The Inverse Relationship between 25-Hydroxyvitamin D and Cancer Survival: Discussion of Causation
Abstract
:1. Introduction
2. Review of Survival Studies
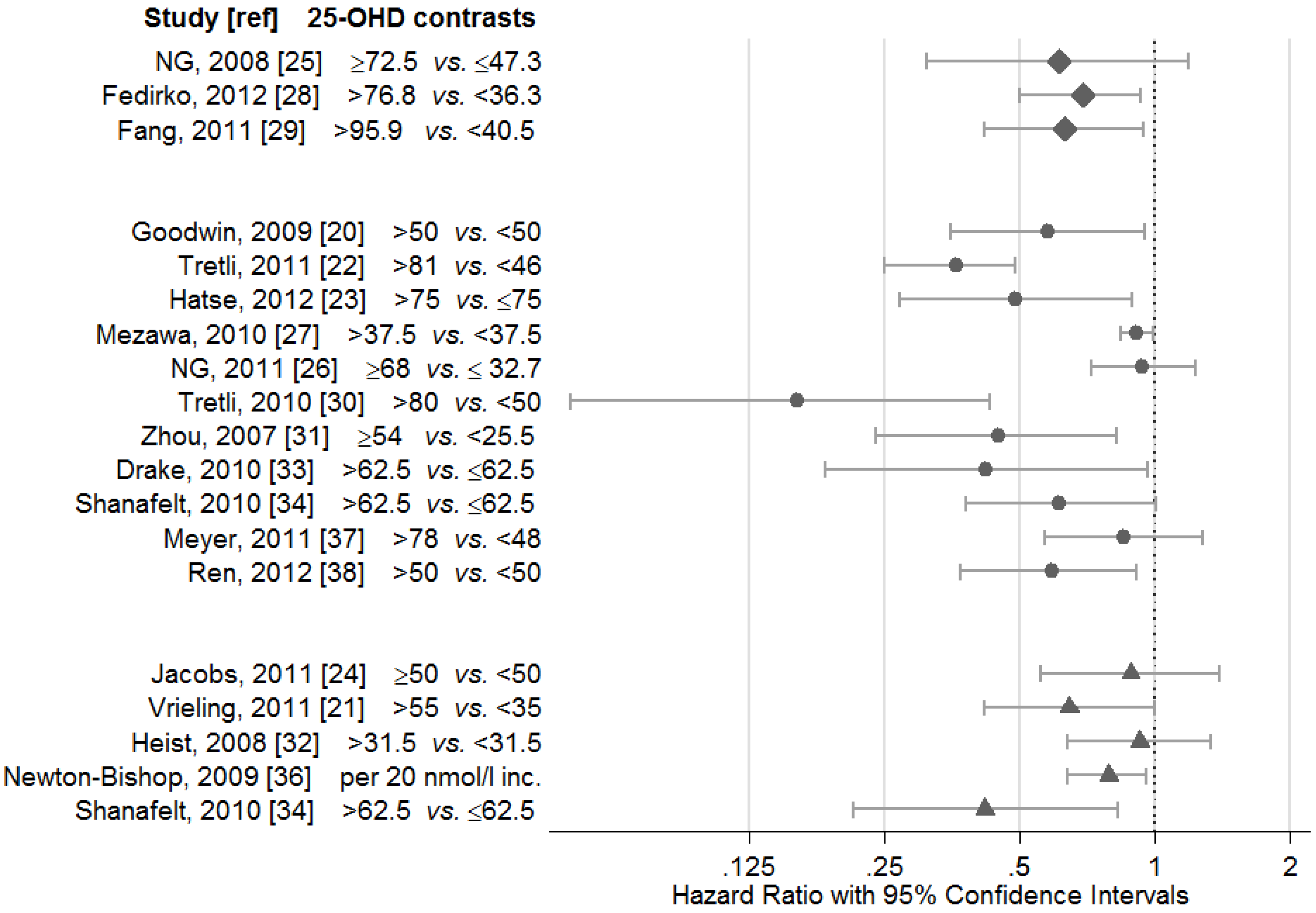
2.1. Breast Cancer
2.2. Colorectal Cancer
2.3. Prostate Cancer
2.4. Lung Cancer
2.5. Hematologic Cancers
2.6. Other Cancers
| Study [ref] | Cancer 1 | Size (n) | Follow-up, years 4 | Adjustments 5 | Relation 25-OHD/survival | Main findings |
|---|---|---|---|---|---|---|
| Studies based on serum collected prior to diagnosis | ||||||
| NG, 2008 [25] | CRC | 304 | 0–14 c | 1,2,5,6,11,12,15–17 | (+) | Non-significant reduced risk of cancer death for levels ≥72.5 nmol/L (HR 0.61,95% CI 0.31–1.19) compare with levels ≤47.3 nmol/L |
| Fedirko, 2012 [28] | CRC | 1202 | 73 a months | 1,2,5,6,9,11,12, 15–17,20 | + | Reduced risk of cancer death in patients with levels >76.8 nmol/L (HR 0.69, 95% CI 0.50–0.93) compared with levels <36.3 nmol/L |
| Fang, 2011 [29] | PC | 185 | 10 a | 1,2,6,9,11 | (+) | Increased risk of cancer death if 25-OHD <40.5 nmol/L (HR 1.59, 95% CI 1.06–2.39) compared with levels >95.9 nmol/L (not significant when adjust for Gleason score/stage) |
| Studies based on serum collected at or recently after diagnosis and prior to treatment | ||||||
| Goodwin, 2009 [20] | BC 2 | 512 | 11.6 a | 1–5 | + | Increased risk of cancer death (HR 1.73, 95% CI 1.05–2.86) in patients with 25-OHD levels <50 compared with levels >50 nmol/L |
| Tretli, 2011 [22] | 1,2,12,15 | + | Reduced risk of cancer death if high 25-OHD levels | |||
| BC | 251 | 9.3 a | BC: HR 0.42 (95% CI 0.21-0.82), ≥86 versus <50 nmol/l | |||
| CC | 52 | 7.3 a | CC: HR 0.20 (95% CI 0.04-1.10), ≥77 l versus <44 nmol/L | |||
| LC | 210 | 1.6 a | LC: HR 0.18 (95% CI 0.11-0.29), ≥76 versus <41 nmol/L | |||
| NHL/HL | 145 | 6.3 a | NHL/HL: HR 0.39 (95% CI 0.18-0.83), ≥77 versus <44 nmol/L | |||
| Hatse, 2012 [23] | BC 2 | 1800 | 0–9c | 1–3,5,6,12,15 | + | Reduced risk of cancer death when 25-OHD levels >75 nmol/L (HR 0.49, 95% CI 0.27-0.89) compared with levels ≤75 nmol/L (significant for postmenopausal BC only) |
| Mezawa, 2010 [27] | CRC | 257 | 0–5 c | 1–3,12,15,17 | + | Reduced risk of death if 25-OHD levels >37.5 nmol/L (HR 0.91, 95% CI 0.84-0.99) compared with levels <37.5 nmol/L |
| NG, 2011 [26] | CRC 3 | 515 | 1,2,6,12,15,18,19 | 0 | No association between 25-OHD levels and survival. RR was 0.94 (95% CI 0.72–1.23) for 25-OHD levels ≥68 compared with levels ≤32.7 nmol/L (82% were <75 nmol/L) | |
