New Guanidine-Pyridine Copper Complexes and Their Application in ATRP
Abstract
:1. Introduction
2. Experimental Section
| 1 [Cu(DMEGpy)2Cl][CuCl2] | 2 [Cu(DMEGpy)2Br][CuBr2] | 3 [Cu(DMEGpy)Cl2] | |
|---|---|---|---|
| Empirical formula | C22H32Cl3Cu2N8 | C22H32Br3Cu2N8 | C11H16Cl2CuN4 |
| Form. mass/g·mol−1 | 641.99 | 775.37 | 338.72 |
| Crystal Size/mm | 0.25 × 0.13 × 0.12 | 0.22 × 0.11 × 0.07 | 0.13 × 0.09 × 0.04 |
| T/K | 173(2) | 173(2) | 173(2) |
| Crystal system | Triclinic | Monoclinic | Orthorhombic |
| Space group | P1 | C2/c | Pna21 |
| a/Å | a = 11.265(1) | a = 22.783(2) | a = 8.783(1) |
| b/Å | b = 11.777(1) | b = 11.613(1) | b = 10.793(1) |
| c/Å | c = 12.135(1) | c = 22.834(3) | c = 14.605(1) |
| α/° | α = 114.5(1) | α = 90 | α = 90 |
| β/° | β = 104.1(1) | β = 113.6(2) | β = 90 |
| γ/° | γ = 99.6(1) | γ = 90 | γ = 90 |
| V/Å3 | 1,354.0(2) | 5,536.4(10) | 1,384.5(2) |
| Z | 2 | 8 | 4 |
| ρcalc./g/cm3 | 1.575 | 1.860 | 1.625 |
| µ/mm−1 | 1.894 | 5.894 | 1.950 |
| λ/Å | 0.71073 | 0.71073 | 0.71073 |
| F(000) | 658 | 3,064 | 692 |
| Range in hkl | ±13, ±14, ±14 | ±27, −14 ≤ k ≤ 12, −26 ≤ l ≤ 27 | ±10, ±13, ±17 |
| Reflections collected | 13,335 | 15,612 | 12,453 |
| Independent reflections | 5,037 | 5,159 | 2,562 |
| Rint. | 0.0344 | 0.0532 | 0.0494 |
| Reflections observed | 5,037 | 5,159 | 2,562 |
| No. parameters | 323 | 320 | 165 |
| R1 [I ≥ 2σ(I)] | 0.0301 | 0.0338 | 0.0251 |
| wR2 (all data) | 0.0631 | 0.0513 | 0.0415 |
| Goodness-of-fit | 0.893 | 0.854 | 0.893 |
