Earthworm-Derived Pore-Forming Toxin Lysenin and Screening of Its Inhibitors
Abstract
:1. Introduction
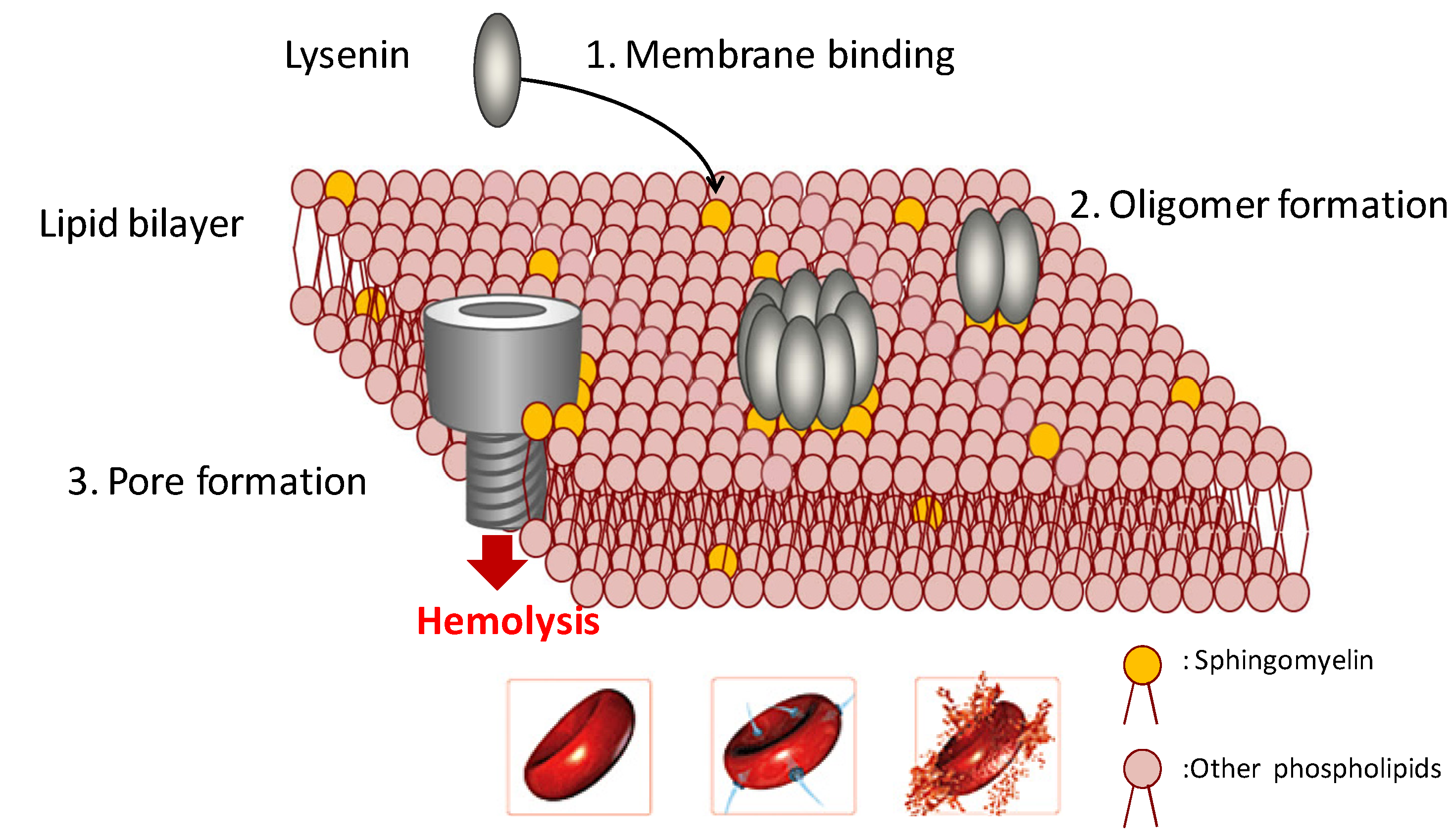
2. Structure of Lysenin and Induction of Hemolysis

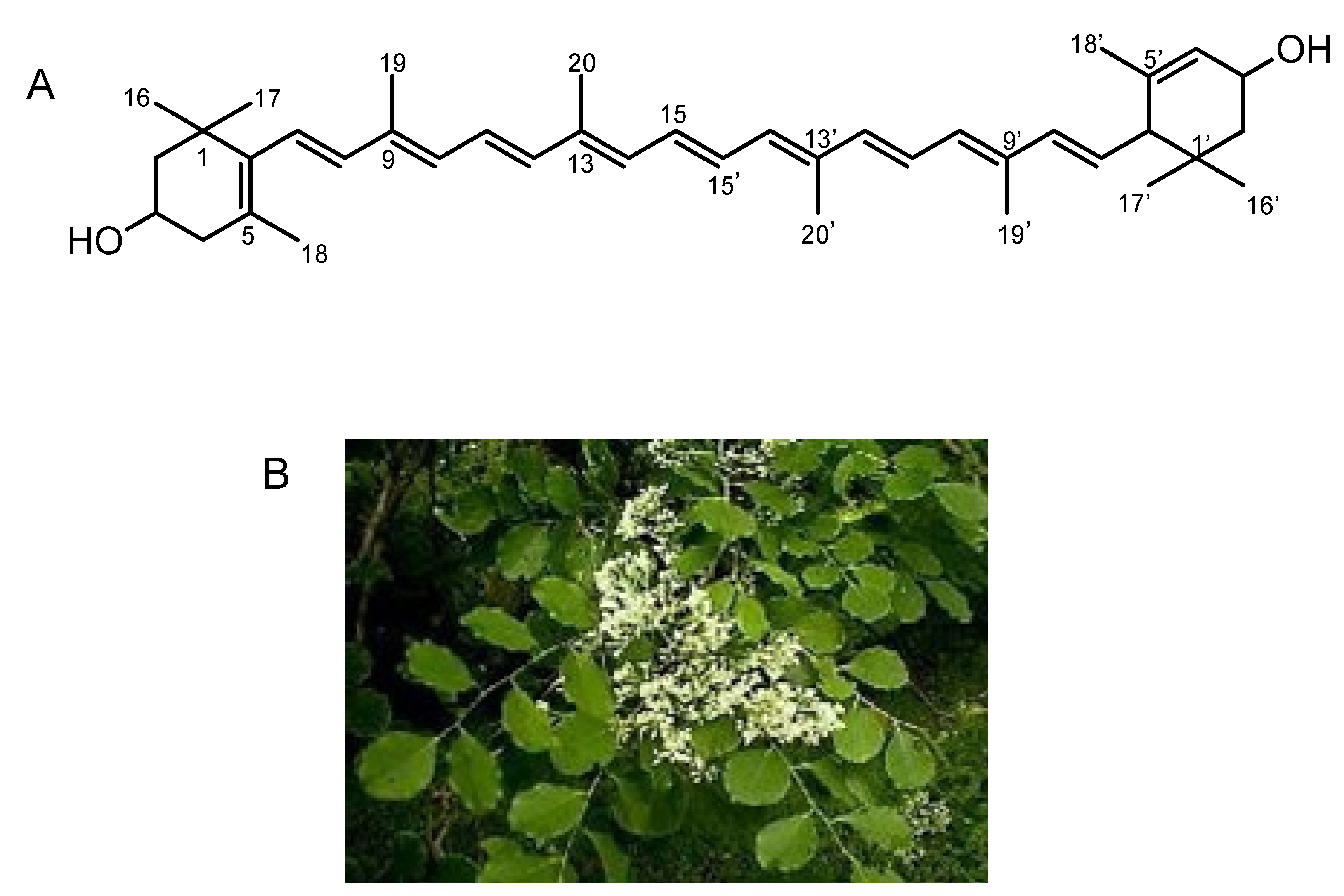
3. Isolation of All-E-Lutein as an Inhibitor of Lysenin-Induced Hemolysis
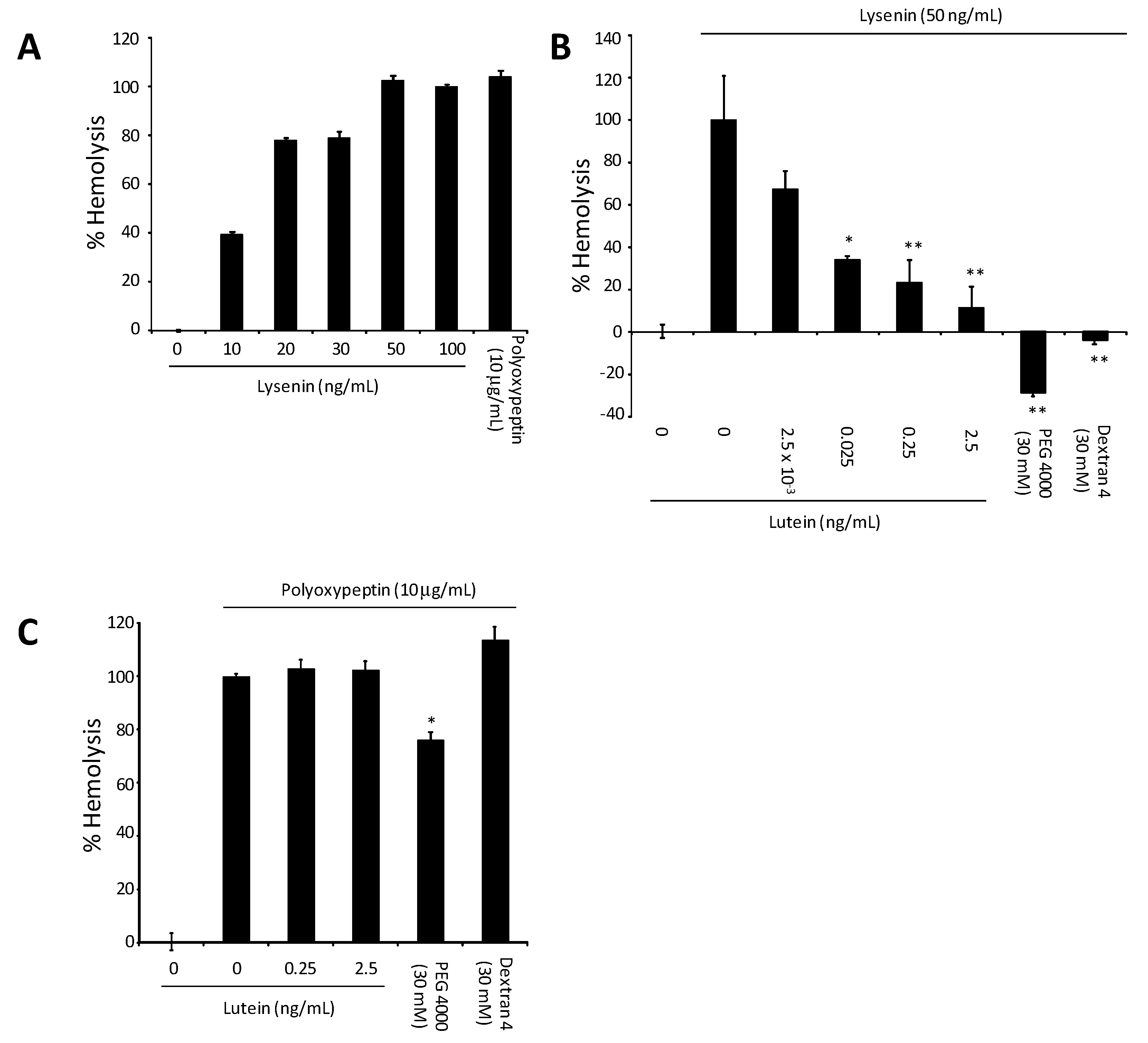
4. Lysenin-Induced Hemolysis and the Effect of All-E-Lutein
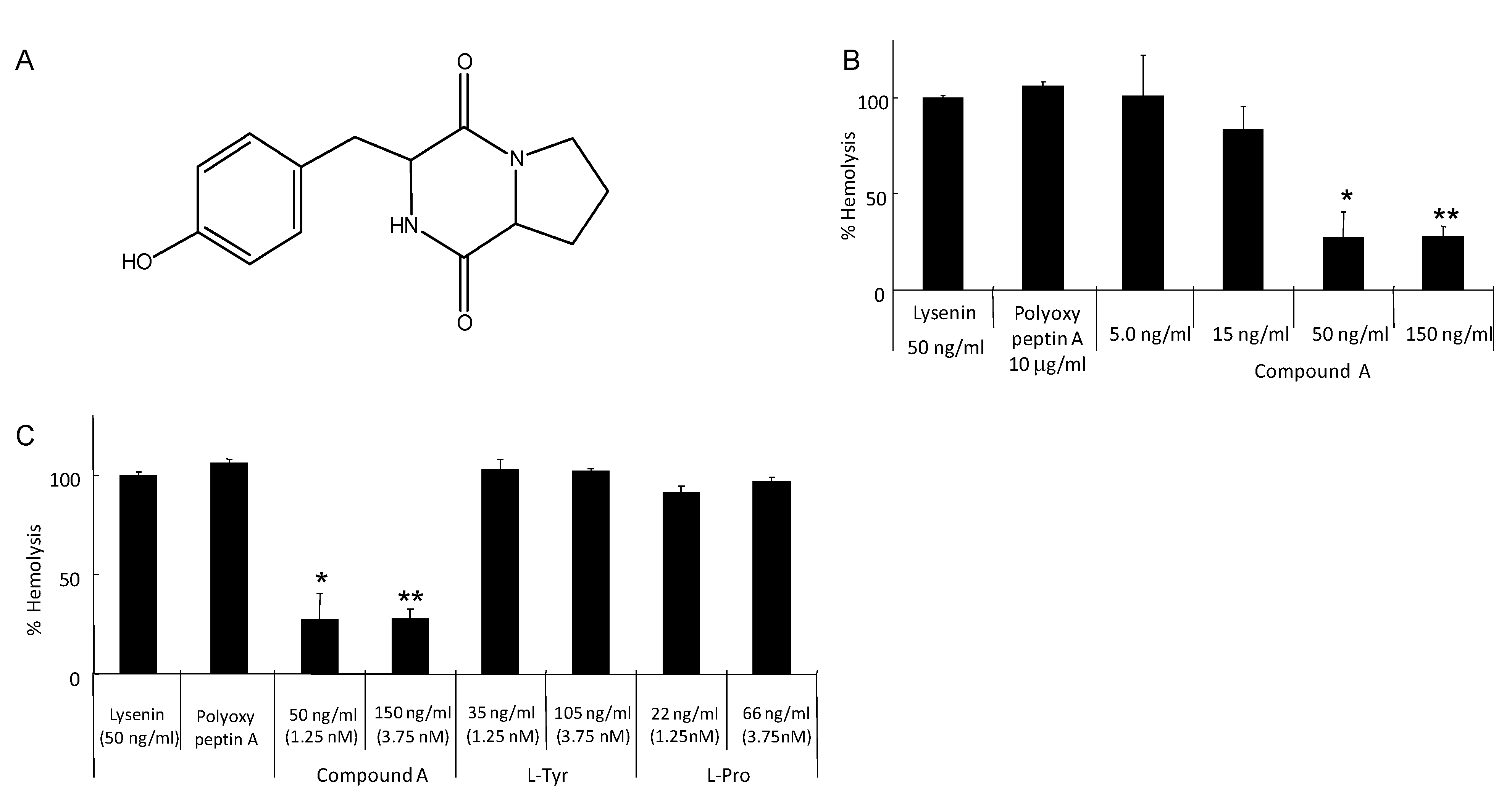
5. Isolation of Tyrosylproline Anhydride as an Inhibitor of Lysenin-Induced Hemolysis
6. Conclusions
Acknowledgments
Conflict of Interest
References
- Bashford, C.L. Pore-forming toxins: Attack and defence at the cell surface. Cell Biol. Mol. Lett. 2001, 6, 328–333. [Google Scholar]
- Bernheimer, A.W.; Rudy, B. Interactions between membranes and cytolytic peptides. Biochim. Biophys. Acta 1986, 864, 123–141. [Google Scholar] [CrossRef]
- Huffman, D.L.; Abrami, L.; Sasik, R.; Corbeil, J.; van der Goot, F.G.; Aroian, R.V. Mitogen-activated protein kinase pathways defend against bacterial pore-forming toxins. Proc. Natl. Acad. Sci. USA 2004, 101, 10995–11000. [Google Scholar]
- Aneiros, A.; Garateix, A. Bioactive peptides from marine sources: Pharmacological properties and isolation procedures. J. Chromatogr. B 2004, 803, 41–53. [Google Scholar] [CrossRef]
