Influence of the Hydrophobicity of Polyelectrolytes on Polyelectrolyte Complex Formation and Complex Particle Structure and Shape
Abstract
: Polyelectrolyte complexes (PECs) were prepared by structural uniform and strongly charged cationic and anionic modified alternating maleic anhydride copolymers. The hydrophobicity of the polyelectrolytes was changed by the comonomers (ethylene, isobutylene and styrene). Additionally, the n−/n+ ratio of the molar charges of the polyelectrolytes and the procedure of formation were varied. The colloidal stability of the systems and the size, shape, and structure of the PEC particles were investigated by turbidimetry, dynamic light scattering (DLS) and atomic force microscopy (AFM). Dynamic light scattering indicates that beside large PEC particle aggregates distinct smaller particles were formed by the copolymers which have the highest hydrophobicity (styrene). These findings could be proved by AFM. Fractal dimension (D), root mean square (RMS) roughness and the surface profiles of the PEC particles adsorbed on mica allow the following conclusions: the higher the hydrophobicity of the polyelectrolytes, the broader is the particle size distribution and the minor is the swelling of the PEC particles. Hence, the most compact particles are formed with the very hydrophobic copolymer.1. Introduction
In the last decades, nanoparticles have become more important due to their potential applications in many areas of industry, biotechnology and medicine.
Special types of such particles are the polyelectrolyte complex (PECs), which are formed by strong electrostatic interactions between oppositely charged polyelectrolytes (PELs) in aqueous solution. Extensive studies have been performed by several authors on the different types of PECs that can be formed: insoluble and amorphous PECs, soluble PECs and stable colloidal dispersions. The type of nanoparticle that a PEC belongs to depends on many different parameters such as pH, ionic strength, the molar ratio of the mixed anionic to cationic charges of the PELs (n−/n+) and on the polymer concentration CP in the solution. Besides the solution properties structural properties of the PELs are also important, for example, the chain length of the PELs (molar mass) and the type and distribution of the functional groups along the polymer chain length. The current knowledge of PECs has been reviewed by different authors [1-4].
Recent studies deal with size, structure and stoichiometry of charges of the polyelectrolyte complexes. Burkhardt et al. [5] found a core-shell-structure of the particles and a change of the number of the molecules of the diblockcopolymer inside the complex as a function of the pH for complexes formed by poly(isobutene)-block-poly(methacrylic acid)-micelles with quarternized poly(vinyl-pyridine).
Pergushov et al. [6] investigated polyelectrolyte complexes from star-shaped polyacrylic acid and quarternized poly(4-vinylpyridine). For the homogeneous aqueous mixtures obtained from different conditions, two types of complexes were found. These complexes are considerably different in their particle size. The small-sized species (big fraction) was interpreted as water-soluble complexes and the larger-sized species (smaller fraction) was interpreted as complex aggregates.
Mihai et al. [7] investigated, amongst other factors, the size and shape of polyelectrolyte complexes of random copolymers of sodium 2-acrylamido-2-methylpropanesulfonate with either t-butyl acrylamide or methyl methacrylate with an ionene-type polycation by dynamic light scattering and AFM. It was found, that the mean particle sizes of the complex nanoparticles, measured by AFM, were close but always lower than those measured by DLS.
Sæther et al. [8] also studied the formation of PECs by dynamic light scattering and atomic force microscopy. They found that the particle size of the PECs of alginate and chitosan depends strongly of the speed and diameter of the dispersing element of the homogenizer. AFM images show variable shapes and forms, such as spherical particles, linear chains and aggregating flocs as a function of the mixing ratio.
Our work is focused on the stability of PECs as a function of the formation procedure and of the hydrophobicity of the used polyelectrolytes, which is connected with the size, shape and structure of the PEC particles formed. The PELs used are strong PELs based on alternating maleic anhydride copolymers, which were modified to be cationic as well as anionic. Because of their uniform structure, the distance between charged groups along the polymer chain is nearly the same for cationic and anionic polymers. Normally, commercially available anionic polyelectrolytes used for complex formation carry weak charged functional groups excepting polystyrenesulfonate (PSS). Apart from these PELs, workers synthesize themselves the strongly charged anionic polyelectrolytes. For instance, the research group of Dragan studied intensively the complex formation with self synthesized poly(sodium 2-acrylamido-2-methylpropanesulfonate) (NaPAMPS) [9-11]. In our experience, complex formation with polystyrenesulfonate (PSS) often leads to colloidal unstable complex dispersions. This is because of the relatively strong hydrophobicity of this polyelectrolyte. In the present work we prepared PELs with different hydrophobicity of the comonomers like ethylene, isobutylene and styrene. Our goal was to study the colloidal stability of the PECs systematically as a function of the hydrophobicity.
