Changes in Gene Expression in the Larval Gut of Ostrinia nubilalis in Response to Bacillus thuringiensis Cry1Ab Protoxin Ingestion
Abstract
:1. Introduction
2. Results and Discussion
2.1. Overview of Transcriptional Responses in O. nubilalis Larvae Fed Cry1Ab Protoxin
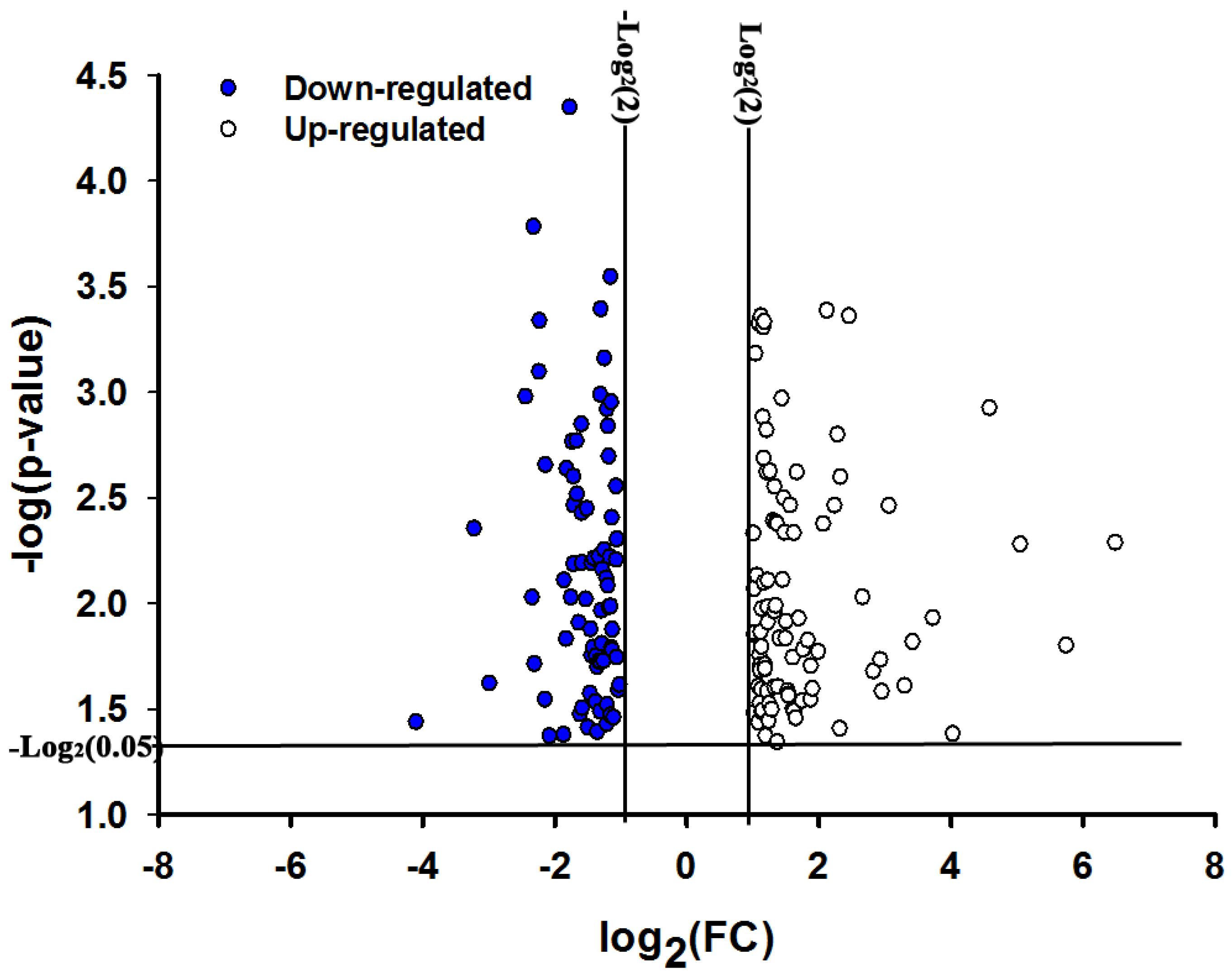
| EST ID | NCBI EST database ID | Gene Homologs | Homolog GenBank Accession No. | Fold Change ± SE * |
|---|---|---|---|---|
| Bt toxin solubilization, activation, degradation or sequestration | ||||
| contig [0243] | GH998064.1 | trypsin precursor | AFM77760.1 | −2.69 ± 0.29 |
| contig [0389] | GH998056.1 | serine protease | AFM77769.1 | 8.39 ± 0.02 |
| contig [0770] | GH997442.1 | trypsin-like serine protease | AFM77762.1 | 3.16 ± 0.14 |
| contig [1207] | GH997507.1 | serine protease | AFM77770.1 | 7.67 ± 0.26 |
| contig [3704] | GH999118.1 | trypsin-like serine protease | AFM77754.1 | 6.36 ± 0.04 |
| contig [4768] | GH998250.1 | trypsin-like serine protease | AFM77753.1 | 10.75 ± 0.12 |
| contig [5740] | GH999046.1 | chymotrypsin-2 (chymotrypsin ii) | AFM77774.1 | 3.26 ± 0.19 |
| ECB-C-18_B11 | GH994018.1 | silk gland derived serine protease | AAR98920.2 | −2.63 ± 0.04 |
| contig [0115] | GH997328.1 | esterase FE4-like (Bombyx mori) | XP_004924612.1 | 3.69 ± 0.13 |
| contig [3820] | GH999448.1 | carboxylesterase (Helicoverpa armigera) | ADE05548.1 | −2.21 ± 0.12 |
| J-ECB-07_G03 | GH991809.1 | carboxylesterase (Loxostege sticticalis) | ACA50924.1 | 3.58 ± 0.06 |
| J-ECB-09_D02 | GH992373.1 | carboxylesterase (Spodoptera litura) | AEJ38204.1 | −3.31 ± 0.22 |
| ECB-17_F12 | GH998536.1 | carboxylesterase (Helicoverpa armigera) | ADD97156.1 | 3.99 ± 0.06 |
| ECB-27_F04 | GH999378.1 | carboxyl/cholinesterase 4A (Bombyx mori) | NP_001116814.1 | −2.83 ± 0.11 |
| Potential Bt toxin binding partners | ||||
| J-ECB-25_B09 | GH990771.1 | cadherin-like protein | ACK37450.1 | 2.85 ± 0.20 |
| ECB-V-05_D12 | GH994582.1 | aminopeptidase n3 | AEO12689.1 | −2.55 ± 0.23 |
| contig [4776] | GH998970.1 | aminopeptidase n2 | ACJ64828.1 | 2.17 ± 0.09 |
| contig [4879] | GH997475.1 | aminopeptidase n8 | ACV04931.1 | −2.31 ± 0.12 |
| contig [5858] | GH998639.1 | membrane-bound alkaline phosphatase (Ostrinia furnacalis) | AEM43806.1 | 2.23 ± 0.06 |
| Signal transduction | ||||
| contig [0492] | GH998299.1 | caspase-4 (Lymantria monacha) | AEK20829.1 | 2.36 ± 0.03 |
| ECB-10_C01 | GH997883.1 | pyridoxal kinase (Bombyx mori) | NP_001037440.1 | −2.99 ± 0.10 |
| contig [5143] | GH995296.1 | ctl2 antioxidant enzyme (Aedes aegypti) | XP_001661235.1 | 2.32 ± 0.04 |
| Transporter | ||||
| contig [0814] | GH998546.1 | sodium-bile acid cotransporter (Danaus plexippus) | EHJ73754.1 | −5.03 ± 0.05 |
| contig [1314] | GH993616.1 | potassium coupled amino acid transporter (Manduca sexta) | AAF18560.1 | −4.68 ± 0.16 |
| contig [4763] | GH998142.1 | sodium-bile acid cotransporter (Aedes aegypti) | XP_001662576.1 | −4.39 ± 0.10 |
| contig [5743] | GH993678.1 | amino acid transporter (Bombyx mori) | NP_001124343.1 | −5.40 ± 0.13 |
| ECB-21_C09 | GH998857.1 | sugar transporter (Danaus plexippus) | EHJ73890.1 | −2.46 ± 0.10 |
| J-ECB-39_E12 | GH992066.1 | sugar transporter (Culex quinquefasciatus) | XP_001862938.1 | −2.65 ± 0.25 |
| J-ECB-55_E04 | GH988996.1 | monocarboxylate transporter (Bombyx mori) | XP_004927805.1 | −3.40 ± 0.10 |
| Transcription factor and gene expression | ||||
