Preliminary Results of the in Vivo and in Vitro Characterization of a Tentacle Venom Fraction from the Jellyfish Aurelia aurita
Abstract
:1. Introduction
2. Results and Discussion
2.1. Jellyfish Specimens, Tentacle Venom Extract and Nematocyst Identification
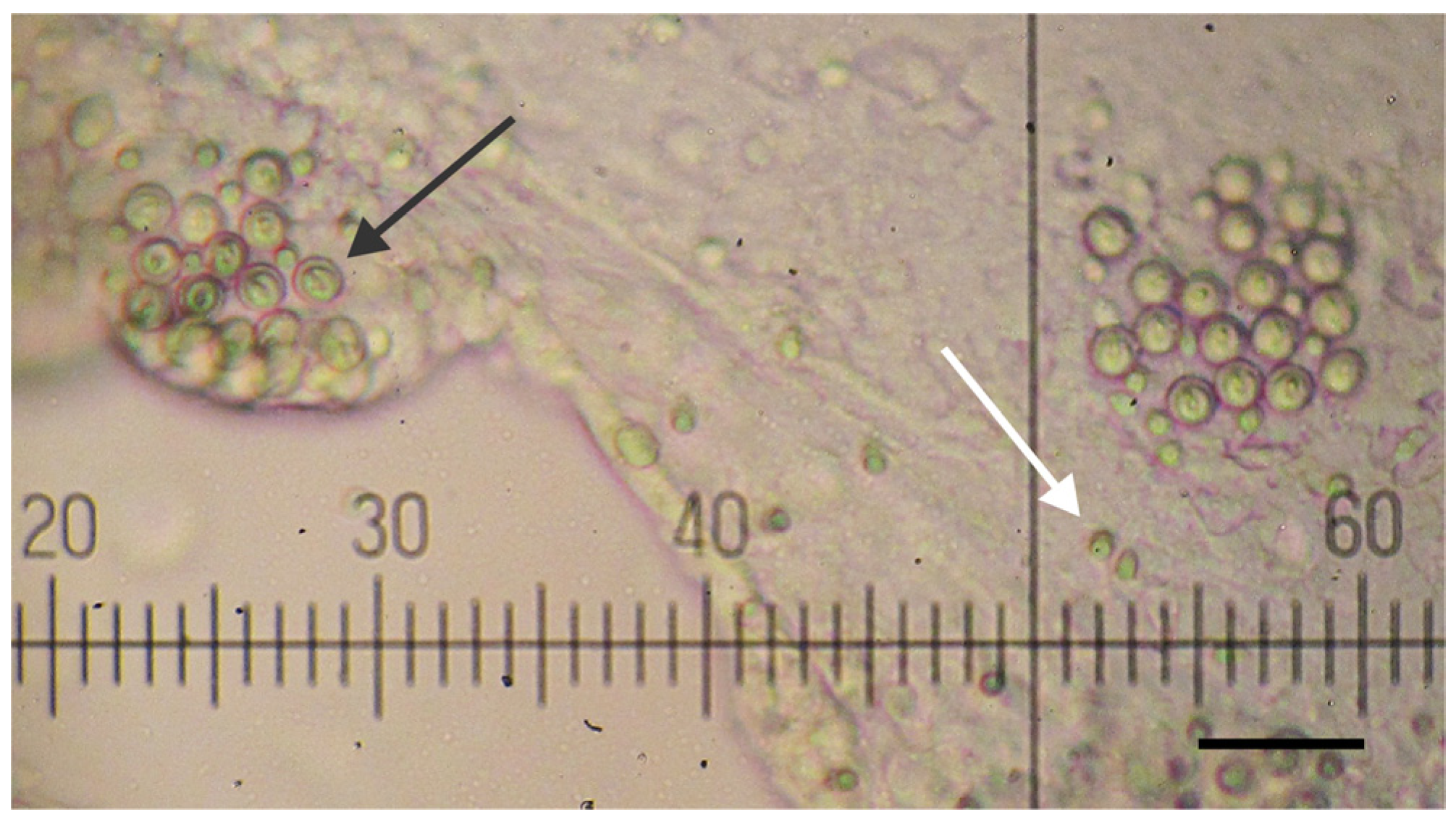
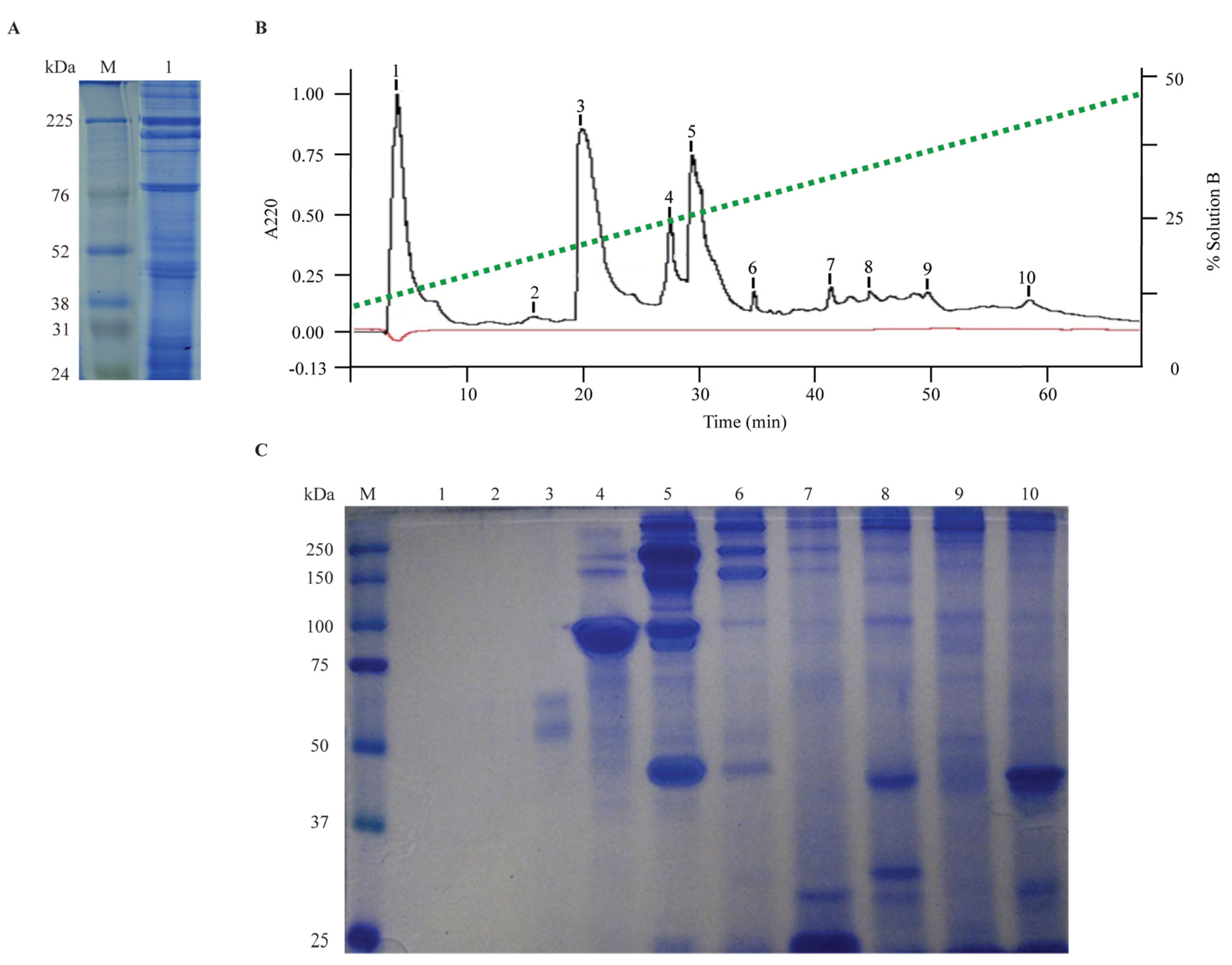
2.2. Protein Content Analysis and Fractionation
2.3. In Vivo Neurotoxic Effects
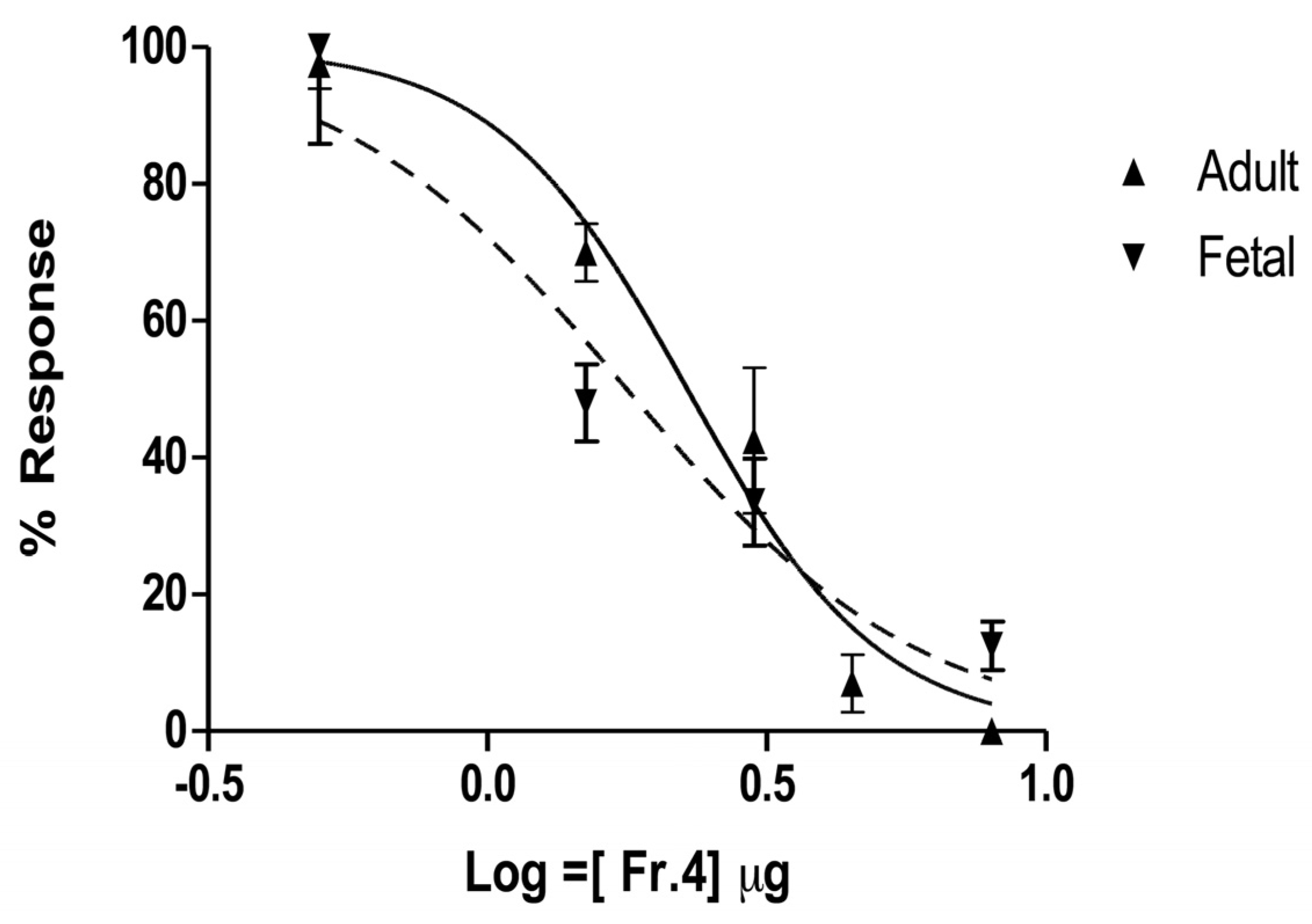
2.4. In Vitro Characterization on Muscle nAChRs

2.5. Phospholipase A Activity
3. Experimental Section
3.1. Reagents
3.2. Specimen Collection
3.3. Preparation of Tentacle Venom Extract
3.4. Chromatography Fractionation of Tentacle Venom Extract
3.5. Protein Concentration
3.6. SDS-PAGE Analysis
3.7. Neurotoxic Activity Bioassay
3.8. Activity on Nicotinic Acetylcholine Receptors
3.9. Phospholipase A (PLA) Assay
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Mariottini, G.L.; Pane, L. Mediterranean jellyfish venoms: A review on scyphomedusae. Mar. Drugs 2010, 8, 1122–1152. [Google Scholar] [CrossRef]
- Fautin, D.G. Structural diversity, systematics, and evolution of cnidae. Toxicon 2009, 54, 1054–1064. [Google Scholar] [CrossRef]
- Šuput, D. In vivo effects of cnidarian toxins and venoms. Toxicon 2009, 54, 1190–1200. [Google Scholar] [CrossRef]
- Turk, T.; Kem, W.R. The phylum Cnidaria and investigations of its toxins and venoms until 1990. Toxicon 2009, 54, 1031–1037. [Google Scholar] [CrossRef]
- Williamson, J.A.; Fenner, P.J.; Burnett, J.W. Principles of Patient Care in Marine Envenomations and Poisonings. In Venomous and Poisonous Marine Animals: A Medical and Biological Handbook, 1st ed.; Williamson, J.A., Burnett, J.W., Fenner, P.J., Rifkin, J.F., Eds.; University of New South Wales Press: Queensland, Australia, 1996; pp. 98–117. [Google Scholar]