| Tretli, 2010 [30] | PC | 160 | 3.7 b | 1,5,12,18 | + | Reduced risk of cancer death in patients with 25-OHD >80 nmol/L (HR 0.16, 95% CI 0.05–0.43) or 50–80 nmol/L (HR 0.33, 95% CI 0.14–0.77) compared with levels <50 nmol/L |
| Zhou, 2007 [31] | NSLC 2 | 447 | 72 b months | 1,2,9,12,15,19 | (+) | All: No association between 25-OHD and survival. IB-IIB stage patients: reduced risk of death if 25-OHD levels ≥54 nmol/L (HR 0.45, 95% CI 0.24–0.82) compared with levels <25.5 nmol/L |
| Drake, 2010 [33] | NHL | 983 | 34.8 b months | 1–3,15,18,20 | + | Increased risk of death for B-cell (HR 1.99, 95% CI 1.27–3.13) and T-cell (HR 2.38, 95% CI 1.04–5.41) lymphoma types, if 25-OHD levels ≤62.5 nmol/L (Serum drawn prior to (n = 649), during or after (n = 334) treatment) |
| Shanafelt, 2010 [34] | CLL | 153 | 9.9 b | 1–3,15 | (+) | The confirmation cohort: non-significant increased risk of cancer death if 25-OHD levels ≤62.5 nmol/L (HR 1.63, 95% CI 0.99–2.69) |
| Meyer, 2011 [37] | HNC | 522 | 4.4 b | 1,2,6,9,10,12,16,19,21 | 0 | No association between 25-OHD level and risk of death. HR for highest (>78 nmol/L) versus lowest quartile (<48 nmol/L) was 0.85 (95% CI 0.57–1.28) |
| Ren, 2012 [38] | GC | 197 | 0–8.3 c | 1,2,5,6,9,12,15,16,19,21 | + | Reduced risk of cancer death in patients with 25-OHD levels >50 nmol/L (HR 0.59, 95% CI 0.37–0.91) compared with levels <50 nmol/L |
| Studies based on serum collected after diagnosis and after start of treatment | ||||||
| Jacobs, 2011 [24] | BC 2 | 512 | 7.3 a | 1,2,4–8,19,22 | 0 | No association with risk of cancer death (OR 1.13, 95% CI 0.72–1.79), comparing 25-OHD levels <50 and ≥50 nmol/L |
| Vrieling, 2011 [21] | BC | 1295 | 5.8 b | 1–5,12–14 | + | Increased risk of cancer death if 25-OHD levels <35 nmol/L (HR 1.55, 95% CI 1.00–2.39) compared with levels >55 (postmenopausal BC only) |
| Heist, 2008 [32] | NSLC 3 | 294 | 42 b months | 2,15,18 | 0 | No associations between 25-OHD and survival, comparing 25-OHD levels <31.5 nmol/L with higher levels (HR 1.08, 95% CI 0.75–1.57) |
| Newton-Bishop, 2009 [36] | CMM | 872 | 4.7 b | 1,2,6,12,15,16 | + | Improved relapse-free survival per 20 nmol/L increase in serum 25-OHD (HR 0.79, 95% CI 0.64–0.96) |
| Shanafelt, 2010 [34] | CLL | 390 | 3 b | 1–3,15 | + | The discovery cohort: Increased risk of cancer death if 25-OHD levels ≤62.5 nmol/L (HR 2.39, 95% CI 1.21–4.70) |
| Pardanani, 2011 [35] | MPN | 409 | 0–300 c months | 1,2,15,16,18 | 0 | No association between 25-OHD levels an survival, comparing patient with 25-OHD levels ≥62.5 and <62.5 nmol/L. Estimates were not possible to abstract (information on pre or post treatment sampling is not given) |
