Role of Topoisomerases in Pediatric High Grade Osteosarcomas: TOP2A Gene Is One of the Unique Molecular Biomarkers of Chemoresponse
Abstract
:1. Introduction
2. Results
| Number of patients | 105 patients |
| Age | 12.9 years (median: 13 years, 4 to 18 years) |
| Tumor sites | 81 tumors in the lower limbs (58 femurs); 14 tumors in the upper limbs (13 humerus); 10 tumors in other bone locations |
| Chemoresponsiveness | 56 GR (53%)/49 PR (47%) |
| Metastases at diagnosis | 18 patients (17%) |
| Histological subtypes | 56 osteoblastic osteosarcomas; 10 fibroblastic osteosarcomas; 11 chondroblastic osteosarcomas; 28 patients with unknown data |
| Overall Survival | 112 mo (median: 127 mo, 7 to 194 mo) |
| Relapse Free Survival | 96 mo (median: 105 mo, 6 to 194 mo) |
| Patients’ relapses | 41 patients |
| Deceased patients | 29 patients |
2.2. A Significant Correlation Was Observed between TOP2A Rearrangements and Good Response to Neo-Adjuvant Chemotherapy
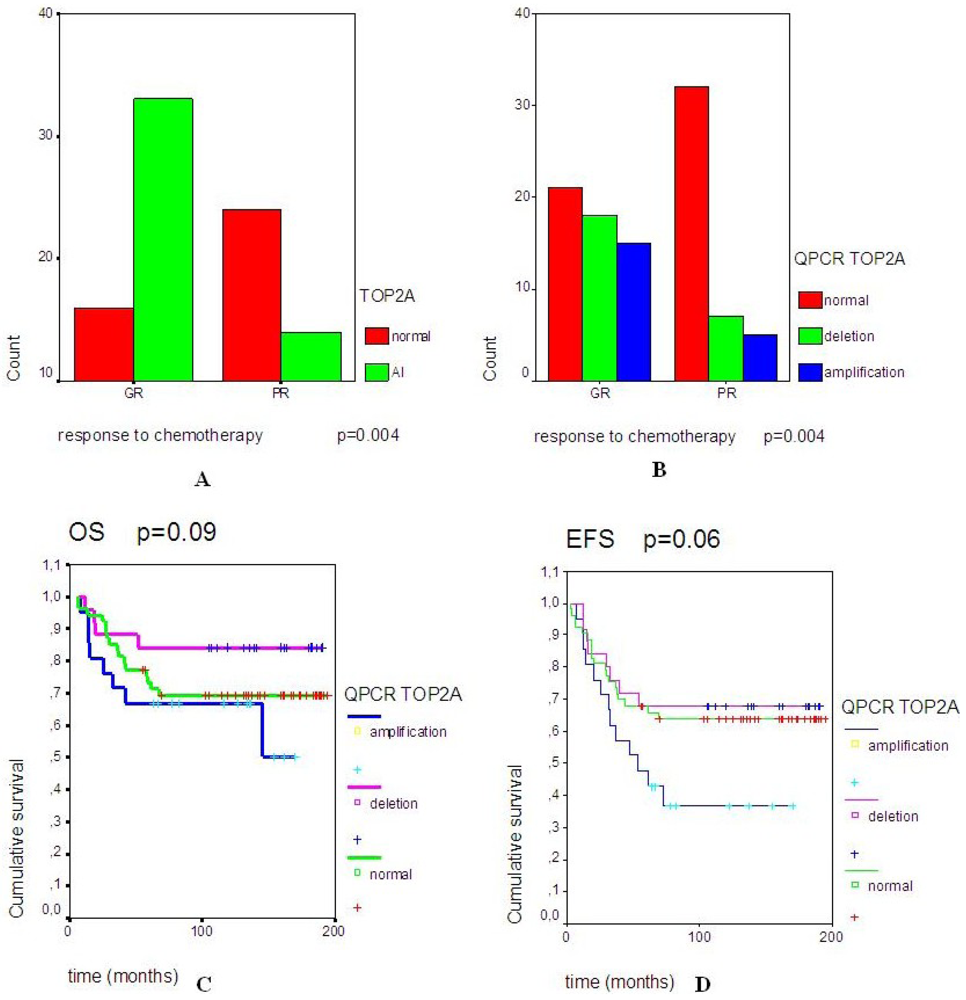
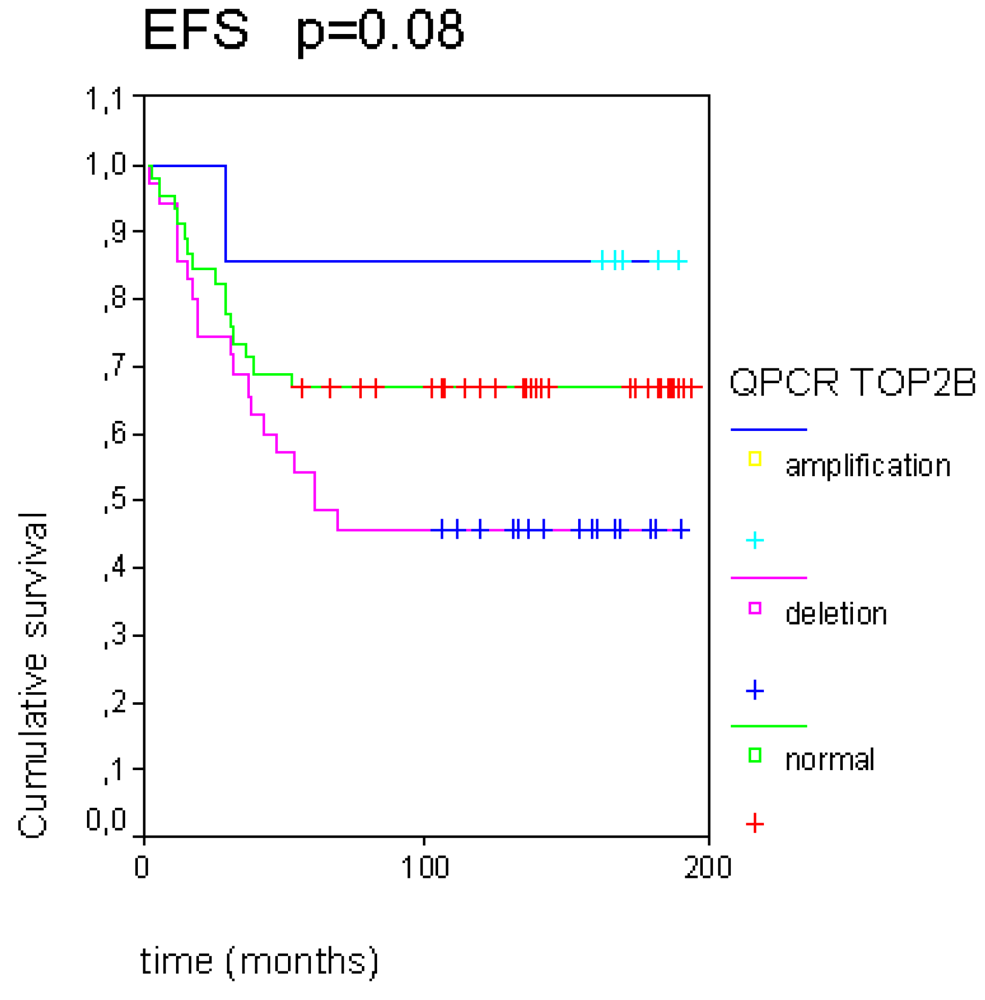
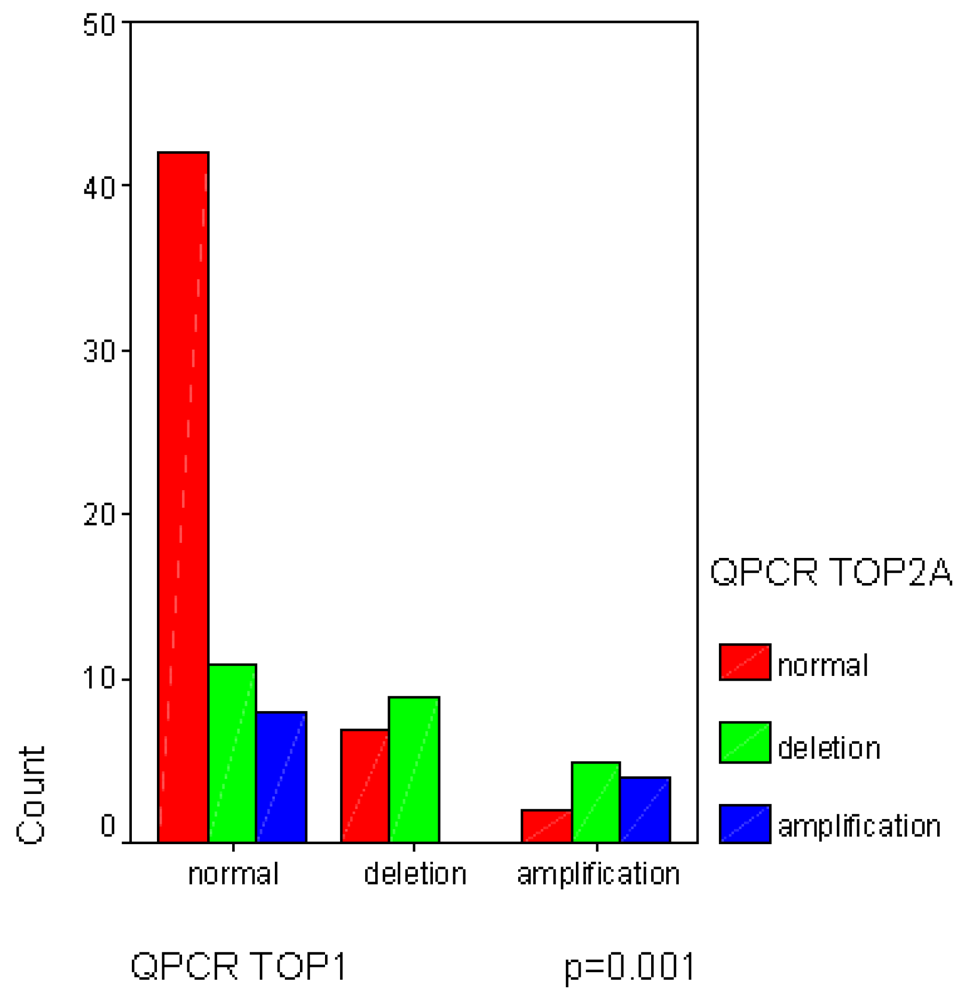
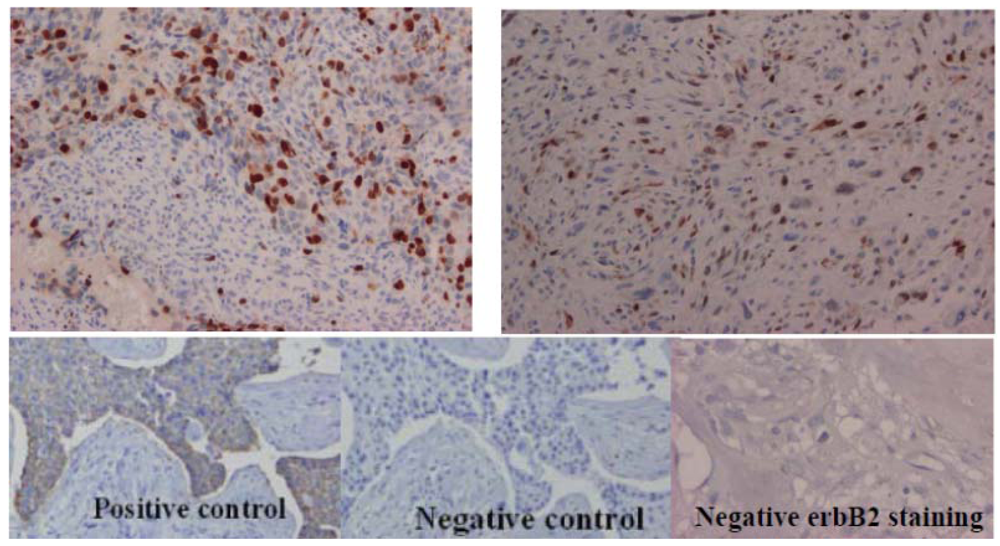
2.3. Topoisomerase IIα Protein Expression and Gene Status
| Patients | Topoisomerase IIα iummunohistochemistry | Allelotyping | QPCR | |||
|---|---|---|---|---|---|---|
| Positive cells (%) | Staining intensity | Localization | Assessment | |||
| Patient 1 | 10% | strong | nucleus | diffuse | homozygous | normal |
| Patient 25 * | 50% (focal) | strong | nucleus | heterogeneous | normal | normal |
| Patient 26 | 10% (focal) | moderate | nucleus | heterogeneous | normal | normal |
| Patient 28 | 40–50% | strong | nucleus | diffuse | allelic imbalance | deletion |
| Patient 29 | 40% (focal) | strong | nucleus | heterogeneous | normal | normal |
| Patient 30 | 40% (focal) | strong | nucleus | heterogeneous | normal | normal |
| Patient 31 | 20% | strong | nucleus | diffuse | allelic imbalance | amplification |
