oring | Palawan | Javanica |
| 10909 | Kinoron | Palawan | Indica |
| 10910 | Pinangpang | Palawan | Indica |
| 10911 | Red Rice | Palawan | Indica |
| 10912 | Binoring-Boring | Palawan | Javanica |
| 10914 | Sagang (Malagkit Puti) | Palawan | Javanica |
| 10915 | Fortuna | Palawan | Indica |
| 10916 | Calubid | Palawan | Indica |
| 10917 | Black Rice | Palawan | Javanica |
| 10918 | Milagrosa | Palawan | Indica |
| 10919 | Benareng | Palawan | Indica |
| 10921 | Minaola | Palawan | Javanica |
| 10923 | Benangkar | Palawan | Javanica |
| 10924 | Mangatas | Palawan | Indica |
| 10925 | Dinayudo | Palawan | Indica |
| 10926 | Dinamit | Palawan | Indica |
| 10927 | Tipak | Palawan | Indica |
| 10928 | Inamoy | Palawan | Indica |
| 10929 | Laok-Laok | Palawan | Indica |
| 10930 | Pinarongpong | Palawan | Javanica |
| 10931 | Kalinayin | Palawan | Indica |
| 10932 | Pinarompong | Palawan | Javanica |
| 10933 | Pinalawan | Palawan | Indica |
| 10934 | Doriat Pula | Palawan | Indica |
| 10939 | Pangpang | Palawan | Indica |
| 10940 | Inantote | Palawan | Indica |
| 10943 | Duriat | Palawan | Indica |
| 11015 | Metao | Negros Occidental | Javanica |
| 11016 | Kutsiam | Negros Occidental | Indica |
| 11017 | Aowot | Negros Occidental | Javanica |
| 11019 | Milagrosa | Negros Occidental | Indica |
| 11020 | Dinorado | Negros Occidental | Indica |
| 11021 | Vietnam Rice | Negros Occidental | Indica |
| 11022 | Pilit | Negros Occidental | Indica |
| 11023 | Azucena | Negros Occidental | Indica |
| 11025 | Unknown 2 | Negros Occidental | Indica |
| 11026 | Kasolid | Negros Occidental | Indica |
| 11027 | Pilit | Negros Occidental | Indica |
| 11032 | M-45 | Negros Occidental | Indica |
| 11035 | Malagaya | Negros Occidental | Javanica |
| 11036 | Dinorado | Negros Occidental | Indica |
| 11037 | Jao Dam Khi Kwai | Negros Occidental | Indica |
| 11038 | Ngacheik | Negros Occidental | Indica |
| 11040 | Tapol | Agusan Del Norte | Indica |
| 11041 | Kapakra | Agusan Del Norte | Indica |
| 11042 | Pilit-Tapol | Agusan Del Norte | Indica |
| 11043 | Señorita | Agusan Del Norte | Indica |
| 11044 | Red Cotabato | Misamis Oriental | Javanica |
| 11045 | Mimis | Misamis Oriental | Indica |
| 11046 | Mal-Os | Misamis Oriental | Javanica |
| 11047 | Kutong | Misamis Oriental | Indica |
| 11049 | Dinorado | Misamis Oriental | Javanica |
| 11050 | Kayatan | Misamis Oriental | Javanica |
| 11051 | Unknown | Misamis Oriental | Indica |
| 11052 | Kabuyog | Misamis Oriental | Javanica |
| 11053 | Gakit | Misamis Oriental | Javanica |
| 11055 | Dinorado | Misamis Oriental | Indica |
| 11056 | Speaker | Misamis Oriental | Javanica |
| 11057 | Dinorado | Misamis Oriental | Indica |
| 11058 | Gakit | Misamis Oriental | Javanica |
| 11059 | Unknown | Misamis Oriental | Indica |