| Largest difference peak, hole/e·Å−3 | 0.332 and −0.535 | 0.957 and −0.803 | 0.502 and −0.277 |
3. Results and Discussion
3.1. Complex Synthesis
3.1.1. Bis(chelate) Complexes 1 and 2
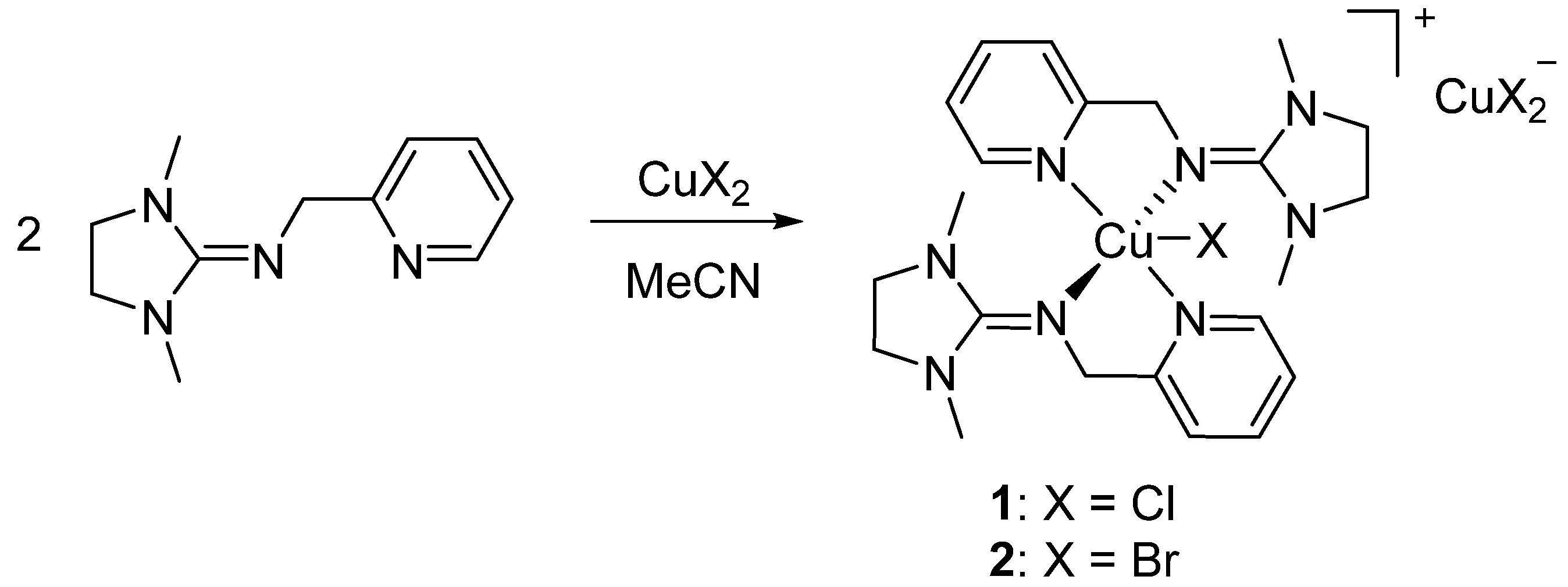
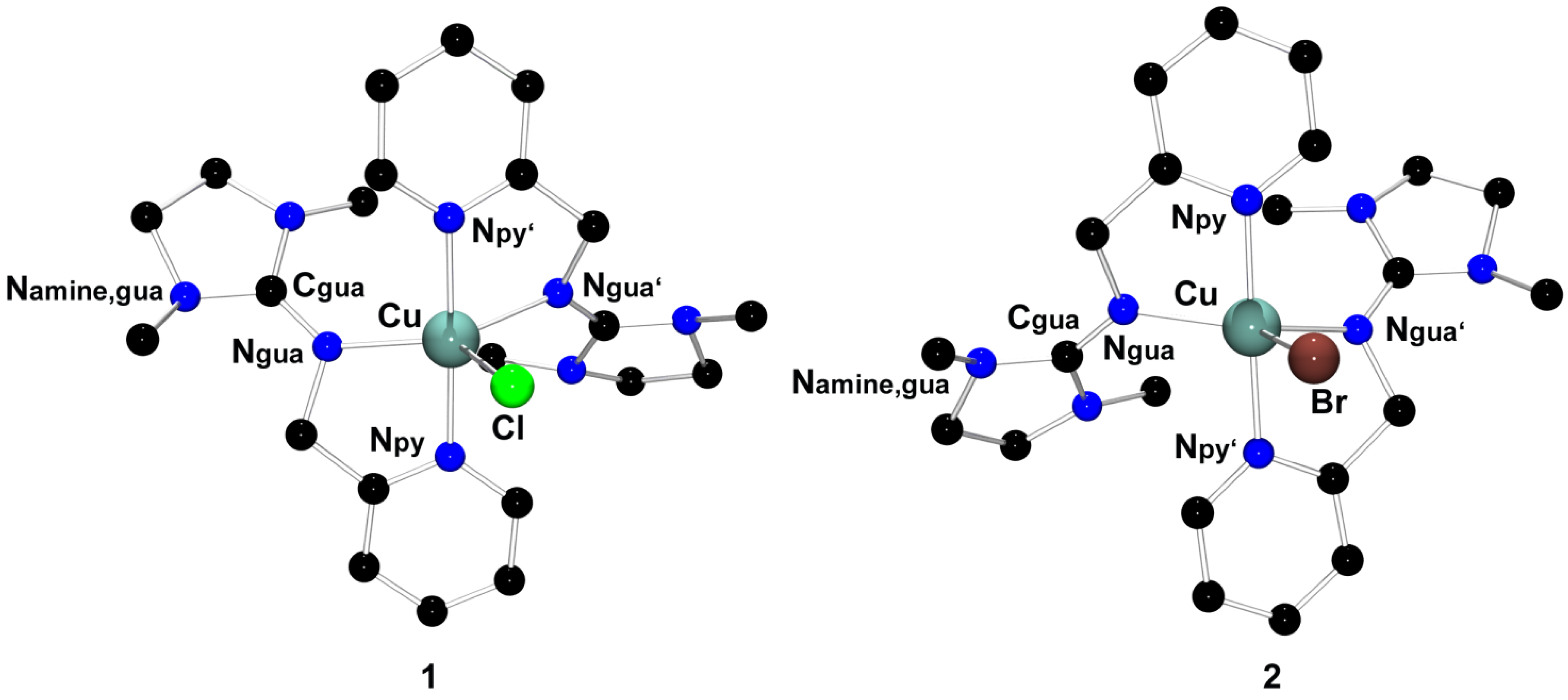
| 1 [Cu(DMEGpy)2Cl][CuCl2] | 2 [Cu(DMEGpy)2Br][CuBr2] | 3 [Cu(DMEGpy)Cl2] | |
|---|---|---|---|
| Bond lengths (Å) | |||
| Cu-Ngua | 2.041(2), 2.133(2) | 2.029(4), 2.065(3) | 1.956(3) |
| Cu-Npy | 1.988(2), 1.994(2) | 1.991(4), 1.993(1) | 2.016(3) |
| Cu-X | 2.398(2) | 2.589(1) | 2.210(1), 2.243(1) |
| Bond angles (°) | |||
| Ngua-Cu-Npy | 81.3(1), 80.7(1) | 81.7(2), 81.1(2) | 81.7(1) |
| Ngua-Cu-Ngua´ | 132.5(1) | 136.9(2) | |
| Npy-Cu-Npy´ | 178.6(1) | 177.6(2) | |
| Ngua-Cu-X | 125.5(1), 102.1(1) | 104.3(1), 118.8(1) | 97.1(1), 152.6(1) |
| Npy-Cu-X | 89.8(1), 90.8(1) | 87.5(1), 90.1(1) | 98.0(1), 138.5(1) |
| X-Cu-X | 100.8(1) | ||
| Angles between planes (°) | |||
| ∠(CuNax,CuNeq) | 85.2(1) | 80.3(1) | |
| ∠(CuN2,CuCl2) | 48.3(1) | ||
| ∠(Namine,guaC3,CguaN3) | 14.7(av) | 14.6(av) | 14.8(av) |
| Structural parameter ρ and τ5 | |||
| ρ | 0.95 | 0.95 | 0.95 |
| τ5 | 0.77 | 0.68 | 0.49 |
3.1.2. Mono(chelate) Complex 3
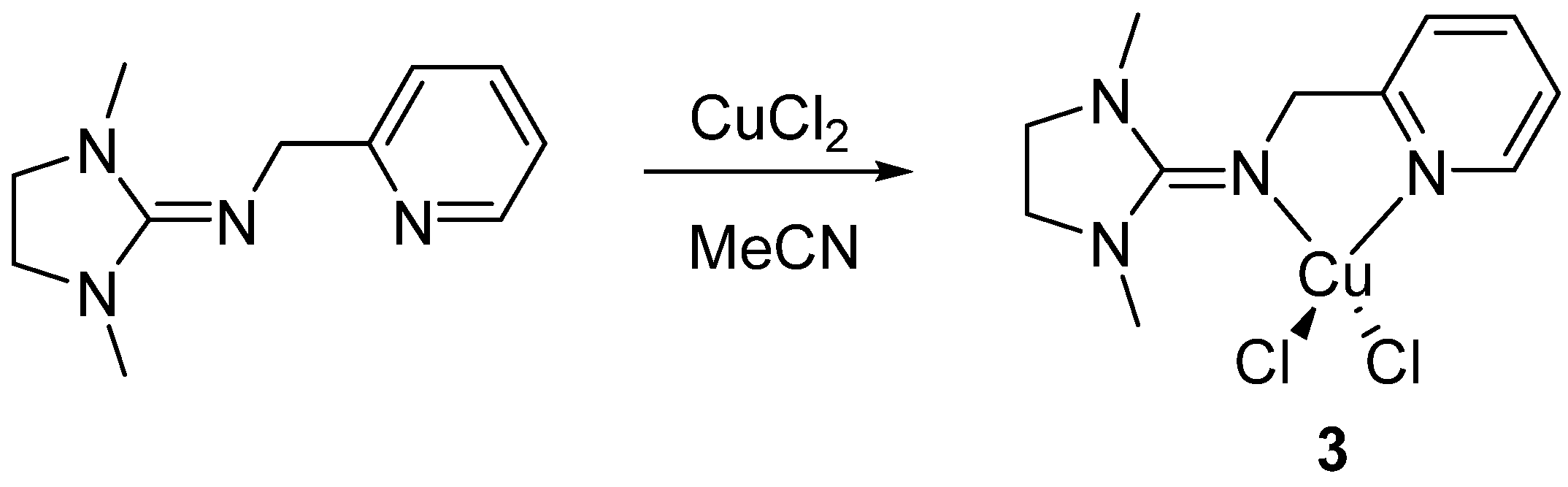

3.1.3. Comparative Structural Discussion
| 4 [Cu(bpy)2Cl]Cl·6H2O | 5 [Cu(bpy)2Br]Br | |
|---|---|---|
| Bond lengths (Å) | ||
| Cu-Nax | 1.989(10),1.970(10) | 1.977(6), 1.978(6) |
| Cu-Neq | 2.077(10), 2.087(10) | 2.075(8), 2.085(7) |
