Vitamin D: Pharmacokinetics and Safety When Used in Conjunction with the Pharmaceutical Drugs Used in Cancer Patients: A Systematic Review
Abstract
:1. Introduction
2. Results and Discussion
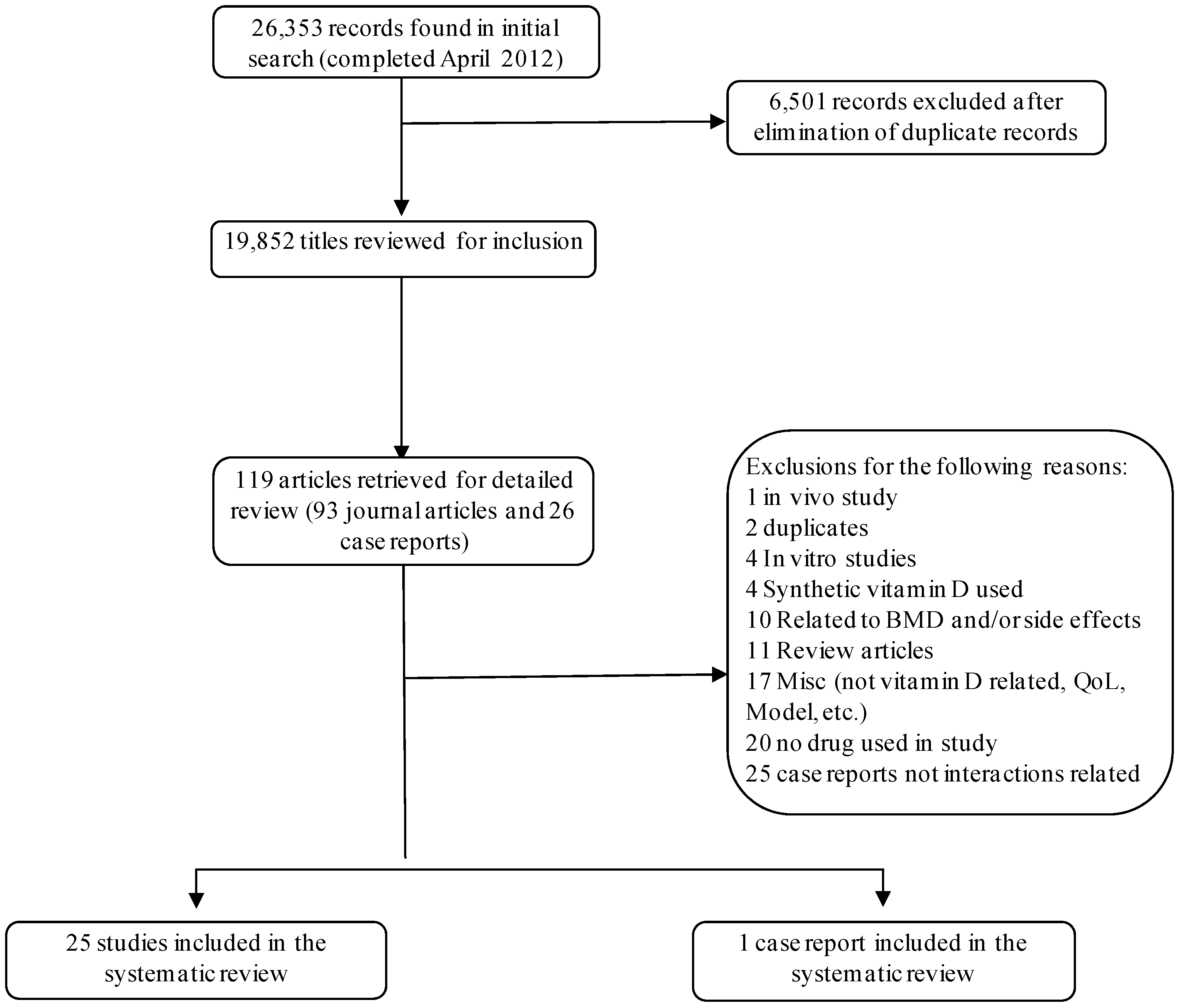
2.1. Overview of the Interactions
2.2. Impact on Pharmacokinetics
| Pharmaceutical drug | Calcitriol/DN-101 | Calciferol | 1α-Hydroxyvitamin D3 |
|---|---|---|---|
| 1,3-bis 1 nitrosurea | 1 | ||
| 13-cis retinoic acid | 1 | ||
| Altizide + spironolactone | 1 | ||
| Carboplatin | 1 | ||
| Cyclophosphamide | 1 | ||
| Cytarabine | 1 | ||
| Cytosine | 1 | ||
| Cytosine arabinoside | 1 | ||
| Dexamethasone | 7 | ||
| Docetaxel | 7 | ||
| Estramustine | 1 | ||
| Gefitinib | 2 | ||
| Hydrochlorothiazide | 1 | ||
| Interferon | 1 | 1 | |
| Melphalan | 1 | ||
| Mitoxantrone | 1 | ||
| Naproxen | 1 | ||
| Paclitaxel | 1 | ||
| Prednisone | 1 | 1 | |
| Zoledronate | 1 |
3. Experimental Section
3.1. Sources
3.2. Study Selection
3.2.1. Inclusion Criteria
3.2.2. Exclusion Criteria
3.3. Data Extraction
4. Conclusions
Appendix
| Study/Year | Cancer type | Drug(s) | Form of Vitamin D | Design Treated/ Control | Study Endpoint | Study Protocol | Outcome |
|---|---|---|---|---|---|---|---|
| Cohen et al. [20] | Multiple myeloma | BCP: 1-3-bis (2-chloro-ethyl) 1-nitrosourea, cyclophosphamide and prednisone | Calciferol | RCT/cross over | (1) Compare directly the BCP regimen with MP | Pt randomized separately to either: | Toxicity of the supplemental drug package did not appear to be of major significance, there was greater GI toxicity for the active regimen. |
| (2) Determine the response of patients initially resistant to one regimen when subsequently treated with the other; | BCP: BCNU, 75 mg/m2 i.v., and cyclophosphamide, 400 mg/m2 i.v., each in single doses, plus prednisone for 75 mg/day p.o. × 7 days; or MP: melphalan, 8 mg/m2/day p.o., for 4 days, and prednisone, 75 mg/day p.o., for 7 days. Each regimen was given every 4 week for 6 courses. | ||||||
| MP: melphalan and prednisone | 373/0 | (3) Determine if the combination of sodium fluoride, calcium gluconate, vitamin D, and fluoxymesterone could produce useful clinical benefit by repairing or strengthening bone structure. | Based on response to the above treatment, patients were then randomized to receive the active drug package (fluoxymesterone, 25 mg/m2 daily, sodium fluoride, 150 mg/day, calcium gluconate, 2 g t.i.d., and vitamin D (Calciferol), 50,000 U tabs twice a week, all given orally (p.o.), or a placebo package. | There was no significant difference between patients receiving the placebo package vs. the active agents in terms of bone pain, tenderness, or development of new fractures. | |||
| Hellstrom et al. [43] | Acute leukemia myelo-dysplastic syndromes | Cytosine arabinoside (Ara-c) | 1α-hydroxy-vitamin D3 | RCT | Study the efficacy and toxicity of each combination | 1ST arm: IFN 3 million units per day, 13-cis-RA 1 mg/kg po per day, D3 start with 1 µg per day, increasing dose until mild hypercalcemia develops. | High rate of side effects due to IFN |
| Alpha interferon (IFN) | |||||||
| 13-cis-retinoic acid (13-cis-RA) | 16/37/28/7 | 2ND arm: Ara-c 15 mg/m2 per day, if no pt response, increased to 25 mg/m2. 3RD arm: all four drugs given simultaneously | 13-cis-RA and D3 were well tolerated and s/e (hypercalcemia) easily controlled with dose adjustment. | ||||
| Slapak et al. [44] | AML | Cytarabine | Calcitriol | Uncontrolled study | Treatment of patients with AML over the age of 65 years. | Cytarabine was administered by continuous intravenous infusion at a dose of 20 mg/m2/day for 21 days. | Thirteen patients (45%) obtained a complete remission, and 10 patients (34%) had a partial response for an overall 79% response rate. There were three early deaths. The median remission duration was 9.8 months. |
| Hydroxyurea 500 mg orally (po) q12 h was instituted 24 h prior to cytarabine and continued through day 21. | |||||||
