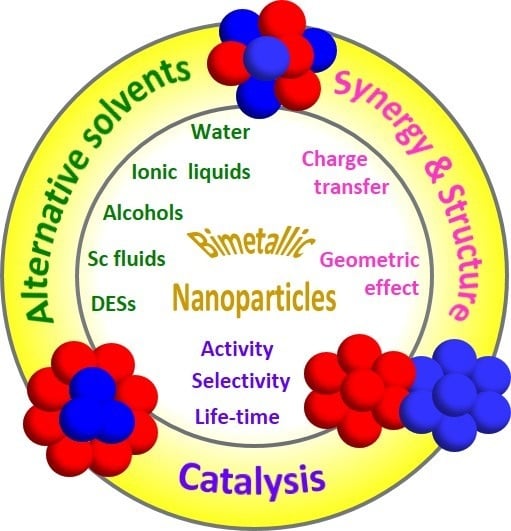
Bimetallic Nanoparticles in Alternative Solvents for Catalytic Purposes
Abstract
:1. Introduction
2. Water
2.1. Unsupported BMNPs in Water
2.2. Supported BMNPs in Water
3. Alcohols
3.1. Unsupported BMNPs in Alcohols
3.2. Supported BMNPs in Alcohols
4. Ionic Liquids and Deep Eutectic Solvents
4.1. Ionic Liquids
4.1.1. Synthesis of BMNPs in Ionic Liquids by Co-Decomposition of Metal Precursors
4.1.2. Synthesis of BMNPs in Ionic Liquids by Sequential Process
4.2. Deep Eutectic Solvents
5. Supercritical Fluids
5.1. Synthesis of BMNPs in Supercritical Fluids
5.2. Applications in Catalysis of BMNPs in Supercritical Fluids
6. Outlook
Acknowledgments
Conflicts of Interest
References
- Schmid, G. (Ed.) Nanoparticles. From Theory to Application; Wiley-VCH: Weinheim, Germany, 2005. [Google Scholar]
- Gao, W.; Hood, Z.D.; Chi, M. Interfaces in Heterogeneous Catalysts: Advancing Mechanistic Understanding through Atomic-Scale Measurements. Acc. Chem. Res. 2017, 50, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, M.; Mistry, H.; Roldan Cuenya, B. Tailoring the Catalytic Properties of Metal Nanoparticles via Support Interactions. J. Phys. Chem. Lett. 2016, 7, 3519–3533. [Google Scholar] [CrossRef] [PubMed]
- Picos, A.; Riva, M.; Brambilla, A.; Calloni, A.; Bussetti, G.; Finazzi, M.; Ciccacci, F.; Duo, L. Reactive metal–oxide interfaces: A microscopic view. Surf. Sci. Rep. 2016, 71, 32–76. [Google Scholar] [CrossRef]
- An, K.; Somorjai, G.A. Nanocatalysis I: Synthesis of Metal and Bimetallic Nanoparticles and Porous Oxides and Their Catalytic Reaction Studies. Catal. Lett. 2015, 145, 233–248. [Google Scholar] [CrossRef]
- Navalon, S.; Dhakshinamoorthy, A.; Alvaro, M.; Garcia, H. Metal nanoparticles supported on two-dimensional graphenes as heterogeneous catalysts. Coord. Chem. Rev. 2016, 312, 99–148. [Google Scholar] [CrossRef]
- Peng, L.; Ringe, E.; Van Duyne, R.P.; Marks, L.D. Segregation in bimetallic nanoparticles. Phys. Chem. Chem. Phys. 2015, 17, 27940–27951. [Google Scholar] [CrossRef] [PubMed]
- Notar Francesco, I.; Fontaine-Vive, F.; Antoniotti, S. Synergy in the Catalytic Activity of Bimetallic Nanoparticles and New Synthetic Methods for the Preparation of Fine Chemicals. ChemCatChem. 2014, 6, 2784–2791. [Google Scholar] [CrossRef]
- Tao, F.F. Synthesis, catalysis, surface chemistry and structure of bimetallic nanocatalysts. Chem. Soc. Rev. 2012, 41, 7977–7979. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Qin, R.; Fu, Q.; Zheng, N. Surface Coordination Chemistry of Metal Nanomaterials. J. Am. Chem. Soc. 2017, 139, 2122–2131. [Google Scholar] [CrossRef] [PubMed]
- Zaleska-Medynska, A.; Marchelek, M.; Diak, M.; Grabowska, E. Noble metal-based bimetallic nanoparticles: The effect of the structure on the optical, catalytic and photocatalytic properties. Adv. Colloid Interface 2016, 229, 80–107. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Rodionov, V.O. Controllable Catalysis with Nanoparticles: Bimetallic Alloy Systems and Surface Adsorbates. ACS Catal. 2016, 6, 4025–4033. [Google Scholar] [CrossRef]
- Gu, J.; Zhang, Y.-W.; Tao, F.F. Shape control of bimetallic nanocatalysts through well-designed colloidal chemistry approaches. Chem. Soc. Rev. 2012, 41, 8050–8065. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Li, J.; Li, Y. A review of plasma–liquid interactions for nanomaterial synthesis. J. Phys. D Appl. Phys. 2015, 48, 424005–424031. [Google Scholar] [CrossRef]
- Weiner, R.G.; Kunz, M.R.; Skrabalak, S.E. Seeding a New Kind of Garden: Synthesis of Architecturally Defined Multimetallic Nanostructures by Seed-Mediated Co-Reduction. Acc. Chem. Res. 2015, 48, 2688–2695. [Google Scholar] [CrossRef] [PubMed]
- Shukla, A.K.; Iravami, S. Metallic nanoparticles: Green synthesis and spectroscopic characterization. Environ. Chem. Lett. 2017, 15, 223–231. [Google Scholar] [CrossRef]
- De, S.; Dutta, S.; Sha, B. Critical design of heterogeneous catalysts for biomass valorization: Current thrust and emerging prospects. Catal. Sci. Technol. 2016, 6, 7364–7385. [Google Scholar] [CrossRef]
- Kim, D.; Xie, C.; Becknell, N.; Yu, Y.; Karamad, M.; Chan, K.; Crumlin, E.J.; Nørskov, J.K.; Yang, P. Electrochemical Activation of CO2 through Atomic Ordering Transformations of Au-Cu Nanoparticles. J. Am. Chem. Soc. 2017. [Google Scholar] [CrossRef] [PubMed]
- Noël, S.; Léger, B.; Ponchel, A.; Philippot, K.; Denicourt-Nowicki, A.; Roucoux, A.; Monflier, E. Cyclodextrin-based systems for the stabilization of metallic(0) nanoparticles and their versatile applications in catalysis. Catal. Today 2014, 235, 20–32. [Google Scholar] [CrossRef]
- Denicourt-Nowicki, A.; Roucoux, A. Metallic nanoparticles in neat water for catalytic applications. In Nanomaterials in Catalysis; Serp, P., Philippot, K., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2013; Chapter 2; pp. 55–95. [Google Scholar] [CrossRef]
- Yang, J.; Lee, J.Y.; Ying, J.Y. Phase transfer and its applications in nanotechnology. Chem. Soc. Rev. 2011, 40, 1672–1696. [Google Scholar] [CrossRef] [PubMed]
