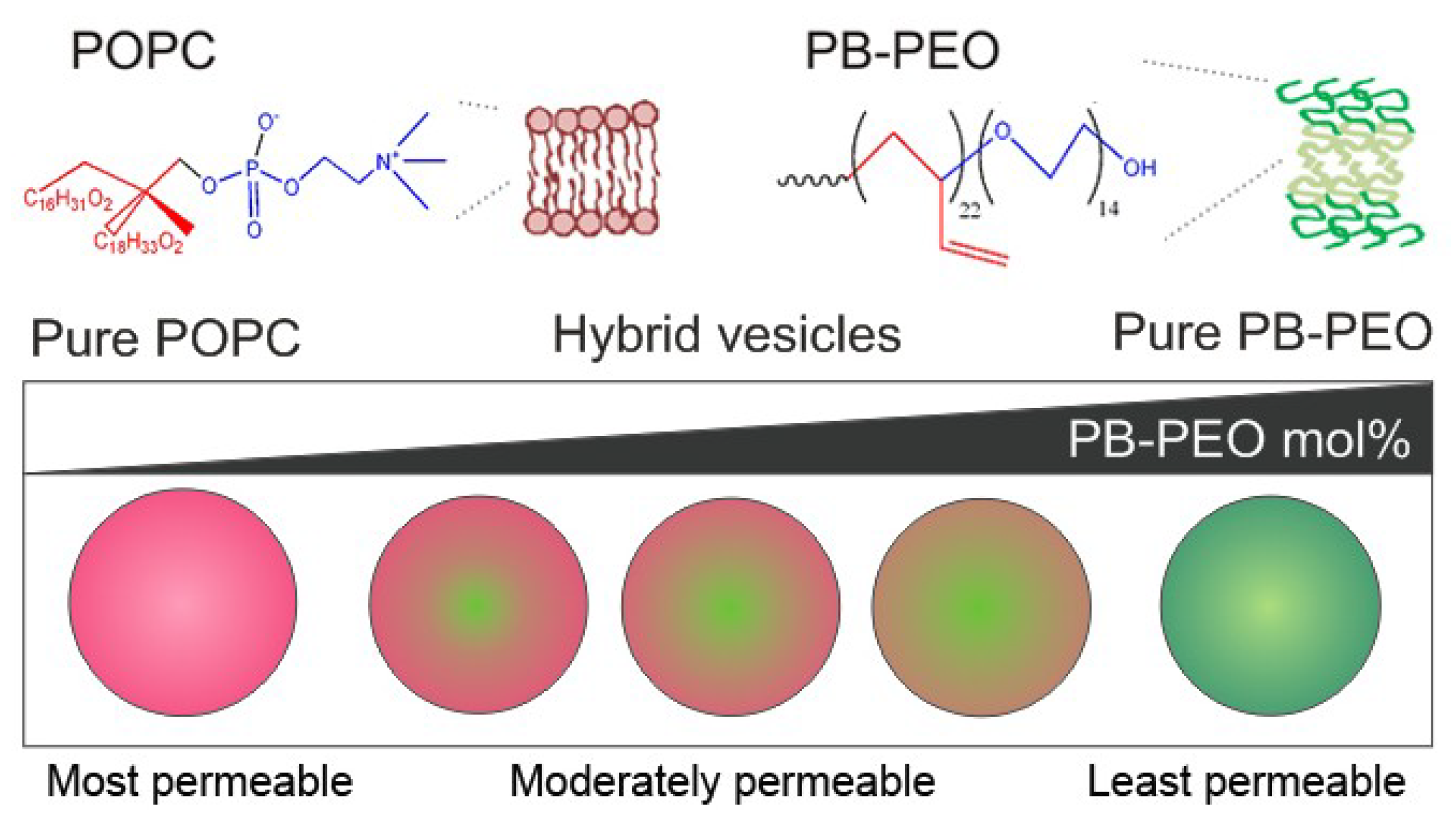
1. Introduction
Polymersomes are hollow vesicles formed by self-assembly and bilayer formation of amphiphilic block copolymers. Since the initial work by the groups of Discher and Eisenberg [
1,
2], sophisticated polymersome designs have been explored involving degradable or non-degradable systems [
3], or those that are responsive towards external stimuli [
4]. Potential applications of polymersomes include delivery of drugs [
5] and other bioactive substances such as siRNA [
6] and antibodies [
7], and they have been used as carriers for probes in bioimaging [
8,
9] or for biocatalysts in the form of nanoreactors [
10,
11]. In general, the polymersome membrane displays enhanced stability as compared to liposomes [
8,
12] and they can, hence, more readily be modified. In more detail, both the thickness and hydrophobicity of the polymer vesicle membrane confer enhanced stability and a concurrent lower permeability [
1]. The chemical versatility of block copolymer allows the tuning of the membrane properties and functionality of the polymersomes by simple variation of the molecular weight or by rigorous chemical design [
12]. Nevertheless, because they are formed from synthetic building blocks, polymersomes are considered to display a reduced biocompatibility and biofunctionality when compared to natural phospholipids. The potential toxicity of long term accumulation of synthetic polymers in the body remains unknown, as is the functionality of bioactive moieties incorporated into the polymeric membrane. On the other hand, over the past decades, liposomes have been studied and employed as drug carriers, leading to commercial formulations such as Doxil [
13]. Despite their inherent instability and limited shelf life, they currently form a major drug carrier, with polymersomes so far lagging behind in applications.
