Listeriolysin O Causes ENaC Dysfunction in Human Airway Epithelial Cells
Abstract
:1. Introduction
2. Results
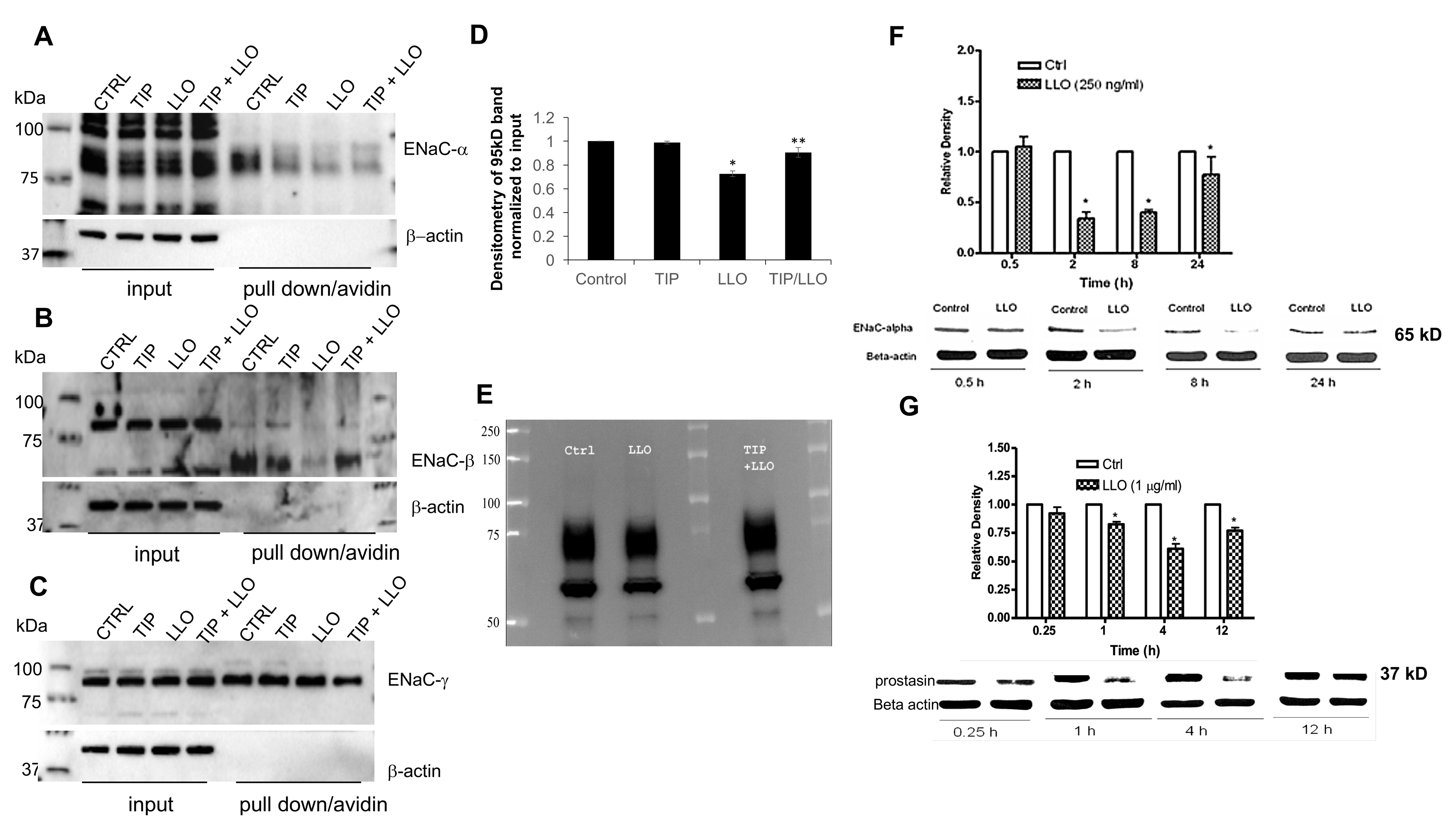
2.1. LLO Decreases ENaC-α Subunit Expression Levels in H441 Cells in a Time-Dependent Manner
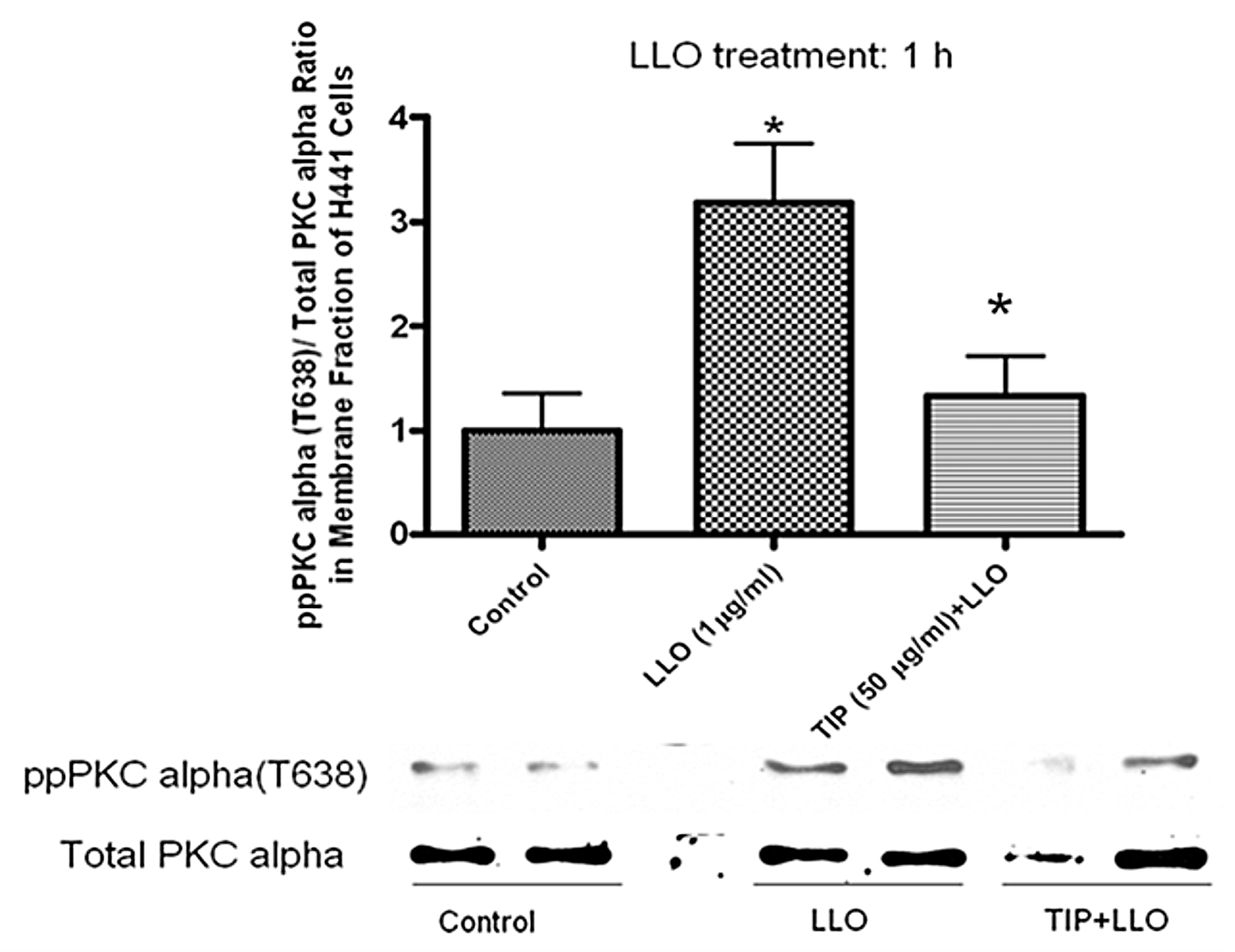
2.2. LLO-Induces PKC-α Activation in H441 Cells
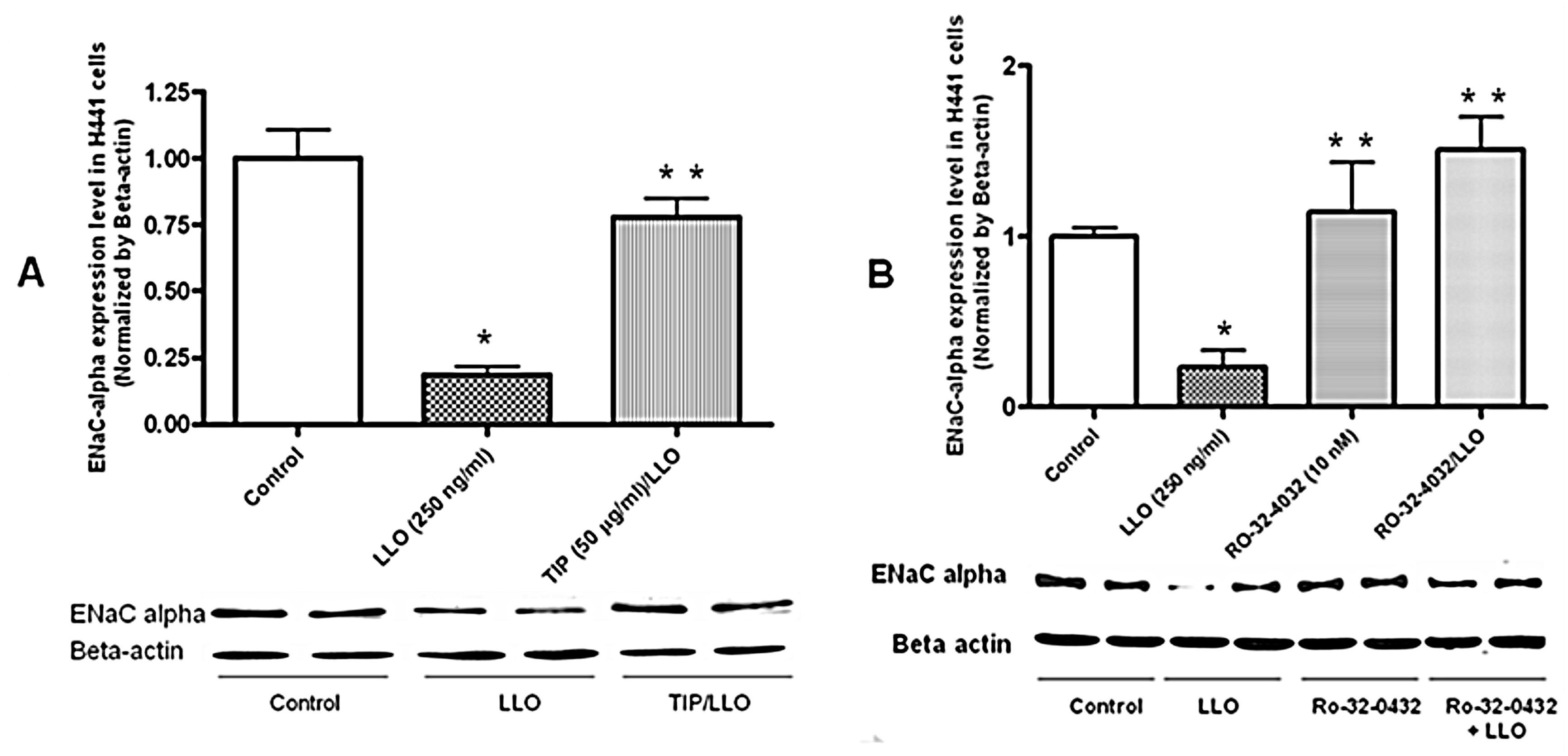
2.3. The Protein Kinase C-α Inhibitor Ro-32-0432 Significantly Prevents LLO-Mediated Reduction of ENaC-α Expression in H441 Cells
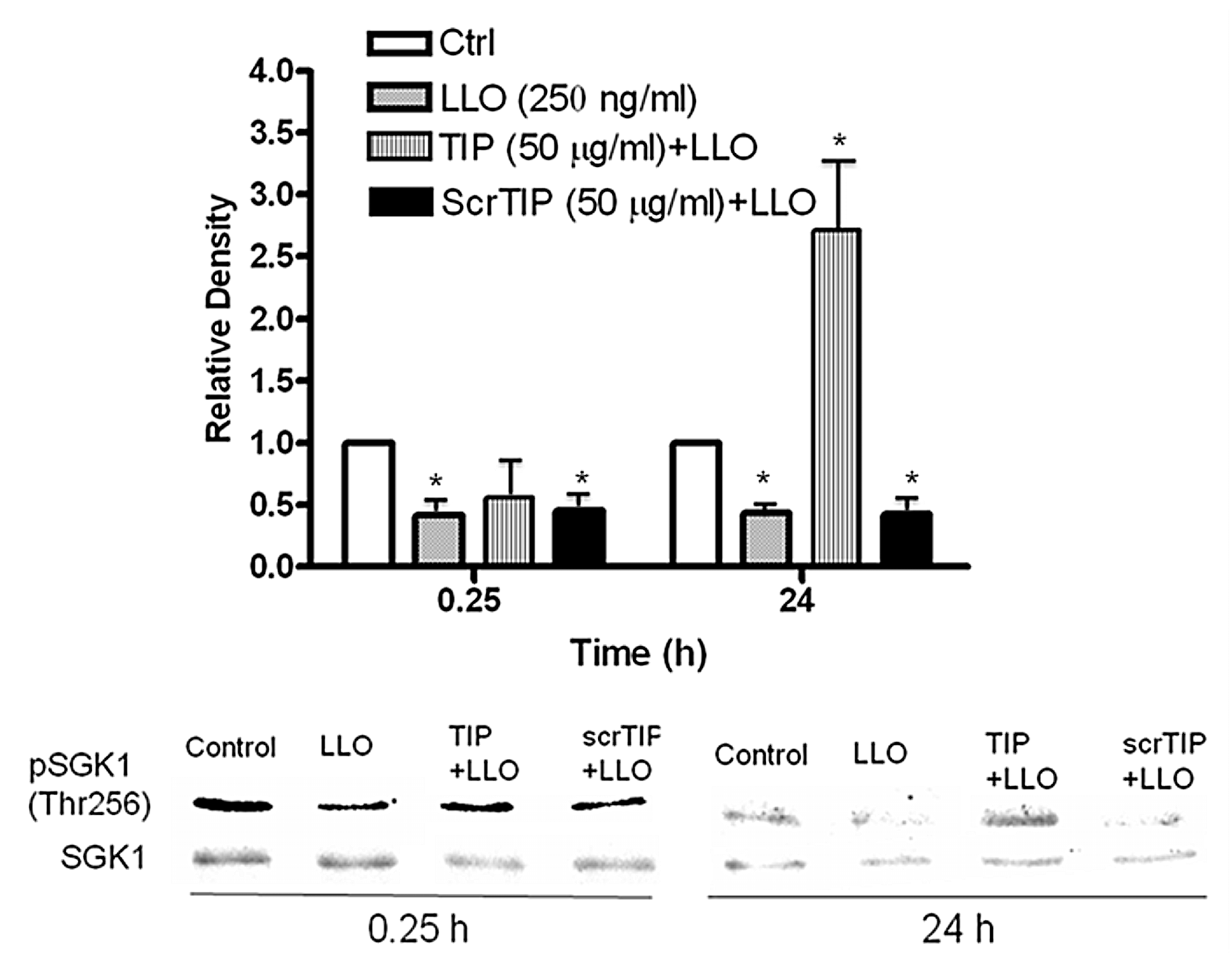
2.4. LLO Reduces Sgk-1 Phosphorylation at Residue T256 and Akt-1 Phosphorylation at Residue S473
2.5. The TNF-Derived TIP Peptide Significantly Restores pT256Sgk-1 Expression Level at 24 h in LLO-Treated H441 Cells
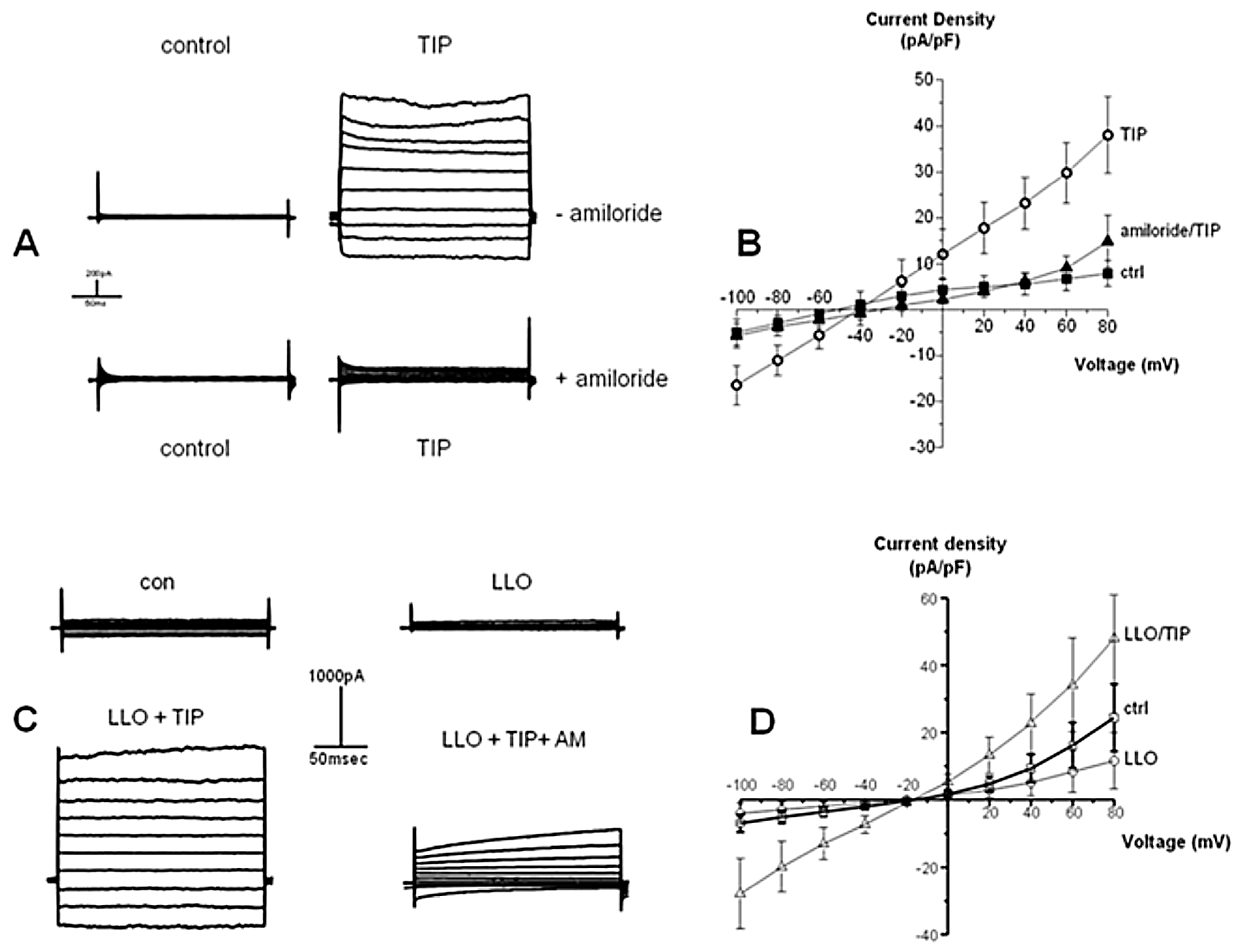
2.6. LLO Decreases Amiloride-Sensitive Sodium Current in H441 Cells
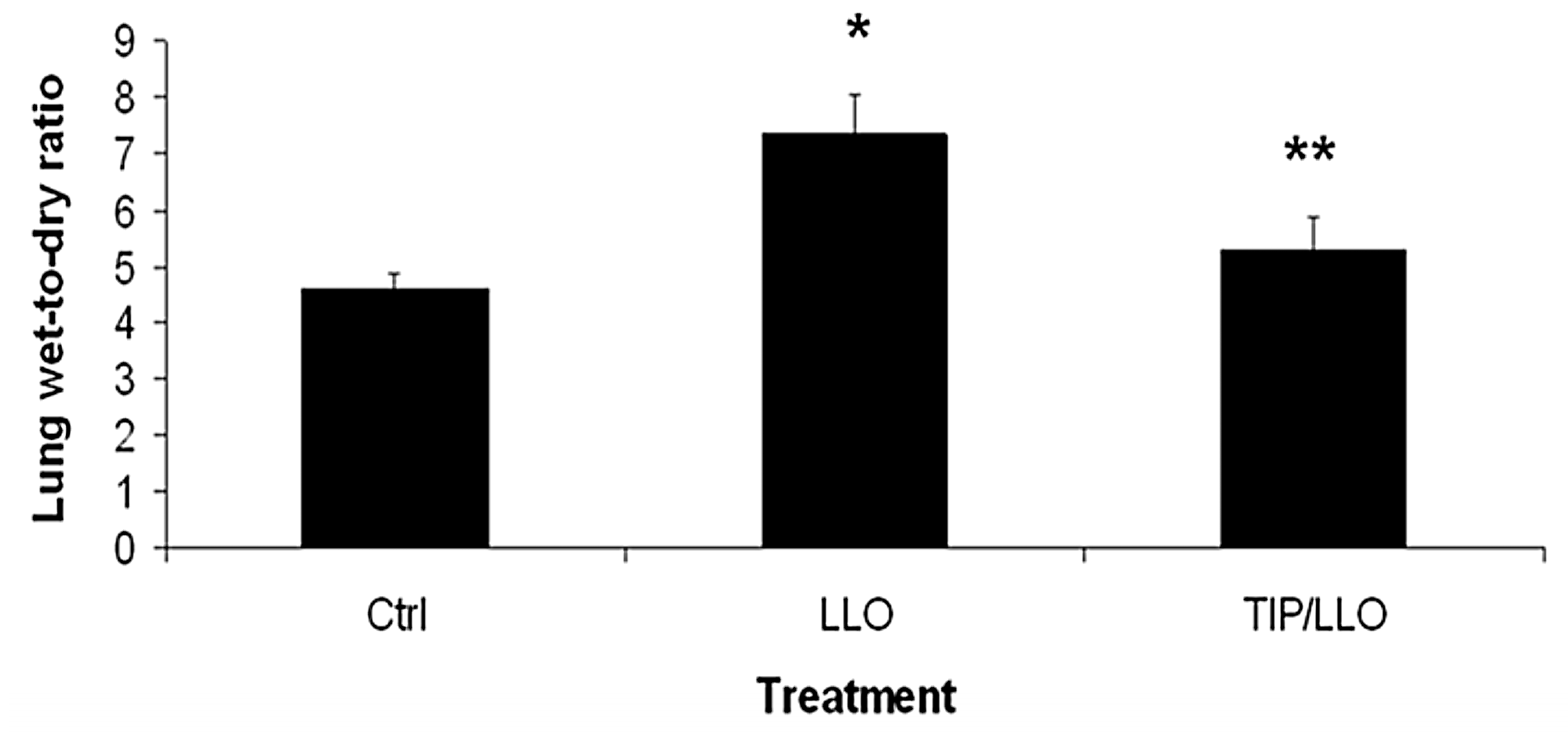
2.7. The TNF-Derived TIP Peptide Reduces Edema Formation in LLO-Treated C57BL/6 Mice
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Cells
5.2. Animals
5.3. Peptides
- human TIP (hTIP) peptide: CGQRETPEGAEAKPWYC
- scrambled TIP peptide: CGTKPWEAGEQERPAYC
5.4. Chemicals and Antibodies
5.5. Purification of LLO
5.6. Immunoblotting
5.7. Surface Biotinylation
5.8. Measurement of PKC-α Activation in H441 Cells
5.9. Whole Cell Patch Clamp Recording
5.10. Determination of the Lung Wet-to-Dry Weight Ratio
5.11. Data Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Allerberger, F.; Wagner, M. Listeriosis: A resurgent foodborne infection. Clin. Microbiol. Infect. 2010, 16, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Ananthraman, A.; Israel, R.H.; Magnussen, C.R. Pleural-pulmonary aspects of Listeria monocytogenes infection. Respiration 1983, 44, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Lerolle, N.; Zahar, J.R.; Duboc, V.; Tissier, F.; Rabbat, A. Pneumonia involving Legionella pneumophila and Listeria monocytogenes in an immunocompromised patient: An unusual coinfection. Respiration 2002, 69, 359–361. [Google Scholar] [CrossRef] [PubMed]
- Alouf, J.E. Pore-forming bacterial protein toxins: An overview. Curr. Top. Microbiol. Immunol. 2001, 257, 1–14. [Google Scholar] [PubMed]