- Rai, R.K.; Gupta, K.; Behrens, S.; Li, J.; Xu, Q.; Singh, S.K. Highly active bimetallic nickel-palladium alloy nanoparticle catalyzed Suzuki-Miyaura reactions. ChemCatChem 2015, 7, 1806–1812. [Google Scholar] [CrossRef]
- Gupta, K.; Rai, R.K.; Dwivedi, A.D.; Singh, S.K. Catalytic aerial oxidation of biomass-derived furans to furan carboxylic acids in water over bimetallic Ni-Pd alloy nanoparticle. ChemCatChem 2016, 8, 1–9. [Google Scholar] [CrossRef]
- Rai, R.K.; Gupta, K.; Tyagi, D.; Mahata, A.; Behrens, S.; Yang, X.; Xu, Q.; Pathakad, B.; Singh, S.K. Access to highly active Ni-Pd bimetallic nanoparticle catalysts for C-C coupling reactions. Catal. Sci. Technol. 2016, 6, 5567–5579. [Google Scholar] [CrossRef]
- Zhang, Q.; Wu, X.-P.; Zhao, G.; Li, Y.; Wang, C.; Liu, Y.; Gong, X.-Q.; Lu, Y. High-performance PdNi alloy structured in situ on monolithic metal foam for coalbed methane deoxygenation via catalytic combustion. Chem. Commun. 2015, 51, 12613–12616. [Google Scholar] [CrossRef] [PubMed]
- Harikumar, K.R.; Ghosh, S.; Rao, C.N.R. X-ray Photoelectron Spectroscopic Investigations of Cu-Ni, Au-Ag, Ni-Pd, and Cu-Pd Bimetallic Clusters. J. Phys. Chem. A 1997, 101, 536–540. [Google Scholar] [CrossRef]
- Zhu, N.-M.; Li, Y.; Zhang, F.-S. Catalytic dechlorination of polychlorinated biphenyls in subcritical water by Ni/Fe nanoparticles. Chem. Eng. J. 2011, 171, 919–925. [Google Scholar] [CrossRef]
- Cervantes, F.J.; Rodríguez-López, J.L.; Peña-Martínez, M.; Ascacio-Valdes, J.A.; Aguilar, C.N.; Bahena, D. Enhanced Reduction of P-Nitrophenol by a Methanogenic Consortium Promoted by Metallic Nanoparticles. Water Air Soil Poll. 2016, 227, 368/1–368/9. [Google Scholar] [CrossRef]
- Heshmatpour, F.; Abazari, R.; Balalaie, S. Preparation of monometallic (Pd, Ag) and bimetallic (Pd/Ag, Pd/Ni, Pd/Cu) nanoparticles via reversed micelles and their use in the Heck reaction. Tetrahedron 2012, 68, 3001–3011. [Google Scholar] [CrossRef]
- Heshmatpour, F.; Abazari, R. Formation of dispersed palladium–nickel bimetallic nanoparticles in microemulsions: Synthesis, characterization, and their use as efficient heterogeneous recyclable catalysts for the amination reactions of aryl chlorides under mild conditions. RSC Adv. 2014, 4, 55815–55826. [Google Scholar] [CrossRef]
- Singh, H.P.; Gupta, N.; Sharma, S.K.; Sharma, R.K. Synthesis of bimetallic Pt-Cu nanoparticles and their application in the reduction of rhodamine B. Colloids Sur. A 2013, 416, 43–50. [Google Scholar] [CrossRef]
- Meena Kumari, M.; Jacob, J.; Philip, D. Green synthesis and applications of Au-Ag bimetallic nanoparticles. Spectrochim. Acta Part A 2015, 137, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Nasrollahzadeh, M.; Sajadi, S.M.; Rostami-Vartoonia, A.; Khalaj, M. Journey on greener pathways: Use of Euphorbia condylocarpa M. bieb as reductant and stabilizer for green synthesis of Au/Pd bimetallic nanoparticles as reusable catalysts in the Suzuki and Heck coupling reactions in water. RSC Adv. 2014, 4, 43477–43484. [Google Scholar] [CrossRef]
- Wang, C.-B.; Zhang, W.-X. Synthesizing Nanoscale Iron Particles for Rapid and Complete Dechlorination of TCE and PCBs. Environ. Sci. Technol. 1997, 31, 2154–2156. [Google Scholar] [CrossRef]
- Nagpal, V.; Bokarea, A.D.; Chikate, R.C.; Rodec, C.V.; Paknikar, K.M. Reductive dechlorination of γ-hexachlorocyclohexane using Fe-Pd bimetallic nanoparticles. J. Hazard. Mater. 2010, 175, 680–687. [Google Scholar] [CrossRef] [PubMed]
- Shiha, Y.-H.; Chena, M.-Y.; Su, Y.-F. Pentachlorophenol reduction by Pd/Fe bimetallic nanoparticles: Effects of copper, nickel, and ferric cations. Appl. Catal. B-Environ. 2011, 105, 24–29. [Google Scholar] [CrossRef]
- Tee, Y.-H.; Bachas, L.; Bhattacharyya, D. Degradation of Trichloroethylene by Iron-Based Bimetallic Nanoparticles. J. Phys. Chem. C 2009, 113, 9454–9464. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Hu, S.; Baig, S.A.; Tang, J.; Xu, X. Catalytic dechlorination of Aroclor 1242 by Ni/Fe bimetallic nanoparticles. J. Colloid Interface Sci. 2012, 385, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Lia, J.-R.; Fua, M.-L.; Yuanc, B.; Cuia, H.-J.; Wang, Y.-F. Fabrication and evaluation of Au-Pd core-shell nanocomposites for dechlorination of diclofenac in water. Environ. Technol. 2015, 36, 1510–1518. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yamauchi, Y. Strategic Synthesis of Trimetallic Au@Pd@Pt Core-Shell Nanoparticles from Poly(vinylpyrrolidone)-Based Aqueous Solution towards Highly Active Electrocatalysts. Chem. Mater. 2011, 23, 2457–2465. [Google Scholar] [CrossRef]
- Balcha, T.; Strobl, J.R.; Fowler, C.; Dash, P.; Scott, R.W.J. Selective Aerobic Oxidation of Crotyl Alcohol Using AuPd Core-Shell Nanoparticles. ACS Catal. 2011, 1, 425–436. [Google Scholar] [CrossRef]
- Maclennan, A.; Banerjee, A.; Scott, R.W.J. Aerobic oxidation of α,β-unsaturated alcohols using sequentially-grown AuPd nanoparticles in water and tetraalkylphosphonium ionic liquids. Catal. Today 2013, 207, 170–179. [Google Scholar] [CrossRef]
- Feng, F.; Ye, J.; Cheng, Z.; Xu, X.; Zhang, Q.; Ma, L.; Lu, C.; Li, X. Cu-Pd/γ-Al2O3 catalyzed the coupling of multi-step reactions: Direct synthesis of benzimidazole derivatives. RSC Adv. 2016, 6, 72750–72755. [Google Scholar] [CrossRef]
