Considerations When Deploying Canopy Temperature to Select High Yielding Wheat Breeding Lines under Drought and Heat Stress
Abstract
:1. Introduction
2. Results and Discussion
2.1. Analysis of Check Cultivars across Trials
| Entry | Drought Stress | Heat Stress | ||||||
|---|---|---|---|---|---|---|---|---|
| Yield (tons/ha) | Canopy Temp (°C) | Days to Heading (Julian) | Plant Height (cm) | Yield (tons/ha) | Canopy Temp(°C) | Days to Heading (Julian) | Plant Height(cm) | |
| RoelfsF2007 | 6.0 | 26.7 | 85.2 | 102.4 | 5.4 | 26.2 | 67.9 | 93.8 |
| Waxwing | 5.2 | 27.6 | 89.4 | 88.4 | 5.0 | 26.3 | 67.8 | 82.2 |
| Mean | 5.6 | 27.1 | 87.3 | 95.4 | 5.2 | 26.2 | 67.9 | 88.0 |
| Error | 0.1 | 0.2 | 2.1 | 3.3 | 0.1 | 0.2 | 1.1 | 3.3 |
| LSD (5%) | 0.8 | 1.3 | 4.4 | 5.6 | 0.7 | 1.1 | 3.3 | 5.6 |
| CV (%) | 4.8 | 1.7 | 1.7 | 1.9 | 4.7 | 1.5 | 1.6 | 2.1 |
| Effect | Entry | Rep a | Trial | Incomplete Block | Entry by Trial |
|---|---|---|---|---|---|
| Heat Stress | |||||
| Yield | <0.0001 | 0.1367 | <0.0001 | 0.0024 | 0.0922 |
| CT | 0.1419 | <0.0001 | <0.0001 | 0.0201 | 0.3668 |
| Days to Heading | 0.8811 | 0.3797 | |||
| Plant Height | <0.0001 | 0.0036 | |||
| Drought Stress | |||||
| Yield | <0.0001 | 0.1956 | <0.0001 | 0.7963 | 0.6638 |
| CT | <0.0001 | <0.0001 | <0.0001 | 0.1145 | 0.1147 |
| Days to Heading | <0.0001 | 0.6665 | |||
| Plant Height | <0.0001 | 0.0034 | |||
2.2. Phenotypic Correlations
| Relative Trait | Canopy Temperature | Plant Height | Days to Heading |
|---|---|---|---|
| Heat Stress | |||
| Yield | −0.26 *** | 0.26 *** | −0.18 *** |
| Canopy Temperature | −0.04 | −0.34 *** | |
| Plant Height | 0.07 | ||
| Drought Stress | |||
| Yield | −0.01 | 0.24 *** | −0.50 *** |
| Canopy Temperature | −0.35 *** | −0.09 * | |
| Plant Height | −0.028 | ||
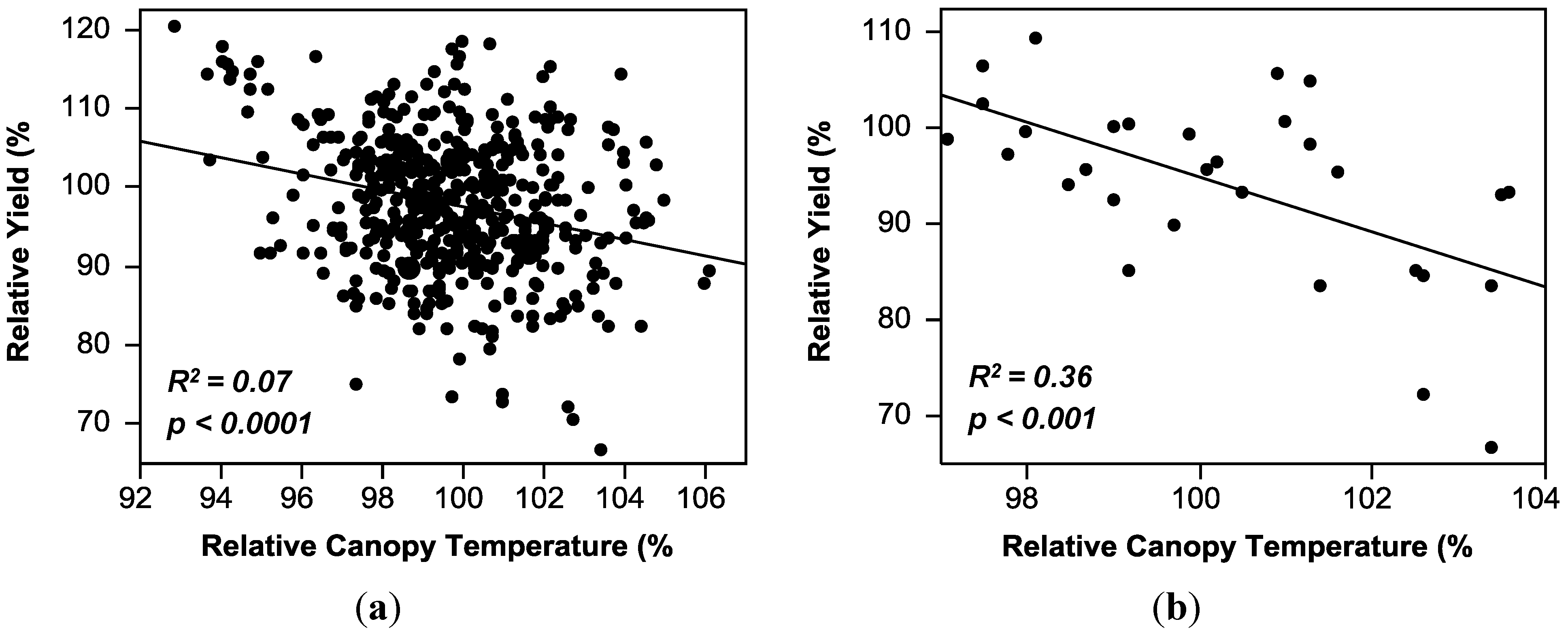
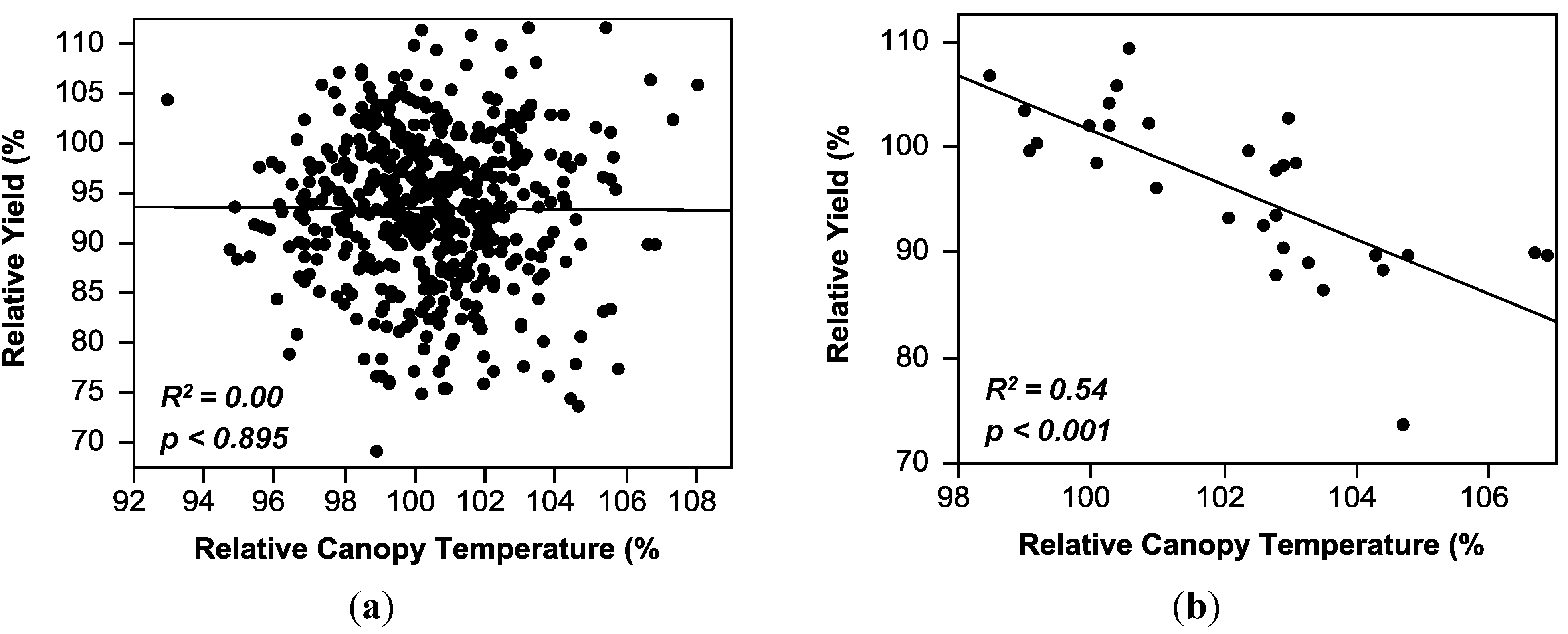
2.3. Regression Analysis