- Repp, H.; Pamukçi, Z.; Koschinski, A.; Domann, E.; Darji, A.; Birringer, J.; Brockmeier, D.; Chakraborty, T.; Dreyer, F. Listeriolysin of Listeria monocytogenes forms Ca2+-permeable pores leading to intracellular Ca2+ oscillations. Cell. Microbiol. 2002, 4, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Eaton, D.C.; Helms, M.N.; Koval, M.; Bao, H.F.; Jain, L. The Contribution of Epithelial Sodium Channels to Alveolar Function in Health and Disease. Annu. Rev. Physiol. 2009, 71, 403–423. [Google Scholar] [CrossRef] [PubMed]
- Garty, H.; Palmer, L.G. Epithelial sodium channels: Function, structure, and regulation. Physiol. Rev. 1997, 77, 359–396. [Google Scholar] [CrossRef] [PubMed]
- Hummler, E.; Barker, P.; Gatzy, J.; Beermann, F.; Verdumo, C.; Schmidt, A.; Boucher, R.; Rossier, B.C. Early death due to defective neonatal lung liquid clearance in alpha-ENaC-deficient mice. Nat. Genet. 1996, 12, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.L.; Zhao, R.Z.; Chen, Z.X.; Shetty, S.; Idell, S.; Matalon, S. δ ENaC: A novel divergent amiloride-inhibitable sodium channel. Am. J. Physiol. Lung Cell. Mol. Physiol. 2012, 303, L1013–L1026. [Google Scholar] [CrossRef] [PubMed]
- Vadasz, I.; Raviv, I.; Sznajder, J.I. Alveolar epithelium and Na,K-ATPase in acute lung injury. Intensiv. Care Med. 2007, 33, 1243–1251. [Google Scholar] [CrossRef] [PubMed]
- Debonneville, C.; Flores, S.Y.; Kamynina, E.; Plant, P.J.; Tauxe, C.; Thomas, M.A.; Munster, C.; Chraibi, A.; Pratt, J.H.; Horisberger, J.D.; et al. Phosphorylation of Nedd4-2 by Sgk1 regulates epithelial Na(+) channel cell surface expression. EMBO J. 2001, 20, 7052–7059. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.Y.; Bhargava, A.; Mastroberardino, L.; Meijer, O.C.; Wang, J.; Buse, P.; Firestone, G.L.; Verrey, F.; Pearce, D. Epithelial sodium channel regulated by aldosterone-induced protein SGK. Proc. Natl. Acad. Sci. USA 1999, 96, 2514–2519. [Google Scholar] [CrossRef] [PubMed]