- Ge, Y.; Gao, T.; Wang, C.; Shah, Z.H.; Lu, R.; Zhang, S. Highly efficient silica coated CuNi bimetallic nanocatalyst from reverse microemulsion. J. Colloid Interface Sci. 2017, 491, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Kadu, B.S.; Hengne, A.M.; Biradar, N.S.; Rode, C.V.; Chikate, R.C. Reductive Cyclization of Levulinic Acid to γ-Valerolactone over Non-Noble Bimetallic Nanocomposite. Ind. Eng. Chem. Res. 2016, 55, 13032–13039. [Google Scholar] [CrossRef]
- Darabdhara, G.; Boruah, P.K.; Borthakur, P.; Hussain, N.; Das, M.R.; Ahamad, T.; Alshehri, S.M.; Malgras, V.; Wu, K.C.-W.; Yamauchid, Y. Reduced graphene oxide nanosheets decorated with Au-Pd bimetallic alloy nanoparticles towards efficient photocatalytic degradation of phenolic compounds in water. Nanoscale 2016, 8, 8276–8287. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Long, C.; Li, A. Selective reduction of nitrate into nitrogen using Fe-Pd bimetallic nanoparticle supported on chelating resin at near-neutral pH. Chem. Eng. J. 2016, 286, 408–415. [Google Scholar] [CrossRef]
- Ma, Y.; Wu, X.; Zhang, G. Core-shell Ag@Pt nanoparticles supported on sepiolite nanofibers for the catalytic reduction of nitrophenols in water: Enhanced catalytic performance and DFT study. Appl. Catal. B-Environ. 2017, 205, 262–270. [Google Scholar] [CrossRef]
- Wang, X.; Wang, Y.-X.; Yuan, B.; Cuia, H.-J.; Fu, M.L. Fabrication of resin supported Au-Pd bimetallic nanoparticle composite to efficiently remove chloramphenicol from water. RSC Adv. 2015, 5, 18806–18812. [Google Scholar] [CrossRef]
- Purushothaman, R.K.P.; Haveren, J.V.; Mayoral, A.; Melia’n-Cabrera, I.; Heeres, H.J. Exploratory Catalyst Screening Studies on the Base Free Conversion of Glycerol to Lactic Acid and Glyceric Acid in Water Using Bimetallic Au-Pt Nanoparticles on Acidic Zeolites. Top. Catal. 2014, 57, 1445–1453. [Google Scholar] [CrossRef]
- Pryadchenko, V.V.; Srabionyan, V.V.; Kurzin, A.A.; Bulat, N.V.; Shemet, D.B.; Avakyan, L.A.; Belenov, S.V.; Volochaev, V.A.; Zizak, I.; Guterman, V.E.; et al. Bimetallic PtCu core-shell nanoparticles in PtCu/C electrocatalysts: Structural and electrochemical characterization. Appl. Catal. A-Gen. 2016, 525, 226–236. [Google Scholar] [CrossRef]
- Fiévet, F.; Brayner, R. The Polyol Process. In Nanomaterials: A Danger or a Promise? Brayner, R., Fiévet, F., Coradin, T., Eds.; Springer: London, UK, 2013; Chapter 1; pp. 1–25. [Google Scholar] [CrossRef]
- Biacchi, A.J.; Schaak, R.E. The Solvent Matters: Kinetic versus Thermodynamic Shape Control in the Polyol Synthesis of Rhodium Nanoparticles. ACS Nano 2011, 5, 8089–8099. [Google Scholar] [CrossRef] [PubMed]
- Chadoura, F.; Pradel, C.; Gómez, M. Palladium Nanoparticles in Glycerol: A Versatile Catalytic System for C-X Bond Formation and Hydrogenation Process. Adv. Synth. Catal. 2013, 355, 3648–3660. [Google Scholar] [CrossRef]
- Chadoura, F.; Pradel, C.; Gómez, M. Copper(I) Oxide Nanoparticles in Glycerol: A Convenient Catalyst for Cross-Coupling and Azide-Alkyne Cycloaddition Processes. ChemCatChem 2014, 6, 2929–2936. [Google Scholar] [CrossRef]
- Reina, A.; Pradel, C.; Martin, E.; Teuma, E.; Gómez, M. Palladium nanoparticles stabilised by cinchona based alkaloids in glycerol: Efficient catalysts for surface assisted processes. RSC Adv. 2016, 6, 93205–93216. [Google Scholar] [CrossRef]
- Dang-Bao, T.; Pradel, C.; Favier, I.; Gómez, M. Making Copper(0) Nanoparticles in Glycerol: A Straightforward Synthesis for a Multipurpose Catalyst. Adv. Synth. Catal. 2017. [Google Scholar] [CrossRef]
- Biswas, M.; Saha, A.; Dule, M.; Mandal, T.K. Polymer-Assisted Chain-like Organization of CuNi Alloy Nanoparticles: Solvent-Adoptable Pseudohomogeneous Catalysts for Alkyne-Azide Click Reactions with Magnetic Recyclability. J. Phys. Chem. C 2014, 118, 22156–22165. [Google Scholar] [CrossRef]
- Rakap, M. Hydrogen generation from hydrolysis of ammonia borane in the presence of highly efficient poly(N-vinyl-2-pyrrolidone)-protected platinum-ruthenium nanoparticles. Appl. Catal. A-Gen. 2014, 478, 15–20. [Google Scholar] [CrossRef]
- Vu, K.B.; Bukhryakov, K.V.; Anjum, D.H.; Rodionov, V.O. Surface-Bound Ligands Modulate Chemoselectivity and Activity of a Bimetallic Nanoparticle Catalyst. ACS Catal. 2015, 5, 2529–2533. [Google Scholar] [CrossRef]
- Dabiri, M.; Vajargahy, M.P. PdCo bimetallic nanoparticles supported on three-dimensional graphene as a highly active catalyst for Sonogashira cross-coupling reaction. Appl. Organometal. Chem. 2017, 31, 3594. [Google Scholar] [CrossRef]
- Jia, L.; Yu, J.; Chen, Y.; Ning, P.; Guan, Q.; Gu, J.; Miao, R.; Chen, Q. Noble-metal-free bimetallic alloy nanoparticle-catalytic gasification of phenol in supercritical water. J. Supercrit. Fluids 2017, 126, 79–88. [Google Scholar] [CrossRef]
- Shen, Y.; Lua, A.C. Polyol synthesis of nickel-copper based catalysts for hydrogen production by methane decomposition. Int. J. Hydrogen Energy 2015, 40, 311–321. [Google Scholar] [CrossRef]
