The Correlation of the Presence and Expression Levels of cry Genes with the Insecticidal Activities against Plutella xylostella for Bacillus thuringiensis Strains
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Cell Cultivation
2.2. Bioassay of the Insecticidal Activity
2.3. Genotyping by PFGE and RAPD
2.4. Detection of cry Genes
| Target genes | Primer sequences | Product size (bp) | Reference |
|---|---|---|---|
| cry1 | F: 5'-CTGGATTTACAGGTGGGGATAT-3' | 558 | [19] |
| R: 5'-TGAGTCGCTTCGCATATTTGACT-3' | |||
| cry1B | F: 5'-CTTCATCACGATGGAGTAA-3' | 367 | [20] |
| R: 5'-CATAATTTGGTCGTTCTGTT-3' | |||
| cry1E | F: 5'-TAGGGATAAATGTAGTACAG-3' | 1137 | [21] |
| R: 5'-MDATYTCTAKRTCTTGACTA-3' | |||
| cry2Aa1 | F: 5'-GTTATTCTTAATGCAGATGAATGGG-3' | 498 | [22] |
| R: 5'-GAGATTAGTCGCCCCTATGAG-3' | |||
| cry3A | F: 5'-CGTTATCGCAGAGAGATGACATTAAC-3' | 951 | [22] |
| R: 5'-TGGTGCCCCGTCTAAACTGAGTGT-3' | |||
| cry4A2 | F: 5'-GGGTATGGCACTCAACCCCACTT-3' | 1529 | [22] |
| R: 5'-GCGTGACATACCCATTTCCAGGTCC-3' | |||
| cry7 | F: 5'-CAACCAGACCTATTTTATTGGAGT-3' | 476 | [23] |
| R: 5'-ATTTTTACAGCTGGAATTTTGTG-3' | |||
| cry8D | F: 5'-AGAAACACAAGATAAAATACTCC-3' | 401 | [23] |
| R: 5'-ATACAGCATCCCCTTCTACAATCT-3' | |||
| cry9A | F: 5'-GGTTCACTTACATTGCCGGTTAGC-3' | 1547 | [24] |
| R: 5'-GTTTGAGCCGCTTCACAGCAATCC-3' | |||
| cry9C | F: 5'-CCACCAGATGAAAGTACCGGAAG-3' | 1232 | [24] |
| R: 5'-GTTTGAGCCGCTTCACAGCAATCC-3' | |||
| cry9Ea | F: 5'-GCGGCTGGCTTTACTTTACCGAG-3' | 824 | [24] |
| R: 5'-GTTTGAGCCGCTTCACAGCAATCC-3' | |||
| cry22 | F: 5'-CAGATGAGATAGATGGGGATTTGA-3' | 413 | [23] |
| R: 5'-ATTCGCTTCTATACTTGGCTGTC-3' | |||
| cry32Aa | F: 5'-TGGTCGGGAGAGAATGGATGGA-3' | 676–677 | [25] |
| R: 5'-ATGTTTGCGACACCATTTTC-3' |
2.5. Expression Levels of cry1 Genes
2.6. Statistical Analysis
3. Results
3.1. Insecticidal Activity of Bt Strains
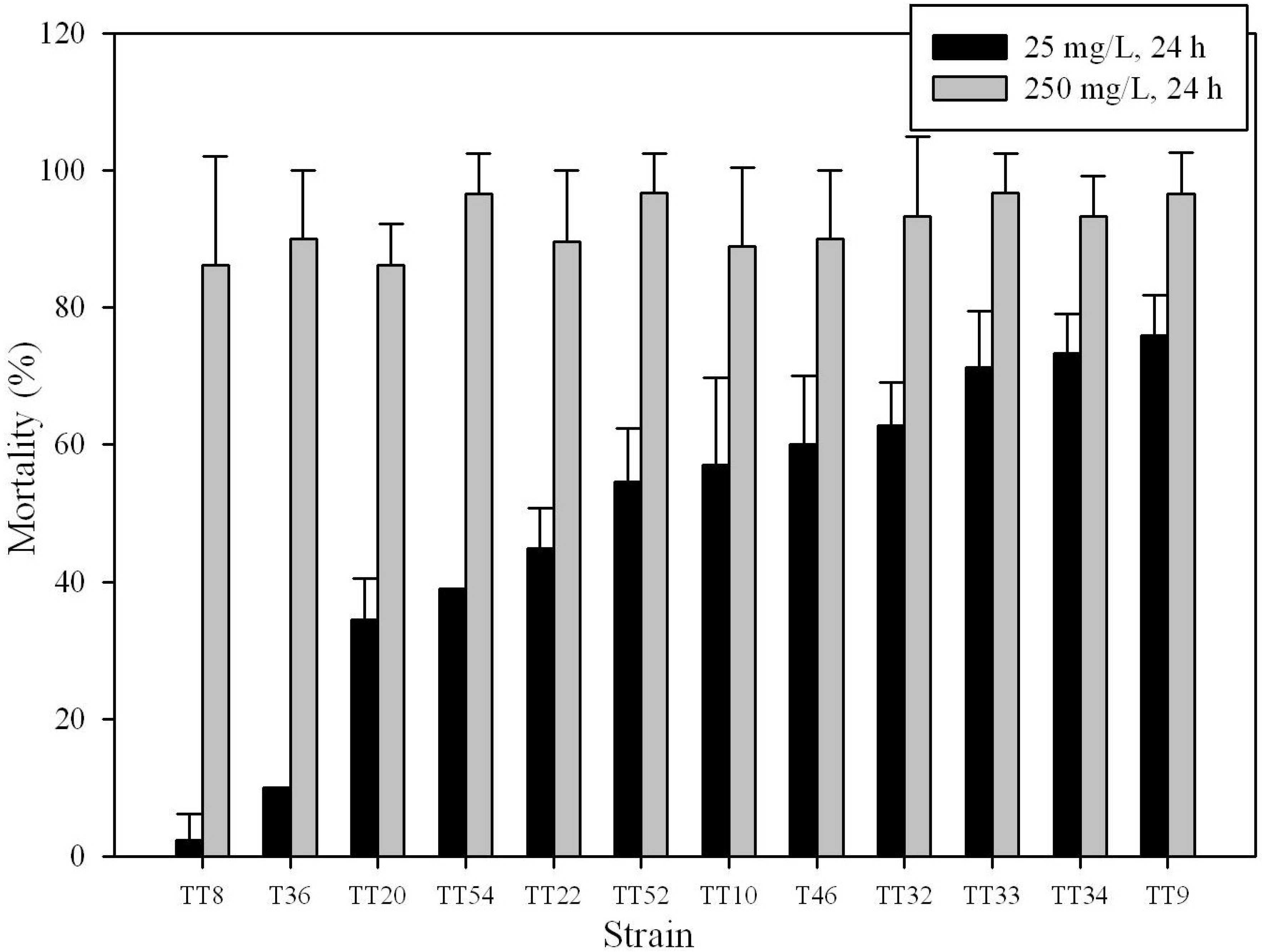
| Strain | RAPD pattern | PFGE pattern | Mortality (%) 1 | |||||
|---|---|---|---|---|---|---|---|---|
| 250 mg/L | 25 mg/L | |||||||
| 24 h | 48 h | 72 h | 24 h | 48 h | 72 h | |||
| TT1 | D11 | PT11 | 6.83 ± 0.00 | 12.57 ± 10.93 | 19.00 ± 12.00 | 4.24 ± 3.71 | 11.91 ± 1.14 | 10.19 ± 4.81 |
| TT2 | D12 | PT12 | 6.90 ± 1.04 | 20.07 ± 7.28 | 22.72 ± 3.70 | 4.55 ± 3.94 | 5.28 ± 6.31 | 4.94 ± 4.28 |
| TT3 | D13 | PT13 | 2.66 ± 4.60 | 1.49 ± 1.43 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| TT4 | D14 | PT14 | 0.00 ± 0.00 | 0.00 ± 0.00 | 12.67 ± 21.94 | 3.33 ± 5.77 | 2.33 ± 4.04 | 4.62 ± 4.00 |
| TT5 | D15 | PT15 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 96.55 ± 5.98 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT6 | D16 | PT16 | 93.10 ± 5.98 | 96.36 ± 6.31 | 96.00 ± 6.93 | 86.20 ± 15.81 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT7 | D17 | PT17 | 96.86 ± 5.44 | 96.68 ± 5.74 | 96.33 ± 6.35 | 86.20 ± 5.98 | 96.33 ± 6.35 | 100.00 ± 0.00 |
