Zearalenone, an Estrogenic Mycotoxin, Is an Immunotoxic Compound
Abstract
:1. Introduction
2. Results
2.1. The Dose-Range Study
2.2. Food Consumption, Body Weight Gain and Haematologic Parameters
| Groups | Food consumption (g) | Body weight gain (g) | RBC (×106/mm3) | WBC (×103/mm3) | Ht (%) | Hg (g/dL) | MCH (pg) | MCV (mm3) |
|---|---|---|---|---|---|---|---|---|
| Control | 448.2 ± 26.1 | 41.8 ± 13.1 | 7.2 ± 0.2 | 6.1 ± 0.5 | 40.8 ± 1.3 | 15.0 ± 0.3 | 20.9 ± 0.4 | 56.8 ± 0.5 |
| 3.0 mg/kg | 390.3 ± 35.7 *** | 22.7 ± 13.9 **,† | 8.2 ±0.3 | 5.8 ± 0.6 | 45.5 ± 1.9 | 16.3 ± 0.6 | 19.9 ± 0.3 | 55.2 ± 0.5 |
| Pair-fed a | - | 35.1 ± 7.8 | 7.8 ± 0.2 | 5.8 ± 0.4 | 44.0 ± 1.3 | 15.8 ± 0.4 | 20.3 ± 0.1 | 57.0 ± 0.4 |
2.3. Evaluation of Lymphoid Organs and Their Cells
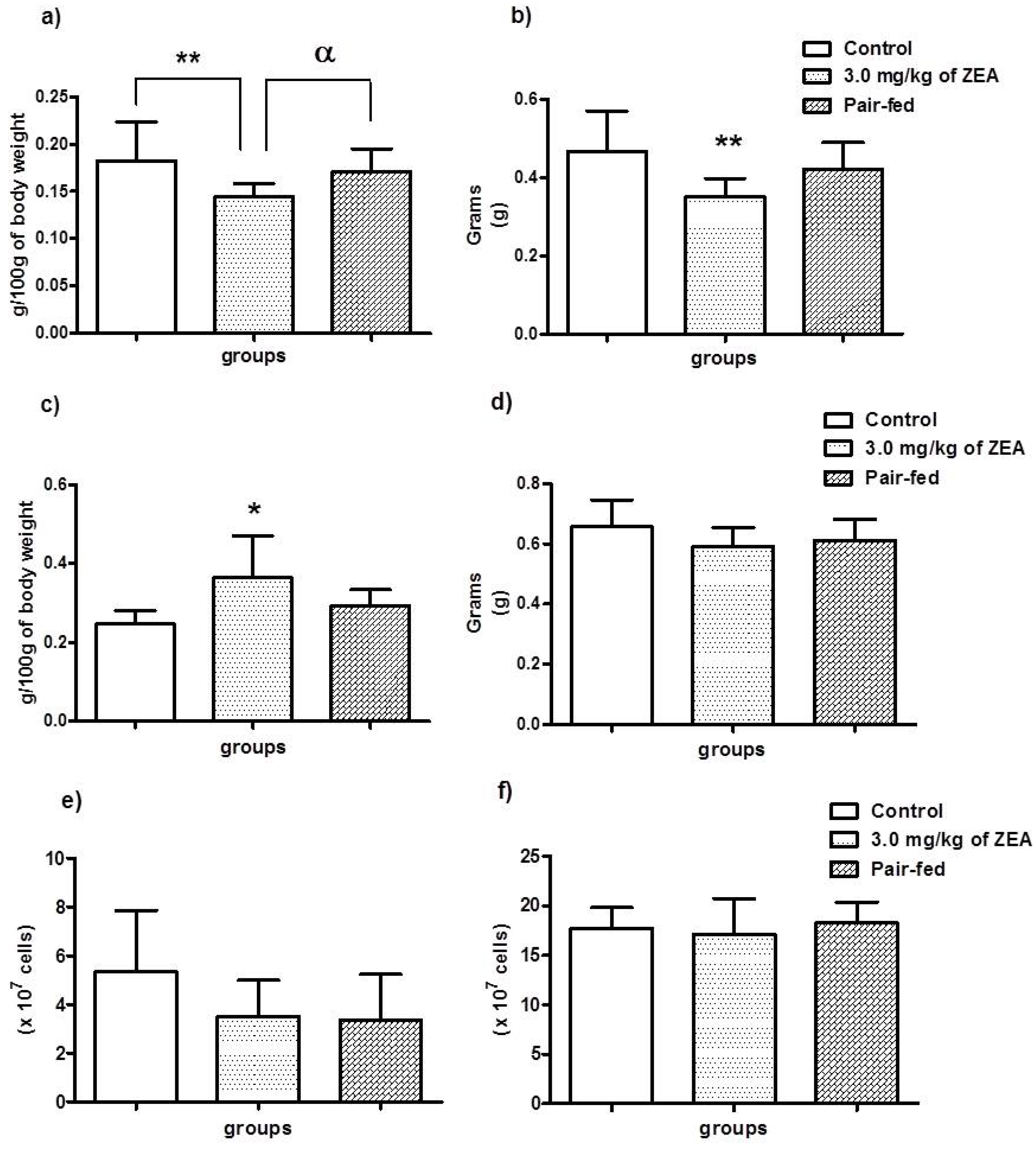

| Treatment (n = 10/group) | Thymus (% total lymphocytes) | Spleen (% total lymphocytes) | |||||
|---|---|---|---|---|---|---|---|
| CD3-CD4+CD8+ | CD3+CD4+CD8+ | CD3+CD4+CD8- | CD3+CD4-CD8+ | IgM+CD45R+ | CD3+CD4+ | CD3+CD8+ | |
| Control | 87.9 ± 6.2 | 13.4 ± 3.0 | 67.0 ± 4.4 | 10.0 ± 2.0 | 29.5 ± 2.9 | 43.7 ± 3.1 | 43.9 ± 3.4 |
| 3.0 mg/kg | 90.2 ± 1.6 | 17.7 ± 3.7 | 59.5 ± 4.3* | 12.2 ± 1.2* | 22.1 ± 3.4* | 43.4 ± 7.6 | 44.3 ± 6.3 |
| Pair-feda | 91.0 ± 1.4 | 18.1 ± 3.9 | 61.6 ± 5.5 | 11.7 ± 1.2 | 29.1 ± 3.2 | 41.6 ± 4.4 | 44.7 ± 3.8 |
2.4. Acquire Immune Responses
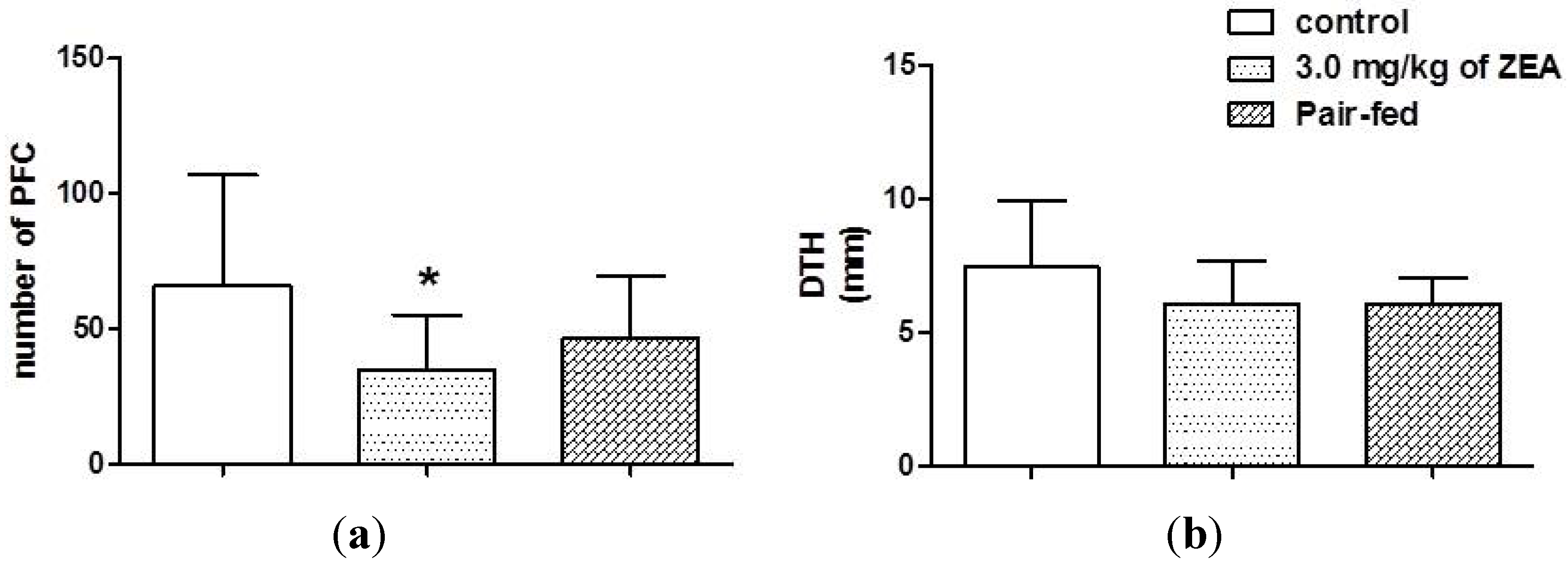
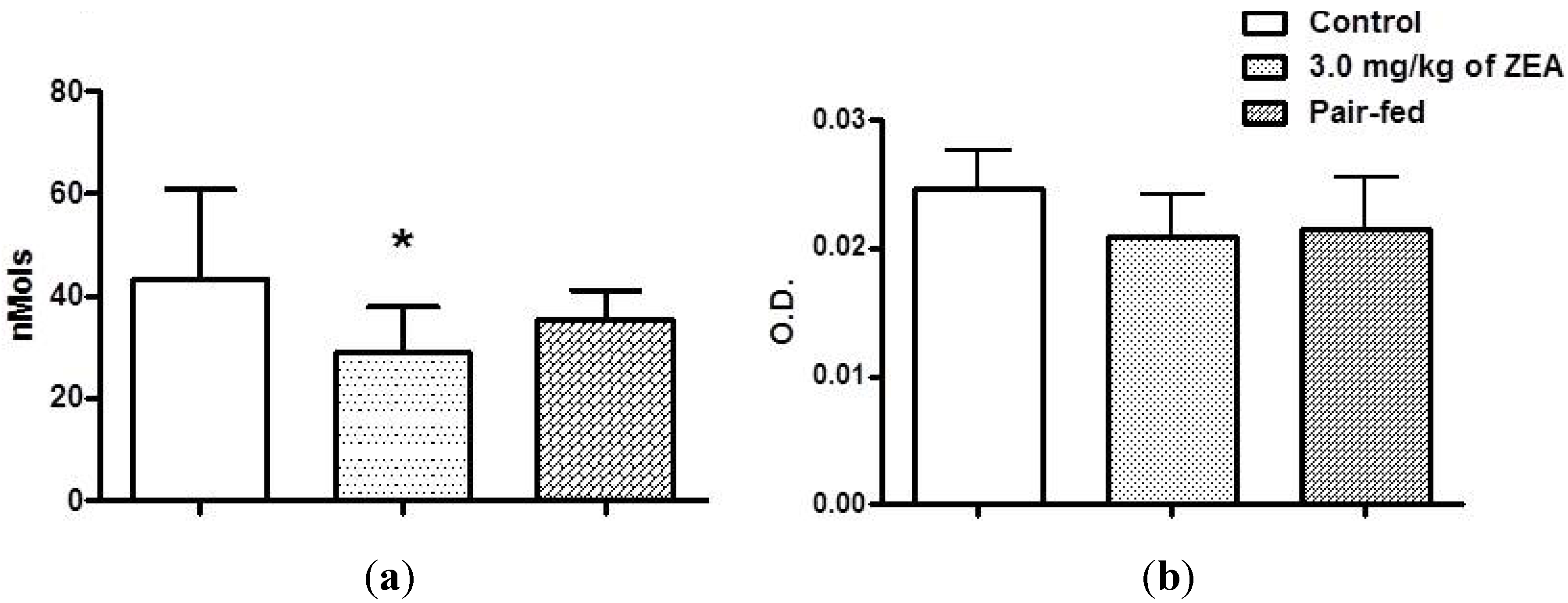
2.5. Innate Immunity: Macrophage Activity
3. Discussion
4. Experimental Section
4.1. Animals and Treatment Regimen
4.2. Reagents
4.3. Haematology, Immune Organ and Lymphoid Cell Analysis
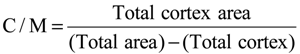
4.4. Humoral Immune Response
4.5. Delayed Type Hypersensitivity (DTH)
4.6. Innate Immunity: Macrophage Activity
4.7. Statistical Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Caldwell, R.W.; Tuite, J.; Stob, M.; Baldwin, R. Zearalenone production by Fusarium species. Appl. Microbiol. 1970, 20, 31–34. [Google Scholar]
- Kuiper-Goodman, T.; Scott, P.M.; Watanabe, H. Risk assessment of the mycotoxin zearalenone. Regul. Toxicol. Pharmacol. 1987, 7, 253–306. [Google Scholar] [CrossRef]