| contig [3833] | GH993952.1 | DNA-binding nuclear protein p8 (Simulium guianense) | ACH56888.1 | 4.89 ± 0.09 |
| contig [4800] | GH998367.1 | endonuclease-reverse transcriptase (Bombyx mori) | ADI61826.1 | 2.49 ± 0.04 |
| ECB-V-26_F03 | GH996285.1 | histone H3.2-like (Meleagris gallopavo) | XP_003202254.1 | −2.04 ± 0.02 |
| contig [3869] | GH997175.1 | cellular repressor of E1A-stimulated genes 1 (Tribolium castaneum) | XP_972946.1 | −3.16 ± 0.06 |
| contig [5038] | GH991382.1 | MluI cell cycle box (MCB) Binding Factor 2 (Samia cynthia) | BAA34219.1 | 2.41 ± 0.11 |
| Metabolism | ||||
| Xenobiotics metabolism | ||||
| contig [0004] | GH992504.1 | glutathione S-transferase (Choristoneura fumiferana) | AAF23078.1 | −3.53 ± 0.45 |
| contig [2246] | GH991501.1 | glutathione S-transferase 16 (Helicoverpa armigera) | ACU09495.1 | −2.59 ± 0.57 |
| contig [0012] | GH987677.1 | microsomal glutathione transferase (Heliothis virescens) | ADH16761.1 | −2.19 ± 0.13 |
| ECB-C-03_D08 | GH992802.1 | cytochrome P450 monooxygenase cyp6ab4 (Bombyx mandarina) | NP_001073135.1 | −2.70 ± 0.08 |
| contig [5080] | GH996933.1 | cytochrome P450 monooxygenase cyp4m5(Bombyx mori) | NP_001103833.1 | 2.81 ± 0.11 |
| J-ECB-21_A02 | GH988690.1 | aliphatic nitrilase (Bombyx mori) | NP_001165388.1 | −2.43 ± 0.43 |
| Lipid metabolism | ||||
| J-ECB-35_D11 | GH990122.1 | alkaline ceramidase-like isoform 1 (Bombus terrestris) | XP_003393007.1 | −3.09 ± 0.37 |
| contig [0029] | GH998728.1 | acidic lipase (Helicoverpa armigera) | AFI64313.1 | −2.08 ± 0.04 |
| contig [0140] | GH998810.1 | neutral lipase (Helicoverpa armigera) | AFI64310.1 | −4.47 ± 0.11 |
| contig [1081] | GH998825.1 | neutral lipase (Helicoverpa armigera) | AFI64314.1 | −2.58 ± 0.05 |
| Lipid metabolism | ||||
| contig [1486] | GH997709.1 | C-5 sterol desaturase erg32-like (Bombyx mori) | XP_004922936.1 | −4.96 ± 0.13 |
| contig [1897] | GH997709.1 | C-5 sterol desaturase-like (Acyrthosiphon pisum) | XP_001947459.1 | −4.48 ± 0.02 |
| J-ECB-11_B07 | GH988922.1 | fatty acid-binding protein, muscle-like isoform 2 (Nasonia vitripennis) | XP_001608053.1 | −2.47 ± 0.10 |
| Carbohydrate metabolism | ||||
| contig [4242] | GH998158.1 | alpha-amylase 2 (Diatraea saccharalis) | AAP97393.1 | −2.21 ± 0.03 |
| contig [4425] | GH988573.1 | enolase (Antheraea pernyi) | ADO40102.1 | −2.36 ± 0.25 |
| ECB-V-12_H04 | GH995176.1 | enolase (Spodoptera litura) | AGQ53952.1 | −2.26 ± 0.02 |
| ECB-28_F02 | GH999466.1 | glucose phosphate dehydrogenase (Axia margarita) | ADW85328.1 | −3.05 ± 0.27 |
| contig [5232] | GH987506.1 | glycoside hydrolases (Aedes aegypti) | XP_001659854.1 | −2.31 ± 0.04 |
| contig [4123] | GH990084.1 | glucose and ribitol dehydrogenase-like (Bombyx mori) | XP_004922759.1 | −2.28 ± 0.10 |
| ECB-V-05_G12 | GH994609.1 | UDP-glycosyltransferase UGT33J1 (Helicoverpa armigera) | AEW43118.1 | −2.27 ± 0.07 |
| ECB-V-08_G03 | GH994852.1 | UDP-glycosyltransferase UGT33F1 (Helicoverpa armigera) | AEW43115.1 | −2.25 ± 0.07 |
| ECB-12_E11 | GH998082.1 | UDP-glycosyltransferase UGT40K1 (Bombyx mori) | AEW43171.1 | −3.62 ± 0.27 |
| ECB-V-19_F07 | GH995711.1 | glycosyltransferase 2 (Chilo suppressalis) | AGG36457.1 | −2.42 ± 0.02 |
| ECB-V-22_H08 | GH995978.1 | UDP-glycosyltransferase UGT40K1 (Bombyx mori) | AEW43171.1 | −2.80 ± 0.07 |
| Amino acid metabolism | ||||
