From Toxins Targeting Ligand Gated Ion Channels to Therapeutic Molecules
Abstract
:1. Introduction and Scope of the Review
1.1. To be Poisonous or Not: The Dose Effect
1.2. Toxins as Biological Poisons
1.3. Scope of the Review
2. Toxins Targeting the Ligand Gated Ion Channels
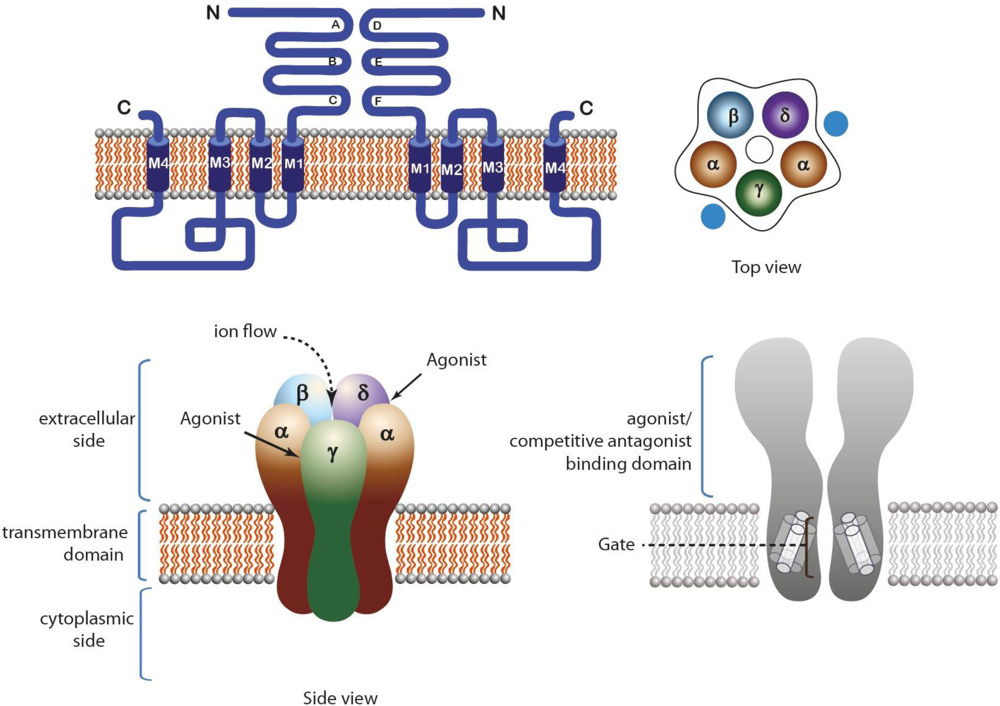
2.1. Nicotinic Acetylcholine Receptors

| Name | Conus specie | Target | References |
|---|---|---|---|
| GI, GIA, GII | geographus | Muscle nAChR | [27] |
| MI | magus | Muscle nAChR | [28] |
| SI, SIA and SII | striatus | Selectivity for the distinct interfaces (α/γ or α/δ) of the muscle-type nAChR | [28,29] |
| ImI, ImII | imperialis | Selective for α7 nAChR but also effective on α3β4, α3β2 | [30] |
| BuIA | bullatus | Highest potency for α3- and α6−containing nAChRs | [31] |
| CnIA, CnIB | consors | Muscle nAChR | [32] |
| Ac1.1a, Ac1.1b | achatinus | Muscle nAChR | [33] |
| EI | ermineus | Selective for muscle nAChR, also effective on α3β4, α4β2 | [34] |
| PnIB, (A10L)-PnIA | pennaceus | Selective for α7, α3β4, α3β2 nAChR | [35,36] |
| GIC | geographus | Selective for α3β2 | [37] |
| MII | magnus | Selective for α3β2, α3β2β3, α6* nAChR | [38,39] |
| PIA | purpurascens | Selective for α6β2, α6β4, α6α3β2(β3), α6α3β4 | [40] |
| PIB | purpurascens | Muscle nAChR | [41] |
| GID | geographus | α7, α3β2, α4β2 | [42,43] |
| AuIA, AuIB and AuIC | aulicus | Selectively blocks α3β4 nAChRs | [44] |
| EPI | episcopatus | Selective for α7, α3β2, α3β4 | [45,46] |
| AnIB | anemone | α7, α3β2 | [47] |
| Vc1.1 | victoriae | α9, α3β4, α3(α5)β2 | [48,49] |
| ArIA, ArIB | arenatus | α7, α3β2α6α3β2β3 | [50] |
| PeIA | pergrandis | α9α10,α6α3β2β3,α3β2 | [51] |
| OmIA | omaria | α7, α3β2 | [52] |
| TxIA | textile | α3β2 | [53] |
| Lp1.1 | leopardus | α3β2,α6α3β2 | [54] |
| SrIA, SrIB | spurious | α4β2, muscle type nAChRs | [55] |
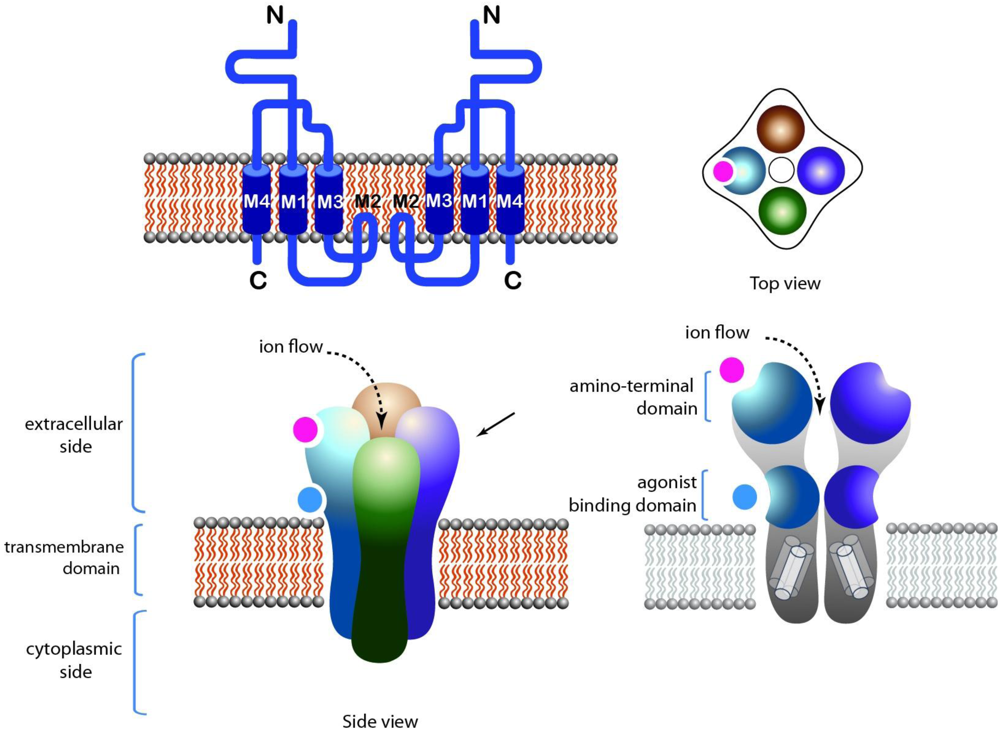
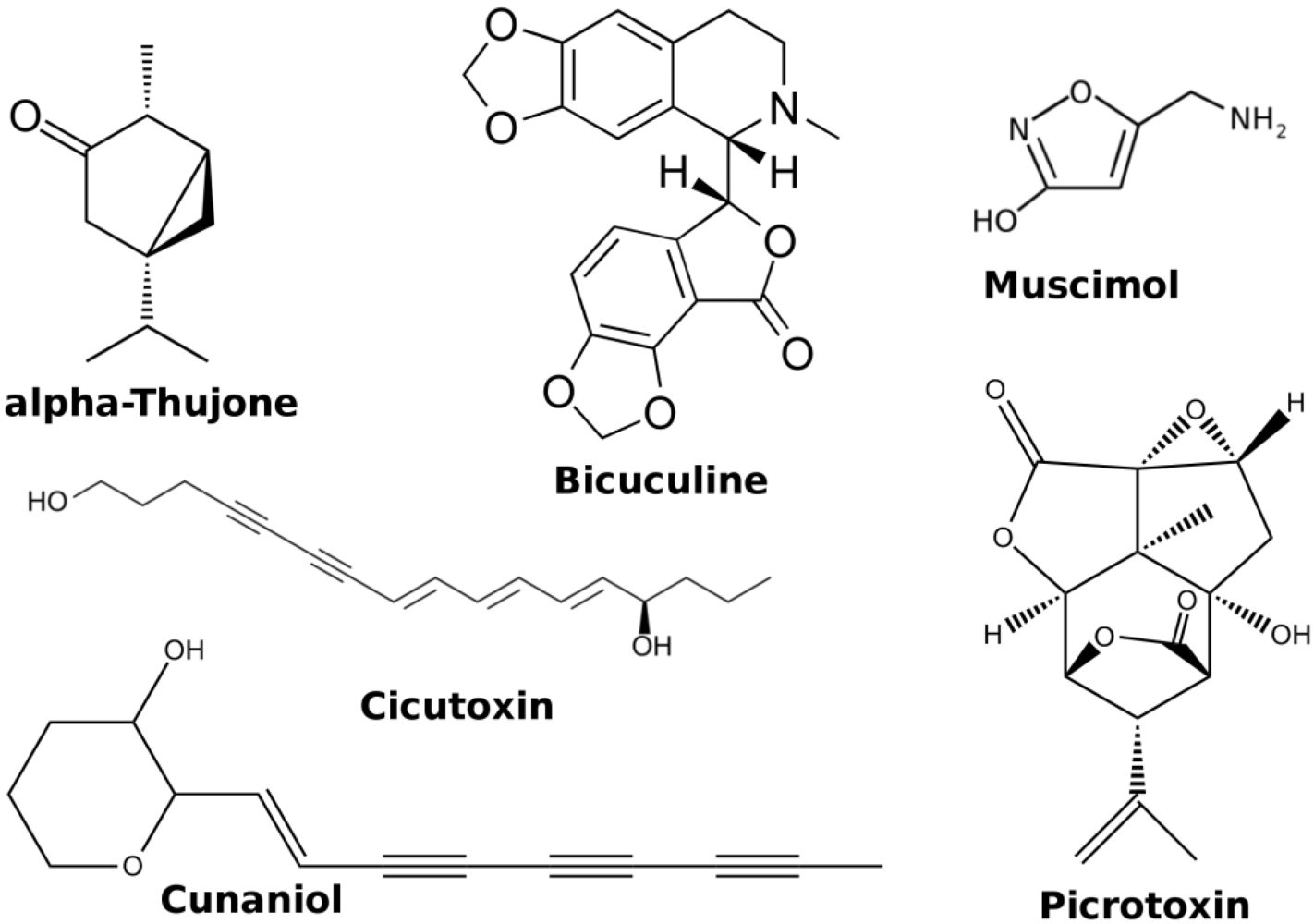
2.2. GABA-A Receptors
2.3. Glycine Receptors
2.4. Serotonin Receptors
2.5. NMDA Receptors
2.6. AMPA Receptors
2.7. Kainate Receptors
2.8. P2X Receptors

3. Current and Proposed Medical Use of Toxins Targeting the LGIC
3.1. nAChR
3.1.1. Agonists
3.1.2. Competitive Antagonists
3.1.3. Non-Competitive Antagonists
3.2. Other Pentameric Ligand Gated Ion Channels
3.3. NMDA Receptors
3.4. AMPA Receptors
3.5. Kainate Receptors
3.6. P2X

4. Conclusion
Appendix: List of Toxins Targeting the LGIC
1. Nicotinic Acetylcholine Receptors
1.1. Peptides and Proteins
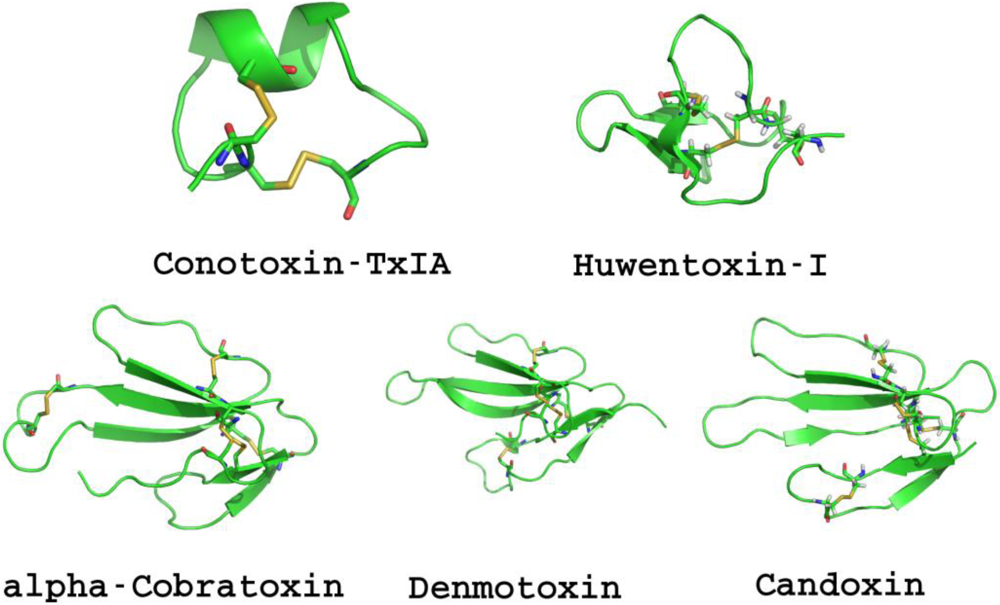
1.1.1. Conotoxins
| Name | Conus Specie | Target | References |
|---|---|---|---|
| GI, GIA, GII | geographus | Muscle nAChR | [27] |
| MI | magus | Muscle nAChR | [28] |
| SI, SIA and SII | striatus | Selectivity for the distinct interfaces (α/γ or α/δ) of the muscle-type nAChR | [28,29] |
| ImI, ImII | imperialis | Selective for α7 nAChR but also effective on α3β4, α3β2 | [30] |
| BuIA | bullatus | Highest potency for α3- and α6−containing nAChRs | [31] |
| CnIA, CnIB | consors | Muscle nAChR | [32] |
| Ac1.1a, Ac1.1b | achatinus | Muscle nAChR | [33] |
| EI | ermineus | Selective for muscle nAChR, also effective on α3β4, α4β2 | [34] |
| PnIB, (A10L)-PnIA | pennaceus | Selective for α7, α3β4, α3β2 nAChR | [35,36] |
| GIC | geographus | Selective for α3β2 | [37] |
| MII | magnus | Selective for α3β2, α3β2β3, α6∗ nAChR | [38,39] |
| PIA | purpurascens | Selective for α6β2, α6β4, α6α3β2(β3), α6α3β4 | [40] |
| PIB | purpurascens | Muscle nAChR | [41] |
| GID | geographus | α7, α3β2, α4β2 | [42,43] |
| AuIA, AuIB and AuIC | aulicus | Selectively blocks α3β4 nAChRs | [44] |
| EPI | episcopatus | Selective for α7, α3β2, α3β4 | [45,46] |
| AnIB | anemone | α7, α3β2 | [47] |
| Vc1.1 | victoriae | α9, α3β4, α3(α5)β2 | [48,49] |
| ArIA, ArIB | arenatus | α7, α3β2, α6α3β2β3 | [50] |
| PeIA | pergrandis | α9α10,α6α3β2β3,α3β2 | [51] |
| OmIA | omaria | α7, α3β2 | [52] |
| TxIA | textile | α3β2 | [53] |
| Lp1.1 | leopardus | α3β2,α6α3β2 | [54] |
| SrIA, SrIB | spurious | α4β2, muscle type nAChRs | [55] |
1.1.2. Snake Neurotoxins
| Name | Source | Target | Structural Group | References |
|---|---|---|---|---|
| α-Bungarotoxin | Bungarus multicinctus | Muscle, α7. | Long-chain α-neurotoxin | [128,129] |
| NmmI | Naja mossambica mossambica | Muscle | short-chain α-neurotoxin | [130] |
| κ−bungarotoxins | Bungarus genus | α3β2 and other β2-containing nAChRs. | long-chain neurotoxin | [131] |
| α-cobratoxin | Naja genus (e.g., Naja kaouthia) | Muscle, α7 | long-chain α-neurotoxins | [132] |
| Erabutoxin-a. | Laticauda semifasciata | Muscle | Short-chain α-toxin | |
| erabutoxin-b | Laticauda semifasciata | Muscle | short-chain neurotoxin | [133] |
| Toxin-α | Naja nigricollis | Muscle | short-chain α-neurotoxin |
1.1.3. Natural Toxic Peptides from Other Species
| Name | Origin | Target | Other specifications | References |
|---|---|---|---|---|
| Candoxin | Bungarus candidus | α7 and muscle. | [ 139] | |
| CM-11, CM-2 | Naja haje haje (Egyptian cobra) | Muscle | [ 140] | |
| CM10, CM12, CM-13b, CM-14 | Naja haje annulifera | Neuromuscular Junction | [ 141] | |
| Cm-9a | N. kaouthia | |||
| S4C11 | N. melanoleucaI. | neurotoxin Homologue | [ 142] | |
| S5C1, S5C10 | Dendroaspis jamesoni kaimosae (Eastern Jameson's mamba) | Muscle | [ 143] | |
| S6C4 | Dendroaspis jamesoni Kaimosae (jameson’s mamba) | 67% sequence identity with Bucandin. | ||
| γ-bungarotoxin | Bungarus multicinctus | post synaptic action | [ 144] | |
| WTX | Naja kaouthia | α7 and muscle. | [ 138] | |
| Wntx-5 | Naja sputatrix | Torpedo α1βγδ, chick α7 | [ 145] | |
| NNA2 | the Taiwan cobra (N. n. atra) | Muscle | long-neurotoxin homologue | [ 146] |
| LSIII | Laticauda Semifasciata | neuromuscular blockade | [ 147] |
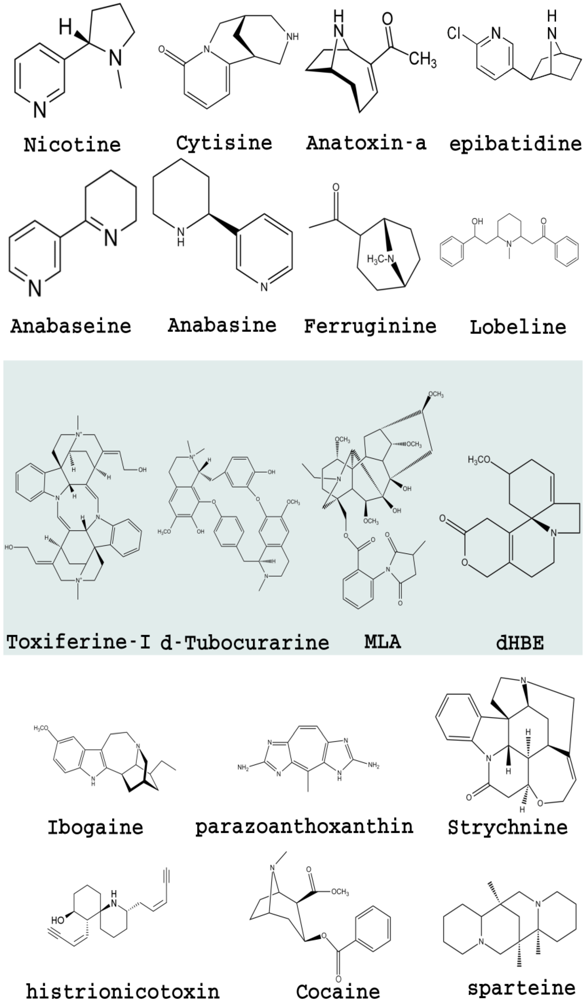
1.2. Alkaloids
1.3. Others
2. GABA-A Receptors
3. Glycine Receptors
4. Serotonin Receptors
5. NMDA
6. AMPA
7. Kainate Receptors
8. P2X Receptors
References
- Bisset, N.G. One man's poison, another man's medicine? J. Ethnopharmacol. 1991, 32, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Rainsford, K.D. Ibuprofen: Pharmacology, efficacy and safety. Inflammopharmacology 2009, 17, 275–342. [Google Scholar]
- Livett, B.G.; Sandall, D.W.; Keays, D.; Down, J.; Gayler, K.R.; Satkunanathan, N.; Khalil, Z. Therapeutic applications of conotoxins that target the neuronal nicotinic acetylcholine receptor. Toxicon 2006, 48, 810–829. [Google Scholar]
- Lewis, R.J.; Garcia, M.L. Therapeutic potential of venom peptides. Nat. Rev. Drug. Discov. 2003, 2, 790–802. [Google Scholar]
- Brieger, L. Zur Kenntniss der Aetiologie des Wundstarrkrampfes nebst Bemerkungen über das Choleraroth. Dtsch. Med. Wochenschr 1887, 13, 303–305. [Google Scholar]
