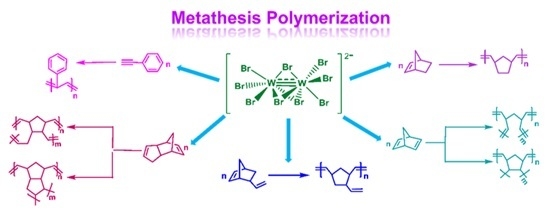
Metathesis Polymerization Reactions Induced by the Bimetallic Complex (Ph4P)2[W2(μ-Br)3Br6]
Abstract
:1. Introduction

2. Materials and Methods
2.1. General
2.2. Catalytic Reactions
3. Results and Discussion
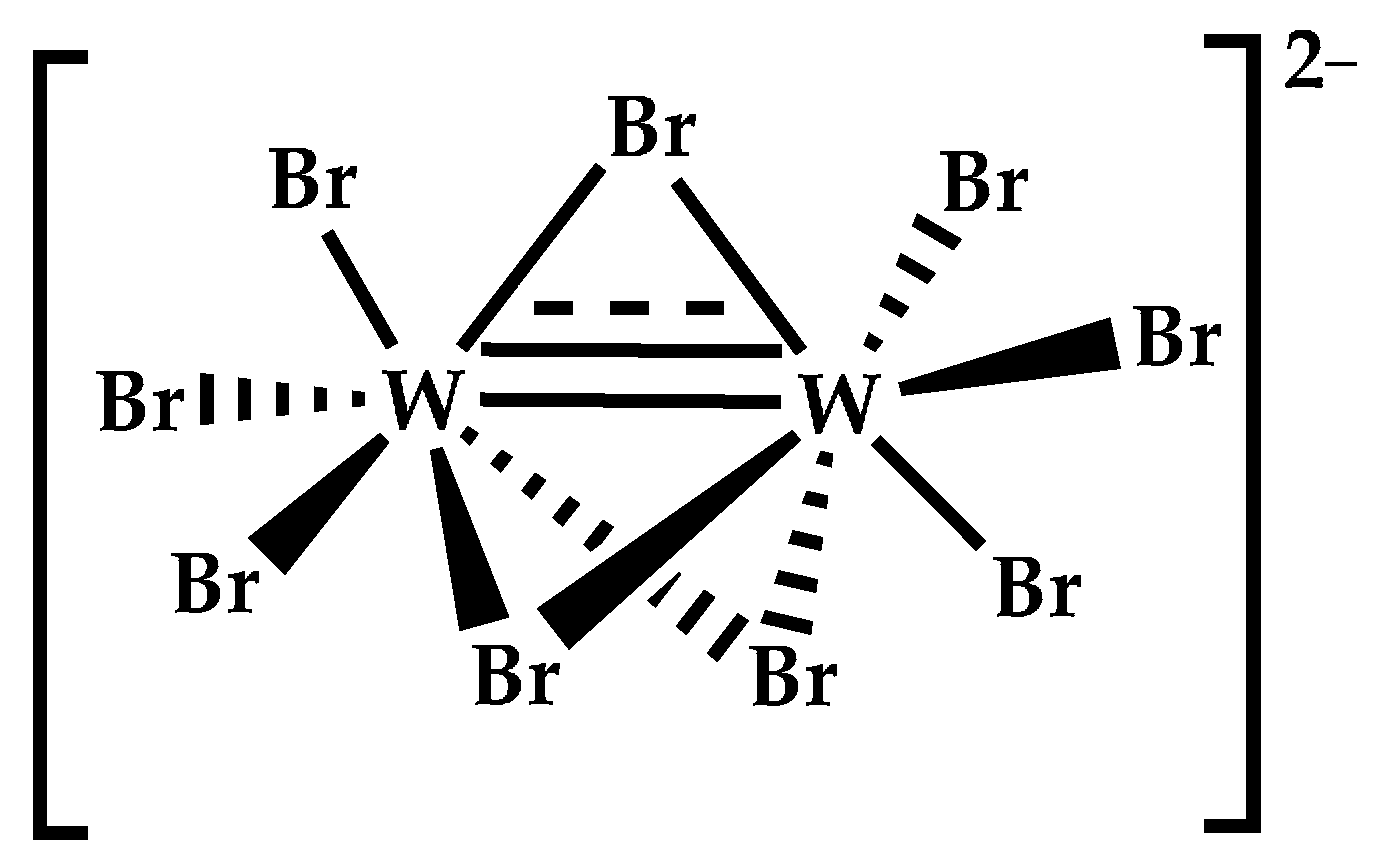
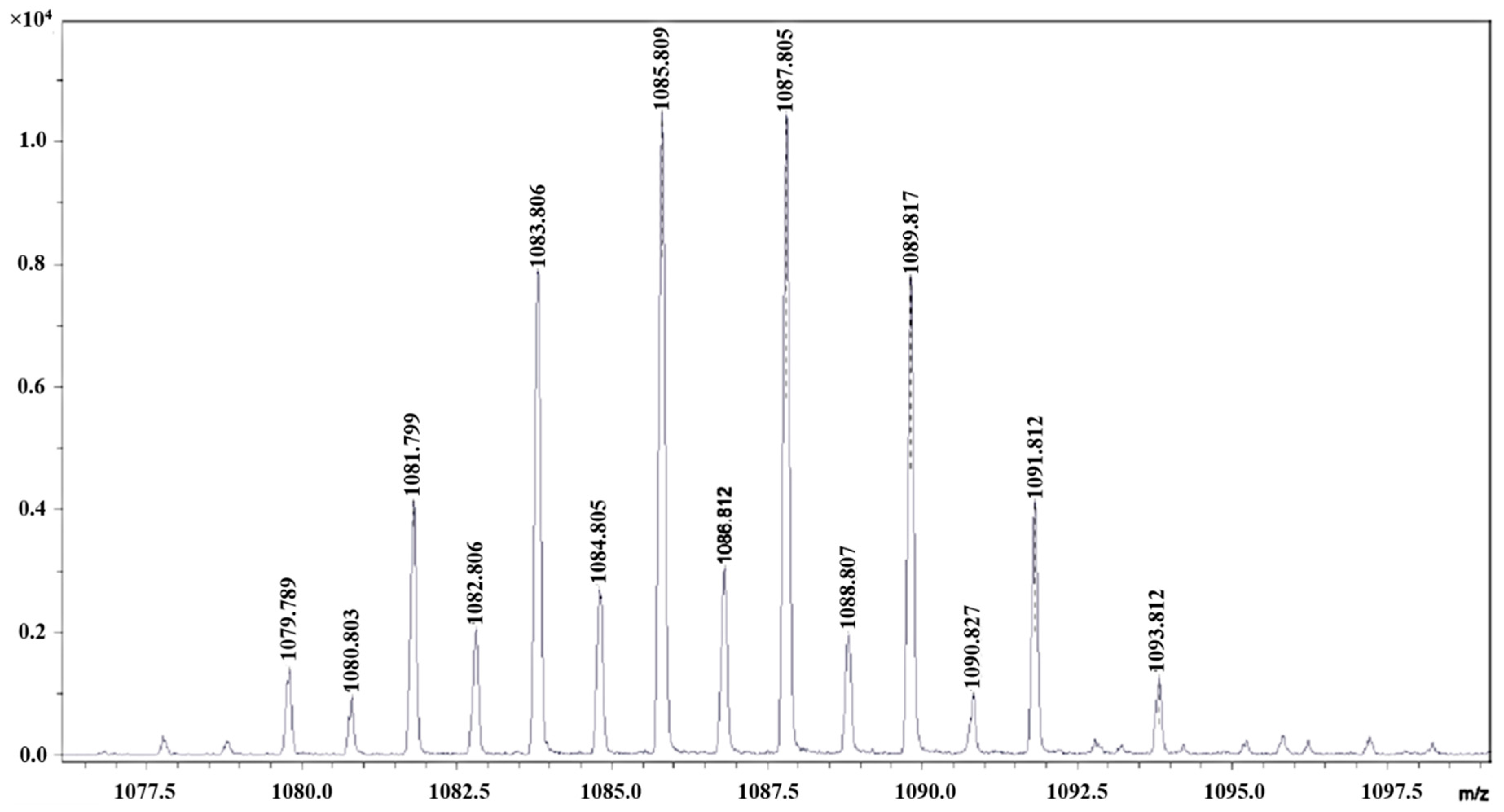
3.1. Catalyst Synthesis and Characterization
| Fragment | m/z (Theoretical) | m/z (Experimental) |
|---|---|---|
| Ph4P+ | 339 | 339.213 |
| [(Ph4P)3W2Br9]+ | 2104 | 2106.932 |
| [W2Br9]− | 1086 | 1085.809 |
| [Ph4PW2Br9]− | 1427 | 1427.563 |
| [WBr5]− | 583 | 582.207 |
| [W2Br7]− | 926 | 925.977 |
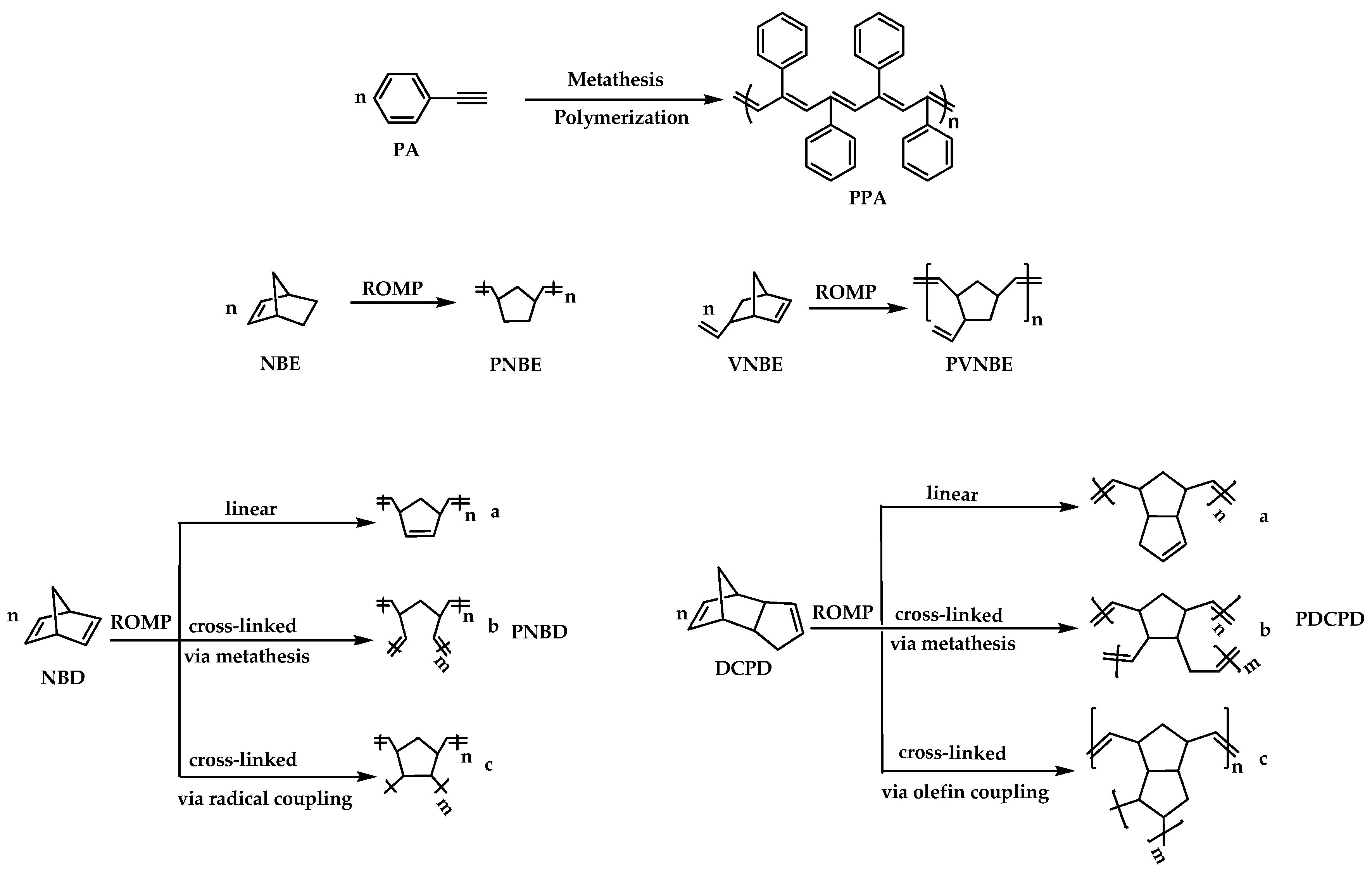
3.2. Polymerization Reactions
| Entry | Solvent | 1/AgBF4/PA Molar ratio | t (h) | Yield (%) | Mn × 10−3 e | Mw/Mn e | cis (%) f |
|---|---|---|---|---|---|---|---|
| 1 | CH2Cl2 | 1/2/350 a | 24 | 9 | 5.2 | 1.45 | 0 |
| 2 | CH2Cl2 | 1/3/350 b | 24 | 12 | 4.6 | 1.37 | 0 |
| 3 | CH2Cl2 | 1/4/350 c | 24 | 20 | 2.8 | 1.89 | 0 |
| 4 | THF | 1/2/350 a | 8 | 4 | 41.8 | 1.86 | 75 |
| 5 | THF | 1/2/350 a | 16 | 88 | 67.8 | 1.60 | 77 |