| contig [4515] | GH987646.1 | gamma-glutamyl hydrolase A-like (Bombyx mori) | XP_004931467.1 | −2.50 ± 0.03 |
| J-ECB-24_G10 | GH990570.1 | methyltransferase (Mesobuthus caucasicus) | CAE53466.1 | 2.14 ± 0.03 |
| contig [5690] | GH988024.1 | farnesoic acid O-methyltransferase (Bombyx mori) | AGS17914.1 | 2.92 ± 0.07 |
| contig [1237] | GH989714.1 | farnesoic acid O-methyltransferase (Bombyx mori) | AGS17915.1 | 2.90 ± 0.05 |
| J-ECB-30_A09 | GH987906.1 | farnesoic acid O-methyltransferase (Bombyx mori) | AGS17914.1 | 2.91 ± 0.08 |
| contig [5679] | GH988679.1 | phosphoserine aminotransferase (Antheraea pernyi) | ADO79970.1 | −2.18 ± 0.08 |
| ECB-09_B04 | GH997795.1 | asparagine synthetase (Bombyx mori) | NP_001037414.1 | −2.38 ± 0.29 |
| Gut chitin metabolism | ||||
| contig [0188] | GH997506.1 | chitinase (Ostrinia nubilalis)chtinase 8 (Drosophila melanogaster) | ADB85578.1 | −2.74 ± 0.23 |
| ECB-V-28_H03 | GH996480.1 | chitin synthase (Ostrinia furnacalis) | ABB97082.1 | 2.16 ± 0.02 |
| ECB-C-05_D05 | GH992955.1 | glucosamine-fructose-6-phosphate aminotransferase 2 (Culex quinquefasciatus) | XP_001848160.1 | −2.02 ± 0.01 |
| Other metabolic enzymes | ||||
| contig [0077] | GH998660.1 | caboxypeptidase 4 (Mamestra configurata) | ACN69214.1 | −2.25 ± 0.04 |
| contig [0009] | GH992549.1 | carboxypeptidase (Bombyx mori) | AFD99126.1 | −2.19 ± 0.05 |
| contig [0019] | GH998697.1 | plasma glutamate carboxypeptidase, partial (Spodoptera exigua) | AFM38216.1 | −3.16 ± 0.19 |
| J-ECB-33_G12 | GH989302.1 | juvenile hormone epoxide hydrolase-like protein 1 (Bombyx mori) | NP_001159617.1 | −3.00 ± 0.06 |
| contig [0557] | GH998460.1 | juvenile hormone epoxide hydrolase (Spodoptera exigua) | ABD85119.1 | −2.17 ± 0.02 |
| contig [1953] | GH997798.1 | NADP-dependent oxidoreductase (Bombyx mori) | NP_001091765.1 | −3.27 ± 0.09 |
| contig [3531] | GH995654.1 | aldo-keto reductase (Aedes aegypti) | XP_001648461.1 | −2.23 ± 0.03 |
| contig [4521] | GH989023.1 | aldo-keto reductase (Bombyx mori) | ADQ89807.1 | −4.40 ± 0.60 |
| J-ECB-37_E05 | GH991174.1 | oxidoreductase (Acromyrmex echinatior) | EGI66780.1 | −2.37 ± 0.05 |
| contig [4410] | GH994481.1 | methionine-R-sulfoxide reductase B1-like isoform X2 (Bombyx mori) | XP_004924661.1 | −2.46 ± 0.03 |
| contig [3814] | GH994966.1 | alcohol dehydrogenase (Bombyx mori) | ADM32152.1 | −2.07 ± 0.03 |
| gi_133906638 | EL929475.1 | retinol dehydrogenase 11-like (Bombyx mori) | XP_004926801.1 | −3.61 ± 0.62 |
| contig [5542] | GH997904.1 | acetyltransferase 1 (Danaus plexippus) | EHJ65205.1 | −2.98 ± 0.27 |
| J-ECB-39_F07 | GH992097.1 | cytidylate kinase (Bombyx mori) | NP_001040356.1 | −2.73 ± 0.06 |
| BM2_M13R_B12 | GH992538.1 | estradiol 17-beta-dehydrogenase 8-like isoform X1 (Bombyx mori) | XP_004928638.1 | −2.09 ± 0.07 |
| Anti-bacterial related protein | ||||
| J-ECB-60_D07 | GH987186.1 | antibacterial protein (Heliothis virescens) | ACI02333.1 | 2.84 ± 0.07 |
| gi_133905829 | EL928679.1 | hinnavin II antibacterial peptides (Pieris rapae) | AAT94287.1 | 7.13 ± 0.22 |
| contig [2223] | GH996406.1 | peptidoglycan recognition protein C (Ostrinia nubilalis) | ADU33186.1 | 5.04 ± 0.07 |
| Others | ||||
| contig [0347] | GH987380.1 | fatty acid binding protein 1 (Manduca sexta) | P31416.1 | 6.89 ± 0.35 |
| ECB-V-18_A08 | GH995588.1 | Fatty acid-binding protein 2 (Danaus plexippus) | EHJ79280.1 | −4.19 ± 0.32 |
| contig [0028] | GH999333.1 | cytochrome b5 (Helicoverpa armigera) | ADU02195.1 | −2.70 ± 0.41 |
