1. Introduction
The field of heterogeneous photocatalysis has expanded rapidly within the last four decades, having undergone various developments especially in relation to energy and the environment. It can be defined as the acceleration of photoreaction in the presence of a catalyst. The two most significant applications of photocatalysis have been in solar water splitting and the purification of air and water containing low concentrations of pollutants. The multidisciplinary nature of the field has also increased significantly and includes semiconductor physics, surface sciences, photo and physical chemistry, materials science and chemical engineering [
1].
Heterogeneous photocatalysis can be described as the acceleration of photoreaction in the presence of a catalyst. In the contexts of history and research, interest in heterogeneous photocatalysis can be traced back to many decades when Fujishima and Honda discovered in 1972 the photochemical splitting of water into hydrogen and oxygen in the presence of TiO2 From this time, extensive research, much of it published, has been carried out to produce hydrogen from water in oxidation reduction reactions using a variety of semiconductor catalyst materials.
In recent years, interest in photocatalysis has focused on the use of semiconductor materials as photocatalysts for the removal of ambient concentrations of organic and inorganic species from aqueous or gas phase systems in environmental clean-up, drinking water treatment, industrial and health applications. This is because of the ability of TiO2 to oxidize organic and inorganic substrates in air and water through redox processes In this context, TiO2 has not only emerged as one of the most fascinating materials in both homogeneous and heterogeneous catalysis, but has also succeeded in engaging the attention of physical chemists, physicists, material scientists and engineers in exploring distinctive semiconducting and catalytic properties.
Inertness to chemical environment and long-term photostability has made TiO
2 an important material in many practical applications, and, in commercial products ranging from drugs to foods, cosmetics to catalysts, paints to pharmaceuticals, and sunscreens to solar cells in which TiO
2 is used as a desiccant, brightener, or reactive mediator [
2]. The U.S. Food and Drug Administration permits up to 1% TiO
2 as an inactive ingredient in food products. While there are no known health effects associated with the use of TiO
2, a recent study found that 3–6 year old children are the most affected group of people that consume TiO
2 particles from food products. Many new properties of TiO
2 have been explored during the past few years [
3]. It should be stated that regulatory frame work for the use of TiO
2 in food products are yet to be firmly established in many countries, especially developing nations. The catalyst itself is unchanged during the process and no consumable chemicals are required. This results in considerable savings and simpler operation of the equipment involved.
Large bandgap semiconductors like TiO
2 are commonly investigated in the rutile (bandgap 3.0 eV) and anatase (bandgap 3.2 eV) phases and TiO
2 response to UV light has not only led to photocatalysis research [
4,
5,
6] but also to an extensive investigation of TiO
2 superhydrophilicity its use in environmental remediation and solar fuel production. Bandgap excitation of TiO
2 causes charge separation followed by scavenging of electrons and holes by surface adsorbed species,
Figure 1:
Figure 1.
Schematic of semiconductor excitation by band gap illumination [
4,
7] leading to the creation of “electrons” in the conduction band and “holes” in the valance band.
Figure 1.
Schematic of semiconductor excitation by band gap illumination [
4,
7] leading to the creation of “electrons” in the conduction band and “holes” in the valance band.
Visible photocatalysis can thus be utilized by doping TiO
2 with a sensitizing dye or short bandgap semiconductors. By controlling the surface treatment and medium conditions, it is possible to fine-tune the photocatalytic properties of TiO
2 to desired applications including the effective mineralization of contaminants in air, as self-cleaning glass and ceramic tiles. On the other hand, the practical use of TiO
2 for remediation of chemical contaminants in wastewater is a challenge because of catalyst poisoning or deactivation. In addition, there remain some challenges with respect to extending the photocatalytic response of TiO
2 in the visible region [
6], an area that merits fundamental research.
In spite of the extensive efforts to dope TiO
2 with C, N, S and transition metal ions such as Ag, Au, Fe, Ru, photocatalytic activity of TiO
2 in the visible region has remained quite low, an issue that has been addressed in recent articles highlighting issues and challenges associated with the application of photocatalysis. Generally, two or more phases are involved in a photocatalytic reaction—a light source and a semiconductor material are used to initiate the photoreaction while the catalyst system can simultaneously carry out oxidation and reduction reactions using long wavelength, UV light as well as sunlight. As a method for contaminant control in water and air,
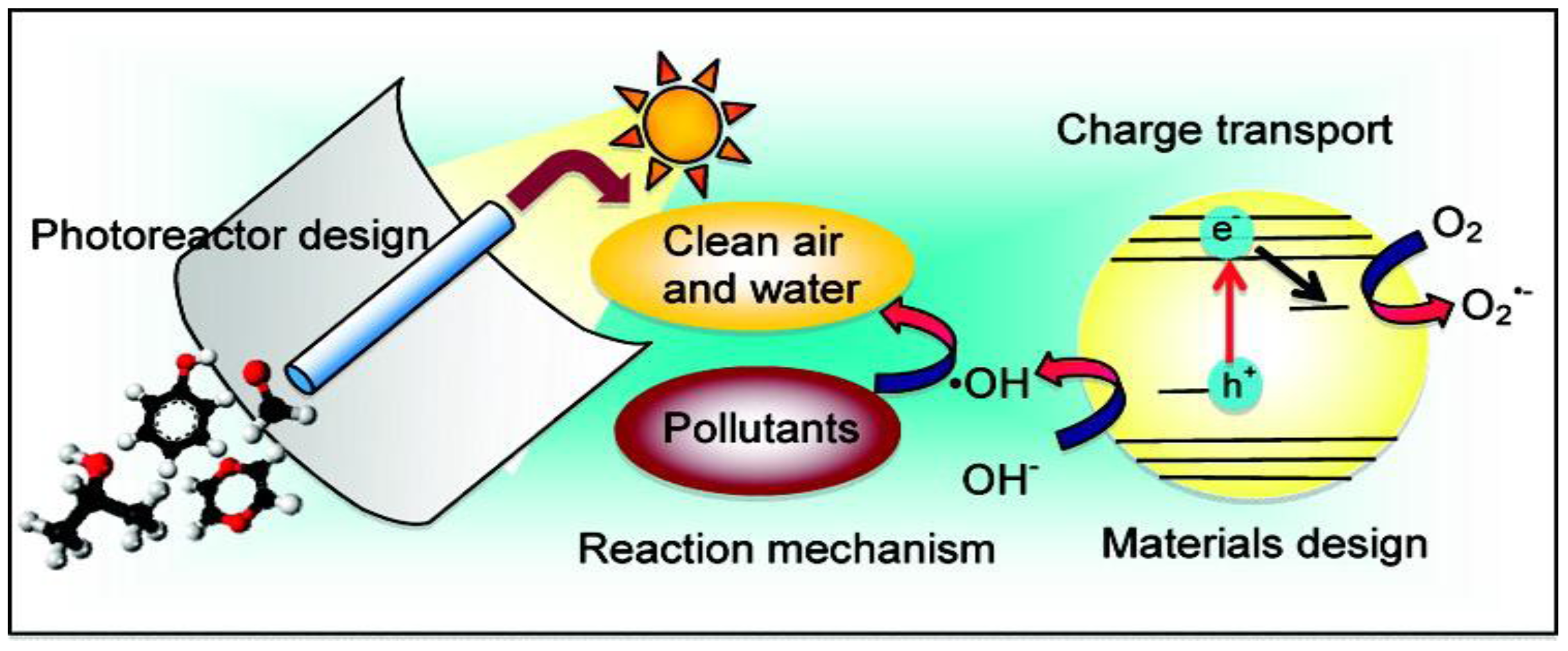
Figure 2, heterogeneous photocatalysis using semiconductors such as titanium dioxide is more efficient than conventional methods. This is because as the photocatalytic process gradually breaks down the contaminant molecules, no residue of the original material remains and therefore no sludge requiring disposal to landfill is produced. The catalyst itself is unchanged during the process and no consumable chemicals are required. This results in considerable savings and a simpler operation of the equipment involved. Additionally, because the contaminant is attracted strongly to the surface of the catalyst, the process will continue to work at very low concentrations. Taken together, these advantages mean that the process results in considerable savings in water production cost and keeping the environment clean.
