Fabrication of Multi-Layered Lidocaine and Epinephrine-Eluting PLGA/Collagen Nanofibers: In Vitro and In Vivo Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. In Vitro Release of Epinephrine and Lidocaine
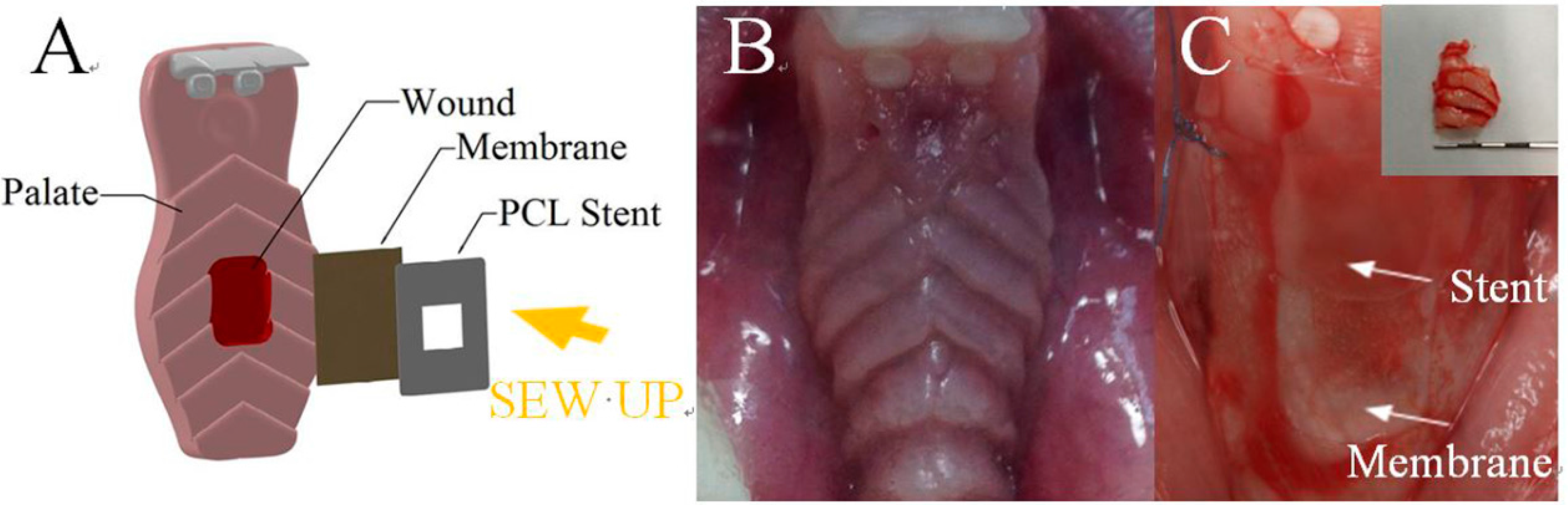
2.1.1. Manufacture of Polycaprolactone Stent and Drugs Loaded PLGA Nanofibers
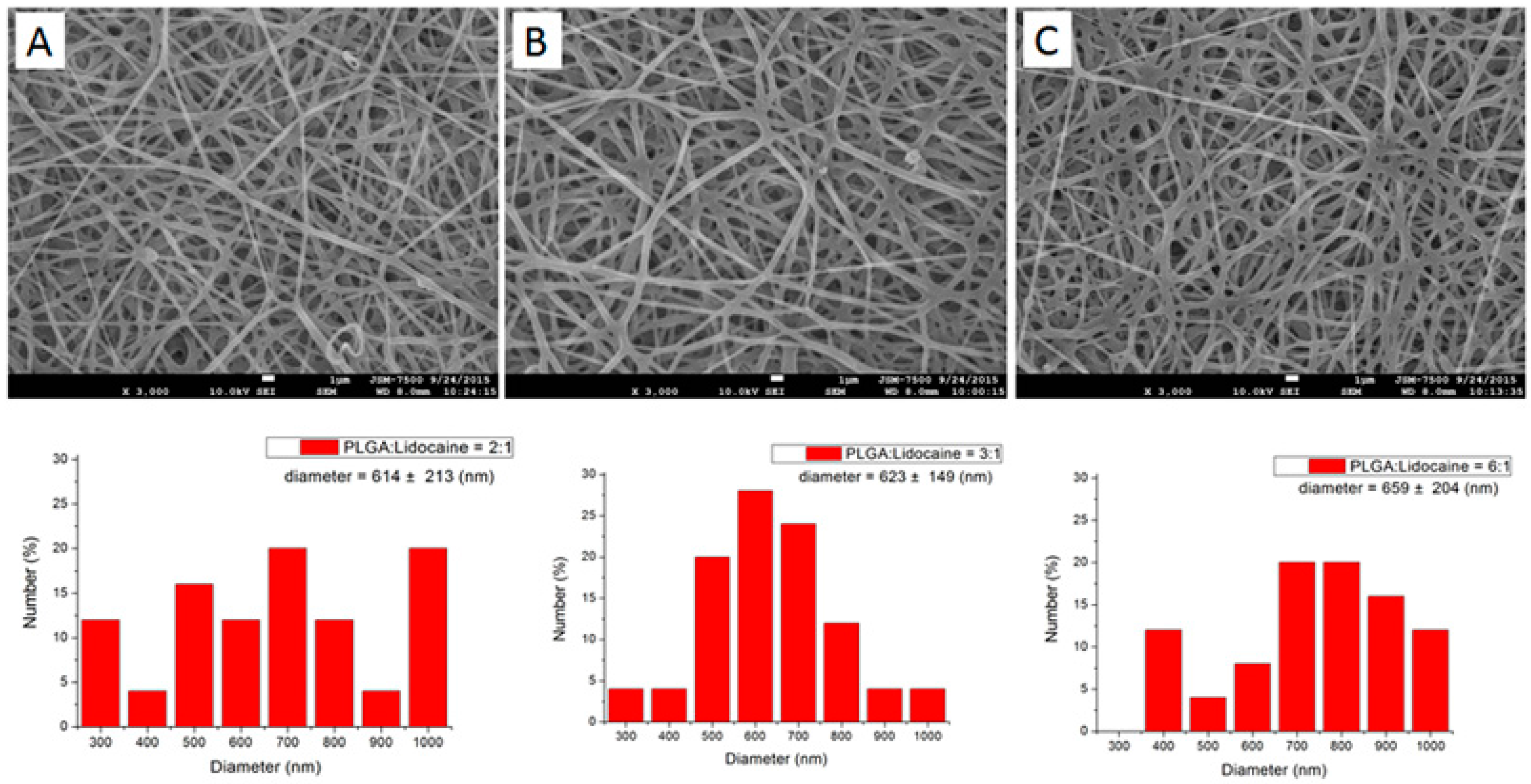
2.1.2. Characterization of Electrospun Nanofibrous Membranes
2.1.3. Drug Concentration Assessment
2.2. Cytotoxicity of Composite Nanofibers
2.3. In Vivo Study of Animal
2.3.1. Animal-Related Procedure
2.3.2. Post-Surgery Evaluation
2.3.3. Wound Healing and Histological Analysis
2.4. Statistical Analysis
3. Results
3.1. In Vitro Characterization of Electrospun Drug-Loaded Nanofibrous Membranes
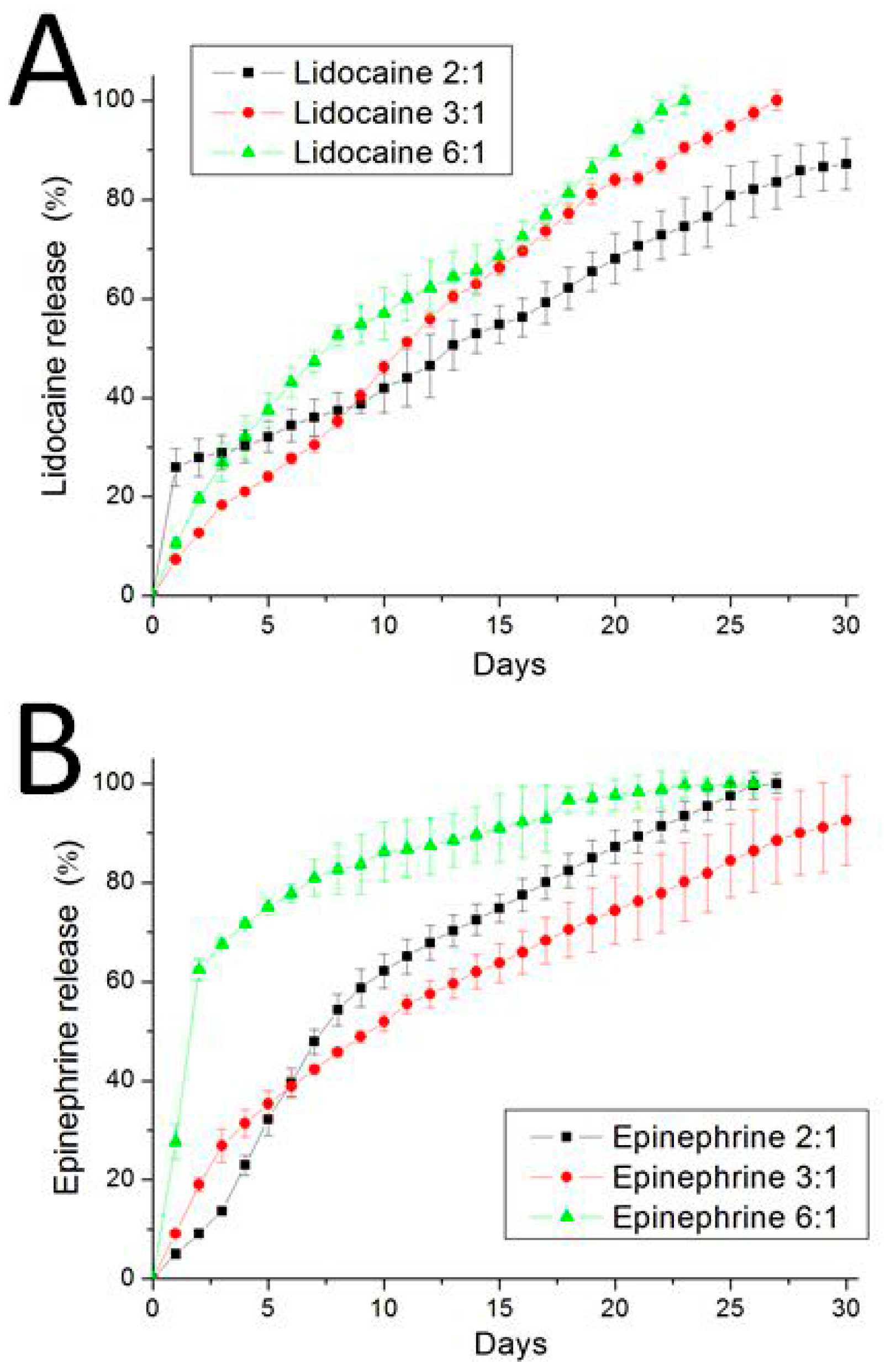
3.2. In Vitro Elution Characteristics of Epinephrine and Lidocaine
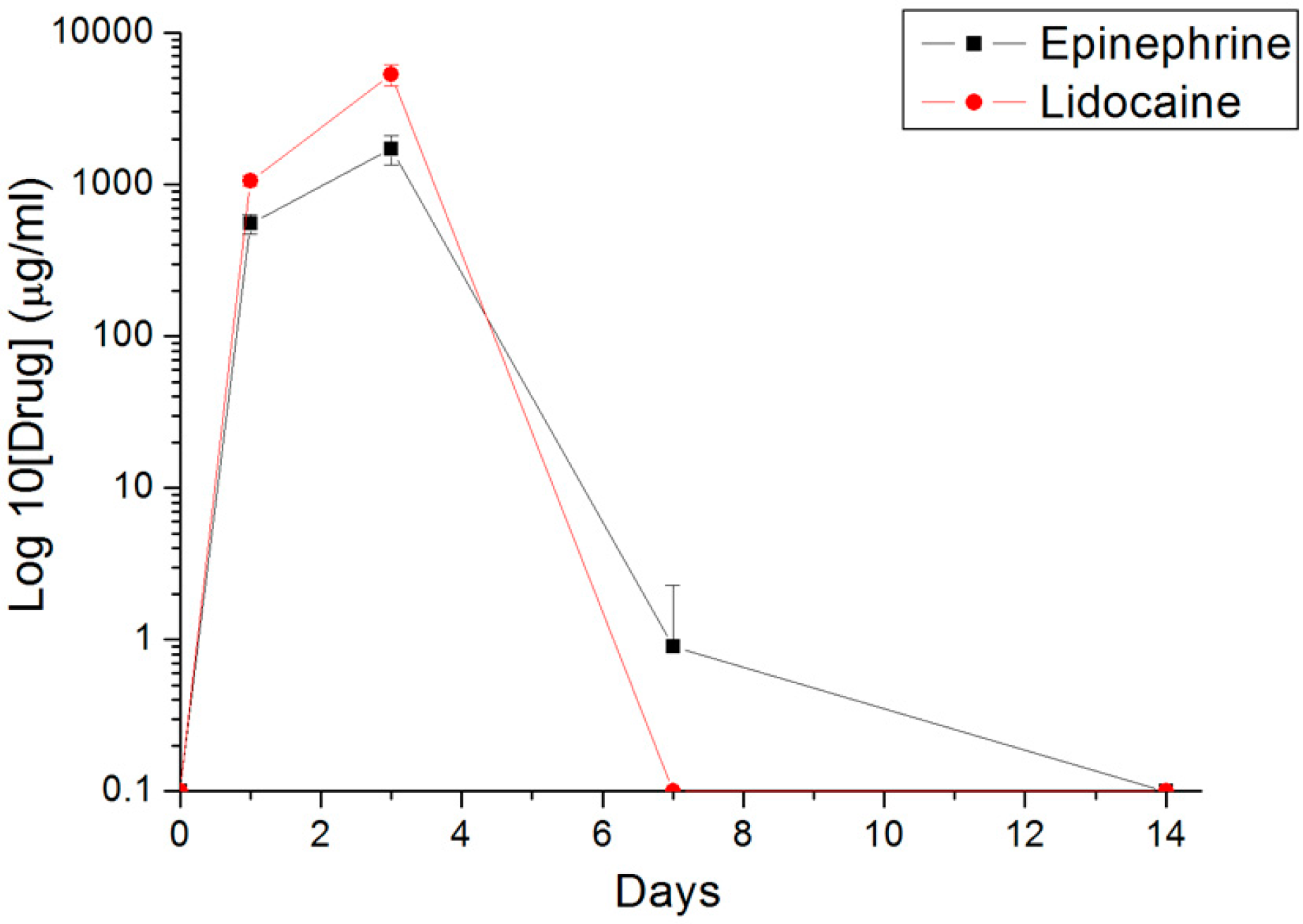
3.3. In Vivo Elution of Lidocaine and Epinephrine
3.4. Efficacy of Released Drugs
3.4.1. Hemostasis Efficacy
3.4.2. Weight Changes
3.4.3. Food Intake and Water Consumption
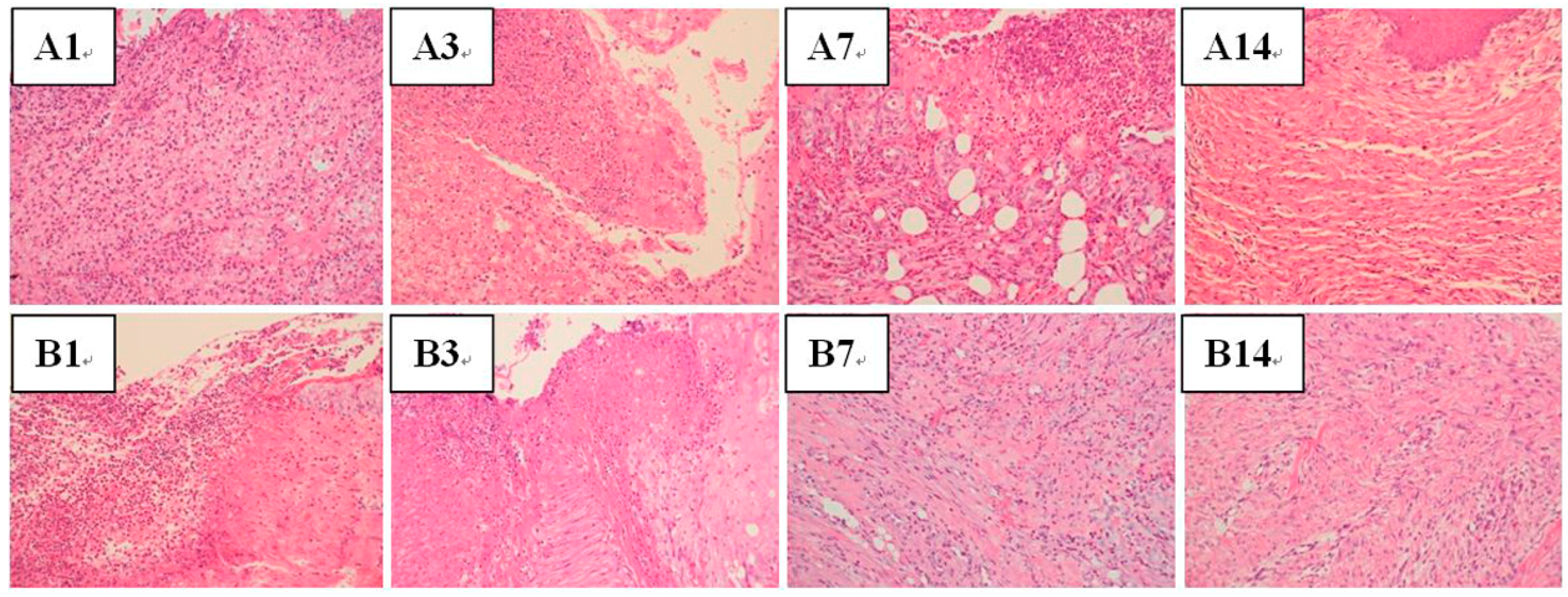
3.4.4. Wound Healing and Histological Analysis
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Matter, J. Free gingival grafts for the treatment of gingival recession. A review of some techniques. J. Clin. Periodontol. 1982, 9, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Bjorn, H. Free transplantation of gingiva propria. Odontol. Revy 1963, 14, 323. [Google Scholar]
