1. Introduction
Polymers form the core of drug delivery systems. Polymers are utilized in drug delivery to provide weight, consistency and volume for the correct administration of the drug and in addition, they are multi-functional providing stability, drug release, targeting, enhanced bioavailability and patient acceptability [
1]. The choice of a polymer due to inherent diversity of structures, necessitates an extensive understanding of the surface and bulk properties of the polymers that can produce the desired functionalities [
2]. Both natural and synthetic polymers are utilized in drug delivery. Natural polymers are materials of large molecular weights from natural origins such as plants, micro-organisms and animals. In comparison to synthetic, natural polymers are preferable due to low toxicity, renewability, flexibility to modification, biodegradability and low cost [
3]. Natural polymers such as polysaccharides are hydrophilic, enzymatically degradable, and are able to retain the stability of protein drugs incorporated in them as well as increase their (proteins) therapeutic effects [
4]. Polysaccharides exhibit good haemocompatibility and interaction with living cells [
5] making them compatible and suitable biomaterials for long systemic circulation and targeting.
Most natural polymers are generally regarded as safe for oral consumption and so find applications in the food and pharmaceutical industries. Extraction and development of polymers from natural sources such as plants utilized as food may reduce regulatory requirement for approval [
6]. Some of the challenges are the seasonality of optimum yields, variations based on locations/climate and soils and stability as they lose viscosity or deform on storage. These challenges and the revolution of petrochemicals led to increasing utilization of synthetic polymers with natural polymers gradually nudged to the background. Synthetic polymers became convenient and their physicochemical properties endeared them to drug delivery scientists. However, the growing concern of their bio-incompatibility, possible toxicity and carcinogenicity has propelled the re-visitation of natural polymers. The use of natural polymers has increased in recent times; also due to the advocacy of “green” materials from “green” chemistry and technologies. Natural polymers are biogenic and their biological properties such as cell recognition and interactions, enzymatic degradability, semblance to the extracellular matrix and their chemical flexibility make them materials of choice for drug delivery.
Natural polymers have been used for diverse applications in drug delivery such as emulsification [
7], suspension [
8], controlled release [
9], film coating [
10], disintegration [
11], solubilization, bioadhesion, gelling, thickening, viscosity modulation, bulking agent [
12], drug devices, drug modification, encapsulation and mechanical strengthening [
13,
14,
15]. Natural polysaccharides are increasingly being used for drug targeting for chronic and site-specific diseases. Natural polymers are continually being explored in innovative approaches to drug delivery and personalized medicines. Increasing application of natural polymers in drug delivery implies increase in demand indicating the need for research and development into new natural polymers for subsequent commercialization. Polymers utilized in pharmaceuticals require regulatory approval. However, before a natural polymer can obtain approval, there are processes to be undertaken to produce pharmaceutical grade natural polymers.
Naturapolyceutics is a word coined from natural polymers and pharmaceutics. It is the art, science and technology of utilizing natural polymers for the design and development of drug delivery systems. Prior to utilization of natural polymers for drug delivery, polymers are first characterized by subjecting the polymers through various analyses. This review is geared towards describing naturapolyceutics—the science of producing a pharmaceutical grade natural polymer from harvest/extraction to in vivo characterization and to advocate the extensive characterization of under-utilized natural polymers for subsequent commercialization. The focus of this paper will be on natural polymers from plants—mucilages and gums.
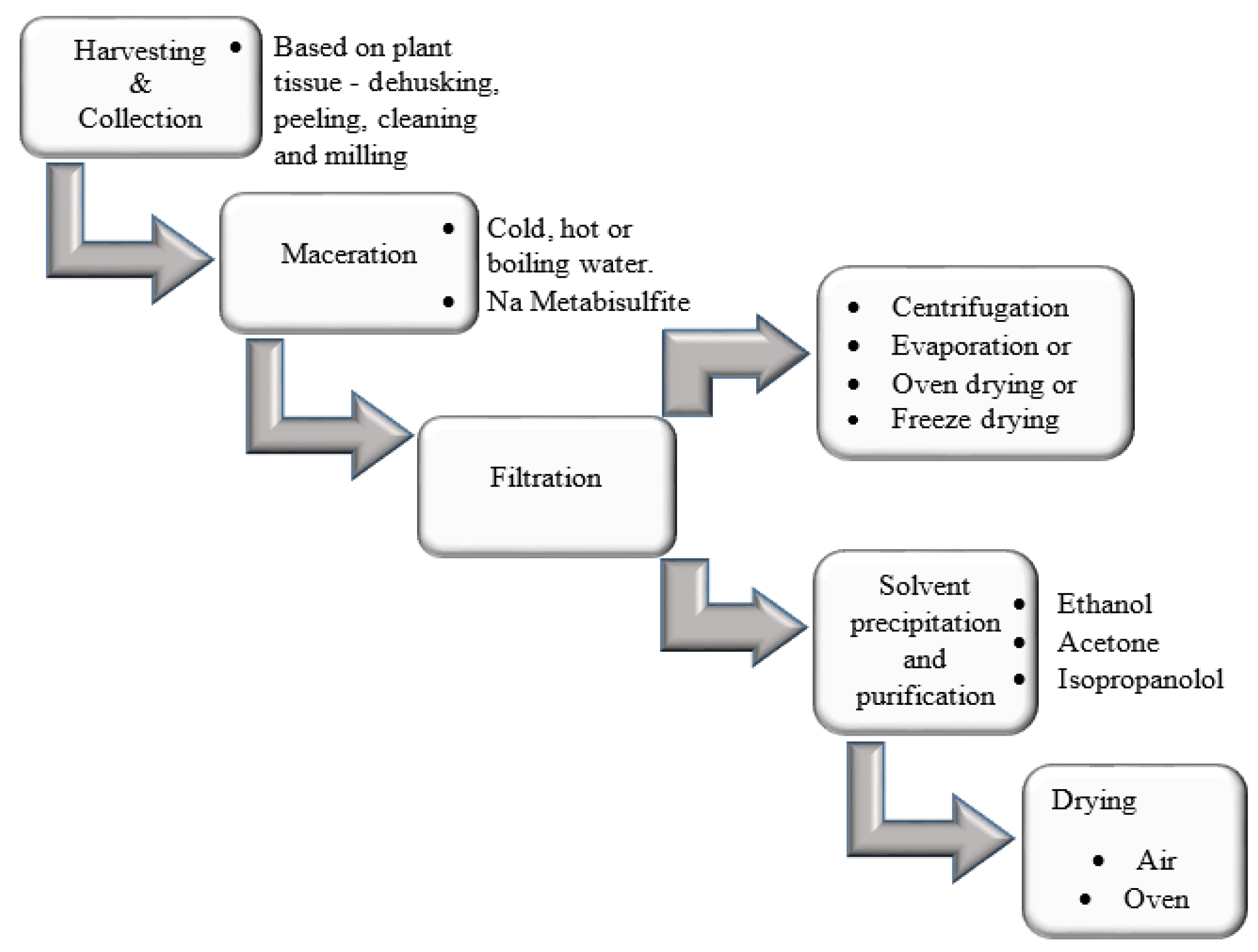
2. Extraction and Purification
