1. Introduction
Hepatocellular carcinoma (HCC) is one of the most destructive cancers. At present, sorafenib is the only drug available that prolongs the life of patients with HCC. However, non-specific uptake leads to high toxicity and serious side effects. Sorafenib is a multikinase inhibitor that targets various receptor tyrosine kinases and RAF kinases; hence, it hampers tumor growth and exerts cytostatic effects and thus demonstrates a significant overall survival rate of patients, e.g., with HCC. However, its water immiscibility results in low bioavailability [
1]; thus, a high dosage is required. Doxorubicin is a common chemotherapeutic agent in numerous cancer therapies [
2]. It is an anthracycline antibiotic. Doxorubicin hydrochloride salt is a water-soluble, hygroscopic, crystalline form of the drug, which possesses better bioavailability. Doxorubicin activation on the nucleic acids of dividing cells can occur by intercalation between the base pairs of the DNA strands, thus inhibiting the synthesis of DNA and RNA by impeding the replication and transcription in the cells and producing iron-mediated free radicals that destroy cell membranes, proteins, and DNA. The most disadvantageous side effects of doxorubicin are myelosuppression and cardiotoxicity.
The drawbacks of the use of anticancer agents could be decreased by the application of a nanocarrier that supports the targeted drug delivery and controls the release of effective agents. Polymeric nanoparticulate drug delivery systems have been shown to be a valid approach to sustain drug liberation and to enable a targeting function. There are some existing papers on sorafenib or doxorubicin microencapsulation using PLGA copolymers. Nevertheless, the sorafenib loading in PLGA nanoparticles is generally rather low. E.g., a 1.4% sorafenib loading in PLGA nanoparticles with an oil-in-water single-emulsion solvent evaporation method was achieved in [
3]. Multiblock polymer nanoparticles consisting of (poly(lactic acid)-poly(ethylene glycol)-poly(
l-lysine)-diethylenetriamine pentaacetic acid and the pH-sensitive material poly(
l-histidine)-poly(ethylene glycol)-biotin could encapsulate 2.4% sorafenib [
4]; however, by a nanoprecipitation-dialysis method using a block copolymer of dextran and poly(
d,
l-lactide-
co-glycolide) the realized drug content was substantially higher, with a maximum of 5.3% [
5]. Doxorubicin-loaded PEG-PLGA-Au nanoparticles with a cytostatic drug content of 3.9% were prepared to enable combined treatment based on chemotherapy and heat-therapy by near-infrared radiation in [
6].
By simultaneous delivery of anticancer drugs to tumor cells, a synergistic effect can be realized by an appropriate composition [
7]. In some studies, co-delivery of sorafenib and doxorubicin has already been successfully done. E.g., a nanocomposite composed of doxorubicin containing a polyvinyl alcohol core and a human serum albumin-sorafenib shell was manufactured by a sequential freeze-thaw method followed by ethanol coacervation in [
8]. The drug loading and the encapsulation efficiency of doxorubicin were 3.0% and 82.0%, respectively, in the nanocore; these values for sorafenib were 2.4% and 91%, respectively, in the albumin nanoshell. Lipid-polymer hybrid nanoparticles decorated with the tumor-homing peptide iRGD were prepared in [
9]. The hybrid nanocomposites possessed synergistic cytotoxicity, a pro-apoptotic ability, and improved uptake by HepG2 human hepatocellular carcinoma cells. The blood circulation time and bioavailability and antitumor effects were also significantly increased in HCC xenograft mouse models. Although the drug loading for sorafenib was rather low (3.6%), high doxorubicin content (13.6%) was realized in this work. Very recently, Xiong et al. [
10] entrapped sorafenib adamantine-terminated doxorubicin using poly(ethylene glycol)-β-cyclodextrin. Their reduction-responsive supramolecular nanosystem was manufactured through host-guest interaction between cyclodextrin and adamantine moieties, which then self-assembled into regular spherical nanoparticles that showed an inhibitory effect against HepG2 hepatocellular carcinoma cells.
In our study, PLGA and PEG-PLGA carriers, respectively, were used to entrap doxorubicin and sorafenib together in nanotherapeutics in order to enable the anticancer drugs to exert a synergistic influence. The double emulsion solvent evaporation method was applied for the simultaneous entrapment of the drugs. After the optimization of size and encapsulation efficiency, the drug release profile was investigated in human blood plasma. In vitro cellular studies in HT-29 cancer cells were performed to study the cellular uptake and cytotoxicity of the drug-loaded nanocomposites.