- Burnett, J.W.; Calton, G.J.; Larsen, J.B. Significant envenomation by Aurelia aurita, the moon jellyfish. Toxicon 1988, 26, 215–217. [Google Scholar] [CrossRef]
- Segura-Puertas, L.; Avila-Soria, G.; Sánchez-Rodríguez, J.; Ramos-Aguilar, M.E.; Burnett, J.W. Some toxinological aspects of Aurelia aurita (Linné) from the Mexican Caribbean. J. Venom. Anim. Toxins Incl. Trop. Dis. 2002, 8, 269–282. [Google Scholar] [CrossRef]
- Helmholz, H.; Johnston, B.D.; Ruhnau, C.; Prange, A. Gill cell toxicity of northern boreal scyphomedusae Cyanea capillata and Aurelia aurita measured by an in vitro cell assay. Hydrobiologia 2010, 645, 223–234. [Google Scholar] [CrossRef]
- Helmholz, H. Selective toxin-lipid membrane interactions of natural, haemolytic Scyphozoan toxins analyzed by surface plasmon resonance. Biochim. Biophys. Acta 2010, 1798, 1944–1952. [Google Scholar] [CrossRef]
- Kihara, H.; Anraku, M.; Ohno, M.; Hashimura, S. Tetrodotoxin-unaffected depolarization of frog muscles induced by the venom of jellyfish (Genus aurelia). Jpn. J. Physiol. 1988, 38, 839–849. [Google Scholar] [CrossRef]
- Nevalainen, T.J.; Peuravuori, H.J.; Quinn, R.J.; Llewellyn, L.E.; Benzie, J.A.H.; Fenner, P.J.; Winkel, K.D. Phospholipase A2 in Cnidaria. Comp. Biochem. Physiol. B 2004, 139, 731–735. [Google Scholar] [CrossRef]
- Radwan, F.F.; Burnett, J.W.; Bloom, D.A.; Coliano, T.; Eldefrawi, M.E.; Erderly, H.; Aurelian, L.; Torres, M.; Heimer-de la Cotera, E.P. A comparison of the toxinological characteristics of two Cassiopea and Aurelia species. Toxicon 2001, 39, 245–257. [Google Scholar] [CrossRef]
