Regioselective Synthesis of Lactulose Esters by Candida antarctica and Thermomyces lanuginosus Lipases
Abstract
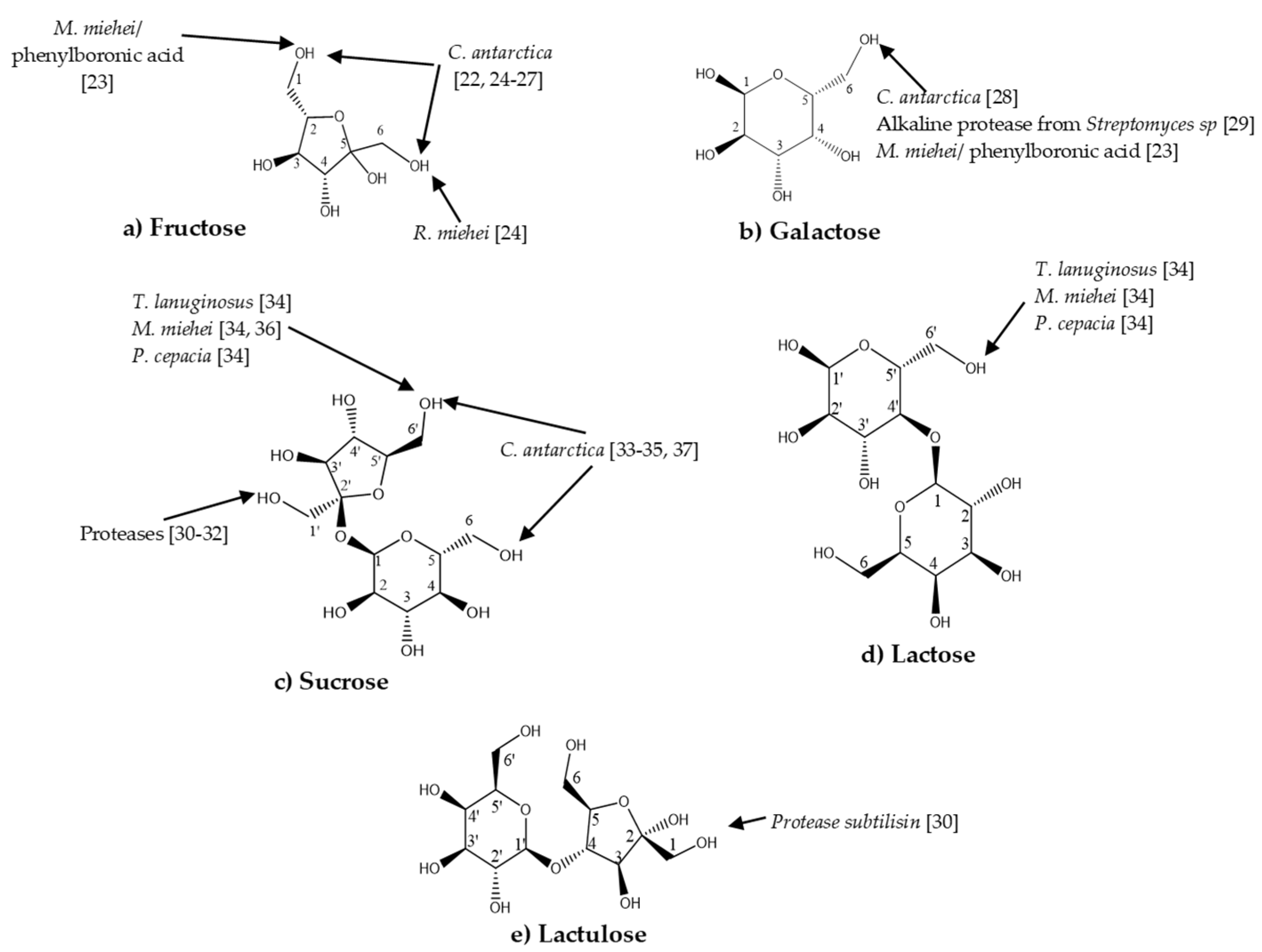
:1. Introduction
2. Results
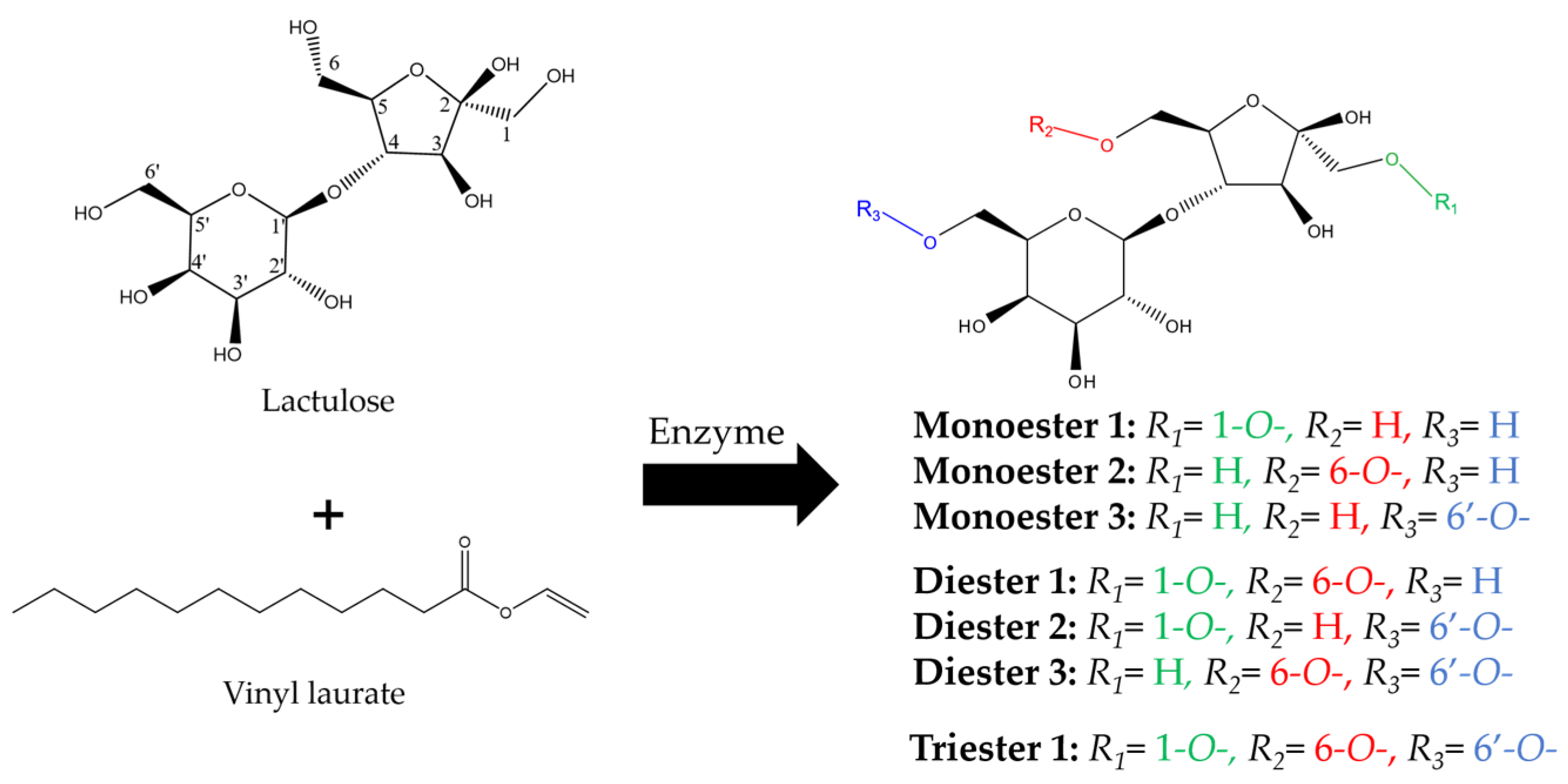
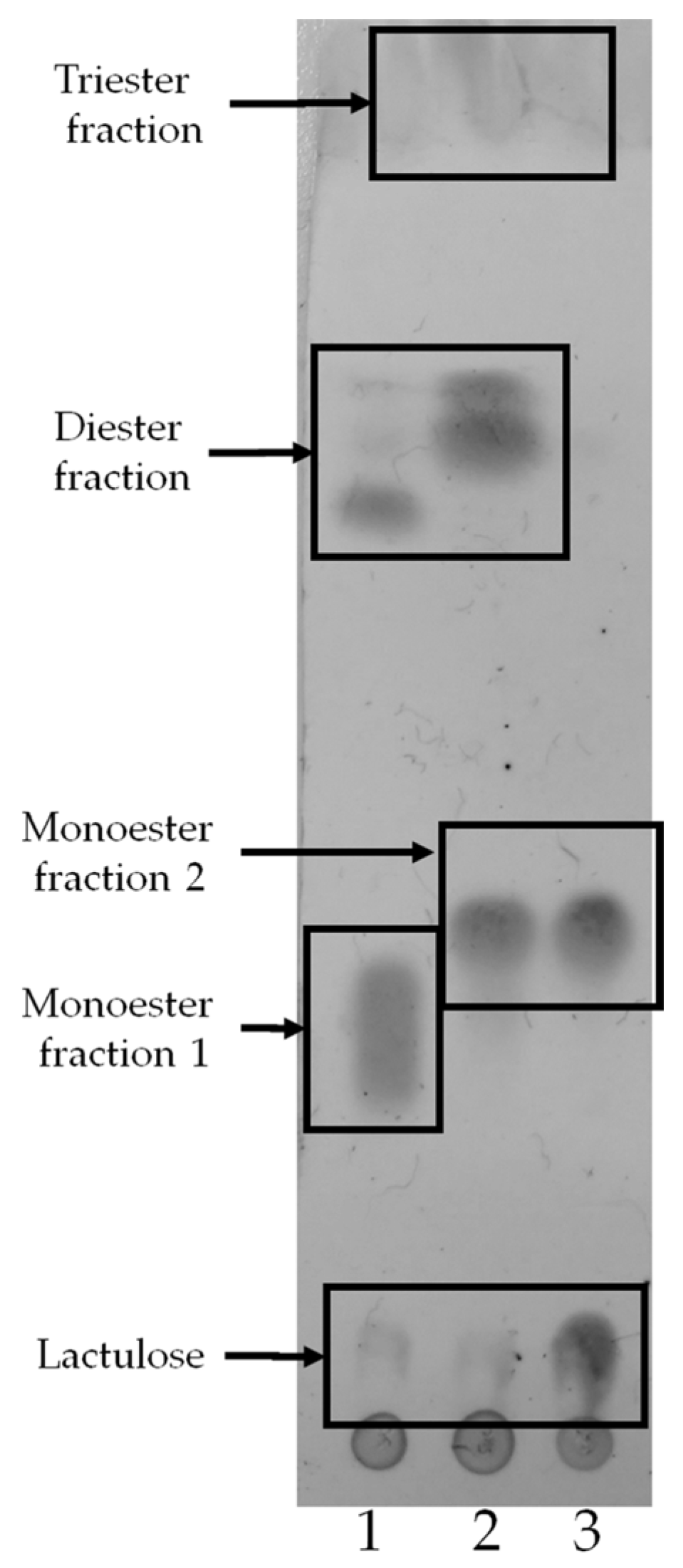
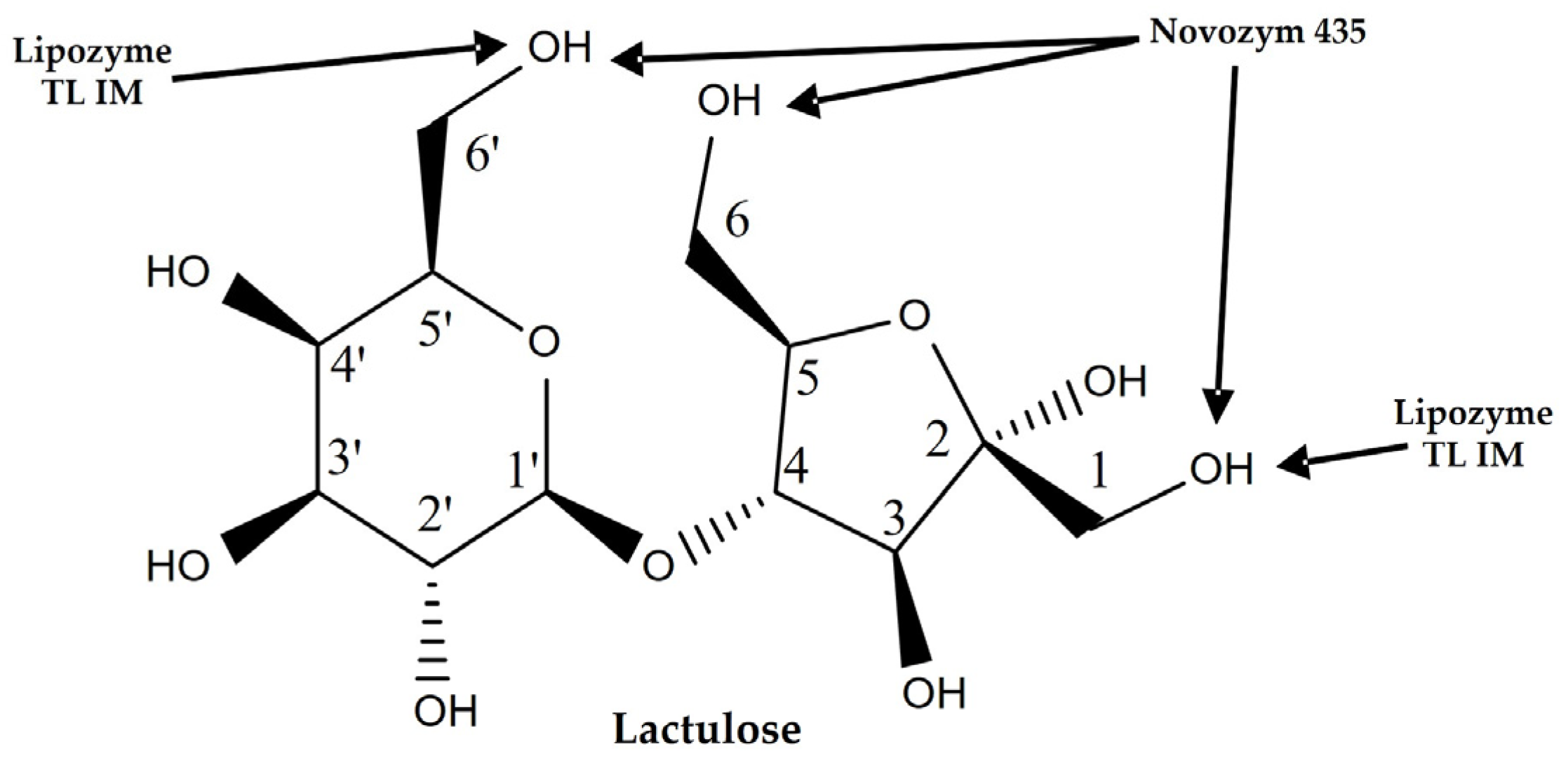
2.1. Enzymatic Synthesis of Lactulose Esters and Screening with Different Biocatalysts
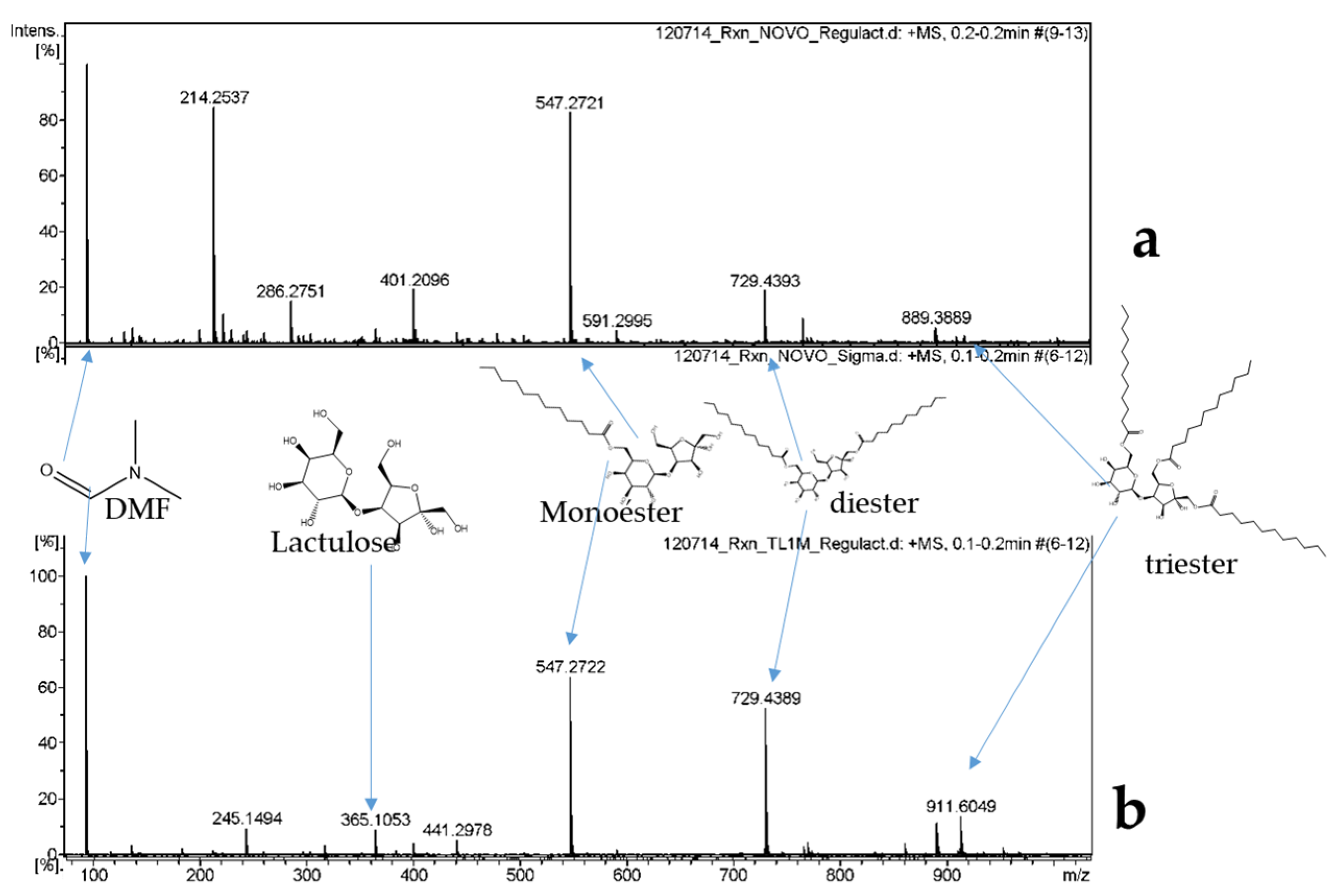
2.2. Characterization by FT-IR, 1H, 13C, and 2D (HMBC) NMR and MS-ESI
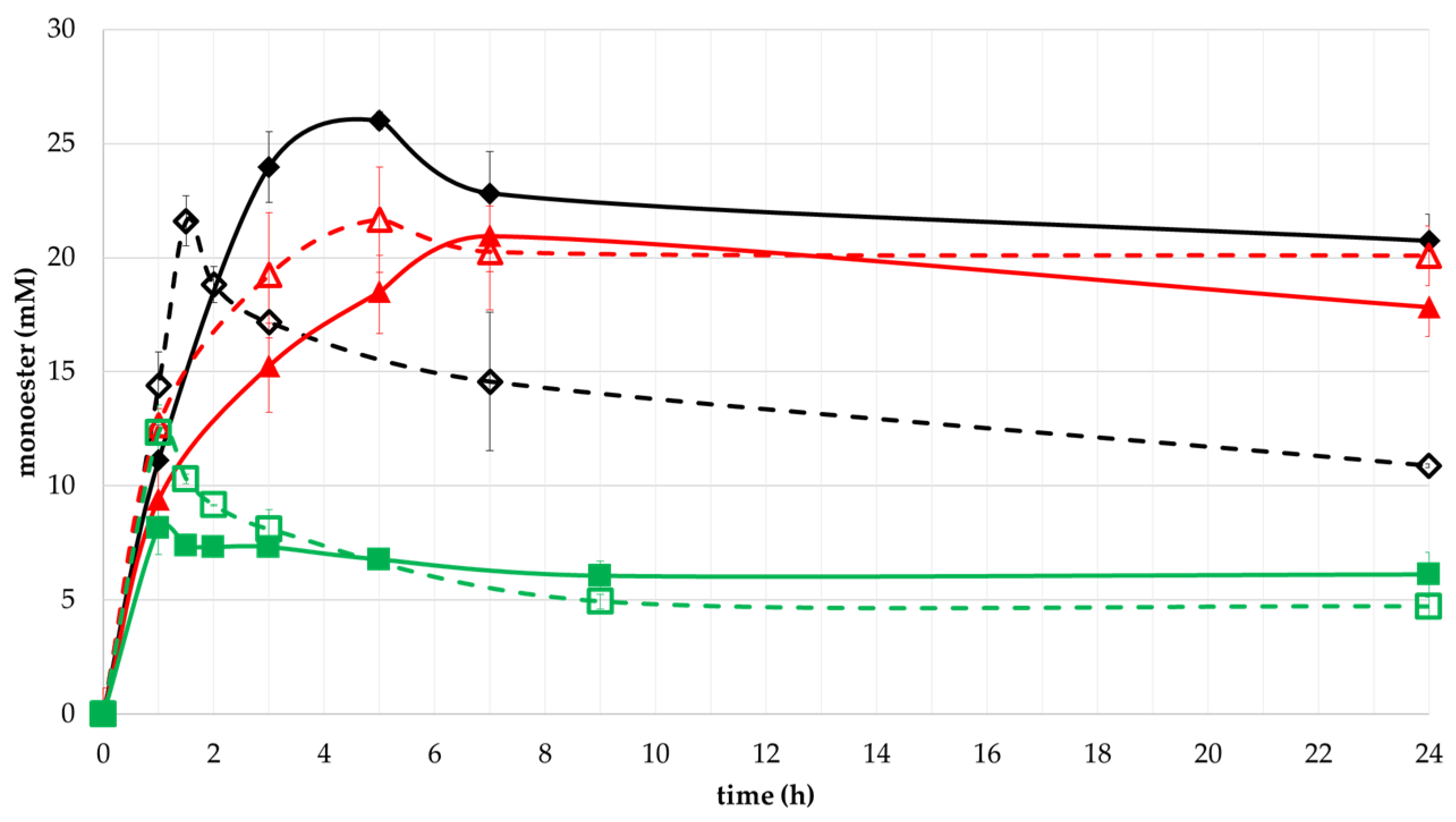
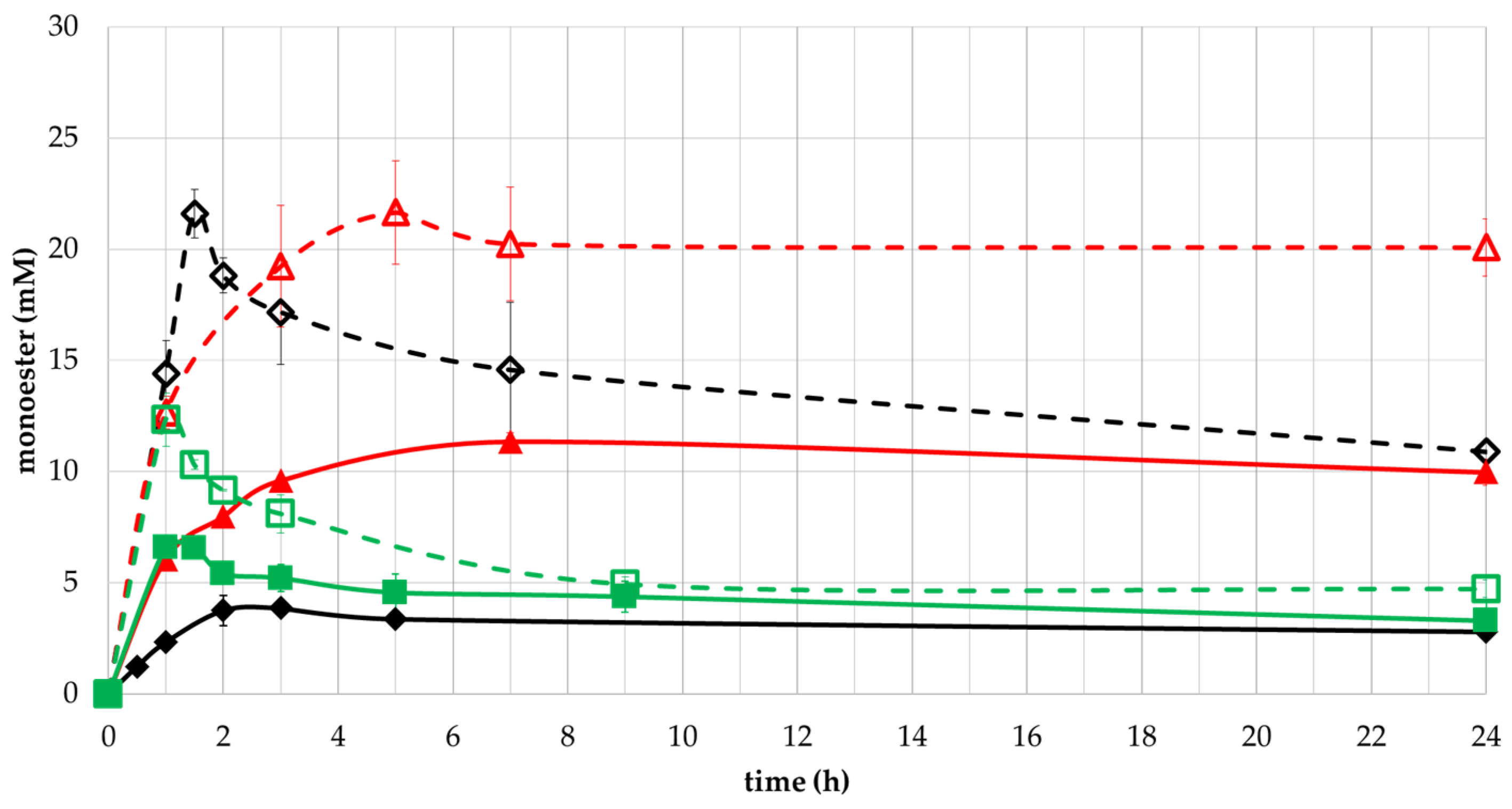
2.3. Conversion to Monoester by Selected Enzymes and Effect of Temperature in the Reaction
Effect of Substrates Concentrations on Monoesters Yield
2.4. Calculation of the Hydrophilic-Lipophilic Balance (HLB)
2.4.1. Griffin Method
- Mh = 341.29 g/mol
- MT-mono = 524.60 g/mol
- MT-di = 706.91 g/mol
- MT-tri = 889.22 g/mol