Figure 2.
Schematic of the interplay of photocatalysis treatment, reactor and material design and photocatalytic reaction mechanism [
1]. Note: This figure is reproduced with permission from [
1], Copyright © 2012 American Chemical Society.
Figure 2.
Schematic of the interplay of photocatalysis treatment, reactor and material design and photocatalytic reaction mechanism [
1]. Note: This figure is reproduced with permission from [
1], Copyright © 2012 American Chemical Society.
In order to activate the degradation process, pure TiO2 requires photo-excitation with light at wavelengths exceeding the band gap of the active anatase phase of 3.2 eV, that is, wavelengths of <387 nm. Indeed, commercially available photocatalytic water treatment plant using artificial UV-light as the energy source is available and can be considered as a developed market.
However, UV in natural sunlight represents only 5%–8% of the solar spectrum at sea level and this provides a limitation and the requirement of artificial illumination of the catalysts in order to achieve degradation of the organic material in times that are practical for water treatment processing. This greatly restricts the application of the technology to areas where abundant electricity supplies are available. The need for more stable, efficient catalysts which can be activated by natural sunlight is now the goal of researchers. In order to achieve significant improvements in catalytic activity using visible light, precise control of the stoichiometry of the catalytic metal oxides and mixed metal oxides, addition of dopants, particle shape and pore topology are all critical factors for catalyst developers.
Advances in the manipulation of matter at the nanoscale are leading to potential solutions for the depollution of water in remote locations. In recent years, the development of nanoscale metal oxides has greatly increased the catalytic activity by virtue of the high specific surface area available for reaction of the smaller particles. In addition, the strategy to alter the band gap of the catalyst is an important approach as this determines the portion of the solar spectrum the catalyst absorbs and, consequently, the amount of energy that is converted to reactive species. Photocatalysts that have high activity using wavelengths of light in the visible spectrum (380 nm < λ < 500 nm) have been demonstrated where potentially greater amounts of energy is available (solar peak energy is around 460 nm). This has been achieved by altering the band gap of, for example, TiO2. Doping of TiO2 with transition metal ions (for example, V, Cr, Mn, Fe and Ni) as well as with Ag, Au and Ru, have been demonstrated to red-shift the TiO2 absorption band from the UV into the visible region, resulting in a great increase in the efficiency of solar-light photocatalysis.
Many industrial waste streams are not suitable for biological processing due to their inherent toxicity, but their treatment by traditional non-catalytic chemical processes or by incineration is energy intensive [
7]. On the other hand, TiO
2 photocatalysts has been shown to decompose organic contaminants in water efficiently because a strong oxidizing ability is generated when the TiO
2 is irradiated by appropriate band gap illumination [
8]. Increased attention is being paid to heterogeneous solution phase catalysis in order to develop an environment-friendly technology to purify polluted air and water without electricity or other energy consuming sources.
Heterogeneous photocatalytic reactions are carried out either in a slurry-type reactor where the catalyst particles are suspended in the contaminated water or in an immobilized-type reactor where the catalyst particles are immobilized onto the surface of various inert substrates of various types and configurations [
9,
10,
11] and the process can be explained on the basis of the following mechanism.
2. Mechanism of Semiconductor Catalysis
A photocatalyst harnesses UV radiation from sunlight or artificial light and uses the energy to break down different substances including organic materials, organic acids, estrogens, pesticides, dyes, crude oil, microbes (including viruses) and chlorine resistant organisms, inorganic molecules such as nitrous oxides (NO
x) and, in combination with precipitation or filtration, can also remove metals such as mercury [
13,
14,
15]. Due to this universal applicability, photocatalysis with nanoparticles as catalysts is used to reduce air pollution, in building materials, for self-cleaning surfaces in addition to water purification. Titanium dioxide (TiO
2) is the most common photocatalyst and comparably little research has been conducted on zinc oxide, ZnO, which could be a viable alternative for some applications. To avoid free nanoparticles in water, TiO
2 nanoparticles are usually immobilized on a substrate [
16] or integrated into thin-films and other materials. For the activation of TiO
2, UV irradiation from sunlight or artificial light is needed, with UVB more efficient than UVA. To allow activation by visible light, TiO
2 can be modified with a second semiconductor, dyes, nitrogen, carbon or sulphur. For example, TiO
2 doped with nitrogen demonstrated superior photocatalytic activities compared to commonly used unmodified TiO
2 nanoparticles in both chemical compound degradation and bactericidal reactions [
17].
