The Effect of Low Carbohydrate Diets on Fertility Hormones and Outcomes in Overweight and Obese Women: A Systematic Review
Abstract
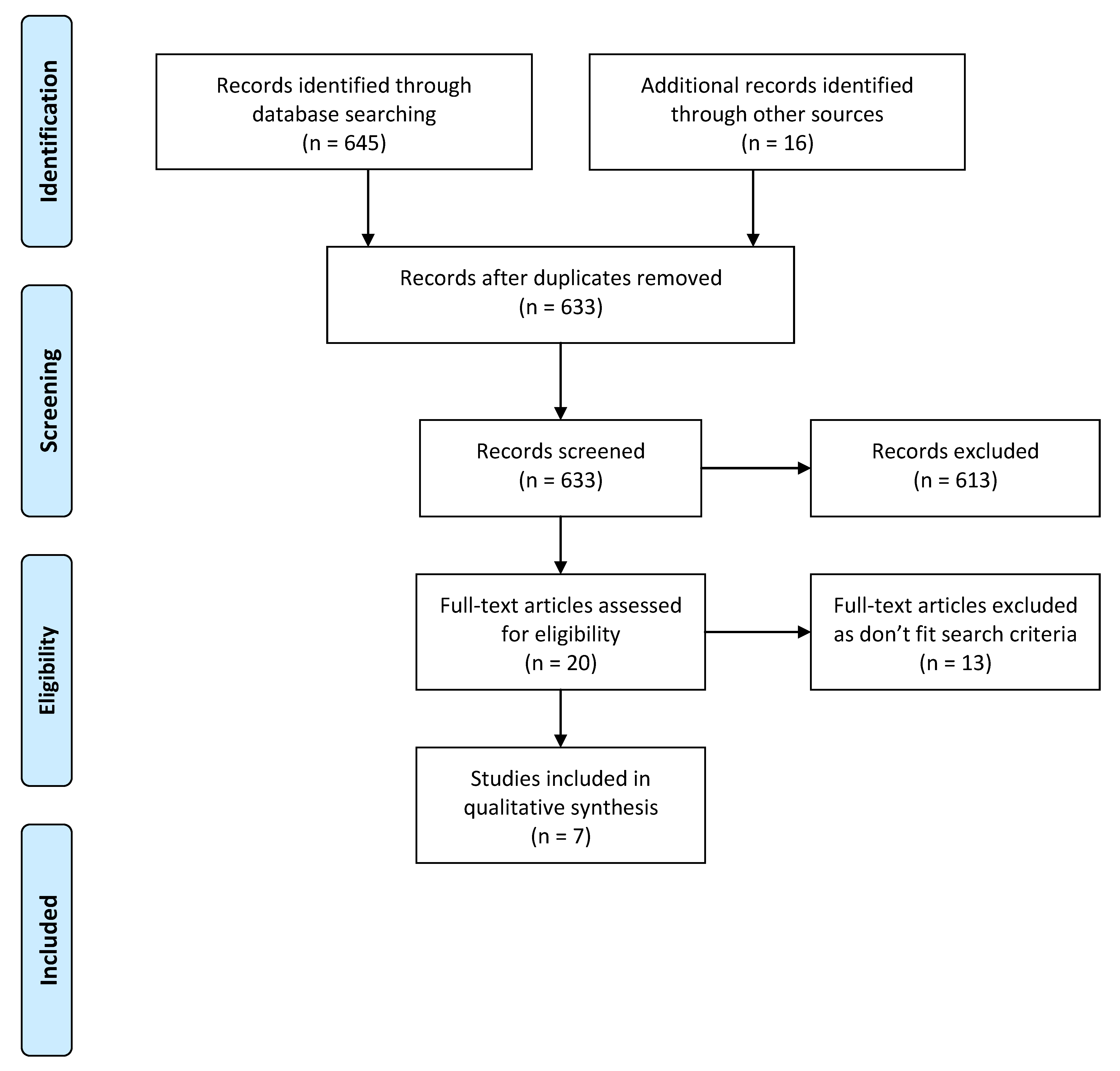
:1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Search Strategy
2.3. Study Selection
2.4. Data Extraction and Quality Assessment
2.5. Analysis
3. Results
3.1. Study Quality
3.2. Outcomes
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kulak, D.; Polotsky, A.J. Should the ketogenic diet be considered for enhancing fertility? Maturitas 2013, 74, 10–13. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Noakes, M.; Norman, R.J. Dietary effects on fertility treatment and pregnancy outcomes. Curr. Opin. Endocrinol. Diabetes Obes. 2007, 14, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Sim, K.A.; Dezarnaulds, G.M.; Denyer, G.S.; Skilton, M.R.; Caterson, I.D. Weight loss improves reproductive outcomes in obese women undergoing fertility treatment: A randomized controlled trial. Clin. Obes. 2014, 4, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Sainsbury, A.; Schwarzer, C.; Couzens, M.; Jenkins, A.; Oakes, S.R.; Ormandy, C.J.; Herzog, H. Y4 receptor knockout rescues fertility in ob/ob mice. Genes Dev. 2002, 16, 1077–1088. [Google Scholar] [CrossRef] [PubMed]
- Sim, K.A.; Partridge, S.R.; Sainsbury, A. Does weight loss in overweight or obese women improve fertility treatment outcomes? A systematic review. Obes. Rev. 2014, 15, 839–850. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Obesity: Preventing and Managing the Global Epidemic. Report of a WHO Consultation. Available online: http://apps.who.int/iris/handle/10665/42330 (accessed on 2 September 2016).
- Nikokavoura, E.A.; Johnston, K.L.; Broom, J.; Wrieden, W.L.; Rolland, C. Weight loss for women with and without polycystic ovary syndrome following a very low-calorie diet in a community-based setting with trained facilitators for 12 weeks. Diabetes Metab. Syndr. Obes. Targets Ther. 2015, 8, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Moragianni, V.A.; Jones, S.M.; Ryley, D.A. The effect of body mass index on the outcomes of first assisted reproductive technology cycles. Fertil. Steril. 