| Patient 32 | 40% (focal) | strong | nucleus | heterogeneous | normal | normal |
| Patient 38 | 20% | strong | nucleus | diffuse | allelic imbalance | amplification |
| Patient 44 | 50% (focal) | moderate | nucleus | heterogeneous | normal | normal |
| Patient 47 | 30% (focal) | strong | nucleus | heterogeneous | normal | normal |
| Patient 55 | 50% (focal) | strong | nucleus | heterogeneous | homozygous | normal |
| Patient 64 | 50% | moderate | nucleus | diffuse | allelic imbalance | amplification |
| Patient 65 | 70% | strong | nucleus | diffuse | normal | normal |
| Patient 66* | 30% | strong | nucleus | diffuse | allelic imbalance | amplification |
| Patient 67 | 60% | strong | nucleus | diffuse | allelic imbalance | amplification |
| Patient 68 | 50% | strong | nucleus | heterogeneous | allelic imbalance | deletion |
3. Discussion
4. Experimental Section
4.1. Tumor Banking and Patient Characteristics
4.2. DNA Extraction of Biopsy Samples
4.3. Microsatellite Analyses
4.4. Semi-Quantitative Real Time PCR (QPCR)
| Genes | Forward primers | Reverse primers |
|---|---|---|
| Target genes | ||
| TOP1 | 5'-ATGGGTACAGTGTGCT-3' (intron 19) | 5-'AGTTTGGAGGTTCCCAG-3' (exon 20) |
| TOP2A | 5'-GCCATTGGCTGTGGTATTG-3' (exon 11) | 5'-GAGAAGCTTCTCGAACATTGAG-3' (exon 12) |
| TOP2B | 5'-GATTGGGTACTAGTACAGCT-3' (exon 16) | 5'-GAATAGAAGGTAGGGGGATG-3' (intron 16) |
| Reference genes | ||
| APP | 5'-TCAGGTTGACGCCGCTGT-3' | 5'-ACCCCAGAGGAGCGCCACCTG-3' |
| DCK | 5'-GCCGCCACAAGACTAAGGAAT-3' | 5'-AGCTGCCCGTCTTTCTCAGCCAGC-3' |
4.5. Immunohistochemical Analyses
4.6. Statistics
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Huvos, A.G. Bone Tumors, Diagnosis, Treatment, and Prognosis, 2nd ed.; W.B. Saunders: Philadelphia, PA, USA, 1991; pp. 85–155, 343–393. [Google Scholar]
- Huvos, A.G.; Rosen, G.; Marcove, R.C. Primary osteogenic sarcoma. Pathologic aspects in twenty patients after treatment with chemotherapy, in bloc resection and prosthetic bone replacement. Arch. Pathol. Lab. Med. 1977, 101, 14–18. [Google Scholar]