Plant-derived polymers are mainly extracted by maceration of the part of plant (calyxes, leaves, stem barks, roots, seeds or fruits) in ambient or hot water over a period. Thereafter, the gum or mucilage is separated from the part of plant by filtration technique such as the use of a muslin bag. The mucilage or gum is precipitated from water by adding alcohol. Absolute ethanol is usually utilized as it precipitates faster giving a higher yield than ethanol: water mixture. In addition, ethanol is a preferred solvent because it is green and FDA approved. Other solvents such as acetone and methanol are also used for precipitation. After precipitation, the mucilage is air- or oven-dried. However, Ahuja and co-workers opted for drying of the mucilage instead of precipitation with alcohol [
16].
Mimosa pudica seeds were macerated in sufficient water for 10 h. The mucilage obtained including the seeds were dried in the oven at 50 °C for 4–5 h. Dried mucilage was separated from the seeds by passing through No 18 mesh. Thereafter, the seed husks were removed by winnowing. Others freeze-dry instead of heating [
17,
18].
Extraction of mucilage from
Plantago psyllium seed was undertaken by adding the seeds to boiling 0.1 M HCl with continuous boiling until the seed husk dissolves. Once all the seeds have changed color, filtration is done while hot. The seeds are washed repeatedly to remove traces of mucilage on them. The collective filtrates are mixed with ethanol to precipitate the mucilage which is subsequently dried [
19]. Mucilage from
Plantage ovate Forsskaol seed husk is obtained by soaking the seed in deionized water kept at 80 °C with occasional stirring for 2 h [
20]. Thereafter it is allowed to cool at room temperature and left overnight. Afterwards, 0.5 M NaOH is added while stirring to separate the mucilage from the seeds and the resultant slurry is filtered. The mucilage is precipitated by addition of 2 M HCl to the filtrate. The precipitate is then washed with water to remove traces of acid and separation is undertaken by centrifugation and subsequent drying of the residue.
Mucilage from
Sesamum indicum/radiatum is extracted by dispersing weighed leaves in hot water and allowed to macerate for 6 h, thereafter filtered using a muslin bag and subsequently precipitated and washed with ethanol [
3]. It is air-dried and milled. However, recent extraction in our laboratory indicated the mucilage can effectively be extracted from the leaves with cold water. It was observed also that browning of the mucilage was less with cold water. Furthermore, sodium metabisulfite (1% w/v) can be added during maceration as a bleaching agent/antioxidant which prevents enzymatic browning.
Cashew gum is an exudate from the bark of the tree
Anacardium occidentale. After collection and possibly drying, gum is milled, dissolved in water and then filtered. Alcohol is added to the filtrate to precipitate the gum which is oven-dried at 45 °C [
21]. This isolates water-soluble cashew gum. Alternatively, the gum can be dissolved in water, filtered and the filtrate centrifuged. The supernatant is collected and evaporated to dryness [
21].
For such parts of the plant as the tubers, defatting may be undertaken with petroleum ether and saponins extracted with methanol first before maceration [
22]. However, Ghule and co-workers neither undertook defatting nor extraction of saponins as they extracted mucilage from tubers of
Eulophia campestris [
23]. Mucilage from tubers of
Eulophia campestris was extracted by boiling freshly dried and coarsely milled tubers in water for 1 h, macerating for 2 h, filtering and precipitating with acetone. Initial defatting and removing of saponins may not be necessary as fat dissolves in acetone and ethanol which are the common solvents used in precipitation of mucilages and gums while saponins dissolve in ethanol and a mixture of water and acetone. This implies that fat and saponins are removed from the mucilage during precipitation. Proteins are also soluble in ethanol. Hence it is possible to obtain purified gums and mucilages by maceration in water and subsequent precipitation using organic solvents such as ethanol, acetone and isopropanol coupled with several soakings and washings in organic solvents over a period. The process of extraction is dependent on the part of plant, location of mucilage within the plant tissue, separation techniques, choice of extraction solvents and choice of drying techniques.