The colloidal stability was followed by means of common turbidity measurements. Using atomic force microscopy, we got information about the structure, size and shape of the PEC particles adsorbed on mica and compared it with results from light scattering.
In a previous paper [12], PECs were characterized by static light scattering and dynamic light scattering at scattering angles of θ = 15°–145° and θ = 30°–100°, respectively. It was found that all PECs seems to be of spherical shape with different internal structure. However, some of these PECs have a very high polydispersity, which complicated the exact determination of the internal structure of the PEC particles with the used methods of characterization.
2. Experimental
2.1. Polyelectrolytes
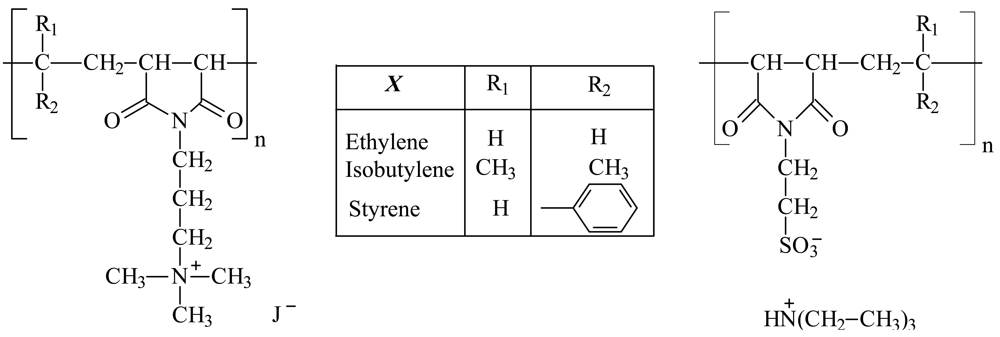
The PELs, polycations (PC) as well as polyanions (PA), used for polyelectrolyte complex formation were synthesized by modification of the alternating maleic anhydride copolymers with different comonomers. For synthesis details see our previous paper [12]. The structures of the used polyelectrolytes are shown in Figure 1. The molecular weight of poly(ethene-alt-maleic anhydride) (Aldrich, Germany) was Mw = 125,000 g/mol with Mw/Mn = 3.12 determined by gel permeation chromatography (GPC). Poly(isobutene-alt-maleic anhydride) (Aldrich, Germany) with Mw = 205,000 g/mol and Mw/Mn = 2.7 was used only for polyanion synthesis. For polycation synthesis poly(isobutene-alt-maleic anhydride) (Aldrich, Germany) with Mw = 76,000 g/mol and Mw/Mn = 3.8 was used. Poly(styrene-alt-maleic anhydride) was obtained from Leuna-Werke AG (Germany) and had a molecular weight of Mw = 300,000 g/mol with Mw/Mn = 2.33.
Molecular weights of the anhydride copolymers were determined by GPC. A mixture of dimethylacetamid (DMAC)/LiCl/water was used as eluent. Poly(propene-alt-maleic anhydride) was used as standard for poly(ethene-alt-maleic anhydride) and poly(styrene-alt-maleic anhydride). Poly(2-vinyl pyridine) was used as standard for poly(isobutene-alt-maleic anhydride).
By means of PEL titration we determined the degree of conversion from anhydride to polyelectrolyte. It was found that the conversion degrees were nearly 100% for all polymers, polycations as well as polyanions. That means that each monomer unit carries one charge and hence the distance between the charges is similar for polycations and polyanions because of its uniform alternating structure.
2.2. Polyelectrolyte Complex Formation
The PECs were prepared by combination of equal amounts (50 mL) of aqueous solutions of the oppositely charged PELs. These solutions were obtained by dilution of definite amounts of stock PEL solutions in Millipore water. The stock PEL solutions had concentrations of CEth−PC = 3.52 g/L, CEth−PA = 3.34 g/L, CIsob−PC = 3.8 g/L, CIsob−PA = 3.62 g/L, CStvr−PC = 4.28 g/L and CStvr−PA = 4.1 g/L, which correspond to a charge concentration of 10−2 mol/L for all PEL stock solutions determined by PEL titration. The PEL titration was carried out by means of a PCD 02 particle charge detector (Mütek GmbH, Germany).