- Pastan, I.; Chaudhary, V.; FitzGerald, D.J. Recombinant toxins as novel therapeutic agents. Annu. Rev. Biochem. 1992, 61, 331–354. [Google Scholar]
- Changeux, J.P.; Taly, A. Nicotinic receptors, allosteric proteins and medicine. Trends. Mol. Med. 2008, 14, 93–102. [Google Scholar]
- Collingridge, G.L.; Olsen, R.W.; Peters, J.; Spedding, M. A nomenclature for ligand-gated ion channels. Neuropharmacology 2009, 56, 2–5. [Google Scholar]
- Karlin, A. Emerging structure of the nicotinic acetylcholine receptors. Nat. Rev. Neurosci. 2002, 3, 102–114. [Google Scholar]
- Le Novere, N.; Changeux, J.P. LGICdb: The ligand-gated ion channel database. Nucleic. Acids. Res. 2001, 29, 294–295. [Google Scholar]
- Baenziger, J.E.; Corringer, P.J. 3D structure and allosteric modulation of the transmembrane domain of pentameric ligand-gated ion channels. Neuropharmacology 2011, 60, 116–125. [Google Scholar]
- Traynelis, S.F.; Wollmuth, L.P.; McBain, C.J.; Menniti, F.S.; Vance, K.M.; Ogden, K.K.; Hansen, K.B.; Yuan, H.; Myers, S.J.; Dingledine, R. Glutamate receptor ion channels: Structure, regulation, and function. Pharmacol. Rev. 2010, 62, 405–496. [Google Scholar] [CrossRef] [PubMed]
- Bernard, C. Leçons sur les effets des substances toxiques et médicamenteuses; Collège de France: Paris, France, 1857. [Google Scholar]
- Angenot, L.; Denoel, A.; Goffart, M. Curare-like effect of African Strychnos: Strychnos usambarensis Gilg du Rwanda. J. Pharm. Belg. 1970, 25, 73–77. [Google Scholar]
- Bisset, N.G. War and hunting poisons of the New World. Part 1. Notes on the early history of curare. J. Ethnopharmacol. 1992, 36, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Langley, J.N. On nerve ending and on special excitable substances in cells. Proc. Roy. Soc. Ser. B 1906, 78, 170–194. [Google Scholar]
- Changeux, J.-P.; Edelstein, S.J. Nicotinic Acetylcholine Receptors: From Molecular Biology to Cognition; Odile Jacob: New York, NY, USA, 2005; p. 284. [Google Scholar]
- Changeux, J.P.; Kasai, M.; Huchet, M.; Meunier, J.C. Extraction from electric tissue of gymnotus of a protein presenting several typical properties characteristic of the physiological receptor of acetylcholine. C. R. Acad. Sci. Hebd. Seances. Acad. Sci. D 1970, 270, 2864–2867. [Google Scholar]
- Changeux, J.P.; Podleski, T.R.; Wofsy, L. Affinity labeling of the acetylcholine-receptor. Proc. Natl. Acad. Sci. USA 1967, 58, 2063–2070. [Google Scholar]
- Favreau, P.; Le Gall, F.; Benoit, E.; Molgo, J. A review on conotoxins targeting ion channels and acetylcholine receptors of the vertebrate neuromuscular junction. Acta. Physiol. Pharmacol. Ther. Latinoam. 1999, 49, 257–267. [Google Scholar]
- Vogel, Z.; Sytkowski, A.J.; Nirenberg, M.W. Acetylcholine receptors of muscle grown in vitro. Proc. Natl. Acad. Sci. USA 1972, 69, 3180–3184. [Google Scholar] [CrossRef]
- Morris, C.E.; Harrison, J.B. Central nervous system features of a nicotine-resistant insect, the tobacco hornworm Manduca sexta. Tissue Cell 1984, 16, 601–612. [Google Scholar]
- Dale, H.H. The action of certain esters and ethers of choline, and their relation to muscarine. J. Pharmacol. Exp. Ther 1914, 6, 147–190. [Google Scholar]
- Tomizawa, M.; Casida, J.E. Molecular recognition of neonicotinoid insecticides: The determinants of life or death. Acc. Chem. Res 2009, 42, 260–269. [Google Scholar]
- Matolcsy, G.; Nádasy, M.; Andriska, V. Pesticide Chemistry; Elsevier: Amsterdam, The Netherlands, 1988; p. 808. [Google Scholar]
- Maskos, U.; Molles, B.E.; Pons, S.; Besson, M.; Guiard, B.P.; Guilloux, J.P.; Evrard, A.; Cazala, P.; Cormier, A.; Mameli-Engvall, M.; Dufour, N.; Cloez-Tayarani, I.; Bemelmans, A.P.; Mallet, J.; Gardier, A.M.; David, V.; Faure, P.; Granon, S.; Changeux, J.P. Nicotine reinforcement and cognition restored by targeted expression of nicotinic receptors. Nature 2005, 436, 103–107. [Google Scholar]
- Marshall, I.G.; Harvey, A.L. Selective neuromuscular blocking properties of alpha-conotoxins in vivo. Toxicon 1990, 28, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Arias, H.R.; Blanton, M.P. Alpha-conotoxins. Int. J. Biochem. Cell Biol. 2000, 32, 1017–1028. [Google Scholar]
- McIntosh, J.M.; Santos, A.D.; Olivera, B.M. Conus peptides targeted to specific nicotinic acetylcholine receptor subtypes. Annu. Rev. Biochem. 1999, 68, 59–88. [Google Scholar]
- Ellison, M.; McIntosh, J.M.; Olivera, B.M. Alpha-conotoxins ImI and ImII. Similar alpha. 7 nicotinic receptor antagonists act at different sites. J. Biol. Chem. 2003, 278, 757–764. [Google Scholar] [PubMed]
- Azam, L.; Dowell, C.; Watkins, M.; Stitzel, J.A.; Olivera, B.M.; McIntosh, J.M. Alpha-conotoxin BuIA, a novel peptide from Conus bullatus, distinguishes among neuronal nicotinic acetylcholine receptors. J. Biol. Chem. 2005, 280, 80–87. [Google Scholar] [PubMed]
- Favreau, P.; Krimm, I.; Le Gall, F.; Bobenrieth, M.J.; Lamthanh, H.; Bouet, F.; Servent, D.; Molgo, J.; Menez, A.; Letourneux, Y.; Lancelin, J.M. Biochemical characterization and nuclear magnetic resonance structure of novel alpha-conotoxins isolated from the venom of Conus consors. Biochemistry 1999, 38, 6317–6326. [Google Scholar]
- Liu, L.; Chew, G.; Hawrot, E.; Chi, C.; Wang, C. Two potent alpha3/5 conotoxins from piscivorous Conus achatinus. Acta Biochim. Biophys. Sin (Shanghai) 2007, 39, 438–444. [Google Scholar] [CrossRef] [PubMed]
- Martinez, J.S.; Olivera, B.M.; Gray, W.R.; Craig, A.G.; Groebe, D.R.; Abramson, S.N.; McIntosh, J.M. alpha-Conotoxin EI, a new nicotinic acetylcholine receptor antagonist with novel selectivity. Biochemistry 1995, 34, 14519–14526. [Google Scholar]
- Hogg, R.C.; Miranda, L.P.; Craik, D.J.; Lewis, R.J.; Alewood, P.F.; Adams, D.J. Single amino acid substitutions in alpha-conotoxin PnIA shift selectivity for subtypes of the mammalian neuronal nicotinic acetylcholine receptor. J. Biol. Chem. 1999, 274, 36559–36564. [Google Scholar]
- Luo, S.; Nguyen, T.A.; Cartier, G.E.; Olivera, B.M.; Yoshikami, D.; McIntosh, J.M. Single-residue alteration in alpha-conotoxin PnIA switches its nAChR subtype selectivity. Biochemistry 1999, 38, 14542–14548. [Google Scholar]
- McIntosh, J.M.; Dowell, C.; Watkins, M.; Garrett, J.E.; Yoshikami, D.; Olivera, B.M. Alpha-conotoxin GIC from Conus geographus, a novel peptide antagonist of nicotinic acetylcholine receptors. J. Biol. Chem. 2002, 277, 33610–33615. [Google Scholar]
- Cartier, G.E.; Yoshikami, D.; Gray, W.R.; Luo, S.; Olivera, B.M.; McIntosh, J.M. A new alpha-conotoxin which targets alpha3beta2 nicotinic acetylcholine receptors. J. Biol. Chem. 1996, 271, 7522–7528. [Google Scholar]
