Weight Management Interventions in Women with and without PCOS: A Systematic Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Selection Criteria
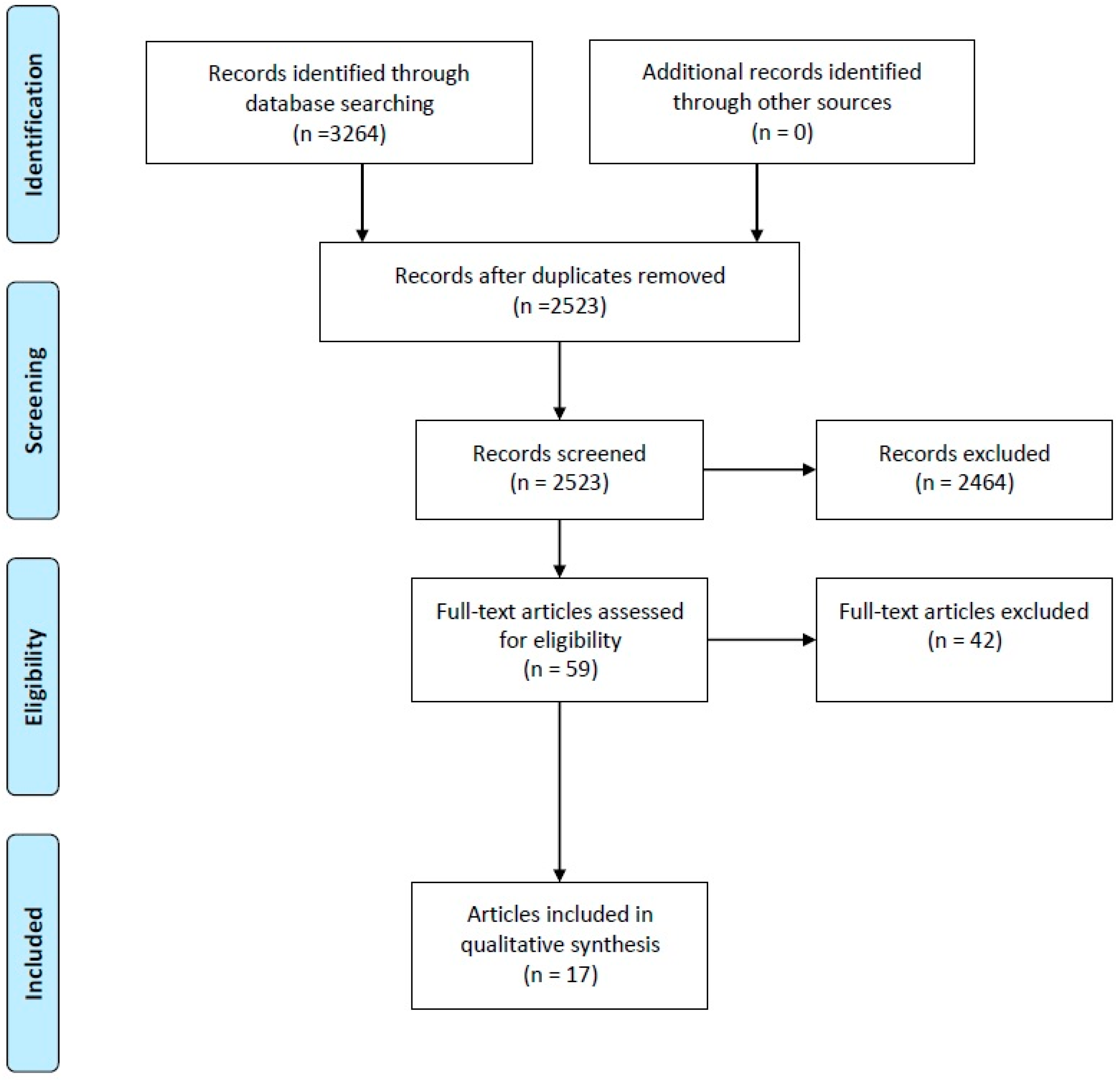
2.2. Systematic Search for Evidence
2.3. Inclusion of Studies
2.4. Quality Appraisal of the Evidence
2.5. Data Extraction
2.6. Data Synthesis
3. Results
3.1. Characteristics and Quality of Included Studies
3.2. Outcomes: Anthropometric
3.2.1. Weight
3.2.2. Body Mass Index (BMI)
3.2.3. Waist Circumference (WC)
3.2.4. Body Composition
3.3. Outcomes: Fertility
Ovulation
3.4. Outcomes: Reproductive Non-Fertility
3.4.1. Total Testosterone
3.4.2. Sex Hormone-Binding Globulin (SHBG)
3.4.3. Free Androgen Index (FAI)
3.5. Outcomes: Metabolic
3.5.1. Fasting Glucose
3.5.2. Oral Glucose Tolerance Test (OGTT-Glucose)
3.5.3. Fasting Insulin
3.5.4. Lipids
3.5.5. Blood Pressure (BP)
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- March, W.A.; Moore, V.M.; Willson, K.J.; Phillips, D.I.; Norman, R.J.; Davies, M.J. The prevalence of polycystic ovary syndrome in a community sample assessed under contrasting diagnostic criteria. Hum. Reprod. 2010, 25, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Asuncion, M.; Calvo, R.M.; San Millan, J.L.; Sancho, J.; Avila, S.; Escobar-Morreale, H.F. A prospective study of the prevalence of the polycystic ovary syndrome in unselected caucasian women from Spain. J. Clin. Endocrinol. Metab. 2000, 85, 2434–2438. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Kouli, C.R.; Bergiele, A.T.; Filandra, F.A.; Tsianateli, T.C.; Spina, G.G.; Zapanti, E.D.; Bartzis, M.I. A survey of the polycystic ovary syndrome in the Greek island of Lesbos: Hormonal and metabolic profile. J. Clin. Endocrinol. Metab. 1999, 84, 4006–4011. [Google Scholar] [CrossRef] [PubMed]
- Stepto, N.K.; Cassar, S.; Joham, A.E.; Hutchison, S.K.; Harrison, C.L.; Goldstein, R.F.; Teede, H.J. Women with polycystic ovary syndrome have intrinsic insulin resistance on euglycaemic-hyperinsulaemic clamp. Hum. Reprod. 2013, 28, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Moran, L.J.; Misso, M.L.; Wild, R.A.; Norman, R.J. Impaired glucose tolerance, type 2 diabetes and metabolic syndrome in polycystic ovary syndrome: A systematic review and meta-analysis. Hum. Reprod. Update 2010, 16, 347–363. [Google Scholar] [CrossRef] [PubMed]
- Teede, H.; Deeks, A.; Moran, L. Polycystic ovary syndrome: A complex condition with psychological, reproductive and metabolic manifestations that impacts on health across the lifespan. BMC Med. 2010, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wild, R.A.; Carmina, E.; Diamanti-Kandarakis, E.; Dokras, A.; Escobar-Morreale, H.F.; Futterweit, W.; Lobo, R.; Norman, R.J.; Talbott, E.; Dumesic, D.A. Assessment of cardiovascular risk and prevention of cardiovascular disease in women with the polycystic ovary syndrome: A consensus statement by the Androgen Excess and Polycystic Ovary Syndrome (AE-PCOS) Society. J. Clin. Endocrinol. Metab. 2010, 95, 2038–2049. [Google Scholar] [CrossRef] [PubMed]
- Broekmans, F.J.; Knauff, E.A.H.; Valkenburg, O.; Laven, J.S.; Eijkemans, M.J.; Fauser, B. PCOS according to the Rotterdam consensus criteria: Change in prevalence among WHO-II anovulation and association with metabolic factors. BJOG 2006, 113, 1210–1217. [Google Scholar] [CrossRef] [PubMed]
- Boomsma, C.M.; Eijkemans, M.J.; Hughes, E.G.; Visser, G.H.; Fauser, B.C.; Macklon, N.S. A meta-analysis of pregnancy outcomes in women with polycystic ovary syndrome. Hum. Reprod. Update 2006, 12, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Barry, J.A.; Kuczmierczyk, A.R.; Hardiman, P.J. Anxiety and depression in polycystic ovary syndrome: A systematic review and meta-analysis. Hum. Reprod. 2011, 26, 2442–2451. [Google Scholar] [CrossRef] [PubMed]
- Vink, J.M.; Sadrzadeh, S.; Lambalk, C.B.; Boomsma, D.I. Heritability of polycystic ovary syndrome in a Dutch twin-family study. J. Clin. Endocrinol. Metab. 2006, 91, 2100–2104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dunaif, A. Insulin resistance in women with polycystic ovary syndrome. Fertil. Steril. 2006, 86 (Suppl. 1), S13–S14. [Google Scholar] [CrossRef] [PubMed]