| Cu-X | 2.361(4) | 2.429(2) |
| Bond angles (°) | ||
| N-Cu-N | 79.3(4), 79.8(4) | 80.4(3), 80.3(3) |
| Nax-Cu-Nax | 178.3(4) | 177.3(3) |
| Neq-Cu-Cl | 118.7(3), 118.6(3) | 128.6(2), 124.7(2) |
| Neq-Cu-Neq | 122.8(4) | 106.7(3) |
| Structural parameter τ5 | ||
| τ5 | 0.93 | 0.81 |
3.2. Atom Transfer Radical Polymerization of Styrene
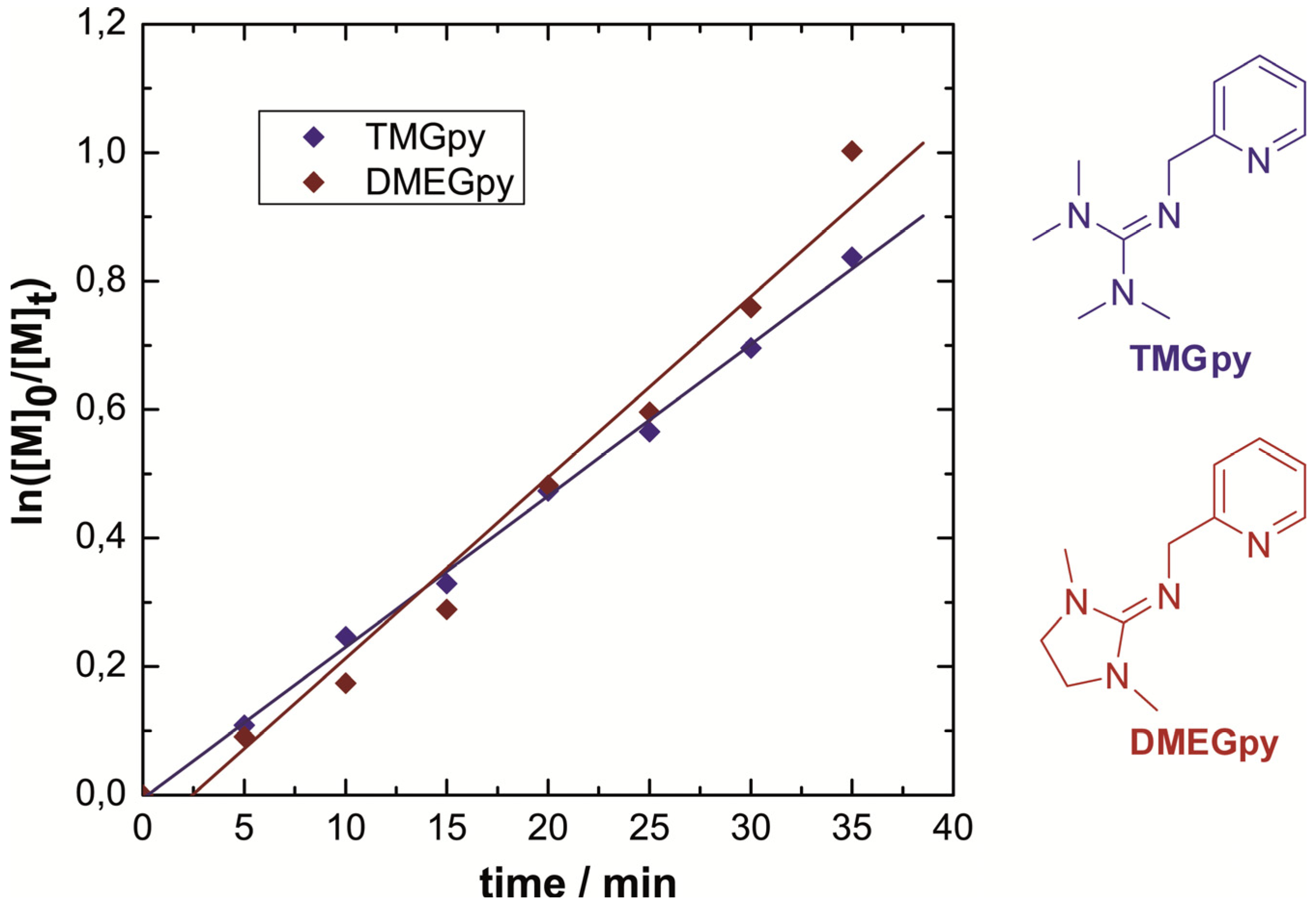
| Catalyst | t (min) | Conversion (%) | Mn,GPC (g/mol) | Mn,th (g/mol) | Mw/Mn |
|---|---|---|---|---|---|
| 2 TMGpy/CuBr | 10 | 22 | 2900 | 2200 | 1.19 |
| 2 TMGpy/CuBr | 35 | 57 | 8500 | 4900 | 1.24 |
| 2 DMEGpy/CuBr | 10 | 16 | 1900 | 1700 | 1.53 |
| 2 DMEGpy/CuBr | 35 | 63 | 9600 | 6600 | 1.25 |

4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Matyjaszewski, K.; Davis, T.P. Handbook of Radical Polymerization; Wiley Interscience: Hoboken, NJ, USA, 2002. [Google Scholar]
- Wang, J.-S.; Matyjaszewski, K. Controlled/“living” radical polymerization. Atom transfer radical polymerization in the presence of transition-metal complexes. J. Am. Chem. Soc. 1995, 117, 5614–5615. [Google Scholar] [CrossRef]
- Patten, T.E.; Matyjaszewski, K. Atom Transfer Radical Polymerization and the Synthesis of Polymeric Materials. Adv. Mater. 1998, 10, 901–915. [Google Scholar] [CrossRef]
- Matyjaszewski, K.; Xia, J. Atom Transfer Radical Polymerization. Chem. Rev. 2001, 101, 2921–2990. [Google Scholar] [CrossRef]