| contig [2048] | GH994666.1 | cytochrome b561 domain-containing protein 2-like (Bombyx mori) | XP_004933387.1 | 2.26 ± 0.03 |
| contig [4527] | GH989618.1 | cytochrome b561 domain-containing protein 1-like (Bombyx mori) | XP_004928254.1 | −7.89 ± 0.34 |
| ECB-11_E06 | GH997996.1 | peroxisomal membrane protein 11C-like (Bombyx mori) | XP_004925254.1 | −3.51 ± 0.23 |
| gi_133907290 | EL930112.1 | interferon-induced very large GTPase 1-like (Danio rerio) | XP_005163746.1 | 4.37 ± 0.27 |
| contig [0566] | GH993617.1 | fatty acid binding protein (Spodoptera litura) | AEH16743.1 | −2.17 ± 0.03 |
| contig [1640] | GH991677.1 | fatty acid-binding protein, adipocyte-like (Bombyx mori) | XP_004930401.1 | −2.99 ± 0.14 |
| contig [2896] | GH987914.1 | lipid storage droplet protein 2 (Manduca sexta) | AEJ33049.1 | 2.28 ± 0.10 |
| contig [0407] | GH997662.1 | sensory appendage protein 3 (Manduca sexta) | AAF16707.1 | −17.04 ± 3.80 |
| J-ECB-08_B02 | GH991953.1 | putative chemosensory protein (Sesamia inferens) | AGY49266.1 | −9.26 ± 0.49 |
| ECB-19_G03 | GH998716.1 | nose resistant to fluoxetine protein 6-like (Bombyx mori) | XP_004929562.1 | 2.75 ± 0.01 |
| ECB-V-07_D03 | GH994735.1 | serine-rich adhesin for platelets-like (Ceratitis capitata) | XP_004536543.1 | 2.66 ± 0.05 |
| contig [5724] | EL928855.1 | silk protein P25 (Corcyra cephalonica) | ACX50393.1 | 33.34 ± 1.92 |
| contig [4952] | GH988655.1 | fibroin light chain (Haritalodes derogata) | AFS32690.1 | 53.91 ± 1.63 |
| ECB-02_H03 | GH997311.1 | saposin-like protein (Bombyx mori) | ADU03994.1 | −2.55 ± 0.35 |
| contig [5293] | GH997917.1 | trypsin inhibitor (Bombyx mori) | NP_001037044.1 | 90.23 ± 1.47 |
| contig [5386] | GH996141.1 | leukocyte surface antigen CD53-like isoform X5 (Bombyx mori) | XP_004926002.1 | 2.33 ± 0.06 |
| ECB-C-04_H06 | GH992916.1 | tetraspanin D107 (Plutella xylostella) | BAD52262.1 | 2.25 ± 0.03 |
| contig [5414] | GH997359.1 | polyubiquitin-C-like isoform X1 (Musca domestica) | XP_005179902.1 | 2.74 ± 0.09 |
| contig [1085] | GH992931.1 | larvae cuticle protein (Choristoneura fumiferana) | AFC88812.1 | −4.92 ± 0.36 |
| J-ECB-12_F09 | GH989631.1 | globin 1 (Bombyx mori) | NP_001136083.1 | −2.54 ± 0.40 |
| J-ECB-29_G03 | GH992468.1 | IST1 homolog (Bombyx mori) | XP_004931988.1 | 2.18 ± 0.07 |
| J-ECB-32_D06 | GH988574.1 | tetratricopeptide repeat protein 27-like (Bombyx mori) | XP_004930370.1 | −2.44 ± 0.01 |
| J-ECB-47_A02 | GH990851.1 | hepatocyte growth factor-regulated tyrosine kinase substrate-like (Bombyx mori) | XP_004932480.1 | 3.09 ± 0.62 |
| Others | ||||
| gi_133905779 | EL928629.1 | pantetheinase (Mamestra configurata) | AEA76314.1 | −3.36 ± 0.13 |
| ECB-19_B09 | GH998666.1 | vanin-like protein 2-like (Bombyx mori) | XP_004928912.1 | 2.56 ± 0.03 |
| J-ECB-14_H06 | GH990255.1 | circadian clock-controlled protein (Harpegnathos saltator) | EFN85083.1 | 2.27 ± 0.10 |
| ECB-V-26_F03 | GH996285.1 | Histone H3c (Culex quinquefasciatus) | XP_001862696.1 | −2.04 ± 0.02 |
| J-ECB-07_A03 | GH991471.1 | circadian clock-controlled protein-like (Bombyx mori) | XP_004932669.1 | 5.02 ± 0.46 |
| J-ECB-39_H09 | GH992207.1 | extracellular domains-containing protein CG31004-like isoform X2 (Bombyx mori) | XP_004925419.1 | 2.15 ± 0.02 |
| ECB-V-25_C10 | GH996179.1 | conserved hypothetical protein (Culex quinquefasciatus) | XP_001845252.1 | 2.29 ± 0.08 |