- Gilling-Smith, C.; Story, H.; Rogers, V.; Franks, S. Evidence for a primary abnormality of thecal cell steroidogenesis in the polycystic ovary syndrome. Clin. Endocrinol. 1997, 47, 93–99. [Google Scholar] [CrossRef]
- Moret, M.; Stettler, R.; Rodieux, F.; Gaillard, R.C.; Waeber, G.; Wirthner, D.; Giusti, V.; Tappy, L.; Pralong, F.P. Insulin modulation of luteinizing hormone secretion in normal female volunteers and lean polycystic ovary syndrome patients. Neuroendocrinology 2009, 89, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Davies, M.J.; Norman, R.J.; Moran, L.J. Overweight, obesity and central obesity in women with polycystic ovary syndrome: A systematic review and meta-analysis. Hum. Reprod. Update 2012, 18, 618–637. [Google Scholar] [CrossRef] [PubMed]
- Yildiz, B.O.; Bozdag, G.; Yapici, Z.; Esinler, I.; Yarali, H. Prevalence, phenotype and cardiometabolic risk of polycystic ovary syndrome under different diagnostic criteria. Hum. Reprod. 2012, 27, 3067–3073. [Google Scholar] [CrossRef] [PubMed]
- Teede, H.J.; Joham, A.E.; Paul, E.; Moran, L.J.; Loxton, D.; Jolley, D.; Lombard, C. Longitudinal weight gain in women identified with polycystic ovary syndrome: Results of an observational study in young women. Obesity 2013, 21, 1526–1532. [Google Scholar] [CrossRef] [PubMed]
- Balen, A.H.; Conway, G.S.; Kaltsas, G.; Techatrasak, K.; Manning, P.J.; West, C.; Jacobs, H.S. Polycystic ovary syndrome: The spectrum of the disorder in 1741 patients. Hum. Reprod. 1995, 10, 2107–2111. [Google Scholar] [CrossRef] [PubMed]
- Kiddy, D.S.; Sharp, P.S.; White, D.M.; Scanlon, M.F.; Mason, H.D.; Bray, C.S.; Polson, D.W.; Reed, M.J.; Franks, S. Differences in clinical and endocrine features between obese and non-obese subjects with polycystic ovary syndrome: An analysis of 263 consecutive cases. Clin. Endocrinol. (Oxf.) 1990, 32, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Hart, R.; Doherty, D.A. The potential implications of a PCOS diagnosis on a woman’s long-term health using data linkage. J. Clin. Endocrinol. Metab. 2015, 100, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Moran, L.J.; Pasquali, R.; Teede, H.J.; Hoeger, K.M.; Norman, R.J. Treatment of obesity in polycystic ovary syndrome: A position statement of the Androgen Excess and Polycystic Ovary Syndrome Society. Fertil. Steril. 2009, 92, 1966–1982. [Google Scholar] [CrossRef] [PubMed]
- Teede, H.J.; Misso, M.L.; Deeks, A.A.; Moran, L.J.; Stuckey, B.G.; Wong, J.L.; Norman, R.J.; Costello, M.F. Assessment and management of polycystic ovary syndrome: Summary of an evidence-based guideline. Med. J. Aust. 2011, 195, S65–S112. [Google Scholar] [CrossRef] [PubMed]
- European Society of Human Reproduction and Embryology; The Thessaloniki, and ASRM-Sponsored PCOS Consensus Workshop Group. Consensus on infertility treatment related to polycystic ovary syndrome. Hum. Reprod. 2008, 23, 462–477. [Google Scholar] [CrossRef]
- Legro, R.S.; Dodson, W.C.; Kris-Etherton, P.M.; Kunselman, A.R.; Stetter, C.M.; Williams, N.I.; Gnatuk, C.L.; Estes, S.J.; Fleming, J.; Allison, K.C.; et al. Randomized controlled trial of preconception interventions in infertile women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2015, 100, 4048–4058. [Google Scholar] [CrossRef] [PubMed]
- Moran, L.J.; Noakes, M.; Clifton, P.M.; Tomlinson, L.; Galletly, C.; Norman, R.J. Dietary composition in restoring reproductive and metabolic physiology in overweight women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2003, 88, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Moran, L.J.; Noakes, M.; Clifton, P.M.; Wittert, G.A.; Le Roux, C.W.; Ghatei, M.A.; Bloom, S.R.; Norman, R.J. Postprandial ghrelin, cholecystokinin, peptide YY, and appetite before and after weight loss in overweight women with and without polycystic ovary syndrome. Am. J. Clin. Nutr. 2007, 86, 1603–1610. [Google Scholar] [PubMed]
- Robinson, S.; Chan, S.-P.; Spacey, S.; Anyaoku, V.; Johnston, D.G.; Franks, S. Postprandial thermogenesis is reduced in polycystic ovary syndrome and is associated with increased insulin resistance. Clin. Endocrinol. 1992, 36, 537–543. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle–Ottawa Scale (NOS) for Assessing the Quality of Nonrandomized Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 7 September 2017).
