Mouse in Vivo Neutralization of Escherichia coli Shiga Toxin 2 with Monoclonal Antibodies
Abstract
:1. Introduction
2. Results
2.1. Detection of Stx2 in Mouse Serum
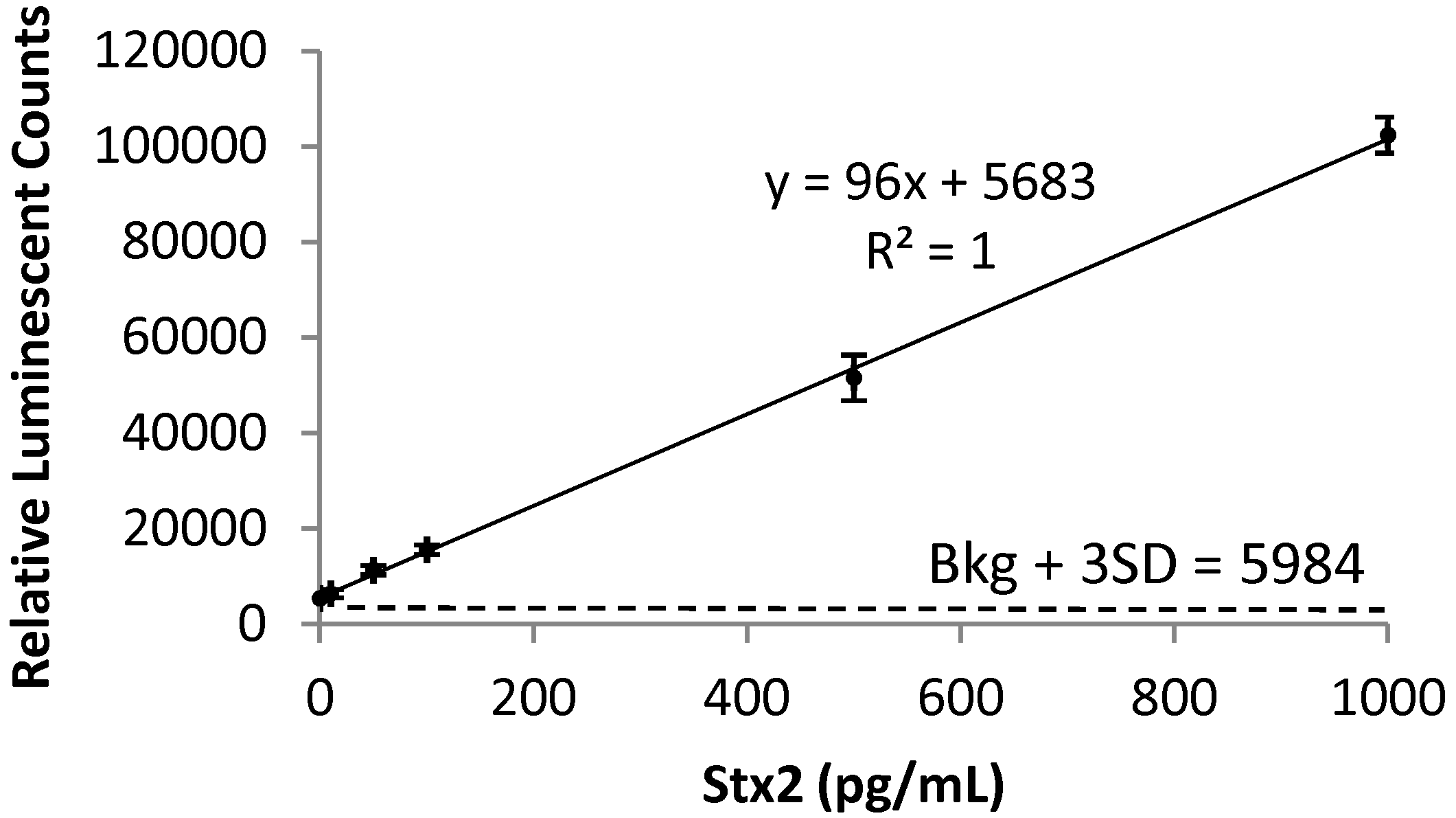
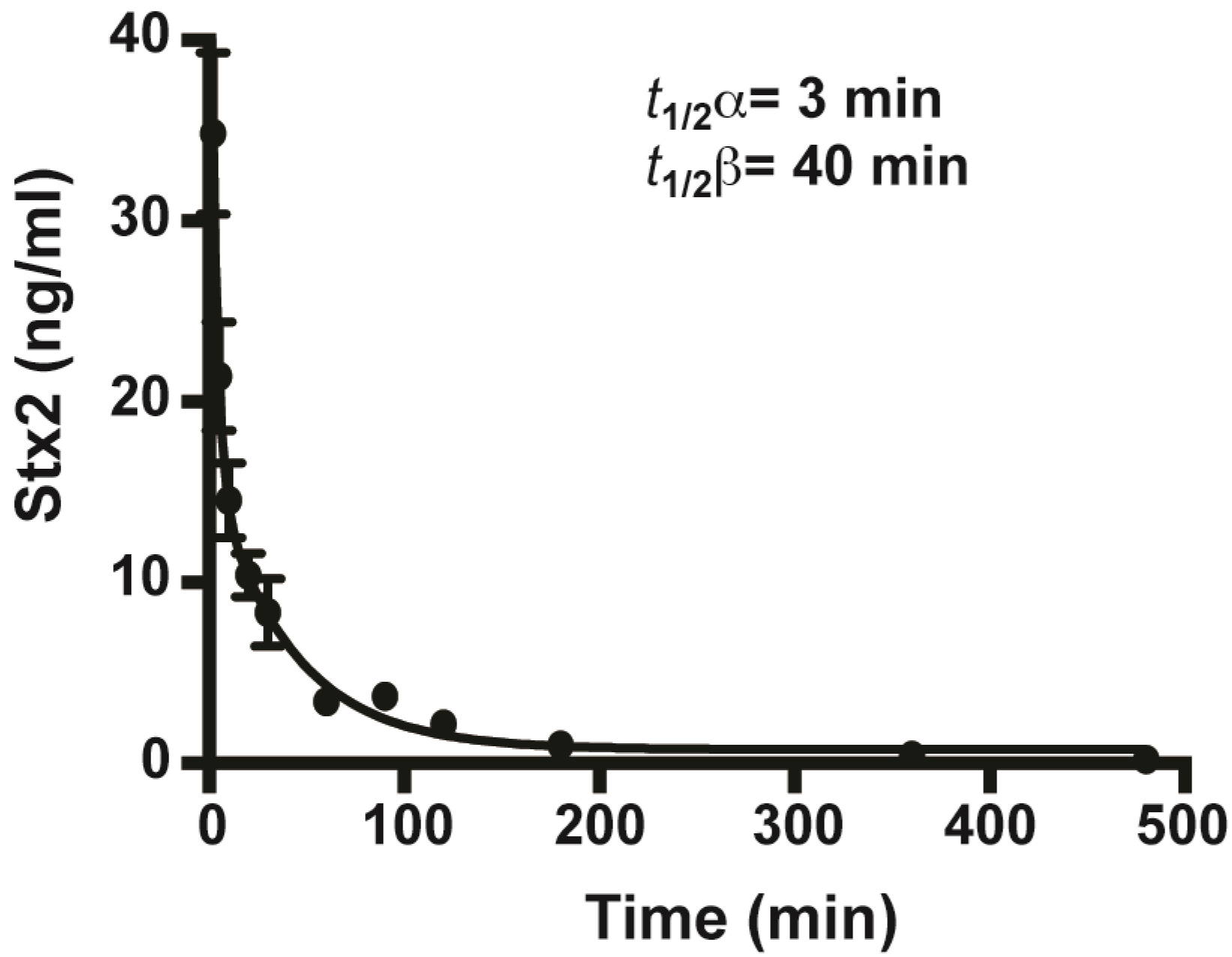
2.2. In Vivo Toxicity and Toxicokinetics of Stx2
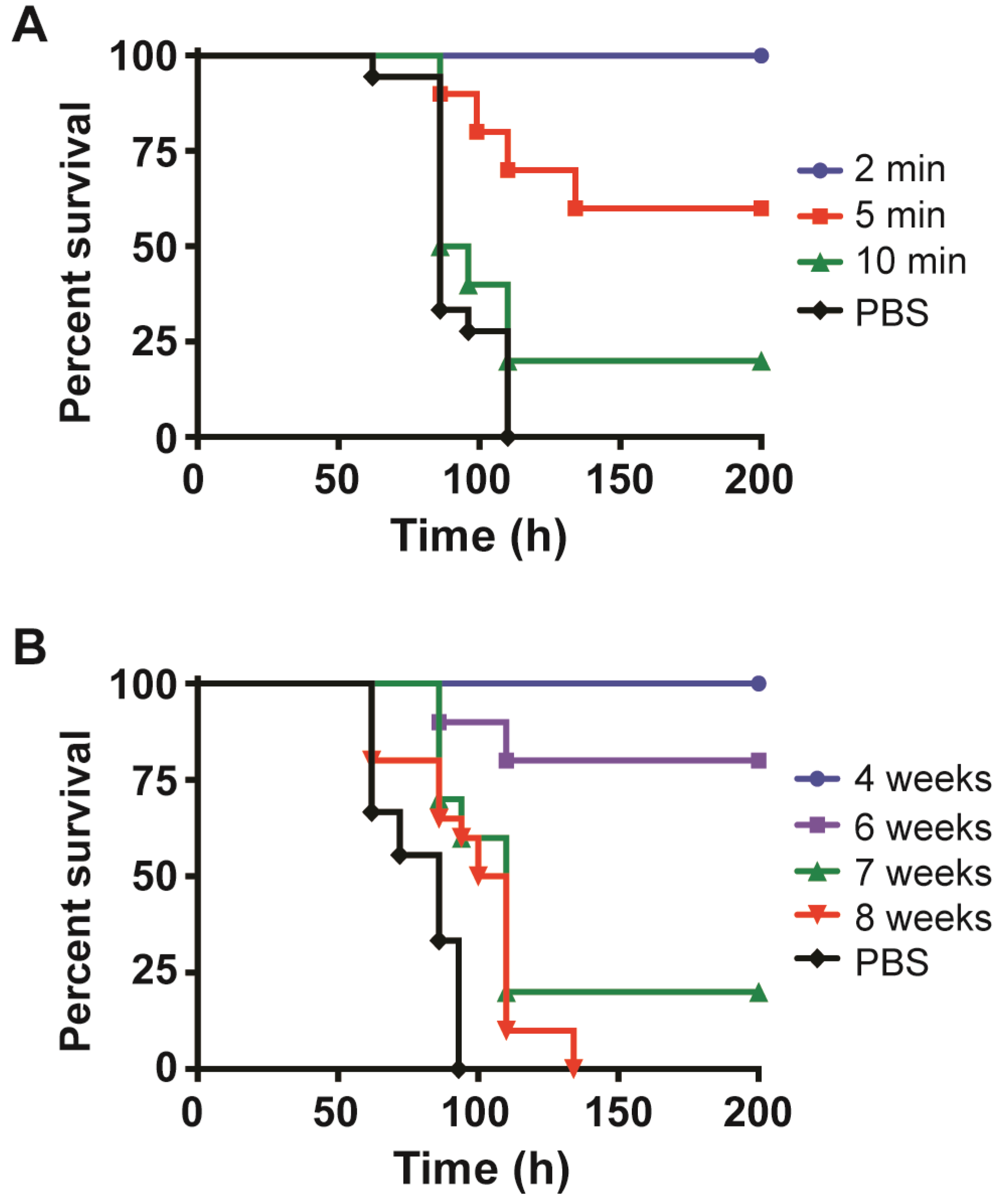
2.3. Protection of Mice from Stx2 with Monoclonal Antibodies
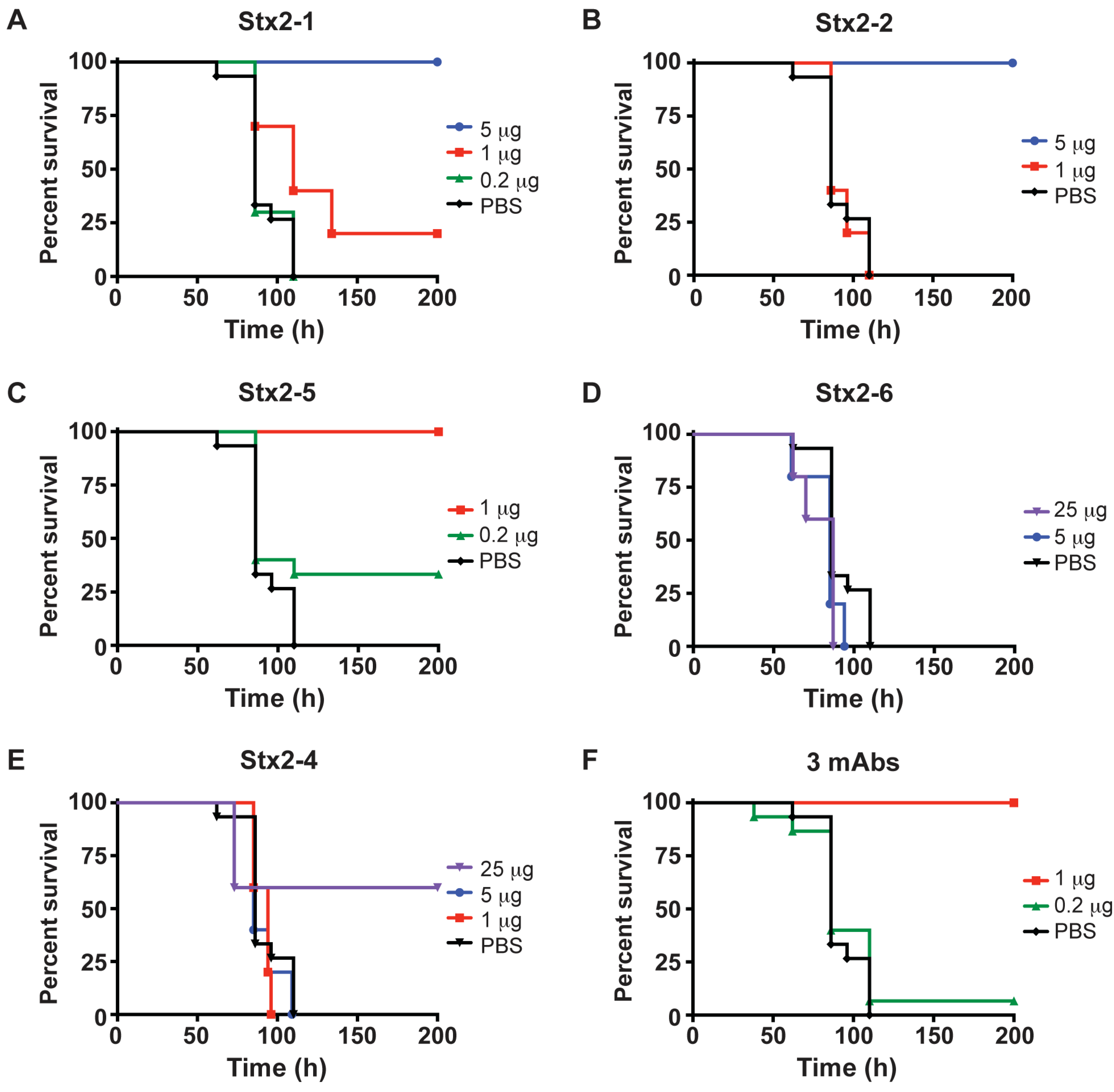
2.4. Survival of Mice Treated with mAbs before and after Intoxication with Stx2
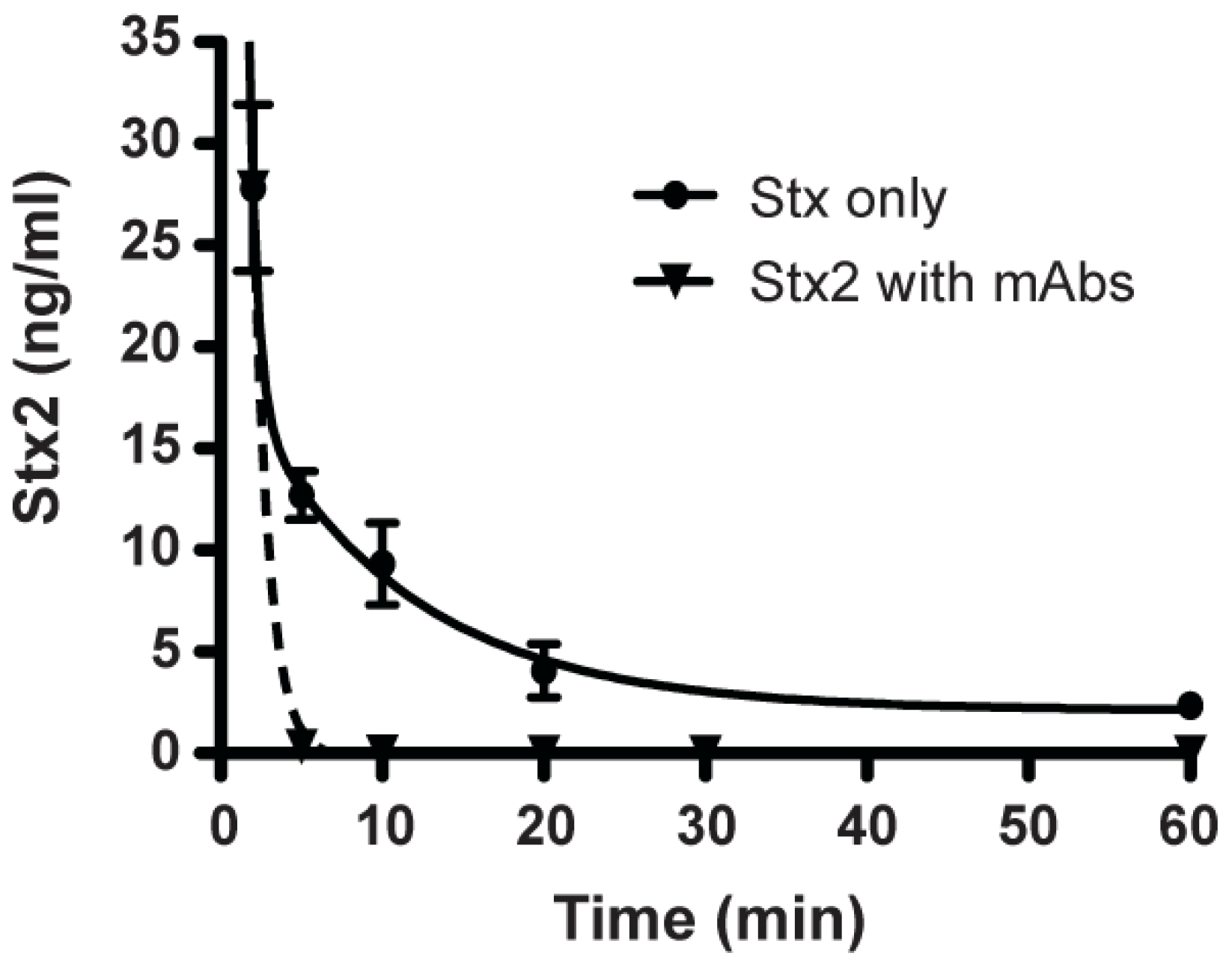
2.5. Clearance of Stx2 by Monoclonal Antibodies
3. Discussion
4. Experimental Section
4.1. Experimental Materials
4.2. Determination of Mean Lethal Dose
4.3. Mouse Protection Assay
4.4. ELISA for Stx2
4.5. Toxicokinetics of Stx2
4.6. Treatment of Mice Post-intoxication or Pre-intoxication with Stx2 mAbs
Acknowledgments
Conflicts of Interest
References
- Karrch, H.; Tarr, P.I.; Bielaszewska, M. Enterohaemorrhagic Escherichia coli in human medicine. Int. J. Med. Microbiol. 2005, 295, 405–418. [Google Scholar] [CrossRef]
- Manning, S.D.; Madera, R.T.; Schneider, W.; Dietrich, S.E.; Khalife, W.; Brown, W.; Whittam, T.S.; Somsel, P.; Rudrik, J.T. Surveillance for Shiga toxin-producing Escherichia coli, Michigan, 2001–2005. Emerg. Infect. Dis. 2007, 13, 318–321. [Google Scholar] [CrossRef]
- Trachtman, H.; Austin, C.; Lewinski, M.; Stahl, R.A. Renal and neurological involvement in typical Shiga toxin-associated HUS. Nat. Rev. Nephrol. 2012, 8, 658–669. [Google Scholar]
- Scheutz, F.; Teel, L.D.; Beutin, L.; Pierard, D.; Buvens, G.; Karch, H.; Mellmann, A.; Caprioli, A.; Tozzoli, R.; Morabito, S.; Strockbine, N.A.; Melton-Celsa, A.R.; Sanchez, M.; Persson, S.; O'Brien, A.D. Multicenter evaluation of a sequence-based protocol for subtyping Shiga toxins and standardizing Stx nomenclature. J. Clin. Microbiol. 2012, 50, 2951–2963. [Google Scholar] [CrossRef]