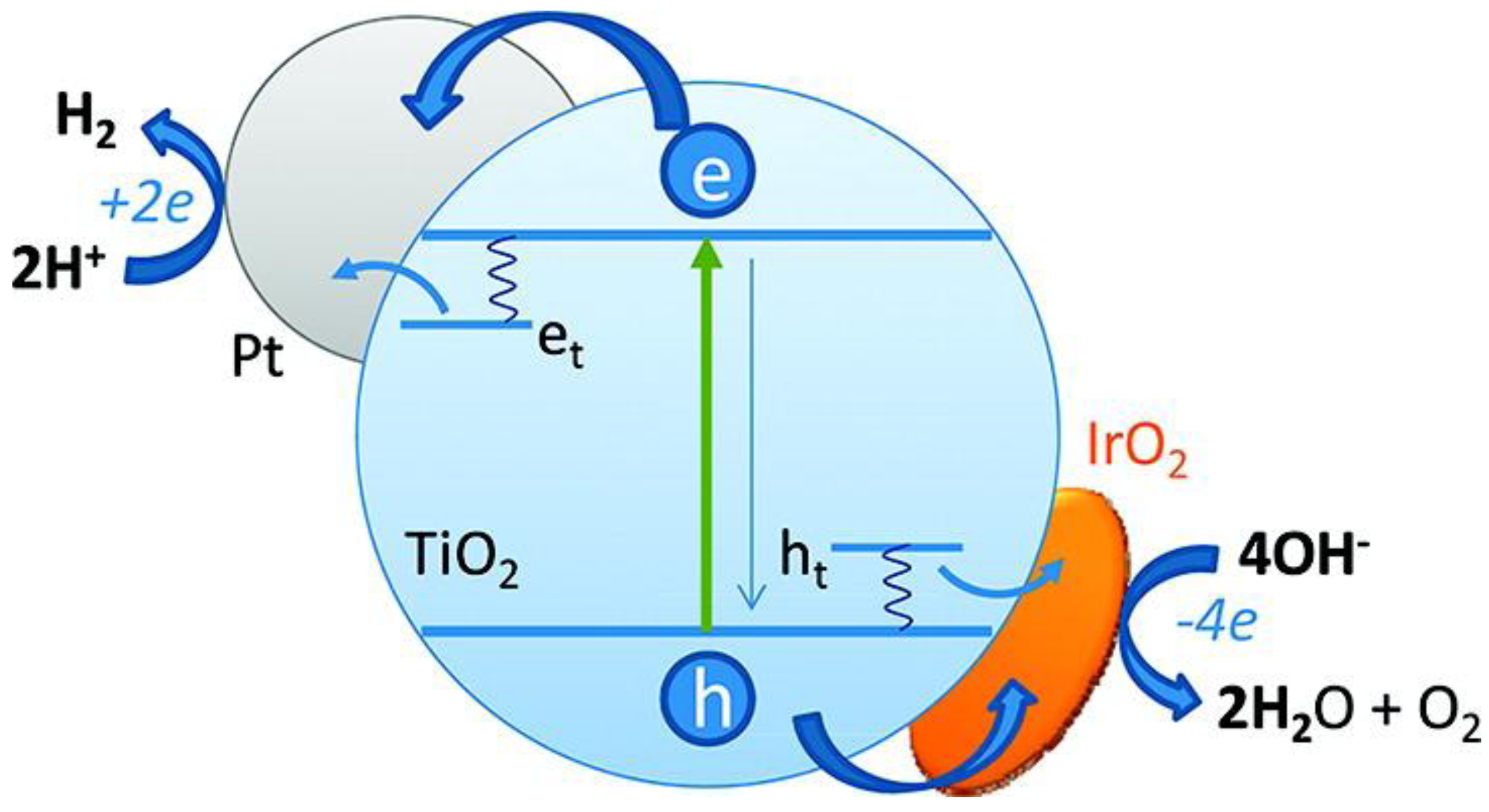
With respect to a semiconductor oxide such as TiO
2, photocatalytic reactions are initiated by the absorption of illumination with energy equal to or greater than the band gap of the semiconductor. This produces electron-hole (e
−/h
+) pairs as in Equation (1),
Figure 3:
where cb is the conduction band and vb is the valence band. Thus, as a result of irradiation, the TiO
2 particle can behave either as an electron donor or acceptor for molecules in contact with the semiconductor. The electron and hole can recombine, releasing the absorbed light energy as heat, with no chemical reaction taking place. On the other hand, they can participate in redox reactions with adsorbed species as the valence band hole is strongly oxidizing while the conduction band electron is strongly reducing. On the semiconductor surface, the excited electron and the hole can participate in redox reactions with water, hydroxide ion (OH
−), organic compounds or oxygen leading to mineralization of the pollutant.
Figure 3.
Schematic of the charge transfer across semicopnductor interface. Note: This figure is reproduced with permission from [
12], Copyright © 2012 American Chemical Society.
Figure 3.
Schematic of the charge transfer across semicopnductor interface. Note: This figure is reproduced with permission from [
12], Copyright © 2012 American Chemical Society.
In fact, research shows that the charges can react directly with adsorbed pollutants, but reactions with water are predominant since the water molecules are more abundant than contaminant molecules. Consequently, oxidation of water or OH
− by the hole produces the hydroxyl radical (·OH), a powerful oxidant. For the purpose of comparison, it is important to compare the oxidation potential of hydroxyl radical (·OH) which is 2.8 V relative to the normal hydrogen electrode. Apart from electron and their holes, other substances that may be used for water disinfection include ozone (2.07 V), H
2O
2 (1.78 V), HOCl (1.49 V) and chlorine (1.36 V). OH radicals are able to rapidly attack pollutants on the semiconductor surface and, as such, are the most important radicals formed in TiO
2 photocatalysis. An important reaction of the conduction band electron is reduction of adsorbed oxygen to oxygen radicals and this prevents the electron from recombining with the hole and results in an accumulation of oxygen radicals that can also participate in degrading contaminants in solution [
11,
18].
Published work indicate that heterogeneous photocatalytic process involves at least five separate reaction steps [
19] and include (1) diffusion of reactants to the surface of semiconductor, (2) adsorption of reactants onto the surface of semiconductor, (3) reaction on the surface of semiconductor, (4) desorption of products from the surface of the semiconductor and (5) diffusion of products from the surface of the semiconductor. There are two routes through which OH radicals can be formed—the reaction of the valence-band holes with either adsorbed H
2O or with the surface OH groups on the TiO
2 particle. These processes have been summarized using appropriate equations [
20].
3. Photocatalyst Materials and Supports
Current and past research in photocatalytic materials has investigated several photocatalysts and their properties. Ideally, a photocatalyst should possess the following properties: photoactivity, biological and chemical inertness, stability toward photo-corrosion, suitability for visible or near UV light energy harnessing, low cost and lack of toxicity [
21]. TiO
2 as a semiconductor photocatalyst, has excellent pigmentary properties, high ultraviolet absorption and high stability which allow it to be used in different applications such as electro-ceramics, glass and in the photocatalytic degradation of chemicals in water and air. It has been used in the form of a suspension, or a thin film in water treatment [
22,
23] and has different crystalline forms, the most common forms being anatase and rutile while the third, brookite, is uncommon, unstable and hence does not feature in discussion of catalyst materials.
Anatase TiO
2, the most stable form of TiO
2 [
24], can be converted to rutile by heating to temperatures above 700 °C. In photocatalytic applications, research has shown that anatase is more efficient than rutile because of its more open structure compared with rutile. Degussa P-25 is the commercially available form of TiO
2 and consists of two forms of approximating 25% rutile and 75% anatase, and has been used in many studies of photocatalytic degradation because of its chemical stability, ready availability, reproducibility and activity as a catalyst for oxidation processes [
13,
25]. However, studies are being carried out to develop existing or prepare new materials which can be used under solar energy and hence shortening the time needed for degradation.