- Pennel, B.M.; Tabor, J.C.; King, K.O.; Towner, J.D.; Fritz, B.D.; Higgason, J.D. Free masticatory mucosa graft. J. Periodontol. 1969, 40, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Griffin, T.J.; Cheung, W.S.; Zavras, A.I.; Damoulis, P.D. Postoperative complications following gingival augmentation procedures. J. Periodontol. 2006, 77, 2070–2079. [Google Scholar] [CrossRef] [PubMed]
- Keceli, H.G.; Aylikci, B.U.; Koseoglu, S.; Dolgun, A. Evaluation of palatal donor site haemostasis and wound healing after free gingival graft surgery. J. Clin. Periodontol. 2015, 42, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Cairo, F.; Nieri, M.; Pagliaro, U. Efficacy of periodontal plastic surgery procedures in the treatment of localized facial gingival recessions. A systematic review. J. Clin. Periodontol. 2014, 41 (Suppl. 15), S44–S62. [Google Scholar] [CrossRef] [PubMed]
- Roccuzzo, M.; Bunino, M.; Needleman, I.; Sanz, M. Periodontal plastic surgery for treatment of localized gingival recessions: A systematic review. J. Clin. Periodontol. 2002, 29 (Suppl. 3), 178–194; discussion 195–176. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Y.; Tsai, S.C.; Yu, M.C.; Lin, Y.F.; Chen, C.C.; Chang, P.C. Light-emitting diode irradiation promotes donor site wound healing of the free gingival graft. J. Periodontol. 2015, 86, 674–681. [Google Scholar] [CrossRef] [PubMed]