Figure 1 summarizes the process of extraction of polymers from the plant.
Figure 1.
Schematic diagram of the general process of extraction of plant polymers.
Figure 1.
Schematic diagram of the general process of extraction of plant polymers.
Several washings and precipitations using solvents such as ethanol, acetone and isopropanol purify the extracted polymer thereby aiding the removal of impurities. However, further purification if desired may be undertaken with a liquid chromatography-mass spectrometry purification system. With regards to microbes, absolute ethanol which is commonly used for extraction and purification does not kill the microbes that may be present. Absolute ethanol inactivates the microbes and in the presence of favorable conditions, the microbes reactivate and may lead to degradation of the polymer. Microbial contamination is one of the setbacks of natural polymers. However, some of the natural polymers such as chitosan and aloe vera mucilage exhibit antimicrobial activity and so may experience no microbial contamination. Conventional microbiological tests can be used to ascertain the microbial load. Consequently, based on application, polymers may be sterilized. Sterilization techniques include filtration, use of ethylene oxide or hydrogen peroxide, 70% ethanol (hydrophilic viruses and bacteria spores are resistant), gamma-irradiation and low-temperature radio-frequency glow discharge (RFGD) plasma treatment [
24]. However, the choice of sterilization technique is paramount to prevent polymer degradation, morphological or chemical damages.
3. Modification of Natural Polymers
Natural polymers are modified as a means to overcome their setbacks such as drop in viscosity, microbial degradation, and partial or low solubility. In addition, modification of natural polymers enhances their drug delivery properties and versatility. Modification should be undertaken such that the natural polymers do not lose their biological properties. Methods of modification include grafting, crosslinking, derivative formation and polymer-polymer blending.
3.1. Grafting and Crosslinking
Singh and Chauhan synthesized a polymer network of Psyllium husk and methacrylamide (MAAm) with ammonium persulfate (APS) as the initiator and
N,
N-methylenebisacrylamide (
N,
N-MBAAm) as the crosslinker [
25]. The synthesis was undertaken by chemically induced polymerization through free radical mechanism. The initiator, APS produced reactive sites on psyllium, methacrylamide and
N,
N-MBAAm. The four reactive sites on
N,
N-MBAAm can be linked with the radicals on psyllium and poly(MAAm) to produce a three-dimensional network, psy-
cl-poly(MAAm)—a hydrogel. Insulin was incorporated into the hydrogel by a swelling equilibrium method whereby the hydrogel is placed into the drug solution and there is an uptake of the drug solution into the hydrogel. The hydrogel swelled in the process and thereafter dried to obtain an insulin-loaded polymer matrix. The polymer matrix was assessed for swelling and drug release at pre-determined times [
25]. Swelling and release of the drug occurred at a faster rate in neutral pH than acidic pH. However, the release of the drug from the hydrogel can further be modulated by varying the composition of the polymer network and the crosslinking ratio.
In addition, polymer network of psyllium and
N-hydroxymethylacrylamide was synthesized from the same laboratory with APS as initiator and
N,
N-MBAAm as crosslinker and salicylic acid and tetracycline hydrochloride as model drugs [
26]. Furthermore, psyllium-
N-vinylpyrrolidone (NVP) based hydrogels was obtained by radiation induced crosslinking and 5-Fluorouracil was the drug used [
27].
In another study, sterculia gum was modified with methacrylic acid using APS as initiator and
N,
N-MBAAm as crosslinker to produce hydrogels [