- McIntosh, J.M.; Azam, L.; Staheli, S.; Dowell, C.; Lindstrom, J.M.; Kuryatov, A.; Garrett, J.E.; Marks, M.J.; Whiteaker, P. Analogs of alpha-conotoxin MII are selective for alpha6-containing nicotinic acetylcholine receptors. Mol. Pharmacol. 2004, 65, 944–952. [Google Scholar]
- Dowell, C.; Olivera, B.M.; Garrett, J.E.; Staheli, S.T.; Watkins, M.; Kuryatov, A.; Yoshikami, D.; Lindstrom, J.M.; McIntosh, J.M. Alpha-conotoxin PIA is selective for alpha6 subunit-containing nicotinic acetylcholine receptors. J. Neurosci. 2003, 23, 8445–8452. [Google Scholar]
- Lopez-Vera, E.; Jacobsen, R.B.; Ellison, M.; Olivera, B.M.; Teichert, R.W. A novel alpha conotoxin (alpha-PIB) isolated from C. purpurascens is selective for skeletal muscle nicotinic acetylcholine receptors. Toxicon 2007, 49, 1193–1199. [Google Scholar] [CrossRef] [PubMed]
- Dutertre, S.; Nicke, A.; Lewis, R.J. Beta2 subunit contribution to. 4/7 alpha-conotoxin binding to the nicotinic acetylcholine receptor. J. Biol. Chem. 2005, 280, 30460–30468. [Google Scholar] [CrossRef] [PubMed]
- Nicke, A.; Loughnan, M.L.; Millard, E.L.; Alewood, P.F.; Adams, D.J.; Daly, N.L.; Craik, D.J.; Lewis, R.J. Isolation, structure, and activity of GID, a novel alpha. 4/7-conotoxin with an extended N-terminal sequence. J. Biol. Chem. 2003, 278, 3137–3144. [Google Scholar] [PubMed]
- Nicke, A.; Wonnacott, S.; Lewis, R.J. Alpha-conotoxins as tools for the elucidation of structure and function of neuronal nicotinic acetylcholine receptor subtypes. Eur. J. Biochem. 2004, 271, 2305–2319. [Google Scholar]
- Loughnan, M.; Bond, T.; Atkins, A.; Cuevas, J.; Adams, D.J.; Broxton, N.M.; Livett, B.G.; Down, J.G.; Jones, A.; Alewood, P.F.; Lewis, R.J. alpha-conotoxin EpI, a novel sulfated peptide from Conus episcopatus that selectively targets neuronal nicotinic acetylcholine receptors. J. Biol. Chem. 1998, 273, 15667–15674. [Google Scholar]
- Nicke, A.; Samochocki, M.; Loughnan, M.L.; Bansal, P.S.; Maelicke, A.; Lewis, R.J. Alpha-conotoxins EpI and AuIB switch subtype selectivity and activity in native versus recombinant nicotinic acetylcholine receptors. FEBS Lett. 2003, 554, 219–223. [Google Scholar]
- Loughnan, M.L.; Nicke, A.; Jones, A.; Adams, D.J.; Alewood, P.F.; Lewis, R.J. Chemical and functional identification and characterization of novel sulfated alpha-conotoxins from the cone snail Conus anemone. J. Med. Chem. 2004, 47, 1234–1241. [Google Scholar]
- Vincler, M.; Wittenauer, S.; Parker, R.; Ellison, M.; Olivera, B.M.; McIntosh, J.M. Molecular mechanism for analgesia involving specific antagonism of alpha9alpha10 nicotinic acetylcholine receptors. Proc. Natl. Acad. Sci. USA 2006, 103, 17880–17884. [Google Scholar]
- Clark, R.J.; Fischer, H.; Nevin, S.T.; Adams, D.J.; Craik, D.J. The synthesis, structural characterization, and receptor specificity of the alpha-conotoxin Vc1.1. J. Biol. Chem. 2006, 281, 23254–23263. [Google Scholar] [PubMed]
- Whiteaker, P.; Christensen, S.; Yoshikami, D.; Dowell, C.; Watkins, M.; Gulyas, J.; Rivier, J.; Olivera, B.M.; McIntosh, J.M. Discovery, synthesis, and structure activity of a highly selective alpha7 nicotinic acetylcholine receptor antagonist. Biochemistry 2007, 46, 6628–6638. [Google Scholar] [PubMed]
- McIntosh, J.M.; Plazas, P.V.; Watkins, M.; Gomez-Casati, M.E.; Olivera, B.M.; Elgoyhen, A.B. A novel alpha-conotoxin, PeIA, cloned from Conus pergrandis, discriminates between rat alpha9 alpha10 and alpha7 nicotinic cholinergic receptors. J. Biol. Chem. 2005, 280, 30107–30112. [Google Scholar] [PubMed]
- Talley, T.T.; Olivera, B.M.; Han, K.H.; Christensen, S.B.; Dowell, C.; Tsigelny, I.; Ho, K.Y.; Taylor, P.; McIntosh, J.M. Alpha-conotoxin OmIA is a potent ligand for the acetylcholine-binding protein as well as alpha3beta2 and alpha7 nicotinic acetylcholine receptors. J. Biol. Chem. 2006, 281, 24678–24686. [Google Scholar]
- Dutertre, S.; Ulens, C.; Buttner, R.; Fish, A.; van Elk, R.; Kendel, Y.; Hopping, G.; Alewood, P.F.; Schroeder, C.; Nicke, A.; Smit, A.B.; Sixma, T.K.; Lewis, R.J. AChBP-targeted alpha-conotoxin correlates distinct binding orientations with nAChR subtype selectivity. EMBO J. 2007, 26, 3858–3867. [Google Scholar]
- Peng, C.; Han, Y.; Sanders, T.; Chew, G.; Liu, J.; Hawrot, E.; Chi, C.; Wang, C. alpha4/7-conotoxin Lp1.1 is a novel antagonist of neuronal nicotinic acetylcholine receptors. Peptides 2008, 29, 1700–1707. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Vera, E.; Aguilar, M.B.; Schiavon, E.; Marinzi, C.; Ortiz, E.; Restano Cassulini, R.; Batista, C.V.; Possani, L.D.; Heimer de la Cotera, E.P.; Peri, F.; Becerril, B.; Wanke, E. Novel alpha-conotoxins from Conus spurius and the alpha-conotoxin EI share high-affinity potentiation and low-affinity inhibition of nicotinic acetylcholine receptors. FEBS J. 2007, 274, 3972–3985. [Google Scholar]
- Dwoskin, L.P.; Crooks, P.A. Competitive neuronal nicotinic receptor antagonists: A new direction for drug discovery. J. Pharmacol. Exp. Ther. 2001, 298, 395–402. [Google Scholar]
- Lukas, R.J. Pharmacological distinctions between functional nicotinic acetylcholine receptors on the PC12 rat pheochromocytoma and the TE671 human medulloblastoma. J. Pharmacol. Exp. Ther. 1989, 251, 175–182. [Google Scholar]
- Decker, M.W.; Anderson, D.J.; Brioni, J.D.; Donnelly-Roberts, D.L.; Kang, C.H.; O'Neill, A.B.; Piattoni-Kaplan, M.; Swanson, S.; Sullivan, J.P. Erysodine, a competitive antagonist at neuronal nicotinic acetylcholine receptors. Eur. J. Pharmacol. 1995, 280, 79–89. [Google Scholar]
- Fitch, R.W.; Xiao, Y.; Kellar, K.J.; Daly, J.W. Membrane potential fluorescence: A rapid and highly sensitive assay for nicotinic receptor channel function. Proc. Natl. Acad. Sci. USA 2003, 100, 4909–4914. [Google Scholar]
- Aiyar, V.N.; Benn, M.H.; Hanna, T.; Jacyno, J.; Roth, S.H.; Wilkens, J.L. The principal toxin of Delphinium brownii Rydb., and its mode of action. Experientia 1979, 35, 1367–1368. [Google Scholar] [CrossRef] [PubMed]
- Macallan, D.R.; Lunt, G.G.; Wonnacott, S.; Swanson, K.L.; Rapoport, H.; Albuquerque, E.X. Methyllycaconitine and (+)-anatoxin-a differentiate between nicotinic receptors in vertebrate and invertebrate nervous systems. FEBS Lett. 1988, 226, 357–363. [Google Scholar]
- Ward, J.M.; Cockcroft, V.B.; Lunt, G.G.; Smillie, F.S.; Wonnacott, S. Methyllycaconitine: A selective probe for neuronal alpha-bungarotoxin binding sites. FEBS Lett. 1990, 270, 45–48. [Google Scholar]
- Kukel, C.F.; Jennings, K.R. Delphinium alkaloids as inhibitors of alpha-bungarotoxin binding to rat and insect neural membranes. Can. J. Physiol. Pharmacol. 1994, 72, 104–107. [Google Scholar]
- Dobelis, P.; Madl, J.E.; Pfister, J.A.; Manners, G.D.; Walrond, J.P. Effects of Delphinium alkaloids on neuromuscular transmission. J. Pharmacol. Exp. Ther. 1999, 291, 538–546. [Google Scholar]