| Hydroxyurea | 28/0 | Calcitriol (0.25 pg, PO Q12 h) was begun on the first day of cytarabine therapy and continued until relapse or the patient went off study | Two patients experienced hypercalcemia, in one patient calcitriol was held until normal levels were reached. | ||||
| Muindi et al. [30] | Advanced solid tumors | Paclitaxel | Calcitriol | Phase I/PK | Determine the maximum tolerated dose and pharmacokinetics of calcitriol when administered with paclitaxel. | Escalating doses of calcitriol were given orally for 3 consecutive days each week, and paclitaxel (80 mg/m2) was given intravenously weekly. |
|
| The starting dose of calcitriol was 4 µg and the maximum dose administered was 38 µg. | |||||||
| 36/0 | However, the study was halted since the study found decreased bioavailability of calcitriol with high dose oral administration.Dose escalation: 4, 6, 8, 11, 13, 17, 22, 29, 38 µg/day | ||||||
| Beer et al. [45] | AIPC | Docetaxel | Calcitriol | Phase I/PK | Determine the safety and efficacy of weekly high-dose oral calcitriol and docetaxel. | Day 1: oral calcitriol (0.5_g/kg) |
|
| Day 2: iv docetaxel (36 mg/m2) | |||||||
| repeated weekly for 6 weeks of an 8-week cycle. | |||||||
| 37/0 | Premedication with dexamethasone 8 mg orally 12 h and 1 h before docetaxel infusion and 12 after docetaxel infusion was given. | ||||||
| Beer et al. [46] | AIPC | Carboplatin | Calcitriol | Uncontrolled trial | PSA response defined as a 50% reduction confirmed 4 weeks later. | Day 1: oral calcitriol (0.5 µg/kg) on day 1 |
|
| Day 2: iv carboplatin (AUC 7 or AUC 6 in patients with prior radiation). | |||||||
| 17/0 | Repeated every 4 weeks. | ||||||
| Morris et al. [47] | Progressive prostate cancer | Zoledronate | Calcitriol | Phase I | Examine the toxicity of pulse-dosed calcitriol with zoledronate and with the addition of dexamethasone at the time of disease progression | Calcitriol was administered for 3 consecutive days per week, starting at a dose of 4 µg per day. D |
|
| Doses were escalated to 30 µg per day. | |||||||
| Intravenous zoledronate (4 mg) was administered monthly. At doses above 6 µg/day. | |||||||
| Dexamethasone | 31/0 | examethasone could be added to the regimen at disease progression. | |||||
| Tiffany et al. [28] | AIPC | Docetaxel | Calcitriol | Phase I/II | Determine the safety and preliminary efficacy of the combination of high dose pulse calcitriol with a standard regimen of docetaxel plus estramustine. | Day 1: 60 µg calcitriol orally, and 8 mg dexamethasone bid for 1st 3 daysCycle repeated every 21 days for up to 12 cycles. |
|
| Day 2: 60 mg/m2 docetaxel on day 2 (70 mg/m2 after cycle 1) | |||||||
| Day 1–5: 280 mg estramustine orally 3 times daily | |||||||
| Cycle repeated every 21 days for up to 12 cycles. | |||||||
| Estramustine | 24/0 | Patients also received 325 mg aspirin and 1 or 2 mg warfarin orally daily. | |||||
| Trump et al. [48] | AIPC | Dexamethasone | Calcitriol | Phase II | Evaluate high-dose calcitriol at a dose of 12 µg daily given X 3 plus dexamethasone weekly. | Oral calcitriol was administered weekly, Monday, Tuesday, and Wednesday (MTW), at a dose of 8 µg, for 1 month, |
|
| at a dose of 10 µg every MTW for 1 month, | |||||||
| and at a dose of 12 µg every MTW thereafter. | |||||||
| 43/0 | Dexamethasone at a dose of 4 mg was administered each Sunday, and MTW weekly. | ||||||
| Petrioli et al. [27] | HRPC | Docetaxel | Calcitriol | 26/0 | Evaluate the activity and tolerability of weekly high-dose calcitriol and docetaxel in patients with metastatic hormone-refractory prostate cancer (HRPC) previously exposed to Docetaxel. | Day 1: The treatment consisted of calcitriol (32 µg) given orally in three divided doses. |
|
| Day 2: iv Docetaxel (30 mg/m2) with dexamethasone 8 mg orally 12 h before, at the time of, and 12 h after docetaxel administration. | |||||||
| Administered on a schedule of six consecutive weekly administrations, followed by a 2-week rest interval. | |||||||
| Fakih et al. [23] | Advanced solid tumors | Gefitinib | Calcitriol | Phase I/PK/PD | Determine the maximum tolerated dose (MTD) of this combination | Calcitriol was given i.v. over 1 h on weeks 1, 3, and weekly thereafter. |