| 6 | THF | 1/2/350 a | 24 | 95 | 59.5 | 1.58 | 88 |
| 7 | THF | 1/3/350 b | 2 | 13 | 70.5 | 1.72 | 85 |
| 8 | THF | 1/3/350 b | 6 | 98 | 207 | 1.16 | 87 |
| 9 | THF | 1/3/350 b | 8 | 79 | 54.2 | 1.61 | 85 |
| 10 | THF | 1/3/350 b | 24 | 59 | 16.0 | 2.07 | 85 |
| 11 | Toluene | 1/3/350 b | 24 | - d | - d | - d | - d |
| 12 | CH3CN | 1/3/350 b | 24 | - d | - d | - d | - d |
| Entry | Solvent | 1/AgBF4/NBE Molar ratio | [NBE] (M) | t (h) | Yield (%) | Mn × 10−3 g | Mw/Mn g | cis (%) h |
|---|---|---|---|---|---|---|---|---|
| 1 | CH2Cl2 | 1/2/350 a | 0.6 | 24 | 6 | 103.8 | 1.60 | 65 |
| 2 | CH2Cl2 | 1/3/350 b | 0.6 | 24 | 8 | 82.3 | 1.80 | 68 |
| 3 | CH2Cl2 | 1/4/350 c | 0.6 | 24 | 22 | 186.7 | 1.25 | 62 |
| 4 | CH2Cl2 | 1/4/500 d | 0.8 | 1 | 15 | 810.5 | 1.34 | 42 |
| 5 | CH2Cl2 | 1/4/500 d | 0.8 | 2 | 94 | 547.1 | 1.28 | 48 |
| 6 | CH2Cl2 | 1/4/500 e | 1.7 | 0.4 | 98 | - i | - i | - i |
| 7 | THF | 1/4/500 e | 1.7 | 24 | - f | - f | - f | - f |
| 8 | CH3CN | 1/4/500 e | 1.7 | 24 | - f | - f | - f | - f |