This development includes increasing the surface area, the active sites, the absorption of photon and reducing the band gap energies of the semiconductor. For industrial applications, high activity, resistance to poisoning and stability for prolonged use at elevated temperatures, mechanical stability, and resistance to attrition, physical and chemical stability in various conditions, are required characteristics of catalysts [
21]. In addition, in the degradation of organic compounds, the redox potential of the H
2O/·OH electrode, OH
− → ·OH + e
−;
E0 = −2.8 V, must lie within the band gap of the photocatalyst as illustrated in
Figure 1. Nowadays, many semiconductors are used as photocatalysts because of their narrow band gap between the valence and conduction bands and they need to absorb energy equal to or more than this energy gap. This involves the movement of electrons from e
−/h
+ or negatively charged electrons to positively charged hole pairs [
26].
Many semiconductors have enough band-gap energies for the effective catalysis of many chemical reactions and this includes materials such as TiO
2, WO
3 and ZnO. Though it is well known that metal oxides are usually less active catalysts than noble metals in the majority of applications, metal oxides are more suitable since they are more resistant to poisoning and deactivation. In addition, combining two or more metal oxide catalysts could improve or enhance catalytic activity. TiO
2 is the most frequently used photocatalyst because of its photostability and low cost, combined with its biological and chemical inertness and resistant to photo and chemical corrosion. On the other hand, binary metal sulfide semiconductors such as CdS and PbS are regarded as insufficiently stable for catalysis and are toxic. ZnO is also unstable in illuminated aqueous solutions while WO
3 has been investigated as a potential photocatalyst, but it is generally less active catalytically than TiO
2. However, these can be combined with other semiconductors including TiO
2 to achieve greater photocatalytic efficiency or stability [
27,
28,
29,
30].
There are many semiconductor support materials that have been investigated. Usually, semiconductor supports are classified by their chemical nature and these can be organic or inorganic supports. They play an important role in immobilizing active catalyst, increase the surface area of catalytic material, decrease sintering and improve hydrophobicity, thermal, hydrolytic and chemical stability of the catalytic material. Examples include glass, carbon fibers and woven fiber cloths and these have been studied as support materials in groundwater denitrification and in the photocatalytic oxidation of water pollutants as well as in other applications. When fibrous supports are applied, loss of pressure is low and pore diffusion resistance is significantly lower than with pellet shaped catalysts. Glass possess an advantage as a catalyst support because of its transparency to UV light in photocatalytic applications [
30].
Studies by Teoh, Kamat and others [
1,
9,
12,
31,
32,
33], show that interest in photocatalysis has also centered on ZnO, which has similar energy characteristic to TiO
2. They report that ZnO however suffers from photo-corrosion problems upon excitation in solution. Hematite (α-Fe
2O
3) on the other hand, which is low cost, abundant and has narrow bandgap for harnessing solar energy, suffers from rapid charge recombination and a short charge carrier diffusion length. As a result of these draw backs, renewed interest in this material has focused on its modification with cationic dopants such as Cr and Mo to improve its charge transport properties [
34] or doping with Si to reduce the charge diffusion path length. Other reviews have also talked about Tungsten trioxide WO
3 as another narrow bandgap material, 2.7 eV, and, as such, it has received renewed interest. Like hermatite, Tungsten trioxide, has the disadvantage of a low electron conduction band, although studies claim that coupling with Pt cocatalyst has been observed to be useful in promoting alternative multiple-electron reduction processes with lower reduction potentials. This has increased the use of WO
3 as one of the very few highly visible-light-active single-phase oxide photocatalysts [
29,
30,
31,
34,
35,
36,
37,
38,
39,
40,
41].
In many cases, the superiority of other photocatalyst materials over P25 is based on a selection of test reactions or only a single organic substrate and this has its problems as all of the important factors cannot be validated in a single experiment or reaction. An example is the pairing of test substrate degradation mechanistic Langmuir-Hinshelwood kinetic model, direct charge transfer, hydroxyl radicals, and superoxide radicals with many physicochemical characteristics of the photocatalyst such as the crystallinity, hydroxyl group density, size, specific surface area, surface defects, surface energetic and aggregation [
12]. Even among flame-made TiO
2 of similar crystallinity, about 80% anatase and 20% rutile, and exact anatase-rutile content, differences in the photocatalytic activity of P25 were observed [
42] due mainly to the class of organic substrate [
43] used and the materials intrinsic efficiencies for ·OH generation and direct charge transfer, which governed the dominant degradation mechanism for each of the substrate functional groups. In a broader context, Ryu and Choi [
44] compared the photocatalytic performance of commercial variants of TiO
2 and observed that one photocatalyst cannot meet all photocatalytic requirements, a fact borne out by several studies [
45].
With respect to the synergy between anatase and rutile, the dominant phases of TiO
2 studies [
46] have suggested that polymorphic characteristics of these materials was one of the reasons for the high photocatalytic efficiency of P25 and that the heterojunction of anatase-rutile promoted efficient charge separation as evidenced by studies indicating discrete trapping of electrons and holes across the different crystal phases. Any synergistic effect, however, is difficult to measure because of the differences in the physicochemical characteristics of pure anatase and rutile (surface area, crystallite size, defect content, surface hydroxylation,
etc.), compared with the individual components in the mixture. Ohtani and other studies [
46,
47,
48] on the other hand, isolated the individual components of P25 by selective acid dissolution and assessed the single components for a range of photocatalytic reactions. The studies concluded, at least for P25, a less probable synergistic effect beyond the photocatalytic activity of the individual components. By carefully synthesizing highly crystalline and physically similar anatase and rutile nanoparticles, Kho [
49,
50] and other studies demonstrated higher rates of hydrogen evolution from aqueous methanol across a wide range of mixed anatase-rutile compositions. The effect, however, was only observed when a strong inter-particle contact existed between the anatase and rutile nanocrystals, supporting the view of an efficient charge separation across heterojunctions in these materials [
1].
Doping of TiO
2 with fine noble metals such as silver (Ag), gold (Au) and palladium (Pd) has been carried out extensively as a means of enhancing charge separation. Doping has been reported [
51] to establish a barrier through equilibration between the photocatalyst and metal deposits and is governed by the difference in work function of the deposits and the electron conduction band of the photocatalyst and does not influence the mechanism of the specific reaction [
1]. Previous studies noted differences in the enhancement by Pt and Ag as well as other noble metals, on TiO
2 over a range of organic classes arising from the combination of substrate or functional group specificity and the dark catalytic oxidation effects [
12,
32,
33,
51,
52]. The latter included the catalytic oxidation of formic acid, oxalic acid, methanol, and the opening of aromatic rings, with each reaction induced by the catalytic properties of the metals. Studies have shown that when a photocatalyst is illuminated, a type of synergistic effect between the catalytic and photocatalytic processes is possible and in fact studies have shown these effects do exist. [
33,
52,
53].