The initial concentration of polycation solution ( ) was kept constant for all prepared PECs, while the amount of anionic charges corresponds to the desired n−/n+ (ratio of charges). n−/n+ is the calculated ratio of charges. Under continuous stirring, one of the PEL solutions was added to the starting PEL solution with a flow rate of 0.2 L/h controlled by a dosing pump. PC solutions (procedure a) as well as PA solutions (procedure b) were used as starting solution, detailed information being given in Table 1. The names of the PECs in Table 1 are as follows: comonomer of PC/comonomer of PA preparation procedure.
It should be mentioned that is the concentration of the polycation solution at the beginning of complex formation ( ). At the end of the complex formation the volume of the formed complex dispersion is twice the volume of the separate initial solutions. The polymer concentration CP of the whole PEC dispersion is related to the end state of the complex formation. Hence the value of CP can be lower than in spite of that the amount of polycation as well as the amount of polyanion is included in CP.
After formation, the complex dispersions were still stirred for 10 min, and characterized after two and 24 h of storage.
2.3. Complex Characterization
2.3.1. Turbidimetry
The Nephelometer 2100AN IS of the HACH Company (Germany) was used to determine the turbidity T of the PECs in order to estimate the stability of the PEC dispersions.
The method is based on the determination of the scattered light intensity measured at a scattering angle of 90°. The scattered light intensity depends on the number of particles, particle size and shape, the wavelength of the incident light beam and whose angle related to the sensor. The device is equipped with an IR-LED (λ = 860 nm) as light source. As the wavelength is out of the visible region, possible coloring of the samples do not perturb the results.
2.3.2. Dynamic Light Scattering
Dynamic light scattering measurements were carried out by using a Zetasizer Nano S (ZEN 1600) of Malvern Instruments, UK. The instrument was equipped with a monochromatic coherent 4 mW Helium Neon laser (λ = 633 nm) as light source and the so-called NIBS®-technology (Non-Invasive Back-Scattering; patent from ALV GmbH, Germany). The light scattered by particles is recorded at an angle of 173°, so the effects of multiple scattering and possible contaminants are greatly reduced. As a result of the very sensitive detector (Avalanche Photodiode), the embedded moveable lens and the automatic laser attenuation (transmission of 100% to 0.0003%), we are able to determine the particle size in a wide size range (0.6 nm to 6 μm) and in a very expanded concentration range. This means that it is not necessary to dilute the PEC dispersions and so we avoid influencing of the PEC dispersion by dilution.
All measurements were performed in disposable cuvettes. Ten autocorrelation functions (ACFs) of 20 s data collection time per scan were combined to a mean ACF and evaluated by the DTS (Dispersion Technology Software) 4.00 appendant to Zetasizer Nano S. This Software includes cumulant analysis and the multimodal size distribution algorithm NNLS.
The analysis of the ACF gives the mean diffusion coefficient DT. From cumulant analysis it is possible to get the z-average hydrodynamic diameter dh, z av of PEC particles, which was calculated by the Stokes-Einstein equation:
Additionally, we used the distribution analysis in order to get some more information about the size distribution of PEC particles in the investigated PEC dispersions.
2.3.3. Atomic Force Microscopy
Except images of PECs composed of PELs with ethylene comonomers and performed in procedure a, all AFM-images were taken by the NanoScope IV- D 3100 from Digital Instruments Veeco (USA) equipped with a silicon cantilever Tap300 from Budget Sensors (Bulgaria). In the case of PECs composed of PELs with ethylene comonomers the AFM images were taken by the MFP-3D AFM from Asylum Research, Santa Barbara (USA) at higher sensitivity of resolution (4,096 × 4,096 pixels) than for the other images (512 × 512 pixels).
All measurements were carried out in tapping mode. The PEC particles were adsorbed onto a freshly cleaved mica surface by positioning the mica pieces vertically in the PEC dispersion for 2 h. Thereafter the mica pieces were rinsed by Millipore water 4 times and dried on air.
With the software WSxM 4.0 Develop 9.3 by Nanotec Electronica S. L. the AFM images were analyzed [13,14].