- Diamanti-Kandarakis, E.; Katsikis, I.; Piperi, C.; Alexandraki, K.; Panidis, D. Effect of long-term orlistat treatment on serum levels of advanced glycation end-products in women with polycystic ovary syndrome. Clin. Endocrinol. 2007, 66, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, S.K.; Stepto, N.K.; Harrison, C.L.; Moran, L.J.; Strauss, B.J.; Teede, H.J. Effects of exercise on insulin resistance and body composition in overweight and obese women with and without polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2011, 96, E48–E56. [Google Scholar] [CrossRef] [PubMed]
- Harrison, C.L.; Stepto, N.K.; Hutchison, S.K.; Teede, H.J. The impact of intensified exercise training on insulin resistance and fitness in overweight and obese women with and without polycystic ovary syndrome. Clin. Endocrinol. (Oxf.) 2012, 76, 351–357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kowalska, I.; Kinalski, M.; Straczkowski, M.; Wolczyski, S.; Kinalska, I. Insulin, leptin, IGF-I and insulin-dependent protein concentrations after insulin-sensitizing therapy in obese women with polycystic ovary syndrome. Eur. J. Endocrinol. 2001, 144, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Moran, L.J.; Noakes, M.; Clifton, P.M.; Wittert, G.A.; Belobrajdic, D.P.; Norman, R.J. C-reactive protein before and after weight loss in overweight women with and without polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2007, 92, 2944–2951. [Google Scholar] [CrossRef] [PubMed]
- Panidis, D.; Tziomalos, K.; Papadakis, E.; Chatzis, P.; Kandaraki, E.A.; Tsourdi, E.A.; Katsikis, I. The role of orlistat combined with lifestyle changes in the management of overweight and obese patients with polycystic ovary syndrome. Clin. Endocrinol. 2014, 80, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Vosnakis, C.; Georgopoulos, N.A.; Rousso, D.; Mavromatidis, G.; Katsikis, I.; Roupas, N.D.; Mamali, I.; Panidis, D. Diet, physical exercise and Orlistat administration increase serum anti-Mullerian hormone (AMH) levels in women with polycystic ovary syndrome (PCOS). Gynecol. Endocrinol. 2013, 29, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Panidis, D.; Farmakiotis, D.; Rousso, D.; Kourtis, A.; Katsikis, I.; Krassas, G. Obesity, weight loss, and the polycystic ovary syndrome: Effect of treatment with diet and orlistat for 24 weeks on insulin resistance and androgen levels. Fertil. Steril. 2008, 89, 899–906. [Google Scholar] [CrossRef] [PubMed]
- Pasquali, R.; Gambineri, A.; Biscotti, D.; Vicennati, V.; Gagliardi, L.; Colitta, D.; Fiorini, S.; Cognigni, G.E.; Filicori, M.; Morselli-Labate, A.M. Effect of long-term treatment with metformin added to hypocaloric diet on body composition, fat distribution, and androgen and insulin levels in abdominally obese women with and without the polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2000, 85, 2767–2774. [Google Scholar] [CrossRef] [PubMed]
- Toscani, M.K.; Mario, F.M.; Radavelli-Bagatini, S.; Wiltgen, D.; Matos, M.C.; Spritzer, P.M. Effect of high-protein or normal-protein diet on weight loss, body composition, hormone, and metabolic profile in southern Brazilian women with polycystic ovary syndrome: A randomized study. Gynecol. Endocrinol. 2011, 27, 925–930. [Google Scholar] [CrossRef] [PubMed]
- Villa, P.; Valle, D.; Mancini, A.; De Marinis, L.; Pavone, V.; Fulghesu, A.M.; Mancuso, S.; Lanzone, A. Effect of opioid blockade on insulin and growth hormone (GH) secretion in patients with polycystic ovary syndrome: The heterogeneity of impaired GH secretion is related to both obesity and hyperinsulinism. Fertil. Steril. 1999, 71, 115–121. [Google Scholar] [CrossRef]
- Kahal, H.; Aburima, A.; Ungvari, T.; Rigby, A.S.; Coady, A.M.; Vince, R.V.; Ajjan, R.A.; Kilpatrick, E.S.; Naseem, K.M.; Atkin, S.L. The effects of treatment with liraglutide on atherothrombotic risk in obese young women with polycystic ovary syndrome and controls. BMC Endocr. Disord. 2015, 15, 14. [Google Scholar] [CrossRef] [PubMed]
- Nikokavoura, E.A.; Johnston, K.L.; Broom, J.; Wrieden, W.L.; Rolland, C. Weight loss for women with and without polycystic ovary syndrome following a very low-calorie diet in a community-based setting with trained facilitators for 12 weeks. Diabetes Metab. Syndr. Obes. Targets Ther. 2015, 8, 495. [Google Scholar] [CrossRef] [PubMed]
- Cheang, K.I.; Sistrun, S.N.; Morel, K.S.; Nestler, J.E. Effect on insulin-stimulated release of D-chiro-inositol-containing inositolphosphoglycan mediator during weight loss in obese women with and without polycystic ovary syndrome. Int. J. Endocrinol. 2016, 2016, 7631804. [Google Scholar] [CrossRef] [PubMed]
- Kogure, G.S.; Miranda-Furtado, C.L.; Silva, R.C.; Melo, A.S.; Ferriani, R.A.; De Sá, M.; Dos Reis, R.M. Resistance exercise impacts lean muscle mass in women with polycystic ovary syndrome. Med. Sci. Sports Exerc. 2016, 48, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, S.; Ganguly, I.; Bhandari, M.; Agarwal, P.; Singh, A.; Gupta, N.; Mishra, A. Effect of sleeve gastrectomy bariatric surgery-induced weight loss on serum AMH levels in reproductive aged women. Gynecol. Endocrinol. 2016, 32, 799–802. [Google Scholar] [CrossRef] [PubMed]
- Al-Eisa, E.; Gabr, S.A.; Alghadir, A.H. Effects of supervised aerobic training on the levels of anti-Mullerian hormone and adiposity measures in women with normo-ovulatory and polycystic ovary syndrome. J. Pak. Med. Assoc. 2017, 67, 499. [Google Scholar] [PubMed]