- Hestbjerg, H.; Nielsen, K.F.; Thrane, U.; Elmholt, S. Production of trichothecenes and other secondary metabolites by Fusarium culmorum and Fusarium equiseti on common laboratory media and a soil organic matter agar: An ecological interpretation. J. Agric. Food Chem. 2002, 50, 7593–7599. [Google Scholar] [CrossRef]
- Zinedine, A.; Soriano, J.M.; Moltó, J.C.; Mañes, J. Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: An oestrogenic mycotoxin. Food Chem. Toxicol. 2007, 45, 1–18. [Google Scholar] [CrossRef]
- Collection of Occurrence Data of Fusarium Toxins in Food Andassessment of Dietary Intake by the Population of EU Member States; SCOOP European Project, task 3.2.10. 2003. Available online: http://ec.europa.eu/food/fs/scoop/task3210.pdf (accessed on 17 October 2013).
- Almeida, M.I.; Almeida, N.G.; Carvalho, K.L.; Gonçalves, G.A.; Silva, C.N.; Santos, E.A.; Garcia, J.C.; Vargas, E.A. Co-occurrence of aflatoxins B1, B2, G1 and G2, ochratoxin A, zearalenone, deoxynivalenol, and citreoviridin in rice in Brazil. Food Addit. Contam. Part A Chem. Anal. Control Exp. Risk. Assess 2012, 29, 694–703. [Google Scholar] [CrossRef]
- Queiroz, V.A.V.; de Oliveira Alves, G.L.; da Conceição, R.R.P.; Guimarães, L.J.M.; Mendes, S.M.; de Aquino Ribeiro, P.E.; da Costa, R.V. Occurrence of fumonisins and zearalenone in maize stored in family farm in Minas Gerais, Brazil. Food Control 2012, 28, 83–86. [Google Scholar] [CrossRef]
- Pleadin, J.; Sokolović, M.; Perši, N.; Zadravec, M.; Jaki, V.; Vulić, A. Contamination of maize with deoxynivalenol and zearalenone in Croatia. Food Control 2012, 28, 94–98. [Google Scholar] [CrossRef]
- Juan, C.; Ritieni, A.; Mañes, J. Occurrence of Fusarium mycotoxins in Italian cereal and cereal products from organic farming. Food Chem. 2013, 141, 1747–1755. [Google Scholar] [CrossRef]
- Lovelace, C.E.; Nyathi, C.B. Estimation of the fungal toxins, zearalenone and aflatoxin, contaminating opaque maize beer in Zambia. J. Sci. Food Agric. 1977, 28, 288–292. [Google Scholar] [CrossRef]