- O'Brien, A.D.; Marques, L.R.; Kerry, C.F.; Newland, J.W.; Holmes, R.K. Shiga-like toxin converting phage of enterohemorrhagic Escherichia coli strain 933. Microb. Pathog. 1989, 6, 381–390. [Google Scholar] [CrossRef]
- Fraser, M.E.; Fujinaga, M.; Cherney, M.M.; Melton-Celsa, A.R.; Twiddy, E.M.; O'Brien, A.D.; James, M.N. Structure of shiga toxin type 2 (Stx2) from Escherichia coli O157:H7. J. Biol. Chem. 2004, 279, 27511–27517. [Google Scholar] [CrossRef]
- Reisbig, R.; Olsnes, S.; Eiklid, K. The cytotoxic activity of Shigella toxin. Evidence for catalytic inactivation of the 60 S ribosomal subunit. J. Biol. Chem. 1981, 256, 8739–8744. [Google Scholar]
- Endo, Y.; Tsurugi, K.; Yutsudo, T.; Takeda, Y.; Ogasawara, T.; Igarashi, K. Site of action of a Vero toxin (VT2) from Escherichia coli O157:H7 and of Shiga toxin on eukaryotic ribosomes. RNA N-glycosidase activity of the toxins. Eur. J. Biochem. 1988, 171, 45–50. [Google Scholar] [CrossRef]
- Lingwood, C.A. Verotoxins and their glycolipid receptors. Adv. Lipid Res. 1993, 25, 189–211. [Google Scholar]
- DeGrandis, S.; Law, H.; Brunton, J.; Gyles, C.; Lingwood, C.A. Globotetraosylceramide is recognized by the pig edema disease toxin. J. Biol. Chem. 1989, 264, 12520–12525. [Google Scholar]
- Boerlin, P.; McEwen, S.A.; Boerlin-Petzold, F.; Wilson, J.B.; Johnson, R.P.; Gyles, C.L. Associations between virulence factors of Shiga toxin-producing Escherichia coli and disease in humans. J. Clin. Microbiol. 1999, 37, 497–503. [Google Scholar]