- Georgopoulos, N.A.; Saltamavros, A.D.; Vervita, V.; Karkoulias, K.; Adonakis, G.; Decavalas, G.; Kourounis, G.; Markou, K.B.; Kyriazopoulou, V. Basal metabolic rate is decreased in women with polycystic ovary syndrome and biochemical hyperandrogenemia and is associated with insulin resistance. Fertil. Steril. 2009, 92, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Hirschberg, A.L.; Naessen, S.; Stridsberg, M.; Bystrom, B.; Holtet, J. Impaired cholecystokinin secretion and disturbed appetite regulation in women with polycystic ovary syndrome. Gynecol. Endocrinol. 2004, 19, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Moran, L.J.; Noakes, M.; Clifton, P.M.; Wittert, G.A.; Tomlinson, L.; Galletly, C.; Luscombe, N.D.; Norman, R.J. Ghrelin and measures of satiety are altered in polycystic ovary syndrome but not differentially affected by diet composition. J. Clin. Endocrinol. Metab. 2004, 89, 3337–3344. [Google Scholar] [CrossRef] [PubMed]
- Moran, L.J.; Noakes, M.; Clifton, P.M.; Wittert, G.A.; Williams, G.; Norman, R.J. Short-term meal replacements followed by dietary macronutrient restriction enhance weight loss in polycystic ovary syndrome. Am. J. Clin. Nutr. 2006, 84, 77–87. [Google Scholar] [PubMed]
- Moran, L.J.; Ranasinha, S.; Zoungas, S.; McNaughton, S.A.; Brown, W.J.; Teede, H.J. The contribution of diet, physical activity and sedentary behaviour to body mass index in women with and without polycystic ovary syndrome. Hum. Reprod. 2013, 28, 2276–2283. [Google Scholar] [CrossRef] [PubMed]
- Legro, R.S.; Kunselman, A.R.; Dodson, W.C.; Dunaif, A. Prevalence and predictors of risk for type 2 diabetes mellitus and impaired glucose tolerance in polycystic ovary syndrome: A prospective, controlled study in 254 affected women. J. Clin. Endocrinol. Metab. 1999, 84. [Google Scholar] [CrossRef]
- Naderpoor, N.; Shorakae, S.; de Courten, B.; Misso, M.L.; Moran, L.J.; Teede, H.J. Metformin and lifestyle modification in polycystic ovary syndrome: Systematic review and meta-analysis. Hum. Reprod. Update 2015, 21, 560–574. [Google Scholar] [CrossRef] [PubMed]
- Apovian, C.M.; Aronne, L.J.; Bessesen, D.H.; McDonnell, M.E.; Murad, M.H.; Pagotto, U.; Ryan, D.H.; Still, C.D. Pharmacological management of obesity: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2015, 100, 342–362. [Google Scholar] [CrossRef] [PubMed]
- Franz, M.J.; VanWormer, J.J.; Crain, A.L.; Boucher, J.L.; Histon, T.; Caplan, W.; Bowman, J.D.; Pronk, N.P. Weight-loss outcomes: A systematic review and meta-analysis of weight-loss clinical trials with a minimum 1-year follow-up. J. Am. Diet. Assoc. 2007, 107, 1755–1767. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.D.; Ryan, D.H.; Apovian, C.M.; Ard, J.D.; Comuzzie, A.G.; Donato, K.A.; Hu, F.B.; Hubbard, V.S.; Jakicic, J.M.; Kushner, R.F.; et al. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. Circulation 2014, 129, S102–S138. [Google Scholar] [CrossRef] [PubMed]
- Yumuk, V.; Tsigos, C.; Fried, M.; Schindler, K.; Busetto, L.; Micic, D.; Toplak, H. European guidelines for obesity management in adults. Obes. Facts 2015, 8, 402–424. [Google Scholar] [CrossRef] [PubMed]
- Harbin Consensus Conference Workshop Group. Improving the Reporting of Clinical Trials of Infertility Treatments (IMPRINT): Modifying the CONSORT statement. Fertil. Steril. 2014, 102, 952–959. [Google Scholar] [CrossRef] [Green Version]
| Study | Design | Country | PCOS Details at Baseline N (Completers) | Non-PCOS Details at Baseline N (Completers) | Attrition Rate | Current Medication | Specific Exclusion Criteria | Intervention | Outcomes | Risk of Bias |
|---|---|---|---|---|---|---|---|---|---|---|
| Diamanti-Kandarakis 2007 [30] | Comparative study | Greece | Age 27.5 ± 5.77 BMI 35.4 ± 5.31 ESHRE/ASRM n = 29 | Age 32.1 ± 5.64 BMI 36.4 ± 6.47 n = 18 | Not reported | Not reported | Galactorrhea | Duration: 24 weeks Normal protein diet, 600 kcal/day energy deficit, Orlistat (Xenical®, Roche) 120 mg × 3 times daily Aim of weight loss: not stated | Anthropo-metric: BMI Reproductive: T, SHBG Metabolic: FG, 2HRG, FI | Moderate |
| Moran 2007 [34] | Comparative study | Australia | Age 31.7 ± 6.2 BMI 35.7 ± 5.8 ESHRE/ASRM n = 15 | Age 37.1 ± 4.7 BMI 35.5 ± 5.1 n = 17 | PCOS 17% Non-PCOS 11% | No hormonal or insulin-sensitising drugs pre study | Pregnancy, breastfeeding | Duration: 8 weeks Energy restricted diet (two meals daily replaced with commercially available meal replacements) Aim of weight loss: Yes | Anthropo-metric: Weight, WC Reproductive: T, SHBG, FAI Metabolic: FI, lipids Others: IL-6, TNF-α | Moderate |
| Hutchison 2011 [31] Harrison 2012 [32] | Comparative study | Australia | BMI > 27 (37.4 ± 1.5) Age 20–40 (29.5 ± 1.4) NIH n = 13 | BMI > 27 (35.7 ± 1.3) Age 20-40 (35.0 ± 1.1) n = 8 | PCOS 35% Non-PCOS 43% | No hormonal or insulin-sensitising drugs pre study | Diabetes, adrenal disorders, recent weight change, regular physical activity, pregnancy, breastfeeding, smoking | Duration: 12 weeks supervised intensified exercise training 60 min three times weekly Aim of weight loss: not stated | Anthropo-metric: WC, Weight, BMI, VF, SCFAT Reproductive: T, SHBG, FAI Metabolic: FG, FI, lipids, BP | Moderate |