2012, 98, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Chavarro, J.E.; Ehrlich, S.; Colaci, D.S.; Wright, D.L.; Toth, T.L.; Petrozza, J.C.; Hauser, R. Body mass index and short-term weight change in relation to treatment outcomes in women undergoing assisted reproduction. Fertil. Steril. 2012, 98, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Musella, M.; Milone, M.; Bellini, M.; Sosa Fernandez, L.M.; Leongito, M.; Milone, F. Effect of bariatric surgery on obesity-related infertility. Surg. Obes. Relat. Dis. 2012, 8, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; Mills, K.T.; Yao, L.; Demanelis, K.; Eloustaz, M.; Yancy, W.S., Jr.; Kelly, T.N.; He, J.; Bazzano, L.A. Effects of low-carbohydrate diets versus low-fat diets on metabolic risk factors: A meta-analysis of randomized controlled clinical trials. Am. J. Epidemiol. 2012, 176, S44–S54. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Psaltopoulou, T.; Orfanos, P.; Hsieh, C.C.; Trichopoulos, D. Low-carbohydrate-high-protein diet and long-term survival in a general population cohort. Eur. J. Clin. Nutr. 2007, 61, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Crowe, T.C. Safety of low-carbohydrate diets. Obes. Rev. 2005, 6, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, T.L.; Sutherland, J.P.; Wolfe, P.; Allian-Sauer, M.; Capell, W.H.; Talley, N.D.; Wyatt, H.R.; Foster, G.D.; Hill, J.O.; Eckel, R.H. Lack of suppression of circulating free fatty acids and hypercholesterolemia during weight loss on a high-fat, low-carbohydrate diet. Am. J. Clin. Nutr. 2010, 91, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The prisma statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The prisma statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef] [PubMed]
- Academy of Nutrition and Dietetics. Evidence Analysis Manual: Steps in the Academy Evidence Analysis Process. Available online: https://www.andeal.org/files/Docs/2012_Jan_EA_Manual.pdf (accessed on 2 September 2016).