- Friedrich, A.W.; Bielaszewska, M.; Zhang, W.L.; Pulz, M.; Kuczius, T.; Ammon, A.; Karch, H. Escherichia coli harboring Shiga toxin 2 gene variants: frequency and association with clinical symptoms. J. Infect. Dis. 2002, 185, 74–84. [Google Scholar] [CrossRef]
- Wong, C.S.; Mooney, J.C.; Brandt, J.R.; Staples, A.O.; Jelacic, S.; Boster, D.R.; Watkins, S.L.; Tarr, P.I. Risk factors for the hemolytic uremic syndrome in children infected with Escherichia coli O157:H7: a multivariable analysis. Clin. Infect. Dis. 2012, 55, 33–41. [Google Scholar] [CrossRef]
- Corogeanu, D.; Willmes, R.; Wolke, M.; Plum, G.; Utermohlen, O.; Kronke, M. Therapeutic concentrations of antibiotics inhibit Shiga toxin release from enterohemorrhagic E. coli O104:H4 from the 2011 German outbreak. BMC Microbiol. 2012, 12, 160. [Google Scholar] [CrossRef]
- Rosales, A.; Hofer, J.; Zimmerhackl, L.B.; Jungraithmayr, T.C.; Riedl, M.; Giner, T.; Strasak, A.; Orth-Holler, D.; Wurzner, R.; Karch, H. Need for long-term follow-up in enterohemorrhagic Escherichia coli-associated hemolytic uremic syndrome due to late-emerging sequelae. Clin. Infect. Dis. 2012, 54, 1413–1421. [Google Scholar] [CrossRef]
- Wen, S.X.; Teel, L.D.; Judge, N.A.; O'Brien, A.D. Genetic toxoids of Shiga toxin types 1 and 2 protect mice against homologous but not heterologous toxin challenge. Vaccine 2006, 24, 1142–1148. [Google Scholar] [CrossRef]
- Paton, A.W.; Morona, R.; Paton, J.C. A new biological agent for treatment of Shiga toxigenic Escherichia coli infections and dysentery in humans. Nat. Med. 2000, 6, 265–270. [Google Scholar] [CrossRef]
- Bitzan, M. Treatment options for HUS secondary to Escherichia coli O157:H7. Kidney Int. Suppl. 2009, S62–S66. [Google Scholar] [CrossRef]