28]. Sterculia gum and the modified, sterculia-
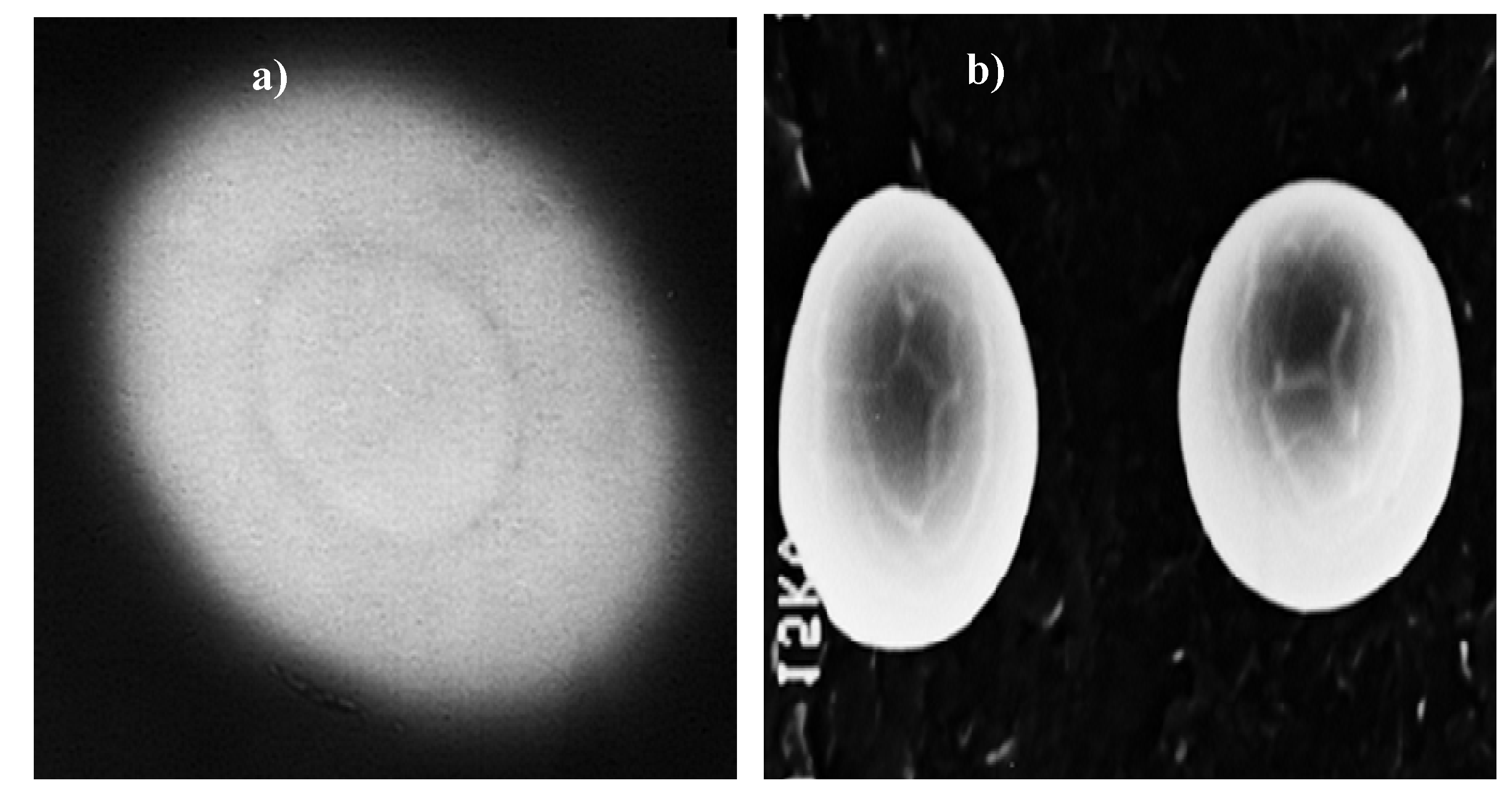
cl-poly(MAAc) were characterized for swellability, drug release, morphology, structural modification. The morphology comparison indicated that sterculia-
cl-poly(MAAc) displayed morphological heterogeneity as opposed to that of sterculia gum which was smooth and homogenous (
Figure 2). Swelling was studied on the basis of reaction parameters. The authors observed that swelling of sterculia-
cl-poly(MAAc) decreased as the monomer concentration in the polymer matrix increased. In addition, swelling of sterculia-
cl-poly(MAAc) increased with increase in concentration of APS, the initiator and decreased with increase in concentration of
N,
N-MBAAm. Also, as would be envisaged, swelling increased with increase in sterculia gum, though the authors stated it increased up to 0.6 g of sterculia gum. Swelling is also a function of time of matrix in the hydrating medium. Furthermore, swelling increased with increase in pH and decreased in 0.9% NaCl. Although swelling increased as pH increased, rate of drug release was higher at lower pH 2.2. This was attributed to the rapid solubility of the drug ranitidine at lower pH. The authors also modified sterculia gum with acrylamide (AAm) using the same initiator and crosslinker [
29].
Other grafting and crosslinking with natural polymers include polyacrylamide grafted to katira gum and katira gum crosslinked with glutaraldehyde [
30,
31]; crosslinking of guar gum with trisodiumtrimetaphosphate [
32]; cashew gum crosslinked with epichlorohydrin [
33].
Figure 2.
Scanning electron micrograph of (
a) sterculia gum and (
b) sterculia-
cl-poly(MAAc) [
28] (reproduced with permission from Elsevier).
Figure 2.
Scanning electron micrograph of (
a) sterculia gum and (
b) sterculia-
cl-poly(MAAc) [
28] (reproduced with permission from Elsevier).
3.3. Polymer-Polymer Blending
Polymer-polymer blending is an easy and convenient approach of modifying polymers without undergoing the procedures for chemical reactions/synthesis for new polymers. The blending may be due to physical bonding whereby polymers interact by Van der Waals, hydrogen bonding and London dispersion forces. The blending may also be due to chemical bonding whereby the polymers interact by covalent bonding (crosslinking) or ionic bonding. Polymer blending is a means of improving the properties of the polymers involved. A blend of alginate, locust bean gum and xanthan gum in fabrication of microspheres enhanced drug entrapment efficiency and further retarded drug release in comparison to the use of alginate and locust bean or alginate and xanthan gum [
47]. Microbeads fabricated with a blend of alginate and irvingia gum significantly enhanced drug entrapment efficiency and controlled the release of diclofenac sodium over 7 h in comparison to microbeads fabricated with alginate alone [
48]. Blends of natural and synthetic gums such as carboxymethyl cellulose/locust bean gum and methacrylate copolymer [
49] to produce hydrogels have also been undertaken. Methacrylate copolymer is unsuitable for controlled release formulations, however, its interaction with carboxymethyl cellulose (CMC) produced a hydrogel thereby enhancing the mechanical strength of CMC.
4. Characterization of Natural Polymers
The mucilages and gums from plants are mainly polysaccharides. To advance the use of mucilages and gums for drug delivery from bench to market, the structure, physicochemical, physicomechanical and drug delivery properties need to be extensively elucidated. It is also important that each natural polymer is distinctly identified from the others. Extensive characterization of a natural polymer will reveal its uniqueness and also assist in its regulatory approval for pharmaceutical use.