- Jakubowski, W.; Matyjaszewski, K. Activators Regenerated by Electron Transfer for Atom-Transfer Radical Polymerization of (Meth)acrylates and Related Block Copolymers. Angew. Chem. Int. Ed. 2006, 45, 4482–4486. [Google Scholar] [CrossRef]
- Matyjaszewski, K.; Jakubowski, W.; Min, K.; Tang, W.; Huang, J.; Braunecker, W.A.; Tsarevsky, N.V. Diminishing catalyst concentration in atom transfer radical polymerization with reducing agents. Proc. Natl. Acad. Sci. USA 2006, 103, 15309–15314. [Google Scholar] [CrossRef]
- Magenau, A.J.D.; Strandwitz, N.C.; Gennaro, A.; Matyjaszewski, K. Electrochemically Mediated Atom Transfer Radical Polymerization. Science 2011, 332, 81–84. [Google Scholar] [CrossRef]
- Bortolamei, N.; Isse, A.A.; Magenau, A.J.D.; Gennaro, A.; Matyjaszewski, K. Controlled Aqueous Atom Transfer Radical Polymerization with Electrochemical Generation of the Active Catalyst. Angew. Chem. Int. Ed. 2011, 50, 11391–11394. [Google Scholar] [CrossRef]
- Konkolewicz, D.; Wang, Y.; Zhong, M.; Krys, P.; Isse, A.A.; Gennaro, A.; Matyjaszewski, K. Reversible-Deactivation Radical Polymerization in the Presence of Metallic Copper. A Critical Assessment of the SARA ATRP and SET-LRP Mechanisms. Macromolecules 2013, 46, 8749–8772. [Google Scholar] [CrossRef]
- Tang, W.; Kwak, Y.; Braunecker, W.; Tsarevsky, N.V.; Coote, M.L.; Matyjaszewski, K. Understanding Atom Transfer Radical Polymerization: Effect of Ligand and Initiator Structures on the Equilibrium Constants. J. Am. Chem. Soc. 2008, 130, 10702–10713. [Google Scholar]
