Protein-Bound Uremic Toxins: New Insight from Clinical Studies
Abstract
:1. Introduction
2. Indoxyl Sulfate and p-Cresyl Sulfate Formation
3. Indoxyl Sulfate, p-Cresyl Sulfate and Renal Toxicity
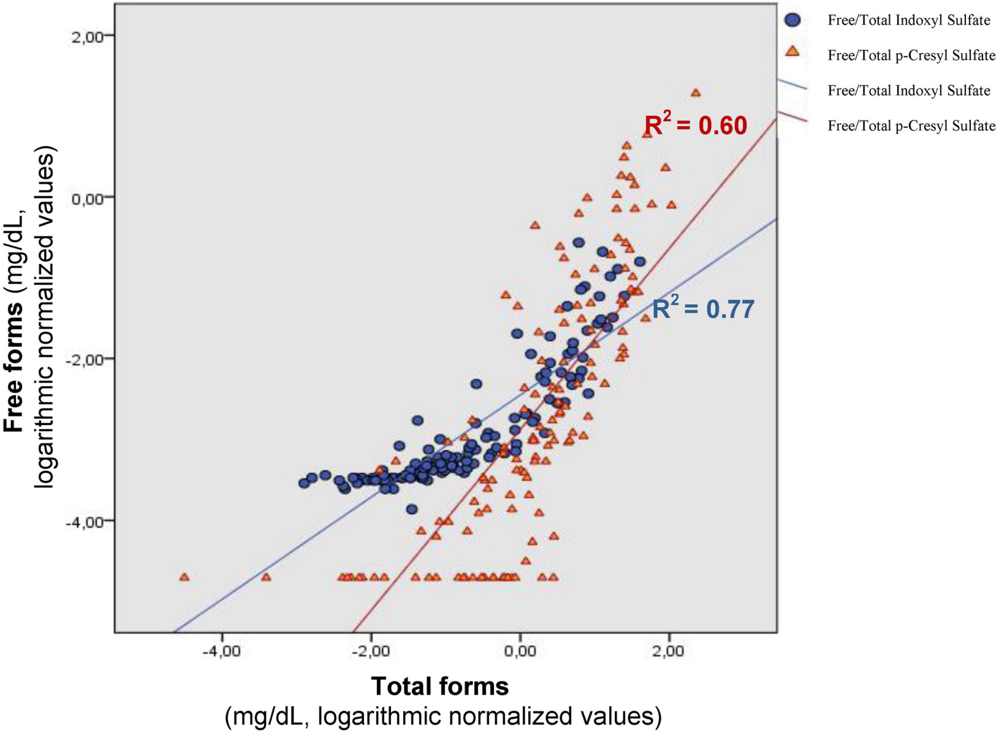
4. Indoxyl Sulfate, p-Cresyl Sulfate and Vascular Toxicity
5. Indoxyl Sulfate, p-Cresyl Sulfate and Clinical Outcomes
| Free Indoxyl Sulfate | p | |||
| Total | ≤0.038 mg/100 mL (n = 70) | >0.038 mg/100 mL (n = 69) | ||
| Binding (%) | 89 ± 8 (88.9) | 83 ± 9 (86) | 91 ± 5 (92) | <0.001 |
| Free p-Cresyl Sulfate | p | |||
| Total | ≤0.051 mg/100 mL ( n = 70) | >0.051 mg/100 mL ( n = 69) | ||
| Binding (%) | 91 ± 11 (95.8) | 97 ± 4 (97.5) | 85 ± 13 (90.2) | <0.01 |
6. Conclusions
References
- Vanholder, R.; de Smet, R. Pathophysiologic effects of uremic retention solutes. J. Am. Soc. Nephrol. 1999, 10, 1815–1823. [Google Scholar]
- Vanholder, R.; de Smet, R.; Hsu, C.; Vogeleere, P.; Ringoir, S. Uremic toxicity: The middle molecule hypothesis revisited. Semin. Nephrol. 1994, 14, 205–218. [Google Scholar]
- Vanholder, R.; Baurmeister, U.; Brunet, P.; Cohen, G.; Glorieux, G.; Jankowski, J. A bench to bedside view of uremic toxins. J. Am. Soc. Nephrol. 2008, 19, 863–870. [Google Scholar]
- Vanholder, R.; van Laecke, S.; Glorieux, G. What is new in uremic toxicity? Pediatr. Nephrol. 2008, 23, 1211–1221. [Google Scholar] [CrossRef] [PubMed]
- Liabeuf, S.; Barreto, D.V.; Barreto, F.C.; Meert, N.; Glorieux, G.; Schepers, E.; Temmar, M.; Choukroun, G.; Vanholder, R.; Massy, Z.A. Free p-cresylsulphate is a predictor of mortality in patients at different stages of chronic kidney disease. Nephrol. Dial. Transplant. 2010, 25, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Barreto, F.C.; Barreto, D.V.; Liabeuf, S.; Meert, N.; Glorieux, G.; Temmar, M.; Choukroun, G.; Vanholder, R.; Massy, Z.A. Serum indoxyl sulfate is associated with vascular disease and mortality in chronic kidney disease patients. Clin. J. Am. Soc. Nephrol. 2009, 4, 1551–1558. [Google Scholar]
- Meijers, B.K.; Claes, K.; Bammens, B.; de Loor, H.; Viaene, L.; Verbeke, K.; Kuypers, D.; Vanrenterghem, Y.; Evenepoel, P. p-Cresol and cardiovascular risk in mild-to-moderate kidney disease. Clin. J. Am. Soc. Nephrol. 2010, 5, 1182–1189. [Google Scholar] [CrossRef] [PubMed]
- Vanholder, R.; Massy, Z.; Argiles, A.; Spasovski, G.; Verbeke, F.; Lameire, N. Chronic kidney disease as cause of cardiovascular morbidity and mortality. Nephrol., Dial., Transplant. 2005, 20, 1048–1056. [Google Scholar] [CrossRef]
- Dou, L.; Bertrand, E.; Cerini, C.; Faure, V.; Sampol, J.; Vanholder, R.; Berland, Y.; Brunet, P. The uremic solutes p-cresol and indoxyl sulfate inhibit endothelial proliferation and wound repair. Kidney Int. 2004, 65, 442–451. [Google Scholar] [CrossRef] [PubMed]
- Meijers, B.K.; Bammens, B.; de Moor, B.; Verbeke, K.; Vanrenterghem, Y.; Evenepoel, P. Free p-cresol is associated with cardiovascular disease in hemodialysis patients. Kidney Int. 2008, 73, 1174–1180. [Google Scholar] [CrossRef] [PubMed]
- Bammens, B.; Evenepoel, P.; Keuleers, H.; Verbeke, K.; Vanrenterghem, Y. Free serum concentrations of the protein-bound retention solute p-cresol predict mortality in hemodialysis patients. Kidney Int. 2006, 69, 1081–1087. [Google Scholar]