It may be concluded that liposomes and polymersomes have their own particular benefits and limitations, which is why the preparation of hybrid membranes consisting of phospholipids and block copolymers is attracting increasing attention [
14,
15,
16,
17,
18,
19,
20,
21]. In this way, the robustness and chemical versatility of polymersomes can be combined with the softness and biocompatibility of liposomes. An important issue in this respect is whether the polymers and lipids assemble to form homogeneously mixed hybrids or rather display heterogeneity, forming a two-phase architecture with lipid-rich domains in a polymer-rich membrane (and
vice versa) or, in the extreme case, form separate populations of liposomes and polymersomes.
Using fluorescence microscopy, several recent studies observed the formation of hybrid lipid/polymer giant unilamellar vesicles (GUVs) of 10–50 µm, prepared via electroformation, over a certain range of compositions [
14,
15,
16,
17,
18]. Parameters controlling the self-assembly of lipids with block copolymers (forming hybrids GUVs), and the resulting phase behavior, are the mole fraction of the constituting amphiphiles, as well as the nature of the amphiphiles themselves [
14,
15,
16,
17]. Nam
et al. [
15] observed efficient mixing of PB
46-PEO
30 [polybutadiene-
b-poly(ethylene oxide)] and POPC (1-palmitoyl-2-oleoyl-
sn-glycero-3-phosphatidylcholine) to occur only at polymer concentrations above 65%, while Schulz
et al. [
17] found hybrid GUVs to form at all investigated molar ratios of 1,2-dipalmitoyl-
sn-glycero-3-phosphocholine (DPPC) and polyisobutylene-
b-PEO. The membrane thickness of the polymers and the melting transition temperature of the lipids have been demonstrated to significantly affect the formation, morphology and/or stability of hybrid GUVs, as well [
14]. It was observed that at high lipid composition, mixing POPC (melting transition temperature = −2 °C) with poly(dimethylsiloxane)-
g-poly(ethylene oxide) (PDMS-
g-PEO) forms separated polymersomes and liposomes as results of budding process, while mixing DPPC (melting transition temperature = 41 °C) with PDMS-
g-PEO under the same condition yields stable hybrid GUVs.
Importantly, it should be stressed that the phase behavior and membrane properties of micron-sized giant GUVs may not necessarily apply to nanoscale vesicles (
ca. 100 nm in diameter) of the same composition, with the latter size range being optimal for
in vivo applications (above
ca. 500 nm particles are rapidly filtered out of the blood stream [
13]). Indeed, membrane characteristics and the mechanical properties of the bilayer may vary significantly with vesicle size, an effect that is especially relevant for nanoscale vesicles [
22].
In previous studies of nanoscale hybrid vesicles, it was found that poly(2-methyloxazoline)-
b-poly(dimethylsiloxane)-
b-poly(2-methyloxazoline) (PMOXA-PDMS-PMOXA) and either phosphatidylethanolamine or DPPC display homogenous mixing [
20]. Very recently, Winzen
et al. [
19] demonstrated the preparation of two different types of nanoscale hybrid vesicles,
i.e., PDMS-
b-PMOXA/1,2-dimyristoyl-
sn-glycero-3-phosphocholine (DMPC) and PDMS-
b-PMOXA/cholesterol hybrids. They found that polymer/cholesterol hybrids exhibit higher membrane packing density and increased bending stiffness compared to pure polymersomes and polymer/lipid hybrids. Another study investigated the efficiency of cancer cell targeting of nanoscale hybrid vesicles of PB
22-PEO
14 and hydrogenated soy phosphatidylcholine [
21]. Unfortunately, no detailed information on the structural properties of the hybrid membrane was provided.
Here, we are specifically interested in the formation and properties of nanometer-sized hybrid phospholipid/block copolymer vesicles formed from PB
22-PEO
14 and POPC. We demonstrate the preparation of nanometer-sized hybrid vesicles composed of PB-PEO and POPC via film-hydration and extrusion. We then study the formation of these PB-PEO/POPC hybrid vesicles using flow cytometry and observe a possible difference in mixing behavior of polymers and lipids in micron and nanometer sized vesicles, prepared by electroformation and film-rehydration/extrusion, respectively. We demonstrate that the size, encapsulation efficiency and content release behavior of nanoscale hybrid vesicles can be tuned by the mixing ratio of the amphiphiles, and is reminiscent of the effect of “PEGylation” on phospholipid vesicles (
Figure 1).
Figure 1.
Schematic diagram of nanoscale phospholipid/block copolymer hybrid vesicles.
Figure 1.
Schematic diagram of nanoscale phospholipid/block copolymer hybrid vesicles.
2. Experimental Section
2.1. Materials
PB-PEO (PB22-PEO14, average molecular weight for the PB and PEO block: 1200 and 600, respectively; PB46-PEO30, average molecular weight for the PB and PEO block: 2500 and 1300, respectively) was purchased from Polymer Source (Dorval, Quebec, Canada). 1-Palmitoyl-2-oleoyl-sn-glycero-3-phosphatidylcholine (POPC), 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine-N-(lissamine rhodamine B sulfonyl) (Rho-PE), and 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine-N-(1-pyrenesulfonyl)(pyrene-PE) were purchased from Avanti Polar Lipids (Alabaster, AL, USA). All other chemicals were purchased from Sigma Aldrich (Singapore).