- Mavropoulos, J.C.; Yancy, W.S.; Hepburn, J.; Westman, E.C. The effects of a low-carbohydrate, ketogenic diet on the polycystic ovary syndrome: A pilot study. Nutr. Metab. 2005, 2, 35. [Google Scholar] [CrossRef] [PubMed]
- Palomba, S.; Giallauria, F.; Falbo, A.; Russo, T.; Oppedisano, R.; Tolino, A.; Colao, A.; Vigorito, C.; Zullo, F.; Orio, F. Structured exercise training programme versus hypocaloric hyperproteic diet in obese polycystic ovary syndrome patients with anovulatory infertility: A 24-week pilot study. Hum. Reprod. 2008, 23, 642–650. [Google Scholar] [CrossRef] [PubMed]
- Moran, L.J.; Noakes, M.; Clifton, P.M.; Tomlinson, L.; Galletly, C.; Norman, R.J. Dietary composition in restoring reproductive and metabolic physiology in overweight women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2003, 88, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Stamets, K.; Taylor, D.S.; Kunselman, A.; Demers, L.M.; Pelkman, C.L.; Legro, R.S. A randomized trial of the effects of two types of short-term hypocaloric diets on weight loss in women with polycystic ovary syndrome. Fertil. Steril. 2004, 81, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Palomba, S.; Falbo, A.; Giallauria, F.; Russo, T.; Rocca, M.; Tolino, A.; Zullo, F.; Orio, F. Six weeks of structured exercise training and hypocaloric diet increases the probability of ovulation after clomiphene citrate in overweight and obese patients with polycystic ovary syndrome: A randomized controlled trial. Hum. Reprod. 2010, 25, 2783–2791. [Google Scholar] [CrossRef] [PubMed]
- Thomson, R.L.; Buckley, J.D.; Noakes, M.; Clifton, P.M.; Norman, R.J.; Brinkworth, G.D. The effect of a hypocaloric diet with and without exercise training on body composition, cardiometabolic risk profile, and reproductive function in overweight and obese women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2008, 93, 3373–3380. [Google Scholar] [CrossRef] [PubMed]
- Clifton, P.M.; Condo, D.; Keogh, J.B. Long term weight maintenance after advice to consume low carbohydrate, higher protein diets—A systematic review and meta analysis. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 224–235. [Google Scholar] [CrossRef] [PubMed]
- Hession, M.; Rolland, C.; Kulkarni, U.; Wise, A.; Broom, J. Systematic review of randomized controlled trials of low-carbohydrate vs. Low-fat/low-calorie diets in the management of obesity and its comorbidities. Obes. Rev. 2009, 10, 36–50. [Google Scholar] [CrossRef] [PubMed]
- Franks, S. Assessment and management of anovulatory infertility in polycystic ovary syndrome. Endocrinol. Metab. Clin. N. Am. 2003, 32, 639–651. [Google Scholar] [CrossRef]
- Moran, L.J.; Ko, H.; Misso, M.; Marsh, K.; Noakes, M.; Talbot, M.; Frearson, M.; Thondan, M.; Stepto, N.; Teede, H.J. Dietary composition in the treatment of polycystic ovary syndrome: A systematic review to inform evidence-based guidelines. J. Acad. Nutr. Diet. 2013, 113, 520–545. [Google Scholar] [CrossRef] [PubMed]