With respect to new materials for use in photocatalytic work, Teoh
et al. [
1] reported in a recent article that a graphene oxide material was demonstrated to act as a mediator for electron transport and for visible-light water splitting in photocatalysis. It has to be stated that prior to this development, electron mediators were largely based on aqueous redox couples such as Fe
2+/Fe
3+ and CO
2+/CO
3+ as well as Au and Ag. A recent study by Wei and coworkers [
54] using hybrid Fe
2O
3-Pd nanoparticle photocatalyst grown by epitaxial growth of Fe
2O
3 on Pd nanoparticles, showed improved photocatalytic efficiency, although this was limited by the presence of organic stabilizers which are difficult to remove. Other materials considered in recent work include ZnS, ZnO, CdSe, Fe
2O
3 and InP [
55]. Some of these materials were reported to show some photocatalytic promise, but with uncertainty over their efficiencies and spectral characteristics [
56]. Fe
2O
3 is particularly interesting because of its stability against photo/chemical corrosion at neutral or basic pH and has band gap energy of about 2.0 to 2.2 eV corresponding to the absorption of 564 to 620 nm light [
54].
5. Doping Mechanisms
Research into photocatalyst doping has spanned several decades. Usually doping involves the use of metals or non-metals and is designed to extend the photocatalytic activity of a semiconductor lower energy excitation. Technically, doping is the introduction of foreign elements into the parent photocatalyst without giving rise to a new crystallographic forms, phases or structures and the aims are [
1] to enhance the net separation of photogenerated charges and thereby efficiently harness the wide visible-light component of about 43% in the solar spectrum as opposed to the narrow ultraviolet component of 5%. It is thus an area of increasing research activity in photocatalysis.
Although discovered as a modification technique in the early 1980s, doping has become a standard and routine method in the design of water-splitting photocatalysts [
12]. Modifying pure photocatalyst materials with metal ions, especially d block metal ions such as Fe
3+ and Cr
3+ results in the insertion of impurity energy levels between the parent conduction and valence bands. In this case, the inserted energy levels provide sub-bandgap irradiation from which electrons can be excited from dopant d-band to conduction band or from valance band to dopant d-band by lower energy photons than are required by the pure photocatalysts. However, there are a number of issues associated with metal ion doping, related to the efficiency of subsequent photocatalytic processes. Whereas the presence of metal dopants was found in some cases to enhance charge separation as well as interfacial charge transfer [
58,
59] in many other cases, the metal dopants actually resulted in rapid charge recombination through their switchable redox states, for example, Fe
3+ ↔ Fe
2+ and thereby reducing the electron diffusion length and lifetime [
1,
9,
12,
60]. This is one of the reasons for the reduced quantum efficiency in many photo response-extended doped photocatalysts.
Several studies have indicated that doping creates structural defects that could be sources for charge recombination and in this sense are potentially negative in their effects. The only exception being doping using high-energy RF and magnetron sputtering, which provide the uncommon existence of tetravalent dopants such as Fe
4+ and Cr
4+ that match the valency of Ti
4+ in TiO
2 [
61]. Co-doping with a conjugate metal cation pair such as Rh
3+/Sb
5+ can preserve charge equality of the doped photocatalysts and result in improved and extended photocatalytic ability. However, this may not necessarily prevent structural defects arising from the differences in cationic radii between dopants and the host photocatalyst. In a recent study, Serpone and co-workers [
62] provided a substantiated alternative argument that the extended photo-response instead of arising from d-band insertion, may actually originate from the color centers and oxygen vacancies that arise from the doping [
63,
64].
Doping involving the use of non metals has also been investigated and studies have reported that the introduction of non-metal such as N, C and S impurity energy levels above the parent photocatalyst valence band has been a popular doping technique and although doping wide bandgap photocatalysts such as TiO
2 and ZrO
2 with F [
64,
65,
66,
67,
68,
69] does not result in coloration, their enhanced photocatalytic efficiencies, even under sub-bandgap excitation, likely results from enhanced stoichiometric defects and this is useful in photocatalytic processes. Higher photocatalytic activity, stability [
28] and nontoxicity of dopant ions are among the advantages of nonmetal over metal dopants and, in general, TiO
2 and several other materials have been studied including the effect of impurity levels in the dopant on photocatalytic efficiency. In addition, results obtained using TiO
2 and oxides such as WO
3, Ta
2O
5 and Ba
5Ta
4O
15 have been published [
69,
70,
71]. By studying nitrogen doped TiO
2 using real-time fluorescence imaging, Tachikawa and coworkers [
72] confirmed the absence of ·OH under sub-bandgap irradiation but retained the formation of O
2•–, which, in turn, became the dominant radicals. Whereas full band gap excitation from the valance to the conduction band may result in the original oxidative potential of the parent photocatalysts, the quantum efficiency is hampered because of the pronounced charge recombination at dopant-induced defect sites [
36].
Similar to metal-doping strategies, anionic co-doping, for example involving N/F-TiO
2 or N/F- and C/N-ZnWO
4 [
73,
74,
75] has been used to lower charge defects, but in many cases, the quantum efficiencies remain unmatched compared with the excited photocatalysts. Many studies, for example [
76,
77], have also reported that new conduction and valence bands are produced in a semiconductor when significant levels of foreign elements are introduced to the photocatalyst. Research using this band gap engineering approach has been carried out successfully and resulted in the emergence of new photocatalytic materials for water-splitting purposes including Bi
3+, Ti
4+ and W
6+ [
78,
79]. From the various studies carried out on doping and the effect of doping, it is obvious that the exploration of new photocatalytic doping materials will continue to be driven mainly by efforts in water splitting reactions although these materials will find relevance in other photocatalytic applications such as PbBi
2Nb
2O
9, for the treatment or remediation of aqueous pollutants, CaBi
2O
4 for the oxidation of gaseous pollutants and ZnGa
2O
4 for the reduction of carbon dioxide and similar materials [
77,
80,
81].
7. Semiconductor Films and Foams
In thin film deposition, a thin layer of material is prepared on a suitable substrate or a previously deposited layer. “Thin” is a relative term, since a film may look operationally thin or thick compared to the wavelength of the light used. Compared to conventional thin film forming processes such as evaporation, or sputtering, sol-gel processing makes it possible to control the microstructure of the deposited film including the pore volume, pore size, and surface area [
89] and uses mainly alkoxide, organic/ inorganic salts, metal oxides and other salts in a liquid phase chemical reaction. The method provides excellent chemical homogeneity and the possibility of deriving unique metastable structures at low reaction temperatures [
90]. The liquid phase reaction enhances the uniformity of the product produced and the process can be operated under ambient conditions. The reactant concentration, temperature and type of solvent will affect the quality of the final product.
In the production of TiO2 particles, calcinations at high temperatures are necessary to change the crystalline structure of TiO2 that possesses photo catalytic activity and the specific surface area, particle size, and other properties of TiO2 can be altered after calcinations at different temperatures. Thus thin films formed by dipping or spinning, use little raw materials and may be processed quickly without cracking and thus overcoming most of the disadvantages of sol-gel processing. In addition, large substrates may be accommodated and it is possible to uniformly coat both sides of planar and axially symmetric substrates such as pipes, tubes, rods and fibers which are not easily handled by more conventional coating processes.