- Anderluh, G.; BarlicÏ, A.; Podlesek, Z.; MacÏek, P.; PungercÏar, J.; GubensÏek, F.; Zecchini, M.L.; Serra, M.D.; Menestrina, G. Cysteine-scanning mutagenesis of an eukaryotic pore-forming toxin. Eur. J. Biochem. 1999, 263, 128–136. [Google Scholar] [CrossRef]
- Sher, D.; Fishman, Y.; Zhang, M.; Lebendiker, M.; Gaathon, A.; Mancheno, J.M.; Zlotkin, E. Hydralysins, a new category of β-pore-forming toxins in cnidaria. J. Biol. Chem. 2005, 280, 22847–22855. [Google Scholar]
- Gilbert, R.J.C. Pore-forming toxins. Cell Mol. Life. Sci. 2002, 59, 832–844. [Google Scholar] [CrossRef]
- Shogomori, H.; Kobayashi, T. Lysenin: A sphingomyelin specific pore-formimg toxin. Biochim. Biophys. Acta 2008, 1780, 612–618. [Google Scholar] [CrossRef]
- Sekizawa, Y.; Kubo, T.; Kobayashi, H.; Nakajima, T.; Natori, S. Molecular cloning of cDNA for lysenin, a novel protein in the earthworm Eisenia foetida that causes contraction of rat vascular smooth muscle. Gene 1997, 191, 97–102. [Google Scholar] [CrossRef]
- Yamaji, A.; Sekizawa, Y.; Emoto, K.; Sakuraba, H.; Inoue, K.; Kobayashi, H.; Umeda, M. Lysenin, a novel sphingomyelin-specific binding protein. J. Biol. Chem. 1998, 273, 5300–5306. [Google Scholar]
- Procházková, P.; Silerová, M.; Felsberg, J.; Josková, R.; Beschin, A.; de baetselier, P.; Bilej, M. Relationship between hemolytic molecules in Eisenia fetida earthworms. Dev. Comp. Immunol. 2006, 30, 381–392. [Google Scholar] [CrossRef]
