1. Introduction
Peripheral nerve injuries arising from trauma, cancer, or congenital defects are challenging clinical issues as they affect an increasing number of people, with dramatic consequences such as the loss of sensory or motor function. Approximately 200,000 peripheral nerve repair procedures are performed every year in the United States alone, which causes a huge social burden [
1]. Direct end-to-end surgical reconnection is a common method for small injury gaps, while large nerve defects are normally treated with autologous nerve grafts. However, complete axonal reconnection and functional regeneration can rarely be achieved. Thus, tissue engineering scaffolds may serve as an alternative choice for peripheral nerve regeneration.
Electrospun nanofibers have been used extensively as substrates for neural tissue engineering and achieved great success [
2,
3,
4]. Various kinds of biomaterials such as poly(
l-lactide acid) (PLLA) [
5], poly(caprolactone) (PCL) [
6] and poly(lactic-
co-glycolic acid) (PLGA) [
7] have been applied to fabricate nanofibrous scaffolds for nerve regeneration using electrospinning technique. Additionally, conductive biomaterials such as polypyrrole (PPy) [
8], polyaniline (PANI) [
9] and Poly(3,4-ethylenedioxythiophene) (PEDOT) [
10] have also been used for scaffold fabrication for nerve regeneration and to achieve a certain level of conductivity. Carbon nanotubes (CNTs) are another group of conducting materials with unique structural, electrical, and mechanical properties. Multi-walled carbon nanotubes (MWCNTs) are composed of layers of graphite sheets with diameters of up to 100 nm, and this unique structure offers excellent physical and chemical properties which enable wide applications of MWCNTs. Numerous studies have investigated the effect of MWCNTs on neuronal behavior, especially on their ability to promote neurite extension and support neural network formation [
11]. Mattson et al. reported the biocompatibility of carbon nanotube for neuronal growth in vitro for the first time, and they demonstrated the ability of MWCNT-layered substrates to support the long-term survival of cultured dissociated hippocampal neurons [
12]. Jin et al. fabricated MWCNT-coated electrospun poly(
l-lactic acid-
co-caprolactone) (PLCL) nanofibers and found that the fibers improved neurite outgrowth of both PC12 cells [
13] and rat dorsal root ganglia (DRG) neurons [
14]. All these results suggest that MWCNTs could be used as a potential scaffold material to improve neural cells response including cell attachment, proliferation and differentiation for neuroregenerative applications.
CNTs were shown to be toxic to cells as a suspension in cell culture media [
15]. Most of the reported toxicological data are based on in vitro experiments with certain cell lines. The cytotoxicity can be regulated by functionalization and safety dosage [
16]. Modifications can enable CNTs increased water miscibility and reduced toxicity. Electrospinning is one of the most efficient ways to embed CNTs into fabricated composite nanomaterials [
17,
18]. In this present study, we aimed to fabricate PLGA nanofibers with various proportions of carboxyl modified MWCNTs (MWCNTs-COOH). The effects of MWCNTs-COOH on the properties of composite nanofibers, including morphology, hydrophilic performance, and mechanical properties were evaluated. Since PLGA served as the main component, and knowing the hydrophobic properties of PLGA, it was necessary to improve the surface properties. Plasma treatment is a favorable method to improve surface hydrophilicity and provide better biocompatibility of scaffolds in tissue engineering [
19]. Various functional chemical groups, such as the hydroxyl, the carboxyl group and the amino group can be introduced onto the surface of materials by different plasma modifying strategies, and then the surface properties such as wettability, surface roughness and cytocompatibility are changed [
20]. In this study, MWCNTs-COOH-containing nanofibers were further treated with air plasma. The change in surface chemistry and hydrophilicity of the scaffolds were characterized by Energy Dispersive X-ray Detector (EDX) and water contact angle, respectively. Furthermore, the influence of plasma treatment on PC12 cells adhesion was investigated. The effect of plasma treated nanofibrous scaffolds with various ratios of MWCNTs-COOH, on the cellular responses of PC12 cells, DRG neurons and Schwann cells, such as cell attachment, proliferation and differentiation were further assessed to investigate the potential of these scaffolds for PNS repair.
2. Materials and Methods
2.1. Materials
The 75/25 poly(lactic-co-glycolic acid) (PLGA) (inherent viscosity 0.75 dL/g) was purchased from Jinan Daigang Biomaterial Co., Ltd., Jinan, China. 1,1,1,3,3,3-Hexafluoro-2-propanol (HFP), glutaraldehyde, Dulbecco’s modified eagle’s medium (DMEM/F12), were purchased from Sigma, St. Louis, MO, USA. Graphitized Multi-Wall Carbon Nanotubes COOH (out diameter: 10–20 nm; length: 1–30 μm; purity: >99.9 wt %; COOH content: 1.0–2.0 wt %) was purchased from Cheaptubes, Cambridgeport, VT, USA. Nerve growth factor (NGF) was purchased from Millipore, Singapore. Rat pheochromocytoma (PC12) cells in the adherent type and rat Schwann cells were obtained from ATCC, Manassas, VA, USA, while fetal bovine serum (FBS), horse serum (HS) and trypsin/EDTA were purchased from GIBCO Invitrogen, Thermo Fisher, Waltham, MA, USA. Alamar Blue (AbD Serotec) was purchased from Chemoscience, Singapore. Schwann cell medium was purchase from Gene-Ethics Asia Pte Ltd., Singapore. Anti-S100 antibody produced in rabbit and anti-NF200 produced in rabbit were purchased from Sigma.