2. Materials and Methods
2.1. Materials
Poly(d,l-lactide-co-glycolide) (PLGA) polymer, Resomer RG 752H (lactide:glycolide: 75:25, inherent viscosity 0.14–0.22 dL/g, Mw = 4000–15,000), and Resomer RG 502H (lactide:glycolide: 50:50, inherent viscosity 0.16–0.24 dL/g, Mw = 7000–17,000 g/mol) were produced by Boehringer Ingelheim (Ingelheim am Rhein, Germany). PEGylated-PLGA (PEG-PLGA) polymer, Resomer, RGP d 5055 (PEG-PLGA) (PEG content: 3–7% (m/m), inherent viscosity: 0.93 dL/g, Mw = 33,500 g/mol) was obtained from Evonik (Essen, Germany). Polyvinyl alcohol (PVA, Mw = 30,000–70,000 g/mol, 87–90% hydrolysed), dichloromethane (DCM), acetone, glacial acetic acid, dimethyl sulfoxide (DMSO), sodium azide, 1-ethyl-3(3-dimethylaminopropyl) carbodiimide (EDC), N-hydroxysuccinimide (NHS), sodium dodecyl sulphate (SDS), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), piroxicam, and RPMI-1640 medium were obtained from Sigma Aldrich (St. Louis, MO, USA). Sorafenib (free base) and doxorubicin HCl were purchased from Active Biochem (Hong Kong, China). Cyanine 5 amine was produced by Lumiprobe GmBH (Hannover, Germany).
2.2. Preparation of Nanocomposites
For the preparation of our dual-agent nanocomposites, the water-in-oil-in-water double emulsion solvent evaporation process was found to be appropriate. Briefly, the inner water phase was composed of 0.2 mL 0.5% (w/v) doxorubicin HCl solution in MilliQ water, which was added to the organic phase that consisted of 20 mg encapsulating polymer (Resomer RG 752H, Resomer RG 502H, or Resomer RGP d5055) dissolved in 1.0 mL DCM combined with 1.0 mg sorafenib dissolved in 0.1 mL acetone. The first emulsification was performed by sonication using a sonicator (Sonics Vibra Cell VCX 130, 130 W, Newtown, CT, USA) at an amplitude of 30% for 30 s. Then, the prepared water-in-oil emulsion was pipetted into the outer water phase that consisted of 1% (w/v) PVA in 5 mL phosphate buffer (pH 8). The water-in-oil-in-water emulsion was formed by another sonication at an amplitude of 50% for 60 s. The organic solvents were evaporated by magnetic stirring for 3 h under atmospheric pressure at room temperature. Nanoparticles were centrifuged by a Hermle Z216 MK microcentrifuge (Schwerte, Germany) at 15,000 rpm for 20 min, washed thrice, and redispersed in MilliQ water or phosphate-buffered saline (PBS, pH 7.4).
2.3. Investigation of Nanocapsules
2.3.1. Morphology and Size Analysis
The morphology of nanocapsules was monitored after centrifuging and redispersing them in distilled water, dropping them onto a grid, and drying them under room temperature. Then, they were examined with an FEI Apreo scanning electron microscope (SEM, Thermofisher, Waltham, MA, USA) at 20 kV.
The size distribution of the obtained nanoparticles was determined by a Zetasizer Nano ZS (Malvern Instruments, Malvern, UK) operated with dynamic light scattering. The particles were characterized by their intensity mean diameter and polydispersity index (PDI).