3. Results and Discussion
3.1. Influence of Polyelectrolyte Hydrophobicity on PEC Dispersion Stability
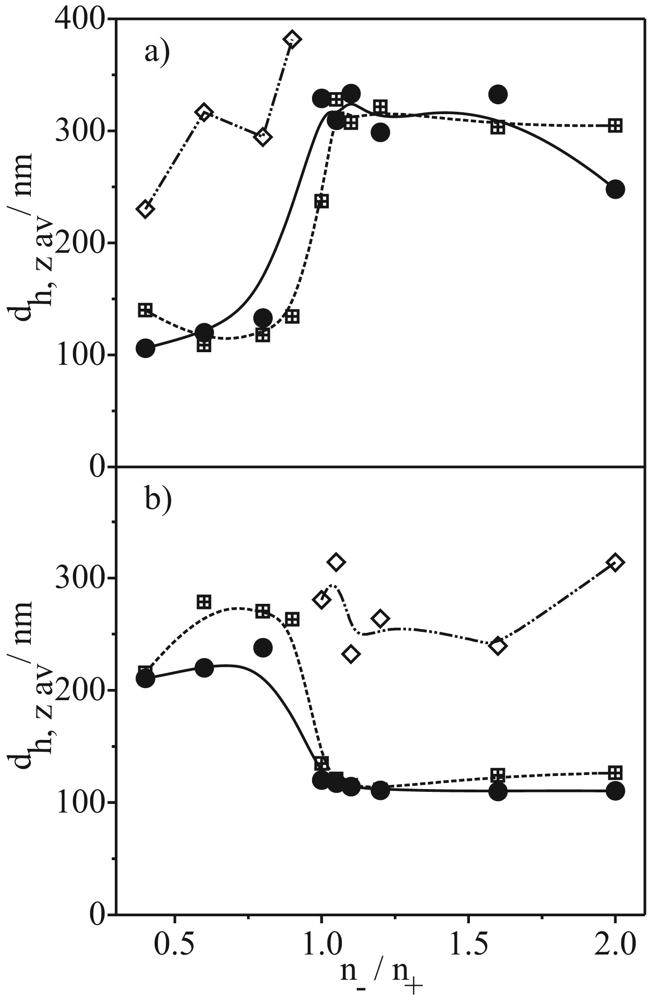
A simple way to get information about the stability of dispersions is to determine the turbidity over a certain time. The turbidity contains information about size, concentration and the shape of particles. Figure 2 shows the turbidity of the formed PEC dispersions in dependence on the n−/n+ ratio, the preparation procedure and on the time. If no difference between turbidity measured after 2 and 24 h is observed, the PEC dispersion is colloidal stable. Around the isoelectric point ((n−/n+)IP) of the systems no or only a small charge excess is available to stabilize the PEC particles. Hence the turbidity decreases in dependence on time.
As already found in former studies [15,16] the turbidity of a PEC dispersion depends also on the fact whether the isoelectric point of the system ((n−/n+)IP = 0.95 ± 0.05 for all systems) is exceeded during the PEC formation or not. If (n−/n+)IP is exceeded then the turbidity of these dispersions is higher, as for the other PECs of the same system. This concerns the PECs with n−/n+ > (n−/n+)IP in the case of preparation procedure a and in procedure b PECs with n−/n+ < (n−/n+)IP. Only the PECs formed of PC and PA with styrene as comonomer in procedure b are exceptions to this finding.
Another result of these studies is that the hydrophobicity of the PELs used for PEC formation have a distinct effect on the colloidal stability of PEC dispersions at least for PECs where (n−/n+)IP is exceeded during formation. Changing the comonomers changed the hydrophobicity of the PELs. The hydrophobicity increases in the order of ethene, isobutene and styrene. One day after PEC formation we observe lower turbidity for PECs where (n−/n+)IP is exceeded during PEC formation (dotted curves in Figure 2) compared to the turbidity only 2 h after PEC formation. This means that the higher the hydrophobicity of the PELs the lower the dispersion stability. The Styr/Styr PECs are even settled down completely after 5 h. This occurs in spite of the same charge excess if we compare PECs with the same n−/n+ ratio but different comonomers of the PELs.
The minimal differences in turbidity of the PECs when (n−/n+)IP is not exceeded during formation and which are stable in turbidity over at least one day (and longer, but not shown here) possibly show also a slight influence of hydrophobicity. If we compare the PECs of the same preparation procedure and n−/n+ ratio as a function of the comonomere of the PC and PA, respectively, we find a minimal increase of the turbidity with higher hydrophobicity of the PELs in most cases.