|
| Gefitinib was given at a fixed oral daily dose of 250 mg starting at week 2 (day 8). | |||||||
| 36/0 | Dose escalation: 10, 15, 20, 26, 24, 44, 57, 74, and 96 µg/week | ||||||
| Beer et al. [49], | AIPC | Docetaxel | Calcitriol (DN-101) | RCT | The primary end point was prostate-specific antigen (PSA) response within 6 months of enrollment, defined as a 50% reduction confirmed at least 4 weeks later. | Weekly: docetaxel 36 mg/m2 intravenously for 3 weeks of a 4-week cycle combined with either 45 µg DN-101 or placebo taken orally 1 day before docetaxel. dexamethasone (4 mg orally 12 h before, 1 before, and 12 h after docetaxel administration). |
|
| This regimen was administered weekly for 3 consecutive weeks of a 4-week cycle. | |||||||
| 125/125 | Primary hormonal therapy with gonadotropin-releasing hormone agonists or antagonists was maintained during the study. | ||||||
| Chan et al. [50] | AIPC | Mitoxantrone | Calcitriol (DN-101) | Phase II | Evaluate the efficacy, safety, and impact on quality of life (QoL) of high dose calcitriol (DN-101) combined with mitoxantrone and glucocorticoids in androgen-independent prostate cancer (AIPC). | Day 1: 180 µg po of DN-101 |
|
| Day 2: iv 12 mg/m2 mitoxantrone | |||||||
| 19/0 | Every 21 days with daily 10 mg po prednisone | ||||||
| Beer et al. [51] | AIPC | Docetaxel | Calcitriol | RCT | Examine outcomes with intermittent chemotherapy in a large multi-institutional trial. | Day 1: calcitriol, oral dose of 45 µg or placebo |
|
| Dexamethasone | 45 of 250 patients participated in intermittent chemotherapy. Approximately 20% of patients treated with high dose calcitriol and 16% of placebo-treated patients received intermittent chemotherapy. | Day 2: docetaxel, iv. dose 36 mg/m2 with dexamethasone (4 mg orally given 12 h before, 1 before, and 12 h after docetaxel administration). | |||||
| Placebo | This regimen was administered weekly for 3 consecutive weeks of a 4-week cycle. | ||||||
| Muindi et al. [26] | Solid tumors | Dexamethasone | Calcitriol | Phase 1 & PK | MTD of weekly iv calcitriol with Gefitinib at 250 mg/day and dexamethasone 4 mg q12 h × 3. | Week1: 4 mg dexamethasone and Iv calcitriol |
|
| Week 2: 250 mg Gefitinib daily | |||||||
| Week3: 4 mg dexamethasone, iv calcitriol with 250 mg Gefitinib daily. | |||||||
| Gefitinib | 20/0 | Escalating doses of calcitriol:57, 74, 96, 125, 163 µg/week | |||||
| Blanke et al. [52] | Pancreatic | Docetaxel | Calcitriol | Phase II | Determine time-to-progression for patients given this combination | Day 1: 0.5 µg/kg calcitriol p.o. |
|
| Day 2: 36 mg/m2 DOX iv. + DEXA 4 mg orally given 12 h before, 1 h before, and 12 h after DOX administration). | |||||||
| Dexamethasone | 25/0 | Weekly for 3 weeks, then 1 week break. | |||||
| Srinivas and Feldman [21] | Prostate | Naproxen | Calcitriol (DN-101) | Single arm, open label Phase II | Determine whether the PSADT was prolonged. Secondary endpoints included: PSA response, defined as the first evidence of a total serum PSA decline of >50% from baseline maintained for at least 28 days and confirmed with two consecutive measurements taken two weeks apart; and duration of sustained response, defined as time from PSA decrease of >50% from baseline to the first evidence of disease progression. | Calcitriol (DN101): 45 µg once per week | The trial was halted after 21 patients were enrolled when a national trial comparing DN101 in combination with weekly docetaxel had a higher death rate in the DN101 arm compared to the new standard docetaxel dosing arm (every 3 weeks) and DN101 use was suspended pending further evaluation. |
| 21/0 | Naproxen: 375 mg twice a day |
| |||||
| Chadha et al. [53] | CRPC | Dexamethasone | Calcitriol | Phase II | Response rate of iv calcitriol plus dexamethasone in CRPC pts. | Weekly treatment cycle: | Study was terminated for due to lack of patient response |
| Day 1: 4 mg dexamethasone | |||||||
| 18/0 | Evaluate toxicity of high-dose iv calcitriol and dexamethasone in patients with CRPC | Day 2: 4 mg dexamethasone, the within 4–8 h later 74 µg calcitriol |