2.2. Preparation of Vesicles
Nanosized vesicles were formed by film rehydration followed by extrusion. The solvent was removed using a gentle stream of nitrogen prior to vacuum desiccation overnight. Vesicles were formed by adding 1.0 mL of 0.1 M phosphate-buffered saline (PBS), pH 7.4 to the dried film, followed by incubation at 45 °C for 20 min and stirring for 2 h at 300 rpm. The vesicles were subsequently extruded 20 times through 450 and 200 nm membranes to form polymersomes. Stock solutions of PB-PEO and POPC in chloroform were mixed in the following molar ratios: 100:0, 75:25, 25:75 and 0:100. The total amount of PB-PEO and POPC for each sample was 5 mg. For flow cytometry studies, 0.5% tetramethylrhodamine-PB-PEO (TMRho-PB-PEO) and/or 1% of pyrene-PE were added before film formation. Preparation of TMRho-PB-PEO was performed by reacting 5(6)-tetramethyl-rhodamine isothiocyanate (TRITC) with amino-terminated PB-PEO following a method reported in literature [
21]. The product was purified by dialysis.
Giant unilamellar vesicles (GUVs) were formed by electroformation using a homemade set-up. Dry lipid/polymer films on conductive indium tin oxide glass slides (70–100 Ω/sq surface resistivity; Sigma Aldrich), cast from 5 µL of 5 mg/mL stock solution, were hydrated with 300 mM sucrose solution at 45 °C at an alternating sine-wave current of 10 Hz and 3 V for 120 min. Imaging of GUVs was conducted using a DeltaVision microscope (Applied Precision Inc., Issaquah, WA, USA) fitted with a PLAPON 60XO/1.42 NA oil-immersion objective (Olympus, Tokyo, Japan), and DAPI (4,6-diamidino-2-phenylindole), TRITC, FITC (fluorescein isothiocyanate), and CY5 (cyanine 5) Semrock filters (New York, NY, USA). Images were processed using ImageJ freeware and DeltaVision software (Softworx 4.1.0, Applied Precision, Inc., Issaquah, WA, USA).
2.3. Instrumentation
Flow cytometry experiments were performed using a Calibur flow cytometer (BD Biosciences, Franklin Lakes, NJ, USA). A volume of 100 µL of 5 mg/mL 0.5% TMRho-labeled and/or 1% pyrene-PE labeled vesicles were added to 1mL of PBS. All experiments analyzed a minimum of 10,000 events. Electronic compensation was conducted using separated color controls. Data were presented in two-dimensional dot plots between TMRho/Pyrene using forward scatter/side scatter (FSC/SSC) gating to select vesicles for analysis.
Dynamic light scattering (DLS) experiments were performed using a Zetasizer Nano ZS (Malvern, Worcestshire, UK) with a 633 nm helium-neon laser using back-scattering detection. A 20× diluted vesicle sample in 400 µL PBS was measured at a fixed 173° scattering angle. Three measurements, consisting of 3 runs of 30 s duration, were performed at 25 °C. Vesicle hydrodynamic size was reported as the average of the three measurements with standard deviation.
2.4. Carboxyfluorescein Leakage Experiments
For carboxyfluorescein (CF) dye encapsulation, 1.0 mL of 50 mM CF in 20 mM sodium phosphate, 10 mM NaCl (pH 7.4) was added to the dried PB-PEO/POPC films, after which vesicle preparation was conducted as described above. Non-entrapped CF was removed via gel filtration using a PD-10 column (GE Healthcare, Singapore) eluting with 0.1 M PBS buffer, pH = 7.4. The encapsulation efficiency was calculated via 100 × Ft/F0, where Ft is the encapsulated CF concentration (as calculated by CF fluorescence after adding 5 µL 10% Triton X-100) and F0 is the initial CF concentration of the stock solution.
For leakage studies, 50 mM CF (self-quenching concentration) was encapsulated in the vesicles. Under these conditions, when CF is released from the vesicles in the surrounding aqueous buffer, fluorescence intensity will increase because of dequenching. Gel-filtered CF-encapsulating vesicles (20 µL) were mixed with 180 µL PBS in a 96 well-plate. The release of CF from the vesicles in PBS at room temperature was monitored as a function of time using a fluorescence plate reader (Infinite 200, Tecan, Salzburg, Austria; λex = 480 nm, λem = 520 nm). The time between gel filtration of the polymersomes and the start of the release experiment was kept within 30 min. The percentage of CF release over time is presented by 100 × (F − F0)/(FT − F0), where F0 is the initial fluorescence of CF, F is the fluorescence of CF at time interval t and FT represents total fluorescence, i.e., after full release of CF upon addition of 0.5% Triton X-100.
3. Results and Discussion
Studies reported to date on hybrid vesicles have not yielded detailed information on the efficiency of hybrid vesicle formation in bulk solution [
14,
15,
16,
17,
18,
19,
20,
21]. Flow cytometry has been demonstrated as a useful tool to study vesicle populations [
23]. To prove that indeed hybrid vesicles, and not separated liposome and polymersome architectures were present in solution, flow cytometry studies were performed using TMRho-labeled PB-PEO and pyrene-labeled PE lipids.