There are two main ways to synthesize gels at room temperature: spin coating and dip coating. Spin coating is used for applications where relatively flat substrates or objects are coated with thin layers of material. On the other hand, the dip-coating process, involves immersion, start-up, deposition, evaporation and drainage. The substrate is slowly dipped into and withdrawn from a tank containing the sol, with a uniform velocity, in order to obtain a uniform coating. The gel samples are dried at 100 °C and then fired at 450 °C to remove organics from the film pyrotically. Once the sample has reached the desired thickness through multiple coatings, it is crystallized by heating. The maximum thickness of crack free coatings that can be achieved is 0.5 microns. Sol-gel and “doctor blade” TiO2 films vary in thickness from 0.2 microns to 0.8 microns.
A foam is a substance that is formed by trapping pockets of gas in a liquid or solid. Particle sizes, porosity differ for various materials depending on the synthetic routes chosen, we have synthesized TiO
2 foams (reported in nano-letters) of 100–500 nm in thickness. TiO
2 films similarly have various thicknesses ranging from a few microns to hundreds or even more depending on the synthetic conditions. We have produced sol-gel and “doctor blade” TiO
2 films varying in thickness from 0.2 microns to 0.8 microns. Generally, the porosity of the foam is larger than that of films (depending on the film and process conditions). Foams usually offer more diffusion pathways than films especially for solution phase degradation. Films on the other hand are usually easier to integrate into structured photocatalytic reactors and light absorption is better than in films. Foams usually get “clogged” at some stage during the photocatalytic process. The synthetic route for TiO
2 foam as reported in the literature [
91]. The thickness of the TiO
2 foam produced by this process was 100 nm and it should be pointed out that particle size of 500 nm is possible depending on the synthetic process. The TiO
2 foam offers extended porosity which is important for “light harvesting”,
Figure 4.
Figure 4.
Degradation of Benzene, Toluene and Xylene in contaminated air with TiO
2 Foam [
82,
86]. The porous structure results in “improved light harvesting” by the catalyst.
Figure 4.
Degradation of Benzene, Toluene and Xylene in contaminated air with TiO
2 Foam [
82,
86]. The porous structure results in “improved light harvesting” by the catalyst.
The TiO
2 foams have been used as catalyst materials in the degradation of low concentrations of benzene, toluene and xylene (BTX) in contaminated air. The degradation of anionic and cationic dyes in industrial effluents [
8,
82,
86] as shown in
Figure 5, an example given with methyl orange has been undertaken.
A metal can affect the surface properties of a semiconductor by generating a “barrier” which acts as an electron trap of the metal in contact with the semiconductor surface. Similarly, doping of transition metal ions to semiconductors improves the trapping of electron and inhibits e
−/h
+ recombination because the relatively low photoactivity of TiO
2 is believed to be due to the fast recombination of photogenerated electrons and holes. In recent years, researchers have focused on the combination of different kinds of metallic oxide particles. Oliviero
et al. [
89] showed that Ru/CeO
2 particle catalysts have very different behavior in the catalytic wet air oxidation of acrylic, succinic and acetic acids depending on the support morphology and the metal/support contact. The use of metal nanoparticles and hybrids has also been researched [
92] and the synthetic route for TiO
2/RuO
2/SiO
2 nanoparticles is reported elsewhere [
92]. These materials have been used in the degradation of low concentrations of anionic and cationic azo-dyes in industrial effluents,
Figure 6.
Figure 5.
The photocatalytic degradation of an anionic azo-dye in a UV irradiated porous titania foam [
8,
86]. The decrease in absorption peaks is an evidence of degradation. This evidence cannot however be conclusive (see section 4-on the use model compounds in this review).
Figure 5.
The photocatalytic degradation of an anionic azo-dye in a UV irradiated porous titania foam [
8,
86]. The decrease in absorption peaks is an evidence of degradation. This evidence cannot however be conclusive (see section 4-on the use model compounds in this review).
Figure 6.
The photocatalytic activity of TiO
2 foam and surface modified binary oxide nanoparticles [
8,
86]. As expected, a higher catalyst concentration results in increased photocatalytic efficiency until a maximum catalyst loading is reached.
Figure 6.
The photocatalytic activity of TiO
2 foam and surface modified binary oxide nanoparticles [
8,
86]. As expected, a higher catalyst concentration results in increased photocatalytic efficiency until a maximum catalyst loading is reached.
Recent work by Wei
et al. [
54] using hybrid Fe
2O
3/Pd nanoparticulate photocatalyst grown by epitaxial growth of Fe
2O
3 on Pd nanoparticles showed improved photocatalytic efficiency, although this was limited by the presence of organic stabilizers which are difficult to remove. Other materials considered in recent work include ZnS, ZnO, CdSe, Fe
2O
3 and InP [
93]. Some of these materials were reported to show some photocatalytic promise, but with uncertainty over their efficiencies and spectral characteristics. Fe
2O
3 is particularly interesting because of its stability against photo and chemical corrosion at neutral to basic pH and has a band gap energy of about 2.0 to 2.2 eV corresponding to the absorption of 564 to 620 nm light.
9. Applications of Photocatalysis
Selected applications of photocatalysis are given in
Table 1,
Table 2,
Table 3. Heterogeneous photocatalysis has been demonstrated as a low cost and sustainable technology for the treatment of a host of pollutants in air and water including organics and heavy metals,
etc., with Japan, USA, India and China as major users of this technology as partly demonstrated by the number of research publications in this area,
Figure 9 [
1]. Unlike reverse osmosis, nano and ultrafiltration, photocatalysis is a cheap and a potential “stand alone” technology for water treatment. As photocatalysis makes use of sunlight or UV radiation, the technology is inexpensive, environmentally friendly and can be applied worldwide. It requires minimal equipment, is highly deployable and appropriate for developing countries and remote sites with no access to electricity. Photocatalysis has also been used successfully in many developing nations to destroy pathogens [
5,
92] and algal blooms in fresh water supplies. Photodisinfection sensitized by TiO
2 has been used to degrade the green algae, treat humic substances [
100] which act as substrates for bacterial growth, inhibit bacterial degradation of impurities in natural water, and aid the transport of metals in the environment and complex with Fe, Pb, Mn, making it harder to remove them. Specific examples of applications are as follows.
Figure 9.
Number of publications pertaining to photocatalysis since 1970. China, Japan, USA and India have the edge in the number of publications on this Subject. Note: This figure is reproduced with permission from [
1], Copyright © 2012 American Chemical Society.
Figure 9.
Number of publications pertaining to photocatalysis since 1970. China, Japan, USA and India have the edge in the number of publications on this Subject. Note: This figure is reproduced with permission from [
1], Copyright © 2012 American Chemical Society.