- Weibel, S.; Jokinen, J.; Pace, N.L.; Schnabel, A.; Hollmann, M.W.; Hahnenkamp, K.; Eberhart, L.H.; Poepping, D.M.; Afshari, A.; Kranke, P. Efficacy and safety of intravenous lidocaine for postoperative analgesia and recovery after surgery: A systematic review with trial sequential analysis. Br. J. Anaesth. 2016, 116, 770–783. [Google Scholar] [CrossRef] [PubMed]
- Hartzell, T.L.; Sangji, N.F.; Hertl, M.C. Ischemia of postmastectomy skin after infiltration of local anesthetic with epinephrine: A case report and review of the literature. Aesthet. Plast. Surg. 2010, 34, 782–784. [Google Scholar] [CrossRef] [PubMed]
- Sinnott, C.J.; Cogswell, I.L.; Johnson, A.; Strichartz, G.R. On the mechanism by which epinephrine potentiates lidocaine’s peripheral nerve block. J. Am. Soc. Anesthesiol. 2003, 98, 181–188. [Google Scholar] [CrossRef]
- Park, S.N.; Lee, H.J.; Lee, K.H.; Suh, H. Biological characterization of EDC-crosslinked collagen-hyaluronic acid matrix in dermal tissue restoration. Biomaterials 2003, 24, 1631–1641. [Google Scholar] [CrossRef]
- Parry, D.A.D. Collagen fibril during development and maturation and their contribution to thr mechanical attributes of connective tissue. Collagen 1988, 2, 1–23. [Google Scholar]
- McGuire, M.K.; Scheyer, E.T. Randomized, controlled clinical trial to evaluate a xenogeneic collagen matrix as an alternative to free gingival grafting for oral soft tissue augmentation. J. Periodontol. 2014, 85, 1333–1341. [Google Scholar] [CrossRef] [PubMed]