- Herres-Pawlis, S.; Neuba, A.; Seewald, O.; Seshadri, T.; Egold, H.; Flörke, U.; Henkel, G. A library of peralkylated bisguanidine ligands for use in biomimetic coordination chemistry. Eur. J. Org. Chem. 2005, 2005, 4879–4890. [Google Scholar]
- Herres-Pawlis, S.; Flörke, U.; Henkel, G. Tuning of Copper(I)-Dioxygen Reactivity by Bisguanidine Ligands. Eur. J. Inorg. Chem. 2005, 2005, 3815–3824. [Google Scholar] [CrossRef]
- Wortmann, R.; Hoffmann, A.; Haase, R.; Flörke, U.; Herres-Pawlis, S. Synthese und Charakterisierung von Cobalt(II)- und Kupfer(I)-Komplexen mit Guanidin-Pyridin-Hybridliganden. Z. Anorg. Allg. Chem. 2009, 635, 64–69. [Google Scholar] [CrossRef]
- Herres-Pawlis, S.; Haase, R.; Henkel, G.; Binder, S.; Eich, A.; Schulz, B.; Rübhausen, M.; Wellenreuther, G.; Meyer-Klaucke, W. Stabilisation of a Highly Reactive Bis-(µ-oxo)dicopper(III) Species at Room Temperature by Electronic and Steric Constraint of an Unconventional Nitrogen Donor Ligand. Chem. Eur. J. 2009, 35, 8678–8682. [Google Scholar]
- Herres-Pawlis, S.; Seshadri, T.; Flörke, U.; Henkel, G. Reactivity of 2,2′-Bis(2N-(1,1′,3,3′-tetramethyl-guanidino))diphenylene-amine with CuI and [Cu(MeCN)4][PF6]: benzimidazole formation vs. Cu oxidation. Z. Anorg. Allg. Chem. 2009, 635, 1209–1214. [Google Scholar] [CrossRef]
- Herres-Pawlis, S.; Berth, G.; Wiedemeier, V.; Schmidt, L.; Zrenner, A.; Warnecke, H.-J. Oxygen sensing by fluorescence quenching of [Cu(btmgp)I]. J. Lumin. 2010, 130, 1958–1962. [Google Scholar] [CrossRef]