2.2. Transcriptional Responses of Genes Potentially Involved in Protoxin Activation or Degradation

2.3. Transcriptional Responses of Genes Potentially Involved in Toxin Binding
2.4. Transcriptional Responses of Genes Potentially Involved in Larval Defense
3. Materials and Methods
3.1. European Corn Borer
3.2. Determination of Median Lethal Concentration of Cry1Ab Protoxin in 7-Day Bioassay
3.3. Cry1Ab Protoxin Bioassay
3.4. Microarray Analysis
3.5. Validation of Expression Changes by RT-qPCR
| Gene name | Primer sequences | Product size (bp) | EST ID |
|---|---|---|---|
| Trypsin-like serine protease | GGACAGTTCTCTGAGCAGTTAC | 109 | contig [4786] |
| ACAGCATGTTGTCAGTGATGG | |||
| Trypsin-like serine protease | ATTCTCAACAACAGGGCTATTTTG | 148 | contig [3704] |
| TGTAGTCAGGGTGGTTAATGATTC | |||
| Trypsin-like serine protease | GCATCATACCCGTCACATCTAC | 148 | contig [0770] |
| GTGAAGTTGCCGTACTGAGTC | |||
| Trypsin precursor | GCCAGCATTACACCTTCCG | 128 | contig [0243] |
| TCGCAGTTCTCGTAGTAAGAC | |||
| Silk gland derived trypsin serine protease | CACAAAGTCCTGGAGGAAGATTC | 125 | ECB-C-18-B11 |
| GTTCACGCCTGTCTGTTGC | |||
| Chymotrypsin-like serine protease | GGTGCTTGTTAGTATGTT | 116 | contig [0389] |
| AAACTTCTTTAATTGCTCAG | |||
| Chymotrypsin-like serine protease | ATAGAGCACCCGAATTACAACG | 123 | contig [1207] |
| GTAGGTTTGCGAGCCAGTG | |||
| Chymotrypsin-2 | CCCCTTCGTCCACGCTAG | 123 | contig [5740] |
| GTCACACCAACCAAGAGTCTC | |||
| Aminopeptidase N | TTCCAAACACATTTTCTTG | 118 | contig [4776] |
| AAGCGTATTGTCCTCTAT | |||
| Aminopeptidase N | CAGTAGCGATAACATCAC | 183 | contig [4879] |
| CCAGTCAAGTCTTCTCTA | |||
| Aminopeptidase N | GTCAACGAAATTGTCATC | 109 | ECB-V-05-D12 |
| AGTCATATTCTGGCTGTA | |||
| Cadherin-like protein | CTATGTGTTCTCAATCCAA | 75 | J-ECB-25-B09 |
| TCGTCGATGTTGACTATC | |||
| Alkaline phosphatase | CGGATTATCTGCTGGGTTTATTTG | 79 | contig [5858] |
| AGTGTGGGCTCGGTAACG |
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Showers, W.B.; Witkowski, J.F.; Mason, C.E.; Calvin, D.D.; Higgins, R.A.; Dively, G.P. European Corn Borer Development and Management; North Central Regional Extension Publication, No. 327; Iowa State University: Ames, IA, USA, 1989. [Google Scholar]
- Clive, J. Global Status of Commercialized Biotech/GM Crops; International Service for the Acquisition of Agri-biotech Application (ISAAA): Ithaca, NY, USA, 2010. [Google Scholar]
- Munkvold, G.P.; Desjardins, A.E. Fumonisins in maize: Can we reduce their occurrence? Plant Dis. 1997, 81, 556–565. [Google Scholar] [CrossRef]
- Witkowski, J.F.; Wedberg, J.L.; Seffey, K.L.; Sloderbeck, P.E.; Siegfried, B.D.; Rice, M.E.; Pilcher, L.C.; Onstad, D.W.; Mason, C.E.; Lewis, L.C.; et al. Bt Corn and European Corn Borer- Long Term Success through Resistance Management; North Central Regional Extension Publication: Ames, IA, USA, 1997. [Google Scholar]
- Hutchison, W.D.; Burkness, E.C.; Mitchell, P.D.; Moon, R.D.; Leslie, T.W.; Fleischer, S.J.; Abrahamson, M.; Hamilton, K.L.; Steffey, K.L.; Gray, M.E.; et al. Areawide suppression of European corn borer with Bt maize reaps savings to non-Bt maize growers. Science 2010, 330, 222–225. [Google Scholar]
- Soberón, M.; Pardo, L.; Muñóz-Garay, C.; Sánchez, J.; Gómez, I.; Porta, H.; Bravo, A. Pore formation by Cry toxins. Adv. Exp. Med. Biol. 2010, 677, 127–142. [Google Scholar] [CrossRef]