| Variable and Covariate(s) | Regression Equation | R2 | R2 Range a |
|---|---|---|---|
| Heat Stress | |||
| Canopy temperature (CT) | y = 201.21 − 1.04(CT) | 0.07 *** | 0.00–0.36 |
| CT + CT × Height | y = 197.05 − 1.30(CT) + 0.003(CT × HGT) | 0.13 *** | 0.05–0.56 |
| CT + CT × Heading | y = 241.47 − 0.98(CT) − 0.005(CT × HD) | 0.14 *** | 0.02–0.52 |
| CT + CT × Height + CT × Heading | y = 239.19 − 1.25(CT) + 0.004(CT × HGT) − 0.005(CT × HD) | 0.21 *** | 0.08–0.57 |
| Drought Stress | |||
| Canopy temperature (CT) | y = 95.47 − 0.02(CT) | 0.00 | 0.00–0.54 |
| CT + CT × Height | y = 61.46 − 0.06(CT) + 0.00394(CT × HGT) | 0.07 *** | 0.06–0.60 |
| CT + CT × Heading | y = 111.36 + 0.46(CT) − 0.0063(CT × HD) | 0.26 *** | 0.12–0.66 |
| CT + CT × Height + CT × Heading | y = 80.84 + 0.42(CT) + 0.0035(CT × HGT) − 0.0061(CT × HD) | 0.31 *** | 0.31–0.69 |
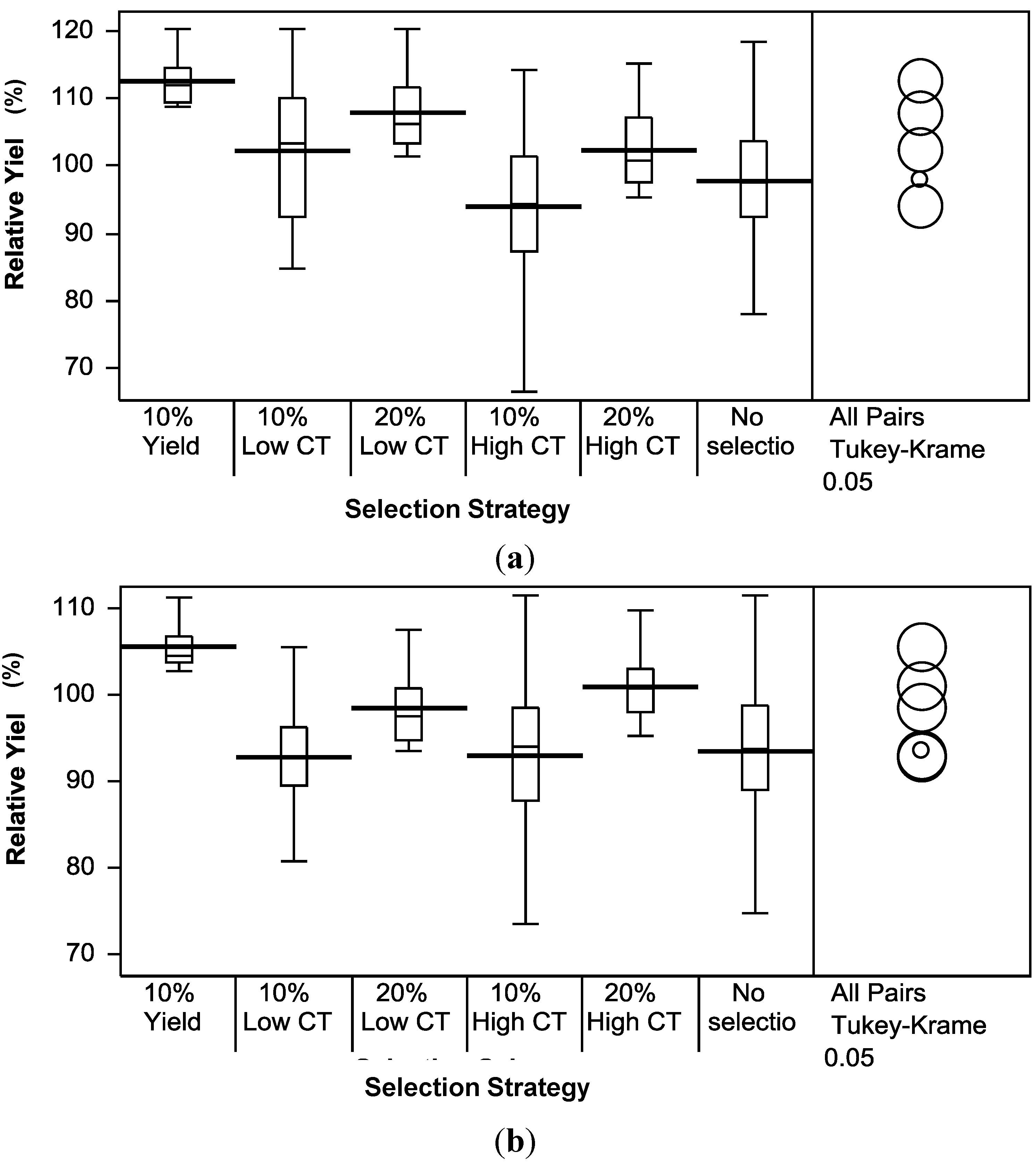
2.4. Comparison of Theoretical Selection Strategies
3. Experimental Section
3.1. Wheat Germplasm and Experimental Design
3.2. Trait Measurement
3.3. Statistical Analysis
3.4. Theoretical Selection Strategies
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Jackson, R.D.; Reginato, R.J.; Idso, S.B. Wheat canopy temperature—Practical tool for evaluating water requirements. Water Resour. Res. 1977, 13, 651–656. [Google Scholar] [CrossRef]
- Reynolds, M.; Manes, Y.; Izanloo, A.; Langridge, P. Phenotyping approaches for physiological breeding and gene discovery in wheat. Ann. Appl. Biol. 2009, 155, 309–320. [Google Scholar] [CrossRef]
- Pask, A.J.D.; Pietragalla, J.; Mullan, D.M.; Reynolds, M.P. Physiological Breeding II: A Field Guide to Wheat Phenotyping; CIMMYT: Mexico City, Mexico, 2012. [Google Scholar]
- Costa, J.M.; Grant, O.M.; Chaves, M.M. Thermography to explore plant-environment interactions. J. Exp. Bot. 2013, 64, 3937–3949. [Google Scholar] [CrossRef]