| Cheang 2016 [43] | Comparative study | USA | Age 26.9 ± 4.6 BMI 36.6 ± 5.1 Modified ESHRE/ASRM n = 16 | Age 27.5 ± 5.7 BMI 35.8 ± 4.8 n = 15 | PCOS 53% Non-PCOS 44% | No unstable medication use for 6 months for disorders such as hypertension or dyslipidemia | Weight loss attempts in 3 months pre study, diabetes, pulmonary, cardiac, renal, neurologic, hepatic, psychiatric, infectious, neoplastic, malignant disease, pregnancy Non-PCOS: history gestational diabetes, family history abnormal glucose tolerance, hypertension, dyslipidemia | Duration: 8 weeks Standardized hypocaloric diet (50% carbohydrate, 20% protein, 30% fat) with 500–1000 kcal/day deficit. No modification of physical activity or other weight loss methods. Aim of weight loss: Yes | Anthropometric: Weight, BMI Metabolic: FG, FI | Moderate |
| Kogure 2016 [44] | Comparative study | Brazil | Age 28.1 ± 5.4 BMI 28.4 ± 6.0 ESHRE/ASRM n = 45 | Age 29.6 ± 5.2 BMI 26.2 ± 5.7 n = 52 | PCOS 38% Non-PCOS 46% | No hormonal contraceptive us drugs pre or during study | Systemic diseases, smoking, pregnancy | Duration: 4 months Progressive resistance training (PRT) for 1 h/day three times per week. Aim of weight loss: not stated | Anthropometric: Weight, BMI, WC, TFFM, % body fat Reproductive: T, SHBG, FAI Metabolic: FG, FI, HOMA-IR | Moderate |
| Villa 1999 [40] | Comparative study | Italy | Age 26.3 ± 5 BMI 27.5 ± 6.8 NIH n = 22 | Age not reported BMI 27.4 ± 6.8 n = 14 | Not reported | Not reported | Not reported | Duration: 4–5 weeks 50 mg Naltrexone daily Aim of weight loss: not stated | Anthropo-metric: BMI Reproductive: T, SHBG, FAI Metabolic: FG, FI | High |
| Kowalska 2001 [33] | Comparative study | Poland | Age 25.3 ± 4.8 BMI 34.7 ± 6.0 NIH n = 11 | Age 27.9 ± 7.3 BMI 36.2 ± 6.0 n = 19 | PCOS 27% Non-PCOS 40% | Not reported | No additional | Duration: 4–5 months Hypocaloric diet (1200–1400 kcal/day), Metformin 500 mg three times daily Aim of weight loss: not stated | Anthropo-metric: BMI, WC Reproductive: T, FAI, SHBG Metabolic: FI | High |
| Panidis 2014 [35] Vosnakis 2013 [36] Panidis 2008 [37] | Comparative study | Greece | Age 26.1 ± 6.4 BMI 34.5 ± 5.9 ESHRE/ASRM n = 101 | Age 31.5 ± 4.7 BMI 34.9 ± 5.4 n = 29 | Not reported | No hormonal or insulin-sensitising drugs pre or during study | Galactorrea | Duration: 6 months Normal protein, energy-restricted diet (600 kcal/day energy deficit, moderate intensity aerobic exercise, 1 h × 3 times/week, Orlistat 120 mg before each meal) Aim of weight loss: Yes | Anthropo-metric: WC, BMI Reproductive: T, FAI, SHBG Metabolic: FG, lipids | High |
| Kahal 2015 [41] | Comparative study | UK | Age 33.9 ± 6.7 BMI 37.9 ± 5.0 ESHRE/ASRM n = 13 | Age 33.5 ± 7.1 BMI 36.5 ± 4.6 n = 12 | PCOS 32% Non-PCOS 29% | No medication | Alcohol intake >14 units/week Non-PCOS: history of hirsutism or menstrual irregularities | Duration: 6 months Liraglutide 0.6 mg o.d subcutaneous injection for 1 week, 1.2 mg o.d for 1 week, and 1.8 mg o.d thereafter for 6 months. No diet or exercise advice given. Aim of weight loss: Yes | Anthropometric: Weight, BMI | High |
| Nikokavora 2015 [42] | Comparative study | UK | Age 35.7 ± 8.9 BMI 40.0 ± 6.3 Diagnostic criteria not reported n = 137 | Age 35.8 ± 8.9 BMI 40.0 ± 6.3 n = 137 | PCOS 73% Non-PCOS 73% | Not reported | Type 1 diabetes, porphyria, lactose intolerance, major cardio- or cerebrovascular disease, history of renal or hepatic disease, cancer, epilepsy, major psychological or eating disorders, breastfeeding, pregnant, birth or miscarriage prior 3 months | Duration: 12 weeks Commercial weight management program (LighterLife Total), 600 kcal (36% protein, 36% carbohydrate, 28% fat) as food packs alongside behavior change program Aim of weight loss: Yes | Anthropometric: Weight, BMI | High |
| Bhandari 2016 [45] | Comparative study | India | Age 27.8 ± 4.50 BMI 42.5 ± 5.71 ESHRE/ASRM n = 43 | Age 29.3 ± 4.96 BMI 45.0 ± 6.11 n = 32 | Not reported | No hormonal, fertility or insulin-sensitising drugs pre or during study | Systemic diseases like hypothyroidism or hyperprolactinaemia, surgical complications intra or post operatively | Duration: 6 months post-surgery Sleeve gastrectomy (bariatric surgery) Aim of weight loss: Yes | Anthropometric: Weight, BMI Other: Abnormal menstrual cycles | High |
| Al-Eisa 2017 [46] | Comparative study | Egypt | Age 27.9 ± 4.1 BMI 33.5 ± 2.75 ESHRE/ASRM n = 30 | Age 27.6 ± 5.7 BMI 31.7 ± 3.8 n = 30 | Not reported | No hormonal drugs pre or during study | Normal BMI, other diseases such as diabetes or viral infections | Duration: 12 weeks treadmill walking, 45 min three times per week for 12 weeks. Aim of weight loss: Yes | Anthropometric: Weight, BMI, WC Metabolic: FG, FI | High |
| Pasquali 2000 [38] | RCT | Italy | BMI > 28 WHR > 0.80 NIH n = 18 | BMI > 28 WHR > 0.80 n = 17 | Diet: PCOS 0% Non-PCOS 25% Metform-in: PCOS 17% Non-PCOS 0% | No hormonal or insulin-sensitising drugs pre study | Diabetes, renal or liver dysfunction | Duration: 6 months One month hypocaloric diet (1200–1400 kcal daily), Metformin 850 mg twice daily. Aim of weight loss: Yes | Anthropo-metric: Weight, BMI, SAT, VAT Reproductive: T, SHBG Metabolic: FG, FI | Moderate |
| Toscani 2011 [39] | RCT | Brazil | Age 22.7 + 5.68 Most participants BMI ≥ 25 NIH n = 18 | Age 29.4 + 5.74 Most participants BMI ≥ 25 n = 22 | Not reported | No hormonal drugs pre study | Diabetes, renal dysfunction | Duration: 2 months Diet 1: HP (30% protein, 40% carbohydrate, 30% fat) Diet 2: NP (15% protein, 55% carbohydrate, 30% fat) Aim of weight loss: Yes | Anthropo-metric: Weight, WC, BMI Reproductive: T, SHBG, FAI Metabolic: BP, FG, 2HRG, FI, 2HRI, lipids | High |