Table 1.
Selected applications of photocatalysis. Note: this Table is reproduced with permission from [
92], Copyright © 2000 Elsevier Science S.A.
Table 1.
Selected applications of photocatalysis. Note: this Table is reproduced with permission from [92], Copyright © 2000 Elsevier Science S.A.
| Property | Category | Application |
|---|
| Self-cleaning | Materials for residential and office buildings | Exterior tiles, kitchen and bathroom components, interior furnishings, plastic surfaces, aluminum siding, building stone and curtains, paper window blinds |
| Indoor and outdoor lamps and related systems | Translucent paper for indoor lamp covers, coatings on fluorescent lamps and highway tunnel lamp cover glass |
| Materials for roads | Tunnel wall, soundproofed wall, traffic signs and reflectors |
| Others | Tent material, cloth for hospital garments and uniforms and spray coatings for cars |
| Air cleaning | Indoor air cleaners | Room air cleaner, photocatalyst-equipped air conditioners and interior air cleaner for factories |
| Outdoor air purifiers | Concrete for highways, roadways and footpaths, tunnel walls, soundproof walls and building walls |
| Water purification | Drinking water | River water, ground water, lakes and water-storage tanks |
| Others | Fish feeding tanks, drainage water and industrial wastewater |
| Antitumor activity | Cancer therapy | Endoscopic-like instruments |
| Self-sterilizing | Hospital | Tiles to cover the floor and walls of operating rooms, silicone rubber for medical catheters and hospital garments and uniforms |
| Others | Public rest rooms, bathrooms and rat breeding rooms |
Table 2.
Applications of super hydrophilic technology. Note: this Table is reproduced with permission from [
92], Copyright © 2000 Elsevier Science S.A.
Table 2.
Applications of super hydrophilic technology. Note: this Table is reproduced with permission from [92], Copyright © 2000 Elsevier Science S.A.
| Property | Category | Application |
|---|
| Self-cleaning | Roads | Tunnel lighting, tunnel walls, traffic signs and sound proof walls |
| Houses | Tiles on kitchen walls and bathrooms, exterior tiles, roofs and windows |
| Buildings | Aluminum panels, tiles, building stone, crystallized glass and glass film |
| Agriculture | Plastic and glass greenhouses |
| Electrical and electronic equipment | Computer displays and cover glass for solar cells |
| Vehicles | Paint work, coatings for exterior surfaces of windows and headlights |
| Daily necessities and consumer products | Tableware, kitchenware and spray-on anti-fouling coatings |
| Paint | General-purpose paints and coatings |
| Anti-fogging property | Roads | Road mirrors |
| Houses | Mirrors for bathrooms and dressers |
| Stores | Refrigerated showcases |
| Electrical and electronic equipment | Heat exchangers for air conditioners and high-voltage electric transmission equipment |
| Vehicles | Inside surfaces of windows, glass films, rear-view mirrors and windshields |
| Daily necessities and consumer products | Spray-on antifogging coatings and films |
| Paint | General-purpose paints and coatings |
| Optical instruments | Optical lenses |
| Bio-compatibility | Medical instruments and supplies | Contact lenses and catheters |
Table 3.
Waterborne microbial species known to be inactivated by solar disinfection [
5].
Table 3.
Waterborne microbial species known to be inactivated by solar disinfection [5].
| Microbe | Species |
|---|
| Bacteria | Campylobacter jejuni |
| Enterococcus sp. |
| Enteropathogenic E. coli |
| Mycobacterium avium |
| Mycobacterium intracellulare |
| Pseudomonas aeruginosa |
| Salmonella typhi |
| Salmonella typhimurium |
| Shigella dysenteriae type 1 |
| Shigella flexneri |
| Streptococcus faecalis |
| Staphylococcus epidermidis |
| Vibrio cholerae |
| Yersinia enterocolitica |
| Viruses | Bacteriophagef2 |
| Encephalomyocarditis virus |
| Polio virus |
| Rotavirus |
| Coxsackie virus A/B |
| Hepatitis A |
| Protozoa | Acanthamoeba polyphaga (cyst ) |
| Cryptosporidium parvum (oocyst) |
| Entamoeba sp. (cysts) |
| Giardia sp.(cysts) |
| Naegleria sp. |
9.1. Water Treatment
Growth in the global population, the diminishing supply of clean water, heightened environmental concerns, and the strong link between water quality and human health require the identification and employment of effective sustainable water treatments to meet the urgent global need for clean water. Advanced oxidation processes (AOPs) have shown tremendous promise in water purification and treatment, including for the destruction of naturally occurring toxins, contaminants of emerging concern, pesticides, and other deleterious contaminants. One of the first references to AOPs was by Glaze in 1987 as processes that involve the generation of hydroxyl radicals in sufficient quantity to affect water purification. The definition and development of AOPs have evolved since the 1990s and include a variety of methods for generating hydroxyl radical and other reactive oxygen species including superoxide anion radical, hydrogen peroxide, and singlet oxygen. However, hydroxyl radical is still the species most commonly tied to the effectiveness of AOPs. Most organic compounds react with hydroxyl radical by addition or hydrogen abstraction pathways to form a carbon-centered radical. The resulting carbon-centred radical reacts with molecular oxygen to form a peroxyl radical that undergoes subsequent reactions, ultimately producing a host of oxidation products such as ketones, aldehydes and alcohols [
21].
Hydroxyl radical can also abstract an electron from electron-rich substrates to form a radical cation, which in aqueous media, is readily hydrolyzed, ultimately leading to an oxidized product. The oxidation products are often less toxic and more susceptible to bioremediation. AOPs include UV/O3, UV/H2O2, Fenton, photo-Fenton, non-thermal plasmas, sonolysis, photocatalysis, radiolysis and supercritical water oxidation processes. Problematic substances in wastewater can include organic matter and/or different trace contaminants and industrial wastewater may also contain heavy loads of metals or organic compounds and these cannot be treated by disinfection. In drinking water production microbe contamination is a problem especially in developing countries and remote locations without access to a centralized drinking water supply.
In Europe, conventional technologies for wastewater treatment are in most cases able to meet the current water quality standards. The chances and potential fields of application of photocatalytic systems with artificial UV-sources include new water treatment plants or plants where conventional methods need to be replaced and the treatment of water contaminated with trace contaminants such as estrogens, the treatment of industrial wastewater contaminated with high loads of organic compounds or metals as well as small scale systems, for example, for the disinfection of swimming pools [
92,
100].
Globally, 1 billion people lack access to safe water supplies and 2.6 billion are without access to basic sanitation [
5]. This is especially true for the least developed regions of Asia, Central and South America, and Africa, innovative methods for water treatment are needed urgently. In the most developed markets such as the USA, Canada, Japan, and most of Western Europe, the success of a new water treatment method is mainly based on its ability to improve the quality of drinking water and/or to reduce water contamination. The beneficial effects of clean water are obvious. Most importantly, the improvement of water supply, sanitation, hygiene, and management of water resources could prevent almost one-tenth of all diseases worldwide. Nanotechnology is one of the most promising emerging technologies for efficient, economical and environmentally friendly water and waste water treatment offering great potential for manufacturers in Europe.
