Appearance of Planktothrix rubescens Bloom with [D-Asp3, Mdha7]MC–RR in Gravel Pit Pond of a Shallow Lake-Dominated Area
Abstract
:1. Introduction
2. Results
2.1. Physicochemical Parameters of the Study Site
| Variables | value | unit |
|---|---|---|
| Lake area | 5.2 | (Ha) |
| Mean depth | 3.2 | (m) |
| Max. depth | 7 | (m) |
| Lake volume | 1.6 × 105 | (m3) |
| Secchi transparency | 1.2 | (m) |
| pH | 8.34 | |
| Specific electrical conductivity | 820 | (µS cm−1) |
| COD(sMn) | 15.8 | (mg L−1) |
| TOC | 22.0 | (mg L−1) |
| DOC | 15.8 | (mg L−1) |
| Inorganic Nitrogen (IN) | 1953 | (µg L−1) |
| Soluble Reactive Phosphorus (SRP) | 3 | (µg L−1) |
| Total Nitrogen (TN) | 3125 | (µg L−1) |
| Total Phosphorus (TP) | 370 | (µg L−1) |

2.2. Morphology-Based Identification of the HAB Causing Organism
2.3. Molecular Phylogenetic Analyses
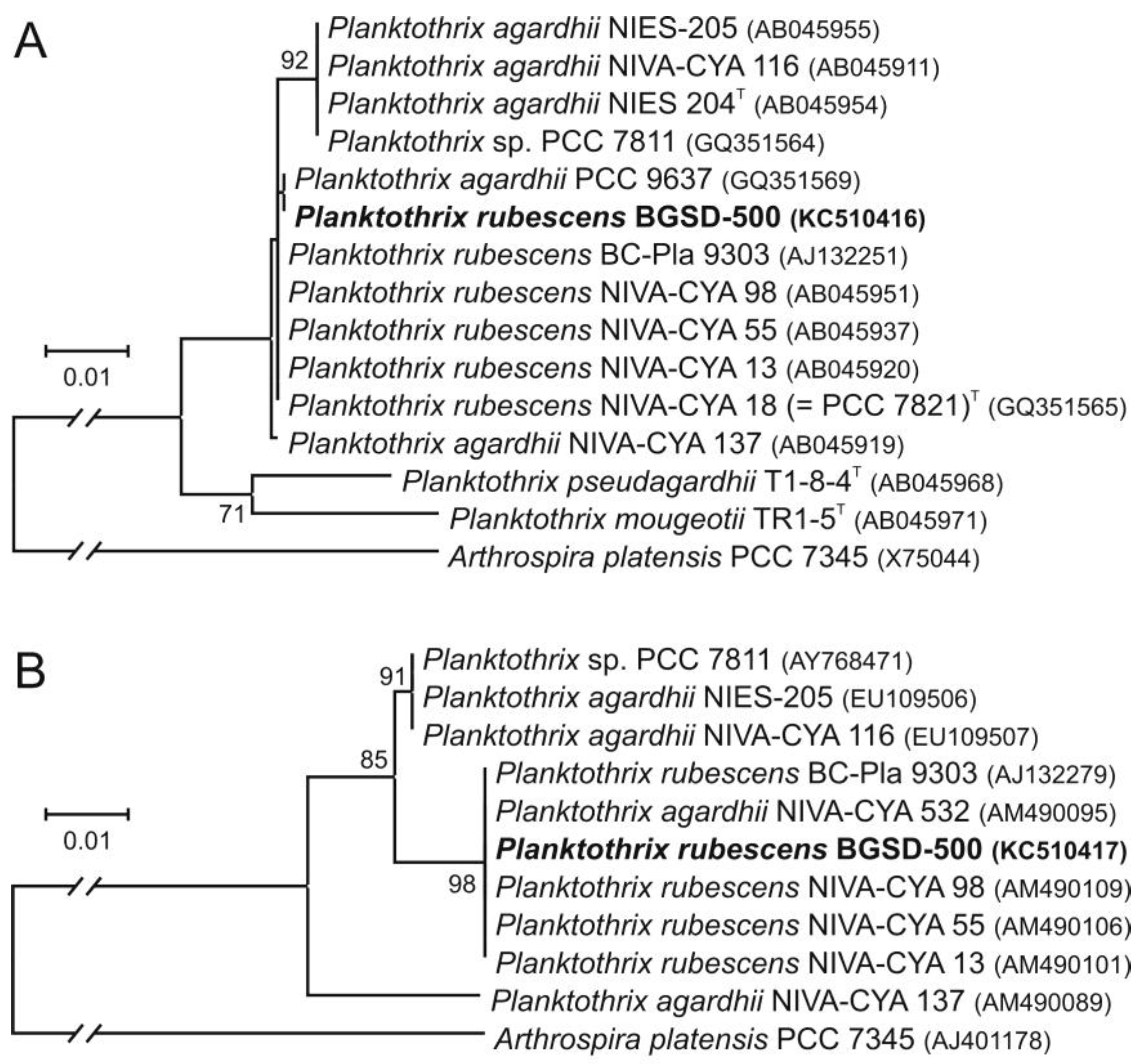
2.4. Identification of MC and Comparative Analysis of Bloom Sample and the Isolated P. rubescens Strain
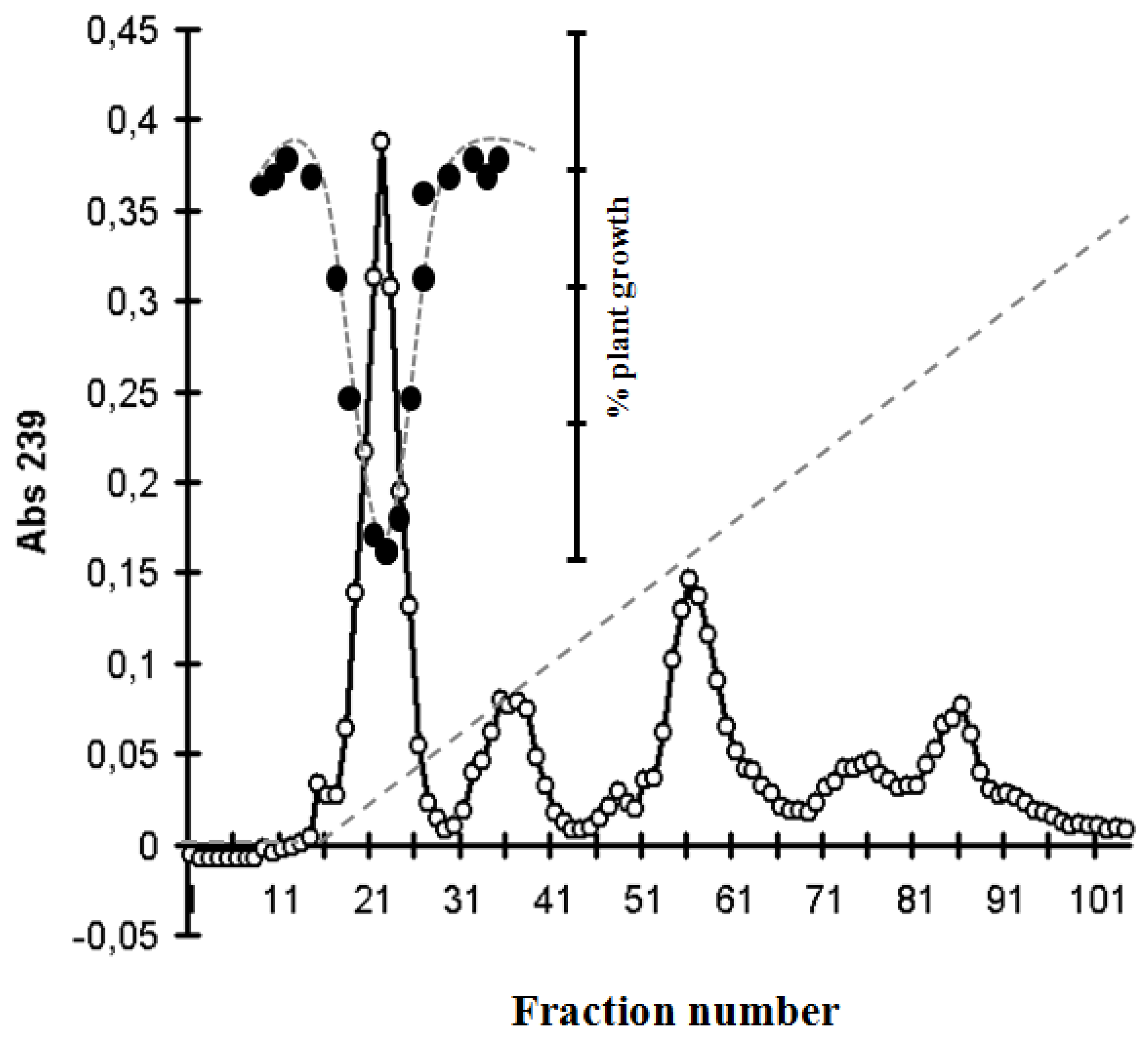

 -) on the growth of Sinapis alba etiolated seedlings (Blue-Green-Sinapis-Test).
-) on the growth of Sinapis alba etiolated seedlings (Blue-Green-Sinapis-Test).
 -) on the growth of Sinapis alba etiolated seedlings (Blue-Green-Sinapis-Test).
