Cancer Stem Cells and Side Population Cells in Breast Cancer and Metastasis
Abstract
: In breast cancer it is never the primary tumour that is fatal; instead it is the development of metastatic disease which is the major cause of cancer related mortality. There is accumulating evidence that suggests that Cancer Stem Cells (CSC) may play a role in breast cancer development and progression. Breast cancer stem cell populations, including side population cells (SP), have been shown to be primitive stem cell-like populations, being long-lived, self-renewing and highly proliferative. SP cells are identified using dual wavelength flow cytometry combined with Hoechst 33342 dye efflux, this ability is due to expression of one or more members of the ABC transporter family. They have increased resistance to chemotherapeutic agents and apoptotic stimuli and have increased migratory potential above that of the bulk tumour cells making them strong candidates for the metastatic spread of breast cancer. Treatment of nearly all cancers usually involves one first-line agent known to be a substrate of an ABC transporter thereby increasing the risk of developing drug resistant tumours. At present there is no marker available to identify SP cells using immunohistochemistry on breast cancer patient samples. If SP cells do play a role in breast cancer progression/Metastatic Breast Cancer (MBC), combining chemotherapy with ABC inhibitors may be able to destroy both the cells making up the bulk tumour and the cancer stem cell population thus preventing the risk of drug resistant disease, recurrence or metastasis.1. Introduction
In breast cancer the primary tumour is never the cause of death; the development of metastatic disease is the major cause of cancer related mortality [1]. In the UK, breast cancer survival rates have improved greatly over the last few decades with a 5-year relative survival rate of 82% between 2001 and 2006 [2]. However this rate is reduced dramatically with the onset of metastatic disease, with a 5-year survival rate of only 12.98% recorded in 2002 for breast cancer patients with stage IV breast cancer [2].
Metastatic breast cancer (MBC) and its causes therefore remains a major concern with approximately 30% of patients with early stage breast cancer eventually progressing to metastatic disease [3]. Currently there is no standardised treatment for MBC. First-line treatment often involves the use of an anthracycline and/or taxane with or without endocrine treatment, and while results with these agents may be initially encouraging, frequently disease progression occurs within 6–10 months [3]. It is thought that the development of chemotherapy resistance can be attributed to treatment failure in over 90% of MBC patients [3]. If progression occurs or the patient is resistant to anthracycline/taxanes they can be offered capecitabine, gemcitabine, vinorelbine or albumin-bound paclitaxel with average response rates of 20–30% [3]. Unfortunately, these second-line agents offer little or no benefit to long term patient outcome [3].
Cancer develops through the progressive accumulation of genetic mutations that transforms normal cells into a malignant phenotype. Carcinogenesis results in cells acquiring more primitive cell characteristics, such as the ability to self-renew and an infinite capacity to divide [4,5], which can occur as a result of mutations affecting self-renewal, cell survival, proliferation and apoptosis [6,7]. There are two main hypotheses on the origin of cancer:
Stochastic theory;
Hierarchy (Cancer Stem Cell) theory.
The stochastic theory predicts that every cell within a tumour can generate a new tumour when in the correct microenvironment [6]. However, the probability of these cells entering the cell cycle is low [8]. This would suggest that tumours are relatively homogeneous in nature. Evidence to support this theory is that cancer cells within the same tumour tend to express a variation of markers representative of normal cellular differentiation [9]. Studies have also shown that mouse fibroblasts can be induced to form pluripotent stem cells [10] and mature rat hepatocytes can form small hepatocyte-like progenitor cells which can regenerate the liver after retrorsine injury [11]. However, primordial germ cells, which form teratocarcinomas were found to be no more differentiated than there malignant counterparts [4,8]. For example, if cancer is believed to be a result of differentiated cells gaining mutations that increase their life span, these cells will not express stem cell markers. Also, these cells are terminally differentiated, proliferate for a short period of time and are subject to a high rate of turnover, which means that they are unlikely to accumulate enough mutations to undergo neoplastic transformation [12].
The cancer stem cell (CSC) theory has been attributed to Cohnheim (1875) who postulated that stem cells misplaced during embryogenesis may be responsible for tumour formation later in life [13-15]. Histological analysis of embryonic and neoplastic tissue and comparison of their growth characteristics shows many similarities [16], for example they are both able to self-renew, differentiate, resist damaging agents, are long-lived, able to migrate and have extensive proliferative potential [17,18]. Studies on teratocarcinomas show that they are heterogeneous tissues containing a variety of undifferentiated and differentiated cell types which is consistent with tissues derived from stem cells. Pierce (1974) expanded on Cohnheim's theory [15] hypothesising that neoplasms develop from normal stem cells which do not escape developmental influences as suggested by Cohnheim but instead develop and function normally until carcinogenesis alters intercellular controls regulating their proliferation [19]. More recently the CSC model has been referred to as the hierarchy theory which predicts that only a small number of cells are capable of initiating a tumour but can do so at a high frequency. This suggests that tumours are heterogeneous in nature [5,6,17,20-22]. There are several reasons to believe that stem cells may be involved in carcinogenesis, for example there is evidence that most tumours develop over months or years which requires the transformational mutations to occur in cells with a long-life span. The longevity of stem cells means that they could accumulate enough mutations to undergo neoplastic transformation [13,18,22-24]. Self-renewal ability of stem cells also means that it would require fewer mutations to become neoplastic compared to more differentiated cell types [25,26].
Over the last few years there has been a surge in the number of studies investigating the role of CSC in tumour progression, however most research has focussed on the involvement in the primary tumour. CSC are tumour cells that are capable of self-renewal and give rise to heterogeneous tumour masses [13,27,28]. They are defined as ‘cells that have the ability to form tumours in immunodeficient mice that recapitulate the heterogeneity of the tumour from which they were isolated [27]’, and thus they share many properties with stem cells, being long-lived, self-renewing and highly proliferative. They have increased resistance to chemotherapeutic agents and apoptotic stimuli and have increased migratory potential above that of the bulk of tumour cells [29-34]. The origin of CSC remains unclear. It is unknown if CSC are dedifferentiated cells that have acquired a more stem cell-like phenotype or if they are stem cells which through longevity have accumulated the sufficient number of mutational hits required for carcinogenic transformation to occur or if they are due to a combination of both these factors. Evidence suggests that conventional chemotherapy targets the bulk of the tumour cells allowing slow cycling cells such as CSC to persist after treatment and promote further/metastatic disease [13].
2. Metastasis
In 1889, Paget devised the “seed and soil hypothesis” of cancer metastasis in which he suggested that the seeds of cancer (the tumour cells) have an ability to survive in specific soils (organs) that are conducive to their growth [35]. This hypothesis could easily refer to the properties of CSC. The metastatic process is complex, non-random and involves a long series of interrelated steps, any of which may be rate-limiting [36-39]. Metastatic steps include: progressive growth of the primary tumour which requires the establishment of a good blood supply through angiogenesis, in which growth factors regulate the breakdown of the extracellular matrix (ECM) and also cause the proliferation and migration of endothelial cells [37,40]. Cells capable of metastasis also acquire the ability to invade [37]. For epithelial cancers this involves cells undergoing an Epithelial-Mesenchymal Transition (EMT). EMT involves epithelial cells losing their epithelial characteristics and acquiring a more mesenchymal phenotype which occurs as a result of cytoskeletal changes within the cells. These changes allow the cell to acquire a more migratory phenotype [36,41], increasing the probability of tumour cells entering the blood and lymphatic systems. This process is influenced by chemokines and their receptors which are thought to play an important role in tumour development by influencing tumour transformation, survival, proliferation, invasion and metastasis and also regulation of angiogenesis and tumour-leukocyte interactions. Despite this, the majority of circulating tumour cells appear to be destroyed [37]. Those that persist may acquire the ability to metastasise and once inside the target organ may undergo Mesenchymal-Epithelial Transition (MET), proliferate and if the environment is conducive the disseminated cells may grow to establish a new tumour thus completing the metastatic process [38].
3. Role of Breast Cancer Stem Cells in Metastasis
There is accumulating evidence which suggests that CSC may play a role in metastasis [42]. Breast cancer stem cells can be identified using several methods [43]:
Flow cytometry—Analysis of breast cancer stem cell markers including CD44 [17], CD24 [47,48], CD49f [49-51] and aldehyde dehydrogenase 1 (ALDH1) [52]. Cells are then sorted and characterised further using clonogenicity, proliferation, differentiation and in vivo tumourigenicity assays;
Label retention studies—For example radioactive thymidine and BrdU [53-55];
Quiescence—Pece and colleagues (2010) have isolated normal human mammary stem cells using the lipophilic dye, PKH26 which is retained by quiescent cells [56];
Functional assays—Side Population (SP) cells which have an increased ability to efflux Hoechst 33342 dye [57] and ALDH-positive cells that are identified using the Aldefluor assay which identifies cells with high aldehyde dehydrogenase activity [52,58];
The use of cell surface markers to identify CSC from solid tumours requires careful optimisation of antibodies and the gated populations must be validated using both in vitro and in vivo functional assays. Discrepancies in marker expression may arise due to the manipulations required to prepare the samples for analysis, for example tumour dissociation into single cell suspensions and culturing of cells may alter cell behaviour and viability. Unlike normal tissue stem cells, CSC are influenced by the specific genetic aberrations of the tumour, by the stage of the disease and also by any therapeutic interventions given to the patient [59]. In patient tumours, CSCs are ‘moving targets’ (cells that are continually evolving) making it difficult to isolate these cells in the clinic. The clinician needs to be able to identify the different CSC populations in the patient throughout all stages of disease progression and needs to be able to target this population without detrimental effects on the normal stem cell populations [27,59].
At present most of what we know about the role of CSC in breast cancer metastasis has been based on ALDH-positive, CD133-positive or CD44+CD24−/low breast CSC populations [58,60-62]. Breast cancer is a heterogeneous disease which can be classified into different subtypes based on histology or molecular profiling [63,64]. It is becoming apparent that different breast cancer subtypes may result from different breast cancer cell populations. ALDH has been used to identify hematopoietic stem cells (HSC) in both mice and humans [52,65-67], normal mammary stem cells, and also CSC populations in AML [68], multiple myeloma [67,69], and malignant human mammary epithelial cell lines [52]. In xenograft human breast tumours, these cell types displayed cancer stem cell properties giving rise to tumours in vivo that recapitulated the heterogeneity of the tumour from which they were isolated [52]. ALDH+ cells have been shown to mediate invasion and metastasis in inflammatory breast cancer [70] and this population is increased in basal breast cancer cell lines and in breast tumours from patients following neoadjuvant chemotherapy [71]. In fact, ALDH1 has been found to be an independent predictive factor for early metastasis and decreased survival in patients [70]. Marcato and co-workers assessed the expression of 19 ALDH isoforms and demonstrated that CSC in breast tumours could be identified using the ALDH1A3 isoform and it was shown that the expression of ALDH1A3 correlated with breast tumour grade and stage and correlated prevalence of CSC with metastatic breast cancer [72].