| 11060 | Mabango | Misamis Oriental | Indica |
| 11061 | 18 (Pula) | Misamis Oriental | Indica |
| 11062 | Ir-9 | Misamis Oriental | Indica |
| 11063 | 18 (Puti) | Misamis Oriental | Indica |
| 11064 | Unknown | Misamis Oriental | Javanica |
| 11065 | Dinorado | Misamis Oriental | Indica |
| 11066 | Speaker | Misamis Oriental | Javanica |
| 11067 | Kabuyok | Misamis Oriental | Javanica |
| 11068 | Pilit | Misamis Oriental | Javanica |
| 11069 | Kabuyok | Misamis Oriental | Javanica |
| 11070 | Mixture from Kabuyok | Misamis Oriental | Javanica |
| 11071 | Dinorado | Misamis Oriental | Indica |
| 11072 | Speaker | Misamis Oriental | Javanica |
| 11074 | Kabuyok | Misamis Oriental | Javanica |
| 11075 | Gakit | Misamis Oriental | Javanica |
| 11076 | Dinorado | Misamis Oriental | Indica |
| 11077 | Speaker | Misamis Oriental | Javanica |
| 11078 | Kayatan | Misamis Oriental | Indica |
| 11079 | Pilit-Tapol | Misamis Oriental | Indica |
| 11080 | Speaker | Misamis Oriental | Javanica |
| 11081 | Dinorado | Misamis Oriental | Indica |
| 11082 | Unknown | Misamis Oriental | Javanica |
| 11083 | Burdagol | Negros Occidental | Indica |
| 11086 | M10-2 | Negros Occidental | Javanica |
| 11089 | M45 | Negros Occidental | Indica |
| 11091 | M119-Nl | Negros Occidental | Indica |
| 11092 | A-G-17 | Negros Occidental | Indica |
| 11094 | Mb-4 | Negros Occidental | Indica |
| 11096 | Inday | Negros Occidental | Indica |
| 11097 | Pangasinan | Negros Occidental | Indica |
| 11098 | M108 | Negros Occidental | Indica |
| 11100 | M11-6-1 | Negros Occidental | Indica |
| 11101 | Tape | Negros Occidental | Indica |
| 11102 | Milagrosa | Negros Occidental | Indica |
| 11105 | Fancy-1 | Negros Occidental | Indica |
| 11106 | Galicia F-1 | Negros Occidental | Indica |
| 11107 | Elon-Elon | Negros Occidental | Indica |
| 11108 | Manabang | Negros Occidental | Javanica |
| 11109 | Solig | Negros Occidental | Indica |
| 11110 | Kabagte | Negros Occidental | Javanica |
| 11111 | Azucena | Negros Occidental | Indica |
| 11112 | Ebod | Negros Occidental | Javanica |
| 11113 | Kotsiam | Negros Occidental | Indica |
| 11114 | Kalisan | Negros Occidental | Javanica |
| 11115 | Katipak | Negros Occidental | Indica |
| 11116 | Palawan | Negros Occidental | Javanica |
| 11117 | Bonlaw | Negros Occidental | Javanica |
| 11118 | Mangasa | Negros Occidental | Indica |
| 11119 | Pula | Agusan Del Norte | Indica |
| 11120 | Denorado | Agusan Del Norte | Indica |
| 11121 | Unknown | Agusan Del Norte | Indica |
| 11122 | M-23 | Agusan Del Norte | Indica |
| 11123 | Dinorado | Agusan Del Norte | Indica |
| 11124 | Dinorado | Agusan Del Norte | Indica |
| 11125 | Dinurado | Agusan Del Norte | Indica |
| 11127 | Tapul-Pilit | Agusan Del Norte | Indica |
