26,26,26,27,27,27-Hexadeuterated-1,25-Dihydroxyvitamin D3 (1,25D-d6) As Adjuvant of Chemotherapy in Breast Cancer Cell Lines
Abstract
:1. Introduction

2. Results and Discussion
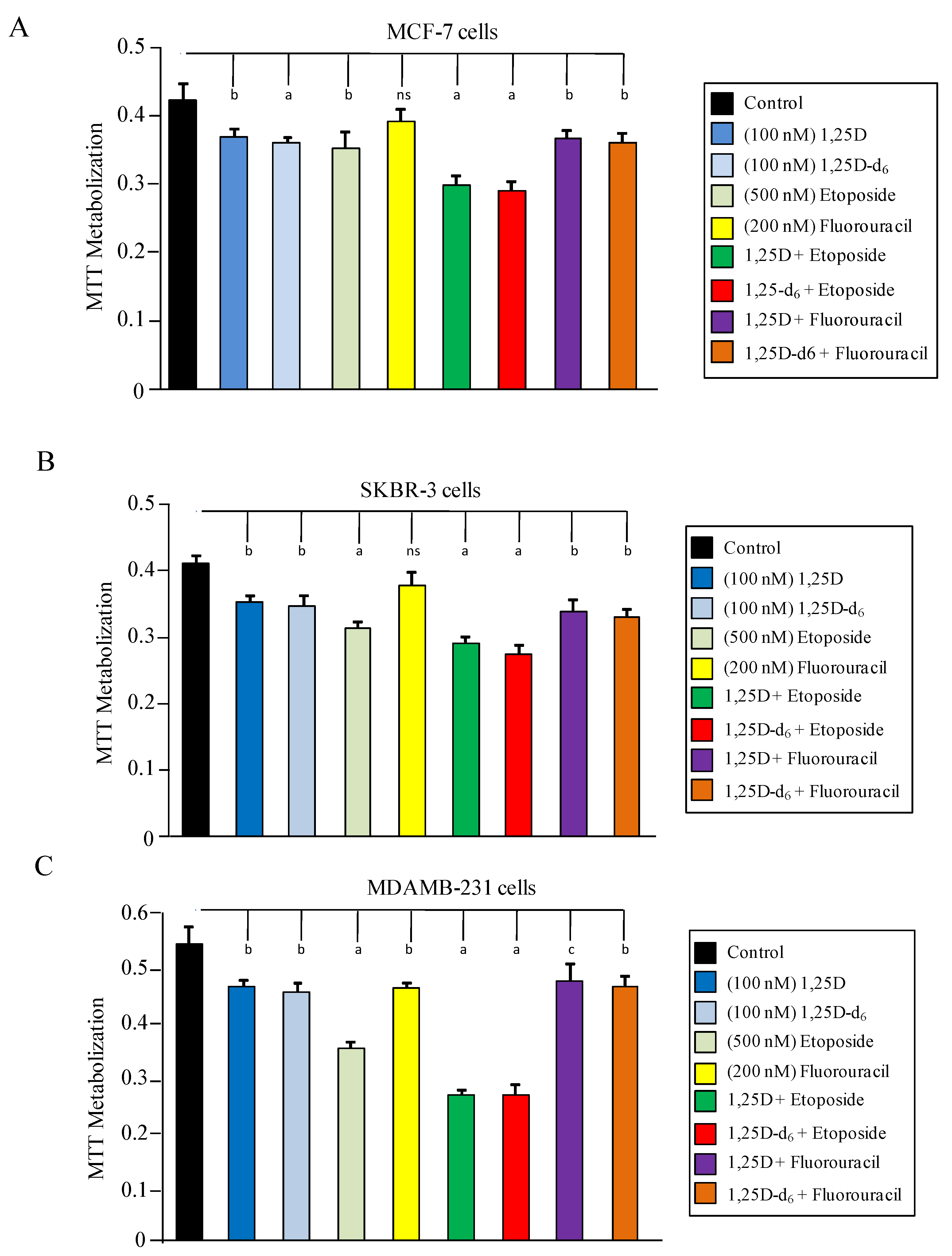
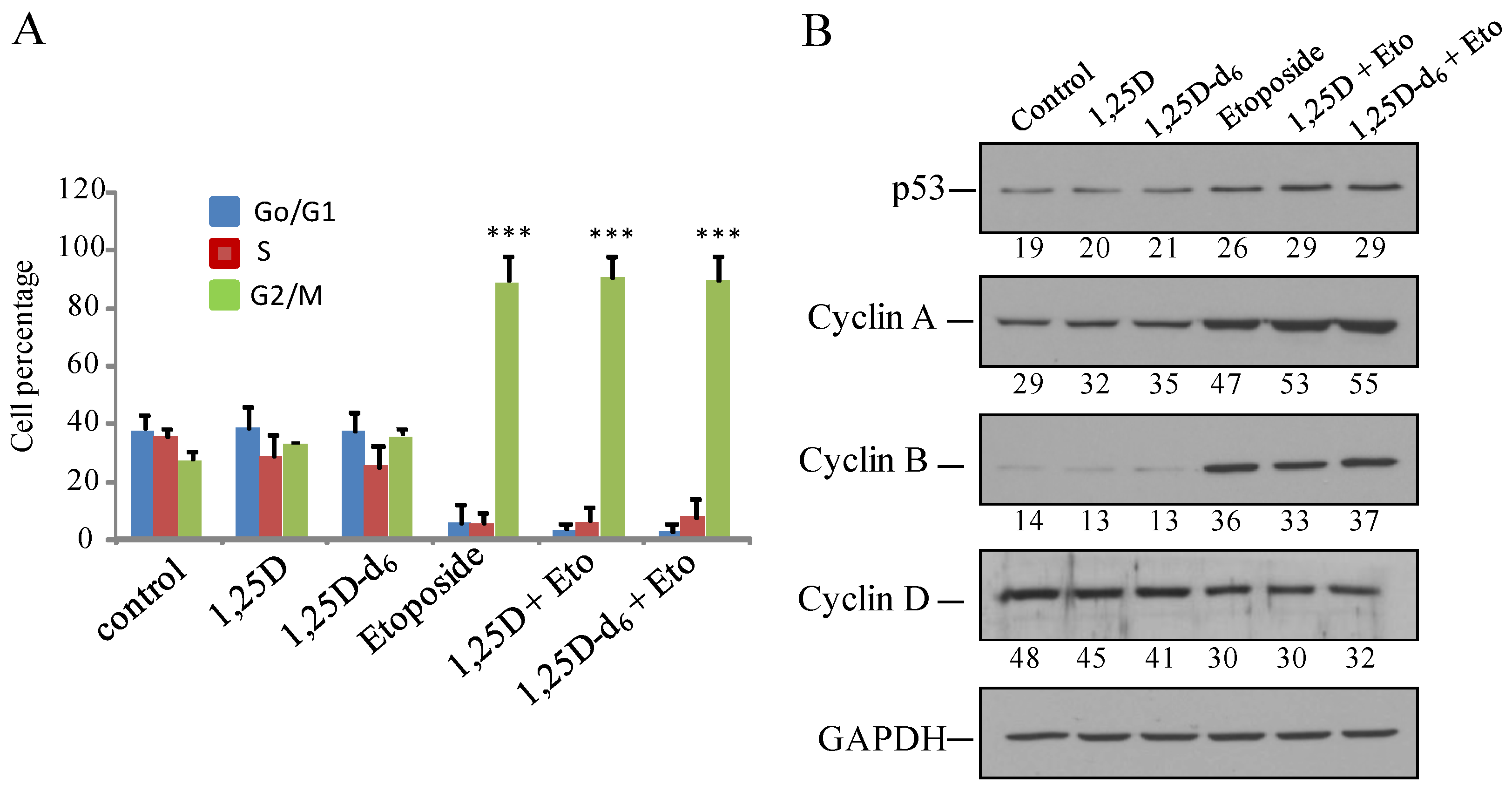


3. Experimental Section
3.1. Cell Culture and Drugs
3.2. Treatments
3.3. MTT Metabolization
3.4. Cell Cycle and Apoptosis Assays
3.5. Western Blot Assays
3.6. Three-Dimensional Cell Culture
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Van Leeuwen, J.; van Driel, M.; Feldman, D.; Muñoz, A. Vitamin D: Cancer and differentiation. In Vitamin D, 3rd ed.; Feldman, D., Pike, J.W., Adams, J.S., Eds.; Elsevier Academic Press: San Diego, CA, USA, 2011; Volume 2, pp. 1591–1624. [Google Scholar]
- Cui, Y.; Rohan, T.E. Vitamin D, calcium, and breast cancer risk: A review. Cancer Epidemiol. Biomarkers Prev. 2006, 15, 1427–1437. [Google Scholar] [CrossRef]
- Colston, K.W.; Lowe, L.C.; Mansi, J.L.; Campbell, M.J. Vitamin D status and breast cancer risk. Anticancer Res. 2006, 26, 2573–2580. [Google Scholar]
- Schwartz, G.; Skinner, H.G. Vitamin D status and cancer: New insights. Curr. Opin. Clin. Nutr. Metab. Care. 2007, 10, 6–11. [Google Scholar]
- Wu, G.; Fan, R.S.; Li, W.; Ko, T.C.; Brattain, M.G. Modulation of cell cycle control by vitamin D3 and its analogue, EB1089, in human breast cancer cells. Oncogene 1997, 15, 1555–1563. [Google Scholar]
- Verlinden, L.; Verstuyf, A.; Convents, R.; Marcelis, S.; van Camp, M.; Bouillon, R. Action of 1,25(OH)2D3 on the cell cycle genes, cyclin D1, p21 and p27 in MCF-7 cells. Mol. Cell Endocrinol. 1998, 142, 57–65. [Google Scholar] [CrossRef]
- Welsh, J. Induction of apoptosis in breast cancer cells in response to vitamin D and antiestrogens. Biochem. Cell Biol. 1994, 72, 537–545. [Google Scholar] [CrossRef]
- Simboli-Campbell, M.; Narvaez, C.J.; Tenniswood, M.; Welsh, J. 1,25-Dihydroxyvitamin D3 induces morphological and biochemical markers of apoptosis in MCF-7 breast cancer cells. J. Steroid. Biochem. Mol. Biol. 1996, 58, 367–376. [Google Scholar] [CrossRef]