- Cobbs, C.S.; Drzymala, R.E.; Shamoo, A.E.; Calton, G.J.; Burnett, J.W. Sea nettle (Chrysaora quinquecirrha) lethal factor: Effect on black lipid membranes. Toxicon 1983, 21, 558–561. [Google Scholar] [CrossRef]
- Dubois, J.M.; Tanguy, J.; Burnett, J.W. Ionic channels induced by sea nettle toxin in the nodal membrane. Biophys. J. 1983, 42, 199–202. [Google Scholar] [CrossRef]
- Toom, P.M.; Phillips, T.D. Effects of purified components of jellyfish toxin (Stomolophus meleagris) on active sodium transport. Toxicon 1975, 13, 261–271. [Google Scholar] [CrossRef]
- Torres, M.; Aguilar, M.B.; Falcon, A.; Sanchez, L.; Radwan, F.F.; Burnett, J.W.; Heimer-de la Cotera, E.P.; Arellano, R.O. Electrophysiological and hemolytic activity elicited by the venom of the jellyfish Cassiopea xamachana. Toxicon 2001, 39, 1297–1307. [Google Scholar] [CrossRef]
- Dutertre, S.; Lewis, R.J. Toxin insights into nicotinic acetylcholine receptors. Biochem. Pharmacol. 2006, 72, 661–670. [Google Scholar] [CrossRef]
- Arias, H.R. Marine toxins targeting ion channels. Mar. Drugs 2006, 4, 37–69. [Google Scholar] [CrossRef]
- Luetje, C.W.; Wada, K.; Rogers, S.; Abramson, S.N.; Tsuji, K.; Heinemann, S.; Patrick, J. Neurotoxins distinguish between different neuronal nicotinic acetylcholine-receptor subunit combinations. J. Neurochem. 1990, 55, 632–640. [Google Scholar] [CrossRef]
- Servent, D.; Antil-Delbeke, S.; Gaillard, C.; Corringer, P.J.; Changeux, J.P.; Menez, A. Molecular characterization of the specificity of interactions of various neurotoxins on two distinct nicotinic acetylcholine receptors. Eur. J. Pharmacol. 2000, 393, 197–204. [Google Scholar] [CrossRef]
- Harvey, A.L.; Hider, R.C.; Hodges, S.J.; Joubert, F.J. Structure-activity studies of homologues of short chain neurotoxins from Elapid snake venoms. Br. J. Pharmacol. 1984, 82, 709–716. [Google Scholar] [CrossRef]