| Entry | Solvent | 1/AgBF4/VNBE Molar ratio | [VNBE] (M) | t (h) | Yield (%) | Mn × 10−3 f | Mw/Mn f |
|---|---|---|---|---|---|---|---|
| 1 | CH2Cl2 | 1/4/500 a | 0.8 | 22 | 10 | - | - |
| 2 | CH2Cl2 | 1/4/500 b | 2.7 | 22 | 20 | - | - |
| 3 | CH2Cl2 | 1/4/500 c | 4.9 | 22 | 17 | - | - |
| 4 | CH2Cl2 | 1/4/1000 d | 2.3 | 26 | Traces | - | - |
| 5 | CH2Cl2 | 1/4/2000 e | 4.9 | 7 | 87 | 8.0 | 1.27 |
| 6 | - | 1/4/2000 e | - | 23 | 39 | 16.7 | 1.16 |
| Entry | Solvent | 1/AgBF4/NBD Molar ratio | [NBD] (M) | t (h) | Yield (%) |
|---|---|---|---|---|---|
| 1 | CH2Cl2 | 1/2/500 a | 1.7 | 0.75 | 72 |
| 2 | CH2Cl2 | 1/4/500 b | 1.7 | 1.5 | 92 |
| 3 | CH2Cl2 | 1/4/500 c | 3.4 | 21 | 98 |
| 4 | CH2Cl2 | 1/8/500 d | 1.7 | 4.5 | 95 |
| 5 | THF | 1/4/500 b | 1.7 | 22 | - h |
| 6 | THF | 1/4/500 c | 3.4 | 22 | 42 |
| 7 | Toluene | 1/4/500 b | 1.7 | 23 | traces |
| 8 | Toluene | 1/4/1000 e | 3.4 | 23 | traces |
| 9 | Toluene | 1/-/1000 g | 3.4 | 4.5 | 80 |
| 10 | Toluene | 1/-/1000 f | 3.4 | 4.5 | traces |
| 11 | - | 1/4/500 b | - | 22 | traces |
| 12 | - | 1/4/1000 e | - | 3 | traces |
| 13 | - | 1/-/1000 g | - | 22 | traces |