- Reynolds, M.P.; Pask, A.J.D.; Mullan, D.M. Physiological Breeding I: Interdisciplinary Approaches to Improve Crop Adaptation; CIMMYT: Mexico City, Mexico, 2012. [Google Scholar]
- Rebetzke, G.J.; Rattey, A.R.; Farquhar, G.D.; Richards, R.A.; Condon, A.G. Genomic regions for canopy temperature and their genetic association with stomatal conductance and grain yield in wheat. Funct. Plant Biol. 2013, 40, 14–33. [Google Scholar] [CrossRef]
- Reynolds, M.P.; Saint Pierre, C.; Saad, A.S.I.; Vargas, M.; Condon, A.G. Evaluating potential genetic gains in wheat associated with stress-adaptive trait expression in elite genetic resources under drought and heat stress. Crop Sci. 2007, 47, S172–S189. [Google Scholar]
- Lopes, M.S.; Reynolds, M.P. Partitioning of assimilates to deeper roots is associated with cooler canopies and increased yield under drought in wheat. Funct. Plant Biol. 2010, 37, 147–156. [Google Scholar] [CrossRef]
- Fischer, R.A.; Rees, D.; Sayre, K.D.; Lu, Z.M.; Condon, A.G.; Saavedra, A.L. Wheat yield progress associated with higher stomatal conductance and photosynthetic rate, and cooler canopies. Crop Sci. 1998, 38, 1467–1475. [Google Scholar]
- Lopes, M.S.; Reynolds, M.P.; Jalal-Kamali, M.R.; Moussa, M.; Feltaous, Y.; Tahir, I.S.A.; Barma, N.; Vargas, M.; Mannes, Y.; Baum, M. The yield correlations of selectable physiological traits in a population of advanced spring wheat lines grown in warm and drought environments. Field Crop Res. 2012, 128, 129–136. [Google Scholar] [CrossRef]
- Pinto, R.S.; Reynolds, M.P.; Mathews, K.L.; McIntyre, C.L.; Olivares-Villegas, J.J.; Chapman, S.C. Heat and drought adaptive QTL in a wheat population designed to minimize confounding agronomic effects. Theor. Appl. Genet. 2010, 121, 1001–1021. [Google Scholar] [CrossRef]