3.2. Average Hydrodynamic Particle Size and Particle Size Distribution
In order to get more information, we determined the average hydrodynamic particle size dh, z av and the particle size distribution. Figure 3 shows the average hydrodynamic particle size dh, z av in dependence on the n−/n+ ratio for the stable PECs prepared in procedure a and b. If we compare the values of turbidity (Figure 2) and the dh, z av values, we observe a similar behavior in these curves. In the case of the Eth/Eth and Isob/Isob PECs we find low turbidity when the average particle size is low and vice versa. In these cases one can assume that the turbidity values primarily come from the particle size. For the Styr/Styr PECs, the relations are not so clear. As mentioned above, the turbidity of these PECs is slightly higher than for the PECs with the other comonomers in the PELs so that we would expect a slightly higher average particle size. However, we observe much higher values for dh, z av than expected, which could be ascribed to the aggregation of particles.
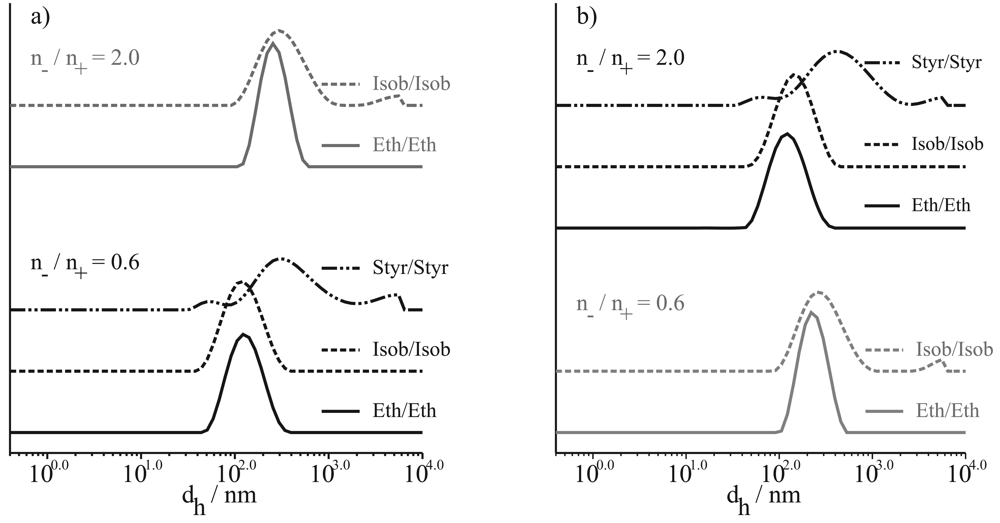
This fact can also be supported by the result shown in Figure 4, in which the particle size distributions of stable PECs with selected n−/n+ ratios are shown. Figure 4(a) demonstrates the size distributions of PECs prepared by procedure a, and Figure 4(b) the size distributions of PECs prepared by procedure b. Independent of the n−/n+ ratio and the preparation procedure the broadness of particle size distribution increases with increasing hydrophobicity of the comonomers of PELs used for PEC preparation.
It is important to note that we observed a little peak at very small particle sizes in the most cases of the broad distributions of the Styr/Styr PECs. As we could not decide between an artifact or a particle, we additionally investigated the PEC particles by means of atomic force microscopy.
3.3. Atomic Force Microscopy
Figure 5 shows the AFM images of PECs adsorbed on mica prepared in procedure a (a) and b (b), respectively. All PECs had an n−/n+ ratio of 0.6 and their dispersions were stable over a longer time. The original image size is 10 μm × 10 μm. For a better recognition of particles, 5 μm × 5 μm zooms are shown.
It is necessary to show selected profiles additionally because from the 2D images one could assume that the Eth/Eth PEC form very large particles in contrast to the others. But if the z direction of the profiles is considered another sight is revealed. The extent of the adsorbed PEC particles and aggregates, respectively, is higher for Styr/Styr PEC than for Eth/Eth PEC. Isob/Isob PEC is in the middle of the two other PECs. That means that the higher the hydrophobicity of the comonomers of PELs used in PEC formation the bigger the particles and aggregates, respectively, in average. This should be one of the reasons for increasing the RMS roughness (Root Mean Square, RMS), summarized in Table 2, with increasing hydrophobicity. Additionally, we observed many small particles with increased hydrophobicity of the PELs, a fact that is already indicated by the dynamic light scattering. These small particles should be a second reason for higher RMS roughness with increasing hydrophobicity.
RMS roughness presented here is based on calculating the height distribution of an image [14,17].