- Scott, P.M. Mycotoxins Transmitted into beer from contaminated grains during brewing. J. AOAC Int. 1996, 79, 875–882. [Google Scholar]
- Ryu, D.; Hanna, M.A.; Bullerman, L.B. Stability of zearalenone during extrusion of corn grits. J. Food Prot. 1999, 62, 1482–1484. [Google Scholar]
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef]
- European Food Safety Authority. Scientific opinion on the risk for public health related to the presence of zearalenone in food. Eur. Food Saf. Auth. J. 2011, 9, 1–124. [Google Scholar]
- Sáenz de Rodríguez, C.A. Environmental hormone contamination in Puerto Rico. N. Engl. J. Med. 1984, 310, 1741–1742. [Google Scholar] [CrossRef]
- Sáenz de Rodríguez, C.A.; Bongiovanni, A.M.; Conde de Borrego, L. An epidemic of precocious development in Puerto Rican children. J. Pediatr. 1985, 107, 393–396. [Google Scholar] [CrossRef]
- Szüts, P.; Mesterházy, Á.; Falkay, G.Y.; Bartók, T. Early thelarche symptoms in children and their relations to zearalenone contamination in foodstuffs. Cereal Res. Commun. 1997, 25, 429–441. [Google Scholar]
- Massart, F.; Meucci, V.; Saggese, G.; Soldani, G. High growth rate of girls with precocious puberty exposed to estrogenic mycotoxins. J. Pediatr. 2008, 152, 690–695. [Google Scholar] [CrossRef]
- Metzler, M.; Pfeiffer, E.; Hildebrand, A.A. Zearalenone and its metabolites as endocrine disrupting chemicals. World Mycotoxin J. 2010, 3, 385–401. [Google Scholar] [CrossRef]
- Lang, T.J. Estrogen as immunomodulator. Clin. Immunol. 2004, 113, 224–230. [Google Scholar] [CrossRef]
- Ádori, M.; Kiss, E.; Barad, Z.; Barabás, K.; Kiszely, E.; Schneider, A.; Sziksz, E.; Ábrahám, I.M.; Matkó, J.; Sármay, G. Estrogen augments the T cell-dependent but not the T-independent immune response. Cell. Mol. Life Sci. 2010, 67, 1661–1674. [Google Scholar] [CrossRef]
- Cunningham, M.; Gilkeson, G. Estrogen receptors in immunity and autoimmunity. Clin. Rev. Allergy Immunol. 2011, 40, 66–73. [Google Scholar] [CrossRef]
- Cutolo, M.; Sulli, A.; Straub, R.H. Estrogen metabolism and autoimmunity. Autoimmun. Rev. 2012, 11, A460–A464. [Google Scholar] [CrossRef]