- Mason, R.E.; Hays, D.B.; Mondal, S.; Ibrahim, A.M.H.; Basnet, B.R. QTL for yield, yield components and canopy temperature depression in wheat under late sown field conditions. Euphytica 2013, 194, 243–259. [Google Scholar] [CrossRef]
- Lopes, M.S.; Reynolds, M.P. Stay-green in spring wheat can be determined by spectral reflectance measurements (normalized difference vegetation index) independently from phenology. J. Exp. Bot. 2012, 63, 3789–3798. [Google Scholar] [CrossRef]
- Saint Pierre, C.; Crossa, J.; Manes, Y.; Reynolds, M.P. Gene action of canopy temperature in bread wheat under diverse environments. Theor. Appl. Genet. 2010, 120, 1107–1117. [Google Scholar]
- Bennett, D.; Reynolds, M.; Mullan, D.; Izanloo, A.; Kuchel, H.; Langridge, P.; Schnurbusch, T. Detection of two major grain yield QTL in bread wheat (Triticum aestivum L.) under heat, drought and high yield potential environments. Theor. Appl. Genet. 2012, 125, 1473–1485. [Google Scholar] [CrossRef]
- Gutierrez, M.; Reynolds, M.P.; Raun, W.R.; Stone, M.L.; Klatt, A.R. Spectral water indices for assessing yield in elite bread wheat genotypes under well-irrigated, water-stressed, and high-temperature conditions. Crop Sci. 2010, 50, 197–214. [Google Scholar] [CrossRef]
- Reynolds, M.P.; Balota, M.; Delgado, M.I.B.; Amani, I.; Fischer, R.A. Physiological and morphological traits associated with spring wheat yield under hot, irrigated conditions. Australian J. Plant Physiol. 1994, 21, 717–730. [Google Scholar] [CrossRef]
- Gutierrez, M.; Reynolds, M.P.; Raun, W.R.; Stone, M.L.; Klatt, A.R. Indirect selection for grain yield in spring bread wheat in diverse nurseries worldwide using parameters locally determined in north-west Mexico. J. Agr. Sci. 2012, 150, 23–43. [Google Scholar] [CrossRef]
- Giunta, F.; Motzo, R.; Pruneddu, G. Has long-term selection for yield in durum wheat also induced changes in leaf and canopy traits? Field Crops Res. 2008, 106, 68–76. [Google Scholar] [CrossRef]