| 11183 | Farasang | Kalinga | Indica |
| 11197 | Palawan | Nueva Ecija | Indica |
| 11198 | Binaka | Nueva Ecija | Indica |
| 11199 | Inuway | Nueva Ecija | Indica |
| 11200 | Galo | Nueva Ecija | Indica |
| 11201 | Palawan | Nueva Ecija | Javanica |
| 11202 | Ginobyerno | Nueva Ecija | Javanica |
| 11203 | Sinumay | Nueva Ecija | Indica |
| 11204 | Dinorado | Nueva Ecija | Indica |
| 11205 | Galo | Nueva Ecija | Indica |
| 11206 | Palawan | Nueva Ecija | Javanica |
| 11207 | Inuway | Nueva Ecija | Indica |
| 11208 | Galo | Nueva Ecija | Indica |
| 11209 | Palawan | Nueva Ecija | Javanica |
| 11210 | Palawan | Nueva Ecija | Javanica |
| 11211 | Dinorado | Nueva Ecija | Indica |
| 11212 | Inuway | Nueva Ecija | Indica |
| 11213 | Palawan | Nueva Ecija | Javanica |
| 11214 | Minindoro | Nueva Vizcaya | Javanica |
| 11215 | Palawan | Nueva Vizcaya | Javanica |
| 11216 | Brilyante | Nueva Vizcaya | Indica |
| 11217 | Galo | Nueva Vizcaya | Indica |
| 11231 | White Intan | Kalinga | Indica |
| 11232 | Intan (Red) | Kalinga | Indica |
| 11233 | Waray | Kalinga | Indica |
| 11234 | Red Intan | Kalinga | Indica |
| 11236 | Ummunoy | Kalinga | Javanica |
| 11237 | Kintuman | Kalinga | Javanica |
| 11238 | Ingtan (Red) | Kalinga | Indica |
| 11239 | Tuhuwan | Kalinga | Javanica |
| 11240 | Gwayay (Chay-Ot) | Kalinga | Javanica |
| 11241 | Puchagwan | Kalinga | Javanica |
| 11242 | Unoy (Ummunoy) | Kalinga | Javanica |
| 11243 | Unoy (Tu-Par) | Kalinga | Javanica |
| 11244 | Chay-Ot (Ojak) | Kalinga | Javanica |
| 11245 | Unoy (Umangan) | Kalinga | Javanica |
| 11246 | Chay-Ot (Ifuwan) | Kalinga | Javanica |
| 11247 | Chomalingan | Kalinga | Javanica |
| 11248 | Chinannay | Kalinga | Indica |
| 11249 | Inasotiyan | Kalinga | Javanica |
| 11250 | Finongod 2 | Kalinga | Indica |
| 11251 | Finongod | Kalinga | Javanica |
| 11252 | Innoway | Kalinga | Javanica |
| 11253 | Innoyan | Kalinga | Indica |
| 11254 | Mingol | Kalinga | Indica |
| 11255 | Ifo | Kalinga | Indica |
| 11256 | Bolinao | Kalinga | Javanica |
| 11282 | 7 Tonner | Bukidnon | Indica |
| 11283 | Red Rice | Bukidnon | Indica |
| 11284 | Red Tonner | Bukidnon | Indica |
| 11285 | Barako | Bukidnon | Indica |
| 11286 | Red Rice | Bukidnon | Indica |
| 11287 | Burdagol (Pilit) | Bukidnon | Indica |
| 11288 | Pilit | Bukidnon | Indica |
| 11289 | Pilit | Bukidnon | Indica |
| 11290 | Red Tonner | Bukidnon | Indica |
| 11291 | Red Rice | Bukidnon | Javanica |
| 11292 | Brown Rice | Bukidnon | Indica |
| 11293 | Dwarf Variety | Bukidnon | Indica |
| 11294 | M-3 | Bukidnon | Indica |
| 11295 | Elon-Elon | Bukidnon | Indica |
| 11296 | Red Tonner | Bukidnon | Indica |
| 11297 | Baknap | Bukidnon | Indica |
| 11298 | Azucena | Bukidnon | Indica |
| 11299 | Bakiki | Bukidnon | Indica |