- Le Deley, M.C.; Guinebretière, J.M.; Gentet, J.C.; Pacquement, H.; Pichon, F.; Marec-Bérard, P.; Entz-Werlé, N.; Schmitt, C.; Brugières, L.; Vanel, D.; et al. Société Française d’Oncologie Pédiatrique (SFOP). SFOP OS94: A randomised trial comparing preoperative high-dose methotrexate plus doxorubicin to high-dose methotrexate plus etoposide and ifosfamide in osteosarcoma patients. Eur. J. Cancer 2007, 43, 752–761. [Google Scholar]
- Lau, C.C. Molecular classification of osteosarcoma. Cancer Treat. Res. 2009, 152, 59–65. [Google Scholar]
- Jarvinen, T.A.; Tanner, M.; Barlund, M.; Borg, A.; Isola, J. Characterization of topoisomerase II α gene amplification and deletion in breast cancer. Genes Chromosom. Cancer 1999, 26, 142–150. [Google Scholar] [CrossRef]
- Feister, H.A.; Swartz, D.; Odgren, P.R.; Holden, J.; Hock, J.M.; Onyia, J.; Bidwell, J.P. Topoisomerase II expression in osseous tissue. J. Cell Biochem. 1997, 67, 451–465. [Google Scholar] [CrossRef]
- Feister, H.A.; Onyia, J.E.; Miles, R.R.; Yang, X.; Galvin, R.; Hock, J.M.; Bidwell, J.P. The expression of the nuclear matrix proteins NuMA, topoisomerase II-alpha, and -beta in bone and osseous cell culture: Regulation by parathyroid hormone. Bone 2000, 26, 227–234. [Google Scholar] [CrossRef]
- Turley, H.; Comley, M.; Houlbrook, S.; Nozaki, N.; Kikuchi, A.; Hickson, I.D.; Gatter, K.; Harris, A.L. The distribution and expression of the two isoforms of DNA topoisomerase II in normal and neoplastic human tissues. Br. J. Cancer 1997, 75, 1340–1346. [Google Scholar] [CrossRef]
- Harisi, R.; Dudás, J.; Timár, F.; Pogány, G.; Timár, J.; Kovalszky, I.; Szendroi, M.; Jeney, A. Invasive growth and topoisomerase-switch induced by tumorous extracellular matrix in osteosarcoma cell culture. Cell Biol. Int. 2005, 29, 959–967. [Google Scholar] [CrossRef]
- Christiansen, M.; Kveiborg, M.; Kassem, M.; Clark, B.F.; Rattan, S.I. CBFA1 and topoisomerase I mRNA levels decline during cellular aging of human trabecular osteoblasts. J. Gerontol. A Biol. Sci. Med. Sci. 2000, 55, B194–B200. [Google Scholar] [CrossRef]