- Kobayashi, H.; Ohtomi, M.; Sekizawa, Y.; Ohta, N. Toxicity of coelomic fluid of the earthworm Eisenia foetida to vertebrates but not invertebrates: Probable role of sphingomyelin. Comp. Biochem. Physiol. Part C 2001, 128, 401–411. [Google Scholar] [CrossRef]
- Hanada, K.; Hara, T.; Fukasawa, M.; Yamaji, A.; Umeda, M.; Nishijima, M. Mammalian cell mutants resistant to a sphingomyelin-directed cytolysin genetic and biochemical evidence for complex formation of the LCB1 protein with the LCB2 Protein for serine palmitoyltransferase. J. Biol. Chem. 1998, 273, 33787–33794. [Google Scholar]
- Kobayashi, H.; Sekizawa, Y.; Aizu, M.; Umeda, M. Lethal and non-lethal responses of spermatozoa from a wide variety of vertebrates and invertebrates to lysenin, a protein from the coelomic fluid of the earthworm. J. Cell. Biol. 2000, 286, 538–549. [Google Scholar]
- Kobayashi, H.; Suzuki, H.; Ohta, N. Exfoliation of the epidermal cells and defecation by amphibian larvae in response to coelomic fluid and lysenin from the earthworm Eisenia foetida. Biomed. Res. 2006, 27, 169–181. [Google Scholar] [CrossRef]
- Opper, B.; Bognár, A.; Heidt, D.; Németh, P.; Engelmann, P. Revising lysenin expression of earthworm coelomocytes. Dev. Comp. Immunol. 2013, 39, 214–218. [Google Scholar] [CrossRef]
- De Colbis, L.; Sonnen, A.F.-P.; Morris, K.J.; Siebert, A.; Abrusci, P.; Plitzko, J.; Hodnik, V.; Leippe, M.; Volpi, E.; Anderluh, G.; Gilbert, R.J.C. Structures of lysenin reveal a shared evolutionary origin for pore-forming proteins and its mode of sphingomyelin recognition. Structure 2012, 20, 1498–1507. [Google Scholar] [CrossRef]