| TT8 | D18 | PT18 | 86.20 ± 15.81 | 100.00 ± 0.00 | 100.00 ± 0.00 | 2.28 ± 3.94 | 16.33 ± 5.77 | 15.12 ± 6.68 |
| TT9 | D19 | PT19 | 96.55 ± 5.98 | 100.00 ± 0.00 | 100.00 ± 0.00 | 75.85 ± 5.98 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT10 | D20 | PT20 | 88.89 ± 11.51 | 100.00 ± 0.00 | 100.00 ± 0.00 | 57.04 ± 12.75 | 89.33 ± 0.58 | 92.63 ± 6.40 |
| TT11 | D21 | PT21 | 4.55 ± 3.95 | 30.78 ± 6.31 | 26.70 ± 6.68 | 2.28 ± 3.94 | 2.00 ± 0.00 | 0.00 ± 0.00 |
| TT14 | D24 | PT23 | ND 2 | ND | ND | ND | ND | ND |
| TT15 | D25 | PT24 | 93.10 ± 11.95 | 96.36 ± 6.31 | 96.00 ± 6.93 | 14.91 ± 20.19 | 38.00 ± 45.40 | 42.13 ± 40.09 |
| TT16 | D26 | PT25 | 30.99 ± 5.98 | 74.50 ± 12.62 | 76.85 ± 11.57 | 4.55 ± 3.94 | 5.67 ± 6.35 | 2.47 ± 4.28 |
| TT17 | D27 | PT26 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 79.30 ± 10.35 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT18 | D28 | PT27 | 96.17 ± 6.63 | 100.00 ± 0.00 | 100.00 ± 0.00 | 65.49 ± 33.28 | 96.33 ± 6.35 | 96.14 ± 6.68 |
| TT19 | D29 | PT28 | 4.55 ± 3.94 | 1.64 ± 0.00 | 2.47 ± 4.28 | 4.93 ± 4.31 | 17.01 ± 5.74 | 24.54 ± 9.62 |
| TT20 | D30 | PT29 | 86.20 ± 5.98 | 100.00 ± 0.00 | 100.00 ± 0.00 | 34.44 ± 5.98 | 74.33 ± 12.70 | 72.99 ± 13.36 |
| TT21 | D31 | PT30 | 68.94 ± 0.00 | 92.71 ± 6.31 | 92.00 ± 6.93 | 17.18 ± 0.00 | 38.07 ± 6.31 | 45.99 ± 6.68 |
| TT22 | D32 | PT31 | 89.65 ± 10.35 | 100.00 ± 0.00 | 100.00 ± 0.00 | 44.79 ± 5.98 | 92.67 ± 12.70 | 92.28 ± 13.36 |
| TT23 | ND | ND | 3.33 ± 5.57 | 11.51 ± 14.37 | 20.67 ± 6.35 | 3.33 ± 5.77 | 10.07 ± 6.26 | 20.10 ± 6.57 |
| TT24 | D33 | PT32 | 48.13 ± 3.23 | 89.66 ± 10.34 | 89.66 ± 10.34 | 3.33 ± 5.77 | 10.38 ± 5.97 | 13.82 ± 5.97 |
| TT25 | D34 | PT33 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 93.10 ± 11.95 | 96.33 ± 6.35 | 96.14 ± 6.68 |
| TT26 | D35 | PT34 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 93.33 ± 5.77 | 96.67 ± 5.77 | 96.49 ± 6.07 |
| TT27 | D36 | PT35 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 93.33 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT28 | D22 | PT36 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 89.27 ± 11.12 | 96.33 ± 6.35 | 96.11 ± 6.74 |
| TT29 | D34 | PT37 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 93.33 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT30 | D37 | PT38 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 93.33 ± 11.55 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT31 | D38 | PT39 | 96.67 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | 90.00 ± 17.32 | 96.67 ± 5.77 | 96.49 ± 6.07 |