- Shen, Y.; Lua, A.C. Synthesis of Ni and Ni-Cu supported on carbon nanotubes for hydrogen and carbon production by catalytic decomposition of methane. Appl. Catal. B-Environ. 2015, 164, 61–69. [Google Scholar] [CrossRef]
- Hallett, J.P.; Welton, T. Room-Temperature Ionic Liquids: Solvents for Synthesis and Catalysis. 2. Chem. Rev. 2011, 111, 3508–3576. [Google Scholar] [CrossRef] [PubMed]
- Dupont, J. From Molten Salts to Ionic Liquids: A “Nano” Journey. Acc. Chem. Res. 2011, 44, 1223–1231. [Google Scholar] [CrossRef] [PubMed]
- Prechtl, M.H.G.; Campbell, P.S. Metal oxide and bimetallic nanoparticles in ionic liquids: Synthesis and application in multiphase catalysis. Nanotechnol. Rev. 2013, 2, 577–595. [Google Scholar] [CrossRef]
- Scholten, J.D.; Leal, B.C.; Dupont, J. Transition Metal Nanoparticle Catalysis in Ionic Liquids. ACS Catal. 2012, 2, 184–200. [Google Scholar] [CrossRef]
- Janiak, C. Metal Nanoparticle Synthesis in Ionic Liquids. Topics Organomet. Chem. 2015, 51, 17–53. [Google Scholar] [CrossRef]
- Wegner, S.; Saito, M.; Barthel, J.; Janiak, C. Soft wet-chemical synthesis of Ru-Sn nanoparticles from single-source ruthenocene-stannole precursors in an ionic liquid. J. Organomet. Chem. 2016, 821, 192–196. [Google Scholar] [CrossRef]
- Suzuki, S.; Tomita, Y.; Kuwabata, S.; Torimoto, T. Synthesis of alloy AuCu nanoparticles with the L10 structure in an ionic liquid using sputter deposition. Dalton Trans. 2015, 44, 4186–4194. [Google Scholar] [CrossRef] [PubMed]
- Helgadottir, I.S.; Arquilliere, P.P.; Brea, P.; Santini, C.C.; Haumesser, P.-H.; Richter, K.; Mudring, A.-V.; Aouine, M. Synthesis of bimetallic nanoparticles in ionic liquids: Chemical routes vs. physical vapor deposition. Microelectron. Eng. 2013, 107, 229–232. [Google Scholar] [CrossRef]
- Pusch, J.M.E.; Brondani, D.; Luza, L.; Dupont, J.; Vieira, I.C. Pt-Pd bimetallic nanoparticles dispersed in an ionic liquid and peroxidase immobilized on nanoclay applied in the development of a biosensor. Analyst 2013, 138, 4898–4906. [Google Scholar] [CrossRef] [PubMed]
- Tsai, T.-H.; Thiagarajan, S.; Chen, S.-M. Ionic liquid assisted one step green synthesis of Au-Ag bimetallic nanoparticles. J. Appl. Electrochem. 2010, 40, 493–497. [Google Scholar] [CrossRef]
- Favier, I.; Teuma, E.; Gómez, M. Bimetallic Nanoparticles in Ionic Liquids: Synthesis and Catalytic Applications. In Nanocatalysis in Ionic Liquids; Prechtl, M.H.G., Ed.; John Wiley and Sons Ltd. Wiley-VCH Verlag GmbH: Hoboken, NJ, USA, 2016; Chapter 7; pp. 125–146. [Google Scholar]
- Yuan, X.; Sun, G.; Asakura, H.; Tanaka, T.; Chen, X.; Yuan, Y.; Laurenczy, G.; Kou, Y.; Dyson, P.J.; Yan, N. Development of Palladium Surface-Enriched Heteronuclear Au-Pd Nanoparticle Dehalogenation Catalysts in an Ionic Liquid. Chem. Eur. J. 2013, 19, 1227–1234. [Google Scholar] [CrossRef] [PubMed]
- Scott, R.W.J.; Dash, P. Bimetallic Nanoparticles as Catalysts in Imidazolium Ionic Liquids. In Ionic Liquids in Materials Synthesis and Application; Baker, G.A., Wilkes, J.S., Yang, H., Eds.; Materials Research Society Symposia Proceedings: Warrendale, PA, USA, 2008; Volume 1082. [Google Scholar]
- Dash, P.; Dehm, N.A.; Scott, R.W.J. Bimetallic PdAu nanoparticles as hydrogenation catalysts in imidazolium ionic liquids. J. Mol. Catal. A Chem. 2008, 286, 114–119. [Google Scholar] [CrossRef]
- Dash, P.; Miller, S.M.; Scott, R.W.J. Stabilizing nanoparticle catalysts in imidazolium-based ionic liquids: A comparative study. J. Mol. Catal. A Chem. 2010, 329, 86–95. [Google Scholar] [CrossRef]
- Suzuki, S.; Suzuki, T.; Tomita, Y.; Hirano, M.; Okazaki, K.I.; Kuwabata, S.; Torimoto, T. Compositional control of AuPt nanoparticles synthesized in ionic liquids by the sputter deposition technique. CrystEngComm 2012, 14, 4922–4926. [Google Scholar] [CrossRef]
- Hirano, M.; Enokida, K.; Okazaki, K.I.; Kuwabata, S.; Yoshida, H.; Torimoto, T. Composition-dependent electrocatalytic activity of AuPd alloy nanoparticles prepared via simultaneous sputter deposition into an ionic liquid. Phys. Chem. Chem. Phys. 2013, 15, 7286–7294. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.H.; Liu, J.; Zhou, Y.Y.; Cai, X.L.; Lu, Y.; Gao, X.; Wang, S.D. Small and uniform Pd monometallic/bimetallic nanoparticles decorated on multi-walled carbon nanotubes for efficient reduction of 4-nitrophenol. Carbon 2015, 94, 295–300. [Google Scholar] [CrossRef]
- Ding, K.; Liu, L.; Cao, Y.; Yan, X.; Wei, H.; Guo, Z. Formic acid oxidation reaction on a PdxNiy bimetallic nanoparticle catalyst prepared by a thermal decomposition process using ionic liquids as the solvent. Int. J. Hydrogen Energy 2014, 39, 7326–7337. [Google Scholar] [CrossRef]
- Ding, K.; Yang, H.; Cao, Y.; Zheng, C.; Rapole, S.B.; Guo, Z. Using ionic liquid as the solvent to prepare Pd–Ni bimetallic nanoparticles by a pyrolysis method for ethanol oxidation reaction. Mater. Chem. Phys. 2013, 142, 403–411. [Google Scholar] [CrossRef]
- Dietrich, C.; Schild, D.; Wang, W.; Kübel, C.; Behrens, S.Z. Bimetallic Pt/Sn-based Nanoparticles in Ionic Liquids as Nanocatalysts for the Selective Hydrogenation of Cinnamaldehyde. Z. Anorg. Allg. Chem. 2017, 643, 120–129. [Google Scholar] [CrossRef]