- Haase, R.; Beschnitt, T.; Flörke, U.; Herres-Pawlis, S. Bidentate guanidine ligands with ethylene spacer in copper-dioxygen chemistry: Structural characterization of bis(l-hydroxo) dicopper complexes. Inorg. Chim. Acta 2011, 374, 546–557. [Google Scholar] [CrossRef]
- Bienemann, O.; Hoffmann, A.; Herres-Pawlis, S. (Guanidine)copper complexes: structural variety and application in bioinorganic chemistry and catalysis. Rev. Inorg. Chem. 2011, 3, 83–108. [Google Scholar]
- Petrovic, D.; Hill, L.M.R.; Jones, P.G.; Tolman, W.B.; Tamm, M. Synthesis and reactivity of copper(I) complexes with an ethylene-bridged bis(imidazolin-2-imine) ligand. Dalton Trans. 2008, 7, 887–894. [Google Scholar]
- Oakley, S.H.; Coles, M.P.; Hitchcock, P.B. Structural and catalytic properties of bis(guanidine)copper(I) halides. Inorg. Chem. 2003, 42, 3154–3156. [Google Scholar] [CrossRef]
- Brar, A.S.; Kaur, S. Tetramethylguanidino-tris(2-aminoethyl)amine: A novel ligand for copper-based atom transfer radical polymerization. J. Polym. Sci. A Polym. Chem. 2005, 43, 5906–5922. [Google Scholar] [CrossRef]
- Bienemann, O.; Haase, R.; Flörke, U.; Döring, A.; Kuckling, D.; Herres-Pawlis, S. Neue Bisguanidin-Kupfer-Komplexe und ihre Anwendung in der ATRP. Z. Naturforsch. 2010, 65b, 798–806. [Google Scholar]
- Bienemann, O.; Haase, R.; Jesser, A.; Beschnitt, T.; Döring, A.; Kuckling, D.; dos Santos Vieira, I.; Flörke, U.; Herres-Pawlis, S. Synthesis and Application of new Guanidine Copper Complexes in Atom Transfer Radical Polymerisation. Eur. J. Inorg. Chem. 2011, 2011, 2367–2379. [Google Scholar]
