Integrin Activation Contributes to Lower Cisplatin Sensitivity in MV3 Melanoma Cells by Inducing the Wnt Signalling Pathway
Abstract
:1. Introduction
2. Results
2.1. Integrin Activation in MV3 Melanoma Leads to Decreased Sensitivity against Cisplatin Cytotoxicity
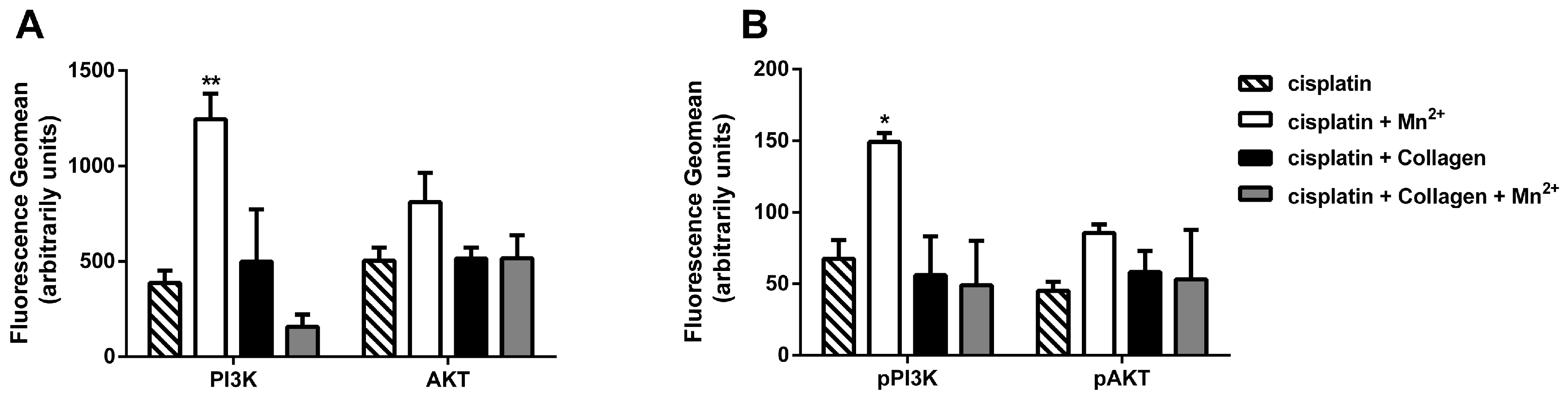
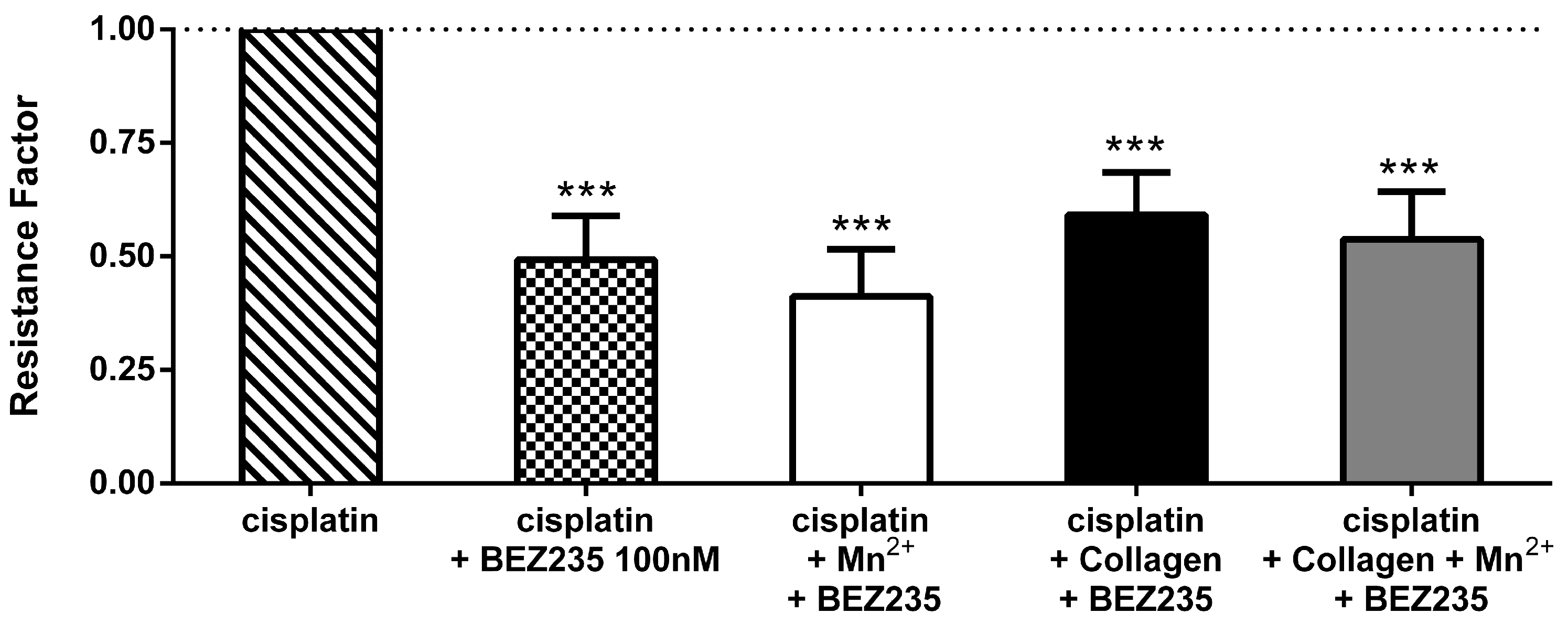
2.2. Resistance of MV3 Cells Is Mediated through the PI3K/AKT pathway
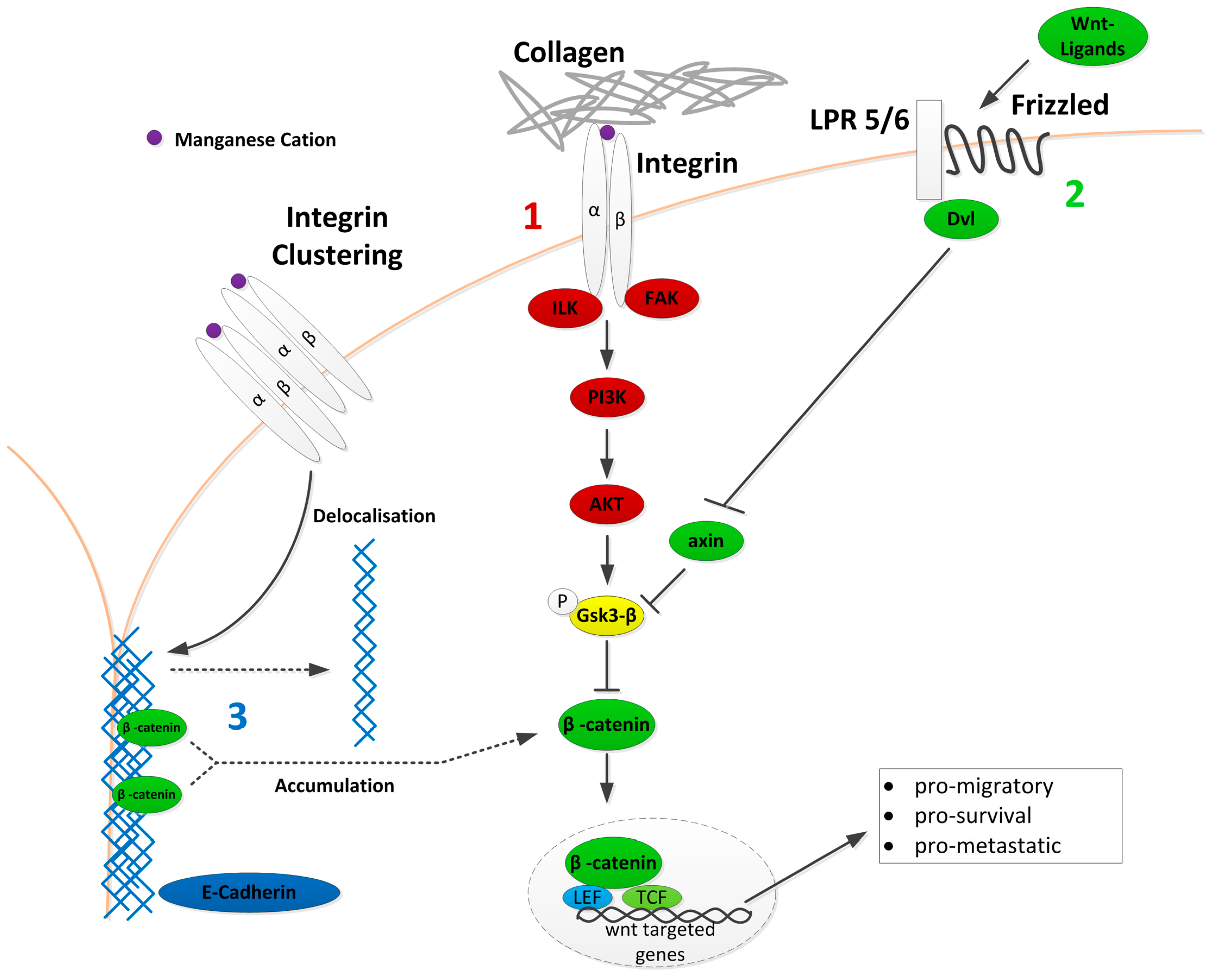
2.3. Wnt Signalling as Keystone between Integrins and Chemoresistance in MV3 Cells
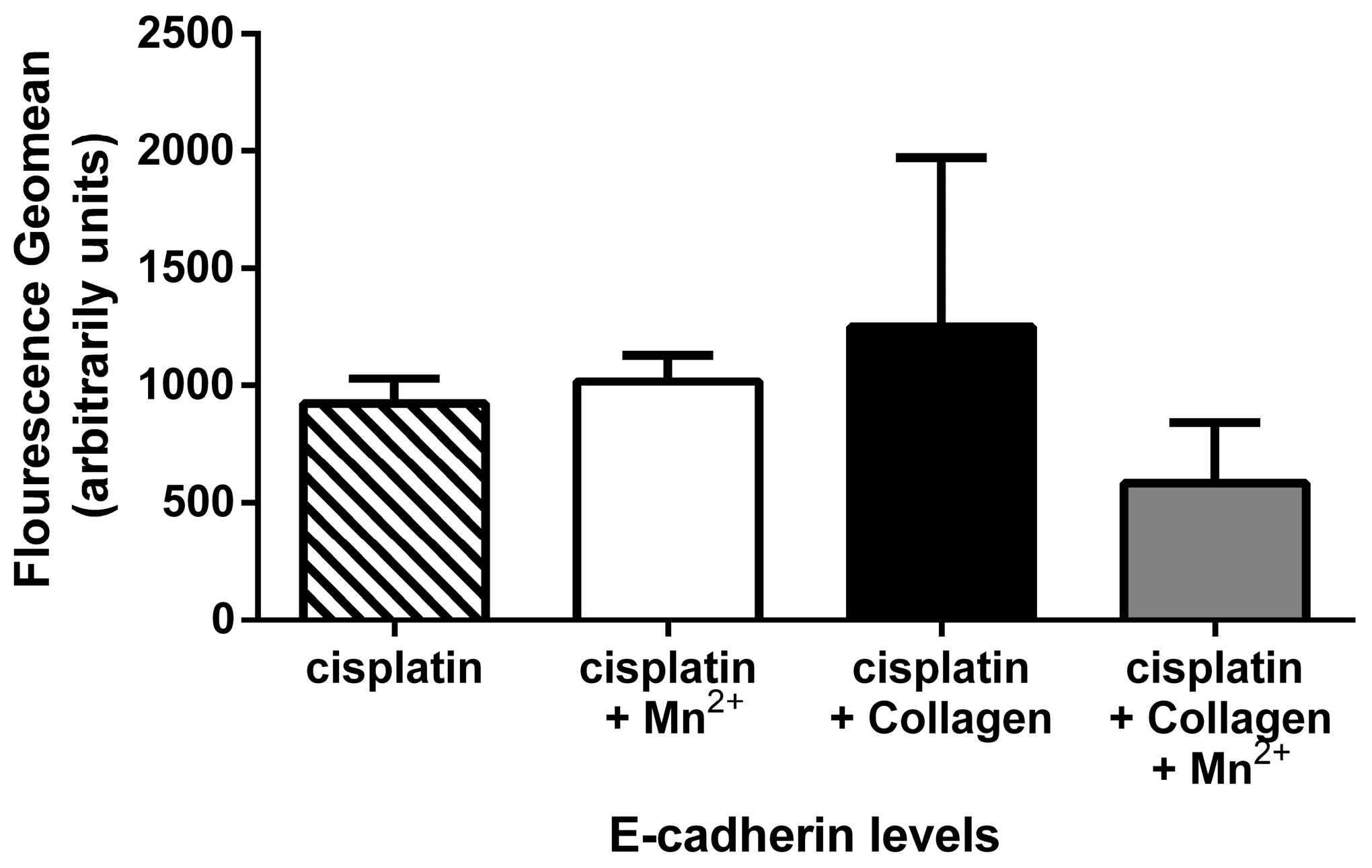
2.4. Integrin Activation and Clustering Does Not Influence E-Cadherin Expression
2.5. Wnt as Mechanism of Integrin Mediated Loss of Sensitivity
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Reagents
4.2. Microscopy
4.3. MTT Assay
4.4. Flow Cytometry
4.5. Proteome Profiler™ Antibody Array
4.6. Western Blot
4.7. TOPflash Assay
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Desgrosellier, J.S.; Cheresh, D.A. Integrins in cancer: Biological implications and therapeutic opportunities. Nat. Rev. Cancer 2010, 10, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Kinashi, T. Intracellular signalling controlling integrin activation in lymphocytes. Nat. Rev. Immunol. 2005, 5, 546–559. [Google Scholar] [CrossRef] [PubMed]
- Shattil, S.J.; Kim, C.; Ginsberg, M.H. The final steps of integrin activation: The end game. Nat. Rev. Mol. Cell Biol. 2010, 11, 288–300. [Google Scholar] [CrossRef] [PubMed]
- Bewick, M.; Lafrenie, R. Adhesion dependent signalling in the tumour microenvironment: The future of drug targetting. Curr. Pharm. Des. 2006, 12, 2833–2848. [Google Scholar] [CrossRef] [PubMed]
- Schmidmaier, R.; Baumann, P. ANTI-ADHESION evolves to a promising therapeutic concept in oncology. Curr. Med. Chem. 2008, 15, 978–990. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, U.B.; Westphal, J.R.; Van Kraats, A.A.; Ruiter, D.J.; Van Muijen, G.N. Expression of integrin alpha(v)beta(3) correlates with activation of membrane-type matrix metalloproteinase-1 (MT1-MMP) and matrix metalloproteinase-2 (MMP-2) in human melanoma cells in vitro and in vivo. Int. J. Cancer 2000, 87, 12–19. [Google Scholar] [CrossRef]