Independently, both CD44+ [73,74] and CD24+ [47,75] breast cancer cell populations have been shown to be involved in the metastatic process. Data on the role of CD44+ or CD24− cells in metastatic tissues is conflicting, Shipitsin and co-workers determined that the CD24+ cell population was increased in distant metastases regardless of the site of metastases [74]. However, no correlation was observed when comparing CD44+ cells in primary invasive tumours and matched samples taken from metastatic sites [74]. CD44 is subject to extensive alternative splicing and different splice variants of CD44 have been shown to be involved in metastasis in a variety of different cancers and have been associated with poor patient outcome [76]. For example CD44v3, CD44v6 and CD44v7/8 variants have been shown to correlate with more aggressive breast cancer subtypes [76]. Brown and colleagues have demonstrated that it is essential for a switch in CD44 expression from CD44 variants (CD44v) to CD44 standard isoform (CD44s) for Epithelial-Mesenchymal Transition (EMT) to occur in vivo [77]. In 2003, Al-Hajj and colleagues identified a population of cells in human breast cancers with a Lin−ESA+CD44+CD24−/low phenotype that were highly tumourigenic in vivo and were the only cell population capable of forming tumours in NOD/SCID mice that recapitulated the heterogeneity of the tumour from which they were isolated [17]. Sheridan and co-workers (2006) discovered CD44+CD24−/low populations constituted a high proportion of the total cell population in basal or myoepithelial breast cancer cell lines [47]. These cell lines expressed high levels of the proinvasive genes Il-1α, Il-6 and Il-8 and were more invasive than cell lines containing less than 30% CD44+CD24−/low cells [47]. Subsequently, studies have shown that CD44+CD24−/low cells are slow cycling, chemoresistant, capable of forming mammospheres and are enriched in drug resistant cell lines [78,79].
Studies correlating the presence of CD44+CD24−/low cells in breast tumours with patient follow up have produced conflicting data. Abraham and colleagues determined that 78% of breast cancer cases assessed contained a CD44+CD24−/low population constituting less than 10% of the total cell population [80]. No significant correlation between the prevalence of CD44+CD24−/low expressing cells and response to different therapeutic regimes was observed [80]. Conversely, Li and co-workers showed that the proportion of cells with the CD44+CD24−/low phenotype was significantly increased following neoadjuvant chemotherapy [81]. The majority of studies investigating CD44+CD24−/low cells have utilised antibodies that do not discriminate between CD44 isoforms. In the future it would be beneficial to investigate expression of specific CD44 isoforms and their affect on the CD44+CD24−/low phenotype.
In breast cancer, CD133 appears to play less of a role as a stem cell marker than in other cancers [61]. However, CD133+ cells have been identified in breast cancer cell lines created from Brca1-deficient murine mammary tumours and these cells were highly tumourigenic, expressed common stem cell markers and were resistant to DNA-damaging chemotherapy [79]. A population of cells with a CD44+CD29hiCD133/2hi phenotype has also been identified in ERα-negative human breast tumours that was highly tumourigenic, capable of self-renewal and also expressed common stem cell markers [82].
4. Side Population Cells
Another putative CSC population are Side population (SP) cells. SP cells are identified using dual wavelength flow cytometry combined with Hoechst 33342 dye efflux [57,83]. SP cells have an elevated rate of Hoechst efflux which is attributed to the expression of one or more members of the ABC transporter family [34,84]. The SP phenotype is a functional ability within this cell population and is not reliant on the use of cell surface markers. Many groups are assessing the role of SP cells in different cancers however, results need to be analysed cautiously. Hoechst 33342 is a DNA binding dye which is toxic to cells at high concentrations and this is exacerbated by exposure to UV light. It is important to optimize the Hoechst 33342 dye concentration, the incubation time and cell counting in order to minimise cell toxicity [85,86]. Some studies have shown that Hoechst 33342 dye can impair cell differentiation [87], clonogenicity [88,89] and tumourigenicity [87,90-92]. Cell viability may also be affected by Hoechst 33342 dye [88] or by the cell sorting process [89,90] and therefore it is important to perform a functional assessment of cell viability to overcome this issue [86].
SP cells were first identified in murine bone marrow and were thought to be a stem cell (a cell type that has the ability to self-renew and differentiate to give rise to more restricted cell types [13]) population as they expressed the phenotypic markers of multipotent Haematopoietic Stem Cell (HSC; Lin−SCA-1+), were capable of contributing to both the lymphoid and myeloid cell lineages and were highly enriched in HSC activity [57]. SP cells have since been identified in a diverse array of normal tissues including, lung, heart, endometrium and mammary gland [93-96]. Several cancer cell lines have also been shown to contain an SP population including breast cancer, lung cancer, prostate cancer and ovarian cancer cell lines [20,97-99]. SP cells have also been identified in solid tumours including mesenchymal tumours, breast tumours, neuroblastomas and ascites extracted from ovarian cancer patients [20,91,99-101]. SP have been shown to possess stem cell-like properties within these tissues [20,91,93-100]. However in some studies, SP cells have not been shown to have stem-cell properties. CSC-like cells have been shown to play an important role in glioblastoma multiforme (GBM) [102]. Broadley and colleagues found that neurosphere formation enriched for stem cell-like activity [102]. GBM SP cells were not enriched in neurospheres, were unable to self-renew and had a lower tumourigenic potential than non-SP (NSP) cells [102]. SP cells in adrenocortical tumours did not have increased ability to proliferate or self-renew and were not more chemoresistant than the NSP population [103]. In 2006, Shackleton and co-workers demonstrated that a single cell with a Lin− CD29hiCD24+ phenotype could function as a mammary gland stem cell reconstituting an entire functional murine mammary gland [48]. SP cells were also identified in the murine mammary gland but they did not identify the Lin−CD29hiCD24+ population [48].
In haematopoietic cells, the SP phenotype has been defined by the expression of ABCG2 [34,84], an ABC half-transporter involved in xenobiotic protection [104] and commonly involved in multidrug resistance [105]. The importance of ABCG2 expression on the SP phenotype is demonstrated by studies on Abcg2 null mice which have been shown to have reduced numbers or fewer detectable SP cells than wild-type mice. Haematopoietic cells from Abcg2−/− null mice were shown to be more sensitive to mitoxantrone, a drug commonly used in chemotherapy [84]. Incorporating the green fluorescent protein (GFP) reporter gene into the Abcg2 locus in mice showed that 91% of haematopoietic SP cells were found to express the Abcg2/GFP allele supporting the role of ABCG2 in the SP phenotype [106].
5. Characterisation of SP in the Normal Mammary Gland
SP cells have been identified in both mouse and human mammary gland tissue [93,107]. Analysis of the murine mammary gland of Abcg2/Abcb1a/1b triple knockout mice showed almost complete loss of mammary gland SP cells, demonstrating in the murine mammary gland that both ABCG2 and ABCB1 contribute to the mammary gland SP phenotype [108]. It is interesting to note that higher percentages of SP cells have been identified in hyperplastic mammary glands from MMTV-Wnt-1 and MMTV-ΔN-catenin murine models compared to the wild-type control [109]. This suggests that the percentage of SP cells increases during breast cancer development and progression in some breast cancer subtypes.
At present there is a debate whether mammary gland SP cells are stem cells or a more-restricted mammary gland progenitor cell population. However, regardless of their status, studies have shown that human mammary gland SP cells are able to generate luminal and myoepithelial lineages [93], to form branching structures in matrigel and preferentially form spheres in vitro [110,111].
6. SP cells: A Presence in Numerous Cancers and Cancer Cell Lines
Patrawala and colleagues (2005) found that 30% of human cancer cells and xenograft tumours contain SP cells [98]. Extensive studies have attempted to characterise SP cells isolated from cancer cell lines and have identified shared properties between cancer SP cells. SP cells isolated from cancer cell lines have been found to be capable of self-renewal [99], can undergo asymmetric division [20,31,97-99,112,113] and express common stem cell markers including Notch1 and Bmi1 [20,97,98,114]. In the majority of studies, SP have also been found to be chemoresistant [20,97,99,112,115], radioresistant [114,116] and have increased invasive potential in vitro [117] compared to NSP cells [97,112,115]. In vivo studies have also demonstrated that in most cancer cell lines SP are more tumourigenic [97,99,113-115,118,119] and have greater metastatic potential [118,120,121] than their NSP counterparts [122].
7. SP in Breast Cancer Cell Lines
Extensive work has been performed to characterise SP cells in breast cancer cell lines [20,92,98,123-126]. Purified SP from the MCF7 breast carcinoma cell line had increased expression of the ‘stemness genes’ Notch1 and β-catenin [98], have a large nuclear-to-cytoplasmic ratio compared to NSP cells (a stem cell characteristic) and breast cancer cell line SP populations are capable of asymmetric division in vitro. In contrast, NSP cells could only generate NSP cells in vitro [92,113,123]. In vivo, breast cancer SP cells have been shown to be more tumourigenic than NSP cells when transplanted into immunodeficient mice [92,98,113,121].
The importance of ABC transporters in defining the SP phenotype in normal tissues has led to an abundance of studies investigating the role of these transporters in breast cancer SP cells. Many studies have reported increased transcriptional expression of ABCG2 in SP compared to NSP cells [92,98,121,123] in several breast cancer cell lines, and this has been confirmed at the protein level [113]. Targeting ABCG2 with ABCG2 inhibitors or siRNA has been shown to eliminate the SP population in the MCF7 cell line [113]. Studies have determined that MCF7 SP are more radioresistant [116] and chemoresistant to agents including mitoxantrone and carboplatin [92,113,121] than the NSP cell population. MCF7 SP cells have also been shown to have a CD44+CD24−CK18+EpCAM+CK19+ phenotype [121,124] and both MCF7 SP and CD44+CD24−/low cells are enriched in mammosphere formation and both populations are preferentially inhibited by NF-κβ pathway inhibitors compared to their non-stem cell counterparts [126]. These properties would suggest that SP cells may play an important role in breast cancer progression in some breast cancer subtypes. Perhaps the presence of an SP population may be indicative of a cohort of patients within a breast cancer subtype that have a poorer response to particular therapeutic options and thus these individuals may have a poorer prognosis. Conversely, in other breast cancer subtypes where SP are not prevalent another breast cancer stem cell population may be responsible for poor prognosis/chemotherapeutic resistance.
There is evidence to suggest that SP cells may undergo EMT and numerous studies have demonstrated an association between EMT, chemoresistance and stem cells [41,44,127,128]. Inducing EMT in the breast cancer cell line MCF7 by the addition of EGFR has been correlated with increased invasive potential and an increased resistance to tamoxifen [129]. Also, doxorubicin induced EMT in murine 4T1 breast cancer cells has been shown to lead to an enrichment of Sca-1-positive cells with an increased invasive potential [130]. SP cells have been shown to be more responsive to TGFβ signalling than NSP cells; with TGFβ treatment increasing the invasive potential and inducing EMT in the SP population in pancreatic cancer cell lines [31]. Upon removal of TGFβ treatment, pancreatic cancer SP cells were also capable of MET [31].