- Wang, J.C. DNA topoisomerases. Annu. Rev. Biochem. 1996, 65, 635–692. [Google Scholar] [CrossRef]
- Entz-Werle, N.; Schneider, A.; Kalifa, C.; Voegeli, A.C.; Tabone, M.D.; Marec-Berard, P.; Marcellin, L.; Pacquement, H.; Terrier, P.; Boutard, P.; et al. Genetic alterations in primary osteosarcoma from 54 children and adolescents by targeted allelotyping. Br. J. Cancer 2003, 88, 1925–1931. [Google Scholar] [CrossRef]
- Entz-Werlé, N.; Stoetzel, C.; Berard-Marec, P.; Kalifa, C.; Brugieres, L.; Pacquement, H.; Schmitt, C.; Tabone, M.D.; Gentet, J.C.; Quillet, R.; et al. Frequent genomic abnormalities at TWIST in human pediatric osteosarcomas. Int. J. Cancer 2005, 117, 349–355. [Google Scholar] [CrossRef]
- Entz-Werle, N.; Lavaux, T.; Metzger, N.; Stoetzel, C.; Lasthaus, C.; Marec, P.; Kalifa, C.; Brugieres, L.; Pacquement, H.; Schmitt, C.; et al. Involvement of MET/TWIST/APC combination or the potential role of ossification factors in pediatric high-grade osteosarcoma oncogenesis. Neoplasia 2007, 9, 678–688. [Google Scholar] [CrossRef]
- Jarvinen, T.A.; Tanner, M.; Rantanen, V.; Barlund, V.; Barlund, A.; Borg, A.; Grenman, S.; Isola, J. Amplification and deletion of topoisomerase IIα associate with ErbB-2 amplification and affect sensitivity to topoisomerase II inhibitor doxorubicin in breast cancer. Am. J. Pathol. 2000, 156, 839–847. [Google Scholar] [CrossRef]
- Knoop, A.S.; Knudsen, H.; Balslev, E. Retrospective analysis of topoisomerase IIa amplifications and deletions as predictive markers in primary breast cancer patients randomly assigned to cyclophosphamide, methotrexate, and fluorouracil or cyclophosphamide, epirubicin, and fluorouracil: Danish Breast Cancer Cooperative Group. J. Clin. Oncol. 2005, 23, 7483–7490. [Google Scholar] [CrossRef]