3. Discussion
3.1. Temporality
3.2. Strength and Exposure-Response Relationship
3.3. Biological Plausibility
3.4. Consistency
3.5. Reversibility
3.6. Coherence
4. Conclusions
Conflicts of Interest
References
- Apperly, F.L. The relation of solar radiation to cancer mortality in North America. Cancer Res. 1941, 1, 191–195. [Google Scholar]
- Hollick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Garland, C.F.; Garland, F.C. Do sunlight and vitamin D reduce the likelihood of colon cancer? Int. J. Epidemiol. 1980, 9, 227–231. [Google Scholar] [CrossRef]
- Schwartz, G.G.; Hulka, B.S. Is vitamin D deficiency a risk factor for prostate cancer? (Hypothesis). Anticancer Res. 1990, 10, 1307–1311. [Google Scholar]
- Grant, W.B. How strong is the evidence that solar ultraviolet B and vitamin D reduce the risk of cancer? An examination using Hill’s criteria for causality. Dermato-endocrinology 2009, 1, 17–24. [Google Scholar] [CrossRef]
- Chen, W.; Armstrong, B.K.; Rahman, B.; Zheng, R.; Zhang, S.; Clements, M. Relationship between cancer survival and ambient ultraviolet B irradiance in China. Cancer Causes Control 2013, 24, 1323–1330. [Google Scholar] [CrossRef]
- Robsahm, T.E.; Tretli, S.; Dahlback, A.; Moan, J. Vitamin D3 from sunlight may improve the prognosis of breast-, colon- and prostate cancer (Norway). Cancer Causes Control 2004, 15, 149–158. [Google Scholar] [CrossRef]
- Porojnicu, A.C.; Robsahm, T.E.; Ree, A.H.; Moan, J. Season of diagnosis is a prognostic factor in Hodgkin’s lymphoma: A possible role of sun-induced vitamin D. Br. J. Cancer 2005, 93, 571–574. [Google Scholar] [CrossRef]
- Lim, H.S.; Roychoudhuri, R.; Peto, J.; Schwartz, G.; Baade, P.; Møller, H. Cancer survival is dependent on season of diagnosis and sunlight exposure. Int. J. Cancer 2006, 119, 1530–1536. [Google Scholar] [CrossRef]
- Porojnicu, A.C.; Robsahm, T.E.; Dahlback, A.; Berg, J.P.; Christiani, D.; Bruland, O.S.; Moan, J. Seasonal and geographical variations in lung cancer prognosis in Norway. Does vitamin D from the sun play a role? Lung Cancer 2007, 55, 263–270. [Google Scholar] [CrossRef]
- Giovannucci, E. The epidemiology of vitamin D and cancer risk. In Vitamin D, 3rd ed.; Feldman, D., Wesley Pike, J., Adams, J.S., Eds.; Academic Press: Amsterdam, The Netherlands, 2011; pp. 1570–1591. [Google Scholar]
- Gorham, E.D.; Garland, C.F.; Garland, F.C.; Grant, W.B.; Mohr, S.B.; Lipkin, M.; Newmark, H.L.; Giovannucci, E.; Wei, M.; Holick, M.F. Optimal vitamin D status for colorectal cancer prevention: A quantitative meta analysis. Am. J. Prev. Med. 2007, 32, 210–216. [Google Scholar] [CrossRef]