| ||||
| Scher et al. [54] | CRPC | Docetaxel | Calcitriol (DN-101) | Phase III/RCT | Compare survival times between weekly DOX+ DN-101 vs. every 3-week DOX + prednisone. | Control: 21-day dosing cycle with 5 mg oral prednisone bid, iv 75 mg/m2 on day 2, and 8 mg dexamethasone 12, 3 and 1 h prior to DOX infusion. | Study halted due to higher death rate in treated vs. control |
| Treated: 28-day dosing cycle of 45 µg oral DN-101 on days 1,8 and 15 | |||||||
| 476/477 | The comparative safety and tolerability was assessed by rates of AEs, grade 3, 4, and 5 AEs, SAEs and gastrointestinal events. | 36 mg/m2 DOX days 2, 9, 16 and 8 mg dexamethasone 12, 3 and 1 h prior to DOX infusion. |
|
| Study/Year | Participants | Drug(s) | DesignTreated/Control | Study Endpoint | Study Protocol | Outcome |
|---|---|---|---|---|---|---|
| Avioli et al. [29] | Healthy subjects | Prednisone | PK study | Demonstrate that the administration of prednisone leads to alternation in vitamin D metabolism and intestinal absorption of calcium. | Day 1–14: participants consumed a diet with 800 IU vitamin D. |
|
| Day 15: participants took 10 µCi of radiochemically pure 1,2-3H-vitamin D. blood samples were taken at 5,15,30 45 min and at 1,2,4,8,12,16 and 24 h. | ||||||
| Day 16–19: blood samples obtained every 12 h. | ||||||
| Day 20: participants received 30 mg/day of prednisone for 10 days. | ||||||
| 4 participants | Day 25: Day 15–19’s procedure was repeated. | |||||
| Odes et al. [33] | Patients with peptic ulcers | Cimetidine | Uncontrolled open label | Examine the effects if cimetidine on vitamin D hydroxylation in humans. | During spring months | Impact on vitamin D metabolites: |
| Dose: 400 mg cimetidine bid for 4 weeks | ||||||
| 9 participants | Labs: 25 hydroxyvitamin-D, 24,25-dihydroxyvitamin D, 1,25-dihydroxyvitamin D, calcium, phosphorus, potassium, urea, creatinine, uric acid, bilirubin, albumin, globulin, SGOT, SGPT and alkaline phosphatase were obtained at the baseline, at 4 weeks and 4 after the completion of treatment. |
| ||||
| Gao et al. [34] | Gynecological malignancies | Various chemotherapy regimens | Uncontrolled open label | Examine the serially changes in vitamin D metabolites before, during and after chemotherapy. | Each person had a different chemotherapy regimen. Combinations of the following drugs: |
|
| Cisplatin, adriamycin, cyclophosphamide, and/or mitomycin. One participant received radiation after chemotherapy was completed. | ||||||
| 4 participants | Labs: 25 hydroxyvitamin-D, 24,25-dihydroxyvitamin D, 1,25-dihydroxyvitamin D, PTH, calcium, phosphorus, potassium, blood urea nitrogen, creatinine, and urinary creatinine clearance at baseline, and 5 days each course of chemotherapy. | |||||
| Yavuz et al. [31] | Hyperlipidemic | Rosuvastatin | Prospective cohort | Investigate the possible effect of rosuvastatin on vitamin D metabolism | During winter months | There was a significant increase in |
| Dose: Rosuvastatin (10–20 mg doses) was used according to the baseline levels of cholesterol and triglycerides, and according to the index of cardiovascular risk. | ||||||
| 91 participants | Labs: Lipid parameters, 25 hydroxyvitamin-D, 1,25-dihydroxyvitamin D, renal and liver function tests, electrolytes, bone alkaline phosphatase (B-ALP) were obtained at the baseline and after 8 weeks of rosuvastatin treatment. |
| ||||
| Fakih et al. [35] | Colorectal cancer | Various chemotherapy regimens | Retrospective study315 patients | Investigate the vitamin D status in 315 patients with colorectal cancer treated in a single institute. | The first 25-OH vitamin D assay was used as the baseline in patients with multiple 25-OH vitamin D testing. Chemotherapy status was documented in all patients. Colorectal cancer patients were divided into two categories: “no chemotherapy group:” all patients who did not receive any chemotherapy or whose last chemotherapy treatment was at least 3 months prior to 25-OH vitamin D assay. |
|