- Yamaji-Hasegawa, A.; Makino, A.; Baba, T.; Senoh, Y.; Kimura-Suda, H.; Sato, S.B.; Terada, N.; Ohno, S.; Kiyokawa, E.; Umeda, M.; et al. Oligomerization and pore Formation of a sphingomyelin-specific toxin, lysenin. J. Biol. Chem. 2003, 278, 22762–22770. [Google Scholar] [CrossRef]
- Kiyokawa, E.; Makino, A.; Ishii, K.; Otsuka, N.; Yamaji-Hasegawa, A.; Kobayashi, T. Recognition of sphingomyelin by lysenin and lysenin-related proteins. Biochemistry 2004, 43, 9766–9773. [Google Scholar] [CrossRef]
- Ishitsuka, R.; Kobayashi, T. Cholesterol and lipid/protein ratio control the oligomerization of a sphingomyelin-specific toxin, lysenin. Biochemistry 2007, 46, 1495–1502. [Google Scholar] [CrossRef]
- Ishitsuka, R.; Yamaji-Hasegawa, A.; Makino, A.; Hirabayashi, T.; Kobayashi, T. A lipid-specific toxin reveals heterogeneity of sphingomyelin-containing membranes. Biophys. J. 2004, 86, 296–307. [Google Scholar] [CrossRef]
- Ide, T.; Aoki, T.; Takeuchi, Y.; Yanagida, T. Lysenin forms a voltage-dependent channel in artificial lipid bilayer membranes. Biochem. Biophys. Res. Commun. 2006, 346, 288–292. [Google Scholar] [CrossRef]
- Hereć, M.; Gagoś, M.; Kulma, M.; Kwiatkowska, K.; Sobota, A.; Gruszecki, W.I. Secondary structure and orientation of the pore-forming toxin lysenin in a sphingomyelin-containing membrane. Biochim. Biophys. Acta 2008, 1778, 872–879. [Google Scholar]