- Gandini, S.; Boniol, M.; Haukka, J.; Byrnes, G.; Cox, B.; Sneyd, M.J.; Mullie, P.; Autier, P. Meta-analysis of observational studies of serum 25-hydroxyvitamin D levels and colorectal, breast and prostate cancer and colorectal adenoma. Int. J. Cancer 2011, 128, 1414–1424. [Google Scholar] [CrossRef]
- Jenab, M.; Bueno-de-Mesquita, H.B.; Ferrari, P.; van Duijnhoven, F.J.; Norat, T.; Pischon, T.; Jansen, E.H.; Slimani, N.; Byrnes, G.; Rinaldi, S.; et al. Association between pre-diagnostic circulating vitamin D concentration and risk of colorectal cancer in European populations: a nested case-control study. Br. Med. J. 2010, 340. [Google Scholar] [CrossRef]
- Wolcott, C.G.; Wilkens, L.R.; Nomura, A.M.; Horst, L.R.; Goodman, M.T.; Murphy, S.P.; Henderson, B.E.; Kolonel, L.N.; le Marchand, L. Plasma 25-hydroxyvitamin D levels and the risk of colorectal cancer: The multiethnic cohort study. Cancer Epidemiol. Biomarkers Prev. 2010, 19, 130–134. [Google Scholar] [CrossRef]
- Helzlsouer, K.J. Overview of the cohort consortium vitamin D pooling project of rarer cancers. Am. J. Epidemiol. 2010, 172, 4–9. [Google Scholar] [CrossRef]
- Guerrieri-Gonzaga, A.; Gandini, S. Vitamin D and overall mortality. Pigment Cell Melanoma Res. 2013, 26, 16–28. [Google Scholar] [CrossRef]
- Boscoe, F.P.; Schymura, M.J. Solar ultraviolet-B exposure and cancer incidence and mortality in the United States, 1993–2002. BMC Cancer 2006, 10. [Google Scholar] [CrossRef]
- Chen, W.; Clements, M.; Rahman, B.; Zhang, S.; Qiao, Y.; Armstrong, B.K. Relationship between cancer mortality/incidence and ambient ultraviolet B irradiance in China. Cancer Causes Control 2010, 21, 1701–1709. [Google Scholar] [CrossRef]
- Goodwin, P.J.; Ennis, M.; Pritchard, K.I.; Koo, J.; Hood, N. Prognostic effects of 25-hydroxyvitamin D levels in early breast cancer. J. Clin. Oncol. 2009, 27, 3757–3763. [Google Scholar] [CrossRef]
- Vrieling, A.; Hein, R.; Abbas, S.; Schneeweiss, A.; Flesch-Janys, D.; Chang-Claude, J. Serum 25-hydroxyvitamin D and postmenopausal breast cancer survival: A prospective patient cohort study. Breast Cancer Res. 2011, 13. [Google Scholar] [CrossRef]
- Tretli, S.; Schwartz, G.G.; Torjesen, P.A.; Robsahm, T.E. Serum levels of 25-hydroxyvitamin D and survival in Norwegian patients with cancer of breast, colon, lung, and lymphoma: A population-based study. Cancer Causes Control 2012, 23, 363–370. [Google Scholar] [CrossRef]