- Arquillière, P.P.; Helgadottir, I.S.; Santini, C.C.; Haumesser, P.H.; Aouine, M.; Massin, L.; Rousset, J.L. Bimetallic Ru-Cu Nanoparticles Synthesized in Ionic Liquids: Kinetically Controlled Size and Structure. Top. Catal. 2013, 56, 1192–1198. [Google Scholar] [CrossRef]
- Andanson, J.M.; Marx, S.; Baiker, A. Selective hydrogenation of cyclohexenone on iron-ruthenium nano-particles suspended in ionic liquids and CO2-expanded ionic liquids. Catal. Sci. Technol. 2012, 2, 1403–1409. [Google Scholar] [CrossRef]
- Laurenczy, G.; Dyson, P.J. Determination of the Viscosity of the Ionic Liquids [bmim][PF6] and [bmim][TF2N] Under High CO2 Gas Pressure Using Sapphire NMR Tubes. Z. Naturforsch. B Chem. Sci. 2008, 63, 681–684. [Google Scholar] [CrossRef]
- Solinas, M.; Pfaltz, A.; Cozzi, P.G.; Leitner, W. Enantioselective Hydrogenation of Imines in Ionic Liquid/Carbon Dioxide Media. J. Am. Chem. Soc. 2004, 126, 16142–16147. [Google Scholar] [CrossRef] [PubMed]
- Schütte, K.; Meyer, H.; Gemel, C.; Barthel, J.; Fischer, R.A.; Janiak, C. Synthesis of Cu, Zn and Cu/Zn brass alloy nanoparticles from metal amidinate precursors in ionic liquids or propylene carbonate with relevance to methanol synthesis. Nanoscale 2014, 6, 3116–3126. [Google Scholar] [CrossRef] [PubMed]
- Schütte, K.; Doddi, A.; Kroll, C.; Meyer, H.; Wiktor, C.; Gemel, C.; van Tendeloo, G.; Fischer, R.A.; Janiak, C. Colloidal nickel/gallium nanoalloys obtained from organometallic precursors in conventional organic solvents and in ionic liquids: Noble-metal-free alkyne semihydrogenation catalysts. Nanoscale 2014, 6, 5532–5544. [Google Scholar] [CrossRef] [PubMed]
- Weilhard, A.; Abarca, G.; Viscardi, J.; Prechtl, M.H.G.; Scholten, J.D.; Bernardi, F.; Baptista, D.L.; Dupont, J. Challenging Thermodynamics: Hydrogenation of Benzene to 1,3-Cyclohexadiene by Ru@Pt Nanoparticles. ChemCatChem 2017, 9, 204–221. [Google Scholar] [CrossRef]
- Chang, J.B.; Liu, C.H.; Liu, J.; Zhou, Y.Y.; Gao, X.; Wang, S.D. Green-chemistry Compatible Approach to TiO2-supported PdAu Bimetallic Nanoparticles for Solvent-free 1-Phenylethanol Oxidation under Mild Conditions. Nano-Micro Lett. 2015, 7, 307–315. [Google Scholar] [CrossRef]
- Liu, C.H.; Chen, X.Q.; Hu, Y.F.; Sham, T.K.; Sun, Q.J.; Chang, J.B.; Gao, X.; Sun, X.H.; Wang, S.D. One-Pot Environmentally Friendly Approach toward Highly Catalytically Active Bimetal-Nanoparticle-Graphene Hybrids. ACS Appl. Mater. Interfaces 2013, 5, 5072–5079. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.H.; Liu, R.H.; Sun, Q.J.; Chang, J.B.; Gao, X.; Liu, Y.; Lee, S.T.; Kang, Z.H.; Wang, S.D. Controlled synthesis and synergistic effects of graphene-supported PdAu bimetallic nanoparticles with tunable catalytic properties. Nanoscale 2015, 7, 6356–6362. [Google Scholar] [CrossRef] [PubMed]
- Alonso, D.A.; Baeza, A.; Chinchilla, R.; Guillena, G.; Pastor, I.M.; Ramon, D.J. Deep Eutectic Solvents: The Organic Reaction Medium of the Century. Eur. J. Org. Chem. 2016, 612–632. [Google Scholar] [CrossRef]
- Dai, Y.; van Spronsen, J.; Witkamp, G.-J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Hao, J.-W.; Mo, L.-P.; Zhang, Z.-H. Recent advances in the application of deep eutectic solvents as sustainable media as well as catalysts in organic reactions. RSC Adv. 2015, 5, 48675–48704. [Google Scholar] [CrossRef]
- Garcia-Alvarez, J. Deep Eutectic Mixtures: Promising Sustainable Solvents for Metal-Catalyzed and Metal-Mediated Organic Reactions. Eur. J. Inorg. Chem. 2015, 5147–5157. [Google Scholar] [CrossRef]
- Guajardo, N.; Mueller, C.R.; Schrebler, R.; Carlesi, C.; Dominguez de Maria, P. Deep Eutectic Solvents for Organocatalysis, Biotransformations, and Multistep Organocatalyst/Enzyme Combinations. ChemCatChem 2016, 8, 1020–1027. [Google Scholar] [CrossRef]
- Wagle, D.V.; Zhao, H.; Baker, G.A. Deep Eutectic Solvents: Sustainable Media for Nanoscale and Functional Materials. Acc. Chem. Res. 2014, 47, 2299–2308. [Google Scholar] [CrossRef] [PubMed]
- Abo-Hamad, A.; Hayyan, M.; AlSaadi, M.A.; Hashim, M.A. Potential applications of deep eutectic solvents in nanotechnology. Chem. Eng. J. 2015, 273, 551–567. [Google Scholar] [CrossRef]
- Oseguera-Galindo, D.O.; Machorro-Mejia, R.; Bogdanchikova, N.; Mota-Morales, J.D. Silver nanoparticles synthesized by laser ablation confined in urea choline chloride deep-eutectic solvent. Colloid Interface Sci. Commun. 2016, 12, 1–4. [Google Scholar] [CrossRef]
- Hammons, J.A.; Ustarroz, J.; Muselle, T.; Torriero, A.A.J.; Terryn, H.; Suthar, K.; Ilavsky, J. Supported Silver Nanoparticle and Near-Interface Solution Dynamics in a Deep Eutectic Solvent. J. Phys. Chem. C 2016, 120, 1534–1545. [Google Scholar] [CrossRef]
- Raghuwanshi, V.S.; Ochmann, M.; Hoell, A.; Polzer, F.; Rademann, K. Deep Eutectic Solvents for the Self-Assembly of Gold Nanoparticles: A SAXS, UV-Vis, and TEM Investigation. Langmuir 2014, 30, 6038–6046. [Google Scholar] [CrossRef] [PubMed]
- Karimi, M.; Eshraghi, M.J.; Jahangir, V. A facile and green synthetic approach based on deep eutectic solvents toward synthesis of CZTS nanoparticles. Mater. Lett. 2016, 171, 100–103. [Google Scholar] [CrossRef]