2.2. Scaffolds Fabrication
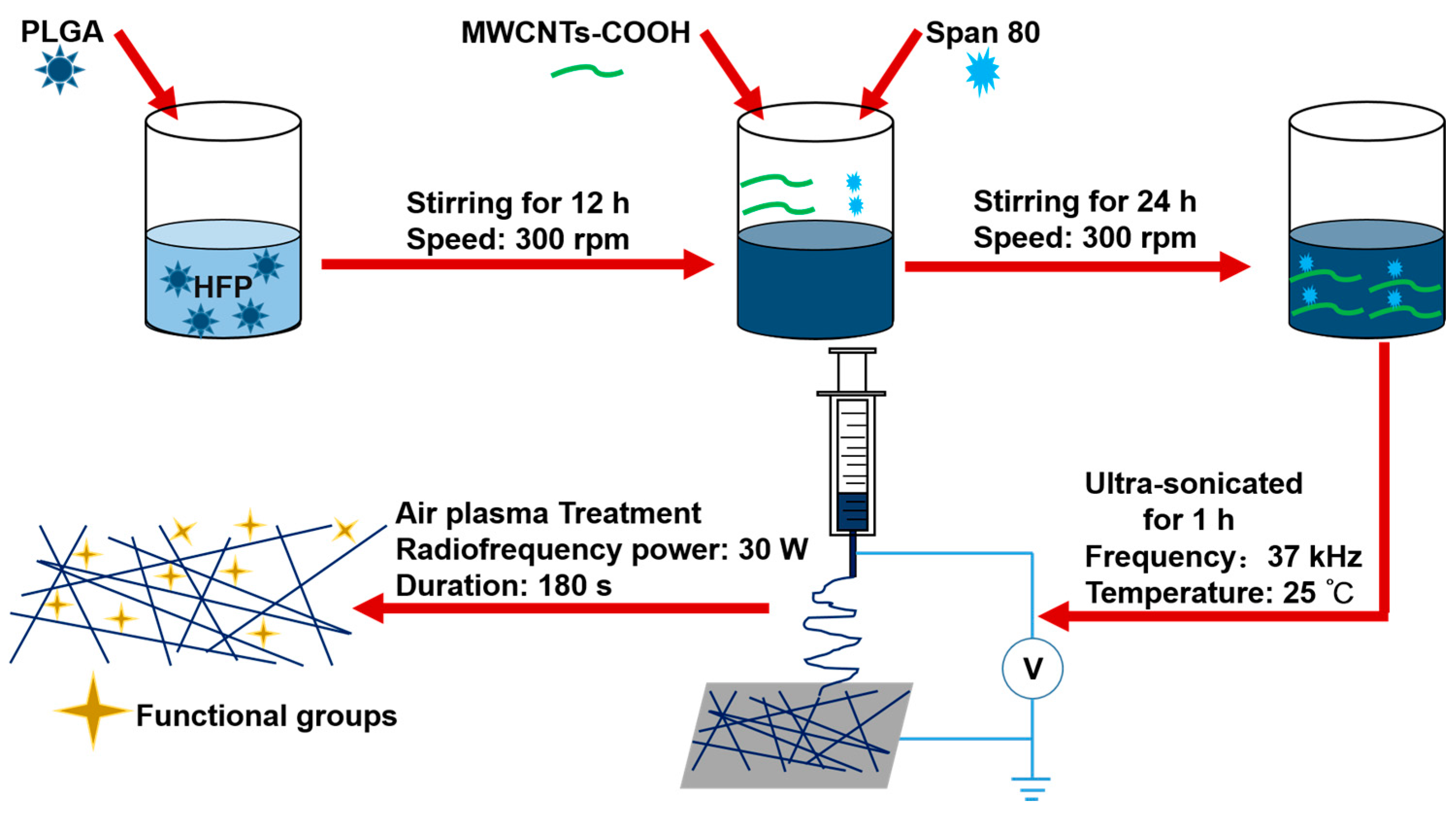
2.2.1. Preparation of the Electrospun Suspensions
PLGA was dissolved into 1,1,1,3,3,3-Hexafluoro-2-propanol (HFP) to form a 25% w/v solution with magnetic stirring of 300 rpm for 12 h. Then the composite PLGA/MWCNTs-COOH suspensions with different ratios of MWCNTs-COOH were obtained by mixing MWCNTs-COOH powder with PLGA solution with a mass ratio of 2:100, 4:100, 8:100 (MWCNTs-COOH:PLGA). Additionally, 100 μL of span 80 was added to improve the dispersion of MWCNTs-COOH. Subsequently, the suspensions were stirred continuously for 24 h. Finally, the suspensions were ultra-sonicated for 1 h before electrospinning.