- Pfister, J.A.; Gardner, D.R.; Panter, K.E.; Manners, G.D.; Ralphs, M.H.; Stegelmeier, B.L.; Schoch, T.K. Larkspur (Delphinium spp.) poisoning in livestock. J. Nat. Toxins 1999, 8, 81–94. [Google Scholar] [PubMed]
- Stromgaard, K. Natural products as tools for studies of ligand-gated ion channels. Chem. Rec. 2005, 5, 229–239. [Google Scholar]
- Hold, K.M.; Sirisoma, N.S.; Ikeda, T.; Narahashi, T.; Casida, J.E. Alpha-thujone (the active component of absinthe): Gamma-aminobutyric acid type A receptor modulation and metabolic detoxification. Proc. Natl. Acad. Sci. USA 2000, 97, 3826–3831. [Google Scholar]
- Deiml, T.; Haseneder, R.; Zieglgansberger, W.; Rammes, G.; Eisensamer, B.; Rupprecht, R.; Hapfelmeier, G. Alpha-thujone reduces. 5-HT3 receptor activity by an effect on the agonist-reduced desensitization. Neuropharmacology 2004, 46, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.K.; Su, C.K. Endogenous activation of nicotinic receptors underlies sympathetic tone generation in neonatal rat spinal cord in vitro. Neuropharmacology 2006, 51, 1120–1128. [Google Scholar] [CrossRef] [PubMed]
- Erkkila, B.E.; Sedelnikova, A.V.; Weiss, D.S. Stoichiometric pore mutations of the GABAAR reveal a pattern of hydrogen bonding with picrotoxin. Biophys. J. 2008, 94, 4299–4306. [Google Scholar]
- Charon, S.; Taly, A.; Rodrigo, J.; Perret, P.; Goeldner, M. Binding Modes of Noncompetitive GABA-Channel Blockers Revisited Using Engineered Affinity-Labeling Reactions Combined with New Docking Studies (dagger). J. Agric. Food Chem. 2010. [Google Scholar]
- Johnson, J.W.; Ascher, P. Glycine potentiates the NMDA response in cultured mouse brain neurons. Nature 1987, 325, 529–531. [Google Scholar]
- England, L.J.; Imperial, J.; Jacobsen, R.; Craig, A.G.; Gulyas, J.; Akhtar, M.; Rivier, J.; Julius, D.; Olivera, B.M. Inactivation of a serotonin-gated ion channel by a polypeptide toxin from marine snails. Science. 1998, 281, 575–578. [Google Scholar]
- Yan, D.; White, M.M. Interaction of d-tubocurarine analogs with mutant 5-HT(3) receptors. Neuropharmacology 2002, 43, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Griffith, H.R.; Johnson, G. The use of curare in general anasthesia. Anesthesiology 1942, 3, 418–420. [Google Scholar]
- Parks, T.N.; Mueller, A.L.; Artman, L.D.; Albensi, B.C.; Nemeth, E.F.; Jackson, H.; Jasys, V.J.; Saccomano, N.A.; Volkmann, R.A. Arylamine toxins from funnel-web spider (Agelenopsis aperta) venom antagonize N-methyl-D-aspartate receptor function in mammalian brain. J. Biol. Chem. 1991, 266, 21523–21529. [Google Scholar] [PubMed]
- Kiskin, N.I.; Chizhmakov, I.V.; Tsyndrenko, A.; Mueller, A.L.; Jackson, H.; Krishtal, O.A. A highly potent and selective N-methyl-D-aspartate receptor antagonist from the venom of the Agelenopsis aperta spider. Neuroscience 1992, 51, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Frydenvang, K.; Lash, L.L.; Naur, P.; Postila, P.A.; Pickering, D.S.; Smith, C.M.; Gajhede, M.; Sasaki, M.; Sakai, R.; Pentikainen, O.T.; Swanson, G.T.; Kastrup, J.S. Full domain closure of the ligand-binding core of the ionotropic glutamate receptor iGluR5 induced by the high affinity agonist dysiherbaine and the functional antagonist. 8,9-dideoxyneodysiherbaine. J. Biol. Chem. 2009, 284, 14219–14229. [Google Scholar] [PubMed]
- Rajendra, W.; Armugam, A.; Jeyaseelan, K. Toxins in anti-nociception and anti-inflammation. Toxicon 2004, 44, 1–17. [Google Scholar]
- Sheng, Z.; Prorok, M.; Castellino, F.J. Specific determinants of conantokins that dictate their selectivity for the NR2B subunit of N-methyl-D-aspartate receptors. Neuroscience 2010, 170, 703–710. [Google Scholar]
- Skjaerbaek, N.; Nielsen, K.J.; Lewis, R.J.; Alewood, P.; Craik, D.J. Determination of the solution structures of conantokin-G and conantokin-T by CD and NMR spectroscopy. J. Biol. Chem. 1997, 272, 2291–2299. [Google Scholar]
- Dai, Q.; Xiao, C.; Dong, M.; Liu, Z.; Sheng, Z.; Castellino, F.J.; Prorok, M. Non-strict strand orientation of the Ca2+-induced dimerization of a conantokin peptide variant with sequence-shifted gamma-carboxyglutamate residues. Peptides 2009, 30, 866–872. [Google Scholar]
- Lodge, D. The history of the pharmacology and cloning of ionotropic glutamate receptors and the development of idiosyncratic nomenclature. Neuropharmacology 2009, 56, 6–21. [Google Scholar]
- Sakai, R.; Minato, S.; Koike, K.; Jimbo, M.; Kamiya, H. Cellular and subcellular localization of kainic acid in the marine red alga Digenea simplex. Cell Tissue Res. 2005, 322, 491–502. [Google Scholar]
- Swanson, G.T.; Green, T.; Sakai, R.; Contractor, A.; Che, W.; Kamiya, H.; Heinemann, S.F. Differential activation of individual subunits in heteromeric kainate receptors. Neuron 2002, 34, 589–598. [Google Scholar]
- Savchenko, H.A.; Vasylevs'kyi, A.A.; Pluzhnykov, K.A.; Korol'kova Iu, V.; Mamenko, M.V.; Volkova, T.M.; Maksymiuk, O.P.; Boichuk Ia, A.; Hrishyn Ie, V.; Kryshtal, O.O. Peptide components of Geolycosa spider venom modulate P2X receptor activity of rat sensory neurons. Fiziol. Zh. 2009, 55, 11–16. [Google Scholar]
- Grishin, E.V.; Savchenko, G.A.; Vassilevski, A.A.; Korolkova, Y.V.; Boychuk, Y.A.; Viatchenko-Karpinski, V.Y.; Nadezhdin, K.D.; Arseniev, A.S.; Pluzhnikov, K.A.; Kulyk, V.B.; Voitenko, N.V.; Krishtal, O.O. Novel peptide from spider venom inhibits P2X3 receptors and inflammatory pain. Ann. Neurol. 2010, 67, 680–683. [Google Scholar]
- Patyar, S.; Joshi, R.; Byrav, D.S.; Prakash, A.; Medhi, B.; Das, B.K. Bacteria in cancer therapy: A novel experimental strategy. J. Biomed. Sci. 2010, 17, 21. [Google Scholar]
- Shapira, A.; Benhar, I. Toxin-Based Therapeutic Approaches. Toxins 2010, 2, 2519–2583. [Google Scholar]
- Tutka, P.; Zatonski, W. Cytisine for the treatment of nicotine addiction: From a molecule to therapeutic efficacy. Pharmacol. Rep. 2006, 58, 777–798. [Google Scholar]
- Cohen, C.; Bergis, O.E.; Galli, F.; Lochead, A.W.; Jegham, S.; Biton, B.; Leonardon, J.; Avenet, P.; Sgard, F.; Besnard, F.; Graham, D.; Coste, A.; Oblin, A.; Curet, O.; Voltz, C.; Gardes, A.; Caille, D.; Perrault, G.; George, P.; Soubrie, P.; Scatton, B. SSR591813, a novel selective and partial alpha4beta2 nicotinic receptor agonist with potential as an aid to smoking cessation. J. Pharmacol. Exp. Ther. 2003, 306, 407–420. [Google Scholar]
- Martin, L.F.; Kem, W.R.; Freedman, R. Alpha-7 nicotinic receptor agonists: Potential new candidates for the treatment of schizophrenia. Psychopharmacology (Berl) 2004, 174, 54–64. [Google Scholar] [PubMed]
- Wei, S.; Qiu, L.; Chen, Z.H.; Xu, M.C. Effect observation of Snake venom point injection in the treatment of scapulohumeral periarthritis. Chin. J. Infor. Traditional Chin. Med. 2007, 14, 68–96. [Google Scholar]