- Nikkola, J.; Vihinen, P.; Vlaykova, T.; Hahka-Kemppinen, M.; Heino, J.; Pyrhönen, S. Integrin chains beta1 and alphav as prognostic factors in human metastatic melanoma. Melanoma Res. 2004, 14, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.C.; Staniszewska, I.; Del Valle, L.; Tuszynski, G.P.; Marcinkiewicz, C. Angiostatic activity of obtustatin as α1β1 integrin inhibitor in experimental melanoma growth. Int. J. Cancer 2008, 123, 2195–2203. [Google Scholar] [CrossRef] [PubMed]
- Pickarski, M.; Gleason, A.; Bednar, B.; Duong, L. Orally active αvβ3 integrin inhibitor MK-0429 reduces melanoma metastasis. Oncol. Rep. 2015, 33, 2737–2745. [Google Scholar] [CrossRef] [PubMed]
- Meads, M.B.; Gatenby, R.A.; Dalton, W.S. Environment-mediated drug resistance: A major contributor to minimal residual disease. Nat. Rev. Cancer 2009, 9, 665–674. [Google Scholar] [CrossRef] [PubMed]
- Shishido, S.; Bönig, H.; Kim, Y.-M. Role of integrin alpha4 in drug resistance of leukemia. Front. Oncol. 2014, 4, 99. [Google Scholar] [CrossRef] [PubMed]
- Engelman, J.A. Targeting PI3K signalling in cancer: Opportunities, challenges and limitations. Nat. Rev. Cancer 2009, 9, 550–562. [Google Scholar] [CrossRef] [PubMed]
- Sulzmaier, F.J.; Jean, C.; Schlaepfer, D.D. FAK in cancer: Mechanistic findings and clinical applications. Nat. Rev. Cancer 2014, 14, 598–610. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Guan, J.L. Signal transduction by focal adhesion kinase in cancer. Cancer Metastasis Rev. 2009, 28, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Parsons, J.T. Focal adhesion kinase: The first ten years. J. Cell Sci. 2003, 116, 1409–1416. [Google Scholar] [CrossRef] [PubMed]
- Schaller, M.D. Cellular functions of FAK kinases: Insight into molecular mechanisms and novel functions. J. Cell Sci. 2010, 123, 1007–1013. [Google Scholar] [CrossRef] [PubMed]