- Niwa, Y.; Matsui, C.; Sukumwang, N.; Iinuma, H.; Ikeda, Y.; Koyano, T.; Kovitayakorn, T.; Simizu, S.; Umezawa, K. Inhibition of lysenin-induced hemolysis by all-E-lutein derived from the plant Dalbergia latifolia. Planta Med. 2012, 78, 957–961. [Google Scholar] [CrossRef]
- Dachtler, M.; Glaser, T.; Kohler, K.; Albert, K. Combined HPLC-MS and HPLC-NMR on-line coupling for the separation and determination of lutein and zeaxanthin stereoisomers in spinach and in Retina. Anal. Chem. 2001, 73, 667–674. [Google Scholar] [CrossRef]
- Khachik, F.; Englert, G.; Daitch, C.E.; Beecher, G.R.; Tonucci, L.H.; Lusby, W.R. Isolation and structural elucidation of the geometrical isomers of lutein and zeaxanthin in extracts from human plasma. J. Chromatogr. 1992, 582, 153–166. [Google Scholar]
- Mercadante, A.Z.; Steck, A.; Pfander, H. Carotenoids from Guava (Psidium guajaca L.): Isolation and structure elucidation. J. Agric. Food. Chem. 1999, 47, 145–151. [Google Scholar] [CrossRef]
- Sekine, N.; Ashitani, A.; Murayama, T.; Shibutani, S.; Hattori, S.; Takahashi, K. Bioactivity of latifolin and its derivatives against termites and fungi. J. Agric. Food. Chem. 2009, 57, 5707–5712. [Google Scholar] [CrossRef]
- Ekka, N.R.; Dixit, V.K. Ethno-pharmacognostical studies of medicinal plants of jashpur district (Chhattisgarh). Int. J. Green Pharm. 2007, 1, 2–4. [Google Scholar]