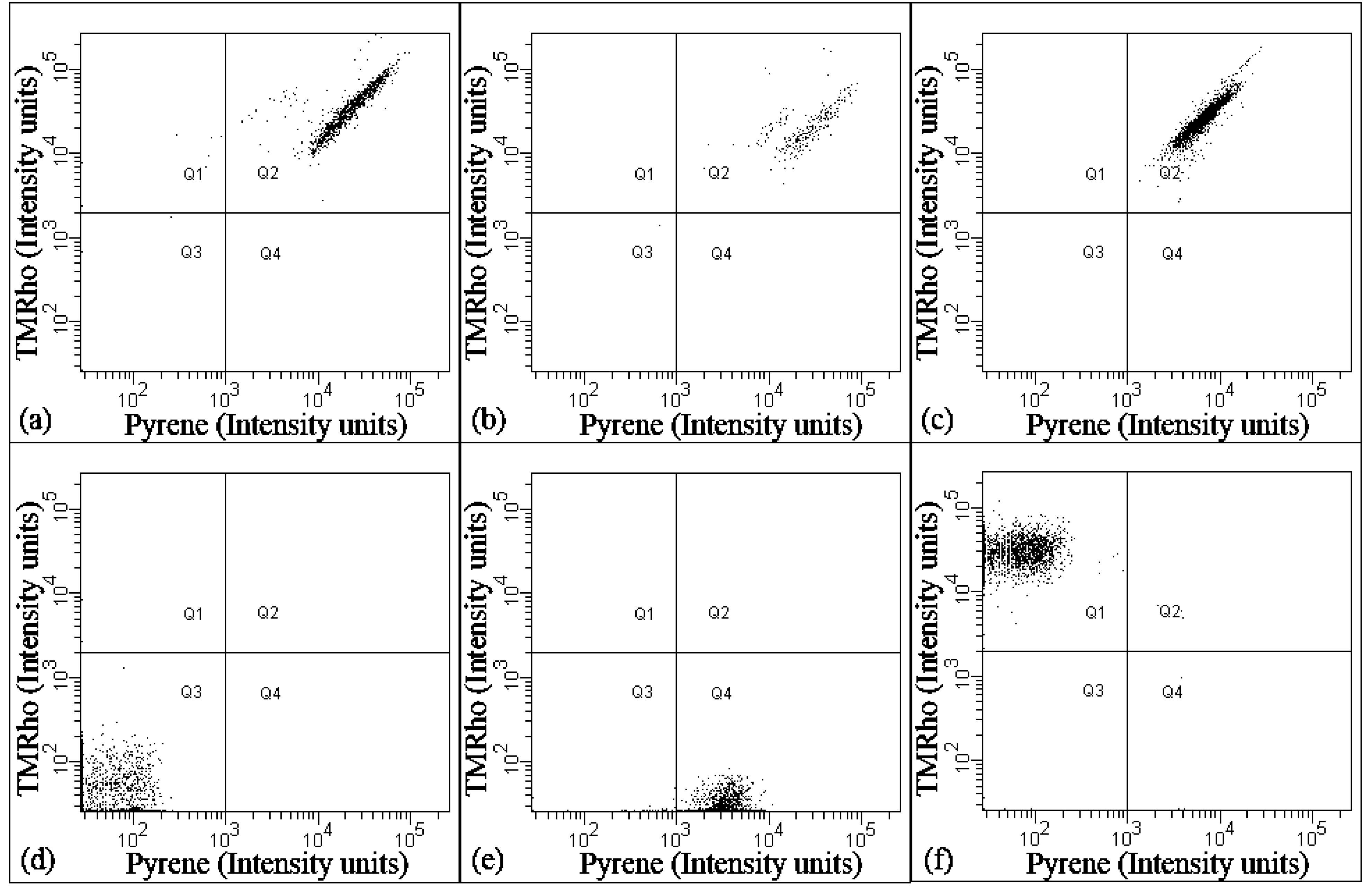
Figure 2 and
Table 1 show that virtually all vesicles (>99% of the population) contain both pyrene and TMRho, suggesting all vesicles to carry both PB-PEO and POPC and constitute hybrids. Single pyrene-labeled vesicles or TMRho-labeled vesicles were not observed. The dot plots of PB-PEO/POPC (75:25) and (50:50) appear to display two populations of hybrid that differ in pyrene fluorescence intensity (
Figure 2a,b). Because we did not observe a heterogeneous population in our light-scattering experiments (see below), we tentatively ascribe this observation to the tendency of pyrene to form excimers [
24], or due to inhomogeneity in lipid compositions, as observed for individual nanoscale liposomes [
25]. In summary, these results imply that film-rehydration of POPC and PB-PEO block copolymers blends yields nanometer-sized hybrid vesicles over the entire range of compositions studied.
Figure 2.
Flow-cytometric analysis of vesicle samples of (a) PB-PEO/POPC (75:25); (b) PB-PEO/POPC (50:50); (c) PB-PEO/POPC (25:75). The samples have been labeled with TMRho-PB-PEO and pyrene-PE to confirm the presence of hybrid vesicles in the population; (d) Control PB-PEO sample (unlabeled); (e) Control PB-PEO sample (pyrene-labeled); and (f) Control PB-PEO sample (TMRho-labeled). The x and y axes of each dot plot represent the fluorescence intensity of pyrene and TMRho, respectively.