- Eke, I. β1 Integrin/FAK/cortactin signalling is essential for human head and neck cancer resistance to radiotherapy. J. Clin. Investig. 2012, 122, 1529–1540. [Google Scholar] [CrossRef] [PubMed]
- Lane, D.; Goncharenko-Khaider, N.; Rancourt, C.; Piche, A. Ovarian cancer ascites protects from TRAIL-induced cell death through alphavbeta5 integrin-mediated focal adhesion kinase and Akt activation. Oncogene 2010, 29, 3519–3531. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y. Role of focal adhesion kinase in regulating YB 1 mediated paclitaxel resistance in ovarian cancer. J. Natl. Cancer Inst. 2013, 105, 1485–1495. [Google Scholar] [CrossRef] [PubMed]
- Halder, J. Therapeutic efficacy of a novel focal adhesion kinase inhibitor TAE226 in ovarian carcinoma. Cancer Res. 2007, 67, 10976–10983. [Google Scholar] [CrossRef] [PubMed]
- Manning, B.D.; Toker, A. AKT/PKB Signalling: Navigating the network. Cell 2017, 169, 381–405. [Google Scholar] [CrossRef] [PubMed]
- Ruan, G.-X.; Kazlauskas, A. Focus on molecules: Akt (PKB). Exp. Eye Res. 2011, 93, 570–571. [Google Scholar] [CrossRef] [PubMed]
- Vanhaesebroeck, B.; Guillermet-Guibert, J.; Graupera, M.; Bilanges, B. The emerging mechanisms of isoform-specific PI3K signalling. Nat. Rev. Mol. Cell Biol. 2010, 11, 329–341. [Google Scholar] [CrossRef] [PubMed]
- Ter Haar, E.; Coll, J.T.; Austen, D.A.; Hsiao, H.M.; Swenson, L.; Jain, J. Structure of GSK3beta reveals a primed phosphorylation mechanism. Nat. Struct. Biol. 2001, 8, 593–596. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, B.T.; Tamai, K.; He, X. Wnt/beta-catenin signalling: Components, mechanisms, and diseases. Dev. Cell 2009, 17, 9–26. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Fukunaga-Kalabis, M.; Li, L.; Herlyn, M. Developmental pathways activated in melanocytes and melanoma. Arch. Biochem. Biophys. 2014, 563, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Spranger, S.; Bao, R.; Gajewski, T.F. Melanoma-intrinsic β-catenin signalling prevents anti-tumour immunity. Nature 2015, 523, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Shah, K.V.; Chien, A.J.; Yee, C.; Moon, R.T. CTLA-4 Is a direct target of Wnt/β-catenin signalling and is expressed in human melanoma tumors. J. Investig. Dermatol. 2008, 128, 2870–2879. [Google Scholar] [CrossRef] [PubMed]