2.2.2. Electrospinning
The electrospinning process was the same as that described in previous work [
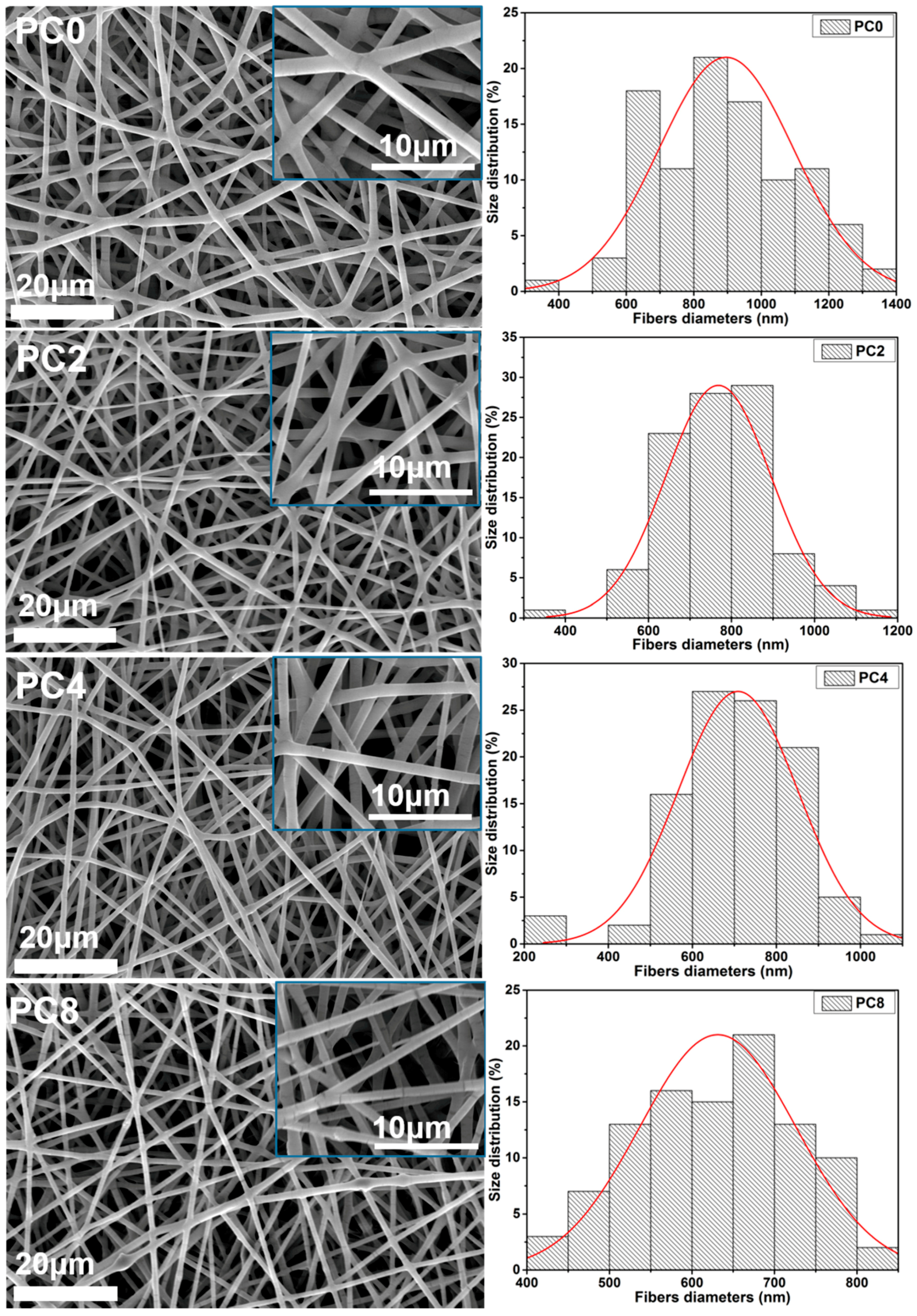
21]. The solution for electrospinning was loaded into a 3 mL syringe with an 18 G blunt stainless needle (diameter 0.84 mm). The high voltage electrostatic generator was supplied by Gamma High Voltage, Ormond Beach, FL, USA. The high voltage for electrospinning was 18 kV, and the flow rate was 1 mL/h. The fabricated nanofibers was collected on a plate covered with aluminum foil, and the distance between the collector and the needle tip was 15 cm. The obtained nanofibrous scaffolds with various proportions of MWCNTs-COOH were dried in a vacuum drying oven (DZF-6050, Liyi, Guangzhou, China) for 24 h and then used for further study. The temperature in the vacuum oven was room temperature. Only the effect of the vacuum was used to further dry the scaffolds. Herein, the labels PC2, PC4 and PC8 correspond to nanofibrous scaffolds with MWCNTs-COOH content of 2, 4 and 8%, respectively. Pure PLGA scaffolds were also fabricated as a control, and labeled as PC0.
2.2.3. Optimization of Plasma Treatment Conditions
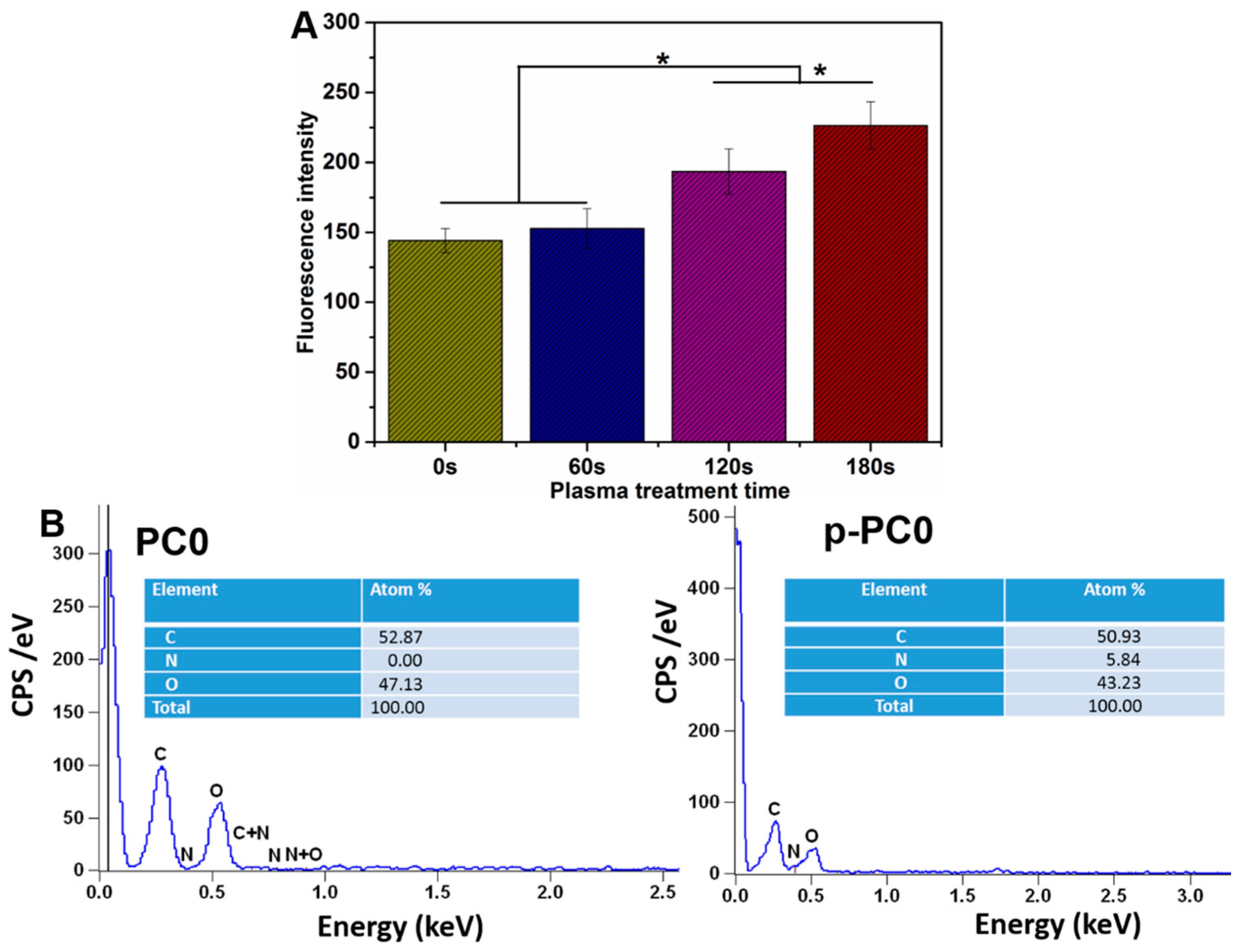
In order to further improve the surface hydrophilicity of the fabricated scaffolds, air plasma treatment was conducted for the scaffolds by a plasma cleaner (Model: PDC-001, Harrick Scientific Corporation, Pleasantville, NY, USA). PC0 scaffolds were used to optimize the treatment duration. The radiofrequency power was set as 30 W under vacuum. The effect of air plasma treatment on the scaffold was controlled by changing the processing period. So, plasma discharge was applied to PC0 scaffolds with various durations, including 60, 120, 180 s. The optimized processing period for air plasma treatment was determined by the evaluation results of plasma treated nanofibers by SEM, water contact angle and tensile mechanical properties. Then the scaffolds with MWCNTs-COOH content of 0, 2, 4 and 8% to be used for further study, were all treated with air plasma using the optimized duration, and the obtained samples were labelled as p-PC0, p-PC2, p-PC4 and p-PC8.
2.3. Scaffold Characterization
2.3.1. Scanning Electron Microscopy (SEM)
The electrospun nanofibers were gold-coated using sputter coating (JEOL JFC-1200 Fine coater, Tokyo, Japan) and the surface topographies were visualized by SEM (Model S-4300, Hitachi, Tokyo, Japan). The gold-coating process was controlled by the current and period. Here, the current for gold-coating was 10 mA, and the process lasted for 80 s. The diameter distribution of the nanofibers was determined from the SEM images using image analysis software (ImageJ, National Institutes of Health, Bethesda, MD, USA). 100 nanofibers were chosen from the SEM images for the diameter measurement.