| 11300 | Lubang | Bohol | Indica |
| 11301 | Lubang | Bohol | Indica |
| 11302 | Unknown | Bohol | Indica |
| 11303 | Muddy Rice | Bohol | Indica |
| 11304 | Kaimpas | Bohol | Indica |
| 11305 | Milagrosa | Bohol | Javanica |
| 11306 | Milagrosa | Bohol | Indica |
| 11307 | Inibi (Red) | Bohol | Indica |
| 11308 | Kaliga | Bohol | Indica |
| 11309 | Muddy Rice | Bohol | Indica |
| 11310 | Malagkit (Puti) | Bohol | Indica |
| 11311 | Kaliga (Red) | Bohol | Indica |
| 11312 | Rc 18 (Pula) | Bohol | Indica |
| 11313 | Kaliso | Bohol | Javanica |
| 11314 | Muddy Rice | Bohol | Indica |
| 11315 | Kaimpas | Bohol | Japonica |
| 11316 | Malagkit | Bohol | Indica |
| 11317 | C-4 Dinorado | Bohol | Indica |
| 11318 | Kabus-Ok | Bohol | Indica |
| 11319 | Rc 10 (Pula) | Bohol | Indica |
| 11320 | Ir 36 | Bohol | Indica |
| 11321 | Ping Rice | Bohol | Indica |
| 11322 | Pilit | Bohol | Indica |
| 11323 | Kabus-Ok | Bohol | Indica |
| 11324 | Kabus-Ok | Bohol | Indica |
| 11325 | 75 (Pula) | Bohol | Indica |
| 11326 | Unknown | Bohol | Indica |
| 11327 | Kabus-Ok | Bohol | Indica |
| 11328 | Unknown | Bohol | Indica |
| 11329 | Japan Red | Bohol | Indica |
| 11330 | Gl-2 | Bohol | Indica |
| 11332 | Rmp/Kaimpas | Bohol | Indica |
| 11333 | Bilar Red | Bohol | Indica |
| 11334 | Kananoy | Bohol | Indica |
| 11337 | Pilit Tapol | Bohol | Indica |
| 11338 | Miracle Pilit | Bohol | Javanica |
| 11339 | 7-7 Red | Bohol | Indica |
| 11340 | Red Rice | Bohol | Indica |
| 11341 | Lubang (M) | Bohol | Indica |
| 11342 | Hubahib | Bohol | Javanica |
| 11343 | Bares | Bohol | Japonica |
| 11345 | Mal-Us | Bohol | Indica |
| 11346 | Elon-Peta | Bohol | Indica |
| 11347 | Pungko | Bohol | Indica |
| 11348 | Melobina | Bohol | Indica |
| 11349 | Inabaka | Bohol | Indica |
| 11350 | Kayupo | Bohol | Indica |
| 11351 | Sulig | Bohol | Indica |
| 11352 | Ceres | Bohol | Javanica |
| 11354 | Torboho Red | Bohol | Javanica |
| 11355 | Pilit Taba | Bohol | Indica |
| 11356 | Lubang | Bohol | Javanica |
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rabara, R.C.; Ferrer, M.C.; Diaz, C.L.; Newingham, M.C.V.; Romero, G.O. Phenotypic Diversity of Farmers’ Traditional Rice Varieties in the Philippines. Agronomy 2014, 4, 217-241. https://doi.org/10.3390/agronomy4020217
Rabara RC, Ferrer MC, Diaz CL, Newingham MCV, Romero GO. Phenotypic Diversity of Farmers’ Traditional Rice Varieties in the Philippines. Agronomy. 2014; 4(2):217-241. https://doi.org/10.3390/agronomy4020217
Chicago/Turabian StyleRabara, Roel C., Marilyn C. Ferrer, Celia L. Diaz, Ma. Cristina V. Newingham, and Gabriel O. Romero. 2014. "Phenotypic Diversity of Farmers’ Traditional Rice Varieties in the Philippines" Agronomy 4, no. 2: 217-241. https://doi.org/10.3390/agronomy4020217