- HLB mono = 20 (341.29/524.60) = 13.01 ≈ 13
- HLB di = 20 (341.29/706.91) = 9.65 ≈ 10
- HLB tri = 20 (341.29/889.22) = 7.67 ≈ 8
- HLBmono: HLB value by monoester
- HLBdi: HLB value by diester
- HLBtri: HLB value by triester
2.4.2. Davies Method
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Methods
4.2.1. Enzymatic Reactions
4.2.2. Products Purification
4.2.3. TLC and HPLC Analysis
4.2.4. Products Characterization by FT-IR, 1H, 13C, and 2D NMR and MS-ESI
4.2.5. Calculation of HLB Values
- Mh = weight fraction of the hydrophilic group
- MT = the molecular weight of the whole molecule
5. Conclusion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ballesteros, A.; Plou, F.J.; Alcalde, M.; Ferrer, M.; García-Arellano, H.; Reyes-Duarte, D.; Ghazi, I. Enzymatic synthesis of sugar esters and oligosaccharides from renewable resources. In Biocatalysis in the Pharmaceutical and Biotechnology Industries; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Mitsubishi-Kagaku-Foods Corporation. Ryoto sugar esters technical information. 2004. Available online: http://www.mfc.co.jp/english/infor.htm (accessed on 3 September 2017).
- Abdulmalek, E.; Mohd Saupi, H.S.; Tejo, B.A.; Basri, M.; Salleh, A.B.; Raja Abd Rahman, R.N.Z.; Abdul Rahman, M.B. Improved enzymatic galactose oleate ester synthesis in ionic liquids. J. Mol. Catal. B Enzym. 2012, 76, 37–43. [Google Scholar] [CrossRef]
- Nobmann, P.; Smith, A.; Dunne, J.; Henehan, G.; Bourke, P. The antimicrobial efficacy and structure activity relationship of novel carbohydrate fatty acid derivatives against Listeria spp. and food spoilage microorganisms. Int. J. Food Microbiol. 2009, 128, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhang, H.; Hao, T.; Li, S. In vitro antibacterial activities and mechanism of sugar fatty acid esters against five food-related bacteria. Food Chem. 