2.3.2. Transmission Electron Microscopy (TEM)
The distribution of MWCNTs-COOH in the fibers was observed using transmission electronic microscopy (JEM-3010, JEOL, Tokyo, Japan) at 200 kV. The samples for TEM observation were prepared by collecting the fibers onto a carbon film-coated copper grid directly for about 8 s. Then the samples were dried in a vacuum drying oven for 24 h to remove residual solvent completely.
2.3.3. Water Contact Angle
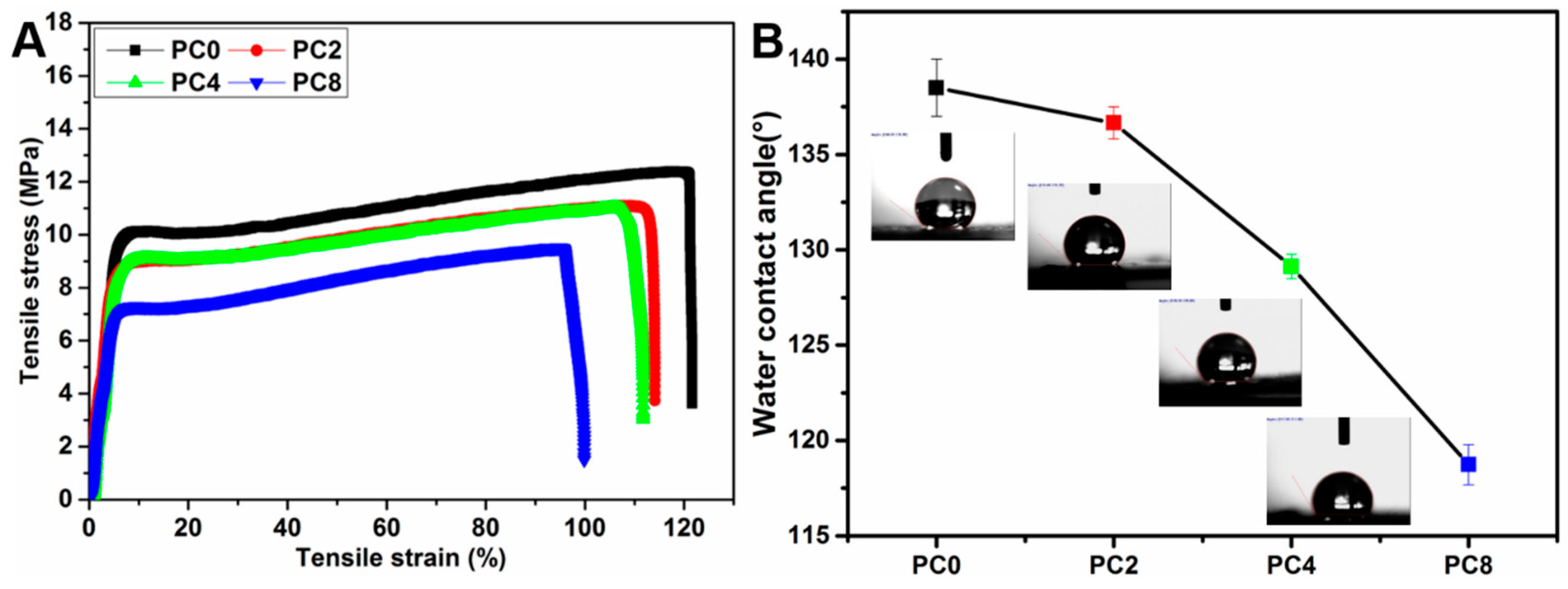
Hydrophilicity of the nanofibers with various ratios of MWCNTs-COOH and pure PLGA nanofibers with different plasma treatment duration was measured by water contact angle using VCA Optima Surface Analysis System (AST products, Billerica, MA, USA). The detailed steps were the same as those described in previous work [
22]. Firstly, the nanofiber mat was placed on the testing plate and kept smooth. Subsequently, 0.05 mL of distilled water was dropped slowly onto the surface of the nanofiber mat. The images of the water drop on the nanofiber mat were recorded by a camera software in the testing system after the droplet was stable. Then, the water contact angle was measured with the measuring tool in the testing system. Three different points of each sample were measured, and the average value was calculated.
2.3.4. Surface Chemistry Analysis
The surface chemistry of pure PLGA nanofibers before and after plasma treatment was analyzed by energy-dispersive X-ray (EDX). The changes in three kinds of elements including carbon, oxygen and nitrogen on the fiber surface were evaluated with EDX. The nanofibers were visualized under SEM without any melt coating. EDX analysis was done by Quantax 70 (Bruker, Berlin, Germany) mounted on a SEM.
2.3.5. Mechanical Properties
The mechanical properties of the nanofibers with various ratios of MWCNTs-COOH and pure PLGA nanofibers with various plasma treatment duration were measured with stress-strain analysis performed using a universal materials tester (H5K-S, Hounsfield, Salfords, UK) at room temperature. The samples were cut into rectangular specimens with dimensions of 30 mm × 10 mm. Then, the thickness was measured with a digital micrometer (Digimatic Micrometer Series 293 MDC-MX Lite, Mitutoyo, Kawasaki, Japan). Subsequently, two sides of the rectangular specimen were fixed with the two clamps of the tester. After that, mechanical testing was conducted with a stretch speed of 10 mm/min. The stretch was stopped until the specimen was completely broken. The reported data of tensile strength, elongation, Young’s Modulus and toughness represent the average results of six tests.