- Naray-Fejes-Toth, A.; Canessa, C.; Cleaveland, E.S.; Aldrich, G.; Fejes-Toth, G. SGK is an aldosterone-induced kinase in the renal collecting duct. Effects on epithelial Na+ channels. J. Biol. Chem. 1999, 274, 16973–16978. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.H.; Dinudom, A.; Sanchez-Perez, A.; Kumar, S.; Cook, D.I. AKT mediates the effect of insulin on epithelial sodium channels by inhibiting Nedd4-2. J. Biol. Chem. 2007, 282, 29866–29873. [Google Scholar] [CrossRef] [PubMed]
- Diakov, A.; Korbmacher, C. A novel pathway of epithelial sodium channel activation involves a serum- and glucocorticoid-inducible kinase consensus motif in the C terminus of the channel’s alpha-subunit. J. Biol. Chem. 2004, 279, 38134–38142. [Google Scholar] [CrossRef] [PubMed]
- Helms, M.N.; Yu, L.; Malik, B.; Kleinhenz, D.J.; Hart, C.M.; Eaton, D.C. Role of SGK1 in nitric oxide inhibition of ENaC in Na+-transporting epithelia. Am. J. Physiol. Cell Physiol. 2005, 289, C717–C726. [Google Scholar] [CrossRef] [PubMed]
- Yamagata, T.; Yamagata, Y.; Masse, C.; Tessier, M.C.; Brochiero, E.; Dagenais, A.; Berthiaume, Y. Modulation of Na+ transport and epithelial sodium channel expression by protein kinase C in rat alveolar epithelial cells. Can. Physiol. Pharmacol. 2005, 83, 977–987. [Google Scholar] [CrossRef] [PubMed]
- Bao, H.F.; Zhang, Z.R.; Liang, Y.Y.; Ma, J.J.; Eaton, D.C.; Ma, H.P. Ceramide mediates inhibition of the renal epithelial sodium channel by tumor necrosis factor-alpha through protein kinase C. Am. J. Physiol. 2007, 293, F1178–F1186. [Google Scholar]
- Kunzelmann, K.; Beesley, A.H.; King, N.J.; Karupiah, G.; Young, J.A.; Cook, D.I. Influenza virus inhibits amiloride-sensitive Na+ channels in respiratory epithelia. Proc. Natl. Acad. Sci. USA 2000, 97, 10282–10287. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.J.; Seth, S.; Yue, G.; Kamat, P.; Compans, R.W.; Guidot, D.; Brown, L.A.; Eaton, D.C.; Jain, L. Influenza virus inhibits ENaC and lung fluid clearance. Am. J. Physiol. Lung Cell. Mol. Physiol. 2004, 287, L366–L373. [Google Scholar] [CrossRef] [PubMed]