| TT32 | D22 | PT40 | 93.33 ± 11.55 | 100.00 ± 0.00 | 100.00 ± 0.00 | 62.77 ± 6.32 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT33 | D39 | PT41 | 96.67 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | 71.20 ± 8.27 | 90.33 ± 0.58 | 89.80 ± 0.55 |
| TT34 | D40 | PT41 | 93.33 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | 73.33 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT35 | D38 | PT42 | 63.33 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | 21.67 ± 2.89 | 77.67 ± 10.97 | 87.94 ± 13.01 |
| TT36 | D41 | PT43 | 90.00 ± 10.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 10.00 ± 0.00 | 44.85 ± 5.97 | 58.63 ± 10.34 |
| TT37 | D42 | PT44 | 80.00 ± 34.64 | 100.00 ± 0.00 | 100.00 ± 0.00 | 46.67 ± 5.77 | 96.67 ± 5.77 | 96.55 ± 5.97 |
| TT38 | D43 | PT45 | 80.00 ± 26.46 | 100.00 ± 0.00 | 100.00 ± 0.00 | 33.33 ± 11.55 | 72.33 ± 11.55 | 72.42 ± 11.94 |
| TT39 | D44 | PT46 | 96.67 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | 33.75 ± 5.30 | 76.73 ± 3.66 | 76.73 ± 3.66 |
| TT40 | D36 | PT47 | 90.00 ± 17.32 | 100.00 ± 0.00 | 100.00 ± 0.00 | 50.00 ± 43.59 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT41 | ND | ND | 10.00 ± 10.00 | 17.00 ± 0.00 | 8.00 ± 6.93 | 6.67 ± 5.77 | 6.93 ± 0.00 | 0.00 ± 0.00 |
| TT42 | D34 | PT48 | 96.67 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | 43.33 ± 5.77 | 86.33 ± 15.82 | 92.03 ± 6.91 |
| TT43 | D45 | PT49 | 13.33 ± 5.77 | 37.95 ± 10.34 | 40.19 ± 11.96 | 0.00 ± 0.00 | 4.62 ± 4.00 | 2.87 ± 2.49 |
| TT44 | D46 | PT50 | 40.00 ± 0.00 | 58.63 ± 10.34 | 52.00 ± 12.00 | 3.33 ± 5.77 | 20.72 ± 5.97 | 12.28 ± 6.91 |
| TT45 | D47 | PT50 | 96.67 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | 83.33 ± 5.77 | 96.67 ± 5.77 | 96.01 ± 6.91 |
| TT46 | D33 | PT51 | 90.00 ± 10.00 | 96.55 ± 5.87 | 96.00 ± 6.93 | 60.00 ± 10.00 | 79.33 ± 10.50 | 80.06 ± 13.81 |
| TT47 | D43 | PT52 | 96.67 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | 74.53 ± 9.47 | 84.00 ± 10.39 | 83.42 ± 10.50 |
| TT48 | D43 | PT52 | 93.33 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | 60.00 ± 10.00 | 82.67 ± 6.35 | 88.04 ± 11.96 |
| TT49 | D43 | PT52 | 88.57 ± 2.48 | 100.00 ± 0.00 | 100.00 ± 0.00 | 80.00 ± 10.00 | 96.67 ± 5.77 | 96.01 ± 6.91 |
| TT50 | D43 | PT52 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 83.33 ± 15.28 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT51 | D48 | PT1 | 70.00 ± 10.00 | 96.55 ± 5.97 | 96.00 ± 6.93 | 3.33 ± 5.77 | 24.33 ± 6.35 | 24.24 ± 6.91 |
| TT52 | D38 | PT53 | 96.67 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | 54.53 ± 7.85 | 84.33 ± 13.80 | 82.06 ± 15.82 |
| TT53 | D41 | PT54 | 96.61 ± 5.87 | 100.00 ± 0.00 | 100.00 ± 0.00 | 86.45 ± 5.87 | 89.33 ± 10.50 | 88.91 ± 11.09 |
| TT54 | D2 | PT2 | 96.61 ± 5.87 | 100.00 ± 0.00 | 100.00 ± 0.00 | 39.02 ± 0.00 | 47.42 ± 10.52 | 51.96 ± 6.40 |
