Ex Vivo Behaviour of Human Bone Tumor Endothelial Cells
Abstract
:1. Introduction
2. Experimental Section
2.1. Human Tissue Samples
2.2. Cytofluorimetric Analysis
2.3. Immunofluorescence Studies
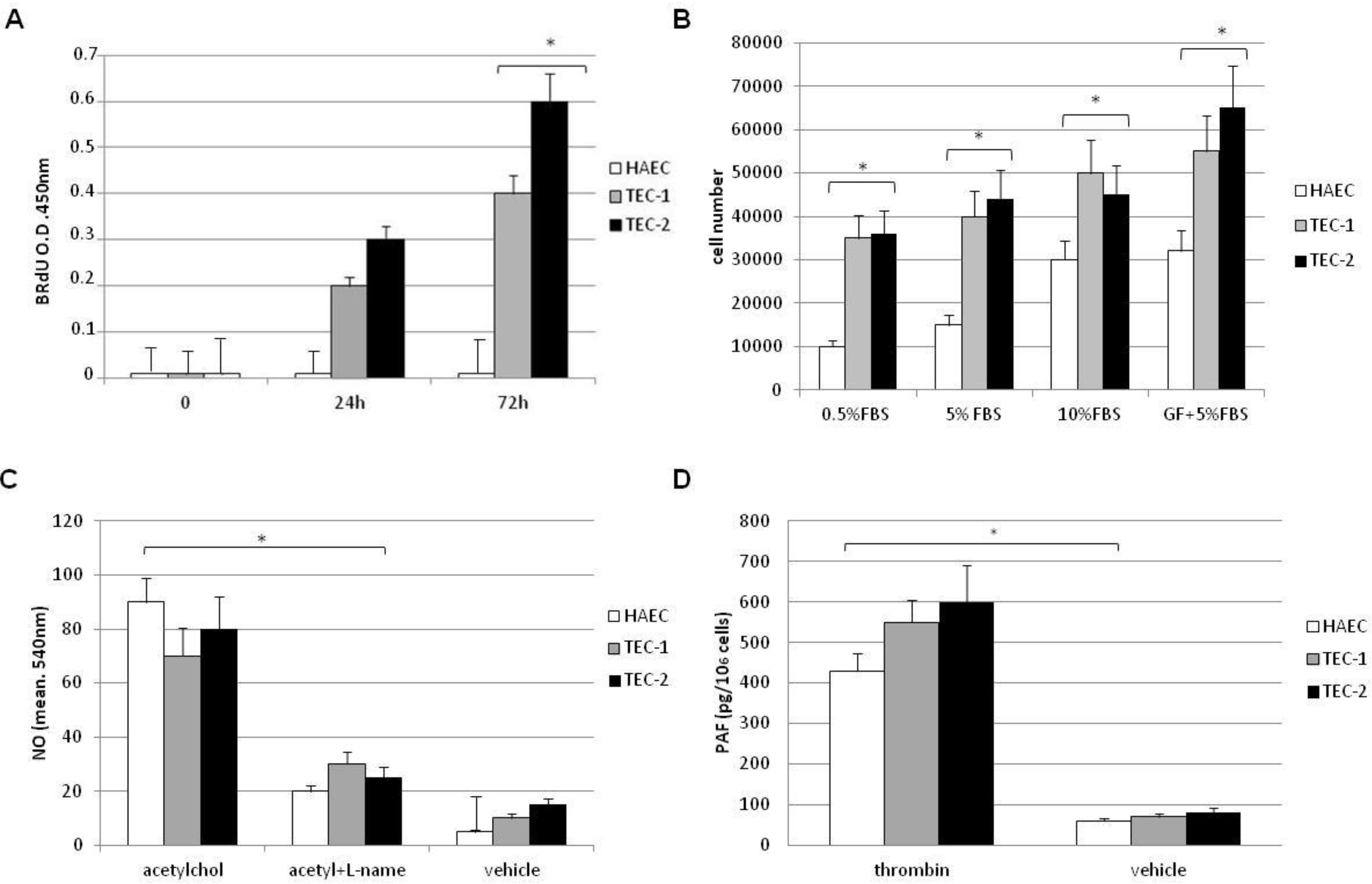
2.4. Nitric Oxide and Platelet Activating Factor (PAF) Assays
2.5. Cell Proliferation Assay
2.6. Tube Formation and Migration Assay
2.7. Statistical Analysis
3. Results
3.1. Characterization of TECs
| Case | Gender | Age | Histotype | Grade |
|---|---|---|---|---|
| 1 | M | 30 | GCT * | I |
| 2 | M | 25 | Chondrosarcoma | II |
| 3 | M | 40 | Fibrosarcoma | I |
| 4 | F | 35 | Osteosarcoma | II |
| 5 | F | 45 | Chondrosarcoma | II |
| 6 | F | 23 | Chondrosarcoma | II |
| 7 | M | 43 | Osteosarcoma | III |
| 8 | F | 31 | Fibrosarcoma | II |
| 9 | M | 26 | Osteosarcoma | II |
| 10 | M | 27 | GCT * | I |
| 11 | F | 22 | Chondrosarcoma | II |
| 12 | F | 20 | GCT * | I |
| 13 | F | 45 | GCT * | I |
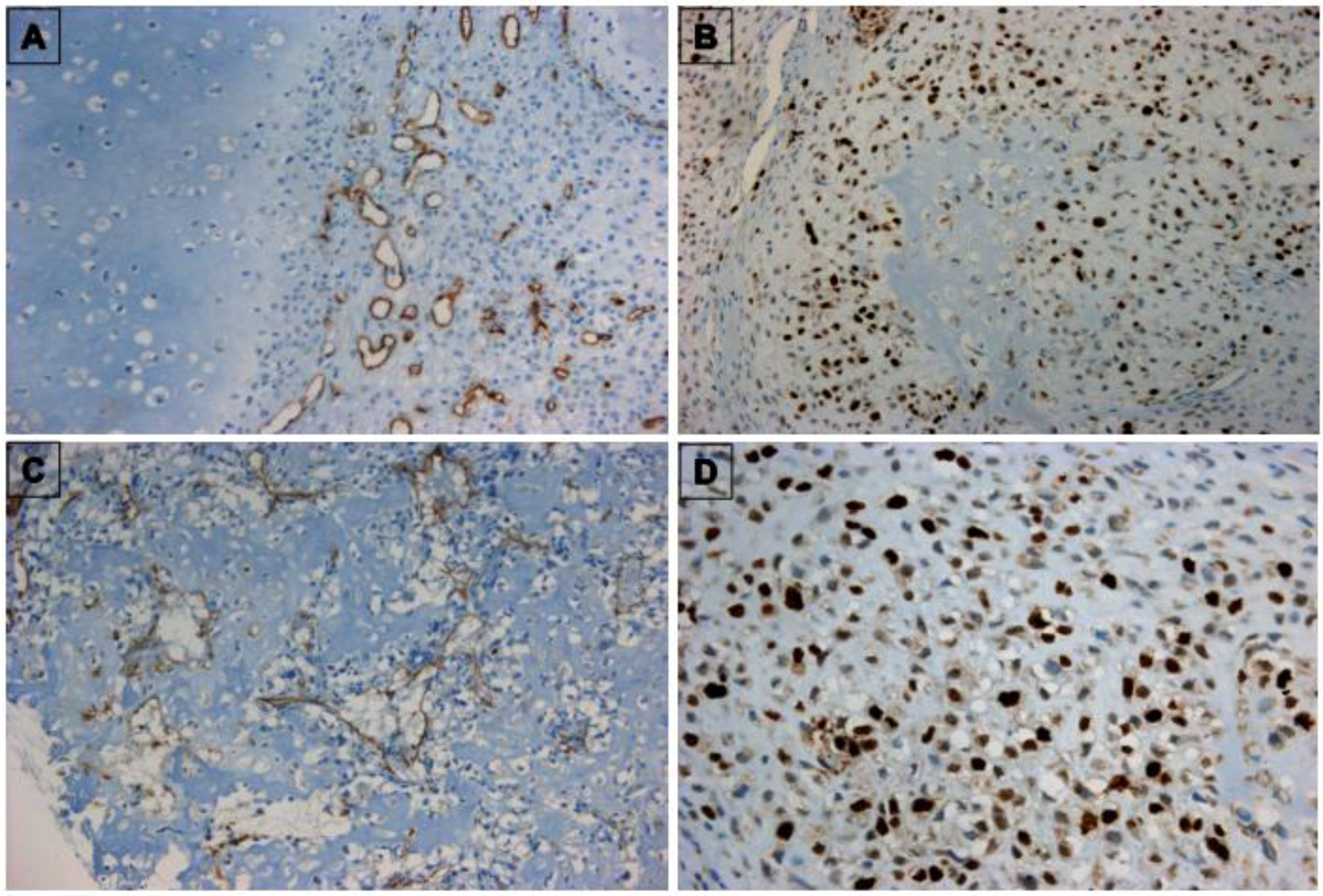
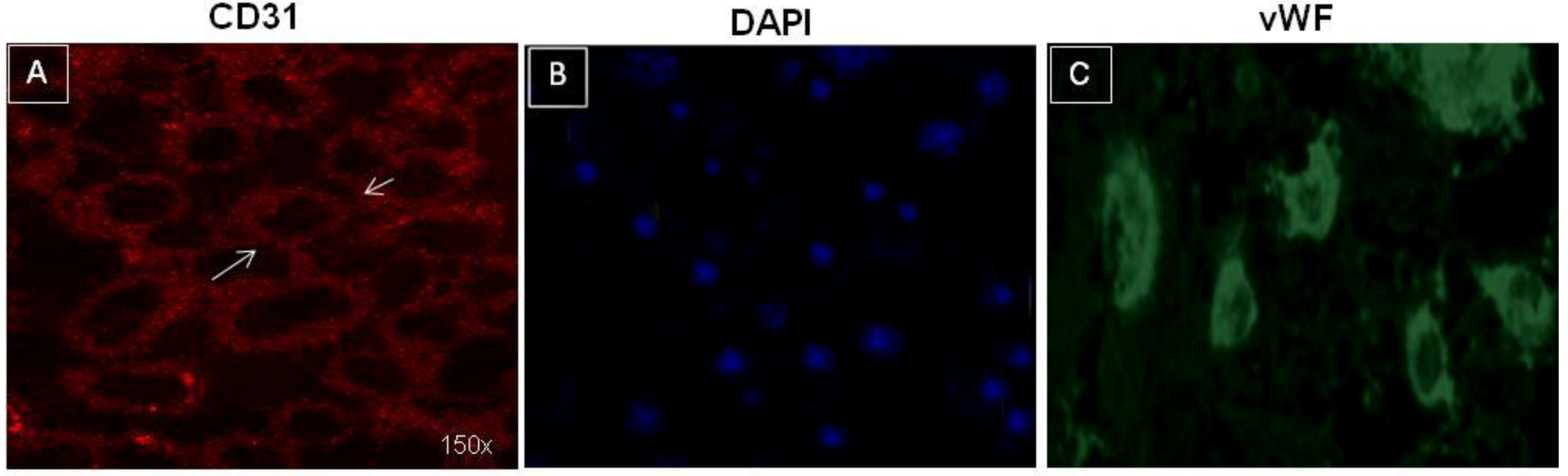

3.2. In Vitro Behaviour of TECs
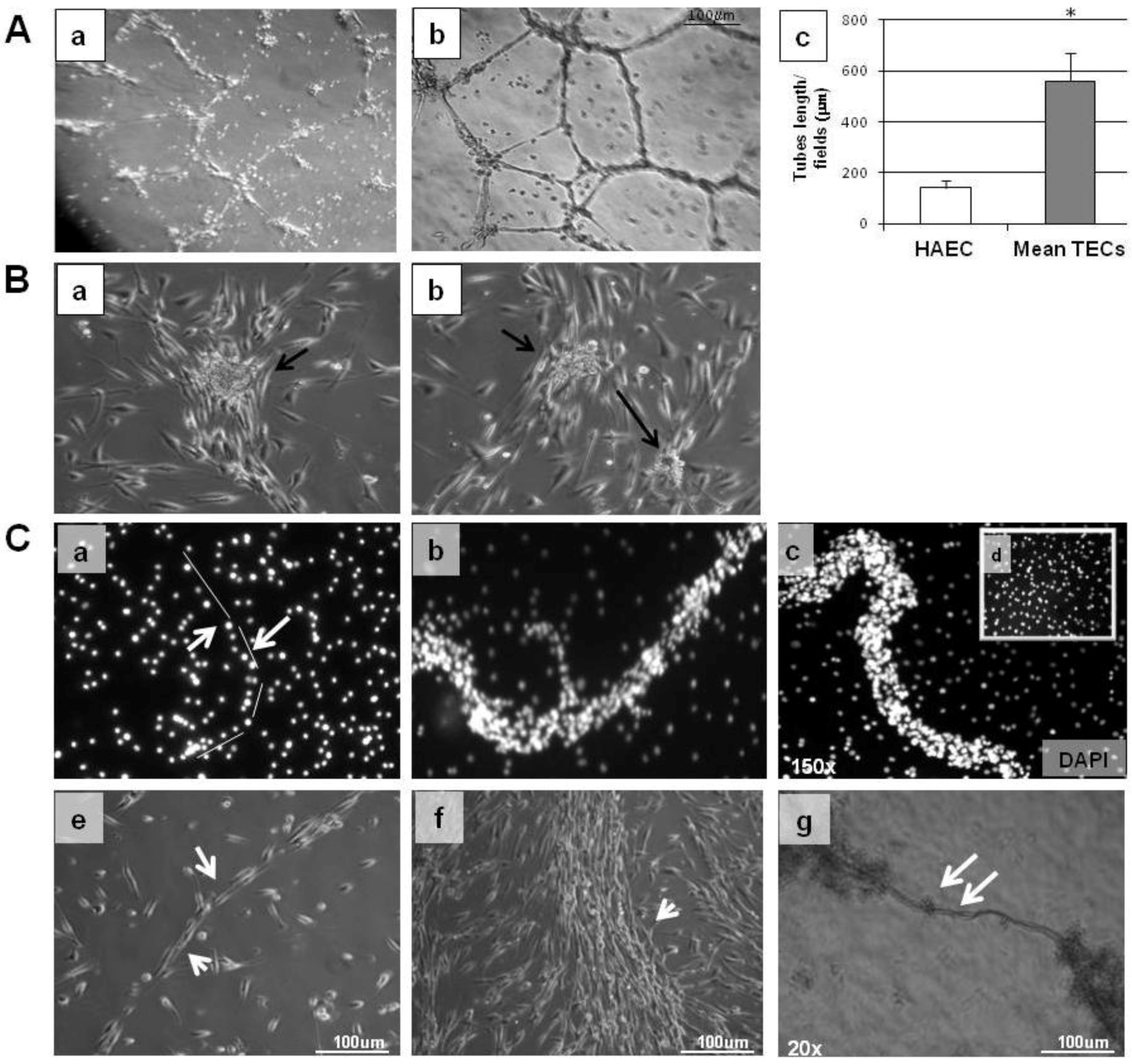
4. Discussion
5. Conclusions
References
- Mundy, G.R. Metastasis to bone: Causes, consequences and therapeutic opportunities. Nat. Rev. Cancer 2002, 2, 584–593. [Google Scholar] [CrossRef]
- Guise, T. Examining the metastatic niche: Targeting the microenvironment. Semin. Oncol. 2010, 37, S2–S14. [Google Scholar] [CrossRef]
- Potente, M.; Gerhardt, H.; Carmeliet, P. Basic and therapeutic aspects of angiogenesis. Cell 2011, 146, 873–887. [Google Scholar] [CrossRef]