-) on the growth of Sinapis alba etiolated seedlings (Blue-Green-Sinapis-Test).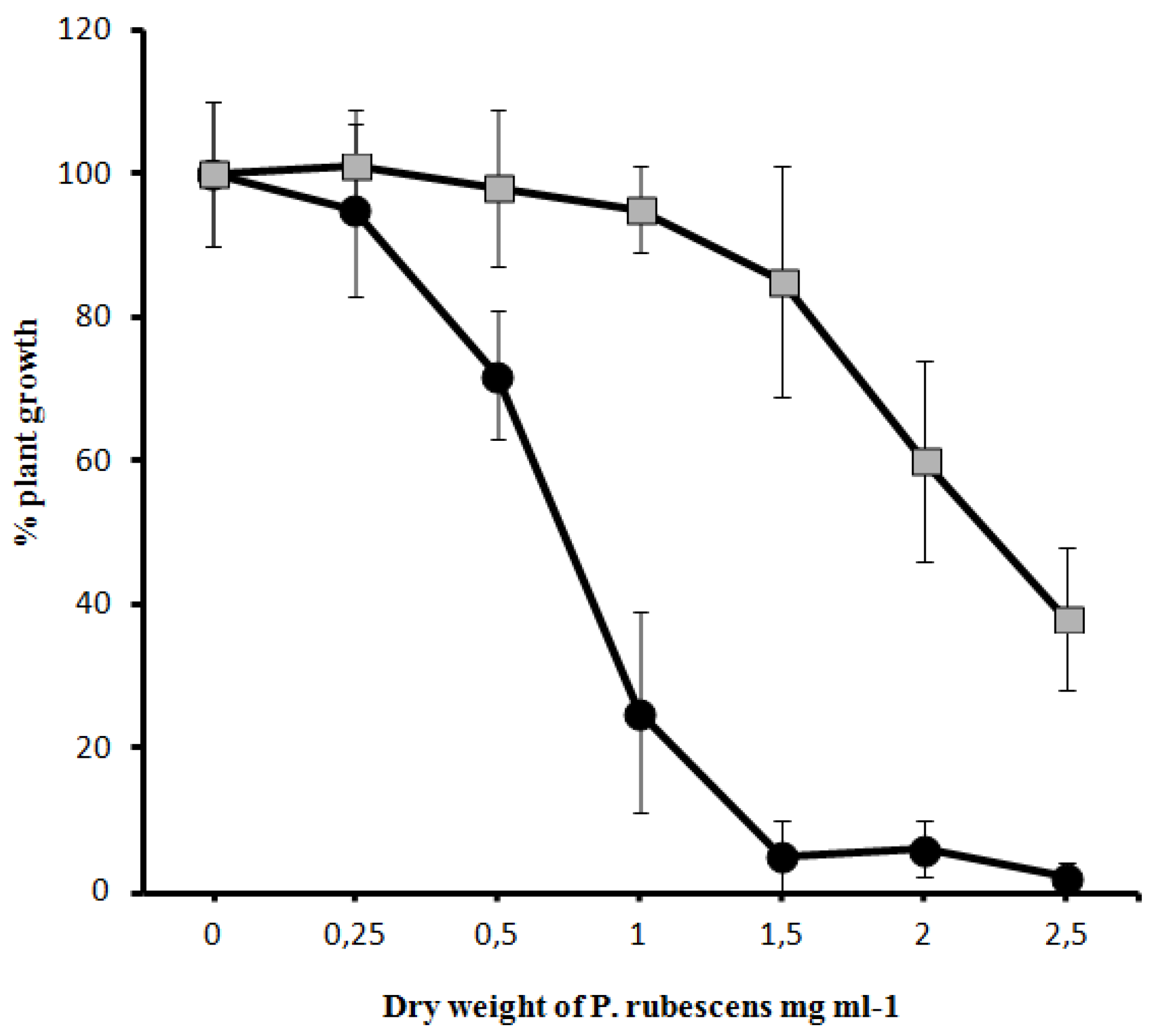

2.5. Analysis of the mcy Gene Cluster
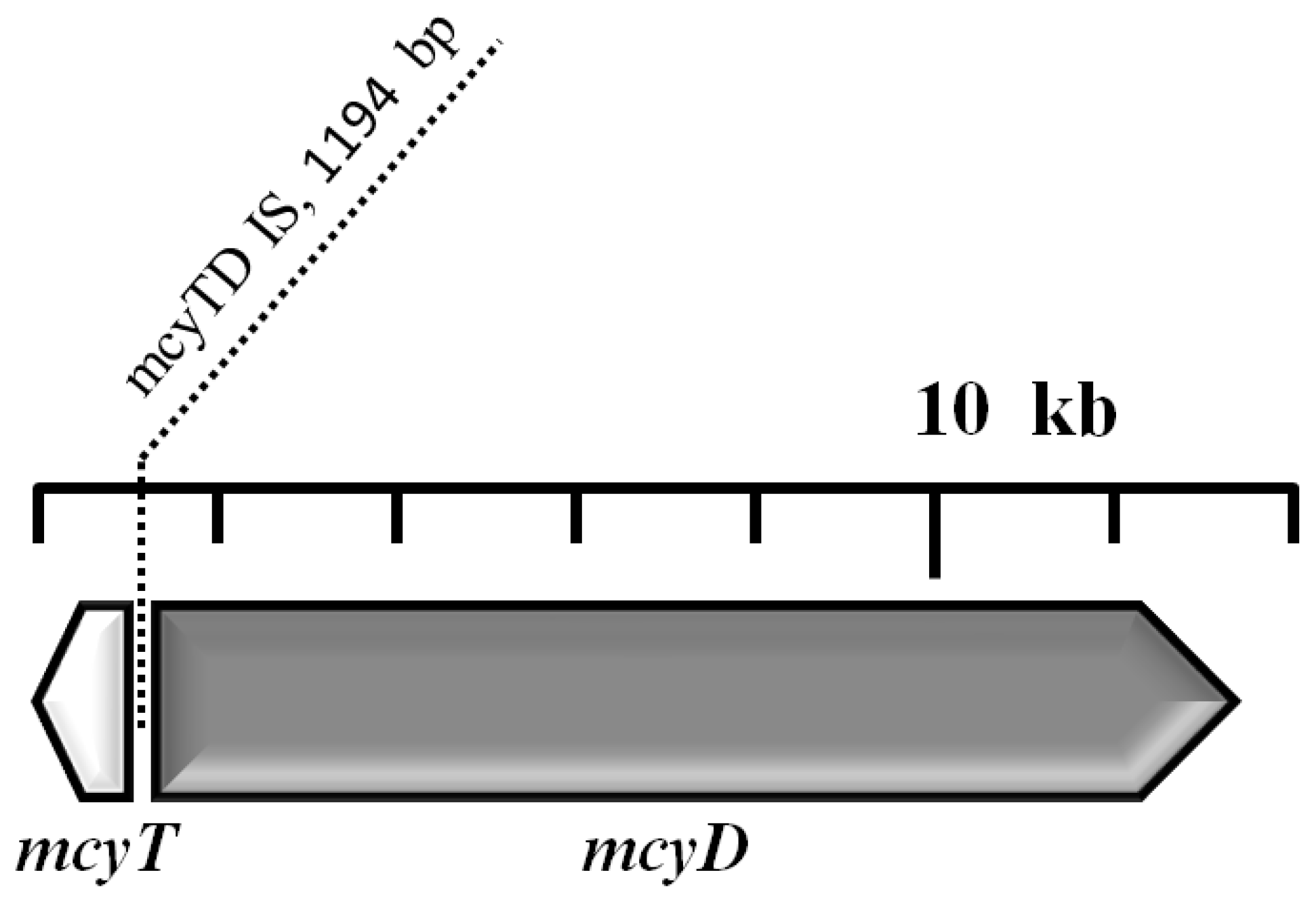
3. Discussion
4. Experimental Section
4.1. Site Description and Sampling
4.2. Physical and Chemical Variables
4.3. Identification of HAB Species
4.4. Molecular Phylogenetic Analyses
4.5. Purification of MC from Field Samples
4.6. Identification of MC Congener
4.6.1. MALDI-TOF MS Analysis
4.6.2. NMR
4.7. Capillary Electrophoresis of Field Samples and Isolated P. rubescens Laboratory Strain Samples
4.8. Genetic Analysis of the mcy Gene Cluster
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Reynolds, C.S.; Walsby, A.E. Water blooms. Biol. Rev. 1975, 50, 437–481. [Google Scholar] [CrossRef]
- Paerl, H.W. Combating the global proliferation of harmful cyanobacterial blooms by integrating conceptual and technological advances in an accessible water management toolbox. Environ. Microbiol. Rep. 2013, 5, 12–14. [Google Scholar]
- Paerl, H.W.; Huisman, J. Climate change: A catalyst for global expansion of harmful cyanobacterial blooms. Environ. Microbiol. Rep. 2009, 1, 27–37. [Google Scholar] [CrossRef]
- Carmichael, W.W. Freshwater cyanobacteria (blue-green algae) toxins. In Natural Toxins; Owby, C.L., Odell, G.V., Eds.; Pergamon Press: Oxford, UK, 1989; pp. 47–82. [Google Scholar]
- Carmichael, W.W. The toxins of cyanobacteria. Sci. Am. 1994, 270, 78–86. [Google Scholar] [CrossRef]
- Paerl, H.W.; Huisman, J. Blooms like it hot. Science 2008, 320, 57–58. [Google Scholar] [CrossRef]
- Chorus, I.; Bartam, J. Toxic Cyanobacteria in Water—A Guide to Their Public Health Consequences, Monitoring and Management; E & FN Spon: London, UK, 1999; pp. 41–111. [Google Scholar]
- Tillett, D.; Dittmann, E.; Erhard, M.; Döhren, H.; Borner, T.; Neilan, B.A. Structural organization of microcystin biosynthesis in Microcystis aeruginosa PCC7806: An integrated peptide-poliketide synthetase system. Chem. Biol. 2000, 7, 753–764. [Google Scholar] [CrossRef]
- Nishizawa, T.; Asayama, M.; Fujii, K.; Harada, K.I.; Shirai, M. Genetic analysis of the peptide synthetase genes for a cyclic heptapeptide microcystin in Microcystis spp. J. Biochem. 1999, 126, 520–529. [Google Scholar] [CrossRef]
- Christiansen, G.; Fastner, J.; Erhard, M.; Börner, T.; Dittmann, E. Microcystin biosynthesis in Planktothrix: Genes, evolution, and manipulation. J. Bacteriol. 2003, 185, 564–572. [Google Scholar] [CrossRef]
- Rouhiainen, L.; Vakkilainen, T.; Siemer, B.L.; Buikema, W.; Haselkorn, R.; Sivonen, K. Genes coding for hepatotoxic heptapeptides (microcystins) in the cyanobacterium Anabaena strain 90. Appl. Environ. Microb. 2004, 70, 686–692. [Google Scholar] [CrossRef]
- Fastner, J.; Erhard, M.; Carmichael, W.W.; Sun, F.; Rinehart, K.L.; Rönicke, H.; Chorus, I. Characterization and diversity of microcystins in natural blooms and strains of the genera Microcystis and Planktothrix from German freshwaters. Arch. Hydrobiol. 1999, 145, 147–163. [Google Scholar]
- Komárek, J.; Komárková, J. Taxonomic review of the cyanoprokaryotic genera Planktothrix and Planktothricoides. Czech. Phycol. Olomouc. 2004, 4, 1–18. [Google Scholar]
- Jann-Para, G.; Schwob, I.; Feuillade, M. Occurrence of toxic Planktothrix rubescens blooms in lake Nantua, France. Toxicon 2004, 43, 279–285. [Google Scholar] [CrossRef]