2.3.2. Nanoparticle Yield and Encapsulation Efficiency
The yield of the nanocomposites was determined by gravimetry after washing and drying of a known volume of nanoparticle suspension. The drug loading and encapsulation efficiency were investigated directly by dissolving 10 mg nanoparticles in 1 mL DMSO, and the solution was diluted to be detectable in the linear calibration range (1–20 mg/L). The absorbance of the solutions was measured spectrophotometrically (PG Instruments T80, Leicestershire, UK) at the absorbance maxima of doxorubicin (480 nm) and sorafenib (270 nm) in DMSO. The encapsulation efficiency of the active agents was calculated as follows:
2.3.3. In Vitro Drug Release Experiment
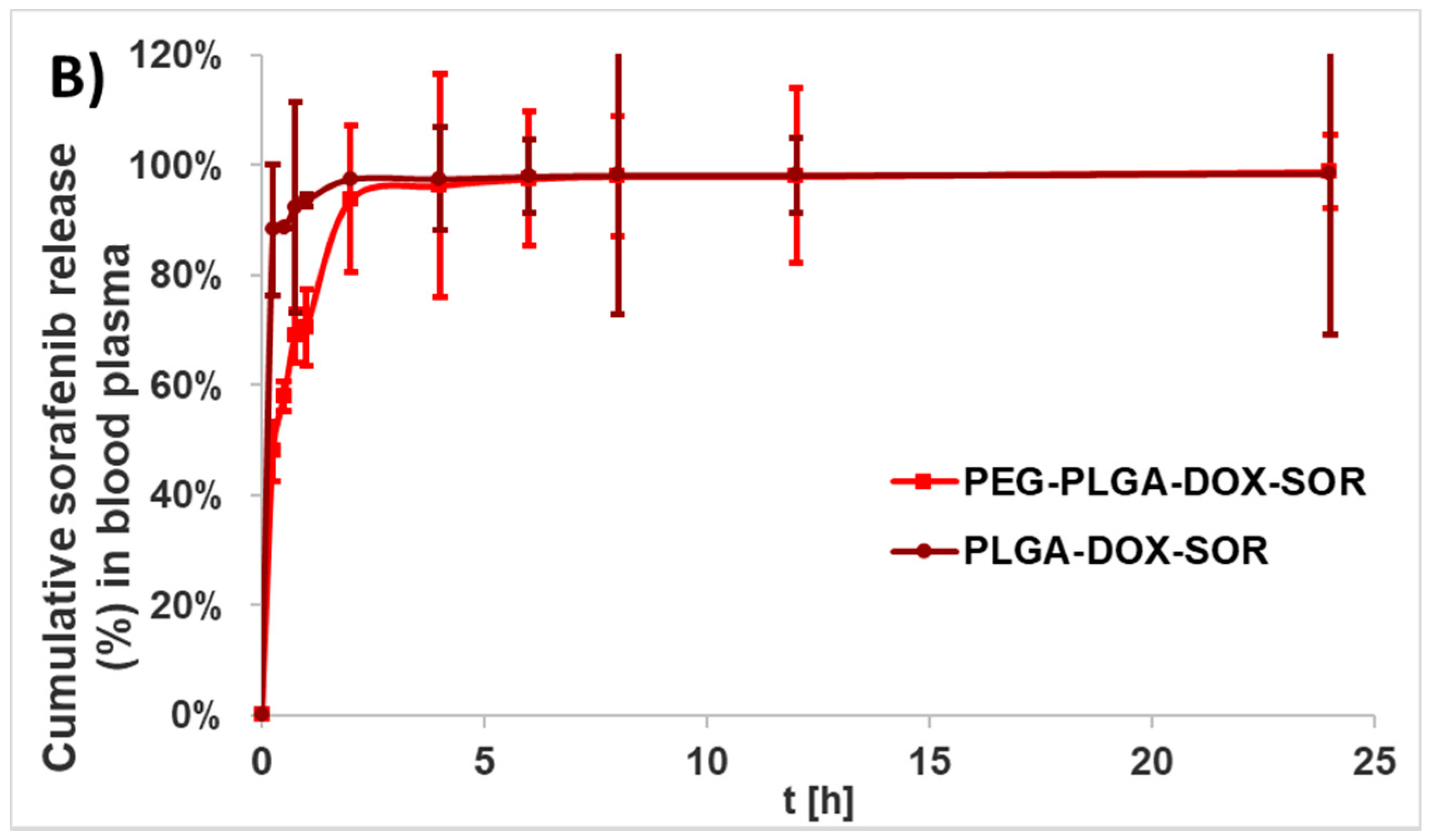
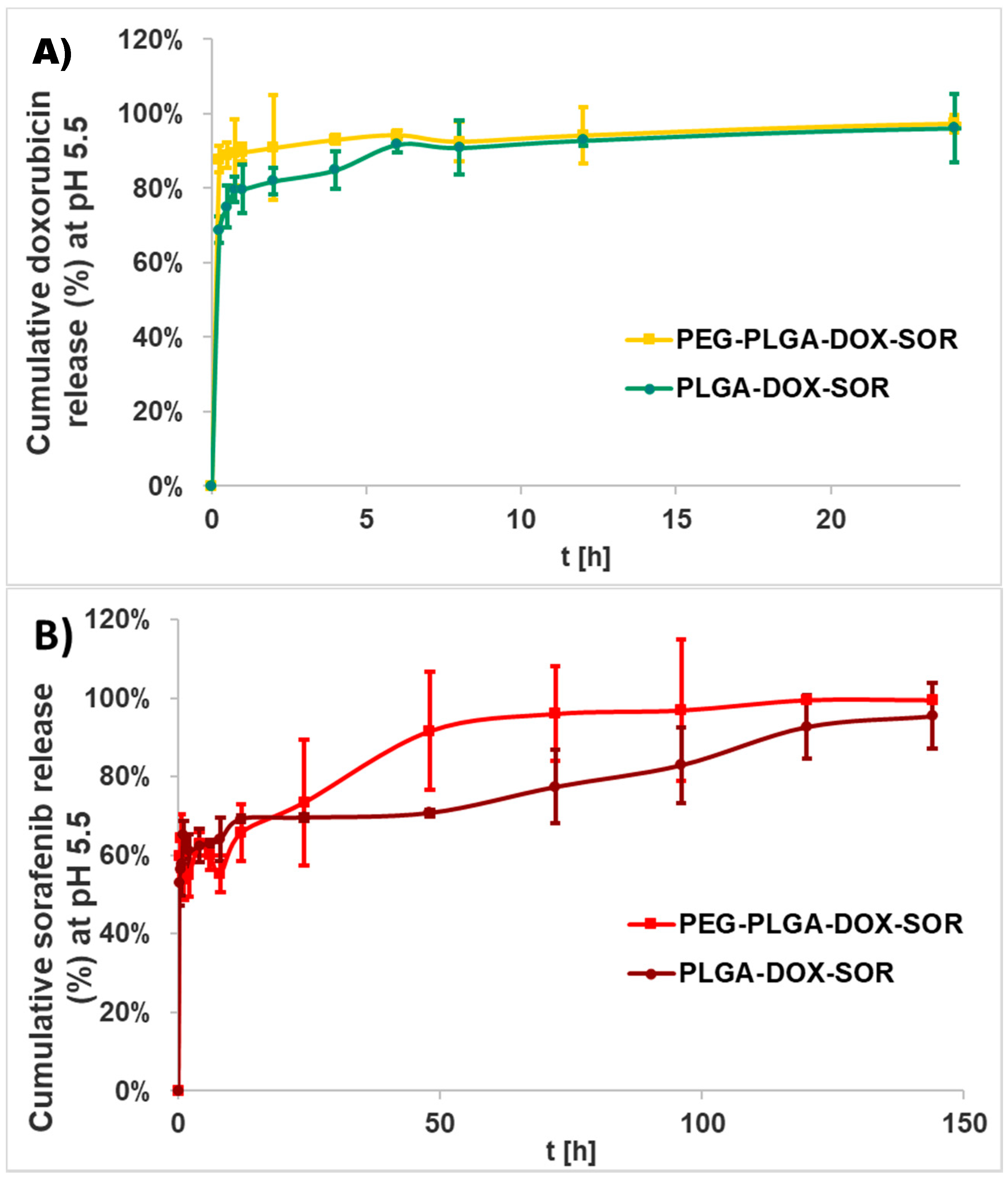
The in vitro drug release of the nanocomposites was investigated in human blood plasma and in ammonium acetate buffer (pH 5.5) because of the acidic tumor microenvironment. For in vitro release experiments after the washing steps, a 2 mL suspension including 4.4 mg PLGA or 6.4 mg PEG-PLGA nanocomposites was resuspended in 15 mL human blood plasma containing 0.03% sodium azide bactericide. Five milliliters (5 mL) of nanoparticle suspension in the release medium were pipetted to 5 mL non-transparent Eppendorf tubes, incubated at 37 °C in a G24 Environmental Incubator Shaker (New Brunswick Scientific Co. Inc., Edison, NJ, USA), and shaken by a BIO RS-24 Mini-rotator (Biosan, Rīga, Latvia) for 7 days at 700 rpm. Three parallel samples per nanocomposite were investigated. After 1 h and every 24 h, 0.5 mL from each sample were centrifuged (Hermle Z216 MK microcentrifuge, Gosheim, Germany) for 20 min at 15,000 rpm, washed three times, and the pellet was dissolved in 0.5 mL DMSO.