Cancer SP cells are known to be chemoresistant and this has been attributed to the expression of ABC transporters [20,99,131]. Unlike the study on pancreatic cancer SP cells [31], it has been shown that in the MCF7 breast cancer cell line, TGFβ induced EMT and depleted the SP population, down-regulated ABCG2 gene expression and increased cell sensitivity to mitoxantrone [113]. The removal of TGFβ restored the SP phenotype and ABCG2 expression [113]. Knockdown of e-cadherin reduced the SP population but did not affect expression of ABCG2 mRNA or protein thus providing evidence that the EMT response reflects post-translational regulation of ABCG2 function by e-cadherin as well as transcriptional repression of the ABCG2 gene [113]. These studies need to be repeated using other cancer cell lines and primary tumour SP cells in order to have a better understanding of the involvement of SP in EMT.
8. SP in Breast Tumours
SP cells have been identified in a diverse array of breast cancer cell lines that are classified within different breast cancer subtypes [123]. However, Clarke and co-workers (2005) have identified SP cells in normal human breast tissue and showed that these cells were ER-positive [111]. This has resulted in it being postulated that SP cells may be predominantly identified in luminal breast tumours [132]. Analysis of putative breast cancer stem cell markers in primary breast cancers demonstrated that different breast cancer stem cell populations were prevalent in different breast cancer subtypes. Basal-like tumours were found to contain a higher proportion of cells with a CD44+CD24−/low [47] or ALDH1+ [52] phenotype whilst Her2-positive cancers predominantly contained ALDH1+ cells. This study did not identify a prevalent breast cancer stem cell population in luminal breast tumours.
To date only Nakanishi and colleagues have identified SP cells in clinical breast tumours which were characterised as luminal cancers [100]. The percentage of SP cells was higher in luminal type (predominantly ER+PR+Her2+) breast cancer cell lines and these cell lines had increased colony forming ability in vitro compared to basal a (CK5+ and CK14+) or basal b (vimentin+) subtypes. A strong correlation was observed between Her2 expression and SP presence with the percentage of SP increasing in cell lines enforced with expression of Her2. Treatment with the Her2 signalling inhibitors, tyrophostin AG825 or trastuzumab, decreased the proportion of SP cells in breast cancer cell lines and also inhibited tumour growth in vivo [100]. The authors concluded that their data highlights the important role that Her2 plays in regulating SP cells and could account for the poor chemotherapeutic response and aggressive nature of Her2-positive breast cancers [100]. Unfortunately the media used in this study selected for primary breast cancer cells that had a luminal nature which means as yet it is unknown if SP cells can be identified in other breast cancer subtypes. This highlights the importance of identifying SP cells in all patient breast tumour subtypes and relating the presence of these cells to patient follow up in order to elucidate the role of SP cells in breast cancer and potentially to highlight new therapeutic targets. It may also be beneficial to use the identification of patient SP cells as a tool to determine which patients require a more aggressive treatment regime due to increased expression of MDR transporters that may be specifically tailored to eradicate this population of cells.
9. SP and Breast Cancer Metastasis
As yet there is no published literature investigating the metastatic potential of SP cells in breast cancer. However, studies in other cancers have shown that SP cells may play a role in metastatic spread of disease [95,120]. Kato and co-workers injected SP and NSP flow cytometrically sorted cells from the Hec-1, human endometrial cancer cell line, subcutaneously into immunodeficient mice and discovered that tumours derived from SP cells were highly invasive with cells invading through the basement membrane into the spine, peritoneum, leg and bone [95]. In contrast, NSP cell derived tumours were encapsulated and clearly separated from the basement membrane of the mouse skin [95]. SP isolated from the pancreatic cancer cell line PANC-1 have been shown to have an increased invasive potential in vitro and increased metastatic potential in vivo when compared to NSP cells using a murine liver metastasis model [31]. Nishii and colleagues (2009) have shown SP from gastric cancer cell lines have increased expression of several adhesion molecules which resulted in SP having a greater potential to form peritoneal metastases [120] and the prevalence of SP cells has been shown to significantly increase in hepatocellular carcinoma cell lines in a stepwise manner dependent on the cell lines metastatic potential, with the highest percentages observed in the most metastatic cell line [131]. In primary mesenchymal neoplasms, the percentage of SP cells has been shown to correlate with the tumours grade [91]. However to date the presence/percentage of SP cells in primary neoplasms has not been correlated to metastasis.
10. CXCR4, SP cells and Breast Cancer
The role of the CXCR4/CXCL12 axis in breast cancer metastasis has been well documented [133-137]. Several studies have shown how acquisition of the chemokine receptor CXCR4 by breast cancer cells can promote specific, chemotactic recruitment to remote sites such as the bone marrow, lymph nodes, liver and lung in response to localised expression of the specific chemokine ligand, CXCL12 [133]. These studies tie in with the distinct pattern of tumour dissemination to regional lymph nodes, lung, liver and bone marrow known to occur as part of the metastatic spread of breast cancer [133]. Stromal cells in all of these sites have been shown to highly express CXCL12 and were able to induce chemotaxis and invasion of breast cancer cells in vitro [133]. Several studies using gene silencing strategies to reduce CXCR4 expression have demonstrated that this results in impaired invasion of breast cancer cells in vitro and inhibits breast cancer metastasis in vivo [133-136]. Primary human breast cancer cells can also respond to CXCL12 [133] and in breast cancer patients, high levels of CXCR4 expression have been correlated with lymph node metastasis in invasive ductal carcinoma and associated with poor overall survival [137,138].
11. SP and CXCR4
SP cells are known to be capable of migration in response to CXCL12 [139,140]. Cardiac SP cells transplanted into non-infarct myocardium were shown to be capable of migrating into damaged myocardial tissue. This migration in vitro and in vivo being due to the interaction between CXCR4 and CXCL12 [140]. Human bone-marrow derived SP cells have also been shown to be CXCR4-positive and capable of migrating in response to CXCL12 in vitro [139]. Perhaps as in the case of pancreatic cancer (where the CD133+CXCR4+ cell population is responsible for metastasis [30]), it is the CXCR4-positive SP population of breast cancer cells that are responsible for breast cancer metastasis.
12. CSC in Primary and Metastatic Breast Disease
Current treatment for MBC is limited and has little clinical success [3]. There is a high rate of disease progression in MBC patients who initially respond to treatment with over 90% of MBC patients developing drug resistant disease [3]. If the SP phenotype in breast cancer is defined by the presence of ABC transporters this would make them an ideal candidate for contributing to drug resistant MBC. If metastases are seeded from SP cells this would result in an abundance of MDR-expressing cells making the tumours highly resistant to therapeutic intervention and therefore extremely difficult to eradicate [141].
To clarify the role of SP cells in MBC, the identification of SP cells needs to be performed in all breast cancer subtypes and their presence related to the tumour characteristics and to patient follow up. Breast cancer SP cells need to be isolated from patients and extensively characterised which will involve the need to transplant these human cells into animal cancer models to elucidate the potential of these cells to contribute to invasion and metastasis. A manuscript published by Michael Clarke's group described the development of a human-in-mouse breast cancer xenograft model in which the metastatic potential of breast cancer stem cells could be determined [142]. This group labelled breast cancer stem cells with Luc2-eGFP or Luc2-Tom enabling as few as 10 labelled cells to be visualised in vivo and ex vivo [142]. It will be necessary to obtain lymph node and primary tumour samples from patients undergoing surgery for breast cancer and to determine the presence of SP within these samples. If SP are indeed present in lymph nodes of breast cancer patients it could be indicative of patients likely to progress with distant metastatic disease. It is already known that the presence of metastases in axillary lymph nodes is associated with more aggressive breast cancers and poor disease-free and overall survival in breast cancer patients [143,144]. A disparate hormone receptor status between primary breast tumour tissue and nodal tissue has been observed demonstrating the importance of assessing breast cancer stem cells in both primary tumour and lymph node samples [145]. Lu et al. (2010) detected higher proportions of CD44-positive cells in primary tumour samples analysed from lymph node positive supraglottic carcinoma patients compared to lymph node negative patients thus showing the importance of identifying CSC in lymph node samples [146].
13. Treatment Strategies to Target Cancer Stem Cells, Including SP Cells
The emerging role of cancer stem cells in breast cancer development and progression has led to intense work to identify drugs that target the CSC population of drug resistant cells [60]. The importance of finding new therapies to target breast cancer stem cells is highlighted by accumulating evidence showing the resistance of cancer stem cells to chemotherapy and radiotherapy. Transcriptional profiling has been used to define several breast cancer subtypes that require different therapeutic regimes [147]. The heterogeneity of breast cancer stem cell populations in different breast cancer subtypes suggests that several targeted therapies may be necessary. It is hoped that increasing our understanding of the molecular biology of cancer stem cells may identify new therapeutic targets. For example, metaplastic breast cancers have been found to display a similar ‘tumourigenic signature’ to that of the CD44+CD24−/low breast cancer stem cell population and MBC have been shown to have aberrant MAPK components which potentially could benefit from treatment with MAPK pathway inhibitors [147].
The majority of studies have focused on targeting the CD44+ and CD44+CD24−/low cell populations, culminating most recently in the use of Salinomycin, a highly selective potassium ionophore, as a compound that could selectively eliminate the CD44+CD24−/low breast cancer stem cell population compared to treatment with paclitaxel alone [148]. In vivo, Salinomycin was able to inhibit the formation of tumours in the 4T1 murine breast cancer cell model and also in the ras-transformed HMLE model. Salinomycin treatment also prevented metastasis of 4T1 cells in vivo [148,149]. The mechanism of action of Salinomycin is the induction of a distinct apoptotic pathway that does not lead to cell cycle arrest and in this way results in massive apoptosis in cancer cells [117]. Fuchs and colleagues demonstrated that Salinomycin could even activate apoptosis in cells that were chemotherapy resistant or resistant to apoptosis due to the overexpression of factors such as bcl-2, MDR1 or 26S proteasomes with increased proteolytic activity [117]. Perhaps the effect of salinomycin on cells overexpressing MDR transporters would mean that it could be used to specifically target breast cancer SP cells.
As the SP cell phenotype is a consequence of the expression of ABC transporters, strategies to inhibit these might be worth revisiting. Nearly all cancer chemotherapy involves a first-line agent known to be a substrate of an ABC transporter [150], thereby increasing the risk of developing drug resistant tumours. If SP cells do play a role in breast cancer progression/MBC, combining chemotherapy with ABC inhibitors may be able to destroy both the cells making up the bulk tumour and the cancer stem cell population thus preventing the risk of drug resistant disease, recurrence or metastasis [104,141,150]. Results from clinical trials of ABC inhibitors have been disappointing with first line ABC transporter inhibitors for example verapamil being too toxic for use in cancer patients. Second line agents such as Zosuquidar were more potent and less toxic but demonstrated no additional clinical benefit in locally recurrent breast cancer or in MBC patients [151]. However, some of these trials have shown limited benefits, for example, clinical trials with PSC 833 showed that it was necessary to reduce the dose of the chemotherapy used compared to conventional chemotherapy, as combining chemotherapy with PSC 833 resulted in a pharmacokinetic interaction that altered the metabolism of the chemotherapeutic agent [150], thus achieving a statistically significant improvement in relapse-free and overall survival [152]. The trial combining PSC 833 with mitoxantrone, etoposide and cytosine arabinoside in AML patients had to terminate early due to toxicity issues [153]. However, analysis did show a statistically significant improvement in the complete remission rate of patients and a trend towards an improved disease-free survival within the treatment arm of the study [154]. However, the use of a broad-spectrum ABC transporter inhibitor will be unlikely as the 3 main transporters, ABCB1, ABCC1 and ABCG2, involved in multidrug resistance are from three evolutionary and structurally distinct ABC transporter families making it difficult to identify a common target [155].