Table 2 shows the fractal dimensions D of PEC particles and aggregates, respectively. The fractal dimensions were calculated applying the slit island method that is included in the WSxM software. In this method, primarily suggested by Mandelbrot [18], the surface of mica coated with the PECs were “flooded” with an imaginary fluid up to a given level. In analog, this can be regarded as a section plane on a given height parallel to the surface. Inside this section plane are the so-called islands—sections of PEC particles. Then the area A and the perimeter P are measured for a set of islands using the same ruler length. These values were plotted in a double logarithmic diagram and fitted by linear regression. The fractal dimension D was then obtained from the slope of the curve with following equation [19]:
In the WSxM software the section plane is automatically set on 50% of the maximum height of the surface topography. It pointed out that this value is quite well suited to our samples, although, mainly in the case of Styr/Styr PEC, the small particles are not included in determination of the fractal dimension. The maximum height values hmax and the height of the section planes hmin used in our samples are also summarized in Table 2. In summary, it can be said that the shape of the larger structures adsorbed onto mica differ clearly from a circle (D = 1) with higher hydrophobicity of the PELs. This result supports the assumption that larger PEC particles formed by the hydrophilic ethylene copolymers are more swollen than the PEC particles formed by the hydrophobic styrene copolymers. In this case, the small strong primary particles aggregate to larger raspberry-like particles.
From the higher resolved AFM images of the Eth/Eth PEC (Figure 5(a)) we can conclude that the PEC particles are of core-shell-form even though the circumstances in dispersion are different from those on a surface and after drying. We interpret this from the fact that, around the larger particles (white areas of about 8 to 12 nm height), we observe gray areas with a height of about 2 nm and then, around this layer, a dark area with a height of nearly zero (Figure 5(a)). A large part of the mica surface is coated with the “gray substance” which should be free polyelectrolyte or soluble primary PEC but in any case accounts with charge. Then, the dark areas should arise from the electrostatic repulsion of the charges between the free polyelectrolyte and primary complex, respectively, and the charge of polyelectrolytes inside the core of PEC particles.
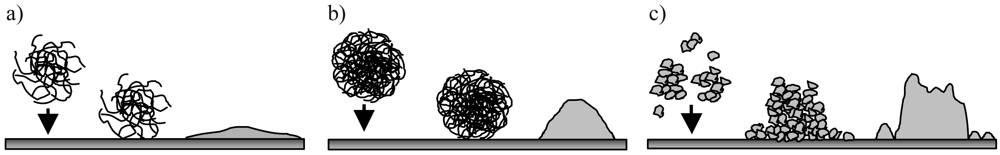
In Figure 6, the schematic construction of the situation during adsorption of the PECs onto the mica surface and after drying is depicted. Each part of Figure 6(a–c) show on the left the particle in solution, in the middle the particle adsorbed onto the surface of mica and on the right the profile detected with AFM. The Eth/Eth PEC particles shown in Figure 6a are symmetrical and very flat. This let us assume that the particles are spherical in solution and more highly swollen than the Isob/Isob and Styr/Styr PEC particles, which are already deduced from the results in [12]. Particles of Isob/Isob PEC are also spherical in x-y-direction (2D). But in the z-direction the particles are distinctly higher than the Eth/Eth PEC particles, which indicate a more compact structure in solution.
In the case of Styr/Styr PEC we found larger asymmetric particles, which can be interpreted as aggregates of the small primary particles as mentioned above. The size range of these primary particles is of the same range as the lowest peak (DLS) in Figure 4a for Styr/Styr with n−/n+ = 0.6 (30 nm–60 nm). Let us assume that these small PEC particles are less swollen than, for instance, the Eth/Eth PEC particles, because the determination of size with DLS is carried out in dispersion and the AFM images are taken on a dried surface.
4. Summary
The colloidal stability of polyelectrolyte complexes (PEC), formed between oppositely charged polyions based on alternating maleic anhydride copolymers, has been investigated by means of turbidimetry. Additionally, the dynamic light scattering and atomic force microscopy provide information about the size, shape and structure of the PEC particles. The main focus of attention was the dependence of the colloidal stability and the structure of the larger PEC particles on the hydrophobicity of the polyelectrolytes used for complex formation. The hydrophobicity was varied by the comonomers in the copolymer backbone (ethylene < isobutylene < styrene).
Furthermore, the n−/n+ ratio and the formation procedure at which in certain cases the isoelectric point of the system is exceeded during complex formation and sometimes not, were varied. It was found that in dependence on the hydrophobicity of the polyelectrolytes the PECs differ clearly in their colloidal stability. Higher hydrophobicity gives lower colloidal stability. In the case of Styr/Styr the PECs are very unstable if the isoelectric point of the system is exceeded during PEC formation despite considerable charge excess.