- Wolk, K.E.; Lazarowski, E.R.; Traylor, Z.P.; Yu, E.N.; Jewell, N.A.; Durbin, R.K.; Durbin, J.E.; Davis, I.C. Influenza A virus inhibits alveolar fluid clearance in BALB/c mice. Am. J. Respir. Crit. Care Med. 2008, 178, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Boncoeur, E.; Tardif, V.; Tessier, M.C.; Morneau, F.; Lavoie, J.; Gendreau-Berthiaume, E.; Grygorczyk, R.; Dagenais, A.; Berthiaume, Y. Modulation of epithelial sodium channel activity by lipopolysaccharide in alveolar type II cells: Involvement of purinergic signaling. Am. J. Physiol. Lung Cell. Mol. Physiol. 2010, 298, L417–L426. [Google Scholar] [CrossRef] [PubMed]
- Sherblom, A.P.; Decker, J.M.; Muchmore, A.V. The lectin-like interaction between recombinant tumor necrosis factor and uromodulin. J. Biol. Chem. 1988, 263, 5418–5424. [Google Scholar] [PubMed]
- Lucas, R.; Magez, S.; De Leys, R.; Fransen, L.; Scheerlinck, J.P.; Rampelberg, M.; Sablon, E.; De Baetselier, P. Mapping the lectin-like activity of tumor necrosis factor. Science 1994, 263, 814–817. [Google Scholar] [CrossRef] [PubMed]
- Daulouède, S.; Bouteille, B.; Moynet, D.; De Baetselier, P.; Courtois, P.; Lemesre, J.L.; Buguet, A.; Cespuglio, R.; Vincendeau, P. Human macrophage tumor necrosis factor (TNF)-alpha production induced by Trypanosoma brucei gambiense and the role of TNF-alpha in parasite control. J. Infect. Dis. 2001, 183, 988–991. [Google Scholar] [CrossRef] [PubMed]
- Magez, S.; Geuskens, M.; Beschin, A.; del Favero, H.; Verschueren, H.; Lucas, R.; Pays, E.; de Baetselier, P. Specific uptake of tumor necrosis factor-alpha is involved in growth control of Trypanosoma brucei. J. Cell Biol. 1997, 137, 715–727. [Google Scholar] [CrossRef] [PubMed]
- Rocha, F.A.C.; Alves, A.M.C.V.; Rocha, M.F.G.; Cordeiro, R.A.; Brilhante, R.S.N.; Pinto, A.C.M.D.; Nunes, R.M.; Girão, V.C.C.; Sidrim, J.J.C. Tumor necrosis factor prevents Candida albicans biofilm formation. Sci. Rep. 2017, 7, 1206. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, N.; Jayr, C.; Lazrak, A.; Wang, Y.; Lucas, R.; Matalon, S.; Matthay, M.A. Mechanisms of TNF-alpha stimulation of amiloride-sensitive sodium transport across alveolar epithelium. Am. J. Physiol. Lung Cell. Mol. Physiol. 2001, 280, L1258–L1265. [Google Scholar] [CrossRef] [PubMed]
- Braun, C.; Hamacher, J.; Morel, D.R.; Wendel, A.; Lucas, R. Dichotomal role of TNF in experimental pulmonary edema reabsorption. J. Immunol. 2005, 175, 3402–3408. [Google Scholar] [CrossRef] [PubMed]