- Joy, P.P.; Thomas, J.; Mathew, S.; Skaria, B.P. Medicinal Plants; Kerala Agricultural University: Kerala, India, 1998; p. 191. [Google Scholar]
- Choi, E.M.; Hwang, J.K. Screening of indonesian medicinal plants for inhibitor activity on nitric oxide production of RAW264.7 cells and antioxidant activity. Fitoterapia 2005, 76, 194–203. [Google Scholar] [CrossRef]
- Stierle, A.C.; Cardellina, J.H., II; Stobel, G.A. Maculosin, a host-specific phytotoxin for spotted knapweed Alternaria alternata. Proc. Natl. Acad. Sci. USA 1988, 85, 8008–8011. [Google Scholar] [CrossRef]
- Umezawa, K.; Weinstein, B.; Shaw, W.V. Staphylococcal delta-hemolysin inhibits cellular binding of epidermal growth factor and induces arachidonic acid release. Biochem. Biophys. Res. Commun. 1980, 94, 625–629. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sukumwang, N.; Umezawa, K. Earthworm-Derived Pore-Forming Toxin Lysenin and Screening of Its Inhibitors. Toxins 2013, 5, 1392-1401. https://doi.org/10.3390/toxins5081392
Sukumwang N, Umezawa K. Earthworm-Derived Pore-Forming Toxin Lysenin and Screening of Its Inhibitors. Toxins. 2013; 5(8):1392-1401. https://doi.org/10.3390/toxins5081392
Chicago/Turabian StyleSukumwang, Neelanun, and Kazuo Umezawa. 2013. "Earthworm-Derived Pore-Forming Toxin Lysenin and Screening of Its Inhibitors" Toxins 5, no. 8: 1392-1401. https://doi.org/10.3390/toxins5081392