Additionally, we observed a higher polydispersity of the PEC particles with higher hydrophobicity of the polyelectrolytes in the PEC dispersion. Styr/Styr PEC dispersion shows a fraction of small particles (about 50 nm), which is indicated by dynamic light scattering and detected by AFM. These small particles form random shaped raspberry-like aggregates (larger PEC particles) showed by fractal dimension in contrast to the Eth/Eth and Isob/Isob PEC particles, which are nearly spherical. From these results we can conclude that, the higher the hydrophobicity of the polyelectrolytes, the less swollen PEC produced particles are. These particles have a more compact structure compared to the PEC particles formed of the less hydrophobic polyelectrolytes.
In the case of Eth/Eth PEC particles from the AFM images measured with higher resolution we can conclude that the structure of these particles corresponds to the core-shell-model. The former studies [12] revealed a spherical shape with a radial density gradient in the whole range of the mixing ratios.


| PEC system | * | * | # | n−/ n+ | CP § | |
|---|---|---|---|---|---|---|
| g/mol | g/mol | mmol/L (g/L) | mmol/L (g/L) | |||
| Eth/Eth | a | 331,000 | 349,000 | 1 (0.352) | 0.4–2.0 | 0.7–1.5 |
| b | (0.22–0.5) | |||||
| Isob/Isob | a | 188,000 | 388,000 | 1 (0.38) | 0.4–2.0 | 0.7–1.5 |
| b | (0.25–0.55) | |||||
| Styr/Styr | a | 635,000 | 610,000 | 1 (0.428) | 0.4–2.0 | 0.7–1.5 |
| b | (0.27–0.6) | |||||
a. starting solution: PC (polycation); b. starting solution: PA (polyanion);*Molar masses of PC or PA (The molar masses were calculated from the corresponding maleic anhydride copolymer with the assumption that the conversion of the anhydride to the PEL is 100%, which was confirmed by PEL titration;#initial concentration PC;§polymer concentration in the whole PEC dispersion.
| RMS roughness / nm | hmax # / nm | hmin * / nm | D | ||
|---|---|---|---|---|---|
| Eth/Eth | a | 1.41 | 12.26 | 6.13 | 1.02 |
| b | 2.79 | 20.21 | 10.10 | 1.07 | |
| Isob/Isob | a | 3.47 | 46.50 | 23.25 | 1.09 |
| b | 8.15 | 91.55 | 45.77 | 1.03 | |
| Styr/Styr | a | 11.36 | 134.86 | 67.43 | 1.19 |
#It is the highest value of height, which was found on the 10 μm × 10 μm area of the AFM image.*The height of section plane in slit island method.
Acknowledgments
We thank K.-F. Arndt and V. Boyko for the interesting and inspiring discussions about light scattering methods.
References
- Thünemann, A.F.; Müller, M.; Dautzenberg, H.; Joanny, J.-H.; Löwen, H. Polyelectrolyte complexes. Adv. Polym. Sci. 2004, 166, 113–171. [Google Scholar]
- Petrak, K. Polyelectrolytes Science and Technology; Hara, M., Ed.; Marcel Dekker: New York, NY, USA, 1993; Chapter 5; pp. 265–298. [Google Scholar]
- Tsuchida, E. Formation of polyelectrolyte complexes and their structures. J. Macromol. Sci. Pure Appl. Chem. 1994, A31, 1–15. [Google Scholar]
- Philipp, B.; Kötz, J.; Dautzenberg, H.; Dawydoff, W.; Linow, K.-J. Applied Polymer Analysis and Characterization, Part III; Mitchell, J., Ed.; Hanser Verlag: München, Germany, 1990; p. 281. [Google Scholar]
- Burkhardt, M.; Ruppel, M.; Tea, S.; Drechsler, M.; Schweins, R.; Pergushov, D.V.; Gradzielski, M.; Zezin, A.B.; Müller, A.H.E. Water-soluble interpolyelectrolyte complexes of polyisobutylene-block-poly(methacrylic acid) micelles: Formation and properties. Langmuir 2008, 24, 1769–1777. [Google Scholar]
- Pergushov, D.V.; Babin, I.A.; Plamper, F.A.; Zezin, A.B.; Müller, A.H.E. Water-soluble complexes of star-shaped poly(acrylic acid) with quaternized poly(4-vinylpyridine). Langmuir 2008, 24, 6414–6419. [Google Scholar]