In addition to chemoresistance, ABC transporters also play a role in cell migration, differentiation and protection against apoptosis [156-158]. In MCF7 breast cancer cells, siRNA knockdown of ABCB1 has been demonstrated to decrease migration and invasion in vitro [159]. ABCC1 has also been seen to be more highly expressed in metastatic lymph nodes compared to the primary tumour of patients with metastatic breast cancer [159]. Therefore gene silencing strategies alone or in combination with the use of chemotherapeutic reagents or anti-CSC drugs might prove more effective at targeting CSC in MBC. However, it will be important to determine if the breast cancer stem cell population can be targeted without detrimental effects on the normal stem cell populations.
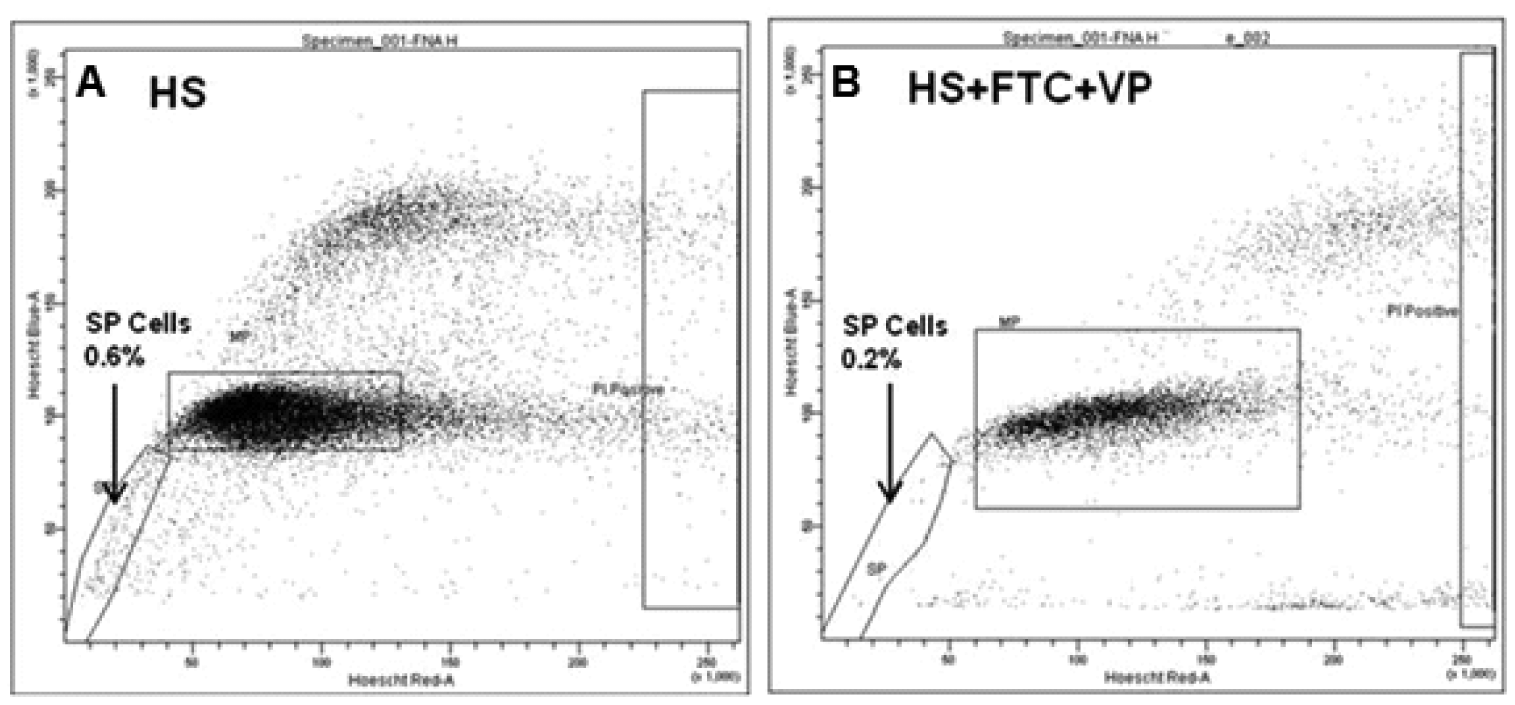
Nakanishi and co-workers (2010) identified SP in primary cells derived from patient breast tumours [100] and we confirm the presence of SP in patient breast tumours through detection of SP cells in Fine Needle Aspirate (FNA) samples from palpable breast tumours. Targeting SP cells may only be relevant to some breast cancer subtypes, such as luminal tumours [100], whilst other therapies may be required for other subtypes shown to be driven by different breast cancer stem cell populations. SP cells have been observed in both female [100] and male breast cancer patients (Meeson laboratory data, Figure 1). This highlights the presence of CSC in male breast cancer, and raises the question can we target CSC in both female and male breast cancer cases using the same strategies? No studies looking at the role of CSC in male breast cancer have as yet been reported.
The importance of Her2 in the regulation of breast cancer SP cells has been demonstrated by Nakanishi and colleagues (2010) and SP cells have been observed to be more prevalent in Her2 overexpressing breast cancer cell lines and luminal breast tumours [100]. The Her2 gene is amplified in approximately 20% of all breast cancer cases and is associated with a more aggressive disease and commonly involves early onset of metastases [29,160]. In fact, breast cancer patients with Her2-positive breast cancer cells in their bone marrow at the time of diagnosis have an increased risk of metastatic relapse and overall have a poorer prognosis [29].
Trastuzumab and Lapatinib are targeted therapies against the Her2 receptor and the Her2 pathway and are usually only prescribed to patients with Her2-positive breast tumours [161] and clinical studies have determined the benefit of giving patients neoadjuvant trastuzumab or lapatinib in both Her2-positive and Her2-negative patients [162,163]. Her2 overexpressing cell lines have increased mammosphere forming efficiency (MSFE) and an increased ALDH+ cell population [164]. Treatment of Her2 overexpressing cell lines with Trastuzumab reduced the number of ALDH-positive cells and this subsequently resulted in decreased invasive and tumourigenic potential of these cells [164,165]. Perhaps the benefit of Her2 inhibitors in both Her2-positive and Her2-negative tumours could be due to the interaction of these therapeutic agents with the CSC population or SP cells within these patient tumours.
14. Conclusion
Reported in vitro studies show promising results for therapeutically targeting breast CSC but they need to be translated into the clinic [117,148,149]. This may be difficult to achieve without standardised methods of detecting and characterising these cell populations both before and after therapy. The knowledge of the particular cancer stem cell properties involved in different breast cancer subtypes may identify new therapeutic targets especially for metastatic breast cancers in which treatment options are currently limited [3,147]. It is important that the role of breast cancer stem cells in metastasis is determined. If cancer stem cells could be targeted in breast cancer therapy the analysis of distant metastases for the presence of cancer stem cell populations may be beneficial in the selection of therapy.
If SP is involved in the metastatic process, therapies to target SP cells will need to be sought in order to minimise toxicity effects often experienced by treatment with systemic therapies. If ABC inhibitors/modulators are to be of use they need to be highly specific in order to reduce systemic effects as ABC transporters play a pivotal role in the blood brain barrier, many tissues and most normal stem cells [155], and interfering with this process could have catastrophic outcomes in patients. Treatments will have to be carefully tested not only on breast cancer SP cells but also on normal SP/stem cell populations to determine the impact they may have on normal body processes [166]. Therapeutic agents will also need to be tested in combination with commonly used breast cancer therapeutics as the effect of each agent alone may be very different to that observed when used in combination.
Acknowledgments
The authors of this manuscript wish to acknowledge the RVI Breast Cancer Appeal for funding this project.
References
- Klein, C.A. Parallel progression of primary tumours and metastases. Nat. Rev. Cancer 2009, 9, 302–312. [Google Scholar]
- CRUK Home page. http://info.cancerresearchuk.org/cancerstats/types/breast/survival/ (accessed17 February 2011).