2015, 187, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Habulin, M.; Šabeder, S.; Knez, Ž. Enzymatic synthesis of sugar fatty acid esters in organic solvent and in supercritical carbon dioxide and their antimicrobial activity. J. Supercrit. Fluids 2008, 45, 338–345. [Google Scholar] [CrossRef]
- Devulapalle, K.S.; Gómez de Segura, A.; Ferrer, M.; Alcalde, M.; Mooser, G.; Plou, F.J. Effect of carbohydrate fatty acid esters on Streptococcus sobrinus and glucosyltransferase activity. Carbohydr. Res. 2004, 339, 1029–1034. [Google Scholar] [CrossRef] [PubMed]
- Karlová, T.; Polakova, L.; Šmidrkal, J.; Filip, V. Antimicrobial effects of fatty acid fructose esters. Czech J. Food Sci. 2010, 28, 146–149. [Google Scholar]
- Ferrer, M.; Soliveri, J.; Plou, F.J.; López-Cortés, N.; Reyes-Duarte, D.; Christensen, M.; Copa-Patiño, J.L.; Ballesteros, A. Synthesis of sugar esters in solvent mixtures by lipases from Thermomyces lanuginosus and Candida antarctica B, and their antimicrobial properties. Enzym. Microb. Technol. 2005, 36, 391–398. [Google Scholar] [CrossRef]
- Puterka, G.J.; Farone, W.; Palmer, T.; Barrington, A. Structure-function relationships affecting the insecticidal and miticidal activity of sugar esters. J. Econ. Entomol. 2003, 96, 636–644. [Google Scholar] [CrossRef] [PubMed]
- Noker, P.E.; Lin, T.H.; Hill, D.L.; Shigeoka, T. Metabolism of 14c-labelled sucrose esters of stearic acid in rats. Food Chem. Toxicol. 1997, 35, 589–595. [Google Scholar] [CrossRef]
- Shigeoka, T.; Izawa, O.; Kitazawa, K.; Yamauchi, F.; Murata, T. Studies on the metabolic fate of sucrose esters in rats. Food Chem. Toxicol. 1984, 22, 409–414. [Google Scholar] [CrossRef]
- Eldred, J.S. Gras Notification for Sucrose Fatty Acid Esters; Center for Food Safety and Applied Nutrition, Food and Drug Administration: Washington, DC, USA, 2003.