- Hatse, S.; Lambrechts, D.; Verstuyf, A.; Smeets, A.; Brouwers, B.; Vandorpe, T.; Brouckaert, O.; Peuteman, G.; Laenen, A.; Verlinden, L.; et al. Vitamin D status at breast cancer diagnosis: Correlation with tumor characteristics, disease outcome, and genetic determinants of vitamin D insufficiency. Carcinogenesis 2012, 33, 1319–1326. [Google Scholar] [CrossRef]
- Jacobs, E.T.; Thomson, C.A.; Flatt, S.W.; Al-Delaimy, W.K.; Hibler, E.A.; Jones, L.A.; Leroy, E.C.; Newman, V.A.; Parker, B.A.; Rock, C.L.; et al. Vitamin D and breast cancer recurrence in the Women’s Healthy Eating and Living (WHEL) Study. Am. J. Clin. Nutr. 2011, 93, 108–117. [Google Scholar] [CrossRef]
- Ng, K.; Meyerhardt, J.A.; Wu, K.; Feskanich, D.; Hollis, B.W.; Giovannucci, E.L.; Fuchs, C.S. Circulating 25-hydroxyvitamin D levels and survival in patients with colorectal cancer. J. Clin. Oncol. 2008, 26, 2984–2991. [Google Scholar] [CrossRef]
- Ng, K.; Sargent, D.J.; Goldberg, R.M.; Meyerhardt, J.A.; Green, E.M.; Pitot, H.C.; Hollis, B.W.; Pollak, M.N.; Fuchs, C.S. Vitamin D status in patients with stage IV colorectal cancer: Findings from intergroup trial N9741. J. Clin. Oncol. 2011, 29, 1599–1606. [Google Scholar] [CrossRef]
- Mezawa, H.; Sugiura, T.; Watanabe, M.; Norizoe, C.; Takahashi, D.; Shimojima, A.; Tamez, S.; Tsutsumi, Y.; Yanaga, K.; Urashima, M. Serum vitamin D levels and survival of patients with colorectal cancer: Post-hoc analysis of a prospective cohort study. BMC Cancer 2010, 10. [Google Scholar] [CrossRef]
- Fedirko, V.; Riboli, E.; Tjønneland, A.; Ferrari, P.; Olsen, A.; Bueno-de-Mesquita, H.B.; van Duijnhoven, F.J.; Norat, T.; Jansen, E.H.J.M.; Dahm, C.C.; et al. Prediagnostic 25-hydroxyvitamin D, VDR and CASR polymorphisms, and survival in patients with colorectal cancerin western European populations. Cancer Epidemiol. Biomarkers Prev. 2012, 21, 582–593. [Google Scholar] [CrossRef]
- Fang, F.; Kasperzyk, J.L.; Shui, I.; Hendrickson, W.; Hollis, B.W.; Fall, K.; Ma, J.; Gaziano, J.M.; Stampfer, M.J.; Mucci, L.A.; Giovannucci, E. Prediagnostic plasma vitamin D metabolites and mortality among patients with prostate cancer. PLoS One 2011, 6, e18625. [Google Scholar] [CrossRef]
- Tretli, S.; Hernes, E.; Berg, J.P.; Hestvik, U.E.; Robsahm, T.E. Association between serum 25(OH)D and death from prostate cancer. Br. J. Cancer 2009, 100, 450–454. [Google Scholar] [CrossRef]
- Zhou, W.; Heist, R.S.; Liu, G.; Asomaning, K.; Neuberg, D.S.; Hollis, B.W.; Wain, J.C.; Lynch, T.J.; Giovannucci, E.; Su, L.; et al. Circulating 25-hydroxyvitamin D levels predict survival in early-stage non-small-cell lung cancerpatients. J. Clin. Oncol. 2007, 25, 479–485. [Google Scholar] [CrossRef]