- Gutierrez, M.; Reynolds, M.P.; Klatt, A.R. Association of water spectral indices with plant and soil water relations in contrasting wheat genotypes. J. Exp. Bot. 2010, 61, 3291–3303. [Google Scholar] [CrossRef]
- Lopes, M.S.; Reynolds, M.P.; Manes, Y.; Singh, R.P.; Crossa, J.; Braun, H.J. Genetic yield gains and changes in associated traits of cimmyt spring bread wheat in a “historic” set representing 30 years of breeding. Crop Sci. 2012, 52, 1123–1131. [Google Scholar] [CrossRef]
- Graybosch, R.A.; Peterson, C.J. Genetic improvement in winter wheat yields in the great plains of North America, 1959–2008. Crop Sci. 2010, 50, 1882–1890. [Google Scholar] [CrossRef]
- Mondal, S.; Singh, R.P.; Crossa, J.; Huerta-Espino, J.; Sharma, I.; Chatrath, R.; Singh, G.P.; Sohu, V.S.; Mavi, G.S.; Sukuru, V.S.P.; et al. Earliness in wheat: A key to adaptation under terminal and continual high temperature stress in South Asia. Field Crops Res. 2013, 151, 19–26. [Google Scholar] [CrossRef]
- Olivares-Villegas, J.J.; Reynolds, M.P.; McDonald, G.K. Drought-adaptive attributes in the Seri/Babax hexaploid wheat population. Funct. Plant Biol. 2007, 34, 189–203. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mason, R.E.; Singh, R.P. Considerations When Deploying Canopy Temperature to Select High Yielding Wheat Breeding Lines under Drought and Heat Stress. Agronomy 2014, 4, 191-201. https://doi.org/10.3390/agronomy4020191
Mason RE, Singh RP. Considerations When Deploying Canopy Temperature to Select High Yielding Wheat Breeding Lines under Drought and Heat Stress. Agronomy. 2014; 4(2):191-201. https://doi.org/10.3390/agronomy4020191
Chicago/Turabian StyleMason, R. Esten, and Ravi P. Singh. 2014. "Considerations When Deploying Canopy Temperature to Select High Yielding Wheat Breeding Lines under Drought and Heat Stress" Agronomy 4, no. 2: 191-201. https://doi.org/10.3390/agronomy4020191