- De Nigris, F.; Schiano, C.; Infante, T.; Napoli, C. CXCR4 inhibitors: Tumor vasculature and therapeutic challenges. Recent Pat. Anticancer Drug Discov. 2012, 7, 251–264. [Google Scholar] [CrossRef]
- Hanahan, D.; Coussens, L.M. Accessories to the crime: Functions of cells recruited to the tumor microenvironment. Cancer Cell 2012, 21, 309–322. [Google Scholar] [CrossRef]
- Jain, R.K.; Duda, D.G.; Willett, C.G.; Sahani, D.V.; Zhu, A.X.; Loeffler, J.S.; Batchelor, T.T.; Sorensen, A.G. Biomarkers of response and resistance to antiangiogenic therapy. Nat. Rev. Clin. Oncol. 2009, 6, 327–338. [Google Scholar] [CrossRef]
- St. Croix, B.; Rago, C.; Velculescu, V.; Traverso, G.; Romans, K.E.; Montgomery, E.; Lal, A.; Riggins, G.J.; Lengauer, C.; Vogelstein, B.; et al. Genes expressed in human tumor endothelium. Science 2000, 289, 1197–1202. [Google Scholar] [CrossRef]
- Bagley, R.G.; Honma, N.; Weber, W.; Boutin, P.; Rouleau, C.; Shankara, S.; Kataoka, S.; Ishida, I.; Roberts, B.L.; Teicher, B.A. Endosialin/TEM 1/CD248 is a pericyte marker of embryonic and tumor neovascularization. Microvasc. Res. 2008, 76, 180–188. [Google Scholar] [CrossRef]
- De Bock, K.; Cauwenberghs, S.; Carmeliet, P. Vessel abnormalization: Another hallmark of cancer? Molecular mechanisms and therapeutic implications. Curr. Opin. Genet. Dev. 2011, 21, 73–79. [Google Scholar] [CrossRef]
- Bussolati, B.; Deregibus, M.C.; Camussi, G. Characterization of molecular and functional alterations of tumor endothelial cells to design anti-angiogenic strategies. Curr. Vasc. Pharmacol. 2010, 8, 220–232. [Google Scholar] [CrossRef]