- Heist, R.S.; Zhou, W.; Wang, Z.; Liu, G.; Neuberg, D.; Su, L.; Asomaning, K.; Hollis, B.W.; Lynch, T.J.; Wain, J.C.; et al. Circulating 25-hydroxyvitamin D, VDR polymorphisms, and survival in advanced non-small-cell lung cancer. J. Clin. Oncol. 2008, 26, 5596–5602. [Google Scholar] [CrossRef]
- Drake, M.T.; Maurer, M.J.; Link, B.K.; Habermann, T.M.; Ansell, S.M.; Micallef, I.N.; Kelly, J.L.; Macon, W.R.; Nowakowski, G.S.; Inwards, D.J.; et al. Vitamin D insufficiency and prognosis in non-Hodgkin’s lymphoma. J. Clin. Oncol. 2010, 28, 4191–4198. [Google Scholar] [CrossRef]
- Shanafelt, T.D.; Drake, M.T.; Maurer, M.J.; Allmer, C.; Rabe, K.G.; Slager, S.L.; Weiner, G.J.; Call, T.G.; Link, B.K.; Zent, C.S.; et al. Vitamin D insufficiency and prognosis in chronic lymphocytic leukemia. Blood 2011, 117, 1492–1498. [Google Scholar] [CrossRef]
- Pardanani, A.; Drake, M.T.; Finke, C.; Lasho, T.L.; Rozell, S.A.; Jimma, T.; Tefferi, A. Vitamin D insufficiency in myeloproliferative neoplasms and myelodysplastic syndromes: Clinical correlates and prognostic studies. Am. J. Hematol. 2011, 86, 1013–1016. [Google Scholar] [CrossRef]
- Newton-Bishop, J.A.; Beswick, S.; Randerson-Moor, J.; Chang, Y.M.; Affleck, P.; Elliott, F.; Chan, M.; Leake, S.; Karpavicius, B.; Haynes, S.; et al. Serum 25-hydroxyvitamin D3 levels are associated with breslow thickness at presentation and survival from melanoma. J. Clin. Oncol. 2009, 27, 5439–5444. [Google Scholar] [CrossRef]
- Meyer, F.; Liu, G.; Douville, P.; Samson, E.; Xu, W.; Adjei, A.; Bairati, I. Dietary vitamin D intake and serum 25-hydroxyvitamin D level in relation to disease outcomes in head and neck cancer patients. Int. J. Cancer 2011, 128, 1741–1746. [Google Scholar]
- Ren, C.; Qiu, M.Z.; Wang, D.S.; Luo, H.Y.; Zhang, D.S.; Wang, Z.Q.; Wang, F.H.; Li, Y.H.; Zhou, Z.W.; Xu, R.H. Prognostic effects of 25-hydroxyvitamin D levels in gastric cancer. J. Transl. Med. 2012, 10. [Google Scholar] [CrossRef]
- Hill, A.B. The environment and disease: Association or causation. Proc. R. Soc. Med. 1965, 58, 295–300. [Google Scholar]
- Zemel, M.B. Calcium modulation of hypertension and obesity: Mechanisms and implications. J. Am. Coll. Nutr. 2001, 20, 428S–435S. [Google Scholar] [CrossRef]
- Thaw, S.S.; Sahmoun, A.E.; Schwartz, G.G. Serum calcium, tumor size, and hormone receptor status in women with untreated breast cancer. Cancer Biol. Therapy 2012, 7, 467–471. [Google Scholar]
- Schwartz, G.G.; Skinner, H.G. Prospective studies of total and ionized serum calcium in relation to incident and fatal ovarian cancer. Gyn. Oncol. 2013, 129, 169–172. [Google Scholar] [CrossRef]