The demand for water treatment products globally reached $44.6 billion in 2008 and it is predicted, by Freedonia Group Inc., to increase annually by 5.7% reaching $59 billion by 2013 [
5]. The fastest annual growth was predicted to be in large developing countries like China and India due to rapid industrialization and increased efforts to expand access to safe water supplies and adequate sanitation facilities especially in rural areas. The worldwide turnover of nanotechnological applications in water and wastewater treatment reached $1.6 billion in 2007 and was predicted to increase to $6.6 billion in 2015. In 2015 the leading countries in water treatment with nanoparticle-based methods are the USA, Germany, Japan, and China [
101]. Disinfection is one of the fastest growing market segments with broad applications and benefits; photocatalysis with nano-catalysts is a promising method for disinfection. In addition, photocatalysts combined with filtration membranes can reduce membrane fouling and thus enhance water cleaning efficiency significantly. Small-scale photocatalytic systems with artificial UV-light have already been on the market for several years whereas solar photocatalytic water treatment plants are at a demonstration phase and pilot projects for drinking water purification in developing countries have only just started.
Recently, humic substance was also decomposed both in highly saline and natural seawater using different photocatalytic materials. The decomposition rate of humic substances in seawater was slow compared with pure water media and no toxic byproducts were detected during the decomposition. Minero and other workers [
29,
100,
102,
103,
104] studied the decomposition of dodecane and toluene in crude oil in seawater media and found that no chlorinated compounds were detected during the irradiation. Complete decomposition was achieved after a few hours of irradiation. Another study conducted on the decomposition of seawater-soluble crude-oil fractions found that it can be decomposed under illumination of nanoparticles of TiO
2 using artificial light [
47].
Photocatalysis can also be used to destroy bacteria and viruses [
92,
100,
105]. The increasing incidence of algal blooms in fresh water supplies and the consequent possibility of cyanobacterial microcystin contamination of potable water
Microcystin toxins are also degraded on immobilized titanium dioxide catalyst. Photodisinfection sensitized by TiO
2 had some effect on the degradation of the green algae which has a thick cell wall. This is being used to great advantage in many developed and developing nations to treat water especially in remote and disaster areas without portable water supply or electricity, as shown in
Figure 10.
Figure 10.
Inactivation of bacteria Ecoli K-12 in drinking water exposed to solar irradiation. Adapted from EU Contract Number INCO-DEV 031650 (2006) [
5].
Figure 10.
Inactivation of bacteria Ecoli K-12 in drinking water exposed to solar irradiation. Adapted from EU Contract Number INCO-DEV 031650 (2006) [
5].
Specific successful applications have been in the following areas, amongst others.
9.3. Destruction of Organics
Photocatalysis has been used for the destruction of organic compounds such as alcohols, carboxylic acids, phenolic derivatives, or chlorinated aromatics, into harmless products, for example, carbon dioxide, water, and simple mineral acids. Water contaminated by oil can be treated efficiently by photocatalytic reaction. Herbicides and pesticides that may contaminate water such as 2,4,5 trichlorophenoxyacetic acid, 2,4,5 trichlorophenol,
S-triazine herbicides and 1,1,1-trichloro-2,2-di(4-chlorophenyl)ethane (DDT) have also been successfully degraded [
22].
9.4. Removal of Inorganic Compounds
In addition to organic compounds, wide ranges of inorganic compounds are sensitive to photochemical transformation on the catalyst surfaces. Inorganic species such as bromate, or chlorate, azide, halide ions, nitric oxide, palladium and rhodium species, and sulfur species can be decomposed. Metal salts such as AgNO
3, HgCl and organometalic compound (e.g., CH
3HgCl) can be removed from water as well as cyanide, thiocyanate, ammonia, nitrates and nitrites [
22].
9.5. Degradation of Natural Organic Matter
Humic substances have also been degraded photochemically. Humic are naturally occurring biogenic heterogeneous organic substances characterized as being yellow brown and having high molecular weights They can also be described as the fraction of filtered water that adsorbs on non-ionic polymeric adsorbent at pH 2. They are the main constituents of the dissolved organic carbon pool in surface and ground waters, imparting a yellowish-brown color to the water system. The concentration of humic substances varies from place to place, the values in seawater being normally from 2–3 mg/L. Humic substances affect the behaviour of some pollutants in natural environments, such as trace metal speciation and toxicity, solubilization and adsorption of hydrophobic pollutants. They act as substrates for bacterial growth; inhibit the bacterial degradation of impurities in natural water, complex with heavy metals such as Fe, Pb, Mn making it harder to remove them. Advanced oxidation has been used to decrease the organic content in water including humic acid [
100] and it has the advantage of not leaving any toxic byproducts or sludge.
Bekbolet and Ozkosemen [
103] investigated the photocatalytic degradation using humic acid as a model and observed that after 1 h irradiation and in the presence of 1.0 g/L TiO
2 (P25), 40% TOC and 75% of the colour (400 nm) were removed. On the other hand, Eggins and coworkers found the suspension of TiO
2 (P25) irradiated by a mercury lamp showed a very efficient reduction of humic acid concentration of about 50% in 12 min. Heterogeneous photocatalysis has also been coupled with other physical methods in order to increase the degradation rate of organic molecules including humic substances [
100,
103].
9.6. Medical Applications
Application in TiO
2 Fabrics is a major and important application of TiO
2 photocatalysis. The ability of TiO
2 to disinfect microbes, viruses and bacteria has been put into good used by Japanese researchers. Hospital garments worn by doctors and nurses have “doses” of TiO
2 added to the fabric during processing operations and the fabric are used to make hospital garments that are worn to control hospital infections, including MRSA. Many lives have been lost because of methicillin resistant
Staphylococcus (MRSA) and research is concentrating efforts on “TiO
2 fabrics” as well as the use of antimicrobial photodynamic therapy (APDT) to decolonize MRSA from patients [
92].
9.7. Application Photodynamic Therapy
Targeting cancer such as colon or kidney cancer (tumor therapy) in an organism with a source of irradiation is easy. However, tumors in rats have shown to respond to PDT treatment. Basically, TiO
2 is introduced in the site of the cancer and illuminated by and is photo sensitized using an optic fiber cable to introduce the illumination [
92]. The activation of the photo sensitizer on illumination creates reactive oxygen species that kill the surrounding cells of the tumor.
9.8. Applications in Construction
TiO
2 cement (“pellite cement”) containing TiO
2 has been used in road construction (tunnels) in some European countries to control exhaust emissions due principally to NO and NO
2. These are especially severe in summer months especially in dense and large urban cities with high traffic levels. This application however, is not new to the market [
106].