- Barco, M.; Flores, C.; Rivera, J.; Caixach, J. Determination of microcystin variants and related peptides present in a water bloom of Planktothrix (Oscillatoria) rubescens in a Spanish drinking water reservoir by LC/ESI-MS. Toxicon 2004, 44, 881–886. [Google Scholar] [CrossRef]
- Jacquet, S.; Briand, J.-F.; Leboulanger, C.; Avois-Jacquet, C.; Oberhaus, L.; Tassin, B.; Vincon-Leite, B.; Paolini, G.; Druart, J.-C.; Anneville, O.; et al. The proliferation of the toxic cyanobacterium Planktothrix rubescens following restoration of the largest natural French lake (Lac du Bourget). Harmful Algae 2005, 4, 651–672. [Google Scholar] [CrossRef] [Green Version]
- Legnani, E.; Copetti, D.; Oggioni, A.; Tartari, G.; Palumbo, M.-T.; Morabito, G. Planktothrix rubescens’ seasonal dynamics and vertical distribution in Lake Pusiano North Italy. J. Limnol. 2005, 64, 61–73. [Google Scholar]
- Ernst, B.; Hoeger, S.J.; O’Brien, E.; Dietrich, D.R. Abundance and toxicity of Planktothrix rubescens in the pre-Alpine Lake Ammersee, Germany. Harmful Algae 2009, 8, 329–342. [Google Scholar] [CrossRef]
- Paulino, S.; Valério, E.; Faria, N.; Fastner, J.; Welker, M.; Tenreiro, R.; Pereira, P. Detection of Planktothrix rubescens (Cyanobacteria) associated with microcystin production in a freshwater reservoir. Hydrobiologia 2009, 621, 207–211. [Google Scholar] [CrossRef]
- Kurmayer, R.; Christiansen, G.; Gumpenberger, M.; Fastner, J. Genetic identification of microcystin ecotypes in toxic cyanobacteria of the genus Planktothrix. Microbiology 2005, 151, 1525–1533. [Google Scholar] [CrossRef]
- Kurmayer, R.; Gumpenberger, M. Diversity of microcystin genotypes among populations of the filamentous cyanobacteria Planktothrix rubescens and Planktothrix agardhii. Mol. Ecol. 2006, 15, 3849–3861. [Google Scholar] [CrossRef]
- Bácsi, I.; Vasas, G.; Surányi, G.; M-Hamvas, M.; Máthé, C.; Tóth, E.; Grigorszky, I.; Gáspár, A.; Tóth, S.; Borbely, G. Alteration of cylindrospermopsin production in sulfate- or phosphate-starved cyanobacterium Aphanizomenon ovalisporum. FEMS Microbiol. Lett. 2006, 259, 303–310. [Google Scholar] [CrossRef]
- Neilan, B.A.; Pearson, L.A.; Muenchhoff, J.; Moffitt, M.C.; Dittmann, E. Environmental conditions that influence toxin biosynthesis in cyanobacteria. Environ. Microbiol. 2013, 15, 1239–1253. [Google Scholar] [CrossRef]
- Kurmayer, R.; Christiansen, G.; Fastner, J.; Börner, T. Abundance of active and inactive microcystin genotypes in populations of the toxic cyanobacterium Planktothrix spp. Environ. Microbiol. 2004, 6, 831–841. [Google Scholar]
- Christiansen, G.; Kurmayer, R.; Liu, Q.; Börner, T. Transposons inactivate the biosynthesis of the nonribosomal peptide microcystin in naturally occurring Planktothrix spp. Appl. Environ. Microbiol. 2006, 72, 117–123. [Google Scholar] [CrossRef]
- Borics, G.; Abonyi, A.; Krasznai, E.; Várbíró, G.; Grigorszky, I.; Szabó, S.; Deák, C.; Tóthmérész, B. Small-scale patchiness of the phytoplankton in a lentic oxbow. J. Plankton Res. 2011, 33, 973–981. [Google Scholar] [CrossRef]
- Reynolds, C.S. Ecology of Phytoplankton; Cambridge University Press: Cambridge, UK, 2006; pp. 42–74. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota, part 2. Oscillatoriales. In Süsswasser Flora von Mitteleuropa Band 19/2; Büdel, B., Gärtner, G., Krienitz, L., Schagerl, M., Eds.; Gustav Fischer: Jena, Germany, 2005; p. 759. [Google Scholar]