- Sutili, F.K.; Ruela, H.S.; Leite, S.G.F.; Miranda, L.S.d.M.; Leal, I.C.R.; de Souza, R.O.M.A. Lipase-catalyzed esterification of steric hindered fructose derivative by continuous flow and batch conditions. J. Mol. Catal. B Enzym. 2013, 85–86, 37–42. [Google Scholar] [CrossRef]
- Van Kempen, S.E.H.J.; Boeriu, C.G.; Schols, H.A.; de Waard, P.; van der Linden, E.; Sagis, L.M.C. Novel surface-active oligofructose fatty acid mono-esters by enzymatic esterification. Food Chem. 2013, 138, 1884–1891. [Google Scholar] [CrossRef] [PubMed]
- Van Kempen, S.E.H.J.; Schols, H.A.; van der Linden, E.; Sagis, L.M.C. Effect of variations in the fatty acid chain on functional properties of oligofructose fatty acid esters. Food Hydrocoll. 2014, 40, 22–29. [Google Scholar] [CrossRef]
- Horchani, H.; Aissa, I.; Ouertani, S.; Zarai, Z.; Gargouri, Y.; Sayari, A. Staphylococcal lipases: Biotechnological applications. J. Mol. Catal. B Enzym. 2012, 76, 125–132. [Google Scholar] [CrossRef]
- Bernal, C.; Illanes, A.; Wilson, L. Heterofunctional hydrophilic-hydrophobic porous silica as support for multipoint covalent immobilization of lipases: Application to lactulose palmitate synthesis. Langmuir 2014, 30, 3557–3566. [Google Scholar] [CrossRef] [PubMed]
- Bernal, C.; Illanes, A.; Wilson, L. Improvement of efficiency in the enzymatic synthesis of lactulose palmitate. J. Agric. Food Chem. 2015, 63, 3716–3724. [Google Scholar] [CrossRef] [PubMed]
- Corzo, N.; Alonso, J.L.; Azpiroz, F.; Calvo, M.A.; Cirici, M.; Leis, R.; Lombo, F.; Mateos-Aparicio, I.; Plou, F.J.; Ruas-Madiedo, P.; et al. [prebiotics: Concept, properties and beneficial effects]. Nutr. Hosp. 2015, 31 (Suppl. 1), 99–118. [Google Scholar] [PubMed]
- Panesar, P.S.; Kumari, S. Lactulose: Production, purification and potential applications. Biotechnol. Adv. 2011, 29, 940–948. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.P.; Kim, H.K. Antibacterial effect of fructose laurate synthesized by Candida antarctica b lipase-mediated transesterification. J. Microbiol. Biotechnol. 2016, 26, 1579–1585. [Google Scholar] [CrossRef] [PubMed]
- Schlotterbeck, A.; Lang, S.; Wray, V.; Wagner, F. Lipase-catalyzed monoacylation of fructose. Biotechnol. Lett. 1993, 15, 61–64. [Google Scholar] [CrossRef]
- Scheckermann, C.; Schlotterbeck, A.; Schmidt, M.; Wray, V.; Lang, S. Enzymatic monoacylation of fructose by two procedures. Enzym. Microb. Technol. 1995, 17, 157–162. [Google Scholar] [CrossRef]
- Li, L.; Ji, F.; Wang, J.; Jiang, B.; Li, Y.; Bao, Y. Efficient mono-acylation of fructose by lipase-catalyzed esterification in ionic liquid co-solvents. Carbohydr. Res. 2015, 416, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Ji, F.; Wang, J.; Li, Y.; Bao, Y. Esterification degree of fructose laurate exerted by Candida antarctica lipase b in organic solvents. Enzym. Microb. Technol. 2015, 69, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Tükel, S.; Sahin, P.B.; Yildirim, D. Optimization of lipase-catalyzed synthesis of fructose stearate using response surface methodology. Artif. Cells Nanomedicine Biotechnol. 2013, 41, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Kimura, Y.; Adachi, S. Continuous synthesis of 6-O-linoleoyl hexose using a packed-bed reactor system with immobilized lipase. Biochem. Eng. J. 2005, 22, 145–149. [Google Scholar] [CrossRef]
- Kitagawa, M.; Fan, H.; Raku, T.; Shibatani, S.; Maekawa, Y.; Hiraguri, Y.; Kurane, R.; Tokiwa, Y. Selective enzymatic preparation of vinyl sugar esters using dmso as a denaturing co-solvent. Biotechnol. Lett. 1999, 21, 355–359. [Google Scholar] [CrossRef]
- Riva, S.; Nonini, M.; Ottolina, G.; Danieli, B. Subtilisin-catalyzed esterification of di-and oligosaccharides containing a d-fructose moiety. Carbohydr. Res. 1998, 314, 259–266. [Google Scholar] [CrossRef]
- Potier, P.; Bouchu, A.; Gagnaire, J.; Queneau, Y. Proteinase N-catalysed regioselective esterification of sucrose and other mono- and disaccharides. Tetrahedron Asymmetry 2001, 12, 2409–2419. [Google Scholar] [CrossRef]
- Wang, X.; Miao, S.; Wang, P.; Zhang, S. Highly efficient synthesis of sucrose monolaurate by alkaline protease protex 6l. Bioresour. Technol. 2012, 109, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Woudenberg-van Oosterom, M.; van Rantwijk, F.; Sheldon, R.A. Regioselective acylation of disaccharides in tert-butyl alcohol catalyzed by Candida antarctica lipase. Biotechnol. Bioeng. 1996, 49, 328–333. [Google Scholar] [CrossRef]
- Walsh, M.K.; Bombyk, R.A.; Wagh, A.; Bingham, A.; Berreau, L.M. Synthesis of lactose monolaurate as influenced by various lipases and solvents. J. Mol. Catal. B Enzym. 2009, 60, 171–177. [Google Scholar] [CrossRef]
- Chen, Y.; Nummer, B.; Walsh, M.K. Antilisterial activity of lactose monolaurate in milk, drinkable yogurt and cottage cheese. Lett. Appl. Microbiol. 2014, 58, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Han, J.J.; Yoon, J.H.; Rhee, J.S. Effect of salt hydrate pair on lipase-catalyzed regioselective monoacylation of sucrose. Biotechnol. Bioeng. 1998, 57, 121–125. [Google Scholar] [CrossRef]
- Ferrer, M.; Cruces, M.A.; Bernabe, M.; Ballesteros, A.; Plou, F.J. Lipase-catalyzed regioselective acylation of sucrose in two-solvent mixtures. Biotechnol. Bioeng. 1999, 65, 10–16. [Google Scholar] [CrossRef]
- Casas-Godoy, L.; Duquesne, S.; Bordes, F.; Sandoval, G.; Marty, A. Lipases: An overview. In Lipases and Phospholipases: Methods and Protocols; Sandoval, G., Ed.; Humana Press: Totowa, NJ, USA, 2012; pp. 3–30. [Google Scholar]
- Pleiss, J.; Fischer, M.; Schmid, R.D. Anatomy of lipase binding sites: The scissile fatty acid binding site. Chem. Phys. Lipids 1998, 93, 67–80. [Google Scholar] [CrossRef]
- Reyes-Duarte, D.; López-Cortés, N.; Ferrer, M.; Plou, F.J.; Ballesteros, A. Parameters affecting productivity in the lipase-catalysed synthesis of sucrose palmitate. Biocatal. Biotransformation 2005, 23. [Google Scholar] [CrossRef] [Green Version]
- Neta, N.S.; Teixeira, J.A.; Rodrigues, L.R. Sugar ester surfactants: Enzymatic synthesis and applications in food industry. Crit. Rev. Food Sci. Nutr. 2015, 55, 595–610. [Google Scholar] [CrossRef] [PubMed]
- Gumel, A.M.; Annuar, M.S.M.; Heidelberg, T.; Chisti, Y. Lipase mediated synthesis of sugar fatty acid esters. Process Biochem. 2011, 46, 2079–2090. [Google Scholar] [CrossRef]
- ICI Americas. The Hlb System: A Time-saving Guide to Emulsifier Selection; ICI Americas, Incorporated: New Castle, DE, USA, 1984; 19p. [Google Scholar]
- Hydrophile-lipophile balance of surfactants. Stud. Interface Sci. 2000, 9, 146–266. Available online: http://dx.doi.org/10.1016/S1383-7303(00)80016-2 (accessed on 3 September 2017).