- Elia, N.; Tapponnier, M.; Matthay, M.A.; Hamacher, J.; Pache, J.C.; Brundler, M.A.; Totsch, M.; De Baetselier, P.; Fransen, L.; Fukuda, N.; et al. Functional identification of the alveolar edema reabsorption activity of murine tumor necrosis factor-alpha. Am. J. Respir. Crit. Care Med. 2003, 168, 1043–1050. [Google Scholar] [CrossRef] [PubMed]
- Hamacher, J.; Stammberger, U.; Roux, J.; Kumar, S.; Yang, G.; Xiong, C.; Schmid, R.A.; Fakin, R.M.; Chakraborty, T.; Hossain, H.M.; et al. The lectin-like domain of tumor necrosis factor improves lung function after rat lung transplantation—Potential role for a reduction in reactive oxygen species generation. Crit. Care Med. 2010, 38, 871–878. [Google Scholar] [CrossRef] [PubMed]
- Vadasz, I.; Schermuly, R.T.; Ghofrani, H.A.; Rummel, S.; Wehner, S.; Muhldorfer, I.; Schafer, K.P.; Seeger, W.; Morty, R.E.; Grimminger, F.; et al. The lectin-like domain of tumor necrosis factor-alpha improves alveolar fluid balance in injured isolated rabbit lungs. Crit. Care Med. 2008, 36, 1543–1550. [Google Scholar] [CrossRef] [PubMed]
- Czikora, I.; Alli, A.; Bao, H.F.; Kaftan, D.; Sridhar, S.; Apell, H.J.; Gorshkov, B.; White, R.; Zimmermann, A.; Wendel, A.; et al. A novel tumor necrosis factor-mediated mechanism of direct epithelial sodium channel activation. Am. J. Respir. Crit. Care Med. 2014, 190, 522–532. [Google Scholar] [CrossRef] [PubMed]
- Lucas, R.; Yue, Q.; Alli, A.; Duke, B.J.; Al-Khalili, O.; Thai, T.L.; Hamacher, J.; Sridhar, S.; Lebedyeva, I.; Su, H.; et al. The Lectin-like Domain of TNF Increases ENaC Open Probability through a Novel Site at the Interface between the Second Transmembrane and C-terminal Domains of the α-Subunit. J. Biol. Chem. 2016, 291, 23440–23451. [Google Scholar] [CrossRef] [PubMed]
- Alli, A.A.; Bao, H.F.; Aldrugh, Y.; Song, J.Z.; Ma, H.P.; Yu, L.; Al-Khalili, O.; Eaton, D.C. Phosphatidylinositol phosphate-dependent regulation of Xenopus ENaC by MARCKS protein. Am. J. Physiol. Ren. Physiol. 2012, 303, F800–F811. [Google Scholar] [CrossRef] [PubMed]