- Townsend-Nicholson, A.; Jayasinghe, S.N. Cell Electrospinning: A Unique Biotechnique for Encapsulating Living Organisms for Generating Active Biological Microthreads/Scaffolds. Biomacromolecules 2006, 7, 3364–3369. [Google Scholar] [CrossRef] [PubMed]
- Saroff, S.A.; Chasens, A.I.; Eisen, S.F.; Levey, S.H. Free soft tissue autografts. Hemostasis and protection of the palatal donor site with a microfibrillar collagen preparation. J. Periodontol. 1982, 53, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Fraceto, L.F.; Pinto Lde, M.; Franzoni, L.; Braga, A.A.; Spisni, A.; Schreier, S.; de Paula, E. Spectroscopic evidence for a preferential location of lidocaine inside phospholipid bilayers. Biophys. Chem. 2002, 99, 229–243. [Google Scholar] [CrossRef]
- Wang, S.F.; Du, D.; Zou, Q.C. Electrochemical behavior of epinephrine at l-cysteine self-assembled monolayers modified gold electrode. Talanta 2002, 57, 687–692. [Google Scholar] [CrossRef]
- Marucha, P.T.; Kiecolt-Glaser, J.K.; Favagehi, M. Mucosal wound healing is impaired by examination stress. Psychosom. Med. 1998, 60, 362–365. [Google Scholar] [CrossRef] [PubMed]
- Kahnberg, K.E.; Thilander, H. Healing of experimental excisional wounds in the rat palate: (I) Histological study of the interphase in wound healing after sharp dissection. Int. J. Oral. Surg. 1982, 11, 44–51. [Google Scholar] [CrossRef]