- Di Tomaso, E.; Capen, D.; Haskell, A.; Hart, J.; Logie, J.; Jain, R.K.; McDonald, D.M.; Jones, R.; Munn, L.L. Mosaic tumor vessels: Cellular basis and ultrastructure of focal regions lacking endothelial cell markers. Cancer Res. 2005, 65, 5740–5749. [Google Scholar] [CrossRef]
- Virrey, J.J.; Golden, E.B.; Sivakumar, W.; Wang, W.; Pen, L.; Schönthal, A.H.; Hofman, F.M.; Chen, T.C. Glioma-associated endothelial cells are chemoresistant to temozolomide. J. Neurooncol. 2009, 95, 13–22. [Google Scholar] [CrossRef]
- Xiong, Y.Q.; Sun, H.C.; Zhang, W.; Zhu, X.D.; Zhuang, P.Y.; Zhang, J.B.; Wang, L.; Wu, W.Z.; Qin, L.X.; Tang, Z.Y. Human hepatocellular carcinoma tumor-derived endothelial cells manifest increased angiogenesis capability and drug resistance compared with normal endothelial cells. Clin. Cancer Res. 2009, 15, 4838–4346. [Google Scholar] [CrossRef]
- Bacci, G.; Longhi, A.; Versari, M.; Mercuri, M.; Briccoli, A.; Picci, P. Prognostic factors for osteosarcoma of the extremity treated with neoadjuvant chemotherapy 15-year experience in 789 patients treated at a single institution. Cancer 2006, 106, 1154–1161. [Google Scholar] [CrossRef]
- Ganjoo, K.; Jacobs, C. Antiangiogenesis agents in the treatment of soft tissue sarcomas. Cancer 2010, 116, 1177–1183. [Google Scholar] [CrossRef]
- De Nigris, F.; Rossiello, R.; Schiano, C.; Arra, C.; Williams-Ignarro, S.; Barbieri, A.; Lanza, A.; Balestrieri, A.; Giuliano, M.T.; Ignarro, L.J.; et al. Deletion of Yin Yang 1 protein in osteosarcoma cells on cell invasion and CXCR4/angiogenesis and metastasis. Cancer Res. 2008, 68, 1797–1808. [Google Scholar] [CrossRef]