- Pendas-Franco, N.; Gonzalez-Sancho, J.M.; Suarez, Y.; Aguilera, O.; Steinmeyer, A.; Gamillo, C.; Berciano, M.T.; Lafarga, M.; Munoz, A. Vitamin D regulates the phenotype of human breast cancer cells. Differentiation 2007, 75, 193–207. [Google Scholar] [CrossRef]
- Deeb, K.K.; Trump, D.L.; Johnson, C.S. Vitamin D signaling pathways in cancer: Potential for anticancer therapeutics. Nat. Rev. Cancer 2007, 7, 684–700. [Google Scholar] [CrossRef]
- Cho, Y.L.; Christensen, C.; Saunders, D.E.; Lawrence, W.D.; Deppe, G.; Malviya, V.K.; Malone, J.M. Combined effects of 1,25-dihydroxyvitamin D3 and platinum drugs on the growth of MCF-7 cells. Cancer Res. 1991, 51, 2848–2853. [Google Scholar]
- Ravid, A.; Rocker, D.; Machlenkinn, A.; Rotem, C.; Hochman, A.; Kessler-Icekson, G.; Liberman, U.A.; Koren, R. 1,25-Dihydroxyvitamin D3 enhances the susceptibility of breast cancer cells to doxorubicin-induced oxidative damage. Cancer Res. 1999, 59, 862–867. [Google Scholar]
- Koshizuka, K.; Koike, M.; Asou, H.; Cho, S.K.; Stephen, T.; Rude, R.K.; Binderup, L.; Uskokovic, M.; Koeffler, H.P. Combined effect of vitamin D3 analogs and paclitaxel on the growth of MCF-7 breast cancer cells in vivo. Breast Cancer Res. Treat. 1999, 53, 113–120. [Google Scholar] [CrossRef]
- Wigington, D.P.; Urben, C.M.; Strugnell, S.A.; Knutson, J.C. Combination study of 1,24-(S)-Dihydroxyvitamin D2 and chemotherapeutic agents on human breast and prostate cancer cell lines. Anticancer Res. 2004, 24, 2905–2912. [Google Scholar]
- Trump, D.L.; Hershberger, P.A.; Bernardi, R.J.; Ahmed, S.; Muindi, J.; Fakih, M.; Yu, W.D.; Johnson, C.S. Anti-tumor activity of calcitriol: Pre-clinical and clinical studies. J. Steroid Biochem. Mol. Biol. 2004, 89–90, 519–526. [Google Scholar] [CrossRef]
- Trump, D.L.; Deeb, K.K.; Johnson, C.S. Vitamin D: Considerations in the continued development as an agent for cancer prevention and therapy. Cancer J. 2010, 16, 1–9. [Google Scholar] [CrossRef]
- Smith, D.C.; Johnson, C.S.; Freeman, C.C.; Muindi, J.; Wilson, J.W.; Trump, D.L. A Phase I trial of calcitriol (1,25-dihydroxycholecalciferol) in patients with advanced malignancy. Clin. Cancer Res. 1999, 5, 1339–1345. [Google Scholar]