- Schwartz, G.G. Prostate cancer, serum Parathyroid Hormone and the progression of skeletal metastases. Cancer Epidemiol. Biomarkers Prev. 2008, 17, 478–483. [Google Scholar] [CrossRef]
- Fakih, M.G.; Trump, D.L.; Johnson, C.S.; Tian, L.; Muindi, J.; Sunga, A.Y. Chemotherapy is linked to severe vitamin D deficiency in patients with colorectal cancer. Int. J. Colorectal. Dis. 2009, 24, 219–224. [Google Scholar] [CrossRef]
- Van Leeuwen, J.P.T.M.; van Driel, M.; Feldman, D.; Muñoz, A. Vitamin D: Cancer and Differentiation. In Vitamin D, 3rd ed.; Feldman, D., Wesley Pike, J., Adams, J.S., Eds.; Academic Press: Amsterdam, The Netherlands, 2011; pp. 1592–1597. [Google Scholar]
- Welsh, J. Vitamin D actions in mammary gland and breast cancer. In Vitamin D, 3rd ed.; Feldman, D., Wesley Pike, J., Adams, J.S., Eds.; Academic Press: Amsterdam, The Netherlands, 2011; pp. 1657–1673. [Google Scholar]
- Krishnan, A.V.; Feldman, D. Vitamin D and prostate cancer. In Vitamin D, 3rd ed.; Feldman, D., Wesley Pike, J., Adams, J.S., Eds.; Academic Press: Amsterdam, The Netherlands, 2011; pp. 1675–1709. [Google Scholar]
- Cross, H.S. The vitamin D system and colorectal cancer prevention. In Vitamin D, 3rd ed.; Feldman, D., Wesley Pike, J., Adams, J.S., Eds.; Academic Press: Amsterdam, The Netherlands, 2011; pp. 1711–1729. [Google Scholar]
- Okamoto, R.; Koeffler, H.P. Hematological malignancy. In Vitamin D, 3rd ed.; Feldman, D., Wesley Pike, J., Adams, J.S., Eds.; Academic Press: Amsterdam, The Netherlands, 2011; pp. 1731–1750. [Google Scholar]
- Trump, D.L.; Johnson, C.S. The anti-tumor effects of vitamin D in other cancers. In Vitamin D, 3rd ed.; Feldman, D., Wesley Pike, J., Adams, J.S., Eds.; Academic Press: Amsterdam, The Netherlands, 2011; pp. 1763–1773. [Google Scholar]
- Jacobs, E.T.; Thomson, C.A.; Flatt, S.W.; Newman, V.A.; Rock, C.L.; Pierce, J.P. Correlates of 25-hydroxyvitamin D and breast cancer stage in the Women’s Healthy Eating and Living Study. Nutr. Cancer 2013, 65, 188–194. [Google Scholar] [CrossRef]
- Schwartz, G.G.; Whitlatch, L.W.; Chen, T.C.; Lokeshwar, B.L.; Holick, M.F. Human prostate cells synthesize 1,25-dihydroxyvitamin D3 from 25-hydroxyvitamin D3. Cancer Epidemiol. Biomarkers Prev. 1998, 7, 391–395. [Google Scholar]
- Bikle, D. Nonclassic actions of vitamin D. J. Clin. Endocrinol. Metab. 2009, 94, 26–34. [Google Scholar] [CrossRef]