| TT55 | D49 | PT55 | 10.00 ± 10.00 | 17.27 ± 10.34 | 20.72 ± 5.97 | 3.33 ± 5.77 | 7.00 ± 0.00 | 13.82 ± 5.97 |
| TT56 | D50 | PT56 | 0.00 ± 0.00 | 3.58 ± 3.10 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.07 ± 0.13 |
| TT57 | D51 | PT57 | 6.54 ± 5.80 | 4.45 ± 4.07 | 1.00 ± 1.73 | 2.85 ± 4.93 | 3.58 ± 3.10 | 0.15 ± 0.13 |
| TT58 | D22 | PT58 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 89.84 ± 10.16 | 96.33 ± 6.35 | 96.30 ± 6.40 |
| TT59 | D38 | PT59 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT60 | D52 | PT60 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 96.61 ± 5.87 | 96.33 ± 6.35 | 96.30 ± 6.40 |
| TT61 | D34 | PT61 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 96.61 ± 5.87 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT62 | D53 | PT62 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| TT12 (positive control) | D22 | PT17 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 79.30 ± 10.35 | 96.33 ± 6.35 | 96.14 ± 6.68 |
| TT13 (positive control) | D23 | PT22 | 96.55 ± 5.98 | 100.00 ± 0.00 | 100.00 ± 0.00 | 50.17 ± 3.35 | 81.33 ± 12.42 | 80.29 ± 13.01 |
| Tris buffer (negative control) | - | - | 1.00 ± 3.05 | 4.67 ± 5.71 | 9.27 ± 7.31 | 1.00 ± 3.05 | 4.67 ± 5.71 | 9.27 ± 7.31 |
3.2. PFGE and RAPD Patterns
3.3. Correlation among Insecticidal Activity, cry Genes, and the Expression Levels of cry1 Genes
| Strain | Mortality (%) 1 | cry genes 2 | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 25 mg/L (global Cry protein) | ||||||||||||||||
| 24 h | 48 h | 72 h | 1 | 1B | 1E | 2Aa1 | 3A | 4A2 | 7 | 8 | 9A | 9C | 9Ea | 22 | 32Aa | |
| TT59 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | + | - | - | + | - | - | - | - | - | - | - | - | - |
| TT62 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | + | - | - | + | - | - | - | - | - | - | - | - | - |
| TT61 | 96.61 ± 5.87 | 100.00 ± 0.00 | 100.00 ± 0.00 | + | - | - | + | - | - | - | - | - | - | - | - | - |
| TT5 | 96.55 ± 5.98 | 100.00 ± 0.00 | 100.00 ± 0.00 | + | - | - | - | - | - | - | - | - | - | + | - | - |
| TT27 | 93.33 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | + | - | - | + | - | - | - | - | - | - | - | - | - |
| TT29 | 93.33 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | + | - | - | + | - | - | - | - | - | - | - | - | - |
| TT30 | 93.33 ± 11.55 | 100.00 ± 0.00 | 100.00 ± 0.00 | + | - | - | - | - | - | - | - | - | - | + | - | - |
| TT25 | 93.10 ± 11.95 | 96.33 ± 6.35 | 96.14 ± 6.68 | + | - | - | + | - | - | - | - | - | - | - | - | - |
| TT50 | 83.33 ± 15.28 | 100.00 ± 0.00 | 100.00 ± 0.00 | + | + | - | + | - | - | - | - | - | - | - | - | - |
| TT49 | 80.00 ± 10.00 | 96.67 ± 5.77 | 96.01 ± 6.91 | + | + | - | + | - | - | - | - | - | - | - | - | - |