- Fakih, M.G.; Trump, D.L.; Muindi, J.R.; Black, J.D.; Bernardi, R.J.; Creaven, P.J.; Schwartz, J.; Brattain, M.G.; Hutson, A.; French, R.; et al. A phase I pharmacokinetic and pharmacodynamic study of intravenous calcitriol in combination with oral gefitinib in patients with advanced solid tumors. Clin. Cancer Res. 2007, 13, 1216–1223. [Google Scholar] [CrossRef]
- Beer, T.M.; Munar, M.; Henner, W.D. A Phase I trial of pulse calcitriol in patients with refractory malignancies: Pulse dosing permits substantial dose escalation. Cancer 2001, 91, 2431–249. [Google Scholar] [CrossRef]
- Muindi, J.R.; Peng, Y.; Potter, D.M.; Hershberger, P.A.; Tauch, J.S.; Capozzoli, M.J.; Egorin, M.J.; Johnson, C.S.; Trump, D.L. Pharmacokinetics of high-dose oral calcitriol: Results from a phase 1 trial of calcitriol and paclitaxel. Clin. Pharmacol. Ther. 2002, 72, 648–659. [Google Scholar] [CrossRef]
- Beer, T.M.; Javle, M.M.; Ryan, C.W.; Garzotto, M.; Lam, G.N.; Wong, A.; Henner, W.D.; Johnson, C.S.; Trump, D.L. Phase I study of weekly DN-101, a new formulation of calcitriol, in patients with cancer. Cancer Chemother. Pharmacol. 2007, 59, 581–587. [Google Scholar] [CrossRef]
- Beer, T.M.; Javle, M.; Lam, G.N.; Henner, W.D.; Wong, A.; Trump, D.L. Pharmacokinetics and tolerability of a single dose of DN-101, a new formulation of calcitriol, in patients with cancer. Clin. Cancer. Res. 2005, 11, 7794–7799. [Google Scholar] [CrossRef]
- Schleicher, R.L.; Encisco, S.E.; Chaudhary-Webb, M.; Paliakov, E.; McCoy, L.F.; Pfeiffer, C.M. Isotope dilution ultra performance liquid chromatography-tandem mass spectrometry method for simultaneous measurement of 25-hydroxyvitamin D2, 25-hydroxyvitamin D3 and 3-epi-25-hydroxyvitamin D3 in human serum. Clin. Chim. Acta 2011, 412, 1594–1599. [Google Scholar] [CrossRef]
- Yazdanpanah, M.; Bailey, D.; Walsh, W.; Wan, B.; Adeli, K. Analytical measurement of serum 25-OH-vitamin D3, 25-OH-vitamin D2 and their C3-epimers by LC-MS/MS in infant and pediatric specimens. Clin. Biochem. 2013, 46, 1264–1271. [Google Scholar] [CrossRef]
- Buras, R.B.; Schumaker, L.M.; Davoodi, F.; Brenner, R.V.; Shabahang, M.; Nauta, R.J.; Evans, S.R.T. Vitamin D receptors in breast cancer cells. Breast Cancer Res. Treat 1994, 31, 191–202. [Google Scholar] [CrossRef]