- Zheng, Y.; Zheng, M.; Ma, Z.; Xin, B.; Guo, R.; Xu, X. Sugar fatty acid esters. In Polar Lipids; Elsevier: Amsterdam, The Netherlands, 2015; pp. 215–243. [Google Scholar]
- Naik, S.; Basu, A.; Saikia, R.; Madan, B.; Paul, P.; Chaterjee, R.; Brask, J.; Svendsen, A. Lipases for use in industrial biocatalysis: Specificity of selected structural groups of lipases. J. Mol. Catal. B Enzym. 2010, 65, 18–23. [Google Scholar] [CrossRef]
- Gennaro, A.R. Remington Farmacia; Editorial Médica Panamericana: Mexico City, Mexico, 2003; Volume 2, p. 2506. [Google Scholar]
- Griffin, W.C. Calculation of HLB values of non-ionic surfactants. Am. Perfum. Essent Oil Rev. 1954, 65, 26–29. [Google Scholar]
- Griffin, W.C. Classification of surface-active agents by "HLB". J. Soc. Cosmet. Chem. 1949, 1, 311–326. [Google Scholar]
- Davies, J. A quantitative kinetic theory of emulsion type, i. Physical chemistry of the emulsifying agent, Gas/Liquid and Liquid/Liquid Interface. In Proceedings of the International Congress of Surface Activity, London, UK, 1957; pp. 426–438. [Google Scholar]

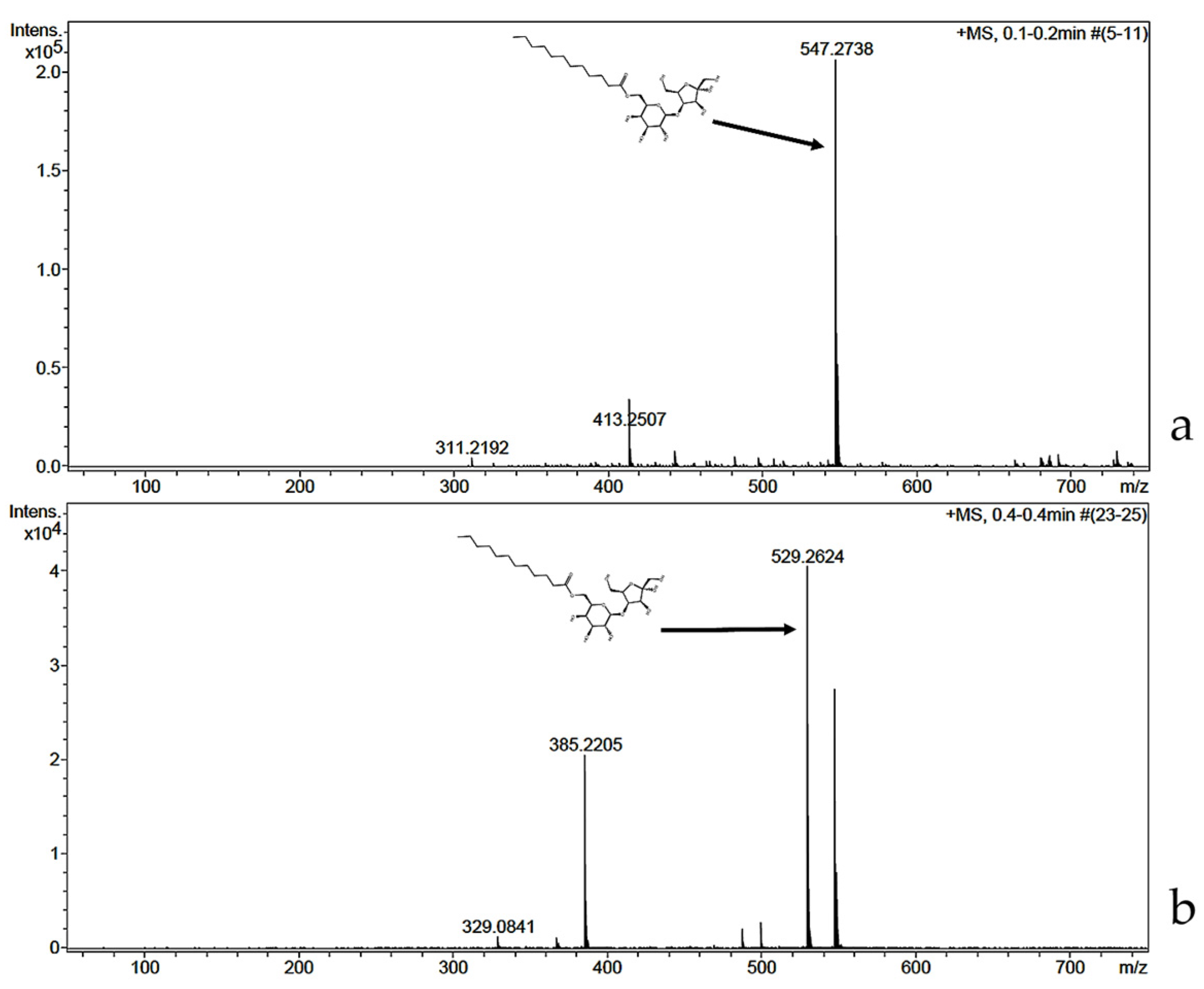
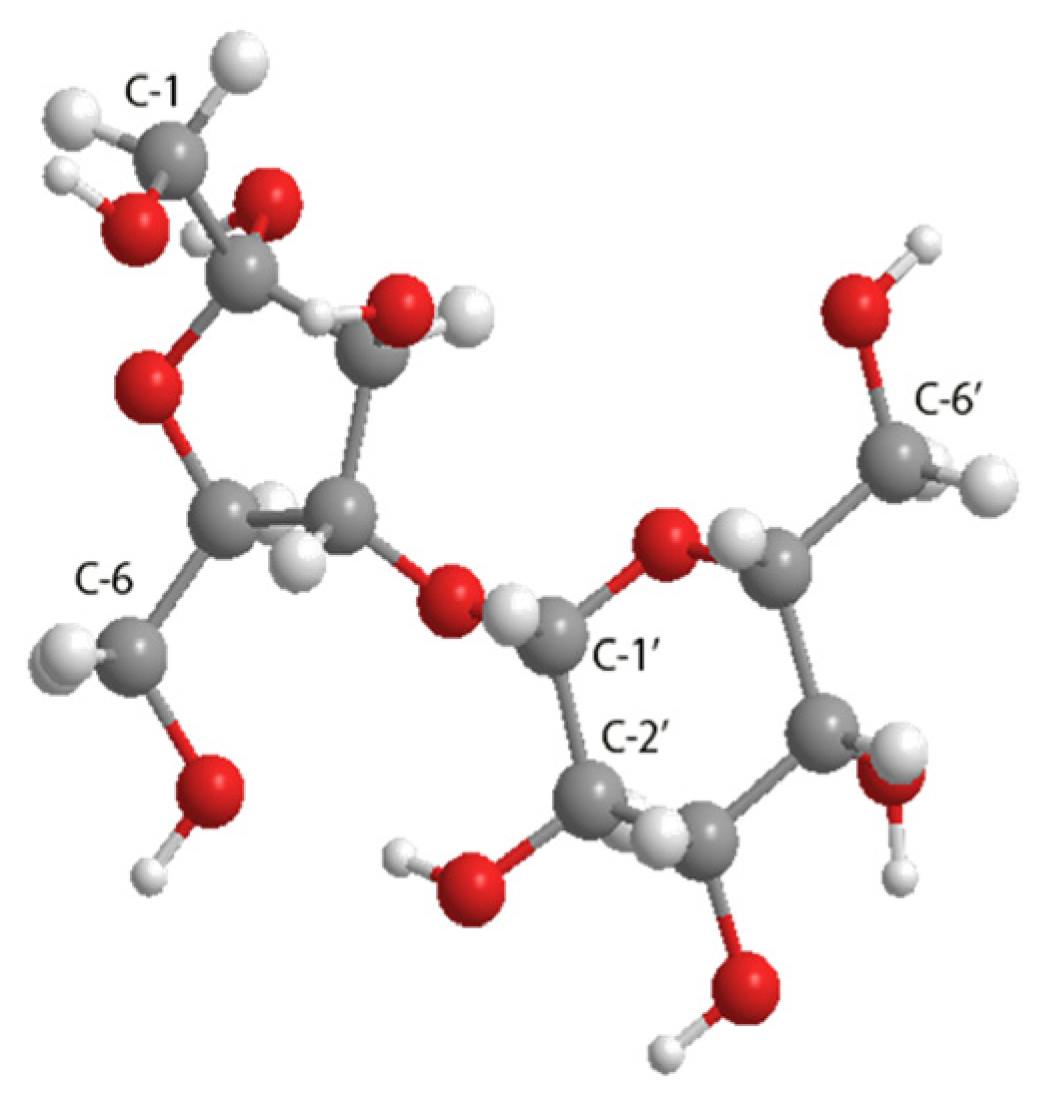

| Monoester Fraction | Monoester-1 (1-O) (ppm) | Monoester-6 (6-O) (ppm) | Monoester-6′ (6′-O) (ppm) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Atom | 13Cexp | 13Ccal | 1Hexp | 1Hcal | 13Cexp | 13Ccal | 1Hexp | 1Hcal | 13Cexp | 13Ccal | |
| Fructose moiety | 1 | 64.00 | 64.04 | 4.39 | 4.46 4.21 | 63.53 | 63.87 | 3.94 (dd) | 3.92 (dd) | 64.21 | 64.42 |