- Mihai, M.; Dragan, E.S.; Schwarz, S.; Janke, A. Dependency of particle sizes and colloidal stability of polyelectrolyte complex dispersions on polyanion structure and preparation mode investigated by dynamic light scattering and atomic force microscopy. J. Phys. Chem. B 2007, 111, 8668–8675. [Google Scholar]
- Sæther, H.V.; Holme, H.K.; Maurstad, G.; Smidsrød, O.; Stokke, B.T. Polyelectrolyte complex formation using alginate and chitosan. Carbohyd. Polym. 2008, 74, 813–821. [Google Scholar]
- Dragan, E.S.; Mihai, M.; Schwarz, S. Complex nanoparticles based on chitosan and ionic/nonionic strong polyanions: formation, stability, and application. ACS Appl. Mater. Interfaces 2009, 1, 1231–1240. [Google Scholar]
- Dragan, E.S.; Schwarz, S. Polyelectrolyte complexes VII. Complex nanoparticles based on poly(sodium 2-acrylamido-2-methylpropansulfonate) tailored by the titrant addition rate. J. Polym.Sci. Part A 2004, 42, 5244–5252. [Google Scholar]
- Dragan, E.S.; Mihai, M.; Schwarz, S. Polyelectrolyte complex dispersions with a high colloidal stability controlled by the polyion structure and titrant addition rate. Colloids Surf. A 2006, 290, 213–221. [Google Scholar]
- Boyko, V.; Richter, S.; Mende, M.; Schwarz, S.; Zschoche, S.; Arndt, K.-F. Characterization of polyelectrolyte complexes based on maleic anhydride alternating copolymers by static and dynamic light scattering. Macromol. Chem. Phys. 2007, 208, 710–717. [Google Scholar]
- Software WSxM 4.0 Develop 9.3 by Nanotec Electronica S. L. Available online: http://www.nanotec.es/ (accessed on 23 August 2011).
- Horcas, I.; Fernandez, R.; Gomez-Rodriguez, J.M.; Colchero, J.; Gomez-Herrero, J.; Baro, A.M. WSXM: A software for scanning probe microscopy and a tool for nanotechnology. Rev. Sci. Instrum. 2007, 78, 013705. [Google Scholar]
- Mende, M.; Buchhammer, H.-M.; Schwarz, S.; Petzold, G.; Jaeger, W. The stability of polyelectrolyte complex systems of poly(diallyldimethylammonium chloride) with different polyanions. Macromol. Symp. 2004, 211, 121–133. [Google Scholar]
- Mende, M.; Petzold, G.; Buchhammer, H.-M. Polyelectrolyte complex formation between poly(diallyldimethyl-ammonium chloride) and copolymers of acrylamide and sodium-acrylate. Colloid Polym. Sci. 2002, 280, 342–351. [Google Scholar]
- Thomas, T.R. Rough Surfaces; Imperial Collage Press: London, UK, 1999; pp. 139–143. [Google Scholar]
- Mandelbrot, B.B.; Passoja, D.E.; Paullay, A.J. Fractal character of fracture surfaces of metals. Nature 1984, 308, 721–722. [Google Scholar]
- Milman, V.Y.; Stelmashenko, N.A.; Blumenfeld, R. Fracture surfaces: A critical review of fractal studies and a novel morphological analysis of scanning tunneling microscopy measurements. Prog. Mater. Sci. 1994, 38, 425–474. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mende, M.; Schwarz, S.; Zschoche, S.; Petzold, G.; Janke, A. Influence of the Hydrophobicity of Polyelectrolytes on Polyelectrolyte Complex Formation and Complex Particle Structure and Shape. Polymers 2011, 3, 1363-1376. https://doi.org/10.3390/polym3031363
Mende M, Schwarz S, Zschoche S, Petzold G, Janke A. Influence of the Hydrophobicity of Polyelectrolytes on Polyelectrolyte Complex Formation and Complex Particle Structure and Shape. Polymers. 2011; 3(3):1363-1376. https://doi.org/10.3390/polym3031363
Chicago/Turabian StyleMende, Mandy, Simona Schwarz, Stefan Zschoche, Gudrun Petzold, and Andreas Janke. 2011. "Influence of the Hydrophobicity of Polyelectrolytes on Polyelectrolyte Complex Formation and Complex Particle Structure and Shape" Polymers 3, no. 3: 1363-1376. https://doi.org/10.3390/polym3031363