- Zhang, X.; Candas, M.; Griko, N.B.; Taussig, R.; Bulla, L.A., Jr. A mechanism of cell death involving an adenylyl cyclase/PKA signaling pathway is induced by the Cry1Ab toxin of Bacillus thuringiensis. Proc. Natl. Acad. Sci. USA 2006, 103, 9897–9902. [Google Scholar]
- Vachon, V.; Laprade, R.; Schwartz, J.L. Current models of the mode of action of Bacillus thuringiensis insecticidal crystal proteins: A critical review. J. Invertebr. Pathol. 2012, 111, 1–12. [Google Scholar] [CrossRef]
- Siqueira, H.A.; González-Cabrera, J.; Ferré, J.; Flannagan, R.; Siegfried, B.D. Analyses of Cry1Ab binding in resistant and susceptible strains of the European corn borer, Ostrinia nubilalis (Hubner) (Lepidoptera: Crambidae). Appl. Environ. Microbiol. 2006, 72, 5318–5324. [Google Scholar]
- Huang, F.; Andow, D.A.; Buschman, L.L. Success of the high-dose/refuge resistance management strategy after 15 years of Bt crop use in North America. Entomol. Exp. Appl. 2011, 140, 1–16. [Google Scholar] [CrossRef]
- Tabashnik, B.E.; Brévault, T.; Carrière, Y. Insect resistance to Bt crops: Lessons from the first billion acres. Nat. Biotechnol. 2013, 31, 510–521. [Google Scholar] [CrossRef]
- Gassmann, A.J.; Petzold-Maxwell, J.L.; Keweshan, R.S.; Dunbar, M.W. Field-evolved resistance to Bt maize by western corn rootworm. PLoS One 2011, 6, e22629. [Google Scholar]
- Bravo, A.; Soberon, M. How to cope with insect resistance to Bt toxins? Trends Biotechnol. 2008, 26, 573–579. [Google Scholar] [CrossRef]
- Yao, J.; Buschman, L.L.; Oppert, B.; Khajuria, C.; Zhu, K.Y. Characterization of cDNAs encoding serine proteases and their transcriptional responses to Cry1Ab protoxin in the gut of Ostrinia nubilalis larvae. PLoS One 2012, 7, e44090. [Google Scholar]
- Li, H.; Oppert, B.; Higgins, R.A.; Huang, F.; Zhu, K.Y.; Buschman, L.L. Comparative analysis of proteinase activities of Bacillus thuringiensis-resistant and -susceptible Ostrinia nabilalis (Lepidoptera:crambidae). Insect Biochem. Mol. Biol. 2004, 34, 753–762. [Google Scholar] [CrossRef]
- Li, H.; Oppert, B.; Higgins, R.A.; Huang, F.; Buschman, L.L.; Gao, J.-R.; Zhu, K.Y. Characterization of cDNAs encoding three trypsin-like proteases and mRNA quantitative analysis in Bt-resistant and -susceptible strains of Ostrinia nubilalis. Insect Biochem. Mol. Biol. 2005, 35, 847–860. [Google Scholar] [CrossRef]
- Khajuria, C.; Zhu, Y.C.; Chen, M.-S.; Buschman, L.L.; Higgins, R.A.; Yao, J.; Cresop, A.L.B.; Siegfried, B.D.; Muthukrishnan, S.; Zhu, K.Y. Expressed sequence tags from larval gut of the European corn borer (Ostrinia nubilalis): Exploring candidate genes potentially involved in Bacillus thuringiensis toxicity and resistance. BMC Genomics 2009, 10, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Kao, C.-Y.; Los, F.C.O.; Huffman, D.L.; Wachi, S.; Kloft, N.; Husmann, M.; Karabrahimi, V.; Schwartz, J.-L.; Bellier, A.; Ha, C.; et al. Global functional analyses of cellular responses to pore-forming toxins. PLoS Pathog. 2011, 7, e1001314. [Google Scholar] [CrossRef]
- Côté, J.C.; Otvos, I.S.; Schwartz, J.-L.; Vincent, C. Gene expression response of the spruce budworm, Choristoneura fumiferana, after exposure to various doses of Bacillus thuringiensis Cry1Ab toxin using microarray technology. In Proceedings of the 6th Pacific Rim Conference on the Biotechnology of Bacillus thuringiensis and its Environmental Impact, Victoria, BC, USA, 30 October 2005.