- Bienemann, O.; Froin, A.-K.; dos Santos Vieira, I.; Wortmann, R.; Hoffmann, A.; Herres-Pawlis, S. Structural Aspects of Copper-Mediated Atom Transfer Radical Polymerisation with a Novel Tetradentate Bisguanidine Ligand. Z. Anorg. Allg. Chem. 2012, 638, 1683–1690. [Google Scholar] [CrossRef]
- Leonard, J.; Lygo, B.; Procter, G. Praxis der Organischen Chemie; Wiley-VCH: Weinheim, Germany, 1996. [Google Scholar]
- Kantlehner, W.; Haug, E.; Mergen, W.W.; Speh, P.; Maier, T.; Kapassakalidis, J.J.; Bräuner, H.-J.; Hagen, H.; Orthoamide, X.L. Herstellung von 1,1,2,3,3-pentasubstituierten und 1,1,2,2,3,3-hexasubstituierten Guanidiniumsalzen sowie von 1,1,2,3,3-Pentaalkylguanidinen. Liebigs Ann. Chem. 1984, 1, 108–125. [Google Scholar]
- Hoffmann, A.; Börner, J.; Flörke, U.; Herres-Pawlis, S. Synthesis and fluorescence properties of guanidine-pyridine hybridligands and structural characterisation of their mono- and bis(chelated) cobalt complexes. Inorg. Chim. Acta 2009, 362, 1185–1193. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Phase annealing in SHELX-90: Direct methods for larger structures. Acta Crystallogr. 1990, A46, 467–473. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXL; University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
- CCDC CIF Depository Request Form. Available online: http://www.ccdc.cam.ac.uk/data_request/cif (accessed on 27 March 2014).