- Ben Salah-Abbès, J.; Abbès, S.; Houas, Z.; Abdel-Wahhab, M.A.; Oueslati, R. Zearalenone induces immunotoxicity in mice: Possible protective effects of radish extract (Raphanussativus). J. Pharm. Pharmacol. 2008, 60, 761–770. [Google Scholar] [CrossRef]
- Choi, B.K.; Cho, J.H.; Jeong, S.H.; Shin, H.S. Zearalenone affects immune-related parameters in lymphoid organs and serum of rats vaccinated with porcine Parvovirus vaccine. Toxicol. Res. 2012, 28, 279–288. [Google Scholar] [CrossRef]
- Marin, D.E.; Taranu, I.; Burlacu, R.; Manda, G.; Motiu, M.; Neagoe, I.; Dragomir, C.; Stancu, M.; Calin, L. Effects of zearalenone and its derivatives on porcine immune response. Toxicol. In Vitro 2011, 25, 1981–1988. [Google Scholar] [CrossRef]
- Cameron, J.K.; Bursian, S.J.; Aulerich, R.J. The effect of zearalenone on reproductive parameters of female mink. Toxicologist 1989, 9, 216. [Google Scholar]
- Kordic, B.; Pribicevic, S.; Muntanola-Cvetkovic, M.; Nikolic, P.; Nikolic, B. Experimental study of the effects of known quantities of zearalenone on swine reproduction. J. Environ. Pathol. Toxicol. Oncol. 1992, 11, 53–55. [Google Scholar]
- Underhill, K.L.; Rotter, B.A.; Thompson, B.K.; Prelusky, D.B.; Trenholm, H.L. Effectiveness of cholestyramine in the detoxification of zearalenone as determined in mice. Bull. Environ. Contam. Toxicol. 1995, 54, 128–134. [Google Scholar]
- Stob, M.; Baldwin, R.S.; Tuite, J.; Andres, F.N.; Gillette, K.G. Isolation of an anabolic, uterotrophic compound from corn infected with Gibberella zeae. Nature 1962, 196, 1318. [Google Scholar]
- Cunningham-Rundles, S.; McNeeley, D.F.; Moon, A. Mechanisms of nutrient modulation of the immune response. J. Allergy Clin. Immunol. 2005, 115, 1119–1129. [Google Scholar] [CrossRef]
- Drewett, R.F. Oestrous and dioestrous components of the ovarian inhibition on hunger in the rat. Anim. Behav. 1973, 21, 772–780. [Google Scholar] [CrossRef]
- Roesch, D.M. Effects of selective estrogen receptor agonists on food intake and body weight gain in rats. Physiol. Behav. 2006, 87, 39–44. [Google Scholar] [CrossRef]
- Tarttelin, M.F.; Gorski, R.A. The effects of ovarian steroids on food and water intake and body weight in the female rat. Acta Endocrinol. Copenh 1973, 72, 551–568. [Google Scholar]