- Tzipori, S.; Sheoran, A.; Akiyoshi, D.; Donohue-Rolfe, A.; Trachtman, H. Antibody therapy in the management of shiga toxin-induced hemolytic uremic syndrome. Clin. Microbiol. Rev. 2004, 17, 926–941, (table of contents). [Google Scholar] [CrossRef]
- Te Loo, D.M.; van Hinsbergh, V.W.; van den Heuvel, L.P.; Monnens, L.A. Detection of verocytotoxin bound to circulating polymorphonuclear leukocytes of patients with hemolytic uremic syndrome. J. Am. Soc. Nephrol. 2001, 12, 800–806. [Google Scholar]
- Nowakowski, A.; Wang, C.; Powers, D.B.; Amersdorfer, P.; Smith, T.J.; Montgomery, V.A.; Sheridan, R.; Blake, R.; Smith, L.A.; Marks, J.D. Potent neutralization of botulinum neurotoxin by recombinant oligoclonal antibody. Proc. Natl. Acad. Sci. USA 2002, 99, 11346–11350. [Google Scholar] [CrossRef]
- Bitzan, M.; Klemt, M.; Steffens, R.; Muller-Wiefel, D.E. Differences in verotoxin neutralizing activity of therapeutic immunoglobulins and sera from healthy controls. Infection 1993, 21, 140–145. [Google Scholar] [CrossRef]
- Mohawk, K.L.; Melton-Celsa, A.R.; Robinson, C.M.; O'Brien, A.D. Neutralizing antibodies to Shiga toxin type 2 (Stx2) reduce colonization of mice by Stx2-expressing Escherichia coli O157:H7. Vaccine 2010, 28, 4777–4785. [Google Scholar] [CrossRef]
- Yamagami, S.; Motoki, M.; Kimura, T.; Izumi, H.; Takeda, T.; Katsuura, Y.; Matsumoto, Y. Efficacy of postinfection treatment with anti-Shiga toxin (Stx) 2 humanized monoclonal antibody TMA-15 in mice lethally challenged with Stx-producing Escherichia coli. J. Infect. Dis. 2001, 184, 738–742. [Google Scholar] [CrossRef]
- Jeong, K.I.; Tzipori, S.; Sheoran, A.S. Shiga toxin 2-specific but not shiga toxin 1-specific human monoclonal antibody protects piglets challenged with enterohemorrhagic Escherichia coli producing shiga toxin 1 and shiga toxin 2. J. Infect. Dis. 2010, 201, 1081–1083. [Google Scholar] [CrossRef]