- Lopes, N.; Sousa, B.; Martins, D.; Gomes, M.; Vieira, D.; Veronese, L.A.; Milanezi, F.; Paredes, J.; Costa, J.L.; Schmitt, F. Alterations in Vitamin D signalling and metabolic pathways in breast cancer progression: A study of VDR, CYP27B1 and CYP24A1 expression in benign and malignant breast lesions. BMC Cancer 2010, 10, 483–493. [Google Scholar] [CrossRef]
- Peng, X.; Jhaveri, P.; Hussain-Hakimjee, E.A.; Mehta, R.G. Overexpression of ER and VDR is not sufficient to make ER-negative MDA-MB231 breast cancer cells responsive to 1α-hydroxyvitamin D5. Carcinogenesis 2007, 28, 1000–1007. [Google Scholar]
- Schonn, I.; Hennesen, J.; Dartsch, D.C. Cellular responses to etoposide: Cell death despite cell cycle arrest and repair of DNA damage. Apoptosis 2010, 15, 162–172. [Google Scholar] [CrossRef]
- Sheikh, M.S.; Li, X.S.; Chen, J.C.; Shao, Z.M.; Ordonez, J.V.; Fontana, J.A. Mechanisms of regulation of WAF1/Cip1 gene expression in human breast carcinoma: Role of p53-dependent and independent signal transduction pathways. Oncogene 1994, 9, 3407–3415. [Google Scholar]
- Wang, Q.; Yang, W.; Uytingco, M.S.; Christakos, S.; Wieder, R. 1,25 Dihydroxyvitamin D3 and all-trans-retinoic acid sensitize breast cancer cells to chemotherapy-induced cell death. Cancer Res. 2000, 60, 2040–2048. [Google Scholar]
- Narvaez, C.J.; Welsh, J.E. Role of mitochondria and caspases in vitamin D-mediated apoptosis of MCF-7 breast cancer cells. J. Biol. Chem. 2001, 276, 9101–9107. [Google Scholar] [CrossRef]
- Weitsman, G.E.; Ravid, A.; Liberman, U.A.; Koren, R. Vitamin D enhances caspase-dependent and -independent TNF-induced breast cancer cell death: The role of reactive oxygen species and mitochondria. Int. J. Cancer 2003, 106, 178–186. [Google Scholar]
- Norman, A.W.; Zhou, J.Y.; Henry, H.L.; Uskokovic, M.R.; Koeffler, H.P. Structure-Function studies on analogues of la,25-dihydroxyvitamin D3: Differential effects on leukemic cell growth, differentiation, and intestinal calcium absorption. Cancer Res. 1990, 50, 6857–6864. [Google Scholar]