4.1. Structural Elucidation of Natural Polymers
Determination of the structure of a polysaccharide polymer gives identity to the polymer. The properties displayed by polymers are dependent on their chemical structure and chain conformations. Although a thorough elucidation of the structure and chain conformation of a polymer may be tasking, a wide range of techniques are available to extensively characterize and uniquely identify a polymer. Structure characterizations are undertaken with Fourier Transform Infrared spectroscopy (FTIR), liquid-state Nuclear Magnetic Resonance (NMR) (One and two dimensions), solid-state NMR, Raman spectroscopy, gas chromatography (GC), GC-Mass spectroscopy (MS), and high performance liquid chromatography [
50]. The chain conformations of polysaccharides in solutions are elucidated with static and dynamic light scattering, viscosity analysis, circular dichroism analysis, single molecular atomic force microscopy (AFM), AFM-based single-molecule force spectroscopy, florescence correlation spectroscopy and NMR spectroscopy [
50].
4.2. Identification of Sugar Constituents
To determine the sugar constituents, the mucilage or gum is hydrolyzed with dilute mineral acids. The different monosaccharides are separated with chromatographic techniques such as Size Exclusion Chromatography Multi-Angle Laser Light Scattering (SEC-MALLS), GC chromatography. Structural elucidation is undertaken with NMR and MS. The mode of monosaccharide-monosaccharide linkage is elucidated by methylation, periodate and lead tetra-acetate oxidation [
14].
4.3. Polymorphism Determination
Crystallinity of polymers influences their chemical properties which impact on their drug delivery capacity. Degree of crystallinity influences properties such as solubility, stability and drug release. Modification of a polymer can change its degree of crystallinity. Techniques such as gel electrophoresis, differential scanning calorimetry, wide angle X-ray diffraction and powder X-ray diffraction are used for polymorphism determination. However, powder X-ray diffraction is the primary technique used to determine the degree of crystallinity of a polymer. Characteristic peaks in the spectrum obtained indicate the degree of crystallinity. Absence of characteristic peaks in the spectrum is indicative of complete amorphous nature of the polymer [
17]. Most of natural polymers are amorphous or semi crystalline. They are more soluble than the highly crystalline polymers [
51].
4.4. Determination of Molecular Weight and Polydispersities of Natural Polymer Fractions
Although determination of molecular weights of polysaccharides may be challenging due to their heterogeneity and polydispersity [
52], the last decade or more has experienced significant progress in techniques employed to determine molecular weights. The molecular weights and polydispersity of polysaccharide fractions of the mucilages and gums can be determined by (preparative) size-exclusion chromatography (SEC), gas chromatography (GC) and viscometry/rheometry [
53]. Viscosity can be utilized to estimate molecular weight as it is a direct reflection of molecular weight. Coupled chromatographic techniques such as Size Exclusion Chromatography Multi-Angle Laser Light Scattering are more rapid and reliable. Recently, Waters
® is promoting a chromatographic technique called Advanced Polymer Chromatograph (APC™) system offering a better resolution of polymer distribution with shortened time run for determination of molecular weight distribution. However, it appears the system is being promoted for low molecular weight polymer samples [
54].
4.5. Surface Characteristics of Natural Polymers
The surface characteristics of a polymer or its derivative/modification when used for drug delivery influence the rate and mechanisms of drug release. Surface modification of a polymer has been utilized for drug targeting [
55]. The morphology of a polymer (modified or native) also influences the degree of drug encapsulation for nano- and micro- drug delivery systems or cell encapsulation of these systems and their circulation duration in the blood as well as polymer-drug interactions or nano- or microparticle-cell interactions [
56]. Therefore, an understanding of the morphology—shape, size and surface chemistry—of natural polymers is paramount. Microscopy such as scanning tunneling microscopy (STM), transmission electron microscopy (TEM), scanning electron microscopy (SEM) and atomic force microscopy (AFM). TEM provides a 2-dimensional view of a material while SEM provides the 3-dimensional view and AFM takes it much higher by revealing surface molecular composition and mechanical properties.
Figure 4 displays the 2D view of TEM and 3D view of SEM both confirming the shape and the inner cavity of the particle. TEM indicated the particle is a nanocapsule with the display of the inner ring and SEM confirmed with the display of hollowness. Combination of techniques such as AFM and Raman spectroscopy provide an enhanced surface characterization of a polymer. High resolution data of both chemical and morphological structures are produced [
57] with topographical, mechanical, electromagnetic, thermal and near-field optical properties elucidated at molecular scale.
Figure 4.
(
a) Transmission electron microscopy (TEM) image and (
b) scanning electron microscopy (SEM) image of polymeric nanoparticles (levodopa-loaded polymethacrylate copolymer/chitosan poly-lipo nanoparticles) [
58].
Figure 4.
(
a) Transmission electron microscopy (TEM) image and (
b) scanning electron microscopy (SEM) image of polymeric nanoparticles (levodopa-loaded polymethacrylate copolymer/chitosan poly-lipo nanoparticles) [
58].