- Sauter, K.A.; Melton-Celsa, A.R.; Larkin, K.; Troxell, M.L.; O'Brien, A.D.; Magun, B.E. Mouse model of hemolytic-uremic syndrome caused by endotoxin-free Shiga toxin 2 (Stx2) and protection from lethal outcome by anti-Stx2 antibody. Infect. Immun. 2008, 76, 4469–4478. [Google Scholar] [CrossRef]
- Mukherjee, J.; Chios, K.; Fishwild, D.; Hudson, D.; O'Donnell, S.; Rich, S.M.; Donohue-Rolfe, A.; Tzipori, S. Production and characterization of protective human antibodies against Shiga toxin 1. Infect. Immun. 2002, 70, 5896–5899. [Google Scholar] [CrossRef]
- Padhye, V.V.; Zhao, T.; Doyle, M.P. Production and characterisation of monoclonal antibodies to Verotoxins 1 and 2 from Escherichia coli of serotype O157:H7. J. Med. Microbiol. 1989, 30, 219–226. [Google Scholar] [CrossRef]
- Parma, Y.R.; Chacana, P.A.; Roge, A.; Kahl, A.; Cangelosi, A.; Geoghegan, P.; Lucchesi, P.M.; Fernandez-Miyakawa, M.E. Antibodies anti-Shiga toxin 2 B subunit from chicken egg yolk: isolation, purification and neutralization efficacy. Toxicon 2011, 58, 380–388. [Google Scholar] [CrossRef]
- Rutjes, N.W.; Binnington, B.A.; Smith, C.R.; Maloney, M.D.; Lingwood, C.A. Differential tissue targeting and pathogenesis of verotoxins 1 and 2 in the mouse animal model. Kidney Int. 2002, 62, 832–845. [Google Scholar] [CrossRef]
- Kimura, T.; Tani, S.; Motoki, M.; Matsumoto, Y. Role of Shiga toxin 2 (Stx2)-binding protein, human serum amyloid P component (HuSAP), in Shiga toxin-producing Escherichia coli infections: assumption from in vitro and in vivo study using HuSAP and anti-Stx2 humanized monoclonal antibody TMA-15. Biochem. Biophys. Res. Commun. 2003, 305, 1057–1060. [Google Scholar] [CrossRef]