- Grozio, A.; Paleari, L.; Catassi, A.; Servent, D.; Cilli, M.; Piccardi, F.; Paganuzzi, M.; Cesario, A.; Granone, P.; Mourier, G.; Russo, P. Natural agents targeting the alpha7-nicotinic-receptor in NSCLC: A promising prospective in anti-cancer drug development. Int. J. Cancer. 2008, 122, 1911–1915. [Google Scholar]
- Spindel, E.R. Is nicotine the estrogen of lung cancer? Am. J. Respir. Crit. Care. Med. 2009, 179, 1081–1082. [Google Scholar] [CrossRef] [PubMed]
- Paleari, L.; Negri, E.; Catassi, A.; Cilli, M.; Servent, D.; D'Angelillo, R.; Cesario, A.; Russo, P.; Fini, M. Inhibition of nonneuronal alpha7-nicotinic receptor for lung cancer treatment. Am. J. Respir. Crit. Care. Med. 2009, 179, 1141–1150. [Google Scholar]
- Catassi, A.; Paleari, L.; Servent, D.; Sessa, F.; Dominioni, L.; Ognio, E.; Cilli, M.; Vacca, P.; Mingari, M.; Gaudino, G.; Bertino, P.; Paolucci, M.; Calcaterra, A.; Cesario, A.; Granone, P.; Costa, R.; Ciarlo, M.; Alama, A.; Russo, P. Targeting alpha7-nicotinic receptor for the treatment of pleural mesothelioma. Eur. J. Cancer. 2008, 44, 2296–2311. [Google Scholar]
- Russo, P.; Catassi, A.; Cesario, A.; Servent, D. Development of novel therapeutic strategies for lung cancer: Targeting the cholinergic system. Curr. Med. Chem. 2006, 13, 3493–3512. [Google Scholar]
- Chen, Z.X.; Zhang, H.L.; Gu, Z.L.; Chen, B.W.; Han, R.; Reid, P.F.; Raymond, L.N.; Qin, Z.H. A long-form alpha-neurotoxin from cobra venom produces potent opioid-independent analgesia. Acta Pharmacol. Sin. 2006, 27, 402–408. [Google Scholar]
- Reid, P.F. Alpha-cobratoxin as a possible therapy for multiple sclerosis: A review of the literature leading to its development for this application. Crit. Rev. Immunol. 2007, 27, 291–302. [Google Scholar]
- Zhang, H.L.; Han, R.; Chen, Z.X.; Gu, Z.L.; Reid, P.F.; Raymond, L.N.; Qin, Z.H. Analgesic effects of receptin, a chemically modified cobratoxin from Thailand cobra venom. Neurosci. Bull. 2006, 22, 267–273. [Google Scholar]
- Nevin, S.T.; Clark, R.J.; Klimis, H.; Christie, M.J.; Craik, D.J.; Adams, D.J. Are alpha9alpha10 nicotinic acetylcholine receptors a pain target for alpha-conotoxins? Mol. Pharmacol. 2007, 72, 1406–1410. [Google Scholar] [CrossRef] [PubMed]
- Callaghan, B.; Haythornthwaite, A.; Berecki, G.; Clark, R.J.; Craik, D.J.; Adams, D.J. Analgesic alpha-conotoxins Vc1.1 and Rg1A inhibit N-type calcium channels in rat sensory neurons via GABAB receptor activation. J. Neurosci. 2008, 28, 10943–10951. [Google Scholar] [PubMed]
- Azam, L.; McIntosh, J.M. Alpha-conotoxins as pharmacological probes of nicotinic acetylcholine receptors. Acta. Pharmacol. Sin. 2009, 30, 771–783. [Google Scholar]
- Tuba, Z.; Maho, S.; Vizi, E.S. Synthesis and structure-activity relationships of neuromuscular blocking agents. Curr. Med. Chem. 2002, 9, 1507–1536. [Google Scholar]
- Hughes, T. The early history of myasthenia gravis. Neuromuscul. Disord. 2005, 15, 878–886. [Google Scholar]
- Vincent, A. Unravelling the pathogenesis of myasthenia gravis. Nat. Rev. Immunol. 2002, 2, 797–804. [Google Scholar]
- Engel, A.G.; Sine, S.M. Current understanding of congenital myasthenic syndromes. Curr. Opin. Pharmacol. 2005, 5, 308–321. [Google Scholar]
- Garcia-Colunga, J.; Miledi, R. Modulation of nicotinic acetylcholine receptors by strychnine. Proc. Natl. Acad. Sci. USA 1999, 96, 4113–4118. [Google Scholar]
- Verkman, A.S.; Galietta, L.J. Chloride channels as drug targets. Nat. Rev. Drug. Discov. 2009, 8, 153–171. [Google Scholar]
- Laube, B.; Maksay, G.; Schemm, R.; Betz, H. Modulation of glycine receptor function: A novel approach for therapeutic intervention at inhibitory synapses? Trends Pharmacol. Sci. 2002, 23, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Walstab, J.; Rappold, G.; Niesler, B. 5-HT(3) receptors: Role in disease and target of drugs. Pharmacol. Ther. 2010, 128, 146–169. [Google Scholar]
- Koller, M.; Urwyler, S. Novel N-methyl-D-aspartate receptor antagonists: A review of compounds patented since. 2006. Expert. Opin. Ther. Pat. 2010, 20, 1683–1702. [Google Scholar] [CrossRef] [PubMed]
- Malmberg, A.B.; Gilbert, H.; McCabe, R.T.; Basbaum, A.I. Powerful antinociceptive effects of the cone snail venom-derived subtype-selective NMDA receptor antagonists conantokins G and T. Pain 2003, 101, 109–116. [Google Scholar]
- Hama, A.; Sagen, J. Antinociceptive effects of the marine snail peptides conantokin-G and conotoxin MVIIA alone and in combination in rat models of pain. Neuropharmacology 2009, 56, 556–563. [Google Scholar]
- Jimenez, E.C.; Donevan, S.; Walker, C.; Zhou, L.M.; Nielsen, J.; Cruz, L.J.; Armstrong, H.; White, H.S.; Olivera, B.M. Conantokin-L, a new NMDA receptor antagonist: Determinants for anticonvulsant potency. Epilepsy Res. 2002, 51, 73–80. [Google Scholar]
- Prorok, M.; Castellino, F.J. The molecular basis of conantokin antagonism of NMDA receptor function. Curr. Drug. Targets. 2007, 8, 633–642. [Google Scholar]
- Swanson, G.T.; Sakai, R. Ligands for ionotropic glutamate receptors. Prog. Mol. SubCell Biol. 2009, 46, 123–157. [Google Scholar]
- Li, Z.; Liang, D.; Chen, L. Potential therapeutic targets for ATP-gated P2X receptor ion channels. Assay Drug. Dev. Technol. 2008, 6, 277–284. [Google Scholar]
- Gunosewoyo, H.; Kassiou, M. P2X purinergic receptor ligands: Recently patented compounds. Expert Opin. Ther. Pat. 2010, 20, 625–646. [Google Scholar]
- Ryan, T.J.; Grant, S.G. The origin and evolution of synapses. Nat. Rev. Neurosci. 2009, 10, 701–712. [Google Scholar]
- Tasneem, A.; Iyer, L.M.; Jakobsson, E.; Aravind, L. Identification of the prokaryotic ligand-gated ion channels and their implications for the mechanisms and origins of animal Cys-loop ion channels. Genome. Biol. 2005, 6, R4. [Google Scholar]
- Bocquet, N.; Prado de Carvalho, L.; Cartaud, J.; Neyton, J.; Le Poupon, C.; Taly, A.; Grutter, T.; Changeux, J.P.; Corringer, P.J. A prokaryotic proton-gated ion channel from the nicotinic acetylcholine receptor family. Nature 2007, 445, 116–119. [Google Scholar]
- Chen, G.Q.; Cui, C.; Mayer, M.L.; Gouaux, E. Functional characterization of a potassium-selective prokaryotic glutamate receptor. Nature 1999, 402, 817–821. [Google Scholar]
- Osborne, R.H. Insect neurotransmission: Neurotransmitters and their receptors. Pharmacol. Ther. 1996, 69, 117–142. [Google Scholar]
- Ryten, M.; Koshi, R.; Knight, G.E.; Turmaine, M.; Dunn, P.; Cockayne, D.A.; Ford, A.P.; Burnstock, G. Abnormalities in neuromuscular junction structure and skeletal muscle function in mice lacking the P2X2 nucleotide receptor. Neuroscience 2007, 148, 700–711. [Google Scholar]
- Adams, D.J.; Alewoodb, P.F.; Craikb, D.J.; Drinkwaterc, R.D.; Lewis, R.J. Conotoxins and Their Potential Pharmaceutical Applications. Drug Dev. Res. 1999, 46, 219–234. [Google Scholar] [CrossRef]