- Lin, C.J.; Wu, C.J.; Pan, C.F.; Chen, Y.C.; Sun, F.J.; Chen, H.H. Serum protein-bound uraemic toxins and clinical outcomes in haemodialysis patients. Nephrol. Dial. Transplant. 2010, 25, 3693–3700. [Google Scholar]
- De Smet, R.; van Kaer, J.; van Vlem, B.; de Cubber, A.; Brunet, P.; Lameire, N.; Vanholder, R. Toxicity of free p-cresol: A prospective and cross-sectional analysis. Clin. Chem. 2003, 49, 470–478. [Google Scholar] [CrossRef] [PubMed]
- Curtius, H.C.; Mettler, M.; Ettlinger, L. Study of the intestinal tyrosine metabolism using stable isotopes and gas chromatography-mass spectrometry. J. Chromatogr. 1976, 126, 569–580. [Google Scholar]
- Burchell, B.; Coughtrie, M.W. Genetic and environmental factors associated with variation of human xenobiotic glucuronidation and sulfation. Environ. Health Perspect. 1997, 105, 739–747. [Google Scholar]
- Vanholder, R.; Bammens, B.; de Loor, H.; Glorieux, G.; Meijers, B.; Schepers, E.; Massy, Z.; Evenepoel, P. Warning: The unfortunate end of p-cresol as a uraemic toxin. Nephrol. Dial. Transplant. 2011, 26, 1464–1467. [Google Scholar] [CrossRef] [PubMed]
- Meert, N.; Eloot, S.; Waterloos, M.A.; van Landschoot, M.; Dhondt, A.; Glorieux, G.; Ledebo, I.; Vanholder, R. Effective removal of protein-bound uraemic solutes by different convective strategies: A prospective trial. Nephrol. Dial. Transplant. 2009, 24, 562–570. [Google Scholar]
- Niwa, T. Phenol and p-cresol accumulated in uremic serum measured by HPLC with fluorescence detection. Clin. Chem. 1993, 39, 108–111. [Google Scholar] [PubMed]
- De Smet, R.; David, F.; Sandra, P.; van Kaer, J.; Lesaffer, G.; Dhondt, A.; Lameire, N.; Vanholder, R. A sensitive HPLC method for the quantification of free and total p-cresol in patients with chronic renal failure. Clin. Chim. Acta 1998, 278, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Niwa, T.; Ise, M. Indoxyl sulfate, a circulating uremic toxin, stimulates the progression of glomerular sclerosis. J. Lab. Clin. Med. 1994, 124, 96–104. [Google Scholar] [PubMed]
- Miyazaki, T.; Ise, M.; Hirata, M.; Endo, K.; Ito, Y.; Seo, H.; Niwa, T. Indoxyl sulfate stimulates renal synthesis of transforming growth factor-beta 1 and progression of renal failure. Kidney Int., Suppl. 1997, 63, S211–S214. [Google Scholar]
- Enomoto, A.; Takeda, M.; Tojo, A.; Sekine, T.; Cha, S.H.; Khamdang, S.; Takayama, F.; Aoyama, I.; Nakamura, S.; Endou, H.; et al. Role of organic anion transporters in the tubular transport of indoxyl sulfate and the induction of its nephrotoxicity. J. Am. Soc. Nephrol. 2002, 13, 1711–1720. [Google Scholar] [PubMed]
- Adijiang, A.; Shimizu, H.; Higuchi, Y.; Nishijima, F.; Niwa, T. Indoxyl sulfate reduces klotho expression and promotes senescence in the kidneys of hypertensive rats. J. Ren. Nutr. 2011, 21, 105–109. [Google Scholar]
- Cerini, C.; Dou, L.; Anfosso, F.; Sabatier, F.; Moal, V.; Glorieux, G.; de Smet, R.; Vanholder, R.; Dignat-George, F.; Sampol, J.; et al. P-cresol, a uremic retention solute, alters the endothelial barrier function in vitro. Thromb. Haemost 2004, 92, 140–150. [Google Scholar] [PubMed]