| TT17 | 79.30 ± 10.35 | 100.00 ± 0.00 | 100.00 ± 0.00 | + | - | - | + | - | - | - | - | - | - | - | - | - |
| TT12 | 79.30 ± 10.35 | 96.33 ± 6.35 | 96.14 ± 6.68 | + | - | - | + | - | - | - | - | - | - | - | - | - |
| TT47 | 74.53 ± 9.47 | 84.00 ± 10.39 | 83.42 ± 10.50 | + | + | - | + | - | - | - | - | - | - | - | - | - |
| TT34 | 73.33 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 | + | - | - | + | - | - | - | - | - | - | - | - | - |
| TT48 | 60.00 ± 10.00 | 82.67 ± 6.35 | 88.04 ± 11.96 | + | + | - | + | - | - | - | - | - | - | - | - | - |
| TT41 | 6.67 ± 5.77 | 6.93 ± 0.00 | 0.00 ± 0.00 | + | - | - | + | - | - | - | - | - | - | - | - | - |
| TT19 | 4.93 ± 4.31 | 17.01 ± 5.74 | 24.54 ± 9.62 | + | - | - | - | - | - | - | - | - | - | - | - | - |
| TT2 | 4.55 ± 3.94 | 5.28 ± 6.31 | 4.94 ± 4.28 | + | - | - | - | - | - | - | - | - | - | - | - | - |
| TT16 | 4.55 ± 3.94 | 5.67 ± 6.35 | 2.47 ± 4.28 | + | - | - | - | - | - | - | - | - | - | - | - | - |
| TT1 | 4.24 ± 3.71 | 11.91 ± 1.14 | 10.19 ± 4.81 | + | - | - | + | - | - | - | - | - | - | - | - | - |
| TT44 | 3.33 ± 5.77 | 20.72 ± 5.97 | 12.28 ± 6.91 | + | - | - | - | - | - | - | - | - | - | + | - | - |
| TT24 | 3.33 ± 5.77 | 10.38 ± 5.97 | 13.82 ± 5.97 | + | - | - | + | - | - | - | - | - | - | - | - | - |
| TT4 | 3.33 ± 5.77 | 2.33 ± 4.04 | 4.62 ± 4.00 | + | - | - | - | - | - | - | - | - | - | - | - | - |
| TT23 | 3.33 ± 5.77 | 10.07 ± 6.26 | 20.10 ± 6.57 | + | - | - | - | - | - | - | - | - | - | - | + | - |
| TT57 | 2.85 ± 4.93 | 3.58 ± 3.10 | 0.15 ± 0.13 | + | - | - | - | - | - | + | - | - | - | - | - | - |
| TT11 | 2.28 ± 3.94 | 2.00 ± 0.00 | 0.00 ± 0.00 | + | - | - | - | - | - | - | - | - | - | - | - | - |
| TT43 | 0.00 ± 0.00 | 4.62 ± 4.00 | 2.87 ± 2.49 | + | - | - | - | - | - | - | - | - | - | - | - | - |
| TT56 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.07 ± 0.13 | + | - | - | - | - | - | + | - | - | - | - | - | - |
| TT3 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | + | - | - | - | - | - | - | - | - | - | - | - | - |
| Total | 29+ 3 | 4+ | 0+ | 16+ | 0+ | 0+ | 2+ | 0+ | 0+ | 0+ | 3+ | 1+ | 0+ | |||
| Variables | Parameters | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 24 h | 48 h | 72 h | |||||||
| Standardized coefficient | t | p | Standardized coefficient | t | p | Standardized coefficient | t | p | |
| cry1B | 0.048 | 0.324 | 0.749 | 0.107 | 0.773 | 0.447 | 0.117 | 0.822 | 0.419 |
| cry2Aa1 | 0.770 | 4.369 | 0.000 | 0.786 | 4.794 | 0.000 | 0.770 | 4.551 | 0.000 |
| cry7 | −0.008 | −0.054 | 0.957 | −0.019 | −0.138 | 0.891 | −0.031 | −0.211 | 0.835 |
| cry9Ea | 0.442 | 2.822 | 0.010 | 0.459 | 3.154 | 0.004 | 0.434 | 2.888 | 0.008 |
| cry22 | 0.002 | 0.016 | 0.988 | 0.019 | 0.143 | 0.888 | 0.058 | 0.414 | 0.683 |