- David, J.-P.; Strode, C.; Vontas, J.; Nikou, D.; Vaughan, A.; Pignatelli, P.; Louis, C.; Hemingway, J.; Ranson, H. The Anopheles gambiae detoxification chip: A highly specific microarray to study metabolic-based insecticide resistance in malaria vectors. Proc. Natl. Acad. Sci. USA 2005, 102, 4080–4084. [Google Scholar] [CrossRef]
- Van Munster, M.; Prefonaine, G.; Meunier, L.; Elias, M.; Mazza, A.; Brousseau, R.; Masson, L. Altered gene expression in Choristoneura fumiferana and Manduca sexta in response to sublethal intoxication by Bacillus thuringiensis Cry1Ab toxin. Insect Mol. Biol. 2007, 16, 25–35. [Google Scholar] [CrossRef]
- Huang, L.; Cheng, T.; Xu, P.; Cheng, D.; Fang, T.; Xia, Q. A genome-wide survey for host response of silkworm, Bombyx mori during pathogen Bacillus bombyseptieus infection. PLoS One 2009, 4, e8098. [Google Scholar]
- Chroma, C.T.; Surewicz, W.K.; Carey, P.R.; Pozsgay, M.; Raynor, T.; Kaplan, H. Unusual proteolysis of the protoxin and toxin from Bacillus thuringiensis: Structural implications. Eur. J. Biochem. 1990, 189, 523–527. [Google Scholar] [CrossRef]
- Bravo, A.; Gill, S.S.; Soberón, M. Mode of action of Bacillus thuringiensis Cry and Cyt toxins and their potential for insect control. Toxicon 2007, 49, 423–435. [Google Scholar] [CrossRef]
- Oppert, B. Protease interactions with Bacillus thuringiensis insecticidal toxins. Arch. Insect Biochem. Physiol. 1999, 42, 1–12. [Google Scholar] [CrossRef]
- Oppert, B.; Kramer, K.J.; Johnson, D.; Upton, S.J.; Mcgaughey, W.H. Luminal proteinases from Plodia interpunctella and the hydrolysis of Bacillus thuringiensis CryIA(c) protoxin. Insect Biochem. Mol. Biol. 1996, 26, 571–583. [Google Scholar]
- Oppert, B.; Kramer, K.J.; Beeman, R.W.; Johnson, D.; McGaughey, W.H. Proteinase-mediated insect resistance to Bacillus thuringiensis toxins. J. Biol. Chem. 1997, 272, 23473–23476. [Google Scholar] [CrossRef]
- Hua, G.; Jurat-Fuentes, J.L.; Adang, M.J. Bt-R1a extracellular cadherin repeat 12 mediates Bacillus thuringiensis Cry1Ab binding and cytotoxicity. J. Biol. Chem. 2004, 279, 28051–28056. [Google Scholar]
- Flannagan, R.D.; Yu, C.G.; Mathis, T.E.; Shi, X.; Siqueira, H.A.; Siegfied, B.D. Identification, cloning and expression of a Cry1Ab cadherin receptor from European corn borer, Ostrinia nubilalis (Hubner) (Lepidoptera: Crabidae). Insect Biochem. Mol. Biol. 2005, 35, 33–40. [Google Scholar] [CrossRef]
- Knight, P.J.K.; Crickmore, N.; Ellar, D.J. The receptor for Bacillus thuringiensis Cry1Ac delta-endotoxin in the brush border membrane of the lepidopteran Manduca sexta is aminpeptidase N. Mol. Microbiol. 1994, 11, 429–436. [Google Scholar] [CrossRef]
- Xu, L.N.; He, K.L.; Wang, Z.Y.; Bai, S.X. Cloning and sequencing of aminopeptidase N genes from Asian corn borer susceptible and resistant to Cry1Ab toxin. J. Agric. Biotechnol. 2011, 19, 164–170. [Google Scholar]
- Yang, Y.; Zhu, Y.C.; Ottea, J.; Husseneder, C.; Leonard, B.R.; Abel, C.; Huang, F. Molecular characterization and RNA interference of three midgut aminopeptidase N isozymes from Bacillus thuringiensis-susceptible and -resistant strains of sugarcane borer, Diatraea saccharalis. Insect Biochem. Mol. Biol. 2010, 40, 592–603. [Google Scholar] [CrossRef]
- Rajagopal, R.; Sivakumar, S.; Agrawal, P.; Malbotra, P.; Bhatnagar, R.K. Silencing of midgut aminopeptidase N of Spodoptera litura by double-stranded RNA establishes role as Bacillus thuringiensis toxin receptor. J. Biol. Chem. 2002, 277, 46849–46851. [Google Scholar]
- Herrero, S.; Gechev, T.; Bakker, P.L.; Moar, W.J.; de Maagd, R.A. Bacillus thuringiensis Cry1Ca-resistant Spodoptera exigua lacks expression of one of four aminopeptidase N genes. BMC Genomics 2005, 6, e96. [Google Scholar] [CrossRef]