- Jeong, K.I.; Chapman-Bonofiglio, S.; Singh, P.; Lee, J.; Tzipori, S.; Sheoran, A.S. In vitro and in vivo protective efficacies of antibodies that neutralize the RNA N-glycosidase activity of Shiga toxin 2. BMC Immunol. 2010, 11, 16. [Google Scholar] [CrossRef]
- March, S.B.; Ratnam, S. Sorbitol-MacConkey medium for detection of Escherichia coli O157:H7 associated with hemorrhagic colitis. J. Clin. Microbiol. 1986, 23, 869–872. [Google Scholar]
- He, X.; McMahon, S.; Skinner, C.; Merrill, P.; Scotcher, M.C.; Stanker, L.H. Development and characterization of monoclonal antibodies against Shiga toxin 2 and their application for toxin detection in milk. J. Immunol. Methods 2013, 389, 18–28. [Google Scholar] [CrossRef]
- Cheng, L.W.; Stanker, L.H.; Henderson, T.D., 2nd; Lou, J.; Marks, J.D. Antibody protection against botulinum neurotoxin intoxication in mice. Infect. Immun. 2009, 77, 4305–4313. [Google Scholar] [CrossRef]
- Liu, Y.N.; Wang, S.H.; Li, T.; Wang, Q.; Tu, W.; Cai, K.; Hou, X.J.; Tian, R.M.; Gao, X.; Liu, H.; Xiao, L.; Shi, J.; Cheng, Y.G.; Li, J.C.; Wang, H. Shiga toxin type 2 (Stx2), a potential agent of bioterrorism, has a short distribution and a long elimination half-life, and induces kidney and thymus lesions in rats. Arch. Toxicol. 2011, 85, 1133–1140. [Google Scholar] [CrossRef]
- Sheoran, A.; Jeong, K.I.; Mukherjee, J.; Wiffin, A.; Singh, P.; Tzipori, S. Biodistribution and elimination kinetics of systemic Stx2 by the Stx2A and Stx2B subunit-specific human monoclonal antibodies in mice. BMC Immunol. 2012, 13, 27. [Google Scholar] [CrossRef]
- Mukherjee, J.; Chios, K.; Fishwild, D.; Hudson, D.; O'Donnell, S.; Rich, S.M.; Donohue-Rolfe, A.; Tzipori, S. Human Stx2-specific monoclonal antibodies prevent systemic complications of Escherichia coli O157:H7 infection. Infect. Immun. 2002, 70, 612–619. [Google Scholar] [CrossRef]