| 2 | 103.11 | 103.01 | 104.62 | 104.43 | 104.12 | 104.43 | |||||
| 4 | 87.00 | 87.18 | 3.61 (dd) | 3.65 (dd) | 87.01 | 87.18 | 3.60 (dd) | 3.63 (dd) | 86.9 | 88.20 | |
| 6 | 63.79 | 63.81 | 4.00, 3.58 | 3.99, 3.58 | 63.80 | 63.81 | 4.45, | 4.32, 4.07 | ---- | ---- | |
| Galactose moiety | 1′ | 103.97 | 103.92 | 3.92 | 4.03 | 103.96 | 103.92 | 3.92 | 4.03 | 3.92 | 4.03 |
| 5′ | 76.08 | 76.69 | 74.96 | 74.94 | |||||||
| 6′ | 61.43 | 61.72 | 3.70, 3.46 | 3.70, 3.50 | 62.63 | 62.56 | 4.14 (dd) | 4.13 (dd) | 4.42 | 4.32 4.07 | |
| Lauroyl moiety | 1″ | 173.5 | 173.5 | 174.1 | 174.1 | 173.8 | 173.5 | ||||
| 2″ | 33.58 | 34.03 | 2.38 (m) | 2.34 (m) | 33.56 | 34.03 | 2.39 | 2.37 (m) | |||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chávez-Flores, L.F.; Beltran, H.I.; Arrieta-Baez, D.; Reyes-Duarte, D. Regioselective Synthesis of Lactulose Esters by Candida antarctica and Thermomyces lanuginosus Lipases. Catalysts 2017, 7, 263. https://doi.org/10.3390/catal7090263
Chávez-Flores LF, Beltran HI, Arrieta-Baez D, Reyes-Duarte D. Regioselective Synthesis of Lactulose Esters by Candida antarctica and Thermomyces lanuginosus Lipases. Catalysts. 2017; 7(9):263. https://doi.org/10.3390/catal7090263
Chicago/Turabian StyleChávez-Flores, Luis F., Hiram Isaac Beltran, Daniel Arrieta-Baez, and Dolores Reyes-Duarte. 2017. "Regioselective Synthesis of Lactulose Esters by Candida antarctica and Thermomyces lanuginosus Lipases" Catalysts 7, no. 9: 263. https://doi.org/10.3390/catal7090263