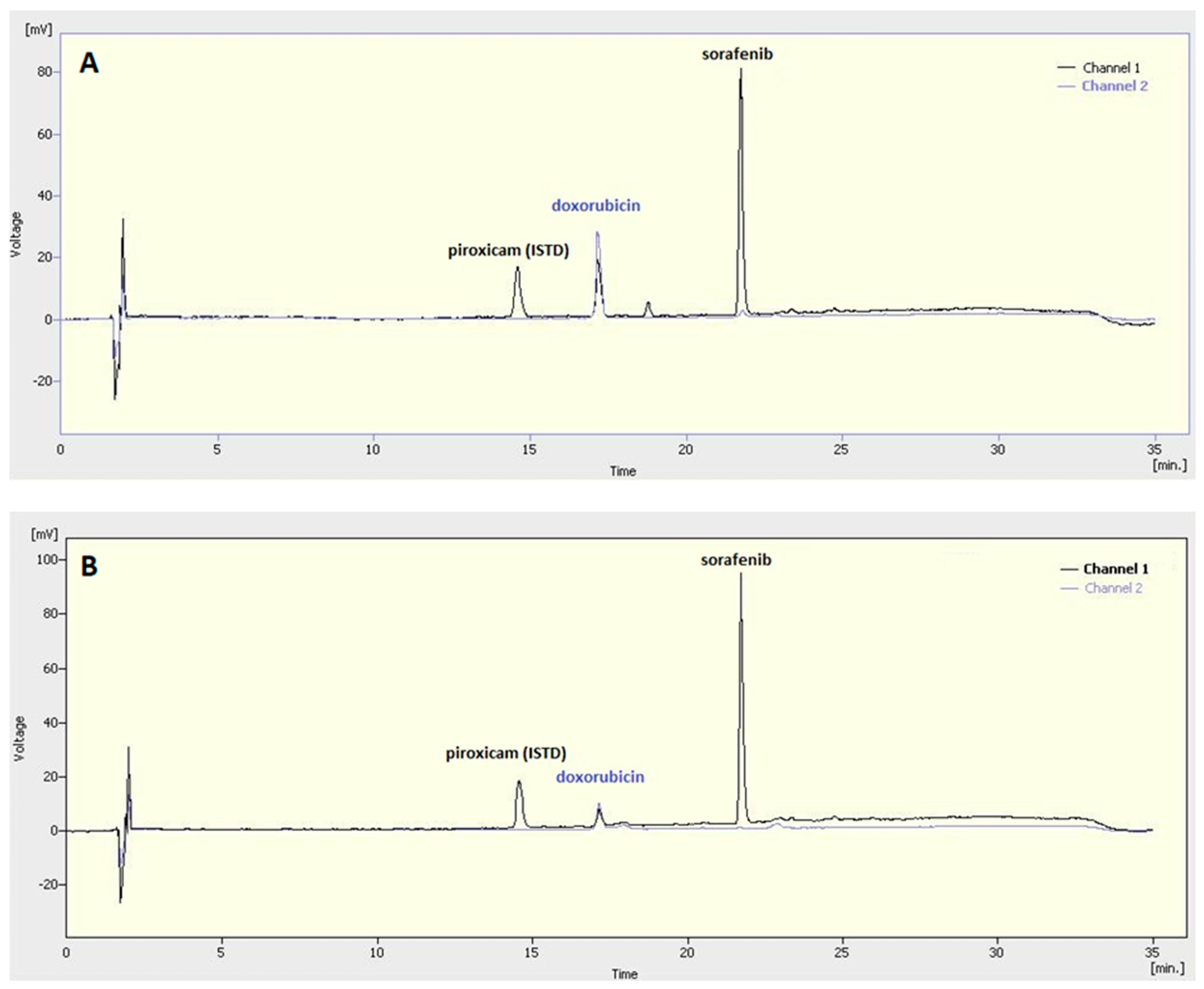
The sorafenib and doxorubicin concentration was measured by a Young Lin YL 9100 HPLC instrument (YL Instruments Co., Ltd., Gyeonggi-do, Korea) at 30 °C. The active agents were separated by a Zorbax SB-Aq column (150 mm × 4.6 mm, 5 μm; Agilent, Santa Clara, CA, USA). The mobile phase composition is given in
Table 1. The flow rate was adjusted to 1 mL/min. The detection wavelength of sorafenib and doxorubicin was 280 and 480 nm, respectively. Piroxicam was used as an internal standard during the measurements.