- Moise, L.; Piserchio, A.; Basus, V.J.; Hawrot, E. NMR structural analysis of alpha-bungarotoxin and its complex with the principal alpha-neurotoxin-binding sequence on the alpha.7 subunit of a neuronal nicotinic acetylcholine receptor. J. Biol. Chem. 2002, 277, 12406–12417. [Google Scholar] [PubMed]
- Samson, A.; Scherf, T.; Eisenstein, M.; Chill, J.; Anglister, J. The mechanism for acetylcholine receptor inhibition by alpha-neurotoxins and species-specific resistance to alpha-bungarotoxin revealed by NMR. Neuron 2002, 35, 319–332. [Google Scholar]
- Osaka, H.; Malany, S.; Molles, B.E.; Sine, S.M.; Taylor, P. Pairwise electrostatic interactions between alpha-neurotoxins and gamma, delta, and epsilon subunits of the nicotinic acetylcholine receptor. J. Biol. Chem. 2000, 275, 5478–5484. [Google Scholar] [PubMed]
- Servent, D.; Menez, A. Snake neurotoxins that interact with nicotinic acetylcholine receptors. In Handbook of Neurotoxicology; Humana Press Inc.: Totowa, NJ, USA, 2001; pp. 385–425. [Google Scholar]
- Zeng, H.; Hawrot, E. NMR-based binding screen and structural analysis of the complex formed between alpha-cobratoxin and an 18-mer cognate peptide derived from the alpha 1 subunit of the nicotinic acetylcholine receptor from Torpedo californica. J. Biol. Chem. 2002, 277, 37439–37445. [Google Scholar] [CrossRef] [PubMed]
- Low, B.W.; Preston, H.S.; Sato, A.; Rosen, L.S.; Searl, J.E.; Rudko, A.D.; Richardson, J.S. Three dimensional structure of erabutoxin b neurotoxic protein: Inhibitor of acetylcholine receptor. Proc. Natl. Acad. Sci. USA 1976, 73, 2991–2994. [Google Scholar]
- Miwa, J.M.; Ibanez-Tallon, I.; Crabtree, G.W.; Sanchez, R.; Sali, A.; Role, L.W.; Heintz, N. lynx1, an endogenous toxin-like modulator of nicotinic acetylcholine receptors in the mammalian CNS. Neuron 1999, 23, 105–114. [Google Scholar]
- Lyukmanova, E.N.; Shenkarev, Z.O.; Shulepko, M.A.; Mineev, K.S.; D'Hoedt, D.; Kasheverov, I.E.; Filkin, S.Y.; Krivolapova, A.P.; Janickova, H.; Dolezal, V.; Dolgikh, D.A.; Arseniev, A.S.; Bertrand, D.; Tsetlin, V.I.; Kirpichnikov, M.P. Nmr Structure and Action on Nicotinic Acetylcholine Receptors of Water-Soluble Domain of Human Lynx1. J. Biol. Chem. 2011. [Google Scholar]
- Pawlak, J.; Mackessy, S.P.; Fry, B.G.; Bhatia, M.; Mourier, G.; Fruchart-Gaillard, C.; Servent, D.; Menez, R.; Stura, E.; Menez, A.; Kini, R.M. Denmotoxin, a three-finger toxin from the colubrid snake Boiga dendrophila (Mangrove Catsnake) with bird-specific activity. J. Biol. Chem. 2006, 281, 29030–29041. [Google Scholar]
- Tsai, M.C.; Hsieh, W.H.; Smith, L.A.; Lee, C.Y. Effects of waglerin-I on neuromuscular transmission of mouse nerve-muscle preparations. Toxicon 1995, 33, 363–371. [Google Scholar]
- Utkin, Y.N.; Kukhtina, V.V.; Kryukova, E.V.; Chiodini, F.; Bertrand, D.; Methfessel, C.; Tsetlin, V.I. "Weak toxin" from Naja kaouthia is a nontoxic antagonist of alpha. 7 and muscle-type nicotinic acetylcholine receptors. J. Biol. Chem. 2001, 276, 15810–15815. [Google Scholar] [PubMed]
- Nirthanan, S.; Charpantier, E.; Gopalakrishnakone, P.; Gwee, M.C.; Khoo, H.E.; Cheah, L.S.; Bertrand, D.; Kini, R.M. Candoxin, a novel toxin from Bungarus candidus, is a reversible antagonist of muscle (alphabetagammadelta ) but a poorly reversible antagonist of neuronal alpha. 7 nicotinic acetylcholine receptors. J. Biol. Chem. 2002, 277, 17811–17820. [Google Scholar] [PubMed]
- Joubert, F.J.; Taljaard, N. Naja haje haje (Egyptian cobra) venom. Some properties and the complete primary structure of three toxins (CM-2, CM-11 and CM-12). Eur. J. Biochem. 1978, 90, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.L.; Hider, R.C.; Hodges, S.J.; Joubert, F.J. Structure-activity studies of homologues of short chain neurotoxins from Elapid snake venoms. Br. J. Pharmacol. 1984, 82, 709–716. [Google Scholar]
- Carlsson, F.H. Snake venom toxins. The primary structure of protein S4C11. A neurotoxin homologue from the venom of forest cobra (Naja melanoleuca). Biochim. Biophys. Acta. 1975, 400, 310–321. [Google Scholar] [PubMed]
- Joubert, F.J.; Taljaard, N. Some properties and the complete primary structures of two reduced and S-carboxymethylated polypeptides (S5C1 and S5C10) from Dendroaspis jamesoni kaimosae (Jameson's mamba) venom. Biochim. Biophys. Acta 1979, 579, 228–233. [Google Scholar]
- Aird, S.D.; Womble, G.C.; Yates, J.R., 3rd.; Griffin, P.R. Primary structure of gamma-bungarotoxin, a new postsynaptic neurotoxin from venom of Bungarus multicinctus. Toxicon 1999, 37, 609–625. [Google Scholar] [CrossRef] [PubMed]
- Poh, S.L.; Mourier, G.; Thai, R.; Armugam, A.; Molgo, J.; Servent, D.; Jeyaseelan, K.; Menez, A. A synthetic weak neurotoxin binds with low affinity to Torpedo and chicken alpha7 nicotinic acetylcholine receptors. Eur. J. Biochem. 2002, 269, 4247–4256. [Google Scholar]
- Chang, L.; Lin, S.; Wang, J.; Hu, W.P.; Wu, B.; Huang, H. Structure-function studies on Taiwan cobra long neurotoxin homolog. Biochim. Biophys. Acta. 2000, 1480, 293–301. [Google Scholar]
- Harvey, A.L.; Rodger, I.W. Reversibility of neuromuscular blockade produced by toxins isolated from the venom of the seasnake Laticauda semifasciata. Toxicon 1978, 16, 219–225. [Google Scholar]
- Brier, T.J.; Mellor, I.R.; Tikhonov, D.B.; Neagoe, I.; Shao, Z.; Brierley, M.J.; Stromgaard, K.; Jaroszewski, J.W.; Krogsgaard-Larsen, P.; Usherwood, P.N. Contrasting actions of philanthotoxin-343 and philanthotoxin-(12) on human muscle nicotinic acetylcholine receptors. Mol. Pharmacol. 2003, 64, 954–964. [Google Scholar]
- Shu, Q.; Liang, S.P. Purification and characterization of huwentoxin-II, a neurotoxic peptide from the venom of the Chinese bird spider Selenocosmia huwena. J. Pept. Res. 1999, 53, 486–491. [Google Scholar]
- Liang, S.P.; Zhang, D.Y.; Pan, X.; Chen, Q.; Zhou, P.A. Properties and amino acid sequence of huwentoxin-I, a neurotoxin purified from the venom of the Chinese bird spider Selenocosmia huwena. Toxicon 1993, 31, 969–978. [Google Scholar]
- Daly, J.W. Nicotinic agonists, antagonists, and modulators from natural sources. Cell Mol. Neurobiol. 2005, 25, 513–552. [Google Scholar] [CrossRef] [PubMed]
- Barlow, R.B.; McLeod, L.J. Some studies on cytisine and its methylated derivatives. Br. J. Pharmacol. 1969, 35, 161–174. [Google Scholar]
- Osswald, J.; Rellan, S.; Gago, A.; Vasconcelos, V. Toxicology and detection methods of the alkaloid neurotoxin produced by cyanobacteria, anatoxin-a. Environ. Int. 2007, 33, 1070–1089. [Google Scholar]
- Merel, S.; Clement, M.; Thomas, O. State of the art on cyanotoxins in water and their behaviour towards chlorine. Toxicon 2010, 55, 677–691. [Google Scholar]