2.4. PC12 Cell Culture In Vitro
PC12 cells were cultured using the same method as described in previous work [
1]. Briefly, PC12 cells were cultured in growth medium which was composed of 84% DMEM/F12, 10% HS, 5% FBS, and 1% antibiotic/antimycotic solution in a 75 cm
2 cell culture flask. Cells were incubated in a CO
2 Incubator (MCO-19 AIC (UV), Panasonic Healthcare, Gunma, Tokyo, Japan) at 37 °C and 5% CO
2. The culture medium was changed once every 2 days. After the cells were grown to confluency, they were detached with trypsin/EDTA and seeded on different nanofibrous scaffolds with a density of 20,000 cells per well for proliferation and differentiation tests and 30,000 cells per well for the adhesion test.
2.5. The Adhesion of PC12 Cells on PC0 Nanofibers with Different Plasma Treatment Duration
Plasma treatment was applied to improve the hydrophilicity and biocompatibility of the electrospun nanofibers. In order to determine the optimized treatment duration, adhesion of PC12 cells on pure PLGA nanofibers with various plasma treatment duration was evaluated. The PC12 cells used here were adherent type. Cell adhesion is the first and most critical step that occurs after seeding the cells on the scaffolds, followed by cell proliferation and differentiation. The adhesion of PC12 cells was evaluated by counting the number of cells attached on the scaffold, while the cell number was measured with Alamar blue assay as specified by the manufacturer. The cells were allowed to be attached to the scaffold for 12 h after cell seeding. Then, the medium was removed and the scaffold with cells was rinsed thrice with PBS to remove the unattached cells. The number of cells attached on the scaffold was further evaluated with Alamar blue. In detail, 800 μL Alamar blue solution (10% Alamar blue, 90% DMEM/F12 medium; v/v) was added to each well followed by incubation at 37 °C for 4 h. Then the Alamar blue solution was pipetted into 96-well plate and the fluorescence at 560 nm (excitation)/590 nm (emission) was read by a Varioskan flash reader (Thermo Fisher Scientific, Waltham, MA, USA). The number of cells was positively proportional to the fluorescence intensity.