- Tsagareli, V.; Noakes, M.; Norman, R.J. Effect of a very-low-calorie diet on in vitro fertilization outcomes. Fertil. Steril. 2006, 86, 227–229. [Google Scholar] [CrossRef] [PubMed]
- Butler, S.T. Nutritional management to optimize fertility of dairy cows in pasture-based systems. Animal 2014, 8, 15–26. [Google Scholar] [CrossRef] [PubMed]
| Author, Year, Location | Participants; Study Duration | Cause of Infertility | Study Design | Intervention | Comparators | Fertility Outcomes (Pregnancy Rate/Other) |
|---|---|---|---|---|---|---|
| Mavropoulos et al. 2005, USA [18] | 11 women with PCOS and BMI >27 from the community; 24 weeks | PCOS | Two arm pre/post study | <20 g carbohydrate/day, ad libitum MJ/day (<6% carbohydrate if consuming 5 MJ/day) | Own control on usual diet (pre-intervention) | Reproductive hormones; Pregnancy rates |
| Moran et al. 2003, Australia [20] | 45 overweight women with PCOS; 12 weeks | PCOS | Matched control trial | High protein, low carbohydrate (6 MJ—40% carbohydrate, 30% protein, 30% fat) | Usual diet (55% carbohydrate, 15% protein, 30% fat) but limited to 6 MJ/day | Reproductive hormones; Ovulation rates |
| Palomba et al. 2008, Italy [19] | 20 obese PCOS patients with anovulatory infertility; 24 weeks | PCOS | Two arm pre/post study | High protein, low carbohydrate diet (35% protein, 45% carbohydrate, 20% fat) with 3.3 MJ deficit | Usual diet and 3 physical activity sessions per week | Reproductive hormones; Ovulation rates; Pregnancy rates |
| Palomba et al. 2010, Italy [22] | 96 overweight or obese Clomiphene citrate resistant women; 2 weeks intervention, 4 weeks clomid (±intervention) | PCOS | Three arm randomised parallel controlled trial | Structured exercise + 35% protein, 45% carbohydrate, 20% fat diet with 4.2 MJ/day deficit + clomiphene citrate | Usual diet followed by clomiphene citrate | Reproductive hormones; Ovulation rate |
| Sim et al. 2014, Australia [3] | 49 obese women planning ART; 12 weeks intervention, 12 month follow up | Mixed | Randomised controlled trial | Very low energy diet (2.5 MJ/day, 34% carbohydrate) for 6/52 followed by 6/52 hypocaloric diet | Usual diet | Pregnancy rates |
| Stamets et al. 2004, USA [21] | 35 obese women with PCOS; 1 month | PCOS | Randomised controlled trial | Diet (40% carbohydrate, 30% protein, 30% fat with 4.2 MJ/day energy deficit) | Usual diet (55% carbohydrate, 15% protein, 30% fat) with a 4.2 MJ/day energy deficit | Reproductive hormones |
| Thomson et al. 2008, Australia [23] | 94 overweight and obese women; 20 weeks | PCOS | Randomised parallel study (only one arm, “Diet Only“ was included in this review) | Diet (5–6 MJ/day, 30% protein, 40% carbohydrate, 30% fat) | Own control on usual diet (pre-intervention) | Reproductive hormones; Ovulation rates |
| Author | Validity Rating b | Overall Rating | Examples of Reasons for Downgrading | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |||