- Khajuria, C.; Buschman, L.L.; Chen, M.-S.; Siegfried, B.D.; Zhu, K.Y. Identification of a novel aminopeptidase P-like gene (OnAPP) possibly involved in Bt toxicity and resistance in a major corn pest (Ostrinia nubilalis). PLoS One 2011, 6, e23983. [Google Scholar]
- Broderick, N.A.; Raffa, K.F.; Handelsman, J. Midgut bacteria required for Bacillus thuringiensis insecticidal activity. Proc. Natl. Acad. Sci. USA 2006, 103, 15196–15199. [Google Scholar] [CrossRef]
- Khajuria, C.; Buschman, L.L.; Chen, M.-S.; Zurek, L.; Zhu, K.Y. Characterization of six antibacterial response genes from the European corn borer (Ostrinia nubilalis) larval gut and their expression in response to bacterial challenge. J. Insect Physiol. 2011, 57, 345–355. [Google Scholar] [CrossRef]
- Chen, C.-S.; Bellier, A.; Kao, C.-Y.; Yang, Y.-L.; Chen, H.-D.; Los, F.C.; Aroian, R.V. WWP-1 is a novel modulator of the DAF-2 insulin-like signaling network involved in pore-forming toxin cellular defenses in Caenorhabditis elegans. PLoS One 2010, 5, e9494. [Google Scholar]
- Khajuria, C.; Buschman, L.L.; Chen, M.-S.; Muthukrishnan, S.; Zhu, K.Y. A gut-specific chitinase gene essential for regulation of chitin content of peritrophic membrane and growth of Ostrinia nubilalis larvae. Insect Biochem. Mol. Biol. 2010, 40, 621–629. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, J.; Zhu, K.Y. Chitosan/double-stranded RNA nanoparticle-mediated RNA interference to silence chitin synthase genes through larval feeding in the African malaria mosquito (Anopheles gambiae). Insect Mol. Biol. 2010, 19, 683–693. [Google Scholar] [CrossRef]
- Kato, N.; Dasgupta, R.; Smartt, C.T.; Christensen, B.M. Glucosamine:fructose-6-phosphate aminotransferase: Gene characterization, chitin biosynthesis and peritrophic matrix formation in Aedes aegypti. Insect Mol. Biol. 2002, 11, 207–216. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, H.; Li, S.; Zhu, K.Y.; Ma, E.; Zhang, J. Characterization of a midgut-specific chitin synthase gene (LmCHS2) responsible for biosynthesis of chitin of peritrophic matrix in Locusta migratoria. Insect Biochem. Mol. Biol. 2012, 42, 902–910. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, J.; Park, Y.; Zhu, K.Y. Identification and characterization of two chitin synthase genes in African malaria mosquito, Anopheles gambiae. Insect Biochem. Mol. Biol. 2012, 42, 674–682. [Google Scholar] [CrossRef]
- Lee, M.K.; Milne, R.E.; Ge, A.Z.; Dean, D.H. Location of Bombyx mori receptor binding region on a Bacillus thuringiensis endotoxin. J. Biol. Chem. 1992, 267, 3115–3121. [Google Scholar]
- Dalman, M.R.; Deeter, A.; Nimishakavi, G.; Duan, Z.-H. Fold change and p-value cutoffs significantly alter microarray interpretations. BMC Bioinform. 2012, 13, S11. [Google Scholar]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yao, J.; Buschman, L.L.; Lu, N.; Khajuria, C.; Zhu, K.Y. Changes in Gene Expression in the Larval Gut of Ostrinia nubilalis in Response to Bacillus thuringiensis Cry1Ab Protoxin Ingestion. Toxins 2014, 6, 1274-1294. https://doi.org/10.3390/toxins6041274
Yao J, Buschman LL, Lu N, Khajuria C, Zhu KY. Changes in Gene Expression in the Larval Gut of Ostrinia nubilalis in Response to Bacillus thuringiensis Cry1Ab Protoxin Ingestion. Toxins. 2014; 6(4):1274-1294. https://doi.org/10.3390/toxins6041274
Chicago/Turabian StyleYao, Jianxiu, Lawrent L. Buschman, Nanyan Lu, Chitvan Khajuria, and Kun Yan Zhu. 2014. "Changes in Gene Expression in the Larval Gut of Ostrinia nubilalis in Response to Bacillus thuringiensis Cry1Ab Protoxin Ingestion" Toxins 6, no. 4: 1274-1294. https://doi.org/10.3390/toxins6041274