- Nielsen, K.V.; Ejlertsen, B.; Moller, S. The value of TOP2A gene copy number ariation as a biomarker in breast cancer: Update of DBCG trial 89D. Acta Oncol. 2008, 47, 725–734. [Google Scholar] [CrossRef]
- O’Malley, F.P.; Chia, S.; Tu, D. Topoisomerase II α and responsiveness of breast cancer to adjuvant chemotherapy. J. Natl. Cancer Inst. 2009, 101, 644–650. [Google Scholar] [CrossRef]
- Somers, G.R.; Ho, M.; Zielenska, M.; Squire, J.A.; Thorner, P.S. HER2 amplification and overexpression is not present in pediatric osteosarcoma: A tissue microarray study. Pediatr. Dev. Pathol. 2005, 8, 525–532. [Google Scholar] [CrossRef]
- Rakesh, K.V.; Gupta, N.; Kakkar, N.; Sharma, S.C. Prognostic and predictive value of c-erbB2 overexpression in osteogenic sarcoma. J. Cancer Res. Ther. 2006, 2, 20–23. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, Y.; Yuan, Z.; Wang, C.; Shi, Y. Predicting chemosensitivity in osteosarcoma prior to chemotherapy: An investigational study of biomarkers with immunohistochemistry. Oncol. Lett. 2012, 3, 1011–1016. [Google Scholar]
- Ma, Y.; Ren, Y.; Han, E.Q.; Li, H.; Chen, D.; Jacobs, J.J.; Gitelis, S.; O’Keefe, R.J.; Konttinen, Y.T.; Yin, G.; et al. Inhibition of the Wnt-β-catenin and Notch signaling pathways sensitizes osteosarcoma cells to chemotherapy. Biochem. Biophys. Res. Commun. 2013, 431, 274–279. [Google Scholar] [CrossRef]
- Qu, J.T.; Wang, M.; He, H.L.; Tang, Y.; Ye, X.J. The prognostic value of elevated vascular endothelial growth factor in patients with osteosarcoma: A meta-analysis and systemic review. J. Cancer Res. Clin. Oncol. 2012, 138, 819–825. [Google Scholar] [CrossRef]
- Romero, A.; Caldés, T.; Díaz-Rubio, E.; Martín, M. Topoisomerase 2 alpha: A real predictor of anthracycline efficacy? Clin. Transl. Oncol. 2012, 14, 163–168. [Google Scholar] [CrossRef]
- Olsen, K.E.; Knudsen, H.; Rasmussen, B.B.; Balslev, E.; Knoop, A.; Ejlertsen, B.; Nielsen, K.V.; Schönau, A.; Overgaard, J.; Danish Breast Cancer Co-operative Group. Amplification of HER2 and TOP2A and deletion of TOP2A genes in breast cancer investigated by new FISH probes. Acta Oncol. 2004, 43, 35–42. [Google Scholar] [CrossRef]