- Schepers, E.; Meert, N.; Glorieux, G.; Goeman, J.; van der Eycken, J.; Vanholder, R. P-cresylsulphate, the main in vivo metabolite of p-cresol, activates leucocyte free radical production. Nephrol. Dial. Transplant. 2007, 22, 592–596. [Google Scholar] [PubMed]
- Yu, M.; Kim, Y.J.; Kang, D.H. Indoxyl sulfate-induced endothelial dysfunction in patients with chronic kidney disease via an induction of oxidative stress. Clin. J. Am. Soc. Nephrol. 2011, 6, 30–39. [Google Scholar]
- Miyamoto, Y.; Watanabe, H.; Otagiri, M.; Maruyama, T. New insight into the redox properties of uremic solute indoxyl sulfate as a pro- and anti-oxidant. Ther. Apher. Dial. 2011, 15, 129–131. [Google Scholar]
- Fujii, H.; Nishijima, F.; Goto, S.; Sugano, M.; Yamato, H.; Kitazawa, R.; Kitazawa, S.; Fukagawa, M. Oral charcoal adsorbent (AST-120) prevents progression of cardiac damage in chronic kidney disease through suppression of oxidative stress. Nephrol. Dial. Transplant. 2009, 24, 2089–2095. [Google Scholar]
- Nakamura, T.; Kawagoe, Y.; Matsuda, T.; Ueda, Y.; Shimada, N.; Ebihara, I.; Koide, H. Oral ADSORBENT AST-120 decreases carotid intima-media thickness and arterial stiffness in patients with chronic renal failure. Kidney Blood Pressure Res. 2004, 27, 121–126. [Google Scholar]
- Wu, I.W.; Hsu, K.H.; Lee, C.C.; Sun, C.Y.; Hsu, H.J.; Tsai, C.J.; Tzen, C.Y.; Wang, Y.C.; Lin, C.Y.; Wu, M.S. P-Cresyl sulphate and indoxyl sulphate predict progression of chronic kidney disease. Nephrol. Dial. Transplant. 2011, 26, 938–947. [Google Scholar] [CrossRef] [PubMed]
- Meijers, B.K.; de Loor, H.; Bammens, B.; Verbeke, K.; Vanrenterghem, Y.; Evenepoel, P. P-Cresyl sulfate and indoxyl sulfate in hemodialysis patients. Clin. J. Am. Soc. Nephrol. 2009, 4, 1932–1938. [Google Scholar] [CrossRef] [PubMed]
- Berge-Lefranc, D.; Chaspoul, F.; Calaf, R.; Charpiot, P.; Brunet, P.; Gallice, P. Binding of p-cresylsulfate and p-cresol to human serum albumin studied by microcalorimetry. J. Phys. Chem., B 2010, 114, 1661–1665. [Google Scholar]
- Ueda, H.; Shibahara, N.; Takagi, S.; Inoue, T.; Katsuoka, Y. AST-120 treatment in pre-dialysis period affects the prognosis in patients on hemodialysis. Renal Fail. 2008, 30, 856–860. [Google Scholar]
- Kikuchi, K.; Itoh, Y.; Tateoka, R.; Ezawa, A.; Murakami, K.; Niwa, T. Metabolomic search for uremic toxins as indicators of the effect of an oral sorbent AST-120 by liquid chromatography/tandem mass spectrometry. J. Chromatogr., B: Anal. Technol. Biomed. Life Sci. 2010, 878, 2997–3002. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liabeuf, S.; Drüeke, T.B.; Massy, Z.A. Protein-Bound Uremic Toxins: New Insight from Clinical Studies. Toxins 2011, 3, 911-919. https://doi.org/10.3390/toxins3070911
Liabeuf S, Drüeke TB, Massy ZA. Protein-Bound Uremic Toxins: New Insight from Clinical Studies. Toxins. 2011; 3(7):911-919. https://doi.org/10.3390/toxins3070911
Chicago/Turabian StyleLiabeuf, Sophie, Tilman B. Drüeke, and Ziad A. Massy. 2011. "Protein-Bound Uremic Toxins: New Insight from Clinical Studies" Toxins 3, no. 7: 911-919. https://doi.org/10.3390/toxins3070911