| Outcome | Reference | Intervention | Baseline PCOS: Mean ± SD | Baseline Non-PCOS: Mean ± SD | p-Value * | Post-Intervention PCOS: Mean ± SD | Post-Intervention Non-PCOS: Mean ± SD | p-Value * |
|---|---|---|---|---|---|---|---|---|
| Weight (kg) | Pasquali 2000 [38] | Hypocaloric diet | 102 ± 19 | 106 ± 13 | NR | 97 ± 18 | 100 ± 13 | NR |
| Cheang 2016 [43] | Hypocaloric diet | 99.2 ± 13.3 | 97.6 ± 15.4 | 0.7508 | −4.08 ± 3.65 | −4.69 ± 2.98 | 0.6281 | |
| Moran 2007 [34] | Hypocaloric diet | 95.1 ± 19.3 | 95.5 ± 16.5 | NS | −3.9 ± 3.6 | −4.5 ± 4.1 | 0.642 | |
| Toscani 2011 [39] | High protein diet | 74.62 ± 18.8 | 75.89 ± 13.49 | NR | 71.4 ± 15.45 | 74.54 ± 13.71 | NR | |
| Toscani 2011 [39] | Normal protein diet | 82.85 ± 15.18 | 77.51 ± 13.31 | NR | 79.82 ± 16.51 | 74.31 ± 13.88 | NR | |
| Nikokavoura 2015 [42] | VLCD + behaviour change | 108.3 ± 18.1 | 107.4 ± 19.8 | 0.713 | 89.8 ± 16.7 | 88.0 ± 17.6 | 0.19 | |
| Pasquali 2000 [38] | Hypocaloric diet + Metformin | 103 ± 18 | 101 ± 8 | NR | 94 ± 17 | 88 ± 7 | NR | |
| Kahal 2015 [41] | AO drug (Liraglutide) | 102.1 ± 17.1 | 100.4 ± 15.1 | NS | −3.0 ± 4.2 | −3.8 ± 3.4 | 0.56 | |
| Bhandari 2016 [45] | Bariatric surgery | 106.89 ± 17.79 | 117.03 ± 19.89 | NR | 77.27 ± 10.72 | 84.89 ± 13.18 | NR | |
| Hutchison 2011 [31] | Intensified exercise training | 100.5 ± 4.5 ! | 96.2 ± 3.5 ! | 0.42 | 95.3 ± 4.8 ! | 96.9 ± 4.5 ! | NR | |
| Kogure 2016 [44] | Progressive resistance training | 73.1 ± 15.6 | 68.1 ± 15.4 | NS | 0.52 [−0.31, 1.36] ^ | 0.13 [−0.64, 0.90] ^ | 0.14 | |
| Al−Eisa 2017 [46] | Aerobic training | 89.8 ± 6.95 | 84.9 ± 7.2 | NR | 84.8 ± 6.42 | 82.2 ± 5.72 | NR | |
| BMI (kg/m2) | Pasquali 2000 [38] | Hypocaloric diet | 39.6 ± 6.9 | 40.1 ± 6.2 | NR | 38 ± 6.2 | 37.8 ± 5.7 | NR |
| Cheang 2016 [43] | Hypocaloric diet | 36.6 ± 5.1 | 35.8 ± 4.8 | 0.6507 | −1.46 [−0.72, −2.20] # | −1.80 [−1.14, −2.45] # | 0.4829 | |
| Nikokavoura 2015 [42] | VLCD + behaviour change | 40.0 ± 6.3 | 40.0 ± 6.3 | 0.955 | 33.2 ± 6.0 | 32.8 ± 5.7 | NR | |
| Kowalska 2001 [33] | Hypocaloric diet + Metformin | 34.7 ± 6.0 | 36.2 ± 6.0 | NS | 31.4 ± 4.8 | 35.8 ± 7.9 | NR | |
| Pasquali 2000 [38] | Hypocaloric diet + Metformin | 39.8 ± 7.9 | 37.4 ± 3.0 | NR | 36.4 ± 7.4 | 32.9 ± 3.4 | NR | |
| Diamanti−Kandarakis 2007 [30] | Hypocaloric diet + AO drug (Orlistat) | 35·43 ± 5·31 | 36·39 ± 6·47 | 0.58 | 29.7 ± 4.57 | 30.15 ± 4.13 | NR | |
| Vosnakis 2013 [36] | Hypocaloric diet + AO drug (Orlistat) + moderate intensity aerobic training | 34.83 ± 6.39 | 36.79 ± 6.98 | NR | 30.21 ± 5.78 | 31.01 ± 4.93 | NR | |
| Kahal 2015 [41] | AO drug (Liraglutide) | 37.9 ± 5.0 | 36.5 ± 4.6 | NS | −1.0 ± 1.5 | −1.4 ± 1.2 | 0.43 | |
| Villa 1999 [40] | AO drug (Naltrexone) | 27.5 ± 6.8 | 27.4 ± 6.8 | NS | 26.8 ± 6.7 | 27 ± 6.8 | NR | |
| Bhandari 2016 [45] | Bariatric surgery | 42.52 ± 5.66 | 45.03 ± 6.3 | 0.0717 | 30.76 ± 2.93 | 32.67 ± 3.51 | 0.013 | |
| Hutchison 2001 [31] | Intensified exercise training | 37.4 ± 1.5 ! | 35.7 ± 1.3 ! | 0.43 | 35 ± 1.6 ! | 35.9 ± 1.8 ! | NR | |
| Kogure 2016 [44] | Progressive resistance training | 28.4 ± 6.0 | 26.2 ± 5.7 | NS | 0.21 [−0.11, 0.54] ^ | 0.05 [−0.25, 0.35] ^ | 0.08 | |
| Al−Eisa 2017 [46] | Aerobic training | 33.45 ± 2.75 | 31.7 ± 3.8 | NR | 28.5 ± 2.25 | 26.8 ± 2.54 | NR | |
| WC (cm) | Pasquali 2000 [38] | Hypocaloric diet | 109 ± 19 | 109 ± 11 | NR | 104 ± 13 | 105 ± 12 | NR |
| Toscani 2011 [39] | High protein diet | 87.74 ± 14.08 | 83.92 ± 9.13 | <0.05 | 86 ± 12.92 | 81.83 ± 9.13 | NR | |
| Toscani 2011 [39] | Normal protein diet | 93.32 ± 8.05 | 84.02 ± 9.03 | <0.05 | 90.03 ± 10.41 | 81.7 ± 11.72 | NR | |
| Kowalska 2001 [33] | Hypocaloric diet + Metformin | 98.1 ± 14.8 | 102.3 ± 13.0 | NS | 93.4 ± 11.8 | 100 ± 19.5 | NR | |
| Pasquali 2000 [38] | Hypocaloric diet + Metformin | 107 ± 16 | 102 ± 6 | NR | 100 ± 15 | 94 ± 6 | NR | |
| Panidis 2008 [37] | Hypocaloric diet + AO drug (Orlistat) + moderate intensity aerobic training | 101.52 ± 2.67 | 100.53 ± 3.94 | NS | 87.86 ± 2.29 | 87.67 ± 2.82 | NR | |
| Hutchison 2011 [31] | Intensified exercise training | 106.8 ± 3.4 ! | 102.8 ± 2.6 ! | 0.39 | 103.1 ± 4 ! | 99.9 ± 4.1 ! | NR | |
| Kogure 2016 [44] | Progressive resistance training | 81.7 ± 12.8 | 76.2 ± 11.3 | <0.05 | 0.86 [0.32, 1.40] ^ | 0.27 [−0.21, 0.75] | 0.21 | |
| Al−Eisa 2017 [46] | Aerobic training | 96.2 ± 3.52 | 94.2 ± 3.82 | NR | 93.8 ± 3.26 | 72.7 ± 2.6 | NR | |
| VAT/VF (cm2) | Pasquali 2000 [38] | Hypocaloric diet | 121 ± 48 | 181 ± 94 | NR | 108 ± 36 | 159 ± 83 | NR |
| Pasquali 2000 [38] | Hypocaloric diet + Metformin | 151 ± 91 | 133 ± 38 | NR | 113 ± 59 | 100 ± 37 | NR | |
| Hutchison 2011 [31] | Intensified exercise training | 129.2 ± 12.8 ! | 121.5 ± 9.4 ! | 0.65 | 107.6 ± 15.1 ! | 132.7 ± 18.1 ! | NR | |
| SAT/SCFAT (cm2) | Pasquali 2000 [38] | Hypocaloric diet | 589 ± 127 | 554 ± 118 | NR | 574 ± 111 | 508 ± 107 | NR |
| Pasquali 2000 [38] | Hypocaloric diet + Metformin | 535 ± 147 | 554 ± 79 | NR | 485 ± 170 | 462 ± 81 | NR | |
| Hutchison 2011 [31] | Intensified exercise training | 590.2 ± 35.2 ! | 550.3 ± 45.2 ! | 0.49 | 538.4 ± 40.2 ! | 558.5 ± 74.5 ! | NR |