- Burch, W.M.; Lopez-Claros, M.; Uskokovic, M.R.; Drezner, M.K. 1,25-dihydroxyvitamin D3 stimulates avian and mammalian cartilage growth in vitro. J. Bone Mineral. Res. 1988, 3, 87–91. [Google Scholar]
- Maehr, H.; Rochel, N.; Lee, H.J.; Suh, N.; Uskokovic, M.R. Diastereotopic and deuterium effects in Gemini. J. Med. Chem. 2013, 56, 3878–3888. [Google Scholar] [CrossRef]
- Tan, J.; Dwivedi, P.P.; Anderson, P.; Nutchey, B.K.; O’Loughlin, P.; Morris, H.A.; May, B.K.; Ferrante, A.; Hii, C.S. Antineoplastic agents target the 25-hydroxyvitamin D3 24-hydroxylase messenger RNA for degradation: Implications in anticancer activity. Mol. Cancer Ther. 2007, 6, 3131–3138. [Google Scholar] [CrossRef]
- Ben-Batalla, I.; Seoane, S.; Garcia-Caballero, T.; Gallego, R.; Macia, M.; Gonzalez, L.O.; Vizoso, F.; Perez-Fernandez, R. Deregulation of the Pit-1 transcription factor in human breast cancer cells promotes tumor growth and metastasis. J. Clin. Invest. 2010, 120, 4289–4302. [Google Scholar] [CrossRef]
- ProgRes C3 camera and the ProgRess Capture Pro, version 2.7; Jenoptik: Jena, Germany, 2009.
- MATLAB, version 7.1.; MathWorks, Inc: Natick, MA, USA, 2011.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Seoane, S.; Bermudez, M.A.; Sendon-Lago, J.; Martinez-Ordoñez, A.; Abdul-Hadi, S.; Maestro, M.; Mouriño, A.; Perez-Fernandez, R. 26,26,26,27,27,27-Hexadeuterated-1,25-Dihydroxyvitamin D3 (1,25D-d6) As Adjuvant of Chemotherapy in Breast Cancer Cell Lines. Cancers 2014, 6, 67-78. https://doi.org/10.3390/cancers6010067
Seoane S, Bermudez MA, Sendon-Lago J, Martinez-Ordoñez A, Abdul-Hadi S, Maestro M, Mouriño A, Perez-Fernandez R. 26,26,26,27,27,27-Hexadeuterated-1,25-Dihydroxyvitamin D3 (1,25D-d6) As Adjuvant of Chemotherapy in Breast Cancer Cell Lines. Cancers. 2014; 6(1):67-78. https://doi.org/10.3390/cancers6010067
Chicago/Turabian StyleSeoane, Samuel, Maria A. Bermudez, Juan Sendon-Lago, Anxo Martinez-Ordoñez, Soraya Abdul-Hadi, Miguel Maestro, Antonio Mouriño, and Roman Perez-Fernandez. 2014. "26,26,26,27,27,27-Hexadeuterated-1,25-Dihydroxyvitamin D3 (1,25D-d6) As Adjuvant of Chemotherapy in Breast Cancer Cell Lines" Cancers 6, no. 1: 67-78. https://doi.org/10.3390/cancers6010067