- Connolly, P.J.; Stern, A.S. Solution structure of LSIII, a long neurotoxin from the venom of Laticauda semifasciata. Biochemistry 1996, 35, 418. [Google Scholar] [CrossRef]
- Nirthanan, S.; Charpantier, E.; Gopalakrishnakone, P.; Gwee, M.C.E.; Khoo, H.E.; Cheah, L.S.; Kini, R.M.; Bertrand, D. Neuromuscular effects of candoxin, a novel toxin from the venom of the Malayan krait (Bungarus candidus). Br. J. Pharmacol. 2003, 139, 832–844. [Google Scholar] [CrossRef]
- Nirthanan, S.; Gopalakrishnakone, P.; Gwee, M.C.E.; Khoo, H.E.; Kini, R.M. Non-conventional toxins from Elapid venoms. Toxicon 2003, 41, 397–407. [Google Scholar] [CrossRef]
- Olivera, B.M.; Teichert, R.W. Diversity of the neurotoxic Conus peptides: A model for concerted pharmacological discovery. Mol. Interv. 2007, 7, 251–260. [Google Scholar] [CrossRef]
- Lewis, R.J.; Dutertre, S.; Vetter, I.; Christie, M.J. Conus venom peptide pharmacology. Pharmacol. Rev. 2012, 64, 259–298. [Google Scholar] [CrossRef]
- Terlau, H.; Olivera, B.M. Conus venoms: A rich source of novel ion channel-targeted peptides. Physiol. Rev. 2004, 84, 41–68. [Google Scholar] [CrossRef]
- Dawson, M.N.; Jacobs, D.K. Molecular evidence for cryptic species of Aurelia aurita (Cnidaria, Scyphozoa). Biol. Bull. 2001, 200, 92–96. [Google Scholar] [CrossRef]
- Di Camillo, C.G.; Betti, F.; Bo, M.; Martinelli, M.; Puce, S.; Bavestrello, G. Contribution to the understanding of seasonal cycle of Aurelia aurita (Cnidaria: Scyphozoa) scyphopolyps in the northern Adriatic Sea. J. Mar. Biol. Assoc. UK 2010, 90, 1105–1110. [Google Scholar] [CrossRef]