| Outcome | References | Intervention | Baseline PCOS: Mean ± SD | Baseline Non-PCOS: Mean ± SD | p-Value * | Post-intervention PCOS: Mean ± SD | Post-intervention Non-PCOS: Mean ± SD | p-Value * |
|---|---|---|---|---|---|---|---|---|
| Number of ovulations | Moran 2007 [34] | Hypocaloric diet | NR | NR | NR | 1.9 ovulations | 1.0 ovulations | <0.001 |
| Total testosterone (nmol/L) | Pasquali 2000 [38] | Hypocaloric diet | 1.77 ± 0.59 | 1.32 ± 0.42 | <0.05 | 1.63 ± 0.45 | 1.14 ± 0.35 | NR |
| Kowalska 2001 [33] | Hypocaloric diet + Metformin | 3.57 ± 1.01 | 1.91 ± 0.42 | <0.05 | 2.39 ± 1.11 | 1.7 ± 0.59 | NR | |
| Pasquali 2000 [38] | Hypocaloric diet + Metformin | 2.36 ± 1.21 | 1.46 ± 0.38 | <0.01 | 1.7 ± 0.87 | 1.25 ± 0.38 | NR | |
| Diamanti-Kandarakis 2007 [30] | Hypocaloric diet + AO drug (Orlistat) | 3.01 ± 0.94 | 1.50 ± 0.43 | <0.001 | 2.28 ± 0.65 | 1.49 ± 0.36 | <0.05 | |
| Panidis 2014 [35] | Hypocaloric diet + AO drug (Orlistat) + moderate intensity aerobic training | 2.56 ± 1.00 | 1.33 ± 0.45 | <0.001 | 2.1 ± 0.78 | 1.41 ± 0.74 | 0.006 | |
| Villa 1999 [40] | AO drug (Naltrexone) | 1.7 ± 0.5 | 1.4 ± 0.5 | NS | 1.9 ± 1.76 | 1.5 ± 1.2 | NR | |
| Hutchison 2011 [31] | Intensified exercise training | 2.9 ± 0.2! | 1.6 ± 0.2 ! | <0.01 | 2.8 ± 0.3 ! | 1.8 ± 0.3 ! | NR | |
| Kogure 2016 [44] | Progressive resistance training | 3.12 ± 1.22 | 2.58 ± 1.02 | <0.05 | 0.59 [0.30, 0.89] ^ | 0.42 [0.16, 0.68] ^ | 0.15 # | |
| SHBG (nmol/L) | Pasquali 2000 [38] | Hypocaloric diet | 16.0 ± 7.04 | 20.2 ± 10.7 | NS | 13.8 ± 2.1 | 28.1 ± 14.7 | NR |
| Kowalska 2001 [33] | Hypocaloric diet + Metformin | 32.0 ± 18.3 | 32.5 ± 16.5 | NS | 38.6 ± 19.3 | 36.5 ± 13.3 | NR | |
| Pasquali 2000 [38] | Hypocaloric diet + Metformin | 18.7 ± 15.0 | 23.4 ± 22.7 | NS | 16.7 ± 8.1 | 28.9 ± 16.5 | NR | |
| Diamanti-Kandarakis 2007 [30] | Hypocaloric diet + AO drug (Orlistat) | 28.72 ± 12.48 | 40.92 ± 19.54 | 0.01 | 37.21 ± 17.59 | 58.6 ± 27.02 | <0.05 | |
| Panidis 2014 [35] | Hypocaloric diet + AO drug (Orlistat) + moderate intensity aerobic training | 30.3 ± 13.2 | 47.8 ± 34.7 | 0.012 | 40.3 ± 20.4 | 62.2 ± 35.5 | NS | |
| Villa 1999 [40] | AO drug (Naltrexone) | 30.2 ± 20.4 | 38.2 ± 16.4 | NS | 32.5 ± 20.9 | 39.2 ± 15.7 | NR | |
| Hutchison 2011 [31] | Intensified exercise training | 29.0 ± 1.8! | 43.6 ± 7.8 ! | 0.04 | 30.7 ± 2.8 ! | 54.3 ± 10.6 ! | NR | |
| Kogure 2016 [44] | Progressive resistance training | 54.9 ± 37.8 | 63.0 ± 35.7 | NS | 0.12 [0.02, 0.23] ^ | 0.09 [−0.01, 0.18] ^ | 0.37 # | |
| FAI | Kowalska 2001 [33] | Hypocaloric diet + Metformin | 14.49 ± 8.49 | 9.97 ± 7.95 | <0.05 | 9.31 ± 9.95 | 5.49 ± 3.9 | NR |
| Panidis 2014 [35] | Hypocaloric diet + AO drug (Orlistat) + moderate intensity aerobic training | 10.25 ± 6.31 | 3_71 ± 2.11 | <0.001 | 6.76 ± 4.32 | 2.71 ± 1.74 | 0.021 | |
| Villa 1999 [40] | AO drug (Naltrexone) | 9.3 ± 6.5 | 4.9 ± 3.1 | <0.05 | 8.9 ± 5.7 | 5.2 ± 3.3 | NR | |
| Hutchison 2011 [31] | Intensified exercise training | 10.7 ± 1.1 ! | 4.6 ± 0.9 ! | <0.01 | 10.1 ± 1.6 ! | 4.1 ± 1.1 ! | NR | |
| Kogure 2016 [44] | Progressive resistance training | 8.3 ± 6.3 | 5.6 ± 4.6 | <0.05 | 0.98 [−0.03, 1.99] ^ | 0.37 [−0.50, 1.24] ^ | 0.25 # |
| Outcome | References | Intervention | Baseline PCOS: Mean ± SD | Baseline Non-PCOS: Mean ± SD | p-Value * | Post-Intervention PCOS: Mean ± SD | Post-Intervention Non-PCOS: Mean ± SD | p-Value * |
|---|---|---|---|---|---|---|---|---|
| Fasting glucose (mmol/L) | Pasquali 2000 [38] | Hypocaloric diet | 5.61 ± 1.0 | 5.11 ± 0.56 | NS | 5.27 ± 0.61 | 5.16 ± 0.94 | NR |
| Cheang 2016 [43] | Hypocaloric diet | 4.75 ± 0.45 | 4.67 ± 0.21 | 0.60 | −0.06 (−0.25, 0.12) % | 0.01 (−0.13, 0.15) % | 0.5041 | |
| Toscani 2011 [39] | High protein diet | 4.97 ± 0.34 | 4.95 ± 0.45 | NS | 5.02 ± 0.40 | 5.05 ± 0.54 | NR | |
| Toscani 2011 [39] | Normal protein diet | 4.90 ± 0.36 | 4.98 ± 0.41 | NS | 4.98 ± 0.35 | 5.02 ± 0.35 | NR | |
| Pasquali 2000 [38] | Hypocaloric diet + Metformin | 5.49 ± 1.61 | 4.94 ± 0.56 | NS | 5 ± 0.94 | 4.94 ± 0.72 | NR | |
| Diamanti-Kandarakis 2007 [30] | Hypocaloric diet + AO drug (Orlistat) | 5.72 ± 0.51 | 5.77 ± 0.61 | 0.92 | 5.52 ± 0.47 | 5.69 ± 0.52 | NR | |
| Panidis 2014 [35] | Hypocaloric diet + AO drug (Orlistat) + moderate intensity aerobic training | 5.58 ± 0.57 | 5.81 ± 0.66 | NS | 5.28 ± 0.56 | 5.58 ± 0.51 | NS | |
| Villa 1999 [40] | AO drug (Naltrexone) | 4.65 ± 0.33 | 4.55 ± 0.22 | NS | 4.33 ± 0.56 | 4.48 ± 0.37 | NR | |
| Hutchison 2011 [31] | Intensified exercise training | 5.0 ± 0.1 ! | 4.8 ± 0.1 ! | 0.57 | 4.9 ± 0.1 ! | 4.9 ± 0.1 ! | NR | |
| Kogure 2016 [44] | Progressive resistance training | 5.34 ± 0.91 | 5.31 ± 0.97 | NS | 0.37 [0.08, 0.66] $ | 0.30 [0.04, 0.56] $ | 0.12 ** | |
| Al-Eisa 2017 [46] | Aerobic training | 4.50 ± 2.80 | 5.59 ± 1.57 | NR | 4.50 ± 2.80 | 5.59 ± 1.57 | NR | |
| OGTT-glucose (mmol/L) | Toscani 2011 [39] | High protein diet | 6.27 ± 1.60 | 5.02 ± 0.99 | NR | 6.92 ± 2.0 | 5.63 ± 1.56 | NR |
| Toscani 2011 [39] | Normal protein diet | 6.55 ± 1.50 | 5.41 ± 1.02 | NR | 6.64 ± 2.16 | 5.18 ± 1.02 | NR | |
| Diamanti-Kandarakis 2007 [30] | Hypocaloric diet + AO drug (Orlistat) | 6.35 ± 1.35 | 6.54 ± 1.54 | 0.65 | 6.17 ± 0.85 | 6.21 ± 1.77 | NR | |
| Fasting insulin (pmol/L) | Pasquali 2000 [38] | Hypocaloric diet | 240.36 ± 214.53 | 149.24 ± 79.64 | NS | 136.33 ± 103.32 | 103.32 ± 76.06 | NR |