- Planès, C.; Randrianarison, N.H.; Charles, R.P.; Frateschi, S.; Cluzeaud, F.; Vuagniaux, G.; Soler, P.; Clerici, C.; Rossier, B.C.; Hummler, E. ENaC-mediated alveolar fluid clearance and lung fluid balance depend on the channel-activating protease 1. EMBO Mol. Med. 2010, 2, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, S.F. Structural basis of protein kinase C isoform function. Physiol. Rev. 2008, 88, 1341–1378. [Google Scholar] [CrossRef] [PubMed]
- Mackay, H.J.; Twelves, C.J. Targeting the protein kinase C family: Are we there yet? Nat. Rev. 2007, 7, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Ling, B.N.; Eaton, D.C. Effects of luminal Na+ on single Na+ channels in A6 cells, a regulatory role for protein kinase C. Am. J. Physiol. 1989, 256, F1094–F1103. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Martinez, J.M.; Alessi, D.R. mTOR complex 2 (mTORC2) controls hydrophobic motif phosphorylation and activation of serum- and glucocorticoid-induced protein kinase 1 (SGK1). Biochem. J. 2008, 416, 375–385. [Google Scholar] [CrossRef] [PubMed]
- Biondi, R.M.; Kieloch, A.; Currie, R.A.; Deak, M.; Alessi, D.R. The PIF-binding pocket in PDK1 is essential for activation of S6K and SGK, but not PKB. EMBO J. 2001, 20, 4380–4390. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.Y.; Zhou, Q.L.; Holik, J.; Patel, S.; Leszyk, J.; Coleman, K.; Chouinard, M.; Czech, M.P. Identification of WNK1 as a substrate of Akt/protein kinase B and a negative regulator of insulin-stimulated mitogenesis in 3T3-L1 cells. J. Biol. Chem. 2005, 280, 21622–21628. [Google Scholar] [CrossRef] [PubMed]
- Ramminger, S.J.; Richard, K.; Inglis, S.K.; Land, S.C.; Olver, R.E.; Wilson, S.M. A regulated apical Na(+) conductance in dexamethasone-treated H441 airway epithelial cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 2004, 287, L411–L419. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.G.; Gallacher, M.; Olver, R.E.; Wilson, S.M. The regulation of selective and nonselective Na+ conductances in H441 human airway epithelial cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 2008, 294, L942–L954. [Google Scholar] [CrossRef] [PubMed]
- Taruno, A.; Niisato, N.; Marunaka, Y. Intracellular calcium plays a role as the second messenger of hypotonic stress in gene regulation of SGK1 and ENaC in renal epithelial A6 cells. Am. J. Physiol. Ren. Physiol. 2008, 294, F177–F186. [Google Scholar] [CrossRef] [PubMed]