The concentration of sorafenib and doxorubicin was calculated using calibration curves and the encapsulation efficiencies were calculated as follows:
2.4. Attachment of Fluorescent Dye
A 1 mL nanoparticle suspension (2.2 mg/mL PLGA and 3.2 mg/mL PEG-PLGA) was centrifuged and washed with MilliQ water, redispersed in 0.5 mL PBS (pH 7.4), mixed with 0.1 mL PBS (pH 7.4) involving a 50× molar excess of EDC and NHS related to the (PEG-)PLGA concentration, then incubated for 60 min at 25 °C, centrifuged and washed with MilliQ water, and redispersed in 1.0 mL PBS (pH 7.4). The obtained carbodiimide-activated nanoparticle dispersion was pipetted to a 0.02 mL PBS (pH 7.4) solution containing 0.5 mg/mL Cyanine 5 amine fluorescent dye and incubated for 1 h at 25 °C. Then, the nanocomposite dispersion was centrifuged, washed three times, and redispersed in 1 mL PBS.
2.5. Cell Cultures
The human cancer cell line HT-29 was grown in RPMI-1640 medium supplemented with 10% fetal calf serum (FCS) and 100 U/mL penicillin. The cells were cultured at 37 °C in a humidified atmosphere containing 5% CO2. They were trypsinized, resuspended, and precultured before use.
2.6. In Vitro Cellular Uptake and Cytotoxicity Studies
The HT-29 cellular uptake of the nanoparticles was evaluated using flow cytometry. The cells were cultured in 24-well plates at a cell density of 2 × 105 cells per well at 37 °C for 24 h. After cultivation, 100 mg of fluorescently labelled nanoparticles/well were added to the cells and incubated for 24 h. Cells grown without nanoparticles were used as a negative control. The cells were washed by PBS, trypsinized, and redispersed in PBS containing 2% BSA. Flow cytometry was performed on a BD FACSAria III Cell sorter (BD Biosciences, San Jose, CA, USA) at an Ex/Em wavelength of 633/660 nm. Every sample was analyzed in triplicate.
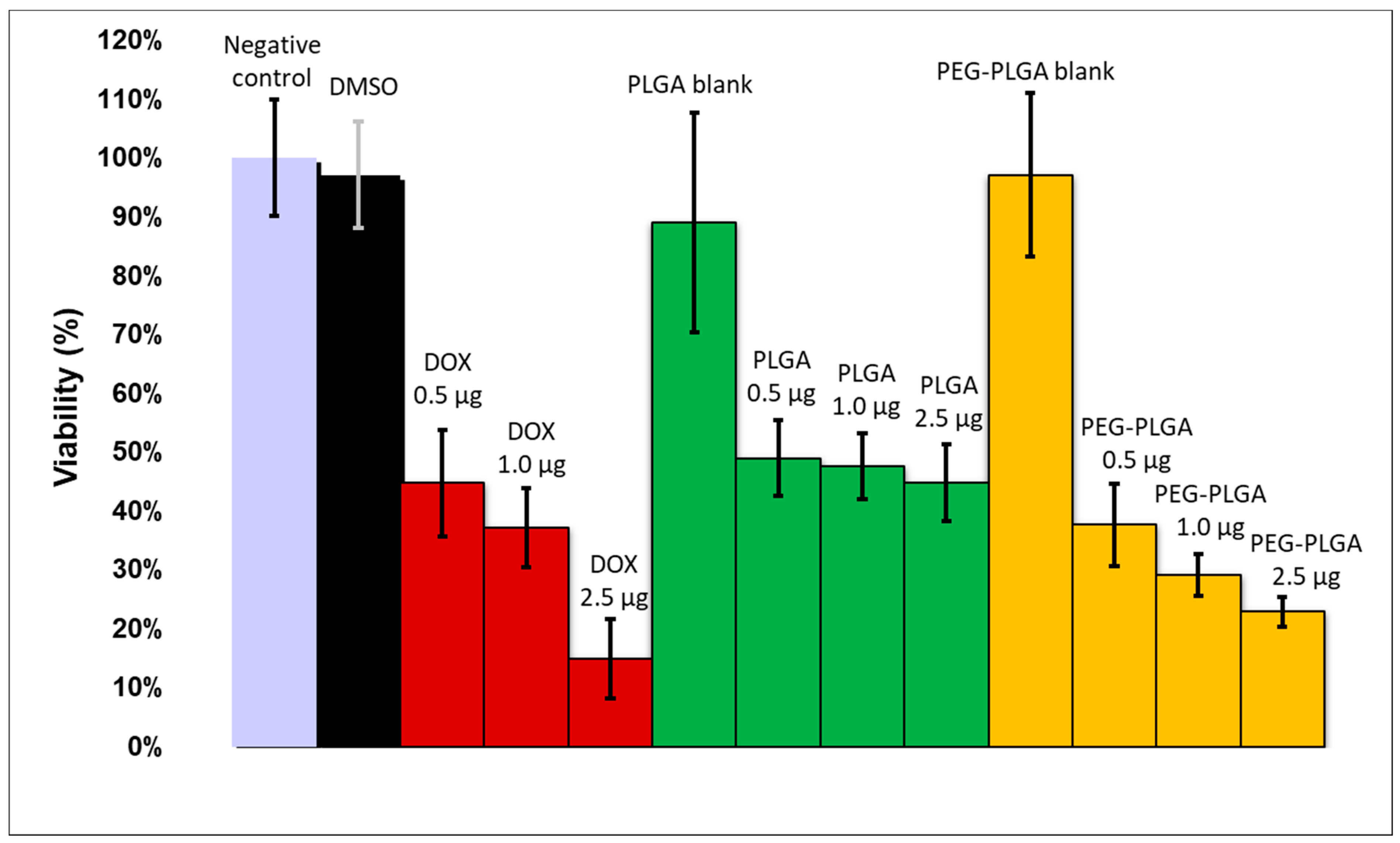