- Arend, R.C.; Londoño-Joshi, A.I.; Straughn, J.M.; Buchsbaum, D.J. The Wnt/β-catenin pathway in ovarian cancer: A review. Gynecol. Oncol. 2013, 131, 772–779. [Google Scholar] [CrossRef] [PubMed]
- Chiurillo, M.A. Role of the Wnt/β-catenin pathway in gastric cancer: An in-depth literature review. World J. Exp. Med. 2015, 5, 84. [Google Scholar] [CrossRef] [PubMed]
- Clevers, H.; Nusse, R. Wnt/β-catenin signalling and disease. Cell 2012, 149, 1192–1205. [Google Scholar] [CrossRef] [PubMed]
- Ohama, T.; Wang, L.; Griner, E.M.; Brautigan, D.L. Protein Ser/Thr phosphatase-6 is required for maintenance of E-cadherin at adherens junctions. BMC Cell Biol. 2013, 14, 42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinto, M.; Sotomayor, P.; Carrasco-Avino, G.; Corvalan, A.; Owen, G. Escaping antiangiogenic therapy: Strategies employed by cancer cells. Int. J. Mol. Sci. 2016, 17, 1489. [Google Scholar] [CrossRef] [PubMed]
- Burkhalter, R.J.; Symowicz, J.; Hudson, L.G.; Gottardi, C.J.; Stack, M.S. Integrin regulation of beta-catenin signalling in ovarian carcinoma. J. Biol. Chem. 2011, 286, 23467–23475. [Google Scholar] [CrossRef] [PubMed]
- Maira, S.-M.; Stauffer, F.; Brueggen, J.; Furet, P.; Schnell, C.; Fritsch, C.; Brachmann, S.; Chene, P.; De Pover, A.; Schoemaker, K.; et al. Identification and characterization of NVP-BEZ235, a new orally available dual phosphatidylinositol 3-kinase/mammalian target of rapamycin inhibitor with potent in vivo antitumor activity. Mol. Cancer Ther. 2008, 7, 1851–1863. [Google Scholar] [CrossRef] [PubMed]
- Kolev, V.N.; Tam, W.F.; Wright, Q.G.; McDermott, S.P.; Vidal, C.M.; Shapiro, I.M.; Xu, Q.; Wicha, M.S.; Pachter, J.A.; Weaver, D.T. Inhibition of FAK kinase activity preferentially targets cancer stem cells. Oncotarget 2017, 8, 51733–51747. [Google Scholar] [CrossRef] [PubMed]