- Fejerskov, O. Excision wounds in palatal epithelium in guinea pigs. Eur. J. Oral Soc. 1972, 80, 139–154. [Google Scholar] [CrossRef]
- Ward, A.W. Postoperative care in the surgical treatment of pyorrhoea. J. Am. Dent. Assoc. 1929, 16, 635–640. [Google Scholar]
- Brasher, W.J.; Rees, T.D.; Boyce, W.A. Complications of free grafts of masticatory mucosa. J. Periodontol. 1975, 46, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Rossmann, J.A.; Rees, T.D. A comparative evaluation of hemostatic agents in the management of soft tissue graft donor site bleeding. J. Periodontol. 1999, 70, 1369–1375. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.V.; Kumar, S.; Vidya, G.D.; Patel, A.; Holmes, J.C.; Kumar, V. Cytological assessment of healing palatal donor site wounds and grafted gingival wounds after application of ozonated oil: An eighteen-month randomized controlled clinical trial. Acta Cytol. 2012, 56, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Maeda, T.; Masaki, C.; Kanao, M.; Kondo, Y.; Ohta, A.; Nakamoto, T.; Hosokawa, R. Low-intensity pulsed ultrasound enhances palatal mucosa wound healing in rats. J. Prosthodont. Res. 2013, 57, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.W.C.; Liao, J.Y.; Liu, S.J.; Chan, E.C. Novel biodegradable sandwich-structured nanofibrous drug-eluting membranes for repair of infected wounds: An in vitro and in vivo study. Int. J. Nanomed. 2012, 7, 763–771. [Google Scholar]