- Rezaiguia, S.; Garat, C.; Delclaux, C.; Meignan, M.; Fleury, J.; Legrand, P.; Matthay, M.A.; Jayr, C. Acute bacterial pneumonia in rats increases alveolar epithelial fluid clearance by a tumor necrosis factor-alpha-dependent mechanism. J. Clin. Investig. 1997, 99, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Meltzer, R.H.; Kapoor, N.; Qadri, Y.J.; Anderson, S.J.; Fuller, C.M.; Benos, D.J. Heteromeric assembly of acid-sensitive ion channel and epithelial sodium channel subunits. J. Biol. Chem. 2007, 282, 25548–25559. [Google Scholar] [CrossRef] [PubMed]
- Trac, P.T.; Thai, T.L.; Linck, V.; Zou, L.; Greenlee, M.M.; Yue, Q.; Al-Khalili, O.; Alli, A.A.; Eaton, A.F.; Eaton, D.C. Alveolar non-selective channels are ASIC1a/α-ENaC channels and contribute to AFC. Am. J. Physiol. Lung Cell. Mol. Physiol. 2017, 312, L797–L811. [Google Scholar] [CrossRef] [PubMed]
- Czikora, I.; Alli, A.A.; Sridhar, S.; Matthay, M.A.; Pillich, H.; Hudel, M.; Berisha, B.; Gorshkov, B.; Romero, M.J.; Gonzales, J.; et al. Epithelial Sodium Channel-α Mediates the Protective Effect of the TNF-Derived TIP Peptide in Pneumolysin-Induced Endothelial Barrier Dysfunction. Front. Immunol. 2017, 8, 842. [Google Scholar] [CrossRef] [PubMed]
- Xiong, C.; Yang, G.; Kumar, S.; Aggarwal, S.; Leustik, M.; Snead, C.; Hamacher, J.; Fischer, B.; Umapathy, N.S.; Hossain, H.; et al. The lectin-like domain of TNF protects from listeriolysin-induced hyperpermeability in human pulmonary microvascular endothelial cells—A crucial role for protein kinase C-alpha inhibition. Vascul. Pharmacol. 2010, 52, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Krenn, K.; Lucas, R.; Croizé, A.; Boehme, S.; Klein, K.U.; Hermann, R.; Markstaller, K.; Ullrich, R. Inhaled AP301 for treatment of pulmonary edema in mechanically ventilated patients with acute respiratory distress syndrome: A phase IIa randomized placebo-controlled trial. Crit. Care 2017, 21, 194. [Google Scholar] [CrossRef] [PubMed]