- Wade, G.N. Gonadal hormones and behavioral regulation of body weight. Physiol. Behav. 1972, 8, 523–534. [Google Scholar] [CrossRef]
- Mauvais-Jarvis, F.; Clegg, D.J.; Hevener, A.L. The role of estrogens in control of energy balance and glucose homeostasis. Endocr. Rev. 2013, 34, 309–338. [Google Scholar] [CrossRef]
- Kuiper, G.; Lemmen, J.; Carlsson, B.; Corton, J.; Safe, S.; van der Saag, P.; van der Burg, B.; Gustafsson, J. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor β. Endocrinology 1998, 139, 4252–4263. [Google Scholar]
- Maaroufi, K.; Chekir, L.; Creppy, E.E.; Ellouz, F.; Bacha, H. Zearalenone induces modifications of haematological and biochemical parameters in rats. Toxicon 1996, 34, 535–540. [Google Scholar] [CrossRef]
- Abbès, S.; Ouanes, Z.; Ben Salah-Abbès, J.; Houas, Z.; Oueslati, R.; Bacha, H.; Othman, O. The protective effect of hydrated sodium calcium aluminosilicate against haematological, biochemical and pathological changes induced by zearalenone in mice. Toxicon 2006, 47, 567–574. [Google Scholar] [CrossRef]
- Novotny, E.A.; Raveche, E.S.; Sharrow, S.; Ottinger, M.; Steinberg, A.D. Analysis of thymocyte subpopulations following treatment with sex hormones. Clin. Immunol. Immunopathol. 1983, 28, 205–217. [Google Scholar] [CrossRef]
- Clarke, A.G.; Kendall, M.D. Histological changes in the thymus during mouse pregnancy. Thymus 1989, 14, 65–78. [Google Scholar]
- Chandra, R.K. Protein-energy malnutrition and immunological responses. J. Nutr. 1992, 122, 597–600. [Google Scholar]
- Prentice, A.M. The thymus: A barometer of malnutrition. Br. J. Nutr. 1999, 81, 345–347. [Google Scholar]
- Kawashima, I.; Seiki, K.; Sakabe, K.; Ihara, S.; Akatsuka, A.; Katsumata, Y. Localization of estrogen receptors and estrogen receptor-mRNA in female mouse thymus. Thymus 1992, 20, 115–121. [Google Scholar]
- Luster, M.I.; Boorman, G.A.; Korach, K.S.; Dietert, M.P.; Hong, L. Mechanisms of estrogen-induced myelotoxicity: Evidence of thymic regulation. Int. J. Immunopharmacol. 1984, 6, 287–297. [Google Scholar]