- Araoz, R.; Vilarino, N.; Botana, L.M.; Molgo, J. Ligand-binding assays for cyanobacterial neurotoxins targeting cholinergic receptors. Anal. Bioanal. Chem. 2010, 397, 1695–1704. [Google Scholar]
- Badio, B.; Daly, J.W. Epibatidine, a potent analgetic and nicotinic agonist. Mol. Pharmacol. 1994, 45, 563–569. [Google Scholar]
- Kem, W.R.; Mahnir, V.M.; Papke, R.L.; Lingle, C.J. Anabaseine is a potent agonist on muscle and neuronal alpha-bungarotoxin-sensitive nicotinic receptors. J. Pharmacol. Exp. Ther. 1997, 283, 979–992. [Google Scholar]
- Spivak, C.E.; Maleque, M.A.; Oliveira, A.C.; Masukawa, L.M.; Tokuyama, T.; Daly, J.W.; Albuquerque, E.X. Actions of the histrionicotoxins at the ion channel of the nicotinic acetylcholine receptor and at the voltage-sensitive ion channels of muscle membranes. Mol. Pharmacol. 1982, 21, 351–361. [Google Scholar]
- Bick, R.C.; Gillard, J.W.; Leow, H.-M. Alkaloids of Darlingia ferruginea. Australian. J. Chem. 1979, 32, 2537–2543. [Google Scholar] [CrossRef]
- Van Maanen, E.F. The antagonism between acetylcholine and the curare alkaloids, d-tubocurarine, c-curarine-I, c-toxiferine-II and beta-erythroidine in the rectus abdominis of the frog. J. Pharmacol. Exp. Ther. 1950, 99, 255–264. [Google Scholar] [PubMed]
- Jensen, A.A.; Zlotos, D.P.; Liljefors, T. Pharmacological characteristics and binding modes of caracurine V analogues and related compounds at the neuronal alpha7 nicotinic acetylcholine receptor. J. Med. Chem. 2007, 50, 4616–4629. [Google Scholar]
- Tay, G. Diallyl nor-Toxiferine -a New Relaxant. Singapore Med. J. 1963, 141, 90–92. [Google Scholar]
- Kosuge, T.; Tsuji, K.; Hirai, K. Isolation of neosurugatoxin from the Japanese ivory shell, Babylonia japonica. Chem. Pharm. Bull (Tokyo) 1982, 30, 3255–3259. [Google Scholar] [PubMed]
- Hayashi, E.; Isogai, M.; Kagawa, Y.; Takayanagi, N.; Yamada, S. Neosurugatoxin, a specific antagonist of nicotinic acetylcholine receptors. J. Neurochem. 1984, 42, 1491–1494. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, E.; Yamada, S. Pharmacological studies on surugatoxin, the toxic principle from Japanese ivory mollusc (Babylonia japonica). Br. J. Pharmacol. 1975, 53, 207–215. [Google Scholar]
- Tornoe, C.; Bai, D.; Holden-Dye, L.; Abramson, S.N.; Sattelle, D.B. Actions of neurotoxins (bungarotoxins, neosurugatoxin and lophotoxins) on insect and nematode nicotinic acetylcholine receptors. Toxicon 1995, 33, 411–424. [Google Scholar]
- Maciulaitis, R.; Kontrimaviciute, V.; Bressolle, F.M.; Briedis, V. Ibogaine, an anti-addictive drug: Pharmacology and time to go further in development. A narrative review. Hum. Exp. Toxicol. 2008, 27, 181–194. [Google Scholar] [CrossRef] [PubMed]
- Mash, D.C.; Kovera, C.A.; Buck, B.E.; Norenberg, M.D.; Shapshak, P.; Hearn, W.L.; Sanchez-Ramos, J. Medication development of ibogaine as a pharmacotherapy for drug dependence. Ann. N.Y. Acad. Sci. 1998, 844, 274–292. [Google Scholar]
- Matsubayashi, H.; Alkondon, M.; Pereira, E.F.; Swanson, K.L.; Albuquerque, E.X. Strychnine: A potent competitive antagonist of alpha-bungarotoxin-sensitive nicotinic acetylcholine receptors in rat hippocampal neurons. J. Pharmacol. Exp. Ther. 1998, 284, 904–913. [Google Scholar]
- Francis, M.M.; Cheng, E.Y.; Weiland, G.A.; Oswald, R.E. Specific activation of the alpha. 7 nicotinic acetylcholine receptor by a quaternary analog of cocaine. Mol. Pharmacol. 2001, 60, 71–79. [Google Scholar] [PubMed]
- Haefely, W. The effects of various "nicotine-like" agents in the cat superior cervical ganglion in situ. Naunyn. Schmiedebergs. Arch. Pharmacol. 1974, 281, 93–117. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, Y.; Takano, Y.; Kohjimoto, Y.; Honda, K.; Kamiya, H.O. Enhancement of [3H]dopamine release and its [3H]metabolites in rat striatum by nicotinic drugs. Brain. Res. 1982, 242, 99–106. [Google Scholar]
- Rozman, K.B.; Araoz, R.; Sepcic, K.; Molgo, J.; Suput, D. Parazoanthoxanthin A blocks Torpedo nicotinic acetylcholine receptors. Chem. Biol. Interact. 2010, 187, 384–387. [Google Scholar]
- Culver, P.; Burch, M.; Potenza, C.; Wasserman, L.; Fenical, W.; Taylor, P. Structure-activity relationships for the irreversible blockade of nicotinic receptor agonist sites by lophotoxin and congeneric diterpene lactones. Mol. Pharmacol. 1985, 28, 436–444. [Google Scholar]
- Culver, P.; Fenical, W.; Taylor, P. Lophotoxin irreversibly inactivates the nicotinic acetylcholine receptor by preferential association at one of the two primary agonist sites. J. Biol. Chem. 1984, 259, 3763–3770. [Google Scholar]
- Culver, P.; Jacobs, R.S. Lophotoxin: A neuromuscular acting toxin from the sea whip (Lophogorgia rigida). Toxicon 1981, 19, 825–830. [Google Scholar] [CrossRef] [PubMed]
- Fenical, W.; Okuda, R.K.; Bandurraga, M.M.; Culver, P.; Jacobs, R.S. Lophotoxin: A novel neuromuscular toxin from Pacific sea whips of the genus Lophogorgia. Science 1981, 212, 1512–1514. [Google Scholar] [PubMed]
- Luetje, C.W.; Wada, K.; Rogers, S.; Abramson, S.N.; Tsuji, K.; Heinemann, S.; Patrick, J. Neurotoxins distinguish between different neuronal nicotinic acetylcholine receptor subunit combinations. J. Neurochem. 1990, 55, 632–640. [Google Scholar]
- Abramson, S.N.; Li, Y.; Culver, P.; Taylor, P. An analog of lophotoxin reacts covalently with Tyr190 in the alpha-subunit of the nicotinic acetylcholine receptor. J. Biol. Chem. 1989, 264, 12666–12672. [Google Scholar]
- Holmskov, U.; Laursen, S.B.; Malhotra, R.; Wiedemann, H.; Timpl, R.; Stuart, G.R.; Tornoe, I.; Madsen, P.S.; Reid, K.B.; Jensenius, J.C. Comparative study of the structural and functional properties of a bovine plasma C-type lectin, collectin-43, with other collectins. Biochem. J. 1995, 305, 889–896. [Google Scholar] [PubMed]
- Johnston, G.A.; Hanrahan, J.R.; Chebib, M.; Duke, R.K.; Mewett, K.N. Modulation of ionotropic GABA receptors by natural products of plant origin. Adv. Pharmacol. 2006, 54, 285–316. [Google Scholar]
- Takemoto, T.; Nakajima, T.; Sakuma, R. Isolation of a Flycidal Constituent "Ibotenic Acid" from Amanita Muscaria and A. Pantherina. Yakugaku. Zasshi. 1964, 84, 1233–1234. [Google Scholar] [PubMed]
- Takemoto, T.; Nakajima, T.; Yokobe, T. Structure of Ibotenic Acid. Yakugaku. Zasshi. 1964, 84, 1232–1233. [Google Scholar]
- Casarett, L.J.; Doull, J.; Klaassen, C.D. Casarett and. Doull's Toxicology: The Basic Science of Poisons, 7th ed; McGraw-Hill Medical: London, UK, 2008; p. 1236. [Google Scholar]
- Shin-ya, K.; Kim, J.-S.; Furihata, K.; Hayakawa, Y.; Seto, H. Structure of kaitocephalin, a novel glutamate receptor antagonist produced by Eupenicillium shearii. Tetrahedron. Lett. 1997, 36, 7079–7082. [Google Scholar]