The in vitro cytotoxicity caused in HT-29 cells was assayed using MTT reagent. Cells were seeded (10,000 cells/well) in 96-well plates. After 24 h of pre-incubation, the growth media were replaced with 200 μL of fresh RPMI-1640 medium containing 10% FCS and PLGA- and PEG-PLGA blank nanoparticles or the dual-drug-entrapping nanocomposites. Three different doxorubicin concentration levels of the added nanopharmaceuticals were applied: 0.5, 1.0, and 2.5 μg per well. The nanocomposites also contained sorafenib; however, it was a higher amount of sorafenib (with 6% and 28% using PLGA and PEG-PLGA, respectively) since its entrapment was more efficacious. The positive control samples were also supplied with the same amount of free doxorubicin and sorafenib in DMSO solution. DMSO cytotoxicity (without drugs) was also investigated. After 48 h of incubation, 20 mL/well MTT solution (5 mg MTT/mL) and 0.2 mL/well supplemented culture media were added followed by further incubation for 2 h. The supernatant was removed, and MTT lysis solution (DMSO, 1% acetic acid, 10% SDS) was added into each well to dissolve the cells with MTT formazan crystals. The absorbance was determined at 492 nm by a Robonik Readwell Touch (Navi Mumbai, India) plate reader. The percentage of viable cells was calculated by comparing the absorbance of treated cells against that of the untreated cells (negative control). The data were presented as the mean and standard deviation with eight replicates.
4. Conclusions
Doxorubicin and sorafenib co-loaded therapeutic nanocomposites were developed using PLGA- and PEG-PLGA-encapsulating polymers, respectively, by the double emulsion solvent evaporation method. The nanoparticles possessed promising physical and chemical properties; that is, a small size, high yield, high drug encapsulation efficiency, and high drug loading. The doxorubicin was released continuously within 6 days, while the sorafenib was released quickly during 24 h under biorelevant conditions. In an acidic tumor-simulated condition, the two agents presented opposing characteristics with accelerated doxorubicin and sustained sorafenib release. The hydrolysis of PEG-PLGA was quicker in both of the media. PEG-PLGA nanocomposites displayed higher cellular uptake, which harmonizes with the higher cytotoxicity experienced.