- Coley, H.M. Mechanisms and strategies to overcome chemotherapy resistance in metastatic breast cancer. Cancer Treat. Rev. 2008, 34, 378–390. [Google Scholar]
- Sell, S.; Pierce, G. Maturation arrest of stem cell differentiation is a common pathway for the cellular origin of teratocarcinomas and epithelial cancers. Lab. Invest. 1994, 70, 6–22. [Google Scholar]
- Al-Hajj, M.; Clarke, M. Self-renewal and solid tumor stem cells. Oncogene 2004, 23, 7274–7282. [Google Scholar]
- Dick, J.E. Breast cancer stem cells revealed. Proc. Natl. Acad. Sci. USA 2003, 100, 3547–3549. [Google Scholar]
- Gudjonsson, T.; Magnusson, M. Stem cell biology and the cellular pathways of carcinogenesis. APMIS 2005, 113, 922–929. [Google Scholar]
- Polyak, K.; Hahn, W.C. Roots and stems: Stem cells in cancer. Nat. Med. 2006, 12, 296–300. [Google Scholar]
- Pierce, G. Teratocarcinoma: Model for a developmental concept of cancer. Curr. Top. Develop. Biol. 1967, 2, 223–246. [Google Scholar]
- Wernig, M.; Meissner, A.; Foreman, R.; Brambrink, T.; Ku, M.; Hochedlinger, K.; Bernstein, B.E.; Jaenisch, R. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature 2007, 448, 318–324. [Google Scholar]
- Avril, A.; Pichard, V.; Bralet, M.-P.; Ferry, N. Mature hepatocytes are the source of small hepatocyte-like progenitor cells in the retrorsine model of liver injury. J. Hepatol. 2004, 4, 737–743. [Google Scholar]
- Pierce, G.; Nakane, P.; Martinez-Herdandez, A.; Ward, J. Ultrastructural comparison of differentiation of stem cells of murine adenocarcinomas of colon and breast with their normal counterparts. J. Nat. Cancer Inst. 1977, 58, 1329–1345. [Google Scholar]
- Reya, T.; Morrison, S.; Clarke, M.; Weissman, I. Stem cells, cancer, and cancer stem cells. Nature 2001, 414, 105–111. [Google Scholar]
- Behbod, F.; Rosen, J. Will cancer stem cells provide new therapeutic targets? Carcinogenesis 2004, 26, 703–711. [Google Scholar]
- Sell, S. Stem cell origin of cancer and differentiation therapy. Crit. Rev. Oncol./Haematol 2004, 51, 1–28. [Google Scholar]
- Sell, S. Cellular origin of cancer: Dedifferentiation or stem cell maturation arrest? Environ. Health Perspect. 1993, 101, 15–26. [Google Scholar]
- Al-Hajj, M.; Wicha, M.; Benito-Hernandez, A.; Morrison, S.J.; Clarke, M.F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl. Acad. Sci. USA 2003, 100, 3983–3988. [Google Scholar]
- Dontu, G.; Al-Hajj, M.; Abdallah, W.; Clarke, M.; Wicha, M. Stem cells in normal breast development and breast cancer. Cell Proliferation 2003, 36, 59–72. [Google Scholar]
- Pierce, G. Neoplasms, differentiations and mutations. Am. J.Pathol 1974, 77, 103–118. [Google Scholar]
- Hirschmann-Jax, C.; Foster, A.; Wulf, G.G.; Nuchtern, J.G.; Jax, T.W.; Gobel, U.; Goodell, M.A. A distinct “side population” of cells with high drug efflux capacity in human tumor cells. Proc. Natl. Acad. Sci. USA 2004, 1010, 14228–14233. [Google Scholar]
- Hirschmann-Jax, C.; Foster, A.; Wulf, G.; Goodell, M.; Brenner, M. A distinct “side population” of cells in human tumor cells: implications for tumor biology and therapy. Cell Cycle 2005, 4, 203–205. [Google Scholar]
- Smalley, M.; Ashworth, A. Stem cells and breast cancer: A field in transit. Nat. Rev. Cancer 2003, 3, 832–844. [Google Scholar]
- Li, Y.; Rosen, J. Stem/progenitor cells in mouse mammary gland development and breast cancer. J. Mammary Gland Biol. Neoplasi. 2005, 10, 17–24. [Google Scholar]
- Waterworth, A. Introducing the concept of breast cancer stem cells. Breast Cancer Res. 2004, 6, 53–54. [Google Scholar]
- Knudson, A. Antioncogenes and human cancer. Proc. Natl. Acad. Sci. USA 1993, 90, 10914–10921. [Google Scholar]
- Benchaouir, R.; Rameau, P.; Decraene, C.; Dreyfus, P.; Israeli, D.; Piétu, G.; Danos, O.; Garcia, L. Evidence for a resident subset of cells with SP phenotype in the C2C12 myogenic line: A tool to explore muscle stem cell biology. Exp. Cell Res. 2004, 294, 254–268. [Google Scholar]
- Clarke, M.F.; Dick, J.E.; Dirks, P.B.; Eaves, C.J.; Jamieson, C.H.M.; Jones, D.L.; Visvader, J.; Weissman, I.L.; Wahl, G.M. Cancer stem cells—Perspectives on current status and future directions: AACR workshop on cancer stem cells. Cancer Res. 2006, 66, 9339–9344. [Google Scholar]
- Visvader, J.; Lindeman, G. Cancer stem cells in solid tumours: Accumulating evidence and unresolved questions. Nat. Rev. Cancer 2008, 8, 755–768. [Google Scholar]
- Freudenberg, J.A.; Wang, Q.; Katsumata, M.; Drebin, J.; Nagatomo, I.; Greene, M.I. The role of HER2 in early breast cancer metastasis and the origins of resistance to HER2-targeted therapies. Exp. Mol. Pathol. 2009, 87, 1–11. [Google Scholar]
- Hermann, P.C.; Huber, S.L.; Herrler, T.; Aicher, A.; Ellwart, J.W.; Guba, M.; Bruns, C.J.; Heeschen, C. Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell 2007, 1, 313–323. [Google Scholar]
- Kabashima, A.; Higuchi, H.; Takaishi, H.; Matsuzaki, Y.; Suzuki, S.; Izumiya, M.; Iizuka, H.; Sakai, G.; Hozawa, S.; Azuma, T.; et al. Side population of pancreatic cancer cells predominates in TGF-β-mediated epithelial to mesenchymal transition and invasion. Int. J. Cancer 2009, 124, 2771–2779. [Google Scholar]
- Peters, R.; Leyvraz, S.; Perey, L. Apoptotic regulation in primitive hematopoietic precursors. Blood 1998, 92, 2041–2052. [Google Scholar]
- Young, M.R.I.; Wright, M.A.; Lathers, D.M.R.; Messingham, K.A. Increased resistance to apoptosis by bone marrow CD34+ progenitor cells from tumor-bearing mice. Int. J. Cancer 1999, 82, 609–615. [Google Scholar]
- Zhou, S.; Schuetz, J.D.; Bunting, K.D.; Colapietro, A.; Sampath, J.; Morris, J.J.; Lagutina, I.; Grosveld, G.C.; Osawa, M.; Nakauchi, H.; et al. The ABC transporter Bcrp1/ABCG2 is expressed in a wide variety of stem cells and is a molecular determinant of the side-population phenotype. Nat. Med. 2001, 7, 1028–1034. [Google Scholar]
- Paget, S. The distribution of secondary growths in cancer of the breast. Lancet 1889, 133, 571–573. [Google Scholar]
- Brabletz, T.; Jung, A.; Spaderna, S.; Hlubek, F.; Kirchner, T. Migrating cancer stem cells— An integrated concept of malignant tumour progression. Nat. Rev. Cancer 2005, 5, 744–749. [Google Scholar]
- Couzin, J. Tracing the steps of metastasis, cancer's menacing ballet. Science 2003, 299, 1002–1006. [Google Scholar]
- Fidler, I.J. The pathogenesis of cancer metastasis: The ‘seed and soil’ hypothesis revisited. Nat. Rev. Cancer 2003, 3, 453. [Google Scholar]
- Geminder, H.; Sagi-Assif, O.; Goldberg, L.; Meshel, T.; Rechavi, G.; Witz, I.P.; Ben-Baruch, A. A possible role for CXCR4 and its ligand, the CXC chemokine stromal cell-derived factor-1, in the development of bone marrow metastases in neuroblastoma. J. Immunol. 2001, 167, 4747–4757. [Google Scholar]
- Ingber, D.E. Can cancer be reversed by engineering the tumor microenvironment? Semin. Cancer Biol. 2008, 18, 356–364. [Google Scholar]
- Hollier, B.; Evans, K.; Mani, S. The epithelial-to-mesenchymal transition and cancer stem cells: A coalition against cancer therapies. J. Mammary Gland Biol. Neoplasi. 2009, 14, 29–43. [Google Scholar]
- Boman, B.M.; Wicha, M.S. Cancer stem cells: A step toward the cure. J. Clin. Oncol. 2008, 26, 2795–2799. [Google Scholar]
- Al-Ejeh, F.; Smart, C.; Morrison, B.; Chenevix-Trench, G.; López, J.; Lakhani, S.; Brown, M.; Khanna, K. Breast cancer stem cells: Treatment resistance and therapeutic opportunities. Carcinogenesis 2011. [Google Scholar] [CrossRef]
- Mani, S.A.; Guo, W.; Mai-Jing, L.; Elinor, E.N.; Ayyanan, A.; Zhou, A.Y.; Brooks, M.; Reinhard, F.; Zhang, C.C.; Shipitsin, M.; et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 2008, 133, 704–715. [Google Scholar]
- Liao, M.; Zhang, C.; Zhou, B.; Zimonjic, D.; Mani, S.; Kaba, M.; Gifford, A.; Reinhardt, F.; Popescu, N.; Guo, W.; et al. Enrichment of a population of mammary gland cells that form mammospheres and have in vivo repopulating activity. Cancer Res. 2007, 67, 8131–9138. [Google Scholar]
- Liu, S.; Dontu, G.; Mantle, I.; Patel, S.; Ahn, N.; Jackson, K.; Suri, P.; Wicha, M. Hedgehog signaling and Bmi-1 regulate self-renewal of normal and malignant human mammary stem cells. Cancer Res. 2006, 66, 6063–6071. [Google Scholar]
- Sheridan, C. CD44+/CD24− breast cancer cells exhibit enhanced invasive properties: An early step necessary for metastasis. Breast Cancer Res. 2006, 8, R59. [Google Scholar]
- Shackleton, M.; Vaillant, F.; Simpson, K.J.; Stingl, J.; Smyth, G.K.; Asselin-Labat, M.-L.; Wu, L.; Lindeman, G.J.; Visvader, J.E. Generation of a functional mammary gland from a single stem cell. Nature 2006, 439, 84–88. [Google Scholar]
- Pommier, S.; Quan, G.; Christante, D.; Muller, P.; Newell, A.; Olson, S.; Diggs, B.; Muldoon, L.; Neuwelt, E.; Pommier, R. Characterizing the HER2/neu status and metastatic potential of breast cancer stem/progenitor cells. 2010, 17, 613–623. [Google Scholar]
- Stingl, J.; Eirew, P.; Ricketson, I.; Shackleton, M.; Vaillant, F.; Choi, D.; Li, H.I.; Eaves, C.J. Purification and unique properties of mammary epithelial stem cells. Nature 2006, 439, 993–997. [Google Scholar]
- Joshi, P.A.; Jackson, H.W.; Beristain, A.G.; Di Grappa, M.A.; Mote, P.A.; Clarke, C.L.; Stingl, J.; Waterhouse, P.D.; Khokha, R. Progesterone induces adult mammary stem cell expansion. Nature 2010, 465, 803–807. [Google Scholar]