Figure 2.
Flow-cytometric analysis of vesicle samples of (a) PB-PEO/POPC (75:25); (b) PB-PEO/POPC (50:50); (c) PB-PEO/POPC (25:75). The samples have been labeled with TMRho-PB-PEO and pyrene-PE to confirm the presence of hybrid vesicles in the population; (d) Control PB-PEO sample (unlabeled); (e) Control PB-PEO sample (pyrene-labeled); and (f) Control PB-PEO sample (TMRho-labeled). The x and y axes of each dot plot represent the fluorescence intensity of pyrene and TMRho, respectively.
Table 1.
Summary of flow-cytometric analysis of vesicle samples of PB-PEO/POPC (25:75); PB-PEO/POPC (50:50); and PB-PEO/POPC (75:25). All vesicles (>99% of vesicle population) contain both pyrene and TMRho, suggesting each vesicle is a hybrid containing PB-PEO [(polybutadiene-b-poly(ethylene oxide)] and POPC (1-palmitoyl-2-oleoyl-sn-glycero-3-phosphatidylcholine).
Table 1.
Summary of flow-cytometric analysis of vesicle samples of PB-PEO/POPC (25:75); PB-PEO/POPC (50:50); and PB-PEO/POPC (75:25). All vesicles (>99% of vesicle population) contain both pyrene and TMRho, suggesting each vesicle is a hybrid containing PB-PEO [(polybutadiene-b-poly(ethylene oxide)] and POPC (1-palmitoyl-2-oleoyl-sn-glycero-3-phosphatidylcholine).
| Vesicles | % of hybrids (Pyrene and TMRho-labeled) | % Pyrene-labeled | % TMRho-labeled |
|---|
| PB-PEO/POPC (25:75) | 99.1 | 0.2 | 0.7 |
| PB-PEO/POPC (50:50) | 99.2 | 0.2 | 0.6 |
| PB-PEO/POPC (75:25) | 100 | 0 | 0 |
The results are significantly different from those obtained in a previous study of micrometer-sized hybrid GUVs formed from POPC and PB-PEO, even though this comprised a higher molecular weight polymer,
i.e., PB
46-PEO
30 (
MW for PB and PEO blocks: 2500 and 1300, respectively), and a different preparation method [
15]. In the respective study, hybrid GUVs formation was inefficient at high lipid composition (>65 mol %), while at intermediate lipid compositions (35–65 mol %) GUVs were not formed at all. This was explained in terms of the energetically unfavorable incorporation of the relatively thick block of the high molecular weight polymer (8–10 nm) into the thin lipid-rich membrane (3–4 nm).
Furthermore, the difference between ours and the system described above is two-fold. Firstly, the molecular weight of the polymer used in this study is significantly less (1800
vs. 3800) and the resulting reduced length may be more favorable for mixing. The increased membrane thickness mismatch could energetically be less favorable. Simulation and experimental studies have suggested block copolymers with molecular weights below 3 kDa or membrane thicknesses below 7 nm adopt more stretched and ordered membrane configuration (compared to more flexible entangled configuration of long block copolymers), which is consistent with the strongly segregated state of lipids [
26,
27], making mixing energetically more favorable. To re-confirm this effect, we also performed flow cytometry on nanoscale vesicles prepared from mixtures of POPC and the longer polymer, PB
46-PEO
30 (
MW = 3800). The dot plots results shows the fraction of hybrids to measure 17%–36%, suggesting that the mixing of this copolymer with lipids is indeed less efficient (Figure S1 and Table S1).