- Malmquist, J.P.; Clemens, S.C.; Oien, H.J.; Wilson, S.L. Hemostasis of oral surgery wounds with the HemCon Dental Dressing. J. Oral. Maxillofac. Surg. 2008, 66, 1177–1183. [Google Scholar] [CrossRef] [PubMed]
- Cunha-Reis, C.; TuzlaKoglu, K.; Baas, E.; Yang, Y.; El Haj, A.; Reis, R.L. Influence of porosity and fibre diameter on the degradation of chitosan fibre-mesh scaffolds and cell adhesion. J. Mater. Sci. Mater. Med. 2007, 18, 195–200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, E.C.; Chou, T.C.; Gau, C.H.; Tu, H.P.; Chen, Y.T.; Fu, E. Releasing growth factors from activated human platelets after chitosan stimulation: A possible bio-material for platelet-rich plasma preparation. Clin. Oral Implant. Res. 2006, 17, 572–578. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Chiono, V.; Carmagnola, I.; Hatton, P.V. An overview of poly(lactic-co-glycolic) acid (PLGA)-based biomaterials for bone tissue engineering. Int. J. Mol. Sci. 2014, 15, 3640–3659. [Google Scholar] [CrossRef] [PubMed]
- Makadia, H.K.; Siegel, S.J. Poly Lactic-co-Glycolic Acid (PLGA) as Biodegradable Controlled Drug Delivery Carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef] [PubMed]
- Weisel, J.W. Structure of fibrin: Impact on clot stability. J. Thromb. Haemost. 2007, 5 (Suppl. 1), 116–124. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, M.R.; Thomas, B.S.; Varghese, J.M.; Bhat, G.S. Platelet-rich fibrin as an adjunct to palatal wound healing after harvesting a free gingival graft: A case series. J. Indian Soc. Periodontol. 2014, 18, 399–402. [Google Scholar] [PubMed]