- Renjith, A.; Lakshminarayanan, V. One step preparation of ‘ready to use’ Au@Pd nanoparticle modified surface using deep eutectic solvents and a study of its electrocatalytic properties in methanol oxidation reaction. J. Mater. Chem. A 2015, 3, 3019–3028. [Google Scholar] [CrossRef]
- Wang, R.X.; Fan, Y.J.; Liang, Z.R.; Zhang, J.M.; Zhou, Z.Y.; Sun, S.G. PdSn nanocatalysts supported on carbon nanotubes synthesized in deep eutectic solvents with high activity for formic acid electrooxidation. RSC Adv. 2016, 6, 60400–60406. [Google Scholar] [CrossRef]
- Gage, S.H.; Ruddy, D.A.; Pylypenko, S.; Richards, R.M. Deep eutectic solvent approach towards nickel/nickel nitride nanocomposites. Catal. Today 2016. [Google Scholar] [CrossRef]
- Qiao, Y.; Said, N.; Rauser, M.; Yan, K.; Qin, F.; Theyssen, N.; Leitner, W. Preparation of SBA-15 supported Pt/Pd bimetallic catalysts using supercritical fluid reactive deposition: How do solvent effects during material synthesis affect catalytic properties? Green Chem. 2017, 19, 977–986. [Google Scholar] [CrossRef]
- Bozbag, S.E.; Erkey, C. Supercritical deposition: Current status and perspectives for the preparation of supported metal nanostructures. J. Supercrit. Fluids 2015, 96, 298–312. [Google Scholar] [CrossRef]
- Bozbag, S.E.; Kostenko, S.O.; Kurykin, M.; Khrustalev, V.N.; Khokhlov, A.R.; Zhang, L.; Aindow, M.; Erkey, C. Aerogel-copper nanocomposites prepared using the adsorption of a polyfluorinated complex from supercritical CO2. J. Nanopart. Res. 2012, 14, 973/1–973/13. [Google Scholar] [CrossRef]
- Bozbag, S.E.; Sanli, D.; Erkey, C. Synthesis of nanostructured materials using supercritical CO2: Part II. Chemical transformations. J. Mater. Sci. 2012, 47, 3469–3492. [Google Scholar] [CrossRef]
- Bozbag, S.E.; Unal, U.; Kurykin, M.A.; Ayala, C.J.; Aindow, M.; Erkey, C. Thermodynamic Control of Metal Loading and Composition of Carbon Aerogel Supported Pt-Cu Alloy Nanoparticles by Supercritical Deposition. J. Phys. Chem. C 2013, 117, 6777–6787. [Google Scholar] [CrossRef]
- Canguel, B.; Zhang, L.C.; Aindow, M.; Erkey, C. Preparation of carbon black supported Pd, Pt and Pd-Pt nanoparticles using supercritical CO2 deposition. J. Supercrit. Fluids 2009, 50, 82–90. [Google Scholar] [CrossRef]
- Wang, J.S.; Pan, H.-B.; Wai, C.M. Deposition of metal nanoparticles on carbon nanotubes via hexane modified water-in-CO2 microemulsion at room temperature. J. Nanosci. Nanotechnol. 2006, 6, 2025–2030. [Google Scholar] [CrossRef] [PubMed]
- Yen, C.H.; Shimizu, K.; Lin, Y.-Y.; Bailey, F.; Cheng, I.F.; Wai, C.M. Chemical Fluid Deposition of Pt-Based Bimetallic Nanoparticles on Multiwalled Carbon Nanotubes for Direct Methanol Fuel Cell Application. Energy Fuel. 2007, 21, 2268–2271. [Google Scholar] [CrossRef]
- Mueller, S.; Tuerk, M. Production of supported gold and gold-silver nanoparticles by supercritical fluid reactive deposition: Effect of substrate properties. J. Supercrit. Fluids 2015, 96, 287–297. [Google Scholar] [CrossRef]
- Hund, J.F.; Bertino, M.F.; Zhang, G.; Sotiriou-Leventis, C.; Leventis, N. Synthesis of homogeneous alloy metal nanoparticles in silica aerogels. J. Non-Cryst. Solids 2004, 350, 9–13. [Google Scholar] [CrossRef]
- Puniredd, S.R.; Weiyi, S.; Srinivasan, M.P. Pd-Pt and Fe-Ni nanoparticles formed by covalent molecular assembly in supercritical carbon dioxide. J. Colloid Interface Sci. 2008, 320, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Fu, L.; Liu, Z.; Han, B.; Liu, Y.; Du, J. Synthesis of noble metal/carbon nanotube composites in supercritical methanol. J. Nanosci. Nanotechnol. 2006, 6, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Prieto, A.; Palomino, M.; Díaz, U.; Corma, A. One-pot two-step process for direct propylene oxide production catalyzed by bi-functional Pd(Au)@TS-1 materials. Appl. Catal. A-Gen. 2016, 523, 73–84. [Google Scholar] [CrossRef]
- Zhai, P.; Sun, G.; Zhu, Q.; Ma, D. Fischer-Tropsch synthesis nanostructured catalysts: Understanding structural characteristics and catalytic reaction. Nanotechnol. Rev. 2013, 2, 547–576. [Google Scholar] [CrossRef]
- Casula, M.F.; Corrias, A.; Paschina, G. FeCo-SiO2 nanocomposite aerogels by high temperature supercritical drying. J. Mater. Chem. 2002, 12, 1505–1510. [Google Scholar] [CrossRef]
- Corrias, A.; Casula, M.F.; Falqui, A.; Paschina, G. Preparation and characterization of FeCo-Al2O3 and Al2O3 aerogels. J. Sol-Gel Sci. Technol. 2004, 31, 83–86. [Google Scholar] [CrossRef]
- Jacobs, G.; Chaudhari, K.; Sparks, D.; Zhang, Y.; Shi, B.; Spicer, R.; Das, T.K.; Li, J.; Davis, B.H. Fischer-Tropsch synthesis: Supercritical conversion by using a Co/Al2O3 catalyst in a fixed bed reactor. Fuel 2003, 82, 1251–1260. [Google Scholar] [CrossRef]
- Irankhah, A.; Haghtalab, A. Fischer-Tropsch synthesis over Co-Ru/γ-Al2O3 catalyst in supercritical media. Chem. Eng. Technol. 2008, 31, 525–536. [Google Scholar] [CrossRef]
- Deal, J.W.; Le, P.; Corey, C.B.; More, K.; West, C.W. Water-gas shift reaction on alumina-supported Pt-CeOx catalysts prepared by supercritical fluid deposition. J. Supercrit. Fluids 2017, 119, 113–121. [Google Scholar] [CrossRef]