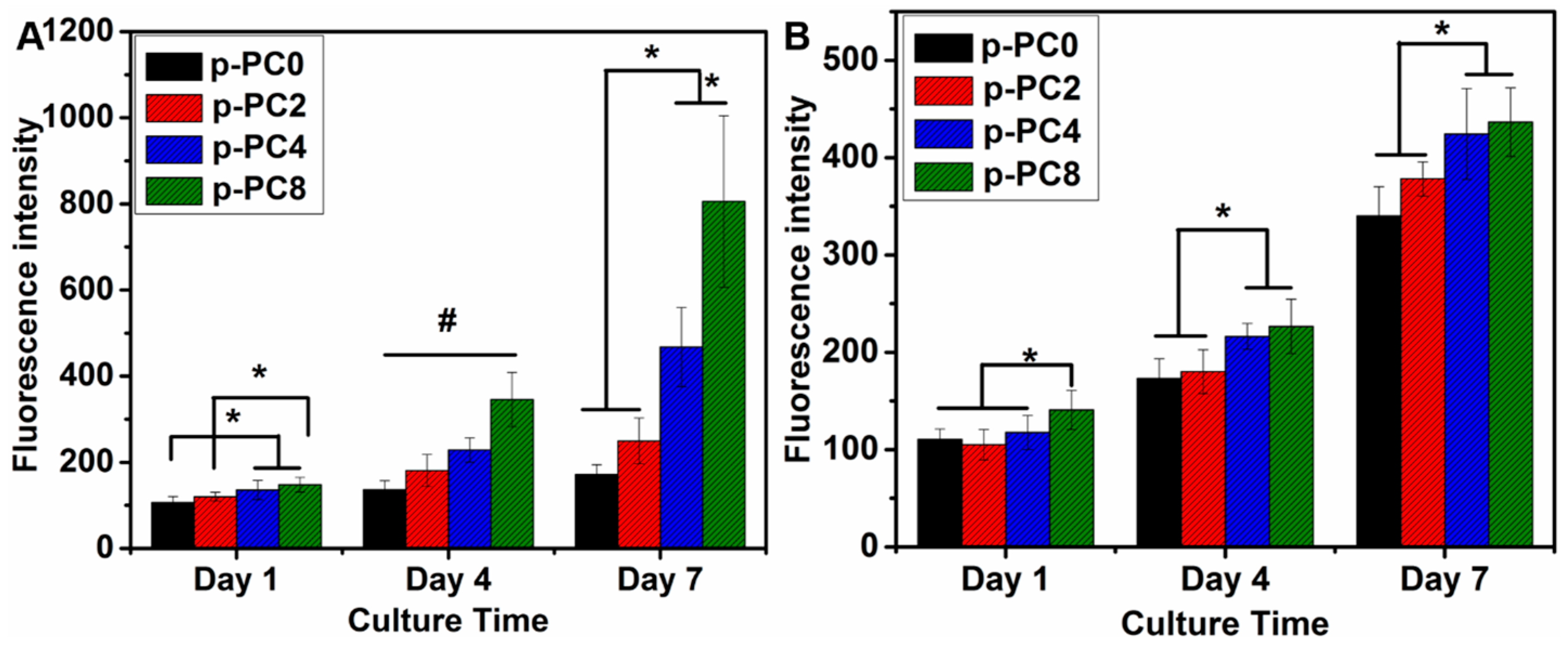
2.6. The Proliferation of PC12 and Schwann Cells
The optimized plasma treatment time was determined based on the above test results. In order to further evaluate the influence of nanofibers with various content of MWCNTs-COOH on nerve cell behavior, all samples for the following research were treated with air plasma for an optimized duration.
Cell proliferation of both PC12 and Schwann cells were evaluated with Alamar blue assay as described above. The cells were seeded on p-PC0, p-PC2, p-PC4, p-PC8 nanofibrous scaffolds which were sterilized for 5 h under UV light in a biological safety cabinet (Bioclassic Class II Series Type A2, Gelman Laminar Airflow Systems, Singapore). The UV lamp for sterilization came with the biological safety cabinet. The UV length was 250–260 nm. After seeding on the scaffolds, the cell culture plate was put into the CO2 incubator (MCO-19AIC (UV), Panasonic Healthcare, Gunma, Japan) at 37 °C and 5% CO2. The culture medium was changed once every 2 days. The cell proliferation was tested after 1, 4 and 7 days of culture in vitro.