- Suda, S.; Watanabe, M.M.; Otsuka, S.; Mahakahant, A.; Yongmanitchai, W.; Nopartnaraporn, N.; Liu, Y.; Day, J.G. Taxonomic revision of water-bloom-forming species of oscillatorioid cyanobacteria. Int. J. Syst. Evol. Micr. 2002, 52, 1577–1595. [Google Scholar] [CrossRef]
- Brudno, M.; Do, C.B.; Cooper, G.M.; Kim, M.F.; Davydov, E.; Green, E.D.; Sidow, A.; Batzoglou, S. NISC comparative sequencing program. LAGAN and Multi-LAGAN: Efficient tools for large-scale multiple alignment of genomic DNA. Genome Res. 2003, 13, 721–731. [Google Scholar] [CrossRef]
- Mayor, C.; Brudno, M.; Schwartz, J.R.; Poliakov, A.; Rubin, E.M.; Frazer, K.A.; Pachter, L.S.; Dubchak, I. VISTA: Visualizing global DNA sequence alignments of arbitrary length. Bioinformatics 2000, 16, 1046–1047. [Google Scholar] [CrossRef]
- Walsby, A.E.; Schanz, F.; Schmid, M. The Burgundy-blood phenomenon: A model of buoyancy change explains autumnal waterblooms by Planktothrix rubescens in Lake Zürich. New Phytol. 2005, 169, 109–122. [Google Scholar] [CrossRef]
- Salmaso, N. Factors affecting the seasonality and distribution of cyanobacteria and chlorophytes: A case study from the large lakes south of the Alps, with special reference to Lake Garda. Hydrobiologia 2000, 438, 43–63. [Google Scholar] [CrossRef]
- Messineo, V.; Mattei, D.; Melchiorre, S.; Salvatore, G.; Bogialli, S.; Salzano, R.; Mazza, R.; Capelli, G.; Bruno, M. Microcystin diversity in a Planktothrix rubescens population from Lake Albano (Central Italy). Toxicon 2006, 48, 160–174. [Google Scholar]
- Halstvedt, C.B.; Rohrlack, T.; Andersen, T.; Skulberg, O.; Edvardsen, B. Seasonal dynamics and depth distribu-tion of Planktothrix spp. in Lake Steinsfjorden (Norway) related to environmental factors. J. Plankton Res. 2007, 29, 471–482. [Google Scholar] [CrossRef]
- Reynolds, C.S. The ecology of the planktonic blue-green algae in the North Shropshire meres. Field Stud. 1971, 3, 409–432. [Google Scholar]
- Borics, G.; Tóthmérész, B.; Lukács, B.A.; Várbíró, G. Functional groups of phytoplankton shaping diversity of shallow lake ecosystems. Hydrobiologia 2012, 698, 251–262. [Google Scholar] [CrossRef]
- Teszárné, N.M.; Márialigeti, K.; Végvári, P.; Csépes, E.; Bancsi, I. Stratification analysis of the Óhalász Oxbow of the River Tisza (Kisköre Reservoir, Hungary). Hydrobiologia 2003, 506–509, 37–44. [Google Scholar] [CrossRef]
- V.-Balogh, K.; Németh, B.; Vörös, L. Specific attenuation coefficients of optically active substances and their contribution to the underwater ultraviolet and visible light climate in shallow lakes and ponds. Hydrobiologia 2009, 632, 91–105. [Google Scholar] [CrossRef]
- Reynolds, C.S.; Huszar, V.; Kruk, C.; Naselli-Flores, L.; Melo, S. Towards a functional classification of the freshwater phytoplankton. J. Plankton Res. 2002, 24, 417–428. [Google Scholar] [CrossRef]
- Dokulil, M.T.; Teubner, K. Deep living Planktothrix rubescens modulated by environmental constraints and climate forcing. Hydrobiologia 2012, 698, 29–46. [Google Scholar] [CrossRef]
- Sundaram, T.R.; Rehm, R.G. The seasonal thermal structure of deep temperature lakes. Tellus 1973, 25, 157–167. [Google Scholar] [CrossRef]
- Berman, T.; Pollinger, U. Annual and seasonal variations of phytoplankton chlorophyll and photosynthesis in Lake Kinneret. Limnol. Oceanogr. 1974, 19, 31–55. [Google Scholar] [CrossRef]