- Cunningham, M.; Gilkeson, G. Estrogen receptors in immunity and autoimmunity. Clin. Rev. Allergy Immunol. 2011, 40, 66–73. [Google Scholar] [CrossRef]
- Pearse, G. Normal structure, function and histology of the thymus. Toxicol. Pathol. 2006, 34, 504–514. [Google Scholar] [CrossRef]
- Nüßlein, H.G.; Huppertz, H.I.; Massenkeil, G.; Mengel, E. Splenomegaly—An important cardinal symptomautoren. Aktuel. Rheumatol. 2009, 34, 213–219. [Google Scholar] [CrossRef]
- Stimson, W.H. Oestrogen and human T lymphocytes: Presence of specific receptors in the T-suppressor/cytotoxic subset. Scand. J. Immunol. 1988, 28, 345–350. [Google Scholar] [CrossRef]
- Stygar, D.; Westlund, P.; Eriksson, H.; Sahlin, L. Identification of wild type and variants of oestrogen receptors in polymorphonuclear and mononuclear leucocytes. Clin. Endocrinol. 2006, 64, 74–81. [Google Scholar] [CrossRef]
- Phiel, K.L.; Henderson, R.A.; Adelman, S.J.; Elloso, M.M. Differential estrogen receptor gene expression in human peripheral blood mononuclear cell populations. Immunol. Lett. 2005, 97, 107–113. [Google Scholar] [CrossRef]
- Zoller, A.L.; Kersh, G.J. Estrogen induces thymic atrophy by eliminating early thymic progenitors and inhibiting proliferation of β-selected thymocytes. J. Immunol. 2006, 176, 7371–7378. [Google Scholar]
- Medina, K.L.; Strasser, A.; Kincade, P.W. Estrogen influences the differentiation, proliferation, and survival of early B-lineage precursors. Blood 2000, 95, 2059–2067. [Google Scholar]
- Sammaritano, L.R. Pregnancy in rheumatic disease patients. J. Clin. Rheumatol. 2013, 19, 259–266. [Google Scholar] [CrossRef]
- Goemaere, S.; Ackerman, C.; Goethals, K.; de Keyser, F.; van der Straeten, C.; Verbruggen, G.; Mielants, H.; Veys, E.M. Onset of symptoms of rheumatoid arthritis in relation to age, sex and menopausal transition. J. Rheumatol. 1990, 17, 1620–1622. [Google Scholar]
- Engdahl, C.; Jochems, C.; Windahl, S.H.; Börjesson, A.E.; Ohlsson, C.; Carlsten, H.; Lagerquist, M.K. Amelioration of collagen-induced arthritis and immune-associated bone loss through signaling via estrogen receptor α, and not estrogen receptor β or G protein-coupled receptor 30. Arthritis Rheum. 2010, 62, 524–533. [Google Scholar]