- Shanmugam, M.; Kumar, T.S.; Arun, K.V.; Arun, R.; Karthik, S.J. Clinical and histological evaluation of two dressing materials in the healing of palatal wounds. J. Indian Soc. Periodontol. 2010, 14, 241–244. [Google Scholar] [PubMed]
- Kumbar, S.G.; Nukavarapu, S.P.; James, R.; Nair, L.S.; Laurencin, C.T. Electrospun poly(lactic acid-co-glycolic acid) scaffolds for skin tissue engineering. Biomaterials 2008, 29, 4100–4107. [Google Scholar] [CrossRef] [PubMed]



© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, F.-Y.; Lee, D.; Lee, T.-C.; Chen, J.-K.; Wu, R.-C.; Liu, K.-C.; Liu, S.-J. Fabrication of Multi-Layered Lidocaine and Epinephrine-Eluting PLGA/Collagen Nanofibers: In Vitro and In Vivo Study. Polymers 2017, 9, 416. https://doi.org/10.3390/polym9090416
Lee F-Y, Lee D, Lee T-C, Chen J-K, Wu R-C, Liu K-C, Liu S-J. Fabrication of Multi-Layered Lidocaine and Epinephrine-Eluting PLGA/Collagen Nanofibers: In Vitro and In Vivo Study. Polymers. 2017; 9(9):416. https://doi.org/10.3390/polym9090416
Chicago/Turabian StyleLee, Fu-Ying, Demei Lee, Tzu-Chia Lee, Jan-Kan Chen, Ren-Chin Wu, Kuan-Chieh Liu, and Shih-Jung Liu. 2017. "Fabrication of Multi-Layered Lidocaine and Epinephrine-Eluting PLGA/Collagen Nanofibers: In Vitro and In Vivo Study" Polymers 9, no. 9: 416. https://doi.org/10.3390/polym9090416