- De Nigris, F.; Crudele, V.; Giovane, A.; Casamassimi, A.; Giordano, A.; Garban, H.J.; Cacciatore, F.; Pentimalli, F.; Marquez-Garban, D.C.; Petrillo, A.; et al. CXCR4/YY1 inhibition impairs VEGF network and angiogenesis during malignancy. Proc. Natl. Acad. Sci. USA 2010, 107, 14484–14489. [Google Scholar] [CrossRef]
- Schiano, C.; Grimaldi, V.; Casamassimi, A.; Infante, T.; Esposito, A.; Giovane, A.; Napoli, C. Different expression of CD146 in human normal and osteosarcoma cell lines. Med. Oncol. 2012, 29, 2998–3002. [Google Scholar] [CrossRef]
- McDonald, D.M.; Foss, A.J. Endothelial cells of tumor vessels: Abnormal but not absent. Cancer Metastasis Rev. 2000, 19, 109–120. [Google Scholar] [CrossRef]
- Zöller, M. CD44: Can a cancer-initiating cell profit from an abundantly expressed molecule? Nat. Rev. Cancer 2011, 24, 254–267. [Google Scholar] [CrossRef]
- Fuchs, S.; Dohle, E.; Kolbe, M.; Kirkpatrick, C.J. Outgrowth endothelial cells: Sources, characteristics and potential applications in tissue engineering and regenerative medicine. Adv. Biochem. Eng. Biotechnol. 2010, 123, 201–217. [Google Scholar]
- Kanczler, J.M.; Oreffo, R.O. Osteogenesis and angiogenesis: The potential for engineering bone. Eur. Cell Mater. 2008, 15, 100–114. [Google Scholar]
- Bussolati, B.; Deambrosis, I.; Russo, S.; Deregibus, M.C.; Camussi, G. Altered angiogenesis and survival in human tumor-derived endothelial cells. FASEB J. 2003, 17, 1159–1161. [Google Scholar]