| 43% of patients: irinotecan-based, 39% of patients: | “Chemotherapy group:” all patients whose baseline 25-OH vitamin D level was obtained during chemotherapy treatment or within 3 months after last dose of chemotherapy. | |||||
| oxaliplatin based, 18% of patients: fluoropyrimidine | ||||||
| Ertugrul et al. [32] | Hyperlipidemic | Rosuvastatin | prospective, randomized design | Compare the influences of rosuvastatin and fluvastatin on the levels of 25-hydroxyvitamin D. | During winter months |
|
| Dose: rosuvastatin 10 mg (Crestor) or fluvastatin 80 mg XL (Lescol XL) for 8 weeks. | ||||||
| Fluvastatin | 134 participants were randomized, 1:1 | Labs: Lipid parameters, 25 hydroxyvitamin-D, 1,25-dihydroxyvitamin D, renal and liver function tests, electrolytes, bone alkaline phosphatase (B-ALP) were obtained at the baseline and after 8 weeks of treatment. |
| No. of patients | Dose (µg/day) | Dose (µg/week) | T1/2 (h) | Cmax (ng/mL) | AUC0–24 h(ng h/mL) | CL/F (mL/min) |
|---|---|---|---|---|---|---|
| 3 | 4 | 28 | 21 (15–29) | 0.21 (0.16–0.29) | 2.4 (2.3–3.6) | 23 (32–50) |
| 3 | 6 | 42 | 21 (8.7–34) | 0.25 (0.23–0.37) | 2.4 (2.1–4.0) | 50 (29–70) |
| 2 | 8 | 56 | 18 (17–19) | 0.27 (0.14–0.41) | 3.2 (2.4–4.0) | 54 (42–65) |
| 2 | 11 | 77 | 20 (16–24) | 0.59 (0.57–0.61) | 7.0 (6.9–7.0) | 30 (30–31) |
| 3 | 13 | 91 | 13 (5.3–27) | 0.37 (0.3–0.9) | 3.7 (3.2–6.5) | 64 (37–80) |
| 2 | 17 | 119 | 34 (2.5–42) | 0.55 (0.39–0.71) | 5.9 (4.5–7.4) | 57 (41–72) |
| 3 | 22 | 154 | 23 (15–36) | 0.46 (0.42–0.54) | 5.5 (5.1–6.3) | 75 (62–109) |
| 2 | 29 | 203 | 25 (25–26) | 0.71 (0.66–0.76) | 8.0 (7.7–8.2) | 66 (65–67) |
| 6 | 38 | 266 | 25 (15–31) | 1.10 (0.32–1.4) | 8.1 (5.8–11.0) | 91 (62–123) |
| Study | No. of patients | Cancer type | Dose (µg/week) | T1/2 (h) | Cmax (ng/mL) | AUC0–24 h(ng h/mL) | AUC0–72 h (ng h/mL) | Other Drugs |
|---|---|---|---|---|---|---|---|---|
| Fakih et al. [23] | 3 | Solid | 10 | 13.5 ± 2.9 | 0.46 ± 0.21 | 4.59 ± 0.91 | ||
| Fakih et al. [23] | 3 | Solid | 15 | 12.3 ± 0.9 | 0.77 ± 0.37 | 5.92 ± 1.00 | ||
| Fakih et al. [23] | 3 | Solid | 20 | 12.5 ± 1.9 | 1.01 ± 0.22 | 8.32 ± 1.04 | ||
| Fakih et al. [23] | 3 | Solid | 26 | 11.6 ± 1.4 | 1.45 ± 0.47 | 12.43 ± 3.64 | ||
| Fakih et al. [23] | 3 | Solid | 34 | 13.3 | 1.44 ± 0.84 | 9.89 ± 3.05 | ||
| Fakih et al. [23] | 3 | Solid | 44 | 19.0 ± 1.5 | 2.72 ± 1.39 | 17.87 ± 10.72 | ||
| Muindi et al. [26] | 3 | Prostate | 57 | 16.3 ± 2.0 | 4.16 ± 1.78 | 26.90 ± 5.00 | dexamethosone | |
| Fakih et al. [23] | 3 | Solid | 57 | 20.9 ± 3.6 | 3.80 ± 2.38 | 24.15 ± 8.62 | ||
| Muindi et al. [26] | 4 | Prostate | 74 | 18.6 ± 3.9 | 4.74 ± 1.13 | 30.94 ± 6.61 | dexamethosone | |
| Fakih et al. [23] | 3 | Solid | 74 | 16.1 ± 4.3 | 6.68 ± 1.42 | 35.65 ± 8.01 | ||
| Muindi et al. [26] | 3 | Prostate | 96 | 8.7 ± 2.3 | 10.12 ± 2.17 | 54.41 ± 15.50 | dexamethosone | |
| Fakih et al. [23] | 3 | Solid | 96 | 18.2 ± 1.9 | 4.23 ± 1.12 | 25.85 ± 4.41 | ||
| Muindi et al. [26] | 6 | Prostate | 125 | 14.6 ± 0.6 | 11.17 ± 2.62 | 53.50 ± 10.49 | dexamethosone | |
| Muindi et al. [26] | 4 | Prostate | 163 | 11.1 ± 1.7 | 12.56 ± 1.31 | 72.22 ± 6.92 | dexamethosone |
References
- Wang, L.; Song, Y.; Manson, J.E.; Pilz, S.; Marz, W.; Michaelsson, K.; Lundqvist, A.; Jassal, S.K.; Barrett-Connor, E.; Zhang, C.; et al. Circulating 25-hydroxy-vitamin d and risk of cardiovascular disease: A meta-analysis of prospective studies. Circ. Cardiovasc. Qual. Outcomes 2012, 5, 819–829. [Google Scholar] [CrossRef]
- Vanoirbeek, E.; Krishnan, A.; Eelen, G.; Verlinden, L.; Bouillon, R.; Feldman, D.; Verstuyf, A. The anti-cancer and anti-inflammatory actions of 1,25(OH)2D3. Baillieres Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 593–604. [Google Scholar] [CrossRef]
- Lang, P.O.; Samaras, N.; Samaras, D.; Aspinall, R. How important is vitamin D in preventing infections? Osteoporos. Int. 2012. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Schottker, B.; Ball, D.; Gellert, C.; Brenner, H. Serum 25-hydroxyvitamin D levels and overall mortality. A systematic review and meta-analysis of prospective cohort studies. Ageing Res. Rev. 2012. [Google Scholar] [CrossRef]