4.6. Thermal Behavioral Analyses of Natural Polymer
Differential scanning calorimetry (DSC) gives insights into the physical and chemical changes that present during thermal processes. Subjecting a polymer through a temperature range assists in identifying its glass transition, crystallization, melting and decomposition temperatures. Materials behave differently below and above their glass transition temperatures. Drug release is impacted by the mechanical properties of a polymer which is dependent on the glass transition temperature which in turn is influenced by the molecular weight of the polymer [
59]. Thermal behavior of polymers is distinct and so each polymer is expected to exhibit a unique thermogram. Other techniques utilized to analyze the thermal behavior of polymers include dynamic mechanical thermal analysis (DMTA), thermally simulated current spectroscopy (TSC), and dilatometry (DIL) [
60].
4.7. Rheological Behavioral Analyses of Natural Polymer
Visco-elastic behavior of a polymer is another property that is dependent on the molecular weight. In drug delivery, visco-elastic properties relate to drug delivery properties such as product hardness, compression, drug release, suspendability and spreadability [
61]. Viscometers measure viscosity—resistance to flow; however, rheometers measure viscosity/flow as well as other parameters such as effects of shear on flow/polymer, effect of varying frequencies and temperatures on polymer and assist in giving insight into the ability of the polymer to behave as a viscous liquid, paste, gel or a 3D network thereby influencing the rate and mechanisms of drug release.
4.8. In vivo Characterization of Natural Polymers Fabricated Drug Delivery Systems
Although some of the mucilages and gums are edible, some are obtained from parts of plants that are not eaten. For instance, cashew nuts and fruits are eaten but cashew gum is obtained from the stem bark. Consequently, the safety should be ascertained through toxicity and histopathology tests. Subsequently, the polymers may be used in formulation of drug delivery systems and
in vivo performance evaluated. The transmucosal delivery of tablets fabricated with hakea gum was assessed
in vivo for its mucoadhesive property and drug delivery [
62]. In this study, white male New Zealand rabbits were used. Mucoadhesive property was undertaken
ex vivo whereby force of detachment of the tablet from the freshly excised rabbit intestinal mucosa was determined after pre-determined contact times between the tablet and hydrated intestinal mucosa. The force required for detachment increased with time and as the concentration of hakea gum increased. To assess transmucosal delivery of the buccal tablets coated on all sides except one flat face (which is the surface attached to the mucosa), the rabbits were anaesthetized, hakea buccal tablets were applied and blood samples were withdrawn from a cannula inserted in the marginal ear at pre-determined time intervals over 5 h. Hakea gum controlled and prolonged the release of CPM over 5 h application period. In addition it provided and sustained an elevated concentration of CPM during this period. However, the more the quantity of hakea gum, the more controlled the release and subsequently lower C
max and bioavailability of the incorporated drug. In summary, the modulation of the rate of release and mucoadhesive strength depends on the variation of the quantity of hakea gum utilized [
62].
For newly explored plant polymers, polymer characterizations, in vitro and in vivo characterizations of the drug delivery systems will be required to follow the procedures and criteria for regulatory approval before they can be approved and given marketing authorization for pharmaceuticals.
5. Natural Polymers for Drug Delivery
Natural polymers have been employed for diverse drug delivery systems. Prajapati and co-workers reviewed the pharmaceutical applications of mucilages, gums and their modified forms [
14]. The review also tabulated the common dosage forms in which gums and mucilages have been utilized. Consequently, in this section, the focus will be on current trends such as BIOMEMS and theranostics.
5.1. Natural Polymers for Controlled Release
Most plant polymers have proven to control the release of the drug over a period of time. Hence, they are used for controlled release matrices.
Terminalia catappa gum is a plant exudate from
Terminalia catappa Linn which was utilized in the formulation of a controlled release tablet of dextromethorphan hydrobromide, a cough suppressant [
63]. The techniques used for formulation of tablets included direct compression, wet granulation and solid dispersion. The formulations were compared with the pure drug filled into capsules. The pure drug filled in capsules releases 100% of the drug in less than 1 h while the formulations with
Terminalia catappa gum controlled the release of dextromethorphan hydrobromide for hours (≥8 h at the ratio 1:6 of drug to polymer). Mucilage from fruits of
Hibiscus esculentus was found to be directly compressible and controlled the release of paracetamol for over 6 h [
64].
Natural gum/polysaccharide from plant
Hakea gibbosa was employed as a sustained release polymer and mucoadhesive agent for buccal tablets [
62,
65,
66]. Another direct compressible agent whose mucoadhesive activity understandably increased as its concentration was increased. Increase in concentration implies increase in molecular chains/bonds of the natural polymer available to interact with the mucus. The attachment/mucoadhesion of hakea gum to the mucus (mucin) may be based on non-covalent bondings such as Van der Waals, H-bonding and electrostatic interactions [
49]. As the polymer hydrates in the presence of saliva, there is an increase in mobility of the polymer chains producing increased surface for adhesion/interactions with mucin. The tablets were directly compressed as well as wetly granulated. Hakea gum retarded the release of salmon calcitonin and chlorpheniramine maleate (CPM) from the tablets matrices [
65,
66]. However, the authors observed that direct compression sustained the release of CPM more than wet granulation [
66]. The tablets were formulated to exhibit unidirectional release of the drug using compression coating technique to applying hydrogenated castor oil on all sides except one flat face. The uncoated face is adhered to the buccal mucosa during the period of drug delivery. This ensures transmucosal delivery of the drug preventing some of the drug being lost in the surrounding oral cavity with subsequent swallowing.