| Entry | Solvent | 1/AgBF4/DCPD Molar ratio | [DCPD] (M) | t (h) | Yield (%) |
|---|---|---|---|---|---|
| 1 | CH2Cl2 | 1/4/250 a | 0.8 | 16 | 95 |
| 2 | CH2Cl2 | 1/4/500 b | 1.4 | 4 | 43 |
| 3 | CH2Cl2 | 1/4/750 c | 1.4 | 22 | 66 |
| 4 | CH2Cl2 | 1/4/750 d | 2.0 | 4 | 9 |
| 5 | CH2Cl2 | 1/4/1000 e | 2.6 | 22 | 15 |
| 6 | Toluene | 1/4/250 a | 0.8 | 22 | Traces |
| 7 | Toluene | 1/4/500 b | 1.4 | 22 | 53 |
| 8 | Toluene | 1/4/750 d | 2.0 | 22 | 52 |
| 9 | Toluene | 1/4/1000 e | 2.6 | 22 | 30 |
| 10 | - | 1/4/250 a | - | 22 | 37 |
| 11 | - | 1/4/500 b | - | 22 | 10 |
| 12 | - | 1/4/750 d | - | 21 | 17 |
| 13 | - | 1/4/1000 e | - | 21 | 47 |
| 14 | Toluene | 1/-/750 f | 2.1 | 24 | 10 |
| 15 | Toluene | 1/-/1250 f | 3.0 | 24 | 10 |
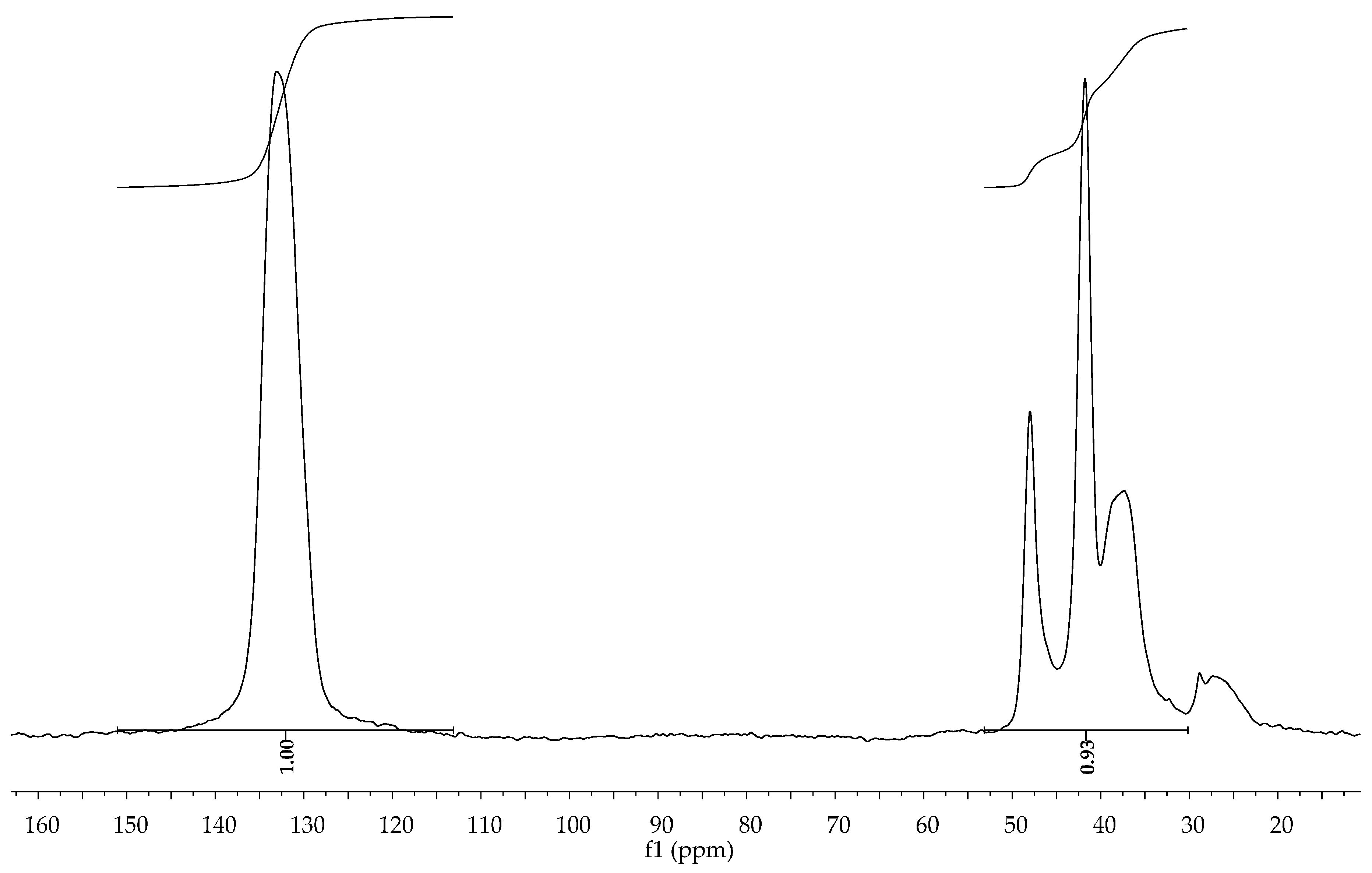
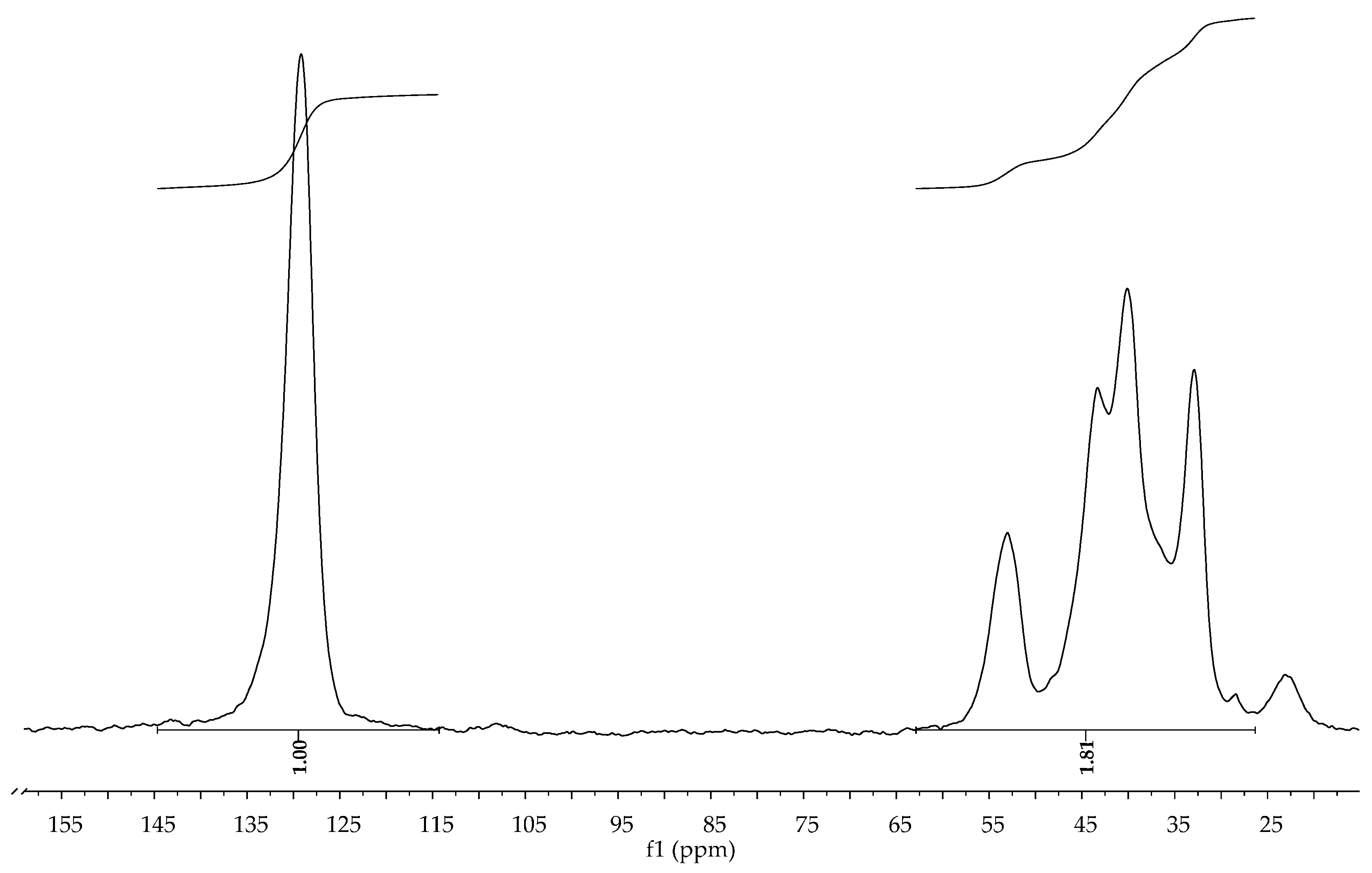
3.3. Kinetic Studies
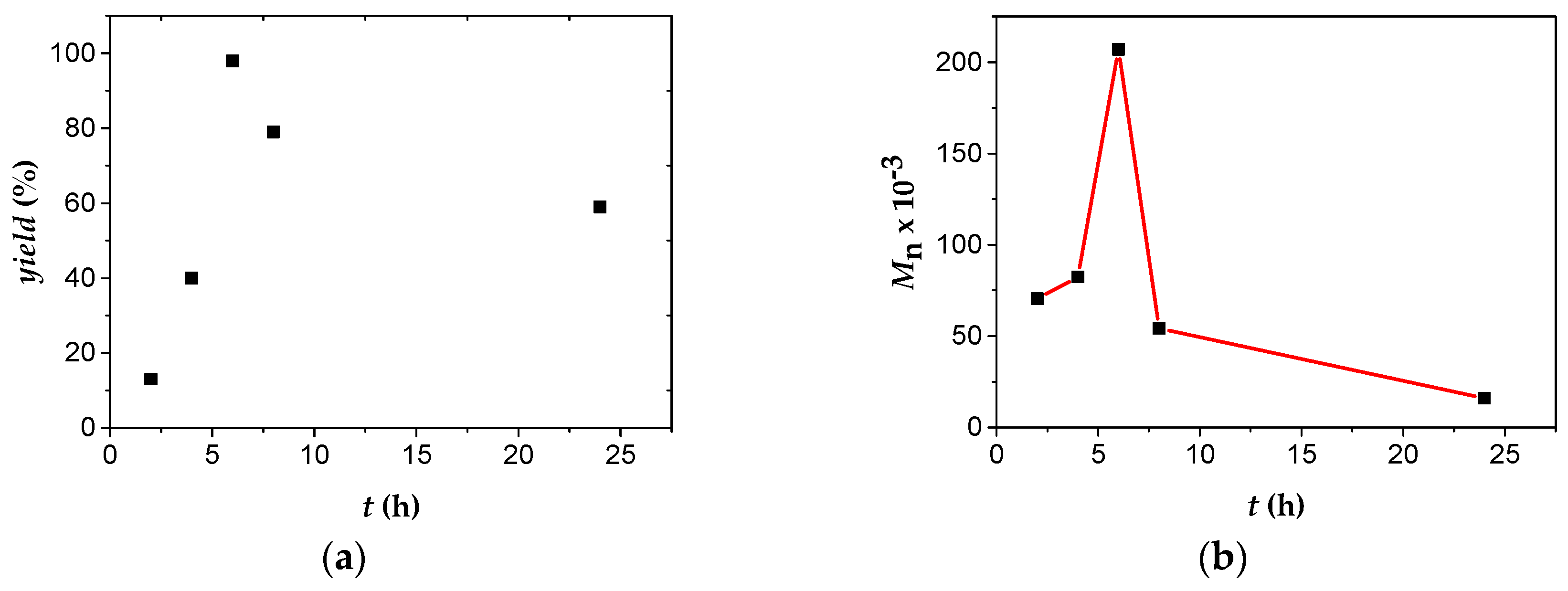
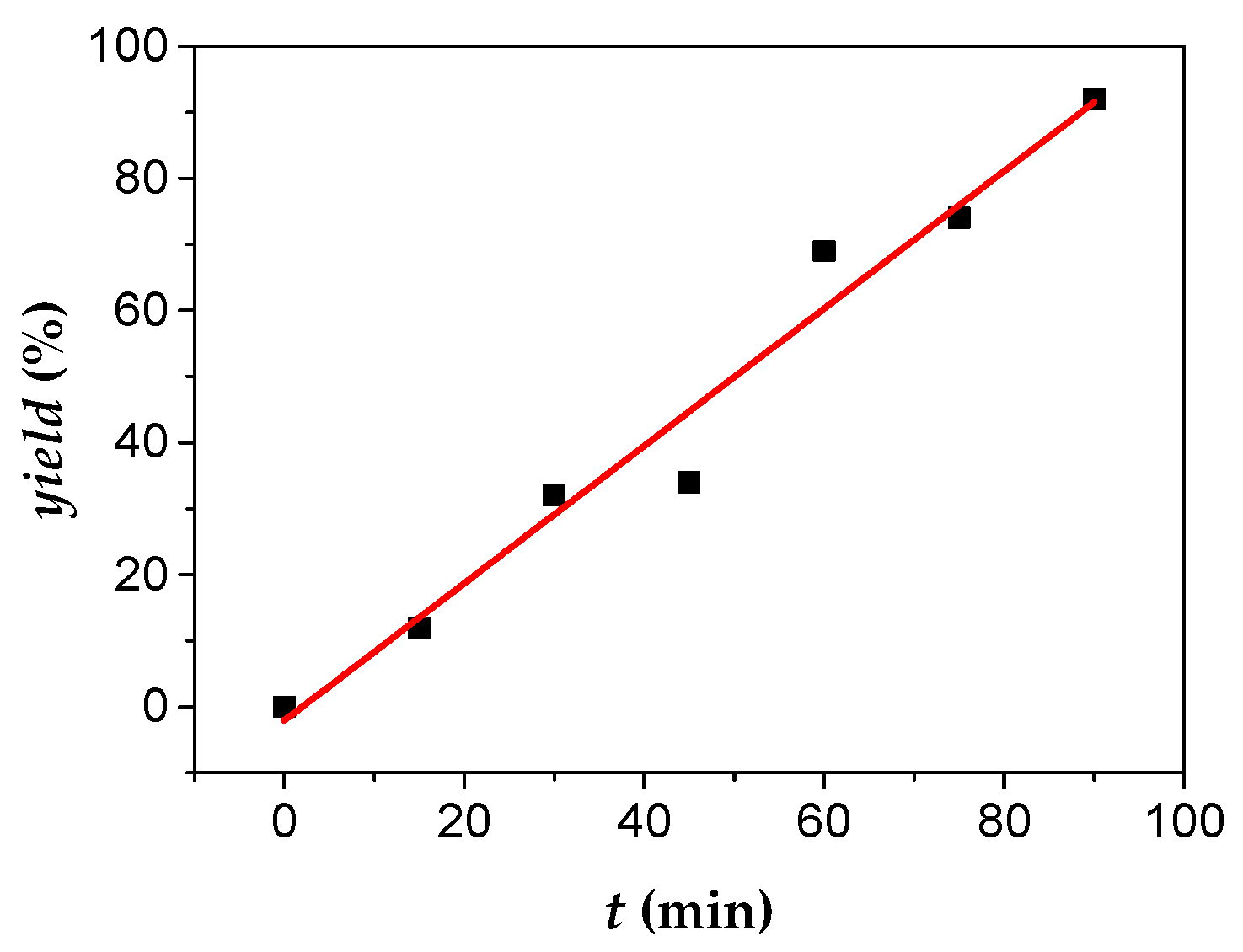
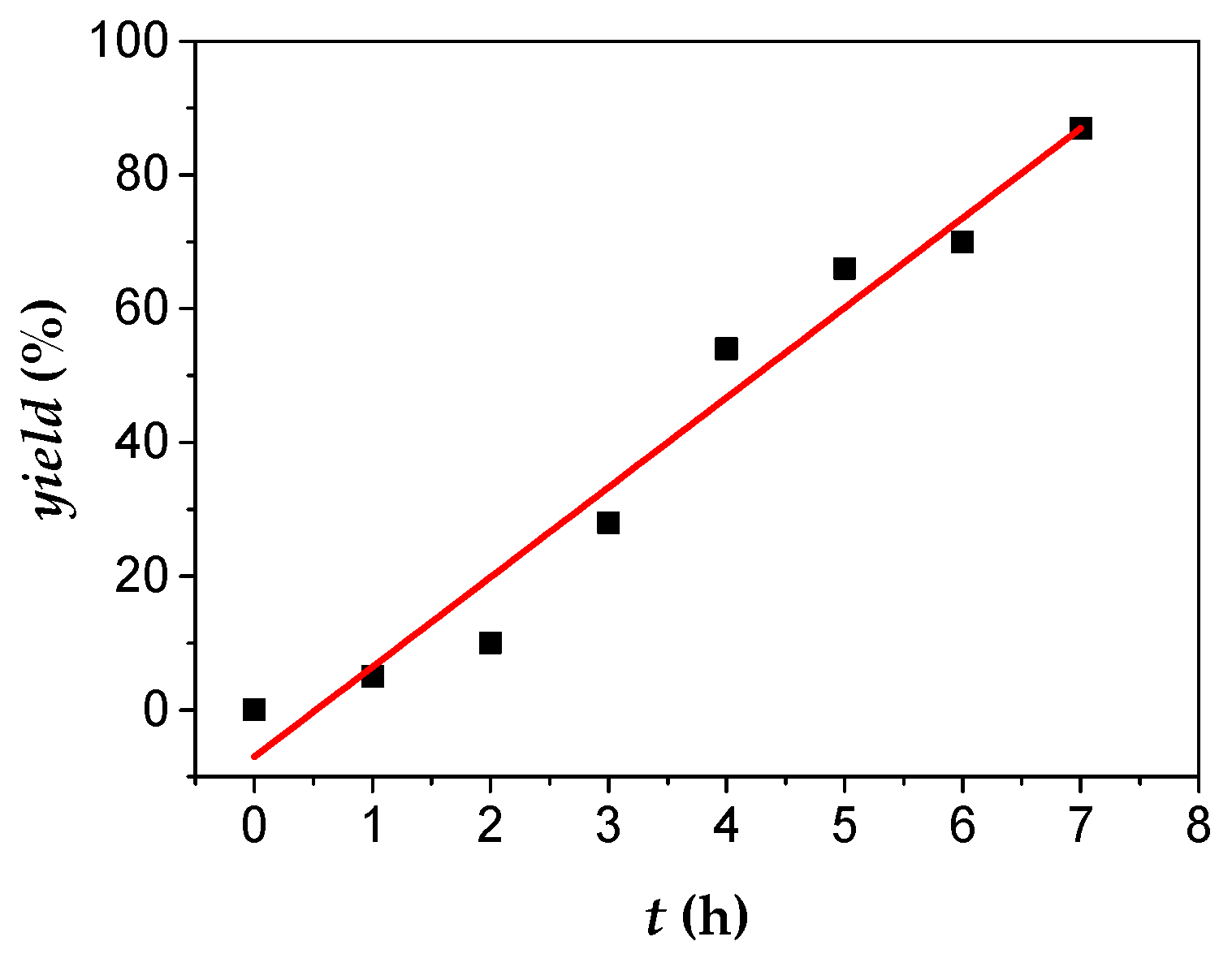
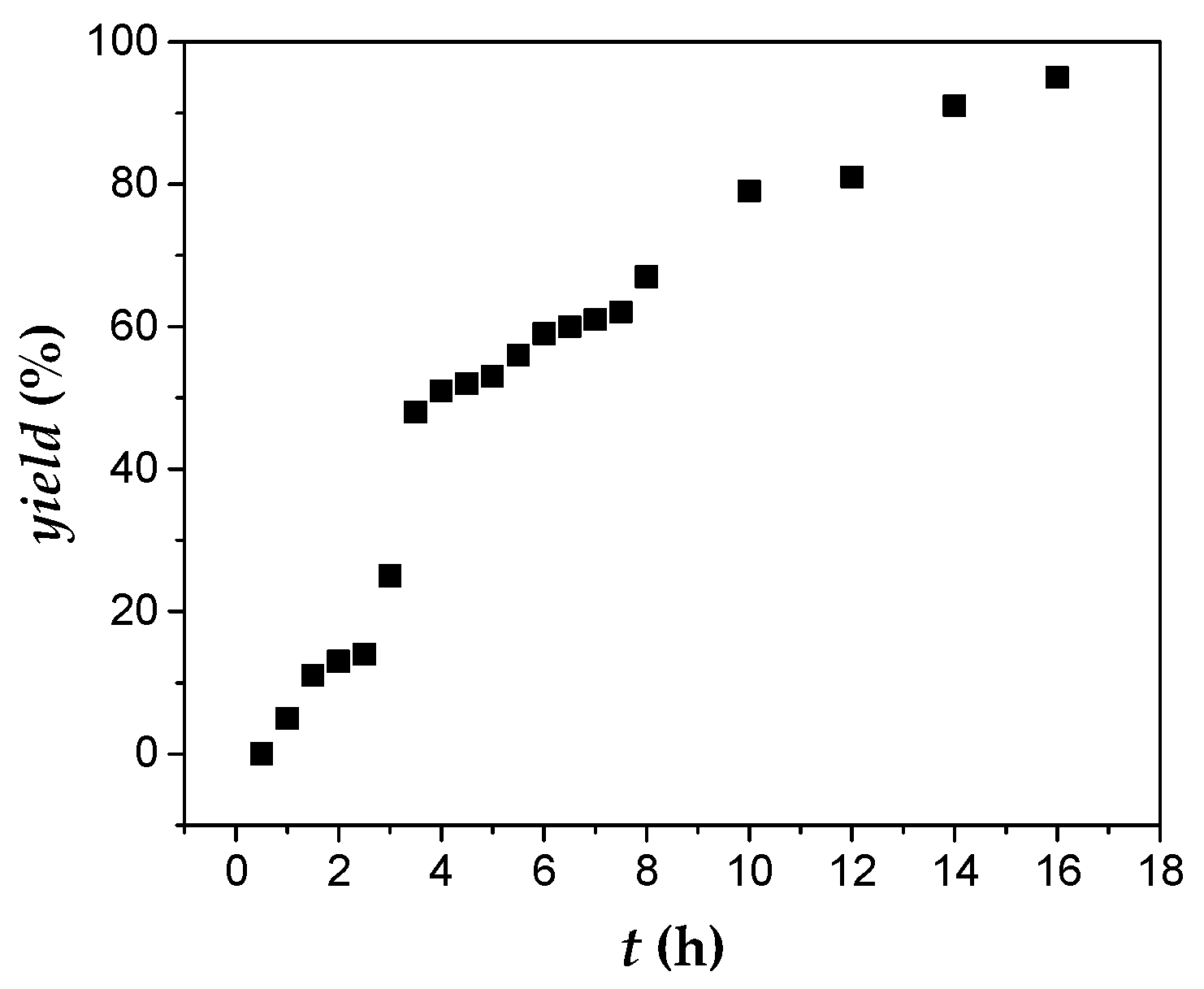
4. Conclusions
- (a)
- Short reaction times (15 min–3 h), of all monomers except for VNBE (23 h).