| R square | 0.559 | 0.619 | 0.594 | ||||||
| df1 | 5 | 5 | 5 | ||||||
| df2 | 23 | 23 | 23 | ||||||
| Sig. F Change | 0.001 | 0.000 | 0.001 | ||||||
| Strain | Mortality (%) | Expression level of cry1 genes (Ct value) | ||
|---|---|---|---|---|
| 25 mg/L (global Cry protein) | ||||
| 24 h | 48 h | 72 h | ||
| TT30 | 93.33 ± 11.55 | 100.00 ± 0.00 | 100.00 ± 0.00 | 17.83 |
| TT59 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 18.92 |
| TT48 | 60.00 ± 10.00 | 82.67 ± 6.35 | 88.04 ± 11.96 | 19.00 |
| TT50 | 83.33 ± 15.28 | 100.00 ± 0.00 | 100.00 ± 0.00 | 22.73 |
| TT12 | 79.30 ± 10.35 | 96.33 ± 6.35 | 96.14 ± 6.68 | 22.76 |
| TT57 | 2.85 ± 4.93 | 3.58 ± 3.10 | 0.15 ± 0.13 | 38.25 |
| TT23 | 3.33 ± 5.77 | 10.07 ± 6.26 | 20.10 ± 6.57 | 39.59 |
| TT4 | 3.33 ± 5.77 | 2.33 ± 4.04 | 4.62 ± 4.00 | 39.79 |
| TT56 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.07 ± 0.13 | 39.80 |
| TT47 | 74.53 ± 9.47 | 84.00 ± 10.39 | 83.42 ± 10.50 | 39.86 |
| TT3 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 39.87 |
| TT11 | 2.28 ± 3.94 | 2.00 ± 0.00 | 0.00 ± 0.00 | 39.88 |
| TT19 | 4.93 ± 4.31 | 17.01 ± 5.74 | 24.54 ± 9.62 | 39.91 |
| TT49 | 80.00 ± 10.00 | 96.67 ± 5.77 | 96.01 ± 6.91 | 39.98 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schnepf, E.; Crickmore, N.; van Rie, J.; Lereclus, D.; Baum, J.; Feitelson, J.; Zeigler, D.R.; Dean, D.H. Bacillus thuringiensis and its pesticidal crystal proteins. Microbiol. Mol. Biol. Rev. 1998, 62, 775–806. [Google Scholar]
- Höfte, H.; Whiteley, H.R. Insecticidal crystal proteins of Bacillus thuringiensis. Microbiol. Rev. 1989, 53, 242–255. [Google Scholar]
- Bravo, A.; Gómez, I.; Porta, H.; García-Gómez, B.I.; Rodriguez-Almazan, C.; Pardo, L.; Soberón, M. Evolution of Bacillus thuringiensis Cry toxins insecticidal activity. Microb. Biotechnol. 2013, 6, 17–26. [Google Scholar] [CrossRef]
- Porcar, M.; Juárez-Pérez, V. PCR-based identification of Bacillus thuringiensis pesticidal crystal genes. FEMS Microbiol. Rev. 2003, 26, 419–432. [Google Scholar] [CrossRef]
- Henderson, I.; Yu, D.; Turnbull, P.C. Differentiation of Bacillus anthracis and other “Bacillus cereus group” bacteria using IS231-derived sequences. FEMS Microbiol. Lett. 1995, 128, 113–118. [Google Scholar] [CrossRef]
- Crickmore, N.; Zeigler, D.R.; Feitelson, J.; Schnepf, E.; van Rie, J.; Lereclus, D.; Baum, J.; Dean, D.H. Revision of the nomenclature for the Bacillus thuringiensis pesticidal crystal proteins. Microbiol. Mol. Biol. Rev. 1998, 62, 807–813. [Google Scholar]
- Crickmore, N.; Baum, J.; Bravo, A.; Lereclus, D.; Narva, K.; Sampson, K.; Schnepf, E.; Sun, M.; Zeigler, D.R. Bacillus thuringiensis toxin nomenclature. Available online: http://www.btnomenclature.info/ (accessed on 15 April 2014).