- Matalon, S.; Bartoszewski, R.; Collawn, J.F. Role of epithelial sodium channels in the regulation of lung fluid homeostasis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2015, 309, L1229–L1238. [Google Scholar] [CrossRef] [PubMed]
- Eaton, D.C.; Malik, B.; Bao, H.F.; Yu, L.; Jain, L. Regulation of epithelial sodium channel trafficking by ubiquitination. Proc. Am. Thorac. Soc. 2010, 7, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Butterworth, M.B.; Edinger, R.S.; Silvis, M.R.; Gallo, L.I.; Liang, X.; Apodaca, G.; Frizzell, R.A.; Johnson, J.P. Rab11b regulates the trafficking and recycling of the epithelial sodium channel (ENaC). Am. J. Physiol. Ren. Physiol. 2012, 302, F581–F590. [Google Scholar] [CrossRef] [PubMed]
- Deribe, Y.L.; Pawson, T.; Dikic, I. Post-translational modifications in signal integration. Nat. Struct. Mol. Biol. 2010, 17, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Hamon, M.A.; Batsche, E.; Regnault, B.; Tham, T.N.; Seveau, S.; Muchardt, C.; Cossart, P. Histone modifications induced by a family of bacterial toxins. Proc. Natl. Acad. Sci. USA 2007, 104, 13467–13472. [Google Scholar] [CrossRef] [PubMed]
- Ribet, D.; Hamon, M.; Gouin, E.; Nahori, M.A.; Impens, F.; Neyret-Kahn, H.; Gevaert, K.; Vandekerckhove, J.; Dejean, A.; Cossart, P. Listeria monocytogenes impairs SUMOylation for efficient infection. Nature 2010, 464, 1192–1195. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, G.; Pillich, H.; White, R.; Czikora, I.; Pochic, I.; Yue, Q.; Hudel, M.; Gorshkov, B.; Verin, A.; Sridhar, S.; et al. Listeriolysin O Causes ENaC Dysfunction in Human Airway Epithelial Cells. Toxins 2018, 10, 79. https://doi.org/10.3390/toxins10020079
Yang G, Pillich H, White R, Czikora I, Pochic I, Yue Q, Hudel M, Gorshkov B, Verin A, Sridhar S, et al. Listeriolysin O Causes ENaC Dysfunction in Human Airway Epithelial Cells. Toxins. 2018; 10(2):79. https://doi.org/10.3390/toxins10020079
Chicago/Turabian StyleYang, Guang, Helena Pillich, Richard White, Istvan Czikora, Isabelle Pochic, Qiang Yue, Martina Hudel, Boris Gorshkov, Alexander Verin, Supriya Sridhar, and et al. 2018. "Listeriolysin O Causes ENaC Dysfunction in Human Airway Epithelial Cells" Toxins 10, no. 2: 79. https://doi.org/10.3390/toxins10020079