| Mavropoulos et al. 2005, USA [18] | Y | Y | N/A | Y | N/A | Y | Y | Y | Y | N | Neutral | Statement of the role of funding source not included. |
| Moran et al. 2003, Australia [20] | Y | Y | Y | Y | N | Y | Y | Y | Y | N | Positive | Statement of the role of funding source not included. |
| Palomba et al. 2008, Italy [19] | Y | Y | Y | N | N | Y | Y | Y | Y | Y | Positive | Patients self-selected their intervention groups. Dropout rates were reported, but no further explanation was provided. |
| Palomba et al. 2010, Italy [22] | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Positive | Source of participants (e.g., whether consecutive) was unclear. |
| Sim et al. 2014, Australia [3] | Y | Y | Y | Y | Y | Y | Y | Y | Y | N | Positive | Two groups didn’t match all aspects of demographics/anthropometry explained by the strict randomisation technique used. Statement of the role of funding source not included. |
| Stamets et al. 2004, USA [21] | Y | Y | Y | Y | N | Y | Y | Y | Y | Y | Positive | No reporting of blinding throughout the study protocol. |
| Thomson et al. 2008, Australia [23] | Y | Y | Y | Y | N | Y | Y | Y | Y | Y | Positive | Unclear whether the study dietitian was blinded. |
| Study (Author, Year) | Intervention Group | Control Group | p-Value |
|---|---|---|---|
| Reproductive hormones | |||
| Mavropoulos, 2005 [18] | Free testosterone ng/dL 1.7 | Free testosterone ng/dL 2.19 | 0.04 |
| Luteinizing Hormone/Follicle Stimulating Hormone ratio 1.21 | Luteinizing Hormone/Follicle Stimulating Hormone ratio 2.23 | 0.03 | |
| Fasting serum insulin mcIU/mL 8.2 | Fasting serum insulin mcIU/mL 23.5 | 0.002 | |
| Moran, 2003 [20] | Fasting glucose (mmol/L) 5.42 ± 0.13 | Fasting glucose (mmol/L) 5.31 ± 0.17 | NS |
| Fasting insulin (mU/L) 16.6 ± 2.4 | Fasting insulin (mU/L) 12.8 ± 2.0 | <0.01 | |
| Sex Hormone Binding Globule (nmol/L) 25 ± 2.5 | Sex Hormone Binding Globule (nmol/L) 35 ± 5 | 0.027 | |
| Testosterone (nmol/L) 1.45 ± 0.2 | Testosterone (nmol/L) 1.3 ± 0.1 | 0.01 | |
| Free Androgen Index (nmol/L) 7 ± 1.5 | Free Androgen Index (nmol/L) 4.5 ± 1 | 0.004 | |
| Palomba, 2008 [19] | Follicle Stimulating Hormone (mIU/mL) 4.2 ± 13.2 | Follicle Stimulating Hormone (mIU/mL) −1.2 ± 3.2 | NS |
| Testosterone (nmol/L) −28.7 ± 11.7 | Testosterone (nmol/L) −33.4 ± 14.3 | <0.05 | |
| Sex Hormone Binding Globule (nmol/L) 41.9 ± 19.1 | Sex Hormone Binding Globule (nmol/L) 82.5 ± 30.6 | <0.05 | |
| Free Androgen Index (%) −18.1 ± 9.7 | Free Androgen Index (%) −27.2 ± 9.2 | <0.05 | |
| Fasting glucose (mmol/L) 1.2 ± 8.6 | Fasting glucose (mmol/L) 0.4 ± 4.1 | NS | |
| Fasting insulin (pmol/L) −13.1 ± 8.6 | Fasting insulin (pmol/L) −23.4 ± 10.0 | <0.05 | |
| Palomba, 2010 [22] | Follicle Stimulating Hormone (mIU/mL) 4.9 ± 3.1 | Follicle Stimulating Hormone (mIU/mL) 4.2 ± 1.2 | NS |
| Testosterone (nmol/L) 2.2 ± 0.6 | Testosterone (nmol/L) 2.51 ± 0.9 | <0.05 | |
| Sex Hormone Binding Globule (nmol/L) 25.3 ± 3.2 | Sex Hormone Binding Globule (nmol/L) 17.4 ± 3.1 | <0.05 | |