- Ferrandis, M.D.; Juárez-Pérez, V.M.; Frutos, R.; Bel, Y.; Ferré, J. Distribution of cryl, cryll and cryV genes within Bacillus thuringiensis isolates from Spain. Syst. Appl. Microbiol. 1999, 22, 179–185. [Google Scholar] [CrossRef]
- Kuo, W.S.; Chak, K.F. Identification of novel cry-type genes from Bacillus thuringiensis strains on the basis of restriction fragment length polymorphism of the PCR-amplified DNA. Appl. Environ. Microbiol. 1996, 62, 1369–1377. [Google Scholar]
- Świȩcicka, I.; Mahillon, J. The clonal structure of Bacillus thuringiensis isolates from north-east Poland does not correlate with their cry gene diversity. Environ. Microbiol. 2005, 7, 34–39. [Google Scholar] [CrossRef]
- Chen, F.C.; Tsai, M.C.; Peng, C.H.; Chak, K.F. Dissection of cry gene profiles of Bacillus thuringiensis isolates in Taiwan. Curr. Microbiol. 2004, 48, 270–275. [Google Scholar] [CrossRef]
- Mohan, M.; Gujar, G.T. Toxicity of Bacillus thuringiensis strains and commercial formulations to the diamondback moth, Plutella xylostella (L.). Crop Prot. 2001, 20, 311–316. [Google Scholar] [CrossRef]
- Kumar, D.; Chaudhary, K.; Boora, K.S. Characterization of native Bacillus thuringiensis strains by PCR-RAPD based fingerprinting. Indian J. Microbiol. 2010, 50, 27–32. [Google Scholar] [CrossRef]
- Frankenhuyzen, K.V. Insecticidal activity of Bacillus thuringiensis crystal proteins. J. Invertebr. Pathol. 2009, 101, 1–16. [Google Scholar] [CrossRef]
- Pang, J.C.; Chen, M.L.; Ho, Y.C.; Yang, C.Y.; Tzeng, C.C.; Kao, S.S.; Tsen, H.Y. Effect of fermentation conditions on the enterotoxigenicity, cytotoxicity and pesticidal activity of Bacillus thuringiensis strains isolated in Taiwan. Bioresour. Technol. 2010, 101, 1871–1876. [Google Scholar] [CrossRef]
- Abbott, W. A method for computing the effectiveness of an insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar]
- Kolstø, A.B.; Gronstad, A.; Oppegaard, H. Physical map of the Bacillus cereus chromosome. J. Bacteriol. 1990, 172, 3821–3825. [Google Scholar]
- Ausubel, F.M.; Kingstone, R.E.; More, D.D.; Seidman, J.G.; Smith, J.A.; Struhl, K. Current Protocols in Molecular Biology; John Wiley & Sons: New York, NY, USA, 1987. [Google Scholar]
- Bravo, A.; Sarabia, S.; Lopez, L.; Ontiveros, H.; Abarca, C.; Ortiz, A.; Ortiz, M.; Lina, L.; Villalobos, F.J.; Pena, G.; et al. Characterization of cry genes in a Mexican Bacillus thuringiensis strain collection. Appl. Environ. Microbiol. 1998, 64, 4965–4972. [Google Scholar]
- Céron, J.; Covarrubis, L.; Quintero, R.; Ortiz, A.; Ortiz, M.; Aranda, E.; Lina, L.; Bravo, A. PCR analysis of the cryI insecticidal crystal family genes from Bacillus thuringiensis. Appl. Environ. Microbiol. 1994, 60, 353–356. [Google Scholar]
- Juárez-Pérez, V.M.; Ferrandis, M.D.; Frutos, R. PCR-based approach for detection of novel Bacillus thuringiensis cry genes. Appl. Environ. Microbiol. 1997, 63, 2997–3002. [Google Scholar]
- Ben-Dov, E.; Zaritsky, A.; Dahan, E.; Barak, Z.; Sinai, R.; Manasherob, R.; Khamraev, A.; Troitskaya, E.; Dubitsky, A.; Berezina, N.; et al. Extended screening by PCR for seven cry-group genes from field-collected strains of Bacillus thuringiensis. Appl. Environ. Microbiol. 1997, 63, 4883–4890. [Google Scholar]
- Ejiofor, A.O.; Johnson, T. Physiological and molecular detection of crystalliferous Bacillus thuringiensis strains from habitats in the South Central United States. J. Ind. Microbiol. Biotechnol. 2002, 28, 284–290. [Google Scholar] [CrossRef]
- Ben-Dov, E.; Wang, Q.; Zaritsky, A.; Manasherob, R.; Barak, Z.; Schneider, B.; Khamraev, A.; Baizhanov, M.; Glupov, V.; Margalith, Y. Multiplex PCR screening to detect cry9 genes in Bacillus thuringiensis strains. Appl. Environ. Microbiol. 1999, 65, 3714–3716. [Google Scholar]