- Yen, C.H.; Lin, H.W.; Phan, T.D.; Tan, C.-S. Chemical fluid deposition of monometallic and bimetallic nanoparticles on ordered mesoporous silica as hydrogenation catalysts. J. Nanosci. Nanotechnol. 2011, 11, 2465–2469. [Google Scholar] [CrossRef] [PubMed]
- Anand, M.; McLeod, M.C.; Bell, P.W.; Roberts, C.B. Tunable solvation effects on the size-selective fractionation of metal NPs in CO2 gas-expanded solvents. J. Phys. Chem. B 2005, 109, 22852–22859. [Google Scholar] [CrossRef] [PubMed]
- Castillejos, E.; Jahjah, M.; Favier, I.; Orejón, A.; Pradel, C.; Teuma, E.; Masdeu-Bultó, A.M.; Serp, P.; Gómez, M. Synthesis of Platinum-Ruthenium Nanoparticles under Supercritical CO2 and their Confinement in Carbon Nanotubes: Hydrogenation Applications. ChemCatChem 2012, 4, 118–122. [Google Scholar] [CrossRef]
- Yu, K.M.K.; Meric, P.; Tsang, S.C. Micelle-hosted bimetallic Pd-Ru nanoparticle for in situ catalytic hydrogenation in supercritical CO2. Catal. Today 2006, 114, 428–433. [Google Scholar] [CrossRef]
- Yu, W.; Hsu, Y.-P.; Tan, C.-S. Synthesis of rhodium-platinum bimetallic catalysts supported on SBA-15 by chemical fluid deposition for the hydrogenation of terephthalic acid in water. Appl. Catal. B-Environ. 2016, 196, 185–192. [Google Scholar] [CrossRef]
- Song, S.; Liu, R.; Zhang, Y.; Feng, J.; Liu, D.; Xing, Y.; Zhao, F.; Zhang, H. Colloidal Noble-Metal and Bimetallic Alloy Nanocrystals: A General Synthetic Method and Their Catalytic Hydrogenation Properties. Chem. Eur. J. 2010, 16, 6251–6256. [Google Scholar] [CrossRef] [PubMed]
- Scott, R.W.J.; Wilson, O.M.; Crooks, R.M. Synthesis, Characterization, and Applications of Dendrimer-Encapsulated Nanoparticles. J. Phys. Chem. B 2005, 109, 692–704. [Google Scholar] [CrossRef] [PubMed]
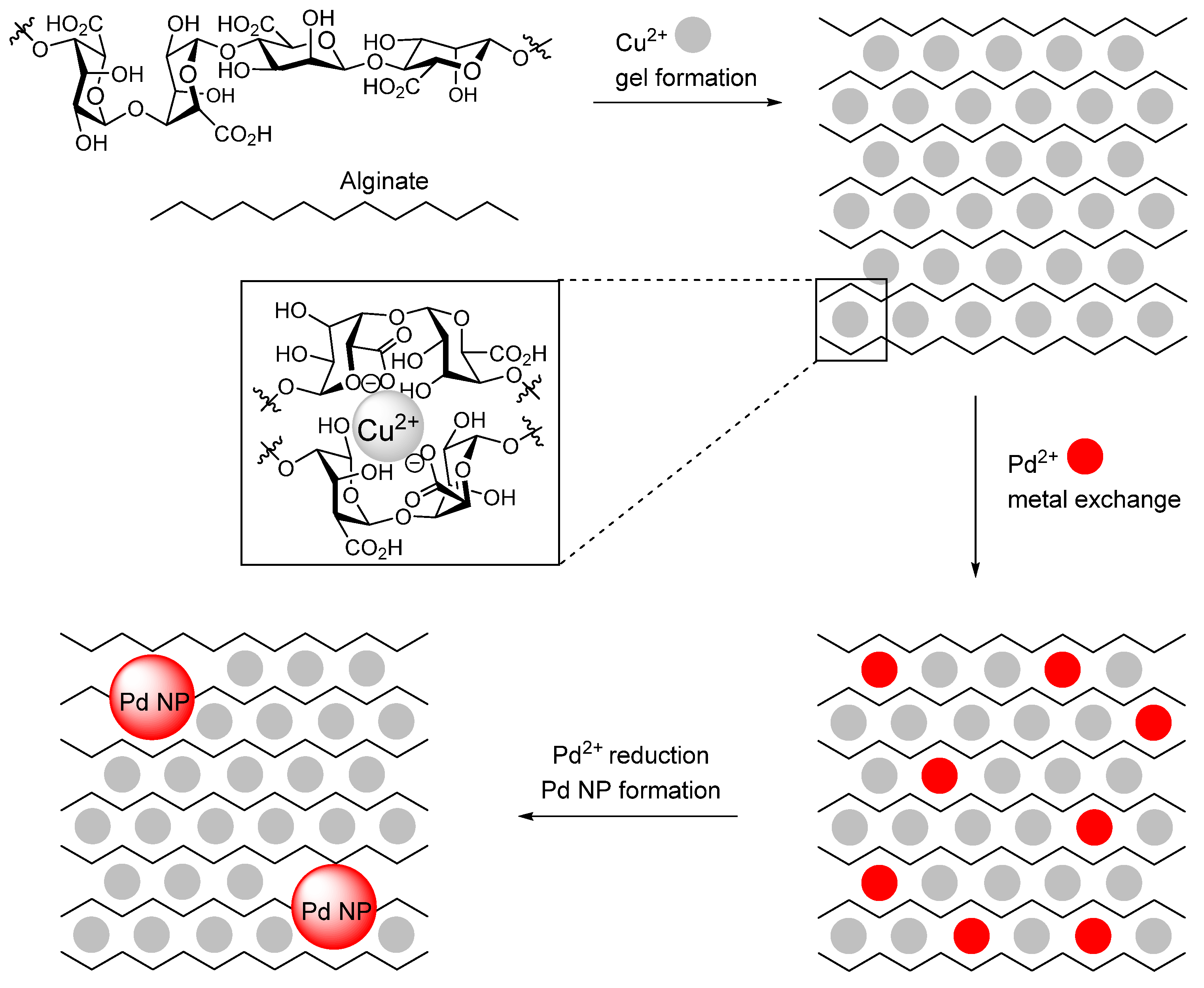
- Chtchigrovsky, M.; Lin, Y.; Ouchaou, K.; Chaumontet, M.; Robitzer, M.; Quignard, F.; Taran, F. Dramatic Effect of the Gelling Cation on the Catalytic Performances of Alginate-Supported Palladium Nanoparticles for the Suzuki-Miyaura Reaction. Chem. Mater. 2012, 24, 1505–1510. [Google Scholar] [CrossRef]
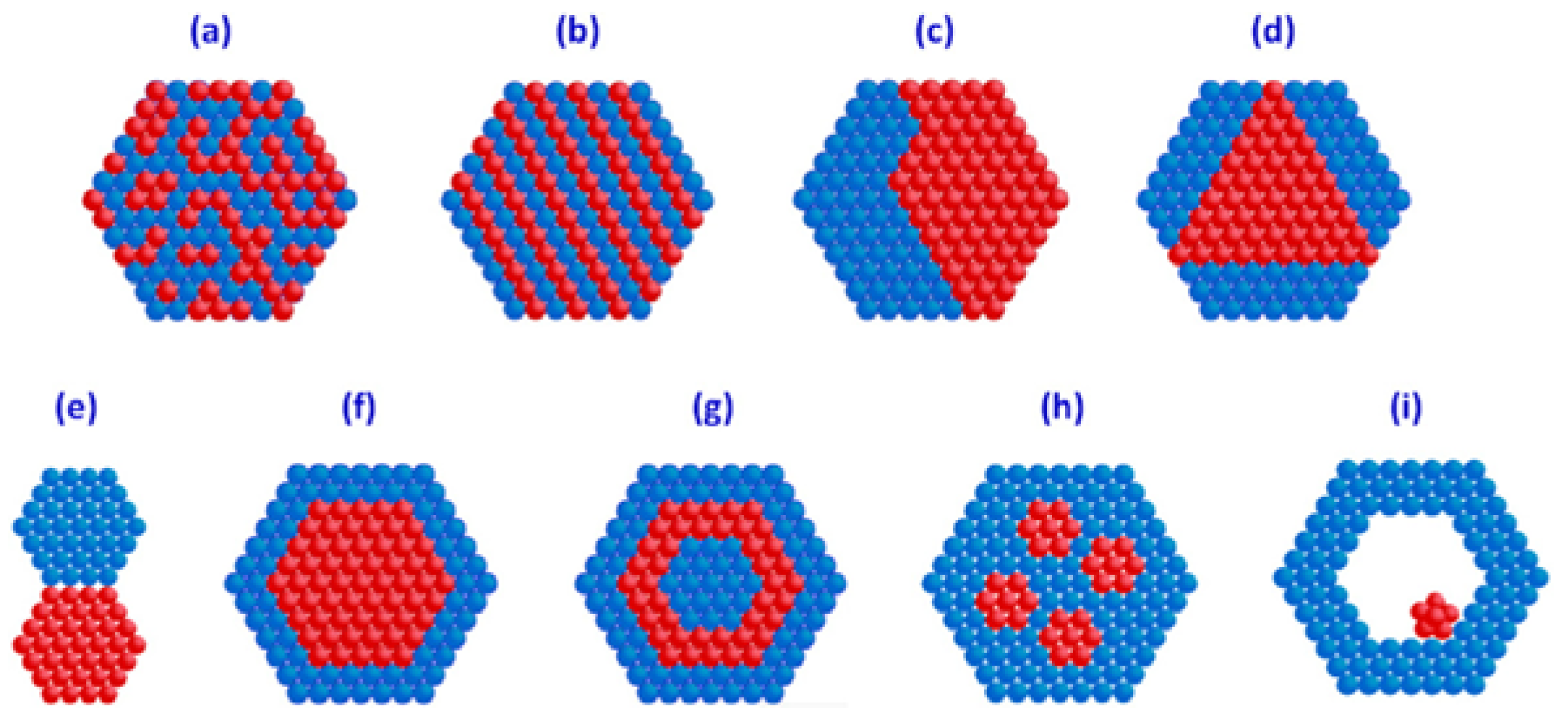
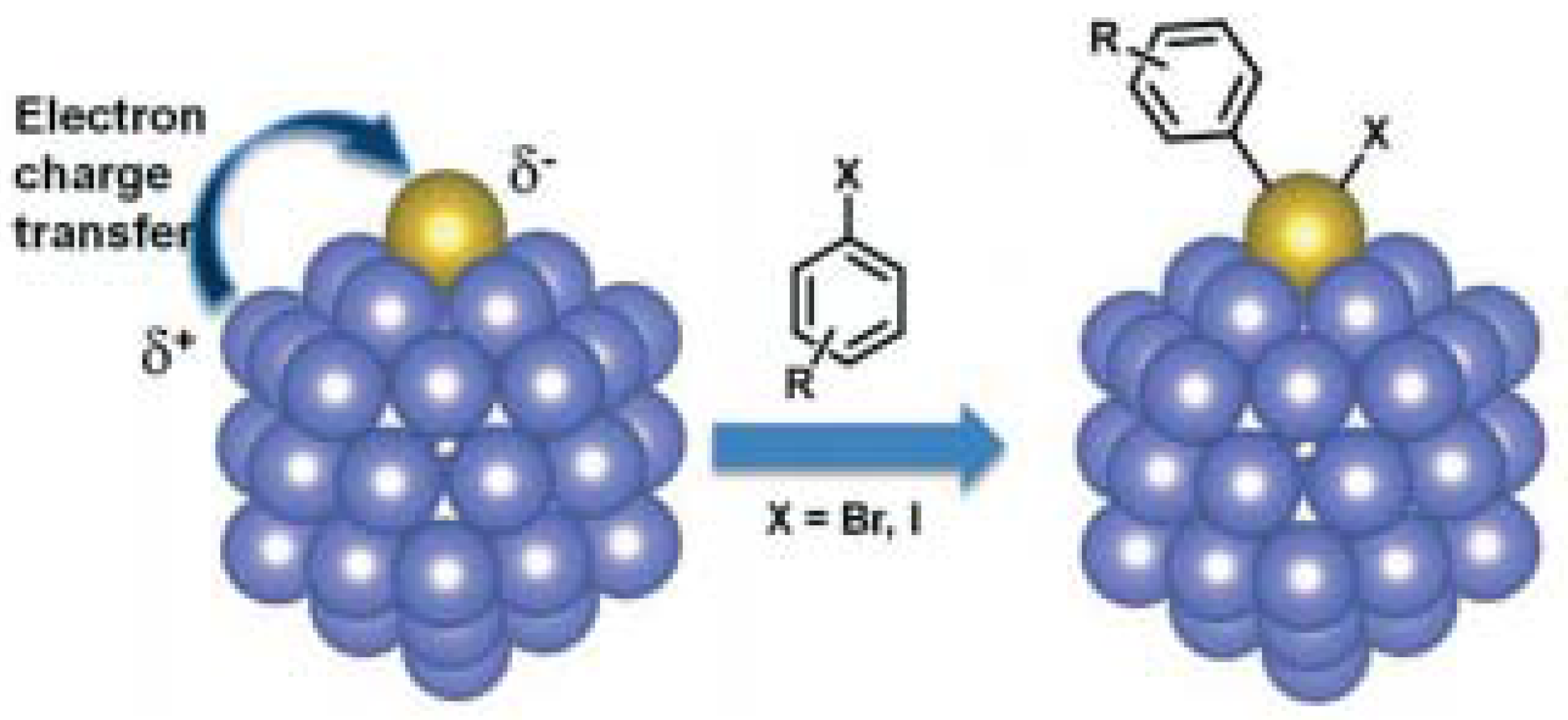
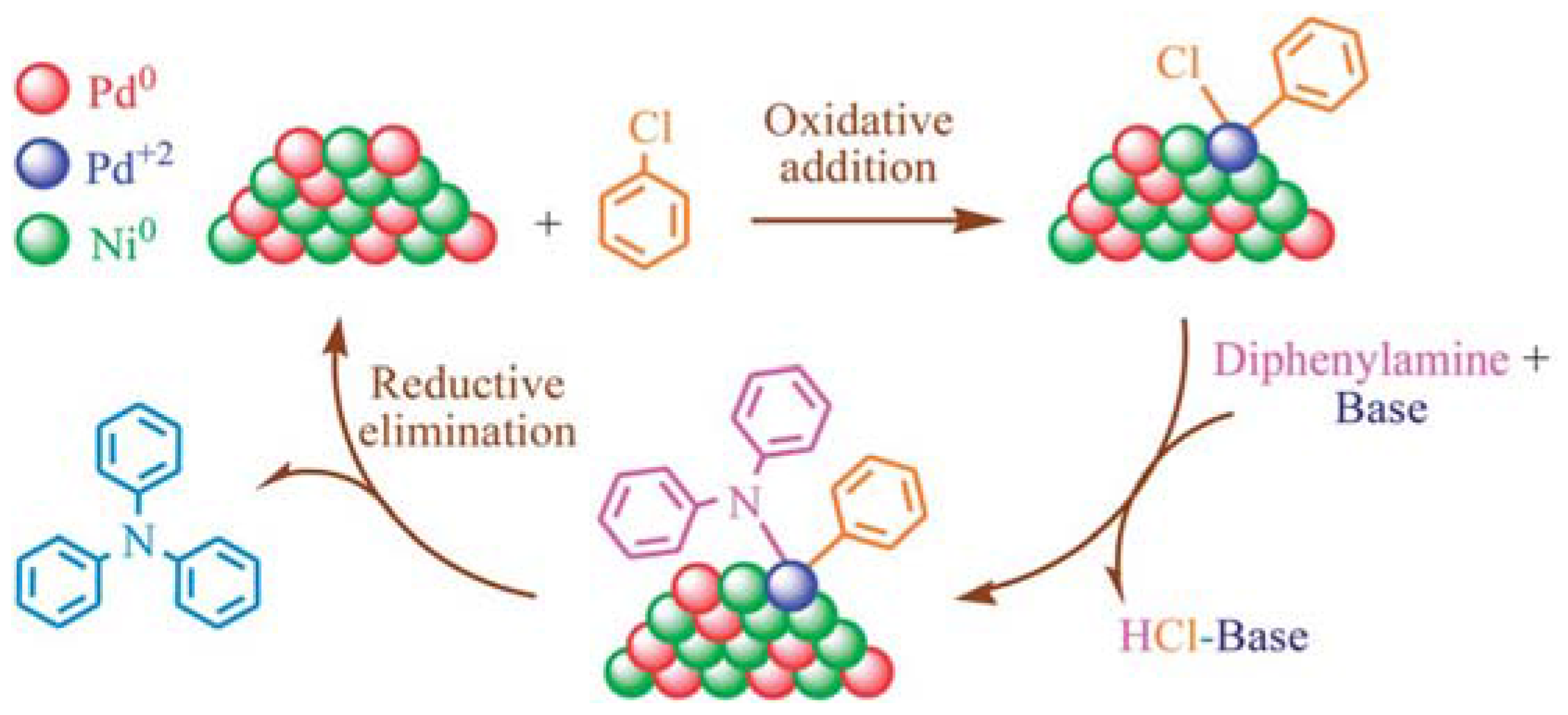
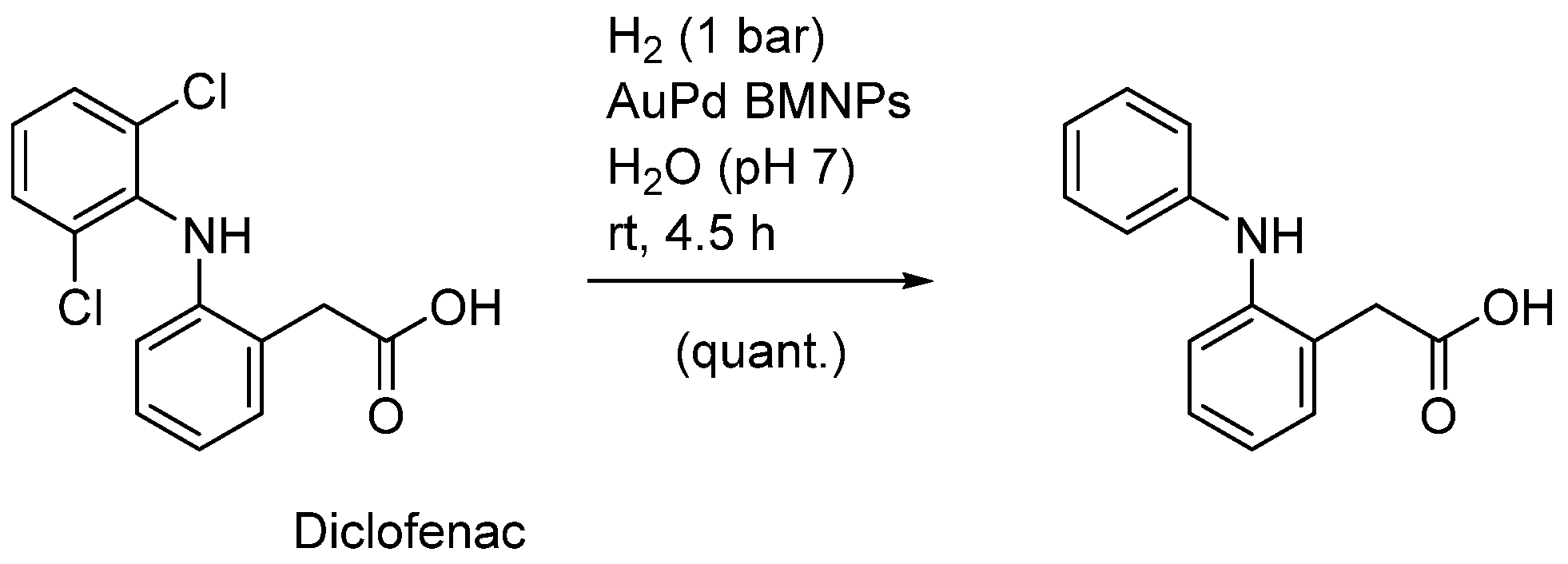
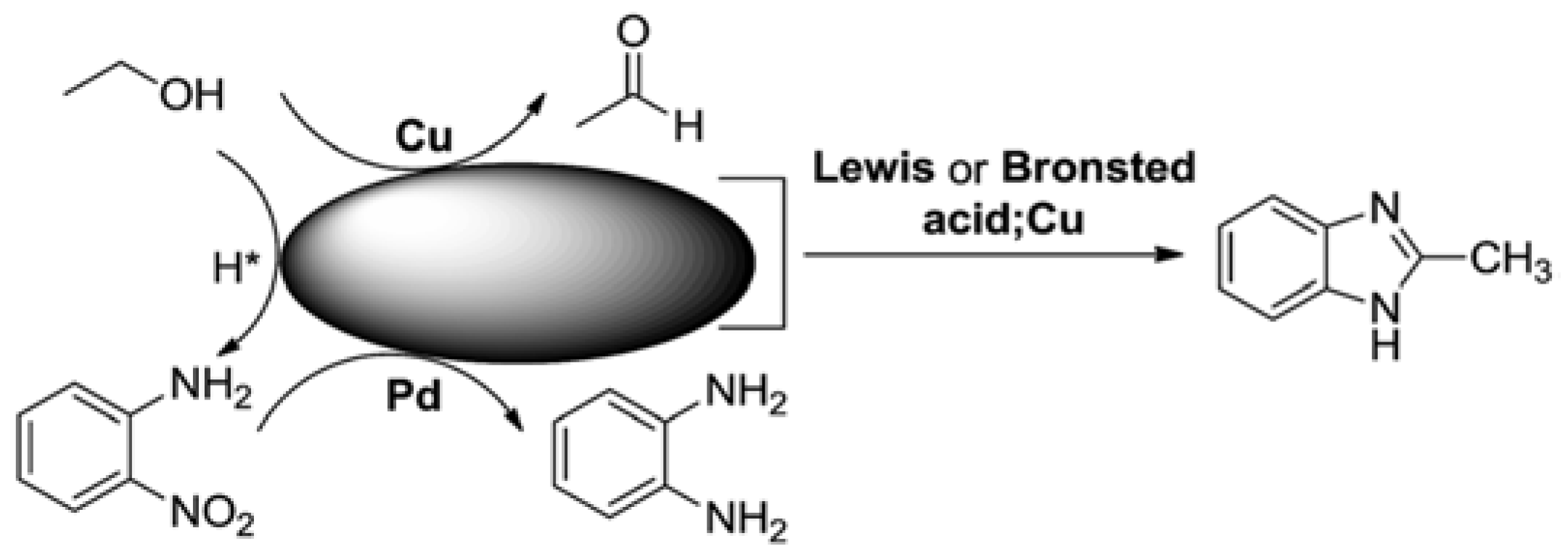
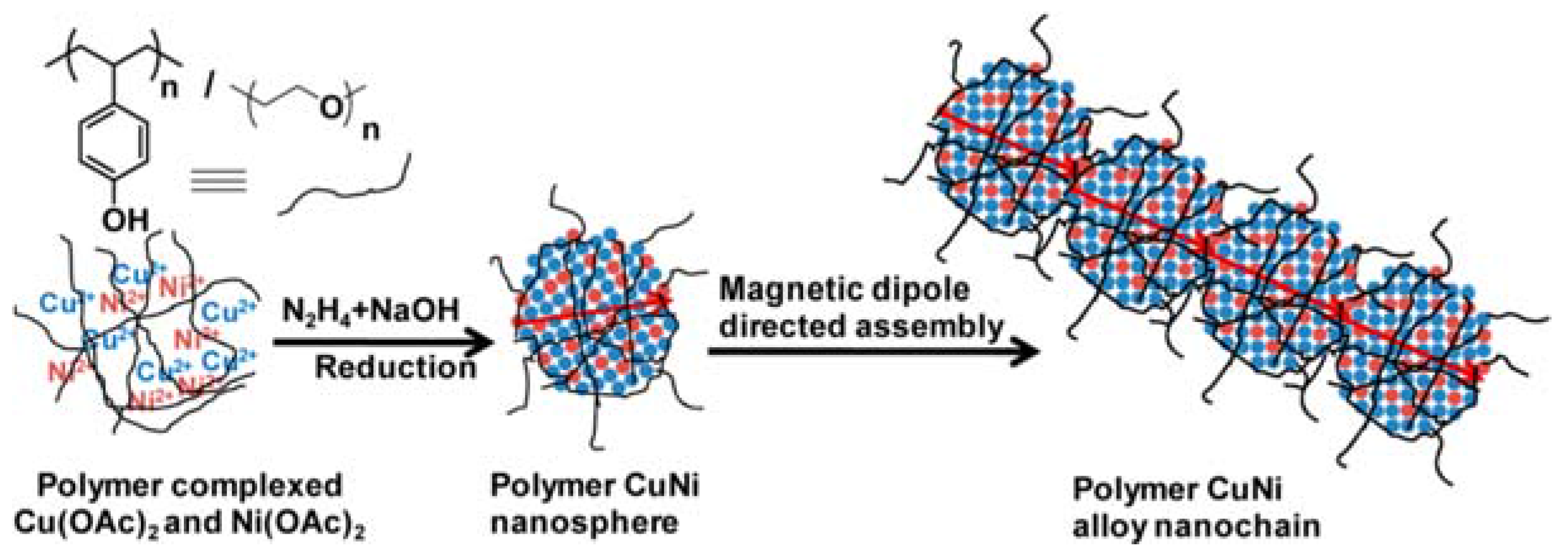

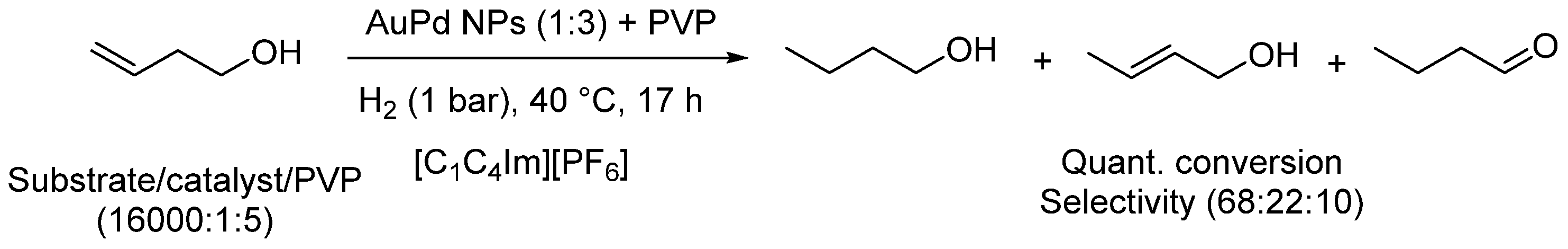
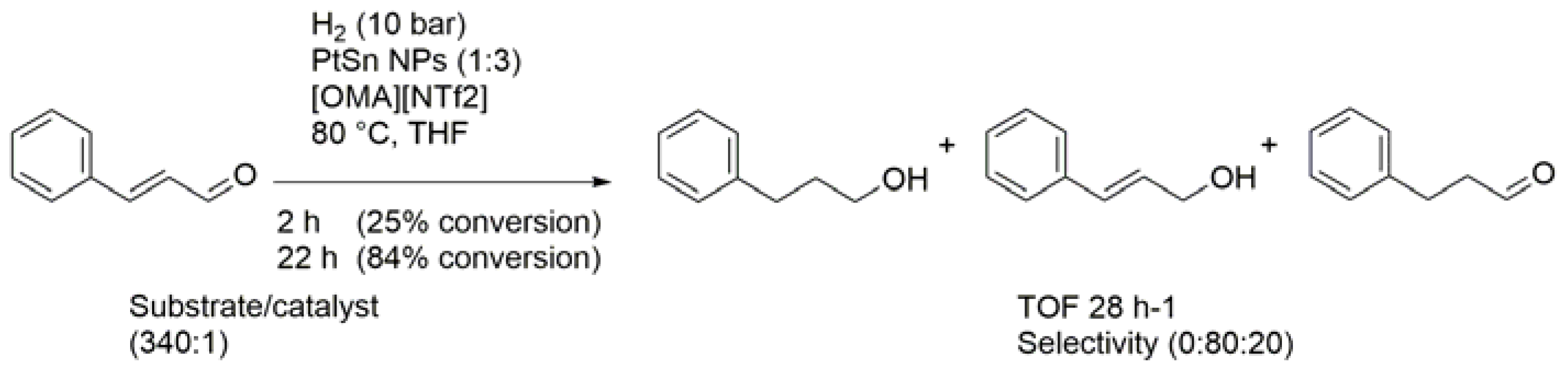

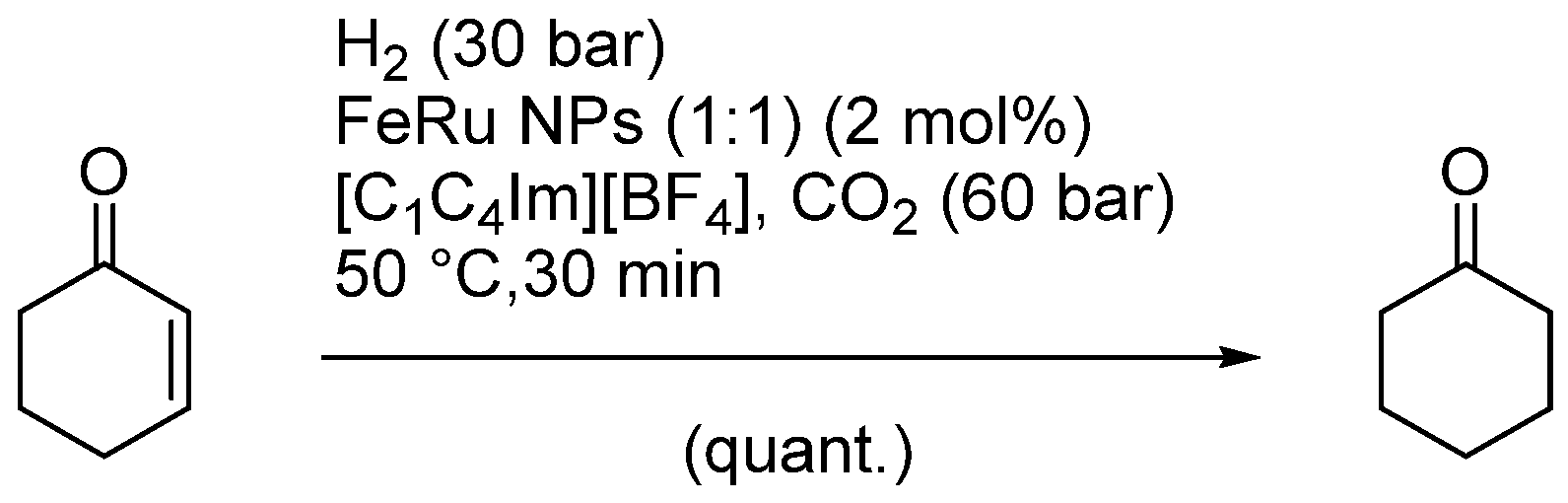
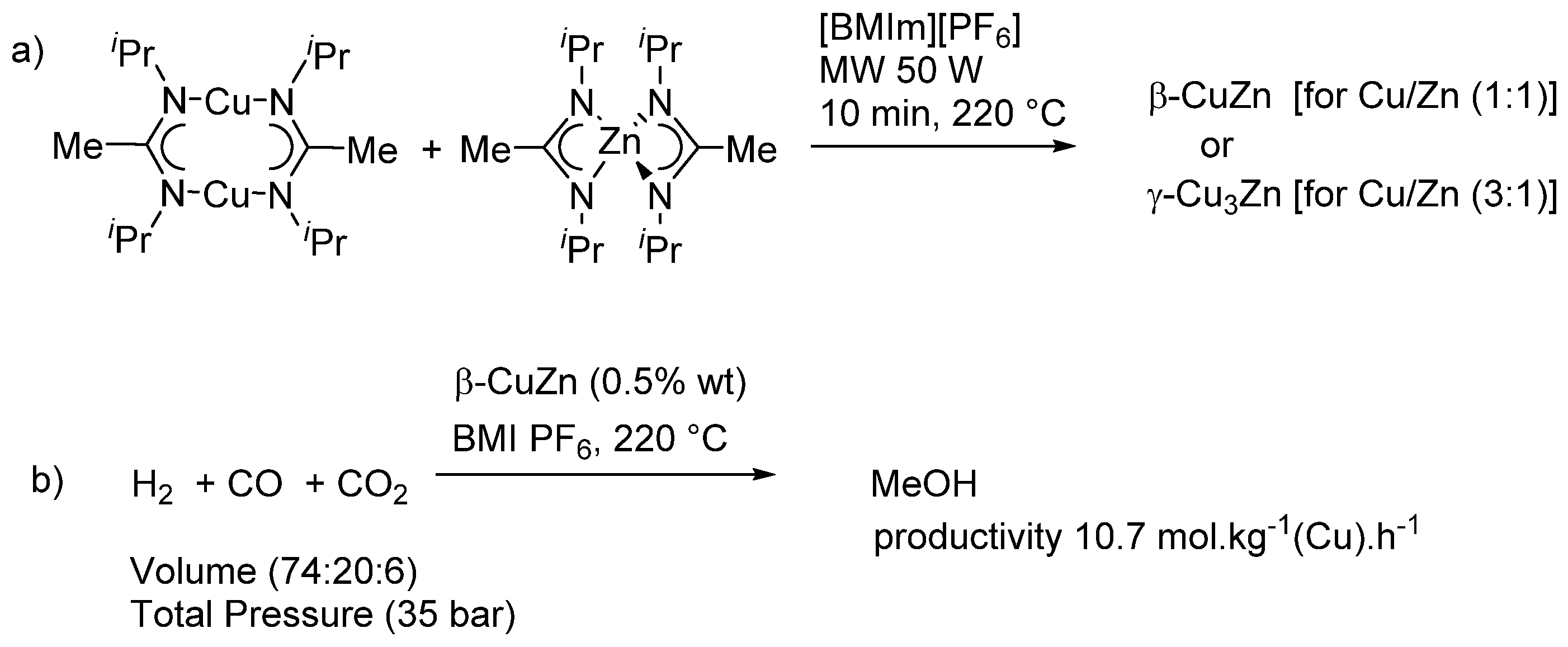

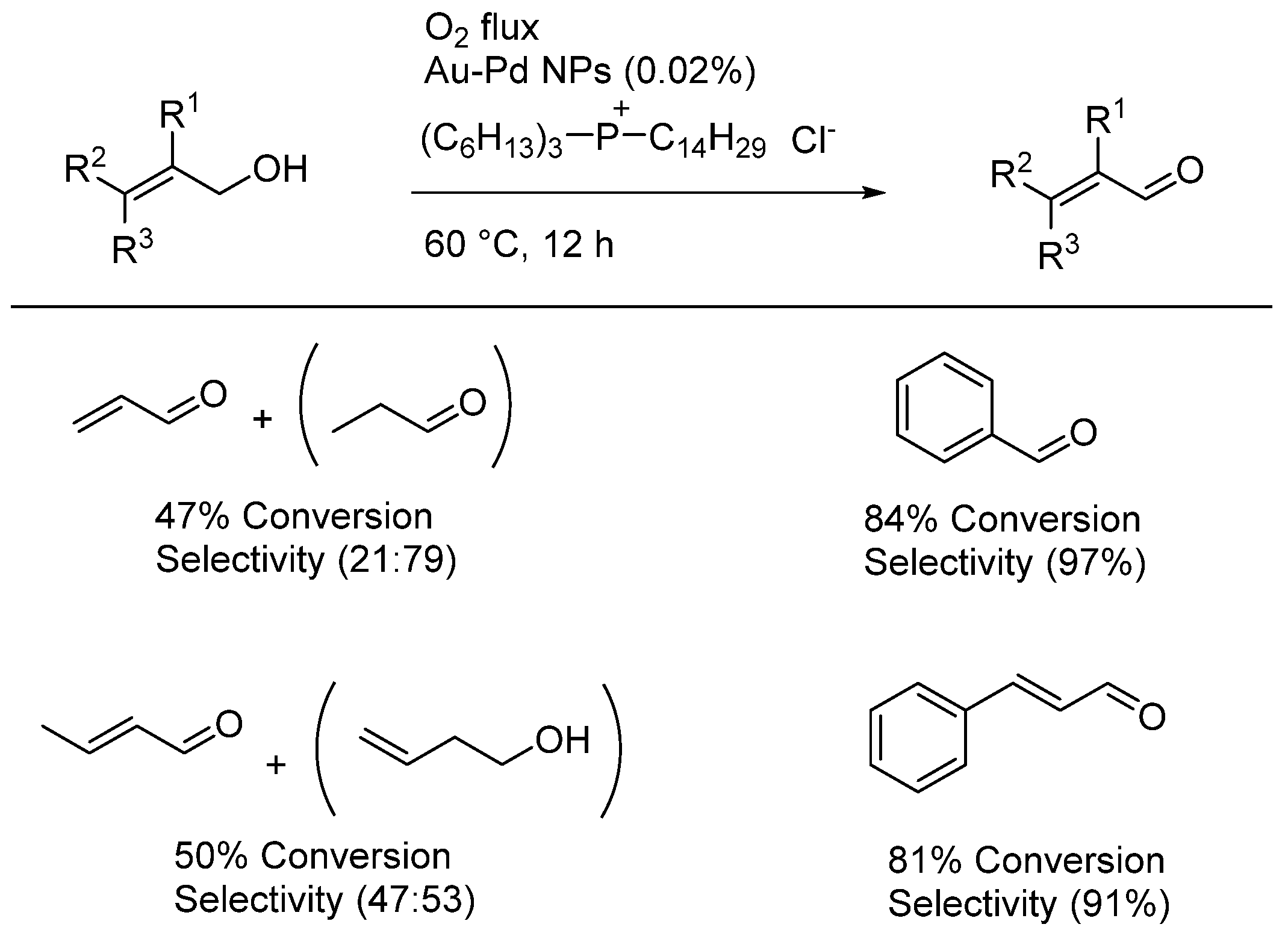
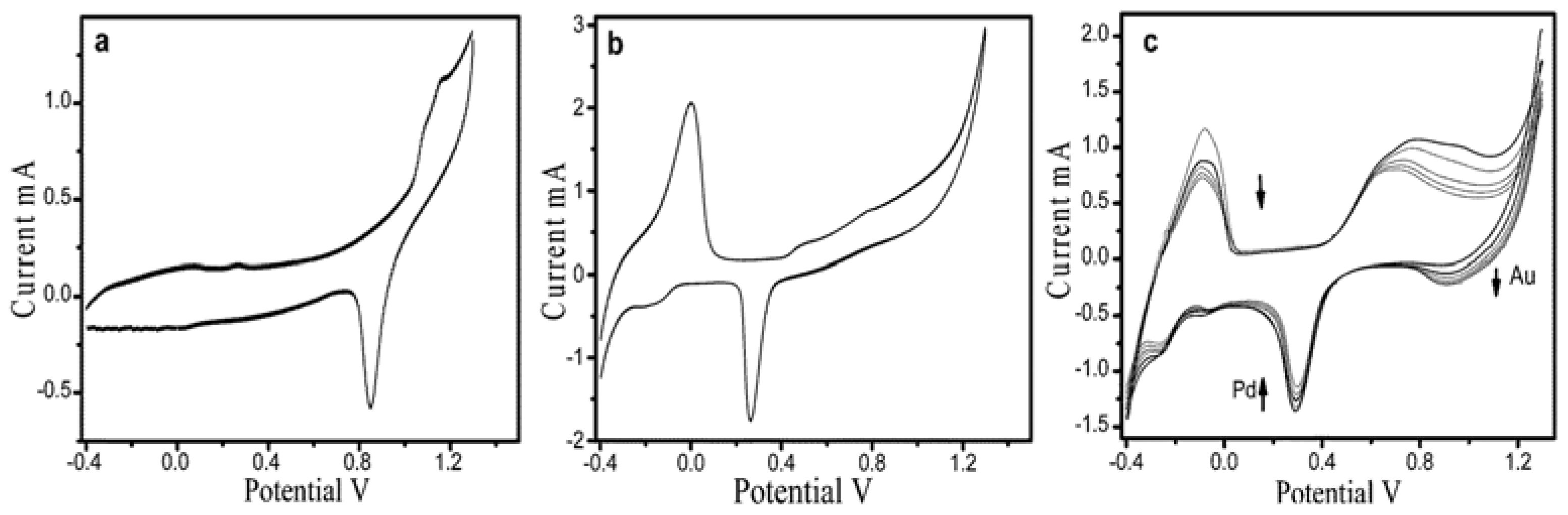
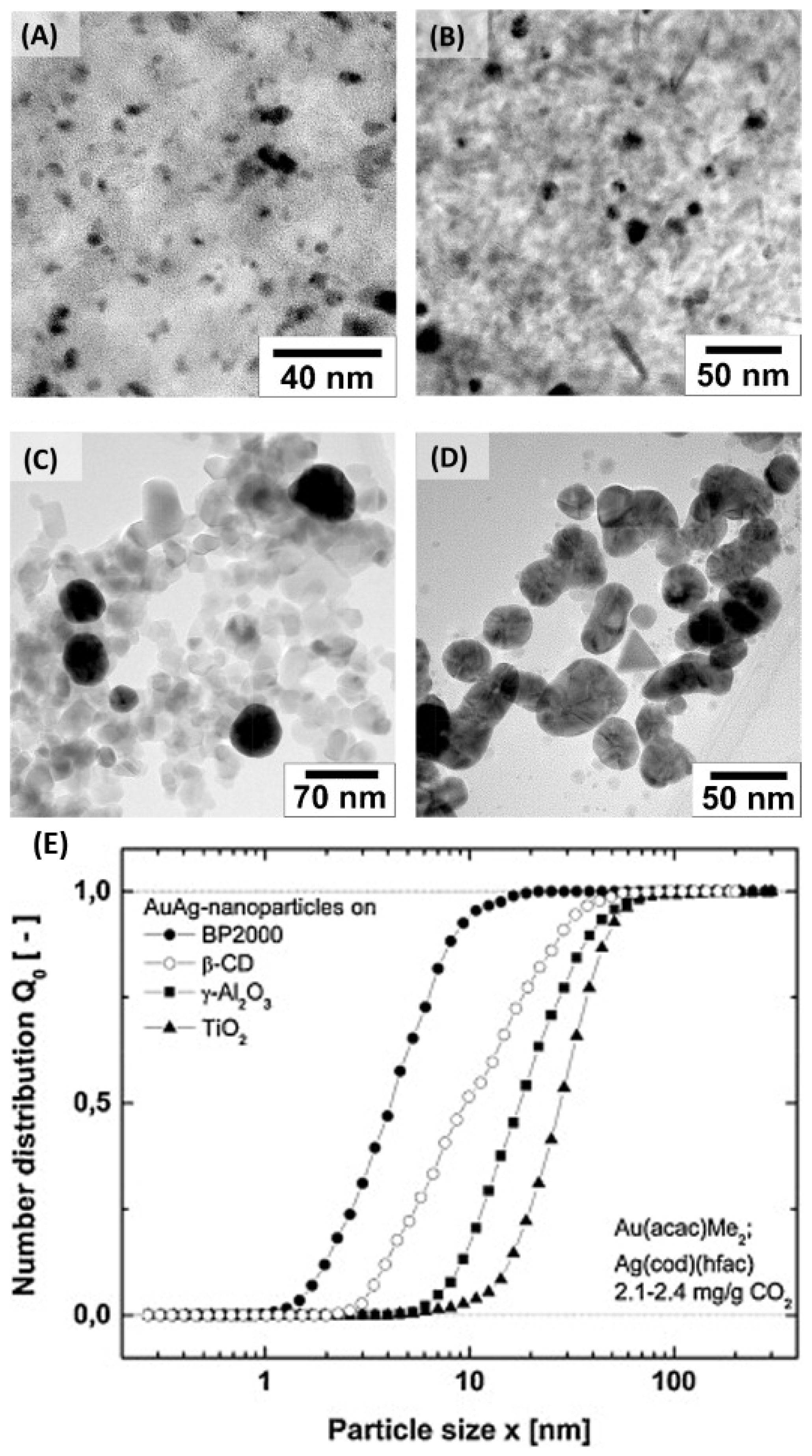
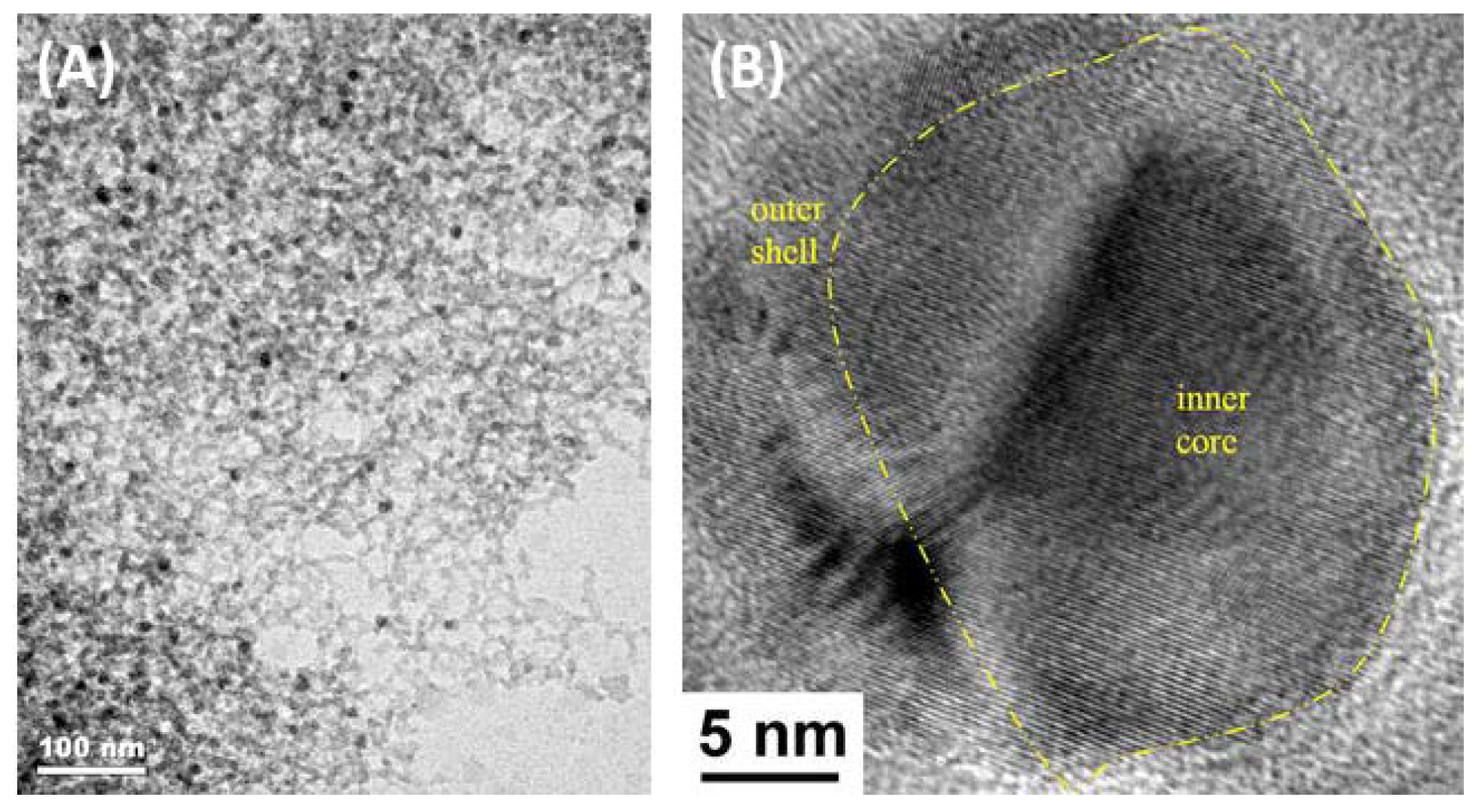

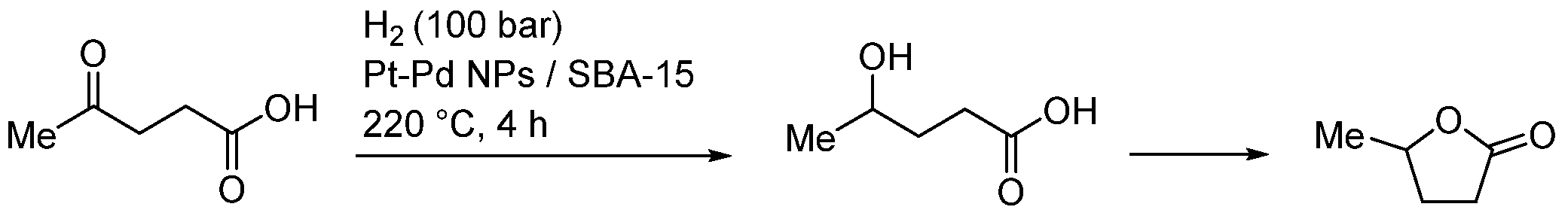
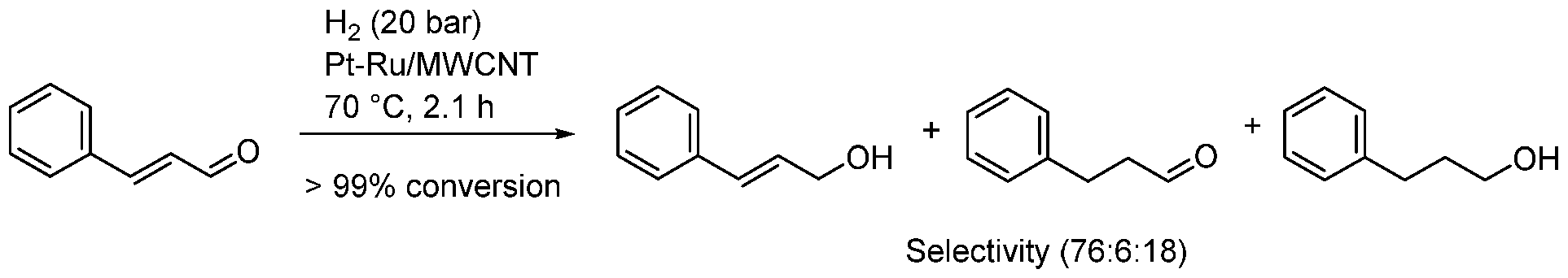
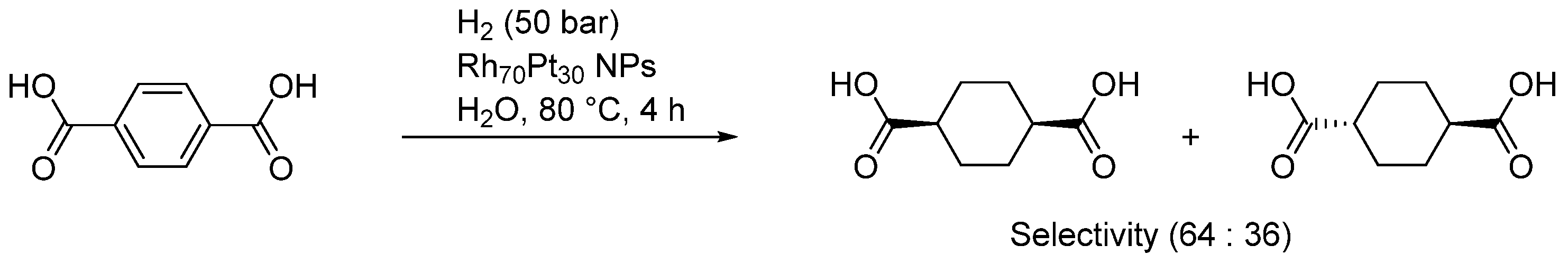
| Entry | BMNPs (Mean Size, nm) | Structure a | Metallic Precursors | Synthetic Procedure | Catalytic Reaction | Refs. |
|---|---|---|---|---|---|---|
| 1 | NiPd (2–4) | alloys | NiCl2·6H2O, K2[PdCl4], | co-reduction | C-C cross-couplings | [22,23,24] |
| 2 | NiFe (10–70) | n.r. | FeSO4·7H2O, Ni(NO3)2·6H2O | co-reduction | dechlorinations | [27] |
| 3 | PdCu (3–7) | alloys | H2[PdCl4], H2[CuCl4] | co-reduction | p-nitrophenol reduction | [28] |
| 4 | PdNi (6–20) | alloys | Pd(OAc)2, NiSO4·6H2O | co-reduction | Buchwald-Hartwig aminations | [30] |
| 5 | PtCu (3) | alloys | H2[PtCl6], Cu(OAc)2 | co-reduction | Rhodamine B degradation | [31] |
| 6 | AuAg (12) | alloys | H[AuCl4], AgNO3 | co-reduction | nitrophenols reduction | [32] |
| 7 | AuAg (8) | core(Au)-shell(Ag) | H[AuCl4], AgNO3 | co-reduction | nitrophenols reduction | [32] |
| 8 | AuPd (80) | n.r. | H[AuCl4], PdCl2 | co-reduction | Suzuki and Heck C-C cross-couplings | [33] |
| 9 | FePd (25–40) | n.r. | Pd(OAc)2, FeSO4·7H2O | sequential reduction | dechlorination | [34,35,36] |
| 10 | FeNi (20–100) | n.r. | NiSO4·6H2O, FeSO4·7H2O | sequential reduction | dechlorination | [38] |
| 11 | AuPd (8) | core(Au)-shell(Pd) | H[AuCl4], H2[PdCl4] | sequential reduction | dechlorination | [39] |
| 12 | AuPd (4.2) | core(Au)-shell(Pd) | H[AuCl4], K2[PdCl4] | sequential reduction | oxidation of unsaturated alcohols | [41,42] |
| 13 | CuPd@γ-Al2O3 (6.9) | alloys | Cu(NO3)2, H2[PdCl4] | co-reduction | transfer hydrogenation | [43] |
| 14 | CuNi@SiO2 (5.7) | alloys | Cu(NO3)2, Ni(NO3)2, | co-reduction | p-nitrophenol reduction | [44] |
| 15 | FeNi@MMT (30–40) | n.r. | FeSO4·7H2O, NiSO4·6H2O | co-reduction | reductive cyclization of levulinic acid | [45] |
| 16 | AuPd@GO (37) | alloys | H[AuCl4], PdCl2 | co-reduction | photocatalytic degradation of phenols | [46] |
| 17 | FePd@DOW® M4195 (3–5) | n.r. | Fe2(SO4)3, PdCl2 | co-reduction | nitrate reduction | [47] |
| 18 | AgPt@sepiolite (30–50) | core(Ag)-shell(Pt) | AgNO3, H2[PtCl6]·6H2O | sequential reduction | nitrophenols reduction | [48] |
| 19 | AuPd@amberlite (8) | core(Au)-shell(Pd) | H[AuCl4], H2[PdCl4] | sequential reduction | hydrogenation | [49] |
| 20 | AuPt@zeolite (4–4.5) | n.r. | H[AuCl4], K2[PtCl4] | sequential reduction | oxidation of glycerol | [50] |