| Toscani 2011 [39] | High protein diet | 76.05 (60.00–123.41) ^ | 57.85 (28.20–84.94) ^ | <0.05 | 61.67 (52.71–108.83) ^ | 56.74 (33.20–97.92) ^ | NR | |
| Toscani 2011 [39] | Normal protein diet | 128.83 (93.06–213.28) ^ | 59.59 (41.04–84.52) ^ | <0.05 | 129.15 (76.99–233.04) ^ | 48.00 (31.14–80.14) ^ | NR | |
| Pasquali 2000 [38] | Hypocaloric diet + Metformin | 308.53 ± 218.12 | 217.40 ± 58.84 | NS | 154.98 ± 223.86 | 102.6 ± 60.99 | NR | |
| Kowalska 2001 [33] | Hypocaloric diet + Metformin | 26.2 ± 13.9 # | 18.3 ± 14.2 # | <0.05 | 16.9 ± 9.1 # | 20.0 ± 13.5 # | NR | |
| Diamanti-Kandarakis 2007 [30] | Hypocaloric diet + AO drug (Orlistat) | 127.37 ± 61.12 | 125.5 ± 87.09 | 0.45 | 76.4 ± 34.93 | 77.02 ± 47.3 | NR | |
| Panidis 2014 [35] | Hypocaloric diet + AO drug (Orlistat) + moderate intensity aerobic training | 129.87 ± 75.01 | 133.34 ± 161.82 | NS | 81.08 ± 50.94 | 68.16 ± 40.18 | NS | |
| Villa 1999 [40] | AO drug (Naltrexone) | 99.7 ± 75 | 104.3 ± 63.1 | NS | 90.4 ± 61 | 85.3 ± 39.4 | NR | |
| Hutchison 2011 [31] | Intensified exercise training | 141.6 (100.8–181.2) ^ | 72.6 (58.8–115.8) ^ | 0.02 | 97.8 (66.6–231.0) ^ | 115.2 (76.2–177.6) ^ | NR | |
| Kogure 2016 [44] | Progressive resistance training | 64.59 ± 47.92 | 36.11 ± 31.25 | <0.05 | 0.90 [−0.21, 1.94] $ | −0.90 [−1.88, 0.07] $ | 0.58 ** | |
| Al-Eisa 2017 [46] | Aerobic training | 17.8 ± 4.20 @ | 20.6 ± 8.2 @ | NR | 14.8 ± 2.9 @ | 16.1 ± 5.1 @ | NR |
| Outcome | References | Intervention | Baseline PCOS: Mean ± SD | Baseline Non-PCOS: Mean ± SD | p-Value * | Post-intervention PCOS: Mean ± SD | Post-intervention Non-PCOS: Mean ± SD | p-Value * |
|---|---|---|---|---|---|---|---|---|
| Total cholesterol (mmol/L) | Toscani 2011 [39] | High protein diet | 4.60 ± 1.15 | 4.24 ± 0.71 | NR | 4.32 ± 1.12 | 4.19 ± 0.82 | NR |
| Toscani 2011 [39] | Normal protein diet | 4.31 ± 1.04 | 4.05 ± 1.13 | NR | 4.02 ± 0.82 | 3.86 ± 1.36 | NR | |
| Panidis 2014 [35] | Hypocaloric diet + AO drug (Orlistat) + moderate intensity aerobic training | 5.04 ± 1.00 | 5.48 ± 0.93 | NS | 4.52 ± 0.82 | 4.55 ± 0.87 | <0.001 | |
| Hutchison 2011 [31] | Intensified exercise training | 5.0 ± 0.3 ! | 4.7 ± 0.2 ! | 0.81 | 4.4 ± 0.2 ! | 4.8 ± 0.4 ! | NR | |
| LDL cholesterol (mmol/L) | Toscani 2011 [39] | High protein diet | 3.91 ± 1.09 | 3.66 ± 0.61 | NR | 3.78 ± 1.1 | 3.54 ± 0.76 | NR |
| Toscani 2011 [39] | Normal protein diet | 3.62 ± 0.95 | 3.30 ± 1.11 | NR | 3.33 ± 0.75 | 3.13 ± 1.34 | NR | |
| Panidis 2014 [35] | Hypocaloric diet + AO drug (Orlistat) + moderate intensity aerobic training | 3.30 ± 0.90 | 3.65 ± 0.68 | NS | 2.87 ± 0.84 | 2.98 ± 0.47 | 0.001 | |
| Hutchison 2011 [31] | Intensified exercise training | 3.3 ± 0.2 ! | 3.0 ± 0.2 ! | 0.48 | 3 ± 0.2 ! | 3.1 ± 0.4 ! | NR | |
| HDL cholesterol (mmol/L) | Toscani 2011 [39] | High protein diet | 1.30 ± 0.19 | 1.35 ± 0.38 | NR | 1.27 ± 0.2 | 1.34 ± 0.39 | NR |
| Toscani 2011 [39] | Normal protein diet | 1.19 ± 0.32 | 1.39 ± 0.26 | NR | 1.18 ± 0.32 | 1.5 ± 0.25 | NR | |
| Panidis 2014 [35] | Hypocaloric diet + AO drug (Orlistat) + moderate intensity aerobic exercise | 1.20 ± 0.22 | 1.32 ± 0.31 | NS | 1.21 ± 0.23 | 1.21 ± 0.26 | 0.006 | |
| Hutchison 2011 [31] | Intensified exercise training | 1.0 ± 0.1 ! | 1.2 ± 0.1 ! | 0.04 | 1 ± 0.1 ! | 1.2 ± 0.1 ! | NR | |
| Triglycerides (mmol/L) | Toscani 2011 [39] | High protein diet | 0.86 (0.47–1.47) ^ | 0.68 (0.47–0.76) ^ | NR | 0.55 (0.49–0.71) ^ | 0.92 (0.55–1.02) ^ | NR |
| Toscani 2011 [39] | Normal protein diet | 0.97 (0.67–1.30) ^ | 0.71 (0.41–1.62) ^ | NR | 0.94 (0.81–1.15) ^ | 0.88 (0.64–1.21) ^ | NR | |
| Panidis 2014 [35] | Hypocaloric diet + AO drug (Orlistat) + moderate intensity aerobic training | 1.17 ± 0.56 | 1.28 ± 0.62 | NS | 0.99 ± 0.44 | 0.87 ± 0.25 | NS | |
| Hutchison 2011 [31] | Intensified exercise training | 1.4 ± 0.2 ! | 1.2 ± 0.2 ! | 0.46 | 0.9 ± 0.1 ! | 1.3 ± 0.1 ! | NR | |
| BP systolic (mmHg) | Toscani 2011 [39] | High protein diet | 125.7 ± 19.0 | 116.1 ± 10.41 | NR | 126 ± 23.1 | 117.85 ± 10.18 | NR |
| Toscani 2011 [39] | Normal protein diet | 119.1 ± 16.4 | 116.43 ± 10.3 | NR | 119.36 ± 15.38 | 110.71 ± 7.32 | NR | |
| Harrison 2012 [32] | Intensified exercise training | 108 ± 14.6 ! | 118 ± 16.7 ! | NS | 109 ± 10.4 ! | 116 ± 16.2 ! | NR | |
| BP diastolic (mmHg) | Toscani 2011 [39] | High protein diet | 77.9 ± 10.75 | 74.6 ± 8.46 | NR | 80 ± 11.2 | 74 ± 8.83 | NR |
| Toscani 2011 [39] | Normal protein diet | 78 ± 11.83 | 75.14 ± 9.6 | NR | 77.82 ± 12.02 | 72.57 ± 7.72 | NR | |
| Harrison 2012 [32] | Intensified exercise training | 72 ± 10.2 ! | 75 ± 8.8 ! | NS | 69 ± 7.4 ! | 73 ± 10.5 ! | NR |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kataoka, J.; Tassone, E.C.; Misso, M.; Joham, A.E.; Stener-Victorin, E.; Teede, H.; Moran, L.J. Weight Management Interventions in Women with and without PCOS: A Systematic Review. Nutrients 2017, 9, 996. https://doi.org/10.3390/nu9090996
Kataoka J, Tassone EC, Misso M, Joham AE, Stener-Victorin E, Teede H, Moran LJ. Weight Management Interventions in Women with and without PCOS: A Systematic Review. Nutrients. 2017; 9(9):996. https://doi.org/10.3390/nu9090996
Chicago/Turabian StyleKataoka, Josefin, Eliza C. Tassone, Marie Misso, Anju E. Joham, Elisabet Stener-Victorin, Helena Teede, and Lisa J. Moran. 2017. "Weight Management Interventions in Women with and without PCOS: A Systematic Review" Nutrients 9, no. 9: 996. https://doi.org/10.3390/nu9090996