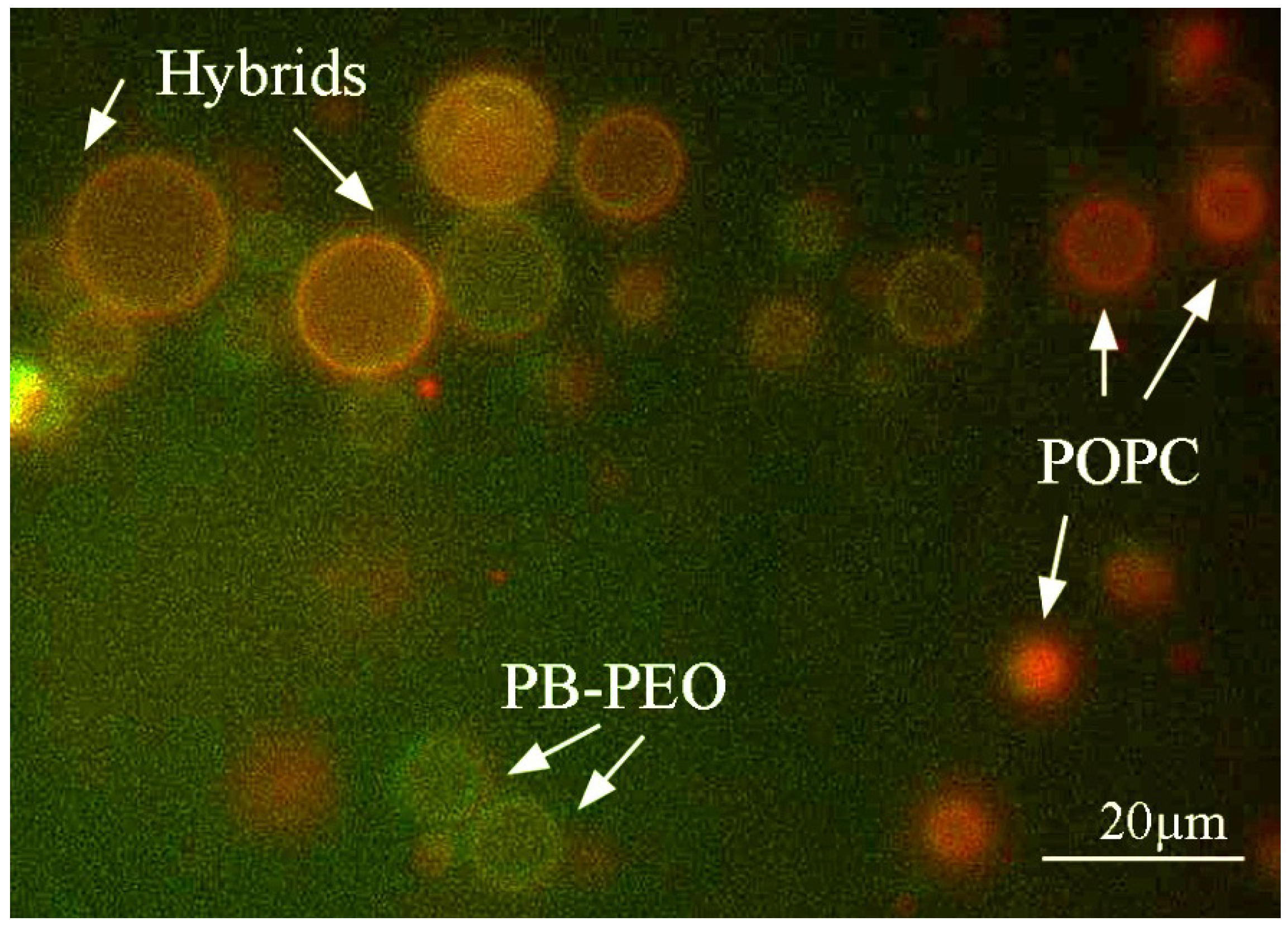
A second difference concerns the preparation method, which may have a significant effect on the formation of hybrids as well [
28]. Indeed, even though application of electroformation to our PB
22-PEO
14:POPC (50:50) films did show formation of hybrid GUVs, not all GUVs were found to be hybrids (
Figure 3). These results thus contrast the observation on the nanosized vesicles, as analyzed by flow cytometry (
Figure 2). This then most likely results from differences in the preparation technique (film rehydration/extrusion
vs. electroformation). We suspect that the multiple extrusion steps allow more thorough mixing of the amphiphiles during vesicles preparation, as compared to electroformation, which may reflect a non-equilibrium state. In nanomedicine and other applications, a high degree of structural and compositional homogeneity is crucial for optimal and safe performance of vesicles. In this aspect, the film rehydration/extrusion could achieve a more homogeneous self-assembling of hybrid lipid/polymer vesicles, as compared to electroformation.
Figure 3.
Hybrid giant unilamellar vesicles (GUVs) of PB-PEO/POPC (50:50) prepared by electroformation observed using fluorescence microscopy. Hybrid GUVs only form part of the GUV population. The green color indicates GUVs formed from PB-PEO, while red indicates POPC. Yellow indicates the presence of both, i.e., hybrid vesicles.
Figure 3.
Hybrid giant unilamellar vesicles (GUVs) of PB-PEO/POPC (50:50) prepared by electroformation observed using fluorescence microscopy. Hybrid GUVs only form part of the GUV population. The green color indicates GUVs formed from PB-PEO, while red indicates POPC. Yellow indicates the presence of both, i.e., hybrid vesicles.
Importantly, our results show that both nanometer-sized and micrometer-sized hybrid vesicles can be formed when employing low molecular weight PB-PEO polymers that more closely match the dimensions of phospholipids.
We then performed a series of experiments to investigate the properties of the hybrid vesicles, as compared to vesicles formed from the pure amphiphiles. DLS was performed to determine the hydrodynamic size distribution of the hybrid vesicles (prepared by film rehydration and extrusion through 200 nm polycarbonate membranes). All extruded vesicles, both hybrids and “pure” vesicles, show a unimodal size distribution with polydispersity indices (PDIs) below 0.3 (
Figure 4). The hybrid vesicles exhibit no significant change in hydrodynamic diameter over 12 days (data not shown), suggesting the excellent colloidal stability of the phospholipid/polymer hybrid vesicles over the period. Note that the extruded PB-PEO polymersomes (
z-average = 196 nm) are slightly larger than extruded POPC liposomes (
z-average = 180 nm), similar to the trend observed for PDMS-
b-PMOXA polymersomes and DMPC liposomes [
19]. This effect may result from the fact that preparation of the PB-PEO/POPC hybrids (and the pure vesicles) involves an extrusion step. For the same applied extrusion pressure, extruded vesicles with higher membrane strength (polymersomes) are expected to have larger vesicle sizes than vesicles with lower lysis tension (
i.e., liposomes) [
29]. Interestingly, hybrid vesicles of PB-PEO/POPC seem to be smaller (
z-average = 66–116 nm) than both pure liposomes (180 nm) and polymersomes (196 nm), with 50:50 hybrids being the smallest (66 nm) (
Figure 4). This size trend is deviates from what was observed previously and this could point to differences in self-assembly behavior at these compositions. We are currently investigating this phenomenon in more detail.