- De Bock, K.; Cauwenberghs, S.; Carmeliet, P. Vessel abnormalization: Another hallmark of cancer? Molecular mechanisms and therapeutic implications. Curr. Opin. Genet. Dev. 2011, 21, 73–79. [Google Scholar] [CrossRef]
- Fadini, G.P.; Losordo, D.; Dimmeler, S. Critical reevaluation of endothelial progenitor cell phenotypes for therapeutic and diagnostic use. Circ. Res. 2012, 110, 624–637. [Google Scholar] [CrossRef]
- Yoder, M.C.; Mead, L.E.; Prater, D.; Krier, T.R.; Mroueh, K.N.; Li, F.; Krasich, R.; Temm, C.J.; Prchal, J.T.; Ingra, D.A. Redefining endothelial progenitor cells via clonal analysis and hematopoietic stem/progenitor cell principals. Blood 2007, 109, 1801–1809. [Google Scholar] [CrossRef]
- Tilki, D.; Hohn, H.-P.; Ergün, B.; Rafii, S.; Ergün, S. Emerging biology of vascular wall progenitor cells in health and disease. Trends Mol. Med. 2009, 15, 501–509. [Google Scholar]
- Zhu, H.; Shao, Q.; Sun, X.; Deng, Z.; Yuan, X.; Yu, D.; Zhou, X.; Ding, Y. The mobilization, recruitment and contribution of bone marrow-derived endothelial progenitor cells to the tumor neovascularization occur at an early stage and throughout the entire process of hepatocellular carcinoma growth. Oncol. Rep. 2012, 28, 1217–1224. [Google Scholar]
- Huang, J.; Kontos, C.D. PTEN modulates vascular endothelial growth factor-mediated signaling and angiogenic effects. J. Biol. Chem. 2002, 277, 10760–10766. [Google Scholar] [CrossRef]