- Johansson, A.G.; Lovdal, T.; Magnusson, K.E.; Berg, T.; Skogh, T. Liver cell uptake and degradation of soluble immunoglobulin G immune complexes in vivo and in vitro in rats. Hepatology 1996, 24, 169–175. [Google Scholar]
- Sepulveda, J.; Mukherjee, J.; Tzipori, S.; Simpson, L.L.; Shoemaker, C.B. Efficient serum clearance of botulinum neurotoxin achieved using a pool of small antitoxin binding agents. Infect. Immun. 2010, 78, 756–763. [Google Scholar] [CrossRef]
- Chow, S.K.; Smith, C.; MacCarthy, T.; Pohl, M.A.; Bergman, A.; Casadevall, A. Disease-enhancing antibodies improve the efficacy of bacterial toxin-neutralizing antibodies. Cell Host Microbe 2013, 13, 417–428. [Google Scholar] [CrossRef]
- Fuller, C.A.; Pellino, C.A.; Flagler, M.J.; Strasser, J.E.; Weiss, A.A. Shiga toxin subtypes display dramatic differences in potency. Infect. Immun. 2011, 79, 1329–1337. [Google Scholar] [CrossRef]
- Donohue-Rolfe, A.; Kondova, I.; Mukherjee, J.; Chios, K.; Hutto, D.; Tzipori, S. Antibody-based protection of gnotobiotic piglets infected with Escherichia coli O157:H7 against systemic complications associated with Shiga toxin 2. Infect. Immun. 1999, 67, 3645–3648. [Google Scholar]
- Vieira, P.; Rajewsky, K. The half-lives of serum immunoglobulins in adult mice. Eur. J. Immunol. 1988, 18, 313–316. [Google Scholar] [CrossRef]
- Ariceta, G.; Besbas, N.; Johnson, S.; Karpman, D.; Landau, D.; Licht, C.; Loirat, C.; Pecoraro, C.; Taylor, C.M.; Van de Kar, N.; Vandewalle, J.; Zimmerhackl, L.B. Guideline for the investigation and initial therapy of diarrhea-negative hemolytic uremic syndrome. Pediatr. Nephrol. 2009, 24, 687–696. [Google Scholar] [CrossRef]