Figure 4.
Intensity-weighted hydrodynamic diameter of extruded vesicles prepared from POPC, PB-PEO/POPC (75:25), PB-PEO/POPC (50:50), PB-PEO/POPC (25:75), and PB-PEO, as measured byDLS. All vesicles show a single unimodal population.
Figure 4.
Intensity-weighted hydrodynamic diameter of extruded vesicles prepared from POPC, PB-PEO/POPC (75:25), PB-PEO/POPC (50:50), PB-PEO/POPC (25:75), and PB-PEO, as measured byDLS. All vesicles show a single unimodal population.
Encapsulation Efficiency and Membrane Permeability
We extended our investigations to hybrid vesicles in which a model compound was encapsulated to gauge their behavior as drug-delivery vehicles. We therefore encapsulated the hydrophilic dye carboxyfluorescein (CF) and determined its encapsulation efficiency. It was found that hybrid vesicles with 25, 50, and 75 mol % POPC displayed a decreased CF encapsulation efficiency (1.8%, 0.4%, and 5%, respectively), with 50:50 hybrids being the lowest (
Figure 5). We hypothesize that this observation is related to the surface area-to-volume ratio, which follows a similar trend if the radii obtained for the (hybrid) vesicles by DLS are used as the input for the calculation of the volumes (Figure S2). A second contribution to the observed phenomenon may come from the excluded-volume effect of the polyethylene glycol (PEG)-block of the polymers, as observed for PEG-ylated liposomes [
30].
Figure 5.
The effect of PB-PEO molar ratio on the encapsulation efficiency of the vesicles. The hydrophilic dye carboxyfluorescein (CF) was used as model compound to calculate the encapsulation efficiency (% encapsulation) of vesicles. The encapsulation efficiency was calculated via 100 × Ft/F0, where Ft is the encapsulated CF concentration (as calculated by CF fluorescence after 0.5% Triton X-100 addition) and F0 is the initial CF concentration of the stock solution.
Figure 5.
The effect of PB-PEO molar ratio on the encapsulation efficiency of the vesicles. The hydrophilic dye carboxyfluorescein (CF) was used as model compound to calculate the encapsulation efficiency (% encapsulation) of vesicles. The encapsulation efficiency was calculated via 100 × Ft/F0, where Ft is the encapsulated CF concentration (as calculated by CF fluorescence after 0.5% Triton X-100 addition) and F0 is the initial CF concentration of the stock solution.
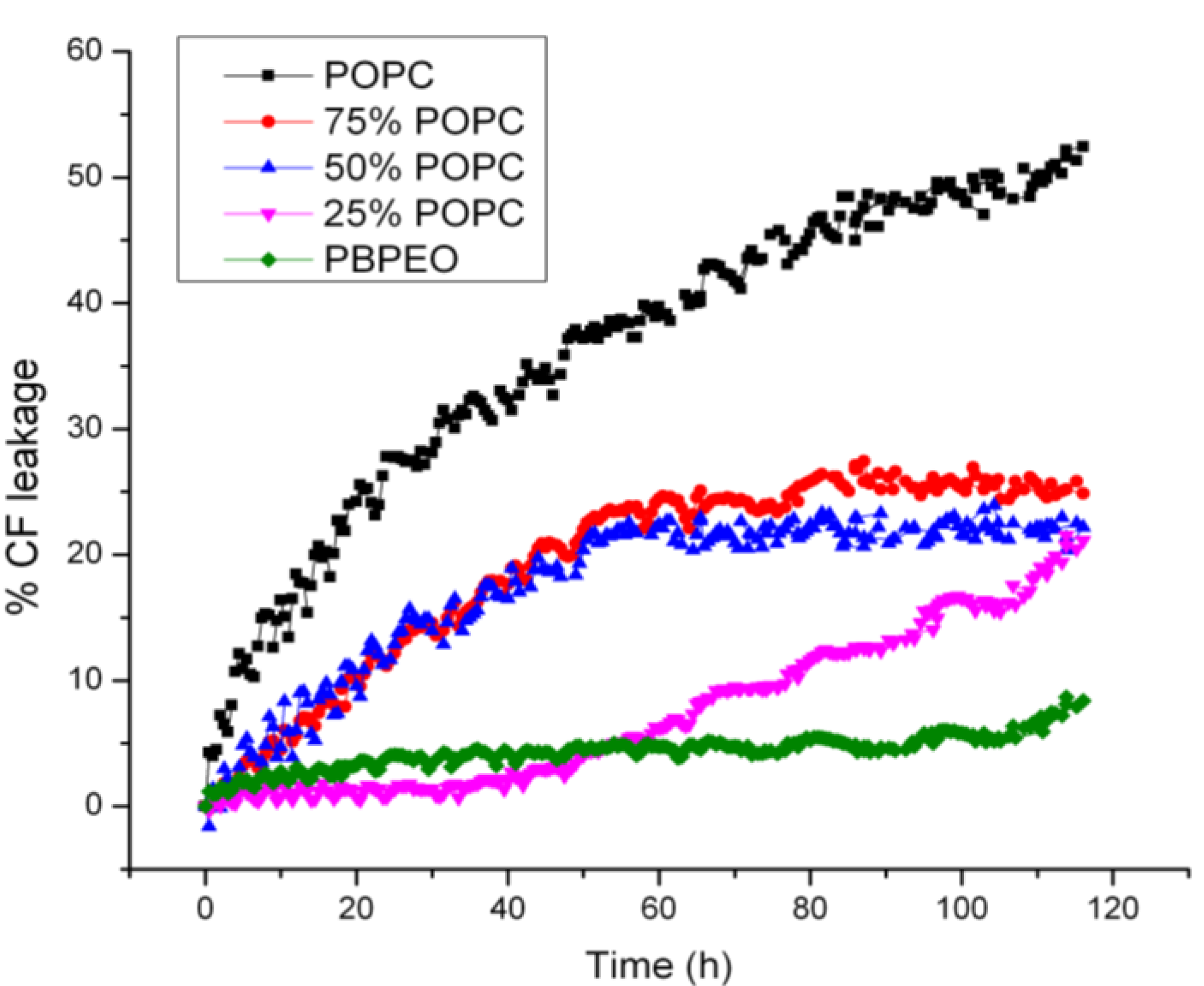
To assess the membrane permeability and content release from the hybrid vesicles, CF release from vesicles in PBS at room temperature was monitored as a function of time, which also provided a means for probing the stability of the hybrid vesicles. It was found that the permeability of the hybrid vesicles depended on the mole fraction of the respective amphiphiles (
Figure 6). A high PB-PEO content significantly delayed CF release, as compared to pure liposomes, showing 50% compound release after
ca. 4 days incubation. Interestingly, a relatively low PB-PEO content of 25% already gave rise to a significantly reduced delay, reaching 25% compound release after 4 days incubation time. At 50% PB-PEO content a similar release profile was observed, while the profile at 75% was clearly different, even though eventual release was similar to that observed at 25% and 50% PB-PEO molar content. Pure PB-PEO polymersomes, as expected, were highly stable, with content release remaining below 10%.