5.2. Natural Polymers for Intelligent Drug Delivery
Some natural polymers and their derivatives respond to certain environmental factors such as ions, pH, enzymes, temperature and electromagnetic field. Such polymers are referred to as intelligent, smart, stimuli- and environmental-responsive polymers. These polymers in response to certain environmental factors trigger specific drug release to affected tissues or cells, or in response to certain physiological events [
67]. Crosslinked
Plantago psyllium mucilage (with methacrylamide) was employed as colon-specific drug delivery system due to its response to pH [
68]. Swellability and drug release was higher at pH 7.4. Other crosslinked
Plantago psyllium mucilage (with polyacrylamide) produced hydrogels that respond to ions as well as pH [
69]. Pectin derived from plant cell walls depending on its degree of esterification responds to ions, pH and enzymes while cellulose derivatives respond to ions, pH and temperature [
67] and have been utilized for colon specific drug delivery [
70]. Animal polysaccharides such as chitosan and hyaluronic acid have been responsive to ions, pH, electrical and magnetic fields, temperature, light and molecules [
67]. However, it is envisaged that plant polymers can also be configured to respond to electromagnetic field, light and molecules through some suitable modifications/derivatizations.
5.4. Natural Polymers for BioMEMS
BioMEMS refers to biomedical or biological microelectromechanical systems. It is a cross-disciplinary process of utilizing and customization of microfabrication technologies for biomedical applications. Microneedles are fabricated for transdermal delivery. The transdermal route is an alternative to the oral route and is utilized for proteins and vaccines which are easily metabolized in the GIT providing limited bioavailability and subsequent sub-optimal therapy. However, the presence of stratum corneum (outermost skin layer) poses a challenge to the delivery of drugs through the skin. Consequently, microneedles overcome this barrier by disrupting the stratum corneum in a fairly painless manner [
72]. Microneedles are needles produced at micron-scale by utilizing the tools of microelectronics. Microneedles assembled into patches incise through the stratum corneum into the epidermis or the superficial dermis to administer drugs into the skin for local or systemic delivery [
72]. The application of microneedles consists of two approaches [
73]. One approach is the permeabilization of skin with microneedles and then application of a transdermal patch for drug delivery over an extended period. The second is coating or encapsulation of the drug on or within the microneedles. As the skin is permeabilized by the microneedles and as the microneedles and or the coating dissolves, the drug is released into the skin.
Figure 5.
Dissolving microneedles for transdermal drug delivery. (
a) Microneedle master-structure (600 μm in height and 300 μm wide at base) used to mold dissolving microneedles made of (
b) carboxymethyl cellulose (CMC), and (
c) amylopectin. The master-structure was imaged by scanning electron microscopy and the molded microneedles were imaged by brightfield microscopy [
73] (reproduced with permission from Elsevier).
Figure 5.
Dissolving microneedles for transdermal drug delivery. (
a) Microneedle master-structure (600 μm in height and 300 μm wide at base) used to mold dissolving microneedles made of (
b) carboxymethyl cellulose (CMC), and (
c) amylopectin. The master-structure was imaged by scanning electron microscopy and the molded microneedles were imaged by brightfield microscopy [
73] (reproduced with permission from Elsevier).
Lee and co-workers utilized carboxymethylcellulose (CMC) and amylopectin for fabrication of microneedles (
Figure 5) using photolithography for the micromolds [
73]. These polymers were dissolved separately in deionized water and concentrated to viscous hydrogels by heating to 60–70 °C. The drug may be incorporated into the hydrogel before filling the micromolds or the drug may be incorporated into the hydrogel used to form the backing layer. The microneedles were assessed for mechanical strength, skin penetration/insertion, drug release and drug (protein) stability. The mechanical behavior of the two polymers indicated amylopectin was stronger than CMC and the presence of protein in CMC microneedles enhanced its mechanical strength. In addition the pyramidal microneedles were stronger than the conical microneedles which the authors suggested may be due to the larger cross-sectional area of the base of the pyramid. Of 600 μm length, 150–200 μm of the length penetrated the skin and the incomplete insertion was due to the broad base (300 μm) of the microneedles. Furthermore, the drug incorporated into the microneedles experienced bolus release due to the microneedles’ rapid dissolution. However, the backing layer provided a sustained rate of release of incorporated drug or protein. In addition, the protein (lysozyme) sustained its structural and functional integrity after incorporation, during release and storage of 2 months at room temperature [
73]. Indeed more plant polymers should be explored for the fabrication of microneedles and other BioMEMS.
5.5. Natural Polymers for Theranostics
A theranostic refers to a delivery system fabricated to deliver both medicine and imaging agent(s) in a single dose, bridging the gap between imaging and therapy, thereby facilitating real-time monitoring of therapeutic efficacy of the incorporated drug [
74]. The multi-functionality of nanoparticles confers on them the ability to deliver medicine and imaging agents. Inorganic and metallic nanoparticles have been employed as theranostics as well as synthetic and natural polymeric nanoparticles. However, natural polysaccharides due to their excellent biocompatibility, low toxicity, biodegradability and functionalities that the body can identify with, make them excellent materials for theranostics.
Figure 6.
(
a)
Ex vivo fluorescence images of normal organs and tumors collected at two days post-injection of HA-NPs and; (
b) Quantification of the
ex vivo tumor targeting characteristics of hyaluronic acid nanoparticles (HA-NPs) in tumor-bearing mice. Error bars represent standard deviation (
n = 3) [
75] (reproduced with permission from Elsevier).
Figure 6.