- Ginestier, C.; Hur, M.H.; Charafe-Jauffret, E.; Monville, F.; Dutcher, J.; Brown, M.; Jacquemier, J.; Viens, P.; Kleer, C.G.; Liu, S.; et al. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell 2007, 1, 555–567. [Google Scholar]
- Chepko, G.; Smith, G.H. Three division-competent, structurally-distinct cell populations contribute to murine mammary epithelial renewal. Tissue Cell 1997, 29, 239–253. [Google Scholar]
- Smith, G.H. Label-retaining epithelial cells in mouse mammary gland divide asymmetrically and retain their template DNA strands. Development 2005, 132, 681–687. [Google Scholar]
- Welm, B.E.; Tepera, S.B.; Venezia, T.; Graubert, T.A.; Rosen, J.M.; Goodell, M.A. Sca-1pos cells in the mouse mammary gland represent an enriched progenitor cell population. Develop. Biol. 2002, 245, 42–56. [Google Scholar]
- Pece, S.; Tosoni, D.; Confalonieri, S.; Mazzarol, G.; Vecchi, M.; Ronzoni, S.; Bernard, L.; Viale, G.; Pelicci, P.G.; Di Fiore, P.P. Biological and molecular heterogeneity of breast cancers correlates with their cancer stem cell content. Cell 2010, 140, 62–73. [Google Scholar]
- Goodell, M.; Brose, K.; Paradis, G.; Conner, A.; Mulligan, R. Isolation and functional properties of murine hematopoietic stem cells that are replicating. in vivo. J. Exp. Med. 1996, 183, 1797–1806. [Google Scholar]
- Alison, M.R.; Guppy, N.J.; Lim, S.M.L.; Nicholson, L.J. Finding cancer stem cells: Are aldehyde dehydrogenases fit for purpose? J. Pathol. 2010, 222, 335–344. [Google Scholar]
- Rosen, J.M.; Jordan, C.T. The increasing complexity of the cancer stem cell paradigm. Science 2009, 324, 1670–1673. [Google Scholar]
- Alison, M.R.; Lim, S.M.L.; Nicholson, L.J. Cancer stem cells: Problems for therapy? J. Pathol. 2011, 223, 148–162. [Google Scholar]
- Mizrak, D.; Brittan, M.; Alison, M.R. CD133: Molecule of the moment. J. Pathol. 2008, 214, 3–9. [Google Scholar]
- Stingl, J. Detection and analysis of mammary gland stem cells. J. Pathol. 2009, 217, 229–241. [Google Scholar]
- Perou, C.M.; Sorlie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; Rees, C.A.; Pollack, J.R.; Ross, D.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular portraits of human breast tumours. Nature 2000, 406, 747–752. [Google Scholar]
- Sørlie, T.; Perou, C.; Tibshirani, R.; Aas, T.; Geisler, S.; Johnsen, H.; Hastie, T.; Eisen, M.; van de Rijn, M.; Jeffrey, S.; et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA 2001, 98, 10869–10874. [Google Scholar]
- Armstrong, L.; Stojkovic, M.; Dimmick, I.; Ahmad, S.; Stojkovic, P.; Hole, N.; Lako, M. Phenotypic characterization of murine primitive hematopoietic progenitor cells isolated on basis of aldehyde dehydrogenase activity. Stem Cells 2004, 22, 1142–1151. [Google Scholar]
- Hess, D.A; Meyerrose, T.E.; Wirthlin, L.; Craft, T.P.; Herrbrich, P.E.; Creer, M.H.; Nolta, J.A. Functional characterization of highly purified human hematopoietic repopulating cells isolated according to aldehyde dehydrogenase activity. Blood 2004, 104, 1648–1655. [Google Scholar]
- Matsui, W.; Huff, C.A.; Wang, Q.; Malehorn, M.T.; Barber, J.; Tanhehco, Y.; Smith, B.D.; Civin, C.I.; Jones, R.J. Characterization of clonogenic multiple myeloma cells. Blood 2004, 10, 2332–2336. [Google Scholar]
- Dick, J. Acute myeloid leukemia stem cells. Ann. NY Acad. Sci. 2005, 1044, 1–5. [Google Scholar]
- Pearce, D.J.; Taussig, D.; Simpson, C.; Allen, K.; Rohatiner, A.Z.; Lister, T.A.; Bonnet, D. Characterization of cells with a high aldehyde dehydrogenase activity from cord blood and acute myeloid leukemia samples. Stem Cells 2005, 23, 752–760. [Google Scholar]
- Charafe-Jauffret, E.; Ginestier, C.; Iovino, F.; Tarpin, C.; Diebel, M.; Esterni, B.; Houvenaeghel, G.; Extra, J.-M.; Bertucci, F.; Jacquemier, J.; et al. Aldehyde dehydrogenase 1–Positive cancer stem cells mediate metastasis and poor clinical outcome in inflammatory breast cancer. Clin. Cancer Res. 2010, 16, 45–55. [Google Scholar]
- Tanei, T.; Morimoto, K.; Shimazu, K.; Kim, S.J.; Tanji, Y.; Taguchi, T.; Tamaki, Y.; Noguchi, S. Association of breast cancer stem cells identified by aldehyde dehydrogenase 1 expression with resistance to sequential paclitaxel and epirubicin-based chemotherapy for breast cancers. Clin. Cancer Res. 2009, 15, 4234–4241. [Google Scholar]
- Marcato, P.; Dean, C.; Pan, D.; Araslanova, R.; Gillis, M.; Joshi, M.; Helyer, L.; Pan, L.; Leidal, A.; Gujar, S.; et al. Aldehyde dehydrogenase activity of breast cancer stem cells is primarily due to isoform ALDH1A3 and its expression is predictive of metastasis. Stem Cells 2011, 29, 32–45. [Google Scholar]
- Günthert, U.; Hofmann, M.; Rudy, W.; Reber, S.; Zöller, M.; Haumann, I.; Matzku, S.; Wenzel, A.; Ponta, H.; Herrlich, P. A new variant of glycoprotein CD44 confers metastatic potential to rat carcinoma cells. Cell 1991, 65, 13–24. [Google Scholar]
- Shipitsin, M.; Campbell, L.; Argani, P.; Weremowicz, S.; Bloushtain-Qimron, N.; Yao, J.; Nikolskaya, T.; Serebryiskaya, T.; Beroukhim, R.; Hu, M.; et al. Molecular definition of breast tumor heterogeneity. Cancer Cell 2007, 11, 259–273. [Google Scholar]
- Baumann, P.; Cremers, N.; Kroese, F.; Orend, G.; Chiquet-Ehrismann, R.; Uede, T.; Yagita, H.; Sleeman, J.P. CD24 expression causes the acquisition of multiple cellular properties associated with tumor growth and metastasis. Cancer Res. 2005, 65, 10783–10793. [Google Scholar]
- Orian-Rousseau, V. CD44, a therapeutic target for metastasising tumours. Eur. J. Cancer 2010, 46, 1271–1277. [Google Scholar]
- Brown, R.; Reinke, L.; Damerow, M.; Perez, D.; Chodosh, L.; Yang, J.; Cheng, C. CD44 splice isoform switching in human and mouse epithelium is essential for epithelial-mesenchymal transition and breast cancer progression. J. Clin. Invest. 2011, 121, 1064–1074. [Google Scholar]
- Fillmore, C.; Kuperwasser, C. Human breast cancer cell lines contain stem-like cells that self-renew, give rise to phenotypically diverse progeny and survive chemotherapy. Breast Cancer Res. 2008, 10, R25. [Google Scholar]
- Wright, M.; Calcagno, A.; Salcido, C.; Carlson, M.; Ambudkar, S.; Varticovski, L. Brca1 breast tumors contain distinct CD44+/CD24− and CD133+ cells with cancer stem cell characteristics. Breast Cancer Res. 2008, 10, R10. [Google Scholar]
- Abraham, B.K.; Fritz, P.; McClellan, M.; Hauptvogel, P.; Athelogou, M.; Brauch, H. Prevalence of CD44+/CD24−/low Cells in breast cancer may not be associated with clinical outcome but may favor distant metastasis. Clin Cancer Res. 2005, 11, 1154–1159. [Google Scholar]
- Li, X.; Lewis, M.T.; Huang, J.; Gutierrez, C.; Osborne, C.K.; Wu, M.-F.; Hilsenbeck, S.G.; Pavlick, A.; Zhang, X.; Chamness, G.C.; et al. Intrinsic Resistance of tumorigenic breast cancer cells to chemotherapy. J. Nat. Cancer Inst. 2008, 100, 672–679. [Google Scholar]
- Meyer, M.J.; Fleming, J.M.; Lin, A.F.; Hussnain, S.A.; Ginsburg, E.; Vonderhaar, B.K. CD44posCD49fhiCD133/2hi defines xenograft-initiating cells in estrogen receptor-negative breast cancer. Cancer Res. 2010, 70, 4624–4633. [Google Scholar]
- Ibrahim, S.F.; Diercks, A.H.; Petersen, T.W.; van den Engh, G. Kinetic analyses as a critical parameter in defining the side population (SP) phenotype. Exp. Cell Res. 2007, 313, 1921–1926. [Google Scholar]
- Zhou, S.; Morris, J.J.; Barnes, Y.; Lan, L.; Schuetz, J.D.; Sorrentino, B.P. Bcrp1 gene expression is required for normal numbers of side population stem cells in mice, and confers relative protection to mitoxantrone in hematopoietic cells in vivo. Proc. Natl. Acad. Sci. USA 2002, 99, 12339–12344. [Google Scholar]
- Montanaro, F.; Liadaki, K.; Schienda, J.; Flint, A.; Gussoni, E.; Kunkel, L.M. Demystifying SP cell purification: Viability, yield, and phenotype are defined by isolation parameters. Exp. Cell Res. 2004, 298, 144–154. [Google Scholar]
- Golebiewska, A.; Brons, N.H.C.; Bjerkvig, R.; Niclou, S.P. Critical appraisal of the side population assay in stem cell and cancer stem cell research. Cell Stem Cell 2011, 8, 136–147. [Google Scholar]
- Adamski, D.; Mayol, J.; Platet, N.; Berger, F.; Hérodin, F.; Wion, D. Effects of hoechst 33342 on C2C12 and PC12 cell differentiation. FEBS Lett. 2007, 581, 3076–3080. [Google Scholar]
- Platet, N.; Mayol, J.-F.; Berger, F.; Hérodin, F.; Wion, D. Fluctuation of the SP/non-SP phenotype in the C6 glioma cell line. FEBS Lett. 2007, 581, 1435–1440. [Google Scholar]
- Zhong, Y.; Zhou, C.; Ma, W.; Wang, D.; Guo, S.; Su, X.; Zhang, S. Most MCF7 and SK-OV3 cells were deprived of their stem nature by Hoechst 33342. Biochem. Biophys. Res. Commun. 2007, 364, 338–343. [Google Scholar]
- Camargo, F.; Chambers, S.; Drew, E.; McNagny, K.; Goodell, M. Hematopoietic stem cells do not engraft with absolute efficiencies. Blood 2006, 107, 501–507. [Google Scholar]
- Wu, C.; Wei, Q.; Utomo, V.; Nadesan, P.; Whetstone, H.; Kandel, R.; Wunder, J.S.; Alman, B.A. Side population cells isolated from mesenchymal neoplasms have tumor initiating potential. Cancer Res. 2007, 67, 8216–8222. [Google Scholar]
- Zhou, J.; Wulfkuhle, J.; Zhang, H.; Gu, P.; Yang, Y.; Deng, J.; Margolick, J.B.; Liotta, L.A.; Petricoin, E.; Zhang, Y. Activation of the PTEN/mTOR/STAT3 pathway in breast cancer stem-like cells is required for viability and maintenance. Proc. Natl. Acad. Sci. USA 2007, 104, 16158–16163. [Google Scholar]