- Salmaso, N. Ecological patterns of phytoplankton assemblages in Lake Garda: Seasonal, spatial and historical features. J. Limnol. 2002, 61, 95–115. [Google Scholar]
- Branco, B.F.; Torgersen, T. Predicting the onset of thermal stratification in shallow inland waterbodies. Aquat. Sci. 2009, 71, 65–79. [Google Scholar] [CrossRef]
- Padisák, J.; Reynolds, C.S. Shallow lakes: The absolute, the relative, the functional and the pragmatic. Hydrobiologia 2003, 506–509, 1–11. [Google Scholar] [CrossRef]
- Scheffer, M.; Nes, E.H. Shallow lakes theory revisited: Various alternative regimes driven by climate, nutrients, depth and lake size. In Shallow Lakes in a Changing World. Developments in Hydrobiology; Gulati, R.D., Lammens, E., Pauw, N., Donk, E., Eds.; Springer: Dordrecht, The Netherlands, 2007; Volume 196, pp. 455–466. [Google Scholar]
- Hutchinson, G.E.; Löffler, H. The thermal classification of lakes. Proc. Natl. Acad. Sci. USA. 1956, 42, 84–86. [Google Scholar] [CrossRef]
- Lewis, W.M., Jr. Tropical limnology. Annu. Rev. Ecol. Syst. 1987, 18, 159–184. [Google Scholar]
- Padisák, J.; G.-Tóth, L.; Rajczy, M. Stir-up effect of wind on a more-or-less stratified shallow lake phyto- plankton community, Lake Balaton, Hungary. Hydrobiologia 1990, 191, 249–254. [Google Scholar] [CrossRef]
- Pithart, D.; Pechar, L. The stratification of pools in the alluvium of the river Lužnice. Int. Rev. Gesamten Hydrobiol. Hydrogr. 1995, 80, 61–75. [Google Scholar] [CrossRef]
- Mischke, U. Cyanobacteria associations in shallow poly- trophic lakes: Influence of environmental factors. Acta Oecol. 2003, 24, 11–23. [Google Scholar] [CrossRef]
- Fonseca, B.M.; Bicudo, C.E.M. Phytoplankton seasonal variation in a shallow stratified eutrophic reservoir (Garcas Pond, Brazil). Hydrobiologia 2008, 600, 267–282. [Google Scholar] [CrossRef]
- Folkard, A.M.; Sherborne, A.J.; Coates, M.J. Turbulence and stratification in Priest Pot, a productive pond in a sheltered environment. Limnology 2007, 8, 113–120. [Google Scholar] [CrossRef] [Green Version]
- Vareli, K.; Briasoulis, E.; Pilidis, G.; Sainis, I. Molecular confirmation of Planktothrix rubescens as the cause of intense, microcystin—Synthesizing cyanobacterial bloom in Lake Ziros, Greece. Harmful Algae 2009, 8, 447–453. [Google Scholar] [CrossRef]
- Micheletti, S.; Schanz, F.; Walsby, A.E. The daily integral of photosynthesis by Planktothrix rubescens during summer stratification and autumnal mixing in Lake Zürich. New Phytol. 1998, 139, 233–246. [Google Scholar]
- Komárek, J. Recent changes (2008) in cyanobacteria taxonomy based on a combination of molecular background with phenotype and ecological consequences (genus and species concept). Hydrobiologia. 2010, 639, 245–259. [Google Scholar] [CrossRef]
- Lin, S.; Wu, Z.; Yu, G.; Zhu, M.; Yu, B.; Li, R. Genetic diversity and molecular phylogeny of Planktothrix (Oscillatoriales, cyanobacteria) strains from China. Harmful Algae 2010, 9, 87–97. [Google Scholar] [CrossRef]
- Konopka, A. Influence of temperature, oxygen, and pH on a metalimnetic population of Oscillatoria rubescens. Appl. Environ. Microbiol. 1981, 42, 102–108. [Google Scholar]
- Akcaalan, R.; Young, F.M.; Metcalf, J.S.; Morrison, L.F.; Albay, M.; Codd, G.A. Microcystin analysis in single filaments of Planktothrix spp. in laboratory cultures and environmental blooms. Water Res. 2006, 40, 1583–1590. [Google Scholar] [CrossRef]