- Liu, W.; Ahmad, S.A.; Reinmuth, N.; Shaheen, R.M.; Jung, Y.D.; Fan, F.; Ellis, L.M. Endothelial cell survival and apoptosis in the tumor vasculature. Apoptosis 2000, 5, 323–328. [Google Scholar] [CrossRef]
- Díez, M.; Musri, M.M.; Ferrer, E.; Barberà, J.A.; Peinado, V.I. Endothelial progenitor cells undergo an endothelial-to-mesenchymal transition- like process mediated by TGFbetaRI. Cardiovasc. Res. 2010, 88, 502–511. [Google Scholar] [CrossRef]
- Kurose, K.; Hoshaw-Woodard, S.; Adeyinka, A.; Lemeshow, S.; Watson, P.H.; Eng, C. Genetic model of multi-step breast carcinogenesis involving the epithelium and stroma: Clues to tumour-microenvironment interactions. Hum. Mol. Genet. 2001, 10, 1907–1913. [Google Scholar] [CrossRef]
- Akino, T.; Hida, K.; Hida, Y.; Tsuchiya, K.; Freedman, D.; Muraki, C.; Ohga, N.; Matsuda, K.; Akiyama, K.; Harabayashi, T.; et al. Cytogenetic abnormalities of tumor-associated endothelial cells in human malignant tumors. Am. J. Pathol. 2009, 175, 2657–2667. [Google Scholar] [CrossRef]
- Secchiero, P.; Zorzet, S.; Tripodo, C.; Corallini, F.; Melloni, E.; Caruso, L.; Bosco, R.; Ingrao, S.; Zavan, B.; Zauli, G. Human bone marrow mesenchymal stem cells display anti-cancer activity in SCID mice bearing disseminated non-Hodgkin’s lymphoma xenografts. PLoS One 2010, 1, e11140. [Google Scholar]
- Ho, I.A.; Toh, H.C.; Ng, W.H.; Teo, Y.L.; Guo, C.M.; Hui, K.M.; Lam, P.Y. Human bone marrow-derived mesenchymal stem cells suppress human glioma growth through inhibition of angiogenesis. Stem. Cells 2013, 31, 146–155. [Google Scholar] [CrossRef]