- (b)
- PVNBE contained all pendant vinyl bonds intact, had low molecular weight (Mw = 8000), but also narrow molecular weight distribution (Mw/Mn = 1.27), the lowest reported so far.
- (c)
- Polymerization of NBD could proceed in toluene without addition of co-catalyst (reflux, less than 5 h and 80% yield). That result was important and will be further investigated, because catalytic systems in which components may coexist, but they do not react unless the system is heated, are of particular industrial interest.
- (d)
- PNBD and PDCPD were insoluble and highly cross-linked, as evidenced by thermogravimetric analysis. 13C CPMAS spectra revealed the operation of two mechanisms (metathetic and radical) for cross-linking, with metathesis being the major pathway (~80%). A full mechanistic study is underway.
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Liu, J.; Lam, J.W.Y.; Tang, B.Z. Acetylenic polymers: Syntheses, structures, and functions. Chem. Rev. 2009, 109, 5799–5867. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T. Substituted polyacetylenes. J. Polym. Sci. A Polym. Chem. 2007, 45, 165–180. [Google Scholar] [CrossRef]
- Masuda, T.; Sanda, F.; Shiotsuki, M. Comprehensive Organometallic Chemistry III; Crabtree, R.H., Mingos, D.M.P., Eds.; Elsevier: Oxford, UK, 2007; Volume 11. [Google Scholar]
- Sutthasupa, S.; Shiotsuki, M.; Sanda, F. Recent advances in ring-opening metathesis polymerization, and application to synthesis of functional materials. Polym. J. 2010, 42, 905–915. [Google Scholar] [CrossRef]
- Leitgeb, A.; Wappel, J.; Slugovc, C. The ROMP toolbox upgraded. Polymer 2010, 51, 2927–2946. [Google Scholar] [CrossRef]
- Nomura, K.; Abdellatif, M.M. Precise synthesis of polymers containing functional end groups by living ring-opening metathesis polymerization (ROMP): Efficient tools for synthesis of block/graft copolymers. Polymer 2010, 51, 1861–1881. [Google Scholar] [CrossRef]
- Schrock, R.R.; Lee, J.K.; O’Dell, R.; Oskam, J.H. Exploring factors that determine cis/trans structure and tacticity in polymers prepared by ring-opening metathesis polymerizations with initiators of the type syn- and anti-Mo(NAr)(CHCMe2Ph)(OR)2. Macromolecules 1995, 28, 5933–5940. [Google Scholar] [CrossRef]
- Schrock, R.R. Synthesis of stereoregular ROMP polymers using molybdenum and tungsten imido alkylidene initiators. Dalton Trans. 2011, 40, 7484–7495. [Google Scholar] [CrossRef] [PubMed]
- Ivin, K.J.; Mol, J.C. Olefin Metathesis and Metathesis Polymerization; Academic Press: San Diego, CA, USA, 1997. [Google Scholar]
- Dragutan, V.; Sterck, R. Catalytic Polymerization of Cycloolefins; Elsevier B.V.: Philadelphia, PA, USA, 2000. [Google Scholar]
- Katz, T.J. Olefin metatheses and related reactions initiated by carbene derivatives of metals in low oxidation states. Angew. Chem. 2005, 117, 3070–3079. [Google Scholar] [CrossRef]
- Katz, T.J.; Lee, S.J. Initiation of acetylene polymerization by metal carbenes. J. Am. Chem. Soc. 1980, 102, 422–424. [Google Scholar] [CrossRef]
- Schrock, R.R. Multiple Metal-Carbon Bonds for Catalytic Metathesis Reactions. Available online: http://www.nobelprize.org/nobel_prizes/chemistry/laureates/2005/schrock-lecture.html.