- Plum, L.; DeLuca, H. The Functional metabolism and molecular biology of vitamin D action, 2nd ed; Humana Press: New York, NY, USA, 2010; pp. 61–97, 1155. [Google Scholar]
- Berry, D.; Hypponen, E. Determinants of vitamin D status: Focus on genetic variations. Curr. Opin. Nephrol. Hypertens. 2011, 20, 331–336. [Google Scholar] [CrossRef]
- DeLuca, H.F. Evolution of our understanding of vitamin D. Nutr. Rev. 2008, 66, S73–S87. [Google Scholar] [CrossRef]
- Vieth, R. The mechanisms of vitamin D toxicity. Bone Miner. 1990, 11, 267–272. [Google Scholar] [CrossRef]
- Jones, G. Pharmacokinetics of vitamin D toxicity. Am. J. Clin. Nutr. 2008, 88, 582S–586S. [Google Scholar]
- Bolland, M.J.; Grey, A.; Avenell, A.; Gamble, G.D.; Reid, I.R. Calcium supplements with or without vitamin D and risk of cardiovascular events: Reanalysis of the Women's Health Initiative limited access dataset and meta-analysis. BMJ 2011, 342, d2040. [Google Scholar]
- Lappe, J.M.; Travers-Gustafson, D.; Davies, K.M.; Recker, R.R.; Heaney, R.P. Vitamin D and calcium supplementation reduces cancer risk: Results of a randomized trial. Am. J. Clin. Nutr. 2007, 85, 1586–1591. [Google Scholar]
- Marshall, D.T.; Savage, S.J.; Garrett-Mayer, E.; Keane, T.E.; Hollis, B.W.; Horst, R.L.; Ambrose, L.H.; Kindy, M.S.; Gattoni-Celli, S. Vitamin D3 supplementation at 4,000 international units per day for one year results in a decrease of positive cores at repeat biopsy in subjects with low-risk prostate cancer under active surveillance. J. Clin. Endocrinol. Metab. 2012, 97, 2315–2324. [Google Scholar] [CrossRef]
- Tretli, S.; Schwartz, G.G.; Torjesen, P.A.; Robsahm, T.E. Serum levels of 25-hydroxyvitamin D and survival in Norwegian patients with cancer of breast, colon, lung, and lymphoma: A population-based study. Cancer Causes Control 2012, 23, 363–370. [Google Scholar] [CrossRef]
- Yin, L.; Grandi, N.; Raum, E.; Haug, U.; Arndt, V.; Brenner, H. Meta-analysis: Circulating vitamin D and ovarian cancer risk. Gynecol. Oncol. 2011, 121, 369–375. [Google Scholar] [CrossRef]
- Yin, L.; Grandi, N.; Raum, E.; Haug, U.; Arndt, V.; Brenner, H. Meta-analysis: Serum vitamin D and breast cancer risk. Eur. J. Cancer 2010, 46, 2196–2205. [Google Scholar] [CrossRef]
- Yin, L.; Grandi, N.; Raum, E.; Haug, U.; Arndt, V.; Brenner, H. Meta-analysis: Longitudinal studies of serum vitamin D and colorectal cancer risk. Aliment. Pharmacol. Ther. 2009, 30, 113–125. [Google Scholar] [CrossRef]
- Trump, D.L.; Deeb, K.K.; Johnson, C.S. Vitamin D: Considerations in the continued development as an agent for cancer prevention and therapy. Cancer J. 2010, 16, 1–9. [Google Scholar] [CrossRef]
- Boulard, J.C.; Hanslik, T.; Alterescu, R.; Baglin, A. Symptomatic hypercalcemia after vitamin D-thiazide diuretics combination. Two cases in elderly women. Presse Med. 1994, 23, 96. [Google Scholar]
- Cohen, H.J.; Silberman, H.R.; Tornyos, K.; Bartolucci, A.A. Comparison of two long-term chemotherapy regimens, with or without agents to modify skeletal repair, in multiple myeloma. Blood 1984, 63, 639–648. [Google Scholar]
- Srinivas, S.; Feldman, D. A phase II trial of calcitriol and naproxen in recurrent prostate cancer. Anticancer Res. 2009, 29, 3605–3610. [Google Scholar]
- Beer, T.M.; Eilers, K.M.; Garzotto, M.; Egorin, M.J.; Lowe, B.A.; Henner, W.D. Weekly high-dose calcitriol and docetaxel in metastatic androgen-independent prostate cancer. J. Clin. Oncol. 2003, 21, 123–128. [Google Scholar] [CrossRef]
- Fakih, M.G.; Trump, D.L.; Muindi, J.R.; Black, J.D.; Bernardi, R.J.; Creaven, P.J.; Schwartz, J.; Brattain, M.G.; Hutson, A.; French, R.; et al. A phase I pharmacokinetic and pharmacodynamic study of intravenous calcitriol in combination with oral gefitinib in patients with advanced solid tumors. Clin. Cancer Res. 2007, 13, 1216–1223. [Google Scholar] [CrossRef]
- Trump, D.L.; Hershberger, P.A.; Bernardi, R.J.; Ahmed, S.; Muindi, J.; Fakih, M.; Yu, W.-D.; Johnson, C.S. Anti-tumor activity of calcitriol: Pre-clinical and clinical studies. J. Steroid. Biochem. Mol. Biol. 2004, 89–90, 519–526. [Google Scholar]