- Ibarra, J.E.; del Rincón, M.C.; Ordúz, S.; Noriega, D.; Benintende, G.; Monnerat, R.; Regis, L.; de Oliveira, C.M.; Lanz, H.; Rodriguez, M.H.; et al. Diversity of Bacillus thuringiensis strains from Latin America with insecticidal activity against different mosquito species. Appl. Environ. Microbiol. 2003, 69, 5269–5274. [Google Scholar] [CrossRef]
- Gaviria Rivera, A.M.; Priest, F.G. Pulsed field gel electrophoresis of chromosomal DNA reveals a clonal population structure to Bacillus thuringiensis that relates in general to crystal protein gene content. FEMS Microbiol. Lett. 2003, 223, 61–66. [Google Scholar] [CrossRef]
- Martínez, C.; Ibarra, J.E.; Caballero, P. Association analysis between serotype, cry gene content, and toxicity to Helicoverpa armiger larvae among Bacillus thuringiensis isolates native to Spain. J. Invertebr. Pathol. 2005, 90, 91–97. [Google Scholar] [CrossRef]
- Ammouneh, H.; Harba, M.; Idris, E.; Makee, H. Isolation and characterization of native Bacillus thuringiensis isolates from Syrian soil and testing of their insecticidal activities against some insect pests. Turk. J. Agric. For. 2011, 35, 421–431. [Google Scholar]
- Kaur, S. Molecular approaches for identification and construction of novel insecticidal genes for crop protection. World J. Microbiol. Biotechnol. 2006, 22, 233–253. [Google Scholar] [CrossRef]
- Carlson, C.R.; Caugant, D.A.; Kolstø, A.B. Genotypic diversity among Bacillus cereus and Bacillus thuringiensis strains. Appl. Environ. Microbiol. 1994, 60, 1719–1725. [Google Scholar]
- Asano, S.; Maruyama, T.; Iwasa, T.; Seki, A.; Takahashi, M.; Soares, G.G., Jr. Evaluation of biological activity of Bacillus thuringiensis test samples using a diet incorporation method with diamondback moth, Plutella xylostella (Linnaeus) (Lepidoptera: Yponomeutidae). Appl. Entomol. Zoolog. 1993, 28, 513–524. [Google Scholar]
- Bauce, É.; Bidon, Y.; Berthiaume, R. Effects of food nutritive quality and Bacillus thuringiensis on feeding behaviour, food utilization and larval growth of spruce budworm Choristoneura fumiferana (Clem.) when exposed as fourth-and sixth-instar larvae. Agric. For. Entomol. 2002, 4, 57–70. [Google Scholar] [CrossRef]
- Hill, T.A.; Foster, R.E. Effect of insecticides on the diamondback moth (Lepidoptera: Plutellidae) and its parasitoid Diadegma insulare (Hymenoptera: Ichneumonidae). J. Econ. Entomol. 2000, 93, 763–768. [Google Scholar] [CrossRef]
- Ayalew, G. Comparison of yield loss on cabbage from Diamondback moth, Plutella xylostella L. (Lepidoptera: Plutellidae) using two insecticides. Crop Prot. 2006, 25, 915–919. [Google Scholar] [CrossRef]
- Poncet, S.; Delécluse, A.; Klier, A.; Rapoport, G. Evaluation of synergistic interactions among the CryIVA, CryIVB, and CryIVD toxic components of B. thuringiensis subsp. israelensis crystals. J. Invertebr. Pathol. 1995, 66, 131–135. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chen, M.-L.; Chen, P.-H.; Pang, J.-C.; Lin, C.-W.; Hwang, C.-F.; Tsen, H.-Y. The Correlation of the Presence and Expression Levels of cry Genes with the Insecticidal Activities against Plutella xylostella for Bacillus thuringiensis Strains. Toxins 2014, 6, 2453-2470. https://doi.org/10.3390/toxins6082453
Chen M-L, Chen P-H, Pang J-C, Lin C-W, Hwang C-F, Tsen H-Y. The Correlation of the Presence and Expression Levels of cry Genes with the Insecticidal Activities against Plutella xylostella for Bacillus thuringiensis Strains. Toxins. 2014; 6(8):2453-2470. https://doi.org/10.3390/toxins6082453
Chicago/Turabian StyleChen, Ming-Lun, Pin-Hsin Chen, Jen-Chieh Pang, Chia-Wei Lin, Chin-Fa Hwang, and Hau-Yang Tsen. 2014. "The Correlation of the Presence and Expression Levels of cry Genes with the Insecticidal Activities against Plutella xylostella for Bacillus thuringiensis Strains" Toxins 6, no. 8: 2453-2470. https://doi.org/10.3390/toxins6082453