- Addison, A.W.; Rao, T.N.; Reedik, J.; van Rijin, J.; Verschoor, G.C. Synthesis, Structure, and Spectroscopic Properties of Copper(II) Compounds containing Nitrogen-Sulphur Donor Ligands; the Crystal and Molecular Structure of Aqua[l,7-bis(N-methylbenzimidazol-2′-yl)-2,6-dithiaheptane]copper(II) Perchlorate. J. Chem. Soc. Dalton Trans. 1984, 1984, 1349–1356. [Google Scholar]
- Gispert, J.R. Coordination Chemistry; Wiley-VCH: Weinheim, Germany, 2008. [Google Scholar]
- Rossi, A.R.; Hoffmann, R. Transition metal pentacoordination. Inorg. Chem. 1975, 14, 365–374. [Google Scholar] [CrossRef]
- Yang, L.; Powell, D.R.; Houser, R.P. Structural variation in copper(I) complexes with pyridylmethylamide ligands: structural analysis with a new four-coordinate geometry index, τ4. Dalton Trans. 2007, 2007, 955–964. [Google Scholar] [CrossRef]
- Stephens, F.S.; Tucker, P.A. Crystal and molecular structure of chlorobis(2,2′-bipyridyl)copper(II) chloride hexahydrate. J. Chem. Soc. Dalton Trans. 1973, 1973, 2293–2297. [Google Scholar] [CrossRef]
- Khan, M.A.; Tucker, D.G. Structure of bis(2,2′-bipyridine)monobromocopper(II) bromide. Acta Cryst. 1981, B37, 1409–1412. [Google Scholar] [CrossRef]
- Matyjaszewski, K.; Patten, T.E.; Xia, J. Controlled/“Living” Radical Polymerization. Kinetics of the Homogeneous Atom Transfer Radical Polymerization of Styrene. J. Am. Chem. Soc. 1997, 119, 674–680. [Google Scholar] [CrossRef]
- Poli, R. Relationship between One-Electron Transition-Metal Reactivity and Radical Polymerization Processes. Angew. Chem. Int. Ed. 2006, 45, 5058–5070. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hoffmann, A.; Bienemann, O.; Vieira, I.D.S.; Herres-Pawlis, S. New Guanidine-Pyridine Copper Complexes and Their Application in ATRP. Polymers 2014, 6, 995-1007. https://doi.org/10.3390/polym6040995
Hoffmann A, Bienemann O, Vieira IDS, Herres-Pawlis S. New Guanidine-Pyridine Copper Complexes and Their Application in ATRP. Polymers. 2014; 6(4):995-1007. https://doi.org/10.3390/polym6040995
Chicago/Turabian StyleHoffmann, Alexander, Olga Bienemann, Ines Dos Santos Vieira, and Sonja Herres-Pawlis. 2014. "New Guanidine-Pyridine Copper Complexes and Their Application in ATRP" Polymers 6, no. 4: 995-1007. https://doi.org/10.3390/polym6040995