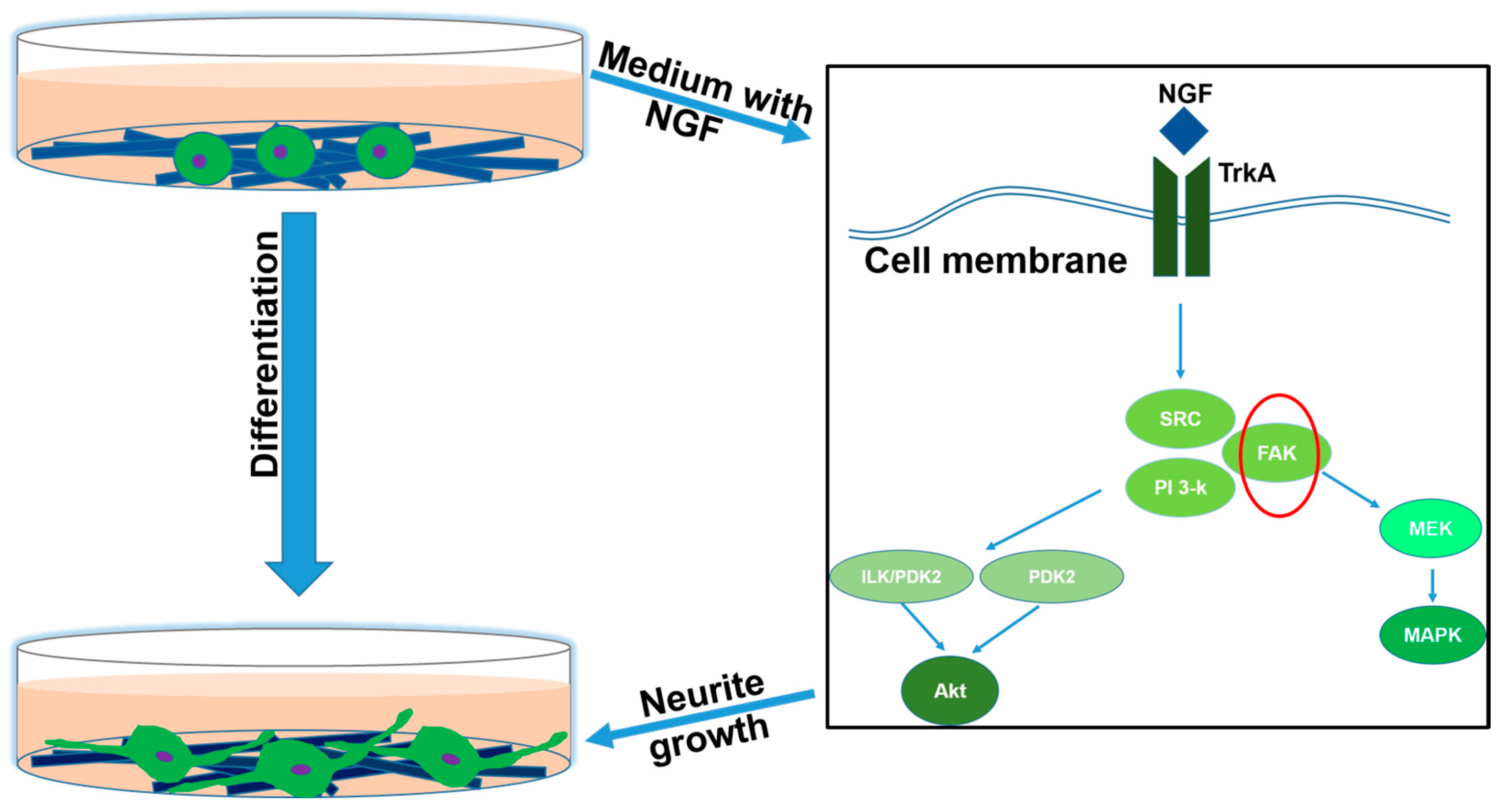
2.7. The Neuronal Differentiation of PC12 Cells
For the differentiation study of PC12 cells cultured on p-PC0, p-PC2, p-PC4, p-PC8 nanofibrous scaffolds, the medium used was changed to a differential medium composed of DMEM/F12 supplemented with 1% HS, 0.5% FBS, 1% antibiotic/antimitotic solution and 100 ng mL−1 NGF 1 day after cell seeding.
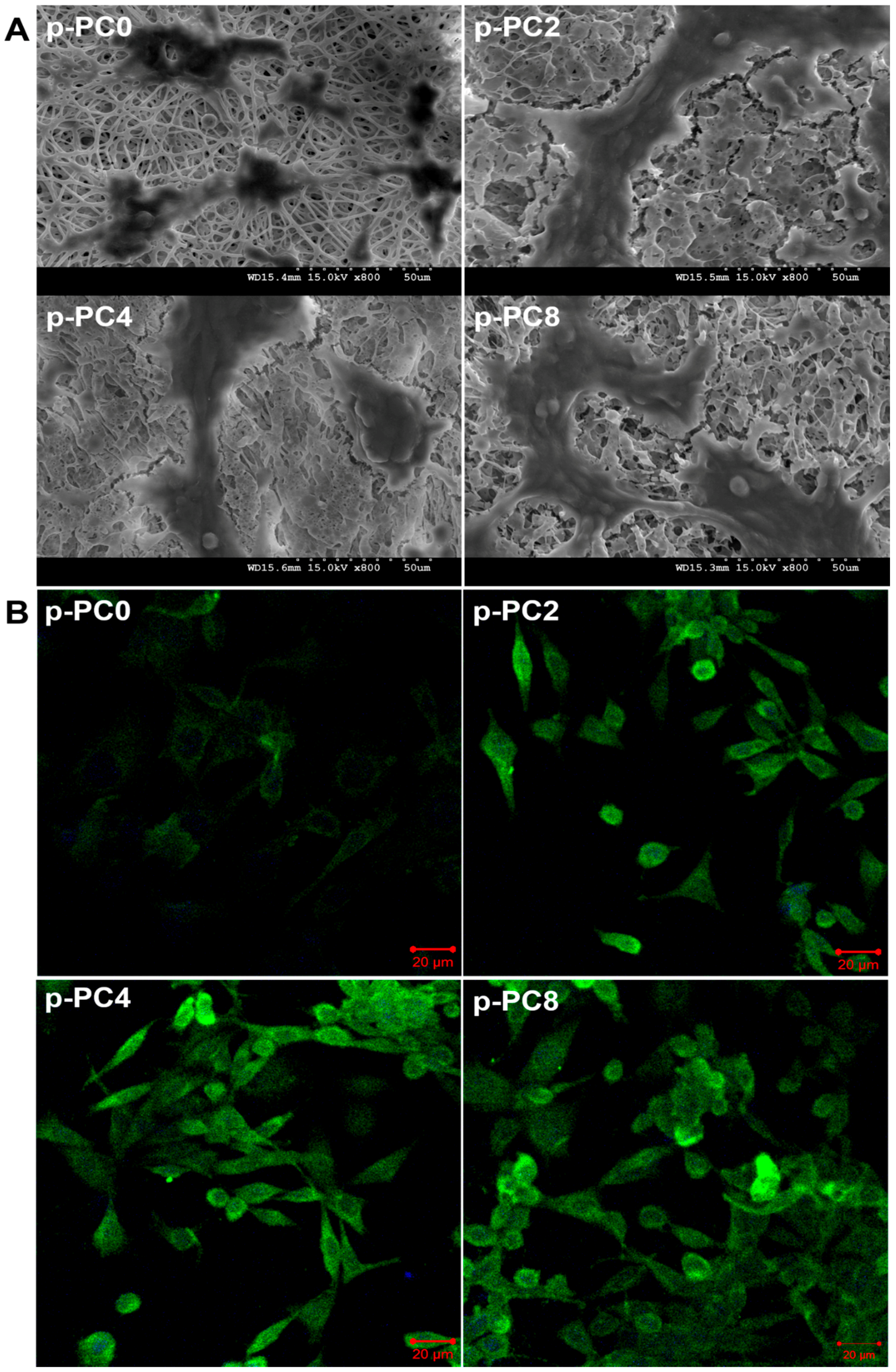
2.7.1. Morphology of Differentiated PC12 Cells
SEM was applied to evaluate the morphology of differentiated PC12 cells cultured on different scaffolds. Firstly, the scaffolds with cells were washed twice with PBS and fixed with 10% formalin for 20 min at room temperature (about 25 °C). The PBS containing 137 mM sodium chloride, 2.7 mM potassium chloride and 10 mM phosphate buffer used here was pH 7.4 ± 0.2. After that, the scaffold-cell constructs were rinsed 3 times with 1 mL distilled water, and dehydrated through a series of graded ethanol (10, 70, 90, and 100%). Finally, the samples were air-dried for 12 h, sputter-coated with gold, and observed by SEM.
2.7.2. Immunostaining of Neurofilament 200 (NF200)
The neural differentiation of PC12 cells was further evaluated by immunofluorescence staining of a nerve specific protein-NF200. After 7 days of cell culture in differentiation medium on different scaffolds, the cell-scaffolds constructs were rinsed thrice with PBS and then fixed with 10% formalin for 20 min. After that, the samples were permeabilized with 0.1% Triton X-100 for 5 min followed by incubation with 3% w/v BSA aqueous solution for 90 min to block the nonspecific binding. Subsequently, the sample were stained with anti-NF200 produced in rabbit as the primary antibody at dilution of 1:100 (Sigma) for 2 h at room temperature. The anti-Rabbit IgG (whole molecule)–FITC antibody was diluted 200 times and used as the secondary antibody to combine with the primary antibody for 1 h. The immunostained samples were mounted onto glass slides with mounting medium with DAPI and observed under laser scanning confocal microscope (Zeiss LSM700, Oberkochen, Germany).
2.8. Neurite Outgrowth of Dorsal Root Ganglia (DRG) Neurons