- Wiebring, A.; Helmholz, H.; Sotje, I.; Lassen, S.; Prange, A.; Tiemann, H. A new method for the separation of different types of nematocysts from scyphozoa and investigation of proteinaceous toxins utilizing laser catapulting and subsequent mass spectrometry. Mar. Biotechnol. 2010, 12, 308–317. [Google Scholar] [CrossRef]
- Xiao, L.; He, Q.; Guo, Y.F.; Zhang, J.; Nie, F.; Li, Y.; Ye, X.F.; Zhang, L.M. Cyanea capillata tentacle-only extract as a potential alternative of nematocyst venom: Its cardiovascular toxicity and tolerance to isolation and purification procedures. Toxicon 2009, 53, 146–152. [Google Scholar] [CrossRef]
- Moran, Y.; Genikhovich, G.; Gordon, D.; Wienkoop, S.; Zenkert, C.; Ozbek, S.; Technau, U.; Gurevitz, M. Neurotoxin localization to ectodermal gland cells uncovers an alternative mechanism of venom delivery in sea anemones. Proc. R. Soc. B 2012, 279, 1351–1358. [Google Scholar] [CrossRef]
- Li, C.P.; Yu, H.H.; Feng, J.H.; Chen, X.L.; Li, P.C. Comparative analysis of methods for concentrating venom from jellyfish Rhopilema esculentum Kishinouye. Chin. J. Oceanol. Limnol. 2009, 27, 172–176. [Google Scholar] [CrossRef]
- Brinkman, D.; Burnell, J. Partial purification of cytolytic venom proteins from the box jellyfish, Chironex fleckeri. Toxicon 2008, 51, 853–863. [Google Scholar]
- Van Wagoner, R.M.; Jacobsen, R.B.; Olivera, B.M.; Ireland, C.M. Characterization and three-dimensional structure determination of psi-conotoxin PIIIF, a novel noncompetitive antagonist of nicotinic acetylcholine receptors. Biochemistry 2003, 42, 6353–6362. [Google Scholar] [CrossRef]
- Shon, K.J.; Grilley, M.; Jacobsen, R.; Cartier, G.E.; Hopkins, C.; Gray, W.R.; Watkins, M.; Hillyard, D.R.; Rivier, J.; Torres, J.; et al. A noncompetitive peptide inhibitor of the nicotinic acetylcholine receptor from Conus purpurascens venom. Biochemistry 1997, 36, 9581–9587. [Google Scholar] [CrossRef]