- Meriluoto, J.A.O.; Sandström, A.; Eriksson, J.E.; Remaud, G.; Craig, A.G.; Chattopadhyaya, J. Structure and toxicity of a peptide hepatotoxin from the cyanobacterium Oscillatoria agardhii. Toxicon 1989, 27, 1024–1034. [Google Scholar]
- Sivonen, K.; Namikoshi, M.; Evans, W.R.; Carmichael, W.W.; Sun, F.; Rouhiainen, L.; Luukkainen, R.; Rinehart, K.L. Isolation and characterization of a variety of microcystins from seven strains of the cyanobacterial genus Anabaena. Appl. Environ. Microb. 1992, 58, 2495–2500. [Google Scholar]
- Luukkainen, R.; Sivonen, K.; Namikoshi, M.; Färdig, M.; Rinehart, K.L.; Niemelä, S.I. Isolation and identification of eight microcystins from thirteen Oscillatoria agardhii strains and structure of a new microcystin. Appl. Environ. Microb. 1993, 59, 2204–2209. [Google Scholar]
- Blom, J.F.; Robinson, J.A.; Jüttner, F. High grazer toxicity of [d-Asp3, (E)-Dhb7]microcystin-RR of Planktothrix rubescens as compared to different microcystins. Toxicon 2001, 39, 1923–1932. [Google Scholar] [CrossRef]
- Blom, J.F.; Jüttner, F. High crustacean toxicity of microcystin congeners does not correlate with high protein phosphatase inhibitory activity. Toxicon 2005, 46, 465–470. [Google Scholar] [CrossRef]
- Sano, T.; Takagi, H.; Kaya, K. A Dhb-microcystin from the filamentous cyanobacterium Planktothrix rubescens. Phytochemistry 2004, 65, 2159–2162. [Google Scholar] [CrossRef]
- Vasas, G.; Gáspár, A.; Surányi, G.; Batta, G.; Gyémánt, G.; M-Hamvas, M.; Máthé, C.; Grigorszky, I.; Molnár, E.; Borbély, G. Capillary electrophoretic assay and purification of cylindrospermopsin, a cyanobacterial toxin from Aphanizomenon ovalisporum by plant test (Blue-Green Sinapis Test). Anal. Biochem. 2002, 302, 95–103. [Google Scholar] [CrossRef]
- Vasas, G.; Gáspár, A.; Páger, C.; Surányi, G.; M-Hamvas, M.; Máthé, C.; Borbély, G. Analysis of cyanobacterial toxins (anatoxin-a, cylindrospermopsin, microcystin-LR) by capillary electrophoresis. Electrophoresis 2004, 25, 108–115. [Google Scholar] [CrossRef]
- Vasas, G.; Szydlowska, D.; Gáspár, A.; Welker, M.; Trojanowicz, M.; Borbély, G. Determination of microcystins in environmental samples using capillary electrophoresis. J. Biochem. Biophys. Methods 2006, 66, 87–97. [Google Scholar] [CrossRef]
- Borics, G.; Grigorszky, I.; Szabó, S.; Padisák, J. Phytoplankton associations under changing pattern of bottom-up vs. top-down control in a small hypertrophic fishpond in East Hungary. Hydrobiologia 2000, 424, 79–90. [Google Scholar] [CrossRef]
- Krasznai, E.; Borics, G.; Várbíró, G.; Abonyi, A.; Padisák, J.; Deák, C.; Tóthmérész, B. Characteristics of the pelagic phytoplankton in shallow oxbows. Hydrobiologia 2010, 639, 173–184. [Google Scholar] [CrossRef]
- Vasas, G.; Bacsi, I.; Suranyi, G.; M Hamvas, M.; Mathe, C.; Nagy, S.A.; Borbely, G. Isolation of viable cell mass from frozen Microcystis viridis bloom containing microcystin-RR. Hydrobiologia 2010, 639, 147–151. [Google Scholar] [CrossRef]
- Farkas, O.; Gyémant, G.; Hajdú, G.; Gonda, S.; Parizsa, P.; Horgos, T.; Mosolygó, Á.; Vasas, G. Variability of microcystins and its synthetase gene cluster in Microcystis and Planktothrix waterblooms in shallow lakes of Hungary. Acta Biol. Hung. 2014, 65, 5–23. [Google Scholar]
- Kurmayer, R.; Christiansen, G. The genetic basis of toxin production in Cyanobacteria. Freshw. Rev. 2009, 2, 31–50. [Google Scholar]