| Free Androgen Index (%) 10.8 ± 3.5 | Free Androgen Index (%) 11.6 ± 3.7 | <0.05 | |
| Fasting glucose (mmol/L) 4.0 ± 1.7 | Fasting glucose (mmol/L) 4.0 ± 1.5 | NS | |
| Fasting insulin (pmol/L) 15.8 ± 3.9 | Fasting insulin (pmol/L) 17.9 ± 4.2 | <0.05 | |
| Stamets, 2004 [21] | Testosterone (ng/dL) −9 ± 21 | Testosterone (ng/dL) −9 ± 18 | 0.96 |
| Luteinizing hormone (mIU/mL) 7 ± 30 | Luteinizing hormone (mIU/mL) 2 ± 11 | 0.59 | |
| Follicle Stimulating Hormone (mIU/mL) −1 ± 5 | Follicle Stimulating Hormone (mIU/mL) 2 ± 4 | 0.12 | |
| Area under Curve Insulin 3 h Oral Glucose Tolerance Test −2912 ± 13,562 | Area Under Curve Insulin 3 h Oral Glucose Tolerance Test −8734 ± 12,218 | 0.26 | |
| Area Under Curve Glucose 3 h Oral Glucose Tolerance Test −87 ± 2803 | Area Under Curve Glucose 3 h Oral Glucose Tolerance Test −93 ± 2049 | 0.99 | |
| Thomson, 2008 [23] | Glucose (mmol/L) 4.96 ± 0.6 | Glucose (mmol/L)5.32 ± 0.49 | <0.01 |
| Insulin (mU/L) 13.5 ± 9.9 | Insulin (mU/L) 17.7 ± 8.2 | <0.01 | |
| Testosterone (nmol/L) 2.09 ± 0.98 | Testosterone (nmol/L) 2.36 ± 0.71 | NS | |
| Sex Hormone Binding Globule (nmol/L) 31.5 ± 17.5 | Sex Hormone Binding Globule (nmol/L) 27.4 ± 15.9 | NS | |
| Free Androgen Index (%) 8.4 ± 6.6 | Free Androgen Index (%) 11.2 ± 5.5 | <0.01 | |
| Ovulation rates | |||
| Moran, 2003 [20] | Improved menstrual cyclicity 6/14 42% | Improved menstrual cyclicity 5/14 35% | NR |
| Palomba, 2008 [19] | Menses frequency (# observed menses/no expected cycles, %) 18/118 15.3% | Menses frequency (# observed menses/no expected cycles, %) 28/107 26.2% | 0.043 |
| Ovulation rate (# ovulatory cycles/# observed cycles, %) 18/119 15.1% | Ovulation rate (# ovulatory cycles/# observed cycles, %) 28/113 24.8% | 0.032 | |
| Palomba, 2010 [22] | Ovulation rate 12/32 37.5% | Ovulation rate 3/32 9.4% | 0.020 |
| Thomson, 2008 [23] | Improved menstrual cyclicity 3/14 21.4% | Improved menstrual cyclicity 0/14 0% | NR |
| Pregnancy rates | |||
| Mavropoulos, 2005 [18] | Pregnancy 2/5 40% | Pregnancy 0/5 0% | NR |
| Moran, 2003 [20] | Pregnancy (# pregnancies/# patients, %) 2/14 14% | Pregnancy (# pregnancies/# patients, %) 1/14 7% | NR |
| Palomba, 2008 [19] | Pregnancy (# pregnancies/# patients, %) 2/20 10% | Pregnancy (# pregnancies/# patients, %) 7/20 35% | 0.058 |
| Sim, 2014 [3] | Natural conception 3/27 0.1% | Natural conception 0/22 0% | 0.11 |
| Pregnancy rate 13/27 48.1% | Pregnancy rate 3/22 13.6% | 0.007 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McGrice, M.; Porter, J. The Effect of Low Carbohydrate Diets on Fertility Hormones and Outcomes in Overweight and Obese Women: A Systematic Review. Nutrients 2017, 9, 204. https://doi.org/10.3390/nu9030204
McGrice M, Porter J. The Effect of Low Carbohydrate Diets on Fertility Hormones and Outcomes in Overweight and Obese Women: A Systematic Review. Nutrients. 2017; 9(3):204. https://doi.org/10.3390/nu9030204
Chicago/Turabian StyleMcGrice, Melanie, and