- Tarr, P.I. Escherichia coli O157:H7: clinical, diagnostic, and epidemiological aspects of human infection. Clin. Infect. Dis. 1995, 20, 1–8, quiz 9–10. [Google Scholar] [CrossRef]
- Safdar, N.; Said, A.; Gangnon, R.E.; Maki, D.G. Risk of hemolytic uremic syndrome after antibiotic treatment of Escherichia coli O157:H7 enteritis: a meta-analysis. JAMA 2002, 288, 996–1001. [Google Scholar] [CrossRef]
- Reed, L.J.; Muench, H. A simple method of estimating fifty per cent endpoints. Am. J. Hyg. 1938, 27, 493. [Google Scholar]
- Weil, C.S. Tables for the convenient calculation of median-effective dose (LD50 or ED50) and instructions in their use. Biometrices 1952, 8, 249–263. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cheng, L.W.; Henderson, T.D., II; Patfield, S.; Stanker, L.H.; He, X. Mouse in Vivo Neutralization of Escherichia coli Shiga Toxin 2 with Monoclonal Antibodies. Toxins 2013, 5, 1845-1858. https://doi.org/10.3390/toxins5101845
Cheng LW, Henderson TD II, Patfield S, Stanker LH, He X. Mouse in Vivo Neutralization of Escherichia coli Shiga Toxin 2 with Monoclonal Antibodies. Toxins. 2013; 5(10):1845-1858. https://doi.org/10.3390/toxins5101845
Chicago/Turabian StyleCheng, Luisa W., Thomas D. Henderson, II, Stephanie Patfield, Larry H. Stanker, and Xiaohua He. 2013. "Mouse in Vivo Neutralization of Escherichia coli Shiga Toxin 2 with Monoclonal Antibodies" Toxins 5, no. 10: 1845-1858. https://doi.org/10.3390/toxins5101845
APA StyleCheng, L. W., Henderson, T. D., II, Patfield, S., Stanker, L. H., & He, X. (2013). Mouse in Vivo Neutralization of Escherichia coli Shiga Toxin 2 with Monoclonal Antibodies. Toxins, 5(10), 1845-1858. https://doi.org/10.3390/toxins5101845