- Nirthanan, S.; Gwee, M.C.E. Three-finger alpha-neurotoxins and the nicotinic acetylcholine receptor, forty years on. J. Pharmacol. Sci. 2004, 94, 1–17. [Google Scholar] [CrossRef]
- Kini, R.M.; Iwanaga, S. Structure-function relationships of phospholipases I: Prediction of presynaptic neurotoxicity. Toxicon 1986, 24, 527–541. [Google Scholar] [CrossRef]
- Hodgson, W.C.; Wickramaratna, J.C. In vitro neuromuscular activity of snake venoms. Clin. Exp. Pharmacol. Physiol. 2002, 29, 807–814. [Google Scholar] [CrossRef]
- Östman, C. A guideline to nematocyst nomenclature and classification, and some notes on the systematic value of nematocysts. Sci. Mar. 2000, 64, 31–46. [Google Scholar]
- Laemmli, U.K. Cleavage of structural proteins during assembly of head of bacteriophage-T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Béress, L.; Béress, R.; Wunderer, G. Isolation and characterization of 3 polypeptides with neurotoxic activity from anemonia-sulcata. FEBS Lett. 1975, 50, 311–314. [Google Scholar]
- Bruhn, T.; Schaller, C.; Schulze, C.; Sanchez-Rodriguez, J.; Dannmeier, C.; Ravens, U.; Heubach, J.F.; Eckhardt, K.; Schmidtmayer, J.; Schmidt, H.; et al. Isolation and characterisation of five neurotoxic and cardiotoxic polypeptides from the sea anemone Anthopleura elegantissima. Toxicon 2001, 39, 693–702. [Google Scholar] [CrossRef]
- Cartier, G.E.; Yoshikami, D.; Gray, W.R.; Luo, S.; Olivera, B.M.; McIntosh, J.M. A new alpha-conotoxin which targets alpha3beta2 nicotinic acetylcholine receptors. J. Biol. Chem. 1996, 271, 7522–7528. [Google Scholar] [CrossRef]