- Regan, E.R.; Aird, W.C. Dynamical systems approach to endothelial heterogeneity. Circ. Res. 2012, 111, 110–130. [Google Scholar] [CrossRef]
- Resch, T.; Pircher, A.; Kähler, C.M.; Pratschke, J.; Hilbe, W. Endothelial progenitor cells: Current issues on characterization and challenging clinical applications. Stem Cell Rev. 2012, 8, 926–939. [Google Scholar] [CrossRef]
- Langley, R.R.; Fidler, I.J. The seed and soil hypothesis revisited-The role of tumor stroma interactions in metastasis to different organs. Int. J. Cancer 2011, 128, 2527–2535. [Google Scholar] [CrossRef]
- Amini, A.R.; Laurencin, C.T.; Nukavarapu, S.P. Differential analysis of peripheral blood- and bone marrow-derived endothelial progenitor cells for enhanced vascularization in bone tissue engineering. J. Orthop. Res. 2012, 30, 1507–1515. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Infante, T.; Cesario, E.; Gallo, M.; Fazioli, F.; De Chiara, A.; Tutucci, C.; Apice, G.; De Nigris, F. Ex Vivo Behaviour of Human Bone Tumor Endothelial Cells. Cancers 2013, 5, 404-417. https://doi.org/10.3390/cancers5020404
Infante T, Cesario E, Gallo M, Fazioli F, De Chiara A, Tutucci C, Apice G, De Nigris F. Ex Vivo Behaviour of Human Bone Tumor Endothelial Cells. Cancers. 2013; 5(2):404-417. https://doi.org/10.3390/cancers5020404
Chicago/Turabian StyleInfante, Teresa, Elena Cesario, Michele Gallo, Flavio Fazioli, Annarosaria De Chiara, Cristina Tutucci, Gaetano Apice, and Filomena De Nigris. 2013. "Ex Vivo Behaviour of Human Bone Tumor Endothelial Cells" Cancers 5, no. 2: 404-417. https://doi.org/10.3390/cancers5020404