- Mor, G.; Sapi, E.; Abrahams, V.M.; Rutherford, T.; Song, J.; Hao, X.-Y.; Muzaffar, S.; Kohen, F. Interaction of the estrogen receptors with the Fas ligand promoter in human monocytes. J. Immunol. 2003, 170, 114–122. [Google Scholar]
- Exon, J.H.; Bussiere, J.L.; Mather, G.G. Immunotoxicity testing in the rat: An improved multiple assay model. Int. J. Immunopharmacol. 1990, 12, 699–701. [Google Scholar] [CrossRef]
- Rabinovitch, M.; Destefano, M.J. Macrophage spreading in vitro: I. Inducers of spreading. Exp. Cell Res. 1973, 77, 323–334. [Google Scholar] [CrossRef]
- Rabinovitch, M.; Destefano, M.J. Macrophage spreading in vitro: II. Manganese and other metals as inducers or as co-factors for induced spreading. Exp. Cell Res. 1973, 79, 423–430. [Google Scholar] [CrossRef]
- Russo, M.; Teixeira, H.C.; Marcondes, M.C.G.; Barbuto, J.A.N. Superoxide independent hydrogen peroxide release by activated macrophages. Braz. J. Med. Biol. Res. 1989, 22, 1271–1273. [Google Scholar]
- Pick, E.; Mizel, D. Rapid microassays for the measurement of superoxide and hydrogen peroxide production by macrophages in culture sing an automatic enzyme immunoassay. J. Immunol. Methods 1981, 46, 211–226. [Google Scholar] [CrossRef]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tanneubaum, S.R. Analysis of nitrate, nitrite and [15N] nitrite in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hueza, I.M.; Raspantini, P.C.F.; Raspantini, L.E.R.; Latorre, A.O.; Górniak, S.L. Zearalenone, an Estrogenic Mycotoxin, Is an Immunotoxic Compound. Toxins 2014, 6, 1080-1095. https://doi.org/10.3390/toxins6031080
Hueza IM, Raspantini PCF, Raspantini LER, Latorre AO, Górniak SL. Zearalenone, an Estrogenic Mycotoxin, Is an Immunotoxic Compound. Toxins. 2014; 6(3):1080-1095. https://doi.org/10.3390/toxins6031080
Chicago/Turabian StyleHueza, Isis M., Paulo Cesar F. Raspantini, Leonila Ester R. Raspantini, Andreia O. Latorre, and Silvana L. Górniak. 2014. "Zearalenone, an Estrogenic Mycotoxin, Is an Immunotoxic Compound" Toxins 6, no. 3: 1080-1095. https://doi.org/10.3390/toxins6031080