- Sine, S.M.; Bren, N.; Quiram, P.A. Molecular dissection of subunit interfaces in the nicotinic acetylcholine receptor: Identification of residues that determine curare selectivity. Proc. Natl. Acad. Sci. USA 1993, 90, 9436–9440. [Google Scholar] [CrossRef]
- Carbajal-Saucedo, A.; López-Vera, E.; Bénard-Valle, M.; Smith, E.N.; Zamudio, F.; De Roodt, A.R.; Olvera-Rodríguez, A. Isolation, characterization, cloning and expression of an alpha-neurotoxin from the venom of the Mexican coral snake Micrurus laticollaris (Squamata: Elapidae). Toxicon 2013, 66, 64–74. [Google Scholar] [CrossRef]
- Haberman, E.; Hardt, K.L. Sensitive and specific plate test for quantitation of phospholipases. Anal. Biochem. 1972, 50, 163–173. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ponce, D.; López-Vera, E.; Aguilar, M.B.; Sánchez-Rodríguez, J. Preliminary Results of the in Vivo and in Vitro Characterization of a Tentacle Venom Fraction from the Jellyfish Aurelia aurita. Toxins 2013, 5, 2420-2433. https://doi.org/10.3390/toxins5122420
Ponce D, López-Vera E, Aguilar MB, Sánchez-Rodríguez J. Preliminary Results of the in Vivo and in Vitro Characterization of a Tentacle Venom Fraction from the Jellyfish Aurelia aurita. Toxins. 2013; 5(12):2420-2433. https://doi.org/10.3390/toxins5122420
Chicago/Turabian StylePonce, Dalia, Estuardo López-Vera, Manuel B. Aguilar, and Judith Sánchez-Rodríguez. 2013. "Preliminary Results of the in Vivo and in Vitro Characterization of a Tentacle Venom Fraction from the Jellyfish Aurelia aurita" Toxins 5, no. 12: 2420-2433. https://doi.org/10.3390/toxins5122420
APA StylePonce, D., López-Vera, E., Aguilar, M. B., & Sánchez-Rodríguez, J. (2013). Preliminary Results of the in Vivo and in Vitro Characterization of a Tentacle Venom Fraction from the Jellyfish Aurelia aurita. Toxins, 5(12), 2420-2433. https://doi.org/10.3390/toxins5122420