To assess the neurite outgrowth of DRG neurons, rat DRG neurons were seeded on different scaffolds at a density of 10,000 cells per well in a 24-well cell culture plate, and cultured in DMEM/F12 with 10% FBS and 50 ng mL−1 NGF in a 37 °C, 5% (v/v) CO2 incubator for 5 days. For immunofluorescence staining of specific protein-NF160, the cells were fixed with 10% formalin for 20 min, treated with 0.1% Triton X-100 for 5 min, and blocked with 3% w/v BSA aqueous solution for 90 min. Then the samples were immunostained with the primary antibody solution overnight at 4 °C, followed by incubation with Goat anti-Rabbit IgG Alexa 488 (Invitrogen, Camarillo, CA, USA) diluted 1:200 in PBS as the secondary antibody for 1 h. After staining with DAPI for 15 min, the outgrowth of immunostained DRG neurons was visualized under LSCM.
2.9. Morphologies of Schwann Cells
The morphological analysis of Schwann cells on p-PC0, p-PC2, p-PC4, p-PC8 nanofibrous scaffolds was evaluated with immunofluorescence staining for S100 protein, a commonly used astrocyte marker. Cells cultured on different scaffolds were fixed with 10% formalin at day 5 followed by permeabilization with 0.1% Trixton-X100 for 5 min. Then, the nonspecific binding was blocked with 3% w/v BSA aqueous solution for 90 min. After that, the samples were incubated with rabbit anti-S100 antibody (dilution 1:100; Sigma) at 4 °C overnight. Subsequently, samples were stained with FITC conjugated secondary antibody (dilution 1:100; Sigma) at room temperature for 1 h, and nuclei was stained with DAPI for 15 min. Finally, the samples were mounted onto glass slides and visualized under a laser scanning confocal microscope (LSCM, Zeiss LSM700).
2.10. Statistical Analysis
Statistical analysis was performed using ANOVA and Student’s t-test. All data were expressed as mean ± standard deviation (SD). In all statistical evaluations, p < 0.05 was considered statistically significant.
4. Conclusions
The enhancement of nerve cell behaviors, such as adhesion, proliferation, differentiation and maturation, should be taken into account in the design of peripheral nerve tissue engineered scaffolds. In this study, we prepared PLGA nanofibrous scaffolds containing various proportions of MWCNTs-COOH via electrospinning, and air plasma treatment was applied to improve the surface hydrophilicity of the scaffolds. Plasma treated scaffolds were more favorable to PC12 cells adhesion. The addition of MWCNTs-COOH in scaffolds was beneficial to PC12 cell proliferation, neuronal differentiation, and Schwann cell maturity. The p-PC8 nanofibrous scaffold provided a superior substrate for neural differentiation of PC12 cells and neurite extension of rat DRG neurons. Collectively, p-PC8 nanofibers could be a promising tissue engineered scaffold for peripheral nerve regeneration.