- Limon, A.; Reyes-Ruiz, J.M.; Vaswani, R.G.; Chamberlin, A.R.; Miledi, R. Kaitocephalin antagonism of glutamate receptors expressed in Xenopus oocytes. ACS. Chem. Neurosci. 2010, 1, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Reis, H.J.; Prado, M.A.; Kalapothakis, E.; Cordeiro, M.N.; Diniz, C.R.; De Marco, L.A.; Gomez, M.V.; Romano-Silva, M.A. Inhibition of glutamate uptake by a polypeptide toxin (phoneutriatoxin. 3-4) from the spider Phoneutria nigriventer. Biochem. J. 1999, 343, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Mafra, R.A.; Figueiredo, S.G.; Diniz, C.R.; Cordeiro, M.N.; Cruz, J.D.; De Lima, M.E. PhTx4, a new class of toxins from Phoneutria nigriventer spider venom, inhibits the glutamate uptake in rat brain synaptosomes. Brain. Res. 1999, 831, 297–300. [Google Scholar] [CrossRef] [PubMed]
- de Figueiredo, S.G.; de Lima, M.E.; Nascimento Cordeiro, M.; Diniz, C.R.; Patten, D.; Halliwell, R.F.; Gilroy, J.; Richardson, M. Purification and amino acid sequence of a highly insecticidal toxin from the venom of the brazilian spider Phoneutria nigriventer which inhibits NMDA-evoked currents in rat hippocampal neurones. Toxicon 2001, 39, 309–317. [Google Scholar]
- Piek, T. Neurotoxins from venoms of the Hymenoptera--twenty-five years of research in Amsterdam. Comp. Biochem. Physiol. C 1990, 96, 223–233. [Google Scholar]
- Clark, R.B.; Donaldson, P.L.; Gration, K.A.; Lambert, J.J.; Piek, T.; Ramsey, R.; Spanjer, W.; Usherwood, P.N. Block of locust muscle glutamate receptors by delta-philanthotoxin occurs after receptor activations. Brain. Res. 1982, 241, 105–114. [Google Scholar]
- Hagan, R.M.; Jones, B.J.; Jordan, C.C.; Tyers, M.B. Effect of 5-HT3 receptor antagonists on responses to selective activation of mesolimbic dopaminergic pathways in the rat. Br. J. Pharmacol. 1990, 99, 227–232. [Google Scholar] [PubMed]
- Andersen, T.F.; Tikhonov, D.B.; Bolcho, U.; Bolshakov, K.; Nelson, J.K.; Pluteanu, F.; Mellor, I.R.; Egebjerg, J.; Stromgaard, K. Uncompetitive antagonism of AMPA receptors: Mechanistic insights from studies of polyamine toxin derivatives. J. Med. Chem. 2006, 49, 5414–5423. [Google Scholar]
- Rash, L.D.; Hodgson, W.C. Pharmacology and biochemistry of spider venoms. Toxicon 2002, 40, 225–254. [Google Scholar]
- Brackley, P.T.; Bell, D.R.; Choi, S.K.; Nakanishi, K.; Usherwood, P.N. Selective antagonism of native and cloned kainate and NMDA receptors by polyamine-containing toxins. J. Pharmacol. Exp. Ther. 1993, 266, 1573–1580. [Google Scholar]
- Chiba, T.; Akizawa, T.; Matsukawa, M.; Pan-Hou, H.; Yoshioka, M. Finding of primitive polyamine toxins in the venom of a joro spider, Nephila clavata. Chem. Pharm. Bull (Tokyo) 1994, 42, 1864–1869. [Google Scholar] [PubMed]
- Sorkin, L.S.; Yaksh, T.L.; Doom, C.M. Pain models display differential sensitivity to Ca2+-permeable non-NMDA glutamate receptor antagonists. Anesthesiology. 2001, 95, 965–973. [Google Scholar]
- Gmelin, R. The free amino acids in the seeds of Acacia willardiana (Mimosaceae). Isolation of willardiin, a new plant amino acid which is probably L-beta-(3-uracil)-alpha-aminopropionic acid. Hoppe. Seylers. Z.Physiol. Chem. 1959, 316, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Evans, R.H.; Jones, A.W.; Watkins, J.C. Willardiine: A potent quisqualate-like excitant. J. Physiol (Lond) 1980, 308, 71–72. [Google Scholar]
- Shinozaki, H.; Ishida, M. Excitatory amino acids: Physiological and pharmacological probes for neuroscience research. Acta. Neurobiol. Exp (Wars) 1993, 53, 43–51. [Google Scholar] [PubMed]
- Scallet, A.C.; Ye, X. Excitotoxic mechanisms of neurodegeneration in transmissible spongiform encephalopathies. Ann. N Y. Acad. Sci. 1997, 825, 194–205. [Google Scholar]
- Agrawal, B.B.; Goldstein, I.J. Protein-carbohydrate interaction. VI. Isolation of concanavalin A by specific adsorption on cross-linked dextran gels. Biochim. Biophys. Acta. 1967, 147, 262–271. [Google Scholar] [PubMed]
- Bowie, D.; Garcia, E.P.; Marshall, J.; Traynelis, S.F.; Lange, G.D. Allosteric regulation and spatial distribution of kainate receptors bound to ancillary proteins. J. Physiol. 2003, 547, 373–385. [Google Scholar]
- Boland, L.M.; Morrill, J.A.; Bean, B.P. omega-Conotoxin block of N-type calcium channels in frog and rat sympathetic neurons. J. Neurosci. 1994, 14, 5011–5027. [Google Scholar]
- Lalo, U.V.; Pankratov, Y.V.; Arndts, D.; Krishtal, O.A. Omega-conotoxin GVIA potently inhibits the currents mediated by P2X receptors in rat DRG neurons. Brain. Res. Bull. 2001, 54, 507–512. [Google Scholar]
- Skals, M.; Jorgensen, N.R.; Leipziger, J.; Praetorius, H.A. Alpha-hemolysin from Escherichia coli uses endogenous amplification through P2X receptor activation to induce hemolysis. Proc. Natl. Acad. Sci. USA 2009, 106, 4030–4035. [Google Scholar]
- Yilmaz, O.; Yao, L.; Maeda, K.; Rose, T.M.; Lewis, E.L.; Duman, M.; Lamont, R.J.; Ojcius, D.M. ATP scavenging by the intracellular pathogen Porphyromonas gingivalis inhibits P2X7-mediated host-cell apoptosis. Cell Microbiol. 2008, 10, 863–875. [Google Scholar]
- Csolle, C.; Sperlagh, B. Peripheral origin of IL-1beta production in the rodent hippocampus under in vivo systemic bacterial lipopolysaccharide (LPS) challenge and its regulation by P2X(7) receptors. J. Neuroimmunol. 2010, 219, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Verstrepen, L.; Bekaert, T.; Chau, T.L.; Tavernier, J.; Chariot, A.; Beyaert, R. TLR-4, IL-1R and TNF-R signaling to NF-kappaB: Variations on a common theme. Cell Mol. Life. Sci. 2008, 65, 2964–2978. [Google Scholar]
- Cameron, D.J. Inhibition of macrophage mediated cytotoxicity by exogenous adenosine. 5'-triphosphate. J. Clin. Lab. Immunol. 1984, 15, 215–218. [Google Scholar] [PubMed]
- Schilling, W.P.; Wasylyna, T.; Dubyak, G.R.; Humphreys, B.D.; Sinkins, W.G. Maitotoxin and P2Z/P2X(7) purinergic receptor stimulation activate a common cytolytic pore. Am. J. Physiol. 1999, 277, C766–776. [Google Scholar]
- Lundy, P.M.; Nelson, P.; Mi, L.; Frew, R.; Minaker, S.; Vair, C.; Sawyer, T.W. Pharmacological differentiation of the P2X7 receptor and the maitotoxin-activated cationic channel. Eur. J. Pharmacol. 2004, 487, 17–28. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nasiripourdori, A.; Taly, V.; Grutter, T.; Taly, A. From Toxins Targeting Ligand Gated Ion Channels to Therapeutic Molecules. Toxins 2011, 3, 260-293. https://doi.org/10.3390/toxins3030260
Nasiripourdori A, Taly V, Grutter T, Taly A. From Toxins Targeting Ligand Gated Ion Channels to Therapeutic Molecules. Toxins. 2011; 3(3):260-293. https://doi.org/10.3390/toxins3030260
Chicago/Turabian StyleNasiripourdori, Adak, Valérie Taly, Thomas Grutter, and Antoine Taly. 2011. "From Toxins Targeting Ligand Gated Ion Channels to Therapeutic Molecules" Toxins 3, no. 3: 260-293. https://doi.org/10.3390/toxins3030260