- Muindi, J.R.; Modzelewski, R.A.; Peng, Y.; Trump, D.L.; Johnson, C.S. Pharmacokinetics of 1α,25-Dihydroxyvitamin D3 in Normal Mice after Systemic Exposure to Effective and Safe Antitumor Doses. Oncology 2004, 66, 62–66. [Google Scholar] [CrossRef]
- Muindi, J.R.; Johnson, C.S.; Trump, D.L.; Christy, R.; Engler, K.L.; Fakih, M.G. A phase I and pharmacokinetics study of intravenous calcitriol in combination with oral dexamethasone and gefitinib in patients with advanced solid tumors. Cancer Chemother. Pharmacol 2009, 65, 33–40. [Google Scholar] [CrossRef]
- Petrioli, R.; Pascucci, A.; Francini, E.; Marsili, S.; Sciandivasci, A.; de Rubertis, G.; Barbanti, G.; Manganelli, A.; Salvestrini, F.; Francini, G. Weekly high-dose calcitriol and docetaxel in patients with metastatic hormone-refractory prostate cancer previously exposed to docetaxel. BJU Int. 2007, 100, 775–779. [Google Scholar] [CrossRef]
- Tiffany, N.M.; Ryan, C.W.; Garzotto, M.; Wersinger, E.M.; Beer, T.M. High dose pulse calcitriol, docetaxel and estramustine for androgen independent prostate cancer: A phase I/II study. J. Urol. 2005, 174, 888–892. [Google Scholar] [CrossRef]
- Avioli, L.V.; Birge, S.J.; Lee, S.W. Effects of prednisone on vitamin D metabolism in man. J. Clin. Endocrinol. Metab. 1968, 28, 1341–1346. [Google Scholar] [CrossRef]
- Muindi, J.R.; Peng, Y.; Potter, D.M.; Hershberger, P.A.; Tauch, J.S.; Capozzoli, M.J.; Egorin, M.J.; Johnson, C.S.; Trump, D.L. Pharmacokinetics of high-dose oral calcitriol: Results from a phase 1 trial of calcitriol and paclitaxel. Clin. Pharmacol. Ther. 2002, 72, 648–659. [Google Scholar]
- Yavuz, B.; Ertugrul, D.T.; Cil, H.; Ata, N.; Akin, K.O.; Yalcin, A.A.; Kucukazman, M.; Dal, K.; Hokkaomeroglu, M.S.; Yavuz, B.B.; et al. Increased levels of 25 hydroxyvitamin D and 1,25-dihydroxyvitamin D after rosuvastatin treatment: A novel pleiotropic effect of statins? Cardiovasc. Drugs Ther. 2009, 23, 295–299. [Google Scholar] [CrossRef]
- Ertugrul, D.T.; Yavuz, B.; Cil, H.; Ata, N.; Akin, K.O.; Kucukazman, M.; Yalcin, A.A.; Dal, K.; Yavuz, B.B.; Tutal, E. STATIN-D Study: Comparison of the Influences of Rosuvastatin and Fluvastatin Treatment on the Levels of 25 Hydroxyvitamin D. Cardiovasc. Ther. 2011, 29, 146–152. [Google Scholar] [CrossRef]
- Odes, H.S.; Fraser, G.M.; Krugliak, P.; Lamprecht, S.A.; Shany, S. Effect of cimetidine on hepatic vitamin D metabolism in humans. Digestion 1990, 46, 61–64. [Google Scholar] [CrossRef]
- Gao, Y.; Shimizu, M.; Yamada, S.; Ozaki, Y.; Aso, T. The effects of chemotherapy including cisplatin on vitamin D metabolism. Endocr. J. 1993, 40, 737–742. [Google Scholar]
- Fakih, M.G.; Trump, D.L.; Johnson, C.S.; Tian, L.; Muindi, J.; Sunga, A.Y. Chemotherapy is linked to severe vitamin D deficiency in patients with colorectal cancer. Int. J. Colorectal Dis. 2009, 24, 219–224. [Google Scholar]
- Jacot, W.; Pouderoux, S.; Thezenas, S.; Chapelle, A.; Bleuse, J.P.; Romieu, G.; Lamy, P.J. Increased prevalence of vitamin D insufficiency in patients with breast cancer after neoadjuvant chemotherapy. Breast Cancer Res. Treat. 2012, 2, 709–711. [Google Scholar]
- Petkovish, M.; Helvig, C.; Epps, T. CYP24A1 Regulation in health and disease. In Vitamin D, 3rd; Feldman, D., Pike, J.W.S., Adams, J., Eds.; Elsevier Science & Technology Books: San Diego, CA, USA, 2011; p. 2144. [Google Scholar]
- Farhan, H.; Wahala, K.; Cross, H.S. Genistein inhibits vitamin D hydroxylases CYP24 and CYP27B1 expression in prostate cells. J. Steroid. Biochem. Mol. Biol. 2003, 84, 423–429. [Google Scholar] [CrossRef]
- Wang, Z.; Lin, Y.S.; Zheng, X.E.; Senn, T.; Hashizume, T.; Scian, M.; Dickmann, L.J.; Nelson, S.D.; Baillie, T.A.; Hebert, M.F.; et al. An inducible cytochrome P450 3A4-dependent vitamin D catabolic pathway. Mol. Pharmacol. 2012, 81, 498–509. [Google Scholar]
- International Clinical Trials Search Portal: Registry Platform. Version 3.2. Available online: http://apps.who.int/trialsearch/ (accessed on 15 February 2013).
- metaRegister of Controlled Trials (mRCT). Available online: http://www.controlled-trials.com/mrct/ (accessed on 15 February 2013).