- Trump, D.L.; Hershberger, P.A.; Bernardi, R.J.; Ahmed, S.; Muindi, J.; Fakih, M.; Yu, W.D.; Johnson, C.S. Antitumor activity of calcitriol: Pre-clinical and clinical studies. J. Steroid Biochem. Mol. Biol. 2004, 89, 519–526. [Google Scholar]
- Townsend, K.; Evans, K.N.; Campbell, M.H.; Colston, K.W.; Adams, J.S.; Hewison, M. Biological actions of extra-renal 25-hydroxyvitamin D-1-alpa-hydroxylase and implications for chemoprevention and treatment. J. Steroid Biochem. Mol. Biol. 2005, 97, 103–109. [Google Scholar] [CrossRef]
- Barreto, A.; Schwartz, G.G.; Woodruff, R.; Cramer, S.D. 25-Hydroxyvitamin D3, the prohormonal form of 1,25-Dihydroxyvitamin D3, inhibits the proliferation of primary prostatic epithelial cells. Cancer Epidemiol. Biomarkers Prev. 2000, 9, 265–270. [Google Scholar]
- Chen, T.C.; Schwartz, G.G.; Burnstein, K.L.; Lokeshwar, B.L.; Holick, M.F. The in vitro evaluation of 25-Hydroxyvitamin D3 and 19-nor-1,25-Dihydroxyvitamin D2 as therapeutic agents for prostate cancer. Clin. Cancer Res. 2000, 6, 901–908. [Google Scholar]
- Swami, S.; Krishnan, A.V.; Wang, J.Y.; Jensen, K.; Horst, R.; Albertelli, M.A.; Feldman, D. Dietary vitamin D3 and 1,25-dihydroxyvitamin D3 (calcitril) exhibit equivalent anticancer activity in mouse xenograft models of breast and prostate cancer. Endocrinolology 2012, 153, 2576–2587. [Google Scholar] [CrossRef]
- Durup, D.; Jørgensen, H.L.; Christensen, J.; Schwartz, P.; Heegard, A.M.; Lind, B. A reverse J-shaped association of all-cause mortality with serum 25-hydroxyvitamin D in general practice: The CopD study. J. Clin. Endocriol. Metab. 2012, 97, 2644–2652. [Google Scholar] [CrossRef]
- Wagner, D.; Trudel, D.; van der Kwast, T.; Nonn, L.; Giangreco, A.A.; Li, D.; Cardoze, M.; Laszlo, S.; Hersey, K.; Klotz, L.; et al. Randomized clinical trial of vitamin D3 doses on prostatic vitamin D metabolite levels and Ki67 labeling in prostate cancer patients. J. Clin. Endocrinol. Metab. 2013, 98, 1498–1507. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Robsahm, T.E.; Schwartz, G.G.; Tretli, S. The Inverse Relationship between 25-Hydroxyvitamin D and Cancer Survival: Discussion of Causation. Cancers 2013, 5, 1439-1455. https://doi.org/10.3390/cancers5041439
Robsahm TE, Schwartz GG, Tretli S. The Inverse Relationship between 25-Hydroxyvitamin D and Cancer Survival: Discussion of Causation. Cancers. 2013; 5(4):1439-1455. https://doi.org/10.3390/cancers5041439
Chicago/Turabian StyleRobsahm, Trude E., Gary G. Schwartz, and Steinar Tretli. 2013. "The Inverse Relationship between 25-Hydroxyvitamin D and Cancer Survival: Discussion of Causation" Cancers 5, no. 4: 1439-1455. https://doi.org/10.3390/cancers5041439