Figure 6.
CF release from vesicles over 120 h at room temperature. 50 mM CF was encapsulated in the vesicles (i.e., at self-quenching concentration). As a result, fluorescence intensity will increase when CF is released from the vesicles in the surrounding aqueous buffer. CF was excited at 480 nm and fluorescence emission at 520 nm was measured. The percentage of CF release over time is presented as 100 × (F − F0)/(FT− F0), where F0 is the initial fluorescence of CF, F is the fluorescence of CF at time interval t and FT is the fluorescence intensity after complete release of CF after adding 0.5% Triton X-100.
Figure 6.
CF release from vesicles over 120 h at room temperature. 50 mM CF was encapsulated in the vesicles (i.e., at self-quenching concentration). As a result, fluorescence intensity will increase when CF is released from the vesicles in the surrounding aqueous buffer. CF was excited at 480 nm and fluorescence emission at 520 nm was measured. The percentage of CF release over time is presented as 100 × (F − F0)/(FT− F0), where F0 is the initial fluorescence of CF, F is the fluorescence of CF at time interval t and FT is the fluorescence intensity after complete release of CF after adding 0.5% Triton X-100.
The effect of PB-PEO on vesicle permeability is reminiscent of the effect that PEG-ylation exerts on the self-assembly and permeability of liposomes [
30,
31,
32,
33]. It was observed that addition of increasing concentrations of DSPE-PEG (DSPE: distearoylphosphatidylethanolamine;
MW(PEG) > 2000) decreases the CF permeability of the PEG-ylated liposomes [
32]. The PEG-block of the PB-PEO polymer, when introduced into the lipid membrane, could have a similar effect. As suggested by our CF leakage assay, vesicles with higher PB-PEO concentration likewise show enhanced stability and a concurrent decreased permeability. It seems, however, somewhat less probable that PB-PEO exerts the same effect on the self-assembly and permeability properties of POPC. Furthermore, for liposomes, addition of PEG-ylated lipids (>10 mol %) leads to micelle formation [
34,
35]. In our PB-PEO/POPC system, addition of 25%, 50% or 75% of PB-PEO did not result in micelle formation. In this regard, the simple mixing of diblock copolymers such as PB-PEO into liposomes could form an alternative method for sterically stabilized liposomal drug carriers, without presenting a similar disruptive effect on the vesicle architecture at higher polymer concentrations.
Considering nanomedicine applications, the hybrid vesicles presented here display several interesting characteristics, such as a decreased, and to some extent tunable, permeability. The observation that encapsulation efficiencies vary considerably with the exact fractions of amphiphiles used should be taken in consideration when formulating hybrid vesicles for
in vivo applications. Finally, the current experiments have only involved hydrophilic compounds. As approximately one-third of newly discovered drugs are hydrophobic [
36], it would be interesting to see to what extent the presented hybrid vesicles perform when loading such drugs in the hydrophobic region of the membrane, in comparison to conventional liposomes.