- Sampson, M.; McGowan, J.; Cogo, E.; Grimshaw, J.; Moher, D.; Lefebvre, C. An evidence-based practice guideline for the peer review of electronic search strategies. J. Clin. Epidemiol. 2009, 62, 944–952. [Google Scholar] [CrossRef]
- Hellstrom, E.; Robert, K.H.; Gahrton, G.; Mellstedt, H.; Lindemalm, C.; Einhorn, S.; Bjorkholm, M.; Grimfors, G.; Uden, A.M.; Samuelsson, J.; et al. Therapeutic effects of low-dose cytosine arabinoside, alpha-interferon, 1 alpha-hydroxyvitamin d3 and retinoic acid in acute leukemia and myelodysplastic syndromes. Eur. J. Haematol. 1988, 40, 449–459. [Google Scholar]
- Slapak, C.A.; Desforges, J.F.; Fogaren, T.; Miller, K.B. Treatment of acute myeloid leukemia in the elderly with low-dose cytarabine, hydroxyurea, and calcitriol. Am. J. Hematol. 1992, 41, 178–183. [Google Scholar] [CrossRef]
- Beer, T.M.; Eilers, K.M.; Garzotto, M.; Egorin, M.J.; Lowe, B.A.; Henner, W.D. Weekly high-dose calcitriol and docetaxel in metastatic androgen-independent prostate cancer. J. Clin. Oncol. 2003, 21, 123–128. [Google Scholar] [CrossRef]
- Beer, T.M.; Garzotto, M.; Katovic, N.M. High-dose calcitriol and carboplatin in metastatic androgen-independent prostate cancer. Am. J. Clin. Oncol. 2004, 27, 535–541. [Google Scholar] [CrossRef]
- Morris, M.J.; Smaletz, O.; Solit, D.; Kelly, W.K.; Slovin, S.; Flombaum, C.; Curley, T.; Delacruz, A.; Schwartz, L.; Fleisher, M.; et al. High-dose calcitriol, zoledronate, and dexamethasone for the treatment of progressive prostate carcinoma. Cancer 2004, 100, 1868–1875. [Google Scholar] [CrossRef]
- Trump, D.L.; Potter, D.M.; Muindi, J.; Brufsky, A.; Johnson, C.S. Phase II trial of high-dose, intermittent calcitriol (1,25 dihydroxyvitamin d3) and dexamethasone in androgen-independent prostate cancer. Cancer 2006, 106, 2136–2142. [Google Scholar] [CrossRef]
- Beer, T.M.; Ryan, C.W.; Venner, P.M.; Petrylak, D.P.; Chatta, G.S.; Ruether, J.D.; Redfern, C.H.; Fehrenbacher, L.; Saleh, M.N.; Waterhouse, D.M.; et al. Double-blinded randomized study of high-dose calcitriol plus docetaxel compared with placebo plus docetaxel in androgen-independent prostate cancer: A report from the ascent investigators. J. Clin. Oncol. 2007, 25, 669–674. [Google Scholar] [CrossRef]
- Chan, J.S.; Beer, T.M.; Quinn, D.I.; Pinski, J.K.; Garzotto, M.; Sokoloff, M.; Dehaze, D.R.; Ryan, C.W. A phase II study of high-dose calcitriol combined with mitoxantrone and prednisone for androgen-independent prostate cancer. BJU Int. 2008, 102, 1601–1606. [Google Scholar] [CrossRef]
- Beer, T.M.; Ryan, C.W.; Venner, P.M.; Petrylak, D.P.; Chatta, G.S.; Ruether, J.D.; Chi, K.N.; Young, J.; Henner, W.D. Intermittent chemotherapy in patients with metastatic androgen-independent prostate cancer: Results from ascent, a double-blinded, randomized comparison of high-dose calcitriol plus docetaxel with placebo plus docetaxel. Cancer 2008, 112, 326–330. [Google Scholar] [CrossRef]
- Blanke, C.D.; Beer, T.M.; Todd, K.; Mori, M.; Stone, M.; Lopez, C. Phase II study of calcitriol-enhanced docetaxel in patients with previously untreated metastatic or locally advanced pancreatic cancer. Invest. New Drugs 2009, 27, 374–378. [Google Scholar] [CrossRef]
- Chadha, M.K.; Tian, L.; Mashtare, T.; Payne, V.; Silliman, C.; Levine, E.; Wong, M.; Johnson, C.; Trump, D.L. Phase 2 trial of weekly intravenous 1,25 dihydroxy cholecalciferol (calcitriol) in combination with dexamethasone for castration-resistant prostate cancer. Cancer 2010, 116, 2132–2139. [Google Scholar]
- Scher, H.I.; Jia, X.; Chi, K.; de Wit, R.; Berry, W.R.; Albers, P.; Henick, B.; Waterhouse, D.; Ruether, D.J.; Rosen, P.J.; et al. Randomized, open-label phase iii trial of docetaxel plus high-dose calcitriol versus docetaxel plus prednisone for patients with castration-resistant prostate cancer. J. Clin. Oncol. 2011, 29, 2191–2198. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kennedy, D.A.; Cooley, K.; Skidmore, B.; Fritz, H.; Campbell, T.; Seely, D. Vitamin D: Pharmacokinetics and Safety When Used in Conjunction with the Pharmaceutical Drugs Used in Cancer Patients: A Systematic Review. Cancers 2013, 5, 255-280. https://doi.org/10.3390/cancers5010255
Kennedy DA, Cooley K, Skidmore B, Fritz H, Campbell T, Seely D. Vitamin D: Pharmacokinetics and Safety When Used in Conjunction with the Pharmaceutical Drugs Used in Cancer Patients: A Systematic Review. Cancers. 2013; 5(1):255-280. https://doi.org/10.3390/cancers5010255
Chicago/Turabian StyleKennedy, Deborah A., Kieran Cooley, Becky Skidmore, Heidi Fritz, Tara Campbell, and Dugald Seely. 2013. "Vitamin D: Pharmacokinetics and Safety When Used in Conjunction with the Pharmaceutical Drugs Used in Cancer Patients: A Systematic Review" Cancers 5, no. 1: 255-280. https://doi.org/10.3390/cancers5010255