- Kasaian, J.; Mosaffa, F.; Behravan, J.; Masullo, M.; Piacente, S.; Iranshahi, M. Modulation of multidrug resistance protein 2 efflux in the cisplatin resistance human ovarian carcinoma cells A2780/RCIS by sesquiterpene coumarins: MRP2 inhibition by sesquiterpene coumarins. Phytother. Res. 2016, 30, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Robinson, A.R.; Duensing, A.; van Drunen, E.; Beverloo, H.B.; Weisberg, D.B.; Hasty, P.; Hoeijmakers, J.H.J.; Niedernhofer, L.J. ERCC1-XPF endonuclease facilitates DNA double-strand break repair. Mol. Cell. Biol. 2008, 28, 5082–5092. [Google Scholar] [CrossRef] [PubMed]
- Chaney, S.G.; Sancar, A. DNA repair: Enzymatic mechanisms and relevance to drug response. J. Natl. Cancer Inst. 1996, 88, 1346–1360. [Google Scholar] [CrossRef] [PubMed]
- Pfankuchen, D.B.; Stölting, D.P.; Schlesinger, M.; Royer, H.-D.; Bendas, G. Low molecular weight heparin tinzaparin antagonizes cisplatin resistance of ovarian cancer cells. Biochem. Pharmacol. 2015, 97, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Beurel, E.; Grieco, S.F.; Jope, R.S. Glycogen synthase kinase-3 (GSK3): Regulation, actions, and diseases. Pharmacol. Ther. 2015, 148, 114–131. [Google Scholar] [CrossRef] [PubMed]
- Luo, J. Glycogen synthase kinase 3β (GSK3β) in tumorigenesis and cancer chemotherapy. Cancer Lett. 2009, 273, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Dormond, O.; Ponsonnet, L.; Hasmim, M.; Foletti, A.; Rüegg, C. Manganese-induced integrin affinity maturation promotes recruitment of αVβ3 integrin to focal adhesions in endothelial cells: Evidence for a role of phosphatidylinositol 3-kinase and Src. Thromb. Haemost. 2004, 92, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Chen, J. The regulation of integrin function by divalent cations. Cell Adhes. Migr. 2012, 6, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Van Muijen, G.N.; Jansen, K.F.; Cornelissen, I.M.; Smeets, D.F.; Beck, J.L.; Ruiter, D.J. Establishment and characterization of a human melanoma cell line (MV3) which is highly metastatic in nude mice. Int. J. Cancer 1991, 48, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Weinreb, P.H.; Li, S.; Gao, S.X.; Liu, T.; Pepinsky, R.B.; Caravella, J.A.; Lee, J.H.; Woods, V.L. Dynamic structural changes are observed upon collagen and metal ion binding to the integrin α1 I domain. J. Biol. Chem. 2012, 287, 32897–32912. [Google Scholar] [CrossRef] [PubMed]
- Sherman-Baust, C.A.; Weeraratna, A.T.; Rangel, L.B.A.; Pizer, E.S.; Cho, K.R.; Schwartz, D.R.; Shock, T.; Morin, P.J. Remodeling of the extracellular matrix through overexpression of collagen VI contributes to cisplatin resistance in ovarian cancer cells. Cancer Cell 2003, 3, 377–386. [Google Scholar] [CrossRef]
- Tang, N.; Wang, X.; Huang, T.; Wu, Y.; Chen, Y. Role of Ets-1 in fibronectin-derived heparin-binding domain polypeptides alleviating melanoma cell invasiveness and chemoresistance. Exp. Dermatol. 2014, 23, 512–513. [Google Scholar] [CrossRef] [PubMed]
- Lydolph, M.C.; Morgan-Fisher, M.; Høye, A.M.; Couchman, J.R.; Wewer, U.M.; Yoneda, A. Alpha9beta1 integrin in melanoma cells can signal different adhesion states for migration and anchorage. Exp. Cell Res. 2009, 315, 3312–3324. [Google Scholar] [CrossRef] [PubMed]
- Gerber, U.; Hoß, S.G.; Shteingauz, A.; Jüngel, E.; Jakubzig, B.; Ilan, N.; Blaheta, R.; Schlesinger, M.; Vlodavsky, I.; Bendas, G. Latent heparanase facilitates VLA-4-mediated melanoma cell binding and emerges as a relevant target of heparin in the interference with metastatic progression. Semin. Thromb. Hemost. 2015, 41, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Hui, T.; Sørensen, E.S.; Rittling, S.R. Osteopontin binding to the alpha 4 integrin requires highest affinity integrin conformation, but is independent of post-translational modifications of osteopontin. Matrix Biol. 2015, 41, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Koch, M.; Krieger, M.L.; Stölting, D.; Brenner, N.; Beier, M.; Jaehde, U.; Wiese, M.; Royer, H.