- Romero, A.; Martín, M.; Cheang, M.C.; López García-Asenjo, J.A.; Oliva, B.; He, X.; de la Hoya, M.; García Sáenz, J.Á.; Arroyo Fernández, M.; Díaz Rubio, E.; et al. Amplification of HER2 and TOP2A and deletion of TOP2A genes in breast cancer investigated by new FISH probes. Am. J. Pathol. 2011, 178, 1453–1460. [Google Scholar]
- GDB Homepage. Available online: http://www.gdb.org/ (accessed on 5 April 2013).
- A New Gene Map of the Human Genome. Available online: http://www.ncbi.nlm.nih.gov/genemap99/ (accessed on 5 April 2013).
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nguyen, A.; Lasthaus, C.; Guerin, E.; Marcellin, L.; Pencreach, E.; Gaub, M.-P.; Guenot, D.; Entz-Werle, N. Role of Topoisomerases in Pediatric High Grade Osteosarcomas: TOP2A Gene Is One of the Unique Molecular Biomarkers of Chemoresponse. Cancers 2013, 5, 662-675. https://doi.org/10.3390/cancers5020662
Nguyen A, Lasthaus C, Guerin E, Marcellin L, Pencreach E, Gaub M-P, Guenot D, Entz-Werle N. Role of Topoisomerases in Pediatric High Grade Osteosarcomas: TOP2A Gene Is One of the Unique Molecular Biomarkers of Chemoresponse. Cancers. 2013; 5(2):662-675. https://doi.org/10.3390/cancers5020662
Chicago/Turabian StyleNguyen, Aurelia, Christelle Lasthaus, Eric Guerin, Luc Marcellin, Erwan Pencreach, Marie-Pierre Gaub, Dominique Guenot, and Natacha Entz-Werle. 2013. "Role of Topoisomerases in Pediatric High Grade Osteosarcomas: TOP2A Gene Is One of the Unique Molecular Biomarkers of Chemoresponse" Cancers 5, no. 2: 662-675. https://doi.org/10.3390/cancers5020662
APA StyleNguyen, A., Lasthaus, C., Guerin, E., Marcellin, L., Pencreach, E., Gaub, M.-P., Guenot, D., & Entz-Werle, N. (2013). Role of Topoisomerases in Pediatric High Grade Osteosarcomas: TOP2A Gene Is One of the Unique Molecular Biomarkers of Chemoresponse. Cancers, 5(2), 662-675. https://doi.org/10.3390/cancers5020662