- Kurmayer, R.; Schober, E.; Tonk, L.; Visser, P.; Christiansen, G. Spatial divergence in the proportions of genes encoding toxic peptide synthesis among populations of the cyanobacterium Planktothrix in European lakes. FEMS Microbiol. Lett. 2011, 317, 127–137. [Google Scholar] [CrossRef]
- Rounge, T.B.; Rohrlack, T.; Nederbragt, A.J.; Kristensen, T.; Jakobsen, K.S. A genome-wide analysis of nonribosomal peptide synthetase gene clusters and their peptides in a Planktothrix rubescens strain. BMC Genomics 2009, 10, 396–406. [Google Scholar] [CrossRef]
- Mbedi, S.; Welker, M.; Fastner, J.; Wiedner, C. Variability of the microcystin synthetase gene cluster in the genus Planktothrix (Oscillatoriales, Cyanobacteria). FEMS Microbiol. Lett. 2005, 245, 299–306. [Google Scholar] [CrossRef]
- Suzuki, T.; Miyauchi, K. Discovery and characterization of tRNAIle lysidine synthetase (TilS). FEBS Lett. 2010, 584, 272–277. [Google Scholar] [CrossRef]
- Loomis, W.F.; Shaulsky, G.; Wang, N. Histidine kinases in signal transduction pathways of eukaryotes. J. Cell Sci. 1997, 110, 1141–1145. [Google Scholar]
- Somogyi, B.; Felföldi, T.; Vanyovszki, J.; Ágyi, Á.; Márialigeti, K.; Vörös, L. Winter bloom of picoeukaryotes in Hungarian shallow turbid soda pans and the role of light and temperature. Aquat. Ecol. 2009, 43, 735–744. [Google Scholar] [CrossRef]
- Lamprinou, V.; Skaraki, K.; Kotoulas, G.; Economou-Amilli, A.; Pantazidou, A. Toxopsis calypsus gen. nov., sp. nov. (Cyanobacteria, Nostocales) from cave ‘Francthi’, Peloponnese, Greece: A morphological and molecular evaluation. Int. J. Syst. Evol. Micr. 2012, 62, 2870–2877. [Google Scholar] [CrossRef]
- Felföldi, T.; Duleba, M.; Somogyi, B.; Vajna, B.; Nikolausz, M.; Présing, M.; Márialigeti, K.; Vörös, L. Diversity and seasonal dynamics of the photoautotrophic picoplankton in Lake Balaton (Hungary). Aquat. Microb. Ecol. 2011, 63, 273–287. [Google Scholar] [CrossRef]
- Pruesse, E.; Peplies, J.; Glöckner, F.O. SINA: Accurate high throughput multiple sequence alignment of ribosomal RNA genes. Bioinformatics 2012, 28, 1823–1829. [Google Scholar] [CrossRef]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef]
- Welker, M.; Fastner, J.; Erhard, M.; Döhren, H. Application of MALDI-TOF MS in cyanotoxin research. Environ. Toxicol. 2002, 17, 367–374. [Google Scholar] [CrossRef]
- Christiansen, G.; Molitor, C.; Philmus, B.; Kurmayer, R. Non-toxic strains of cyanobacteria are the result of major gene deletion events induced by a transposable element. Mol. Biol. Evol. 2008, 25, 1695–1704. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vasas, G.; Farkas, O.; Borics, G.; Felföldi, T.; Sramkó, G.; Batta, G.; Bácsi, I.; Gonda, S. Appearance of Planktothrix rubescens Bloom with [D-Asp3, Mdha7]MC–RR in Gravel Pit Pond of a Shallow Lake-Dominated Area. Toxins 2013, 5, 2434-2455. https://doi.org/10.3390/toxins5122434
Vasas G, Farkas O, Borics G, Felföldi T, Sramkó G, Batta G, Bácsi I, Gonda S. Appearance of Planktothrix rubescens Bloom with [D-Asp3, Mdha7]MC–RR in Gravel Pit Pond of a Shallow Lake-Dominated Area. Toxins. 2013; 5(12):2434-2455. https://doi.org/10.3390/toxins5122434
Chicago/Turabian StyleVasas, Gábor, Oszkár Farkas, Gábor Borics, Tamás Felföldi, Gábor Sramkó, Gyula Batta, István Bácsi, and Sándor Gonda. 2013. "Appearance of Planktothrix rubescens Bloom with [D-Asp3, Mdha7]MC–RR in Gravel Pit Pond of a Shallow Lake-Dominated Area" Toxins 5, no. 12: 2434-2455. https://doi.org/10.3390/toxins5122434