(
a)
Ex vivo fluorescence images of normal organs and tumors collected at two days post-injection of HA-NPs and; (
b) Quantification of the
ex vivo tumor targeting characteristics of hyaluronic acid nanoparticles (HA-NPs) in tumor-bearing mice. Error bars represent standard deviation (
n = 3) [
75] (reproduced with permission from Elsevier).
Self-assembled modified hyaluronic acid (HA) derivatized nanoparticles were fabricated for tumor targeting [
75]. First, amphiphilic HA was conjugated with 5β-cholanic acid to form HA-CA conjugates. HA-CA conjugates were utilized to fabricate nanoparticles (HA-NPs) which were thereafter labeled with NIR dye cyanine 5.5 (Cy5.5). The nanoparticles were then characterized for
in vitro cellular uptake,
ex vivo tissue distribution,
in vivo distribution and tumor targeting. The strong florescent signals detected in the cytoplasm indicated HA-NPs were readily taken by the cancer cells receptor-mediated endocytosis. In addition, the florescent signals were stronger at the tumor site than in the normal tissues (
Figure 6) indicating appreciable tumor targeting, which the authors suggested may be due to enhanced permeation and retention (EPR) effect as well as receptor-mediated endocytosis of HA-NPs [
75]. In summary self-assembled HA-NPs can be utilized for as targeted drug carriers and or imaging agents for therapy and monitoring of therapeutic efficacy of the incorporated drug.
Other natural polymers such as alginate, dextran and chitosan [
74,
76] have been used. However, plant polysaccharides should be explored in fabrication of theranostics as they are exhibit functionalities recognized by the body as comparable to those of the polymers in biological systems. Polysaccharides are the materials of focus for drug targeting and concentration at the site of action. Considering cancer therapy, some polysaccharides have exhibited anti-tumor activity [
77]. An incorporation of chemotherapeutic into a polysaccharide carrier may enhance cancer therapy.
6. Novel and Underutilized Natural Polymers
There are publications on a host of novel polymers utilized as pharmaceutical excipients for drug delivery. These polymers have been evaluated as binders, mucoadhesive agent, disintegrant, emulsifying agent, for controlled release formulation, thickener, suspending agent, coating agent, and film forming agent [
14]. These are conventional drug delivery properties. Although these polymers have been ascertained to have these properties, there appears to be no further research on them. Perhaps researchers have moved on to other polymers or other research interests. Ahuja and team explored
Mimosa pudica seed mucilage as a disintegrant, binder, mucoadhesive agent and sustained release polymer [
16,
78,
79]. However, they went a step further to graft acrylamide on
Mimosa pudica seed mucilage by microwave assisted graft co-polymerization. Perhaps more can be expected from this team.
Plantago psyllium mucilage may have been commercialized as some researchers obtained the mucilage from Sidpur Sat Isabgol factory, India [
25]. However, it is not certain if it has obtained approval as a pharmaceutical excipient.
Consider mucilages and gums from Ocimum gratissimum, Hibiscus rosasinensis, Okra (Abelmoschus esculentus), Leucaena leucocephata, Anogeissus latifolia, Prunus amygdalus, Cissus populnea, Moringa oleifera, Grewia mollis, Sesbania grandiflora, Scaphium scaphigerum, Caesalpinia spinosa, Mimosa pudica, Aloe barbadensis, Prosopis juliflora, etc. Can these polymers be explored for targeting, fabrication of nanoparticles, stimuli response, BioMEMS, and theranostics? They are natural polymers and most biocompatible. Can they be utilized in tissue engineering? These polymers ought to be extensively researched upon. Chitosan is a natural and approved polymer that has been found to have numerous abilities and hence it is employed for controlled release, mucoadhesion, targeting, BioMEMS, etc. When a polymer is extensively studied and so much data produced, it becomes easier for it to be approved by the regulatory authorities. The main obstacle to regulatory approval is cost. There are fees to be paid for any material’s review process before regulatory approval and in addition, the requirements for submission for approval such as scientific data for characterization, ascertainment of safety and clinical trials are capital intensive. Despite the obstacle, more natural polymers need to be approved in order to bring the future to the present or to be steps ahead.
7. Conclusions
Petrochemical resources are dwindling and in the near future, synthetic polymers may be in high demand thereby increasing their cost. In addition, the advocacy of green chemistry, green technology and green environment may precipitate dissatisfaction with synthetic polymers with patients insisting that the pharmaceutical industry use natural materials for the manufacture of drug delivery systems. Furthermore, utilization of natural polymers instead of synthetic polymers may reduce the cost of drug delivery systems. Natural polymers will continue to have increasing impact in drug delivery. As the shift from synthetic polymer to natural polymers continues, naturapolyceutics—extraction, purification, modifications, characterizations and applications of natural polymers in drug delivery—will continue to advance. Polymers are the fundamental facets of drug delivery and as drug delivery technologies continue to evolve and progress, there will be increasing demands on polymers and the need for new polymers. As the quest to meet the unmet needs of patients continues, it will necessitate the exploration of new polymers to meet these needs. However, for regulatory approval, the pharmaceutical industry will justifiably adopt polymers given marketing authorization for pharmaceutical use. New natural polymers are better positioned than new synthetic polymers for approval. In addition, the biological properties of natural polymers give them the edge over synthetic polymers. Natural polymers are the polymers of today and tomorrow. Therefore it behoves the polymer and pharmaceutical scientists to characterize and optimize more natural polymers, to move from bench to market. The study and utilization of natural polymers for drug delivery (Naturapolyceutics) is imperative for the present and the future!