- Grubbs, R.H. Olefin Metathesis Catalysts for the Preparation of Molecules and Materials. Available online: http://www.nobelprize.org/nobel_prizes/chemistry/laureates/2005/grubbs-lecture.html.
- Buchmeiser, M.R. Polymer-supported well-defined metathesis catalysts. Chem. Rev. 2009, 109, 303–321. [Google Scholar] [CrossRef] [PubMed]
- Balcar, H.; Čejka, J. Mesoporous molecular sieves as advanced supports for olefin metathesis catalysts. Coord. Chem. Rev. 2013, 257, 3107–3124. [Google Scholar] [CrossRef]
- Raptopoulos, G.; Grigoropoulos, A.; Mertis, K.; Paraskevopoulou, P.; Pitsikalis, M. Multinuclear transition metal catalysts for metathesis polymerization. Current developments and future perspectives. In Recent Research Developments in Polymer Science; Transworld Research Network: Trivandrum, India, 2014; Volume 12, pp. 83–106. [Google Scholar]
- Saragas, N.; Floros, G.; Paraskevopoulou, P.; Psaroudakis, N.; Koinis, S.; Pitsikalis, M.; Mertis, K. Polymerization of terminal alkynes with a triply bonded ditungsten halo-complex. J. Mol. Catal. A Chem. 2009, 303, 124–131. [Google Scholar] [CrossRef]
- Saragas, N.; Floros, G.; Paraskevopoulou, P.; Psaroudakis, N.; Koinis, S.; Pitsikalis, M.; Hadjichristidis, N.; Mertis, K. Ring opening metathesis polymerization of norbornene and norbornadiene by bimetallic multiply bonded tungsten complexes. Polymers 2012, 4, 1657–1673. [Google Scholar]
- Saragas, N.; Floros, G.; Raptopoulos, G.; Pitsikalis, M.; Paraskevopoulou, P.; Mertis, K. Exploring the reactivity of Na[W2(μ-Cl)3Cl4(THF)2]·(THF)3 towards the polymerization of selected cycloolefins. Molecules 2015. doi:10.3390/molecules201219810. [Google Scholar] [CrossRef]
- Abel, E.W.; Butler, I.S.; Reid, J.G. The anionic halogenopentacarbonyls of chromium, molybdenum, and tungsten. J. Chem. Soc. 1963, 2068–2070. [Google Scholar] [CrossRef]
- Templeton, J.L.; Jacobson, R.A.; Mc Carley, R.E. Synthesis and structure of bis(tetrapropylammonium) tri-μ-bromo-hexabromo-ditungstate(2-). A novel odd-electron dimeric anion showing evidence of Jahn-Teller distortion. Inorg. Chem. 1977, 16, 3320–3328. [Google Scholar] [CrossRef]
- Balcar, H.; Shinde, T.; Lamač, M.; Sedláček, J.; Zedník, J. Ring-opening metathesis polymerization of vinylnorbornene and following polymer modifications. J. Polym. Res. 2014, 21, 1–8. [Google Scholar] [CrossRef]
- Mauldin, T.C.; Kessler, M.R. Enhanced bulk catalyst dissolution for self-healing materials. J. Mater. Chem. 2010, 20, 4198–4206. [Google Scholar] [CrossRef]
- Naumann, S.; Buchmeiser, M.R. Latent and delayed action polymerization systems. Macromol. Rapid Commun. 2014, 35, 682–701. [Google Scholar] [CrossRef] [PubMed]
- Davidson, T.A.; Wagener, K.B.; Priddy, D.B. Polymerization of dicyclopentadiene: A tale of two mechanisms. Macromolecules 1996, 29, 786–788. [Google Scholar] [CrossRef]
- Mohite, D.P.; Mahadik-Khanolkar, S.; Luo, H.; Lu, H.; Sotiriou-Leventis, C.; Leventis, N. Polydicyclopentadiene aerogels grafted with PMMA: I. Molecular and interparticle crosslinking. Soft Matter 2013, 5, 1516–1530. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chriti, D.; Grigoropoulos, A.; Raptopoulos, G.; Charalambidis, G.; Nikolaou, V.; Coutsolelos, A.G.; Pitsikalis, M.; Mertis, K.; Paraskevopoulou, P. Metathesis Polymerization Reactions Induced by the Bimetallic Complex (Ph4P)2[W2(μ-Br)3Br6]. Polymers 2015, 7, 2611-2624. https://doi.org/10.3390/polym7121536
Chriti D, Grigoropoulos A, Raptopoulos G, Charalambidis G, Nikolaou V, Coutsolelos AG, Pitsikalis M, Mertis K, Paraskevopoulou P. Metathesis Polymerization Reactions Induced by the Bimetallic Complex (Ph4P)2[W2(μ-Br)3Br6]. Polymers. 2015; 7(12):2611-2624. https://doi.org/10.3390/polym7121536
Chicago/Turabian StyleChriti, Despoina, Alexios Grigoropoulos, Grigorios Raptopoulos, Georgios Charalambidis, Vasilis Nikolaou, Athanassios G. Coutsolelos, Marinos Pitsikalis, Konstantinos Mertis, and Patrina Paraskevopoulou. 2015. "Metathesis Polymerization Reactions Induced by the Bimetallic Complex (Ph4P)2[W2(μ-Br)3Br6]" Polymers 7, no. 12: 2611-2624. https://doi.org/10.3390/polym7121536
APA StyleChriti, D., Grigoropoulos, A., Raptopoulos, G., Charalambidis, G., Nikolaou, V., Coutsolelos, A. G., Pitsikalis, M., Mertis, K., & Paraskevopoulou, P. (2015). Metathesis Polymerization Reactions Induced by the Bimetallic Complex (Ph4P)2[W2(μ-Br)3Br6]. Polymers, 7(12), 2611-2624. https://doi.org/10.3390/polym7121536