- Clayton, H.; Titley, I.; Vivanco, M. Growth and differentiation of progenitor/stem cells derived from the human mammary gland. Exp. Cell Res. 2004, 297, 444–460. [Google Scholar]
- Hackett, T.-L.; Shaheen, F.; Johnson, A.; Wadsworth, S.; Pechkovsky, D.V.; Jacoby, D.B.; Kicic, A.; Stick, S.M.; Knight, D.A. Characterization of side population cells from human airway epithelium. Stem Cells 2008, 26, 2576–2585. [Google Scholar]
- Kato, K.; Yoshimoto, M.; Kato, K.; Adachi, S.; Yamayoshi, A.; Arima, T.; Asanoma, K.; Kyo, S.; Nakahata, T.; Wake, N. Characterization of side-population cells in human normal endometrium. Hum. Reprod. 2007, 22, 1214–1223. [Google Scholar]
- Martin, C.M.; Meeson, A.P.; Robertson, S.M.; Hawke, T.J.; Richardson, J.A.; Bates, S.; Goetsch, S.; Gallardo, T.D.; Garry, D.J. Persistent expression of the ATP-binding cassette transporter, Abcg2, identifies cardiac SP cells in the developing and adult heart. Develop. Biol. 2004, 265, 262–275. [Google Scholar]
- Ho, M.M.; Ng, A.V.; Lam, S.; Hung, J.Y. Side population in human lung cancer cell lines and tumors is enriched with stem-like cancer cells. Cancer Res. 2007, 67, 4827–4833. [Google Scholar]
- Patrawala, L.; Calhoun, T.; Schneider-Broussard, R.; Zhou, J.; Claypool, K.; Tang, D.G. Side population is enriched in tumorigenic, stem-like cancer cells, whereas ABCG2+ and ABCG2−cancer cells are similarly tumorigenic. Cancer Res. 2005, 65, 6207–6219. [Google Scholar]
- Szotek, P.P.; Pieretti-Vanmarcke, R.; Masiakos, P.T.; Dinulescu, D.M.; Connolly, D.; Foster, R.; Dombkowski, D.; Preffer, F.; MacLaughlin, D.T.; Donahoe, P.K. Ovarian cancer side population defines cells with stem cell-like characteristics and Mullerian Inhibiting Substance responsiveness. Proc. Natl. Acad. Sci. USA 2006, 103, 11154–11159. [Google Scholar]
- Nakanishi, T.; Chumsri, S.; Khakpour, N.; Brodie, A.H.; Leyland-Jones, B.; Hamburger, A.W.; Ross, D.D.; Burger, A.M. Side-population cells in luminal-type breast cancer have tumour-initiating cell properties, and are regulated by HER2 expression and signalling. Br. J. Cancer 2010, 102, 815–826. [Google Scholar]
- Donnenberg, V.S.; Landreneau, R.J.; Donnenberg, A.D. Tumorigenic stem and progenitor cells: Implications for the therapeutic index of anti-cancer agents. J. Control. Release 2007, 122, 385–391. [Google Scholar]
- Broadley, K.; Hunn, M.; Farrand, K.; Price; Grasso, C.; Miller, R.; Hermans, I.; McConnell, M. Side population is not necessary or sufficient for a cancer stem cell phenotype in glioblastoma multiforme. Stem Cells 2011, 29, 452–461. [Google Scholar]
- Lichtenauer, U.; Shapiro, I.; Geiger, K.; Quinkler, M.; Fassnacht, M.; Nitschke, R.; Rückauer, K.; Beuschlein, F. Side population does not define stem cell-like cancer cells in the adrenocortical carcinoma cell line NCI h295R. Endocrinology 2008, 149, 1314–1322. [Google Scholar]
- Polgar, O.; Robey, R.W.; Bates, S.E. ABCG2: Structure, function and role in drug response. Expert Opin. Drug Metab. Toxicol. 2008, 4, 1–15. [Google Scholar]
- Mao, Q. Role of the breast cancer resistance protein (ABCG2) in drug transport. AAPS J. 2005, 7, E118–E133. [Google Scholar]
- Tadjali, M.; Zhou, S.; Rehg, J.; Sorrentino, B.P. Prospective isolation of murine hematopoietic stem cells by expression of an Abcg2/GFP allele. Stem Cells 2006, 24, 1556–1563. [Google Scholar]
- Alvi, A.; Clayton, H.; Joshi, C.; Enver, T.; Ashworth, A.; Vivanco, Md.; Dale, T.; Smalley, M. Functional and molecular characterisation of mammary side population cells. Breast Cancer Res. 2003, 5, R1–R8. [Google Scholar]
- Jonker, J.W.; Freeman, J.; Bolscher, E.; Musters, S.; Alvi, A.J.; Titley, I.; Schinkel, A.H.; Dale, T.C. Contribution of the ABC transporters Bcrp1 and Mdr1a/1b to the side population phenotype in mammary gland and bone marrow of mice. Stem Cells 2005, 23, 1059–1065. [Google Scholar]
- Liu, B.Y.; McDermott, S.P.; Khwaja, S.S.; Alexander, C.M. The transforming activity of Wnt effectors correlates with their ability to induce the accumulation of mammary progenitor cells. Proc. Natl. Acad. Sci. USA 2004, 101, 4158–4163. [Google Scholar]
- Clarke, R.B. Isolation and characterization of human mammary stem cells. Cell Proliferation 2005, 38, 375–386. [Google Scholar]
- Clarke, R.B.; Spence, K.; Anderson, E.; Howell, A.; Okano, H.; Potten, C.S. A putative human breast stem cell population is enriched for steroid receptor-positive cells. Develop. Biol. 2005, 277, 443–456. [Google Scholar]
- Yang, M.; Zhang, R.; Yan, M.; Ye, Z.; Liang, W.; Luo, Z. Detection and characterization of side population in Ewing's sarcoma SK-ES-1 cells. in vitro. Biochem. Biophys. Res. Commun. 2010, 391, 1062–1066. [Google Scholar]
- Yin, L.; Castagnino, P.; Assoian, R.K. ABCG2 Expression and side population abundance regulated by a transforming growth factor {beta}-directed epithelial-mesenchymal transition. Cancer Res. 2008, 68, 800–807. [Google Scholar]
- Wang, J.; Guo, L.-P.; Chen, L.-Z.; Zeng, Y.-X.; Lu, S.H. Identification of cancer stem cell-like side population cells in human nasopharyngeal carcinoma cell line. Cancer Res. 2007, 67, 3716–3724. [Google Scholar]
- Song, J.; Chang, I.; Chen, Z.; Kang, M.; Wang, C.-Y. Characterization of side populations in hnscc: Highly invasive, chemoresistant and abnormal wnt signaling. PLoS One 2010, 5, e11456. [Google Scholar]
- Woodward, W.A.; Chen, M.S.; Behbod, F.; Alfaro, M.P.; Buchholz, T.A.; Rosen, J.M. WNT/beta-catenin mediates radiation resistance of mouse mammary progenitor cells. Proc. Natl. Acad. Sci. USA 2007, 104, 618–623. [Google Scholar]
- Fuchs, D.; Heinold, A.; Opelz, G.; Daniel, V.; Naujokat, C. Salinomycin induces apoptosis and overcomes apoptosis resistance in human cancer cells. Biochem. Biophys. Res. Commun. 2009, 390, 743–749. [Google Scholar]
- Chen, C.-Y.; Chiou, S.-H.; Huang, C.-Y.; Jan, C.-I.; Lin, S.-C.; Tsai, M.-L.; Lo, J.-F. Distinct population of highly malignant cells in a head and neck squamous cell carcinoma cell line established by xenograft model. J. Biomed. Sci. 2009, 16, 100. [Google Scholar]
- Moserle, L.; Indraccolo, S.; Ghisi, M.; Frasson, C.; Fortunato, E.; Canevari, S.; Miotti, S.; Tosello, V.; Zamarchi, R.; Corradin, A.; et al. The side population of ovarian cancer cells is a primary target of IFN-{alpha} antitumor effects. Cancer Res. 2008, 68, 5658–5668. [Google Scholar]
- Nishii, T.; Yashiro, M.; Shinto, O.; Sawada, T.; Ohira, M.; Hirakawa, K. Cancer stem cell-like SP cells have a high adhesion ability to the peritoneum in gastric carcinoma. Cancer Sci. 2009, 100, 1397–1402. [Google Scholar]
- Steiniger, S.C.J.; Coppinger, J.A.; Kruger, J.A.; Yates, J., III.; Janda, K.D. Quantitative mass spectrometry identifies drug targets in cancer stem cell containing side population. Stem Cells 2008, 26, 3037–3046. [Google Scholar]
- Mitsutake, N.; Iwao, A.; Nagai, K.; Namba, H.; Ohtsuru, A.; Saenko, V.; Yamashita, S. Characterization of side population in thyroid cancer cell lines: Cancer stem-like cells are enriched partly but not exclusively. Endocrinology 2007, 148, 1797–1803. [Google Scholar]
- Christgen, M.; Ballmaier, M.; Bruchhardt, H.; von Wasielewski, R.; Kreipe, H.; Lehmann, U. Identification of a distinct side population of cancer cells in the Cal-51 human breast carcinoma cell line. Mol. Cell. Biochem. 2007, 306, 201–212. [Google Scholar]
- Engelmann, K.; Shen, H.; Finn, O.J. MCF7 Side population cells with characteristics of cancer stem/progenitor cells express the tumor antigen MUC1. Cancer Res. 2008, 68, 2419–2426. [Google Scholar]
- Tavaluc, R.; Hart, L.; Dicker, D.; El-Diery, W. Effects of low confluency, serum starvation and hypoxia on the side population of cancer cell lines. Cell Cycle 2007, 6, 2554–2562. [Google Scholar]
- Zhou, J.; Zhang, H.; Gu, P.; Margolick, J.B.; Yin, D.; Zhang, Y. Cancer stem/progenitor cell active compound 8-quinolinol in combination with paclitaxel achieves an improved cure of breast cancer in the mouse model. Breast Cancer Res. Treat. 2008, 115, 269–277. [Google Scholar]
- Santisteban, M.; Reiman, J.M.; Asiedu, M.K.; Behrens, M.D.; Nassar, A.; Kalli, K.R.; Haluska, P.; Ingle, J.N.; Hartmann, L.C.; Manjili, M.H.; et al. Immune-induced epithelial to mesenchymal transition in vivo generates breast cancer stem cells. Cancer Res. 2009, 69, 2887–2895. [Google Scholar]
- Kong, D.; Li, Y.; Wang, Z.; Sarkar, F.H. Cancer stem cells and epithelial-to-mesenchymal transition (EMT)-phenotypic cells: Are they cousins or twins? Cancers 2011, 3, 716–729. [Google Scholar]
- Hiscox, S.; Morgan, L.; Barrow, D.; Dutkowski, C.; Wakeling, A.; Nicholson, R. Tamoxifen resistance in breast cancer cells is accompanied by an enhanced motile and invasive phenotype: Inhibition by gefitinib (‘Iressa’, ZD1839). Clin. Exp. Metastasis 2004, 21, 201–212. [Google Scholar]
- Bandyopadhyay, A.; Wang, L.; Agyin, J.; Tang, Y.; Lin, S.; Yeh, I.T.; De, K.; Sun, L.-Z. Doxorubicin in combination with a small TGFβ inhibitor: A potential novel therapy for metastatic breast cancer in mouse models. PLoS One 2010, 5, e10365. [Google Scholar]