-D.; Bendas, G. Overcoming chemotherapy resistance of ovarian cancer cells by liposomal cisplatin: Molecular mechanisms unveiled by gene expression profiling. Biochem. Pharmacol. 2013, 85, 1077–1090. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, C.M.V.; Bincoletto, C.; Barros, C.C.; Ferreira, A.T.; Paredes-Gamero, E.J. PLCγ2 and PKC are important to myeloid lineage commitment triggered by M-SCF and G-CSF. J. Cell. Biochem. 2014, 115, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Ross, T.; Jakubzig, B.; Grundmann, M.; Massing, U.; Kostenis, E.; Schlesinger, M.; Bendas, G. The molecular mechanism by which saturated lysophosphatidylcholine attenuates the metastatic capacity of melanoma cells. FEBS Open Bio. 2016, 6, 1297–1309. [Google Scholar] [CrossRef] [PubMed]



| Treatment | pIC50 ± SEM | IC50 (μM) | RF |
|---|---|---|---|
| Control | 5.00 ± 0.32 | 10.0 | - |
| Mn2+ | 4.95 ± 0.34 | 11.3 | 1.1 |
| Control | 4.96 ± 0.18 | 10.9 | - |
| Collagen | 4.84 ± 0.21 | 14.5 | 1.2 |
| Control | 4.88 ± 0.27 | 13.1 | - |
| Collagen + Mn2+ | 4.83 ± 0.29 | 14.9 | 1.1 |
| Treatment | pIC50 ± SEM | IC50 (μM) |
|---|---|---|
| Control | 4.55 ± 0.33 | 28.3 |
| BEZ235 100 nM | 4.87 ± 0.31 | 13.4 |
| Mn2+ + BEZ235 | 4.92 ± 0.28 | 12.0 |
| Collagen + BEZ235 | 4.83 ± 0.27 | 14.6 |
| Collagen + Mn2+ + BEZ235 | 4.89 ± 0.31 | 12.8 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piva, M.B.R.; Jakubzig, B.; Bendas, G. Integrin Activation Contributes to Lower Cisplatin Sensitivity in MV3 Melanoma Cells by Inducing the Wnt Signalling Pathway. Cancers 2017, 9, 125. https://doi.org/10.3390/cancers9090125
Piva MBR, Jakubzig B, Bendas G. Integrin Activation Contributes to Lower Cisplatin Sensitivity in MV3 Melanoma Cells by Inducing the Wnt Signalling Pathway. Cancers. 2017; 9(9):125. https://doi.org/10.3390/cancers9090125
Chicago/Turabian StylePiva, Maria B. R., Bastian Jakubzig, and Gerd Bendas. 2017. "Integrin Activation Contributes to Lower Cisplatin Sensitivity in MV3 Melanoma Cells by Inducing the Wnt Signalling Pathway" Cancers 9, no. 9: 125. https://doi.org/10.3390/cancers9090125