| 21 | CuPt@C (10–12) | core(Cu)-shell(Pt) | CuSO4, H2[PtCl6] | sequential reduction | oxygen reduction reaction | [51] |
| Entry | BMNPs (Mean Size, nm) | Structure | Metallic Precursors | Synthetic Procedure | Catalytic Reaction | Refs. |
|---|---|---|---|---|---|---|
| 1 | CuNi (8) | alloys | Ni(OAc)2, Cu(OAc)2 | co-reduction | alkyne-azide cycloaddition | [58] |
| 2 | PtRu (3.2) | alloys | RuCl3·3H2O, H2[PtCl6]·6H2O | co-reduction | H3N-BH3 hydrolysis | [59] |
| 3 | FePt (3.2–3.6) | alloys | [Pt(acac)2], Fe(CO)5 | co-reduction | hydrogenations | [60] |
| 4 | PdCo@3DG (15–25) | alloys | PdCl2, CoCl2 | co-reduction | Sonogashira and Suzuki couplings | [61] |
| 5 | NiCu@CNTs (25–42) | alloys | Ni(NO3)2·6H2O, Cu(NO3)2·3H2O | co-reduction | CH4 decomposition | [63,64] |
| Entry | BMNPs (Mean Size, nm) | Structure | Metallic Precursors | Synthetic Procedure | Catalytic Reaction | Refs. |
|---|---|---|---|---|---|---|
| 1 | AuPd (4–5) | core(Au)-shell(Pd) | Au(OAc)3, Pd(OAc)2 | co-reduction | hydrogenations/hydrodehalogenations | [76] |
| 2 | AuPd (4.6; 3.2) | n.r. | H[AuCl]4 K2[PdCl4] | co-reduction | hydrogenations | [77,78,79] |
| 3 | PtSn (2–3) | alloy | PtCl2, SnCl2 or Sn(OAc)2 | co-reduction | hydrogenations | [85] |
| 4 | RuCu (2–3) | core(Ru)-shell(Cu) | [Ru(cod)(cot)] [CuMes] | co-reduction | hydrogenations | [86] |
| 5 | FeRu (1.65) | n.r. | [Ru3(CO)12] [Fe2(CO)9] | co-decomposition | hydrogenations | [87] |
| 6 | CuZn (45–50) | alloy | Cu(II) and Zn(II) amidinates | co-reduction | methanol synthesis | [90] |
| 7 | NiGa (7–29; 12–19) | alloy | Ni(NO3)2·6H2O, Cu(NO3)2·6H2O | co-pyrolysis | hydrogenations | [91] |
| 8 | AuPd (3.7) | n.r. | K2[PdCl4] H[AuCl]4·4H2O | sequential-reduction | oxidations | [42] |
| Entry | BMNPs (Mean Size, nm) | Structure | Metallic Precursors | Synthetic Procedure | Catalytic Reaction | Refs. |
|---|---|---|---|---|---|---|
| scCO2 | ||||||
| 1 | PdAu@TS-1 (3–8) | alloys | PdCl2, H[AuCl4]·3H2O | co-reduction | olefin epoxidations | [122] |
| 2 | FeCo@SiO2 (10–20) | alloys (bcc) | Fe(NO3)3·9H2O, Co(NO3)2·6H2O | co-reduction | Fischer–Tropsch | [124,125] |
| 3 | CoRu@γ-Al2O3 (n.r.) | n.r. | Co(NO3)2, Ru precursor n.r. | calcination | Fischer–Tropsch | [127] |
| 4 | RuPt@ MWCNTs (2.0–3.6) | hcp and fcc clusters | Pt(acac)2, Ru(acac)3 | co-reduction | combustion | [117] |
| 5 | CuPt@ MWCNTs (3.5–7.9) | alloys (fcc) | Pt(acac)2, Cu(hfa)2 | co-reduction | combustion | [117] |
| 6 | AuPt@ MWCNTs (5.6–13) | alloys (fcc) | Pt(acac)2, AuMe2(acac) | co-reduction | combustion | [117] |
| 7 | PdPt@ MWCNTs (5.9–12.5) | alloys (fcc) | Pt(acac)2, Pd(hfa)2 | co-reduction | combustion | [117] |
| 8 | NiPt@ MWCNTs (3.1–10.1) | alloys (fcc) | Pt(acac)2, Ni(hfa)2 | co-reduction | combustion | [117] |
| 9 | Pt-CeOx@Al2O3 (2–4) | core (Pt)-shell (CeOx) | CeCl3·7H2O, Pt(acac)2 | calcination followed by reduction | Water-gas shift | [128] |
| 10 | RuRh@MCM-41 (2.2–4.6) | hcp and fcc clusters | Ru(acac)3, Rh(acac)3 | co-reduction | arene hydrogenations | [129] |
| 11 | RuPd@MCM-41 (2.3–3.5) | hcp and fcc clusters | Ru(acac)3, Pd(acac)2 | co-reduction | arene hydrogenation | [129] |
| 12 | RhPd@MCM-41 (3.2–4.4) | alloys | Rh(acac)3, Pd(acac)2 | co-reduction | arene hydrogenations | [129] |
| 13 | FeRu (1.3–1.9) | alloys | Fe2(CO)9, [Ru3(CO)12] | co-reduction | olefin hydrogenations | [87] |
| 14 | PtPd@SBA-15 (4.9–6.1) | alloys | [PtMe2(cod)], [PdCp(allyl)] | co-reduction | ketone hydrogenation | [110] |
| 15 | PtRu@MWCNTs (1.0–2.5) | alloys | [Ru(cod)(cot)], [PtMe2(cod)] | co-reduction | ketone and olefin hydrogenations | [131] |
| 16 | PdRu (3.5–4.4) | alloys | Pd(NO3)2, RuCl3 | co-reduction | olefin hydrogenations | [132] |
| 17 | RhPt@SBA-15 (6.1) | alloys | Rh(acac)3, Pt(acac)2 | co-reduction | arene hydrogenations | [133] |
| 18 | AuPd (7) | alloys | PdCl2, H[AuCl4]·4H2O | co-reduction | hydrogenatios | [134] |
| 19 | AuPt (7) | alloys | H2[PtCl6]·6H2O, H[AuCl4]·4H2O | co-reduction | Suzuki-Miyaura C-C cross-couplings | [134] |
| 20 | PdPt (1.5–2.3) | alloys | K2[PdCl4], K2[PtCl4] | co-reduction | Suzuki-Miyaura C-C cross-couplings | [135] |
| 21 | Pd Cu2+/alginate aerogel (2–4) | Pd NPs | Na2[PdCl4], CuCl2 | reduction | Suzuki-Miyaura C-C cross-couplings | [136] |
| scH2O | ||||||
| 22 | NiCo@Cblack (28.2–49.6) | alloys | Ni(NO3)2·6H2O, Co(NO3)2·6H2O | co-reduction | gasification of phenol | [62] |
| 23 | NiZn@Cblack (n.r.) | alloys | Ni(NO3)2·6H2O, Zn(NO3)2·6H2O | co-reduction | gasification of phenol | [62] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dang-Bao, T.; Pla, D.; Favier, I.; Gómez, M. Bimetallic Nanoparticles in Alternative Solvents for Catalytic Purposes. Catalysts 2017, 7, 207. https://doi.org/10.3390/catal7070207
Dang-Bao T, Pla D, Favier I, Gómez M. Bimetallic Nanoparticles in Alternative Solvents for Catalytic Purposes. Catalysts. 2017; 7(7):207. https://doi.org/10.3390/catal7070207
Chicago/Turabian StyleDang-Bao, Trung, Daniel Pla, Isabelle Favier, and Montserrat Gómez. 2017. "Bimetallic Nanoparticles in Alternative Solvents for Catalytic Purposes" Catalysts 7, no. 7: 207. https://doi.org/10.3390/catal7070207