- Shi, G.-M.; Xu, Y.; Fan, J.; Zhou, J.; Yang, X.-R.; Qiu, S.-J.; Liao, Y.; Wu, W.-Z.; Ji, Y.; Ke, A.-W.; et al. Identification of side population cells in human hepatocellular carcinoma cell lines with stepwise metastatic potentials. J. Cancer Res. Clin. Oncol. 2008, 134, 1155–1163. [Google Scholar]
- Nakshatri, H.; Srour, E.; Badve, S. Breast cancer stem cells and intrinsic subtypes: Controversies rage on. Curr. Stem Cell Res. Ther. 2009, 4, 50–60. [Google Scholar]
- Muller, A.; Homey, B.; Soto, H.; Ge, N.; Catron, D.; Buchanan, M.E.; McClanahan, T.; Murphy, E.; Yuan, W.; Wagner, S.N.; et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 2001, 410, 50–56. [Google Scholar]
- Liang, Z.; Wu, H.; Reddy, S.; Zhu, A.; Wang, S.; Blevins, D.; Yoon, Y.; Zhang, Y.; Shim, H. Blockade of invasion and metastasis of breast cancer cells via targeting CXCR4 with an artificial microRNA. Biochem. Biophys. Res. Commun. 2007, 363, 542–546. [Google Scholar]
- Liang, Z.; Yoon, Y.; Votaw, J.; Goodman, M.M.; Williams, L.; Shim, H. Silencing of CXCR4 blocks breast cancer metastasis. Cancer Res. 2005, 65, 967–971. [Google Scholar]
- Smith, M.C.P.; Luker, K.E.; Garbow, J.R.; Prior, J.L.; Jackson, E.; Piwnica-Worms, D.; Luker, G.D. CXCR4 regulates growth of both primary and metastatic breast cancer. Cancer Res. 2004, 64, 8604–8612. [Google Scholar]
- Kato, M.; Kitayama, J.; Kazama, S.; Nagawa, H. Expression pattern of CXC chemokine receptor-4 is correlated with lymph node metastasis in human invasive ductal carcinoma. Breast Cancer Res. 2003, 5, R144–R150. [Google Scholar]
- Lee, B.-C.; Lee, T.-H.; Avraham, S.; Avraham, H.K. Involvement of the chemokine receptor CXCR4 and its ligand stromal cell-derived factor 1α in breast cancer cell migration through human brain microvascular endothelial cells. Mol. Cancer Res. 2004, 2, 327–338. [Google Scholar]
- Nilsson, J.; Ali, S.; Harvey, I.; Kirby, J.A.; Meeson, A.P. Stem cell therapy: A role for CXCR4 in homing bone marrow side population cells to areas of myocardial damage. Int. J. Cardiol. 2010. in press. [Google Scholar]
- Liang, S.X.; Tan, T.Y.L.; Gaudry, L.; Chong, B. Differentiation and migration of Sca1+/CD31− cardiac side population cells in a murine myocardial ischemic model. Int. J. Cardiol. 2010, 138, 40–49. [Google Scholar]
- Dean, M.; Fojo, T.; Bates, S. Tumour stem cells and drug resistance. Nat. Rev. Cancer 2005, 5, 275–284. [Google Scholar]
- Liu, H.; Patel, M.R.; Prescher, J.A.; Patsialou, A.; Qian, D.; Lin, J.; Wen, S.; Chang, Y.-F.; Bachmann, M.H.; Shimono, Y.; et al. Cancer stem cells from human breast tumors are involved in spontaneous metastases in orthotopic mouse models. Proc. Natl. Acad. Sci. USA 2010, 107, 18115–18120. [Google Scholar]
- de Boer, M.; van Dijck, J.A.A.M.; Bult, P.; Borm, G.F.; Tjan-Heijnen, V.C.G. Breast cancer prognosis and occult lymph node metastases, isolated tumor cells, and micrometastases. J. Nat. Cancer Inst. 2010, 102, 410–425. [Google Scholar]
- Jatoi, I.; Hilsenbeck, S.G.; Clark, G.M.; Osborne, C.K. Significance of axillary lymph node metastasis in primary breast cancer. J. Clin. Oncol. 1999, 17, 2334–2340. [Google Scholar]
- Aitken, S.J.; Thomas, J.S.; Langdon, S.P.; Harrison, D.J.; Faratian, D. Quantitative analysis of changes in ER, PR and HER2 expression in primary breast cancer and paired nodal metastases. Ann. Oncol. 2010, 21, 1254–1261. [Google Scholar]
- Lu, S.; Tian, J.; Lv, Z.; Wang, H.; Bai, X.; Liu, W.; Li, J.; Xu, W. The probable role of tumor stem cells for lymph node metastasis in supraglottic carcinoma. Pathol. Oncol. Res. 2010, 17, 33–38. [Google Scholar]
- Hennessy, B.; Gonzalez-Angulo, A.; Stemke-Hale, K.; Gilcrease, M.; Krishnamurthy, S.; Lee, J.; Fridlyand, J.; Sahin, A.; Agarwal, R.; Joy, C.; et al. Characterization of a naturally occurring breast cancer subset enriched in epithelial-to-mesenchymal transition and stem cell characteristics. Cancer Res. 2009, 69, 4116–4124. [Google Scholar]
- Gupta, P.B.; Onder, T.T.; Jiang, G.; Tao, K.; Kuperwasser, C.; Weinberg, R.A.; Lander, E.S. Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell 2009, 138, 645–659. [Google Scholar]
- Beug, M.H. Breast cancer stem cells: Eradication by differentiation therapy? Cell 2009, 138, 623–625. [Google Scholar]
- Polgar, O.; Bates, S.E. ABC transporters in the balance: Is there a role in multidrug resistance? Biochem. Soc. Trans. 2005, 33, 241–245. [Google Scholar]
- Fletcher, J.I.; Haber, M.; Henderson, M.J.; Norris, M.D. ABC transporters in cancer: More than just drug efflux pumps. Nat. Rev. Cancer 2010, 10, 147–156. [Google Scholar]
- Dorr, R.; Karanes, C.; Spier, C.; Grogan, T.; Greer, J.; Moore, J.; Weinberger, B.; Schiller, G.; Pearce, T.; Litchman, M.; et al. Phase I/II study of the p-glycoprotein modulator PSC 833 in patients with acute myeloid leukemia. J. Clin. Oncol. 2001, 19, 1589–1599. [Google Scholar]
- Greenberg, P.L.; Lee, S.J.; Advani, R.; Tallman, M.S.; Sikic, B.I.; Letendre, L.; Dugan, K.; Lum, B.; Chin, D.L.; Dewald, G.; et al. Mitoxantrone, etoposide, and cytarabine with or without valspodar in patients with relapsed or refractory acute myeloid leukemia and high-risk myelodysplastic syndrome: A phase III trial (E2995). J. Clin. Oncol. 2004, 22, 1078–1086. [Google Scholar]
- Baer, M.R.; George, S.L.; Dodge, R.K.; O'Loughlin, K.L.; Minderman, H.; Caligiuri, M.A.; Anastasi, J.; Powell, B.L.; Kolitz, J.E.; Schiffer, C.A.; et al. Phase 3 study of the multidrug resistance modulator PSC-833 in previously untreated patients 60 years of age and older with acute myeloid leukemia: Cancer and Leukemia Group B Study 9720. Blood 2002, 100, 1224–1232. [Google Scholar]
- Dean, M. ABC transporters, drug resistance, and cancer stem cells. J. Mammary Gland Biol. Neoplasi. 2009, 14, 3–9. [Google Scholar]
- Raaijmakers, M.H.G.P. ATP-binding-cassette transporters in hematopoietic stem cells and their utility as therapeutical targets in acute and chronic myeloid leukemia. Leukemia 2007, 21, 2094–2102. [Google Scholar]
- Robbiani, D.F.; Finch, R.A.; Jäger, D.; Muller, W.A.; Sartorelli, A.C.; Randolph, G.J. The leukotriene C4 transporter MRP1 regulates CCL19 (MIP-3[beta], ELC)-dependent mobilization of dendritic cells to lymph nodes. Cell 2000, 103, 757–768. [Google Scholar]
- Smyth, M.J.; Krasovskis, E.; Sutton, V.R.; Johnstone, R.W. The drug efflux protein, P-glycoprotein, additionally protects drug-resistant tumor cells from multiple forms of caspase-dependent apoptosis. Proc. Natl. Acad. Sci. USA 1998, 95, 7024–7029. [Google Scholar]
- Zöchbauer-Müller, S.; Filipits, M.; Rudas, M.; Brunner, R.; Krajnik, G.; Suchomel, R.; Schmid, K.; Pirker, R. P-glycoprotein and MRP1 expression in axillary lymph node metastases of breast cancer patients. Anticancer Res. 2001, 21, 119–124. [Google Scholar]
- Korkaya, H.; Wicha, M.S. Cancer stem cells: Nature versus nurture. Nat. Cell. Biol. 2010, 12, 419–421. [Google Scholar]
- Kruser, T.J.; Wheeler, D.L. Mechanisms of resistance to HER family targeting antibodies. Exp. Cell Res. 2010, 316, 1083–1100. [Google Scholar]
- Johnston, S.; Pippen, J.; Pivot, X.; Lichinitser, M.; Sadeghi, S.; Dieras, V.; Gomez, H.L.; Romieu, G.; Manikhas, A.; Kennedy, M.J.; et al. Lapatinib combined with letrozole versus letrozole and placebo as first-line therapy for postmenopausal hormone receptor-positive metastatic breast cancer. J. Clin. Oncol. 2009, 27, 5538–5546. [Google Scholar]
- Leary, A.F.; Drury, S.; Detre, S.; Pancholi, S.; Lykkesfeldt, A.E.; Martin, L.-A.; Dowsett, M.; Johnston, S.R.D. Lapatinib restores hormone sensitivity with differential effects on estrogen receptor signaling in cell models of human epidermal growth factor receptor 2-negative breast cancer with acquired endocrine resistance. Clin. Cancer Res. 2010, 16, 1486–1497. [Google Scholar]
- Korkaya, H.; Paulson, A.; Iovino, F.; Wicha, M.S. HER2 regulates the mammary stem/progenitor cell population driving tumorigenesis and invasion. Oncogene 2008, 27, 6120–6130. [Google Scholar]
- Magnifico, A.; Albano, L.; Campaner, S.; Delia, D.; Castiglioni, F.; Gasparini, P.; Sozzi, G.; Fontanella, E.; Menard, S.; Tagliabue, E. Tumor-initiating cells of HER2-positive carcinoma cell lines express the highest oncoprotein levels and are sensitive to trastuzumab. Clin. Cancer Res. 2009, 15, 2010–2021. [Google Scholar]
- Donnenberg, V.S.; Donnenberg, A.D. Multiple drug resistance in cancer revisited: the cancer stem cell hypothesis. J. Clin. Pharmacol. 2005, 45, 872–877. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Britton, K.M.; Kirby, J.A.; Lennard, T.W.J.; Meeson, A.P. Cancer Stem Cells and Side Population Cells in Breast Cancer and Metastasis. Cancers 2011, 3, 2106-2130. https://doi.org/10.3390/cancers3022106
Britton KM, Kirby JA, Lennard TWJ, Meeson AP. Cancer Stem Cells and Side Population Cells in Breast Cancer and Metastasis. Cancers. 2011; 3(2):2106-2130. https://doi.org/10.3390/cancers3022106
Chicago/Turabian StyleBritton, Kelly M., John A. Kirby, Thomas W.J. Lennard, and Annette P. Meeson. 2011. "Cancer Stem Cells and Side Population Cells in Breast Cancer and Metastasis" Cancers 3, no. 2: 2106-2130. https://doi.org/10.3390/cancers3022106
APA StyleBritton, K. M., Kirby, J. A., Lennard, T. W. J., & Meeson, A. P. (2011). Cancer Stem Cells and Side Population Cells in Breast Cancer and Metastasis. Cancers, 3(2), 2106-2130. https://doi.org/10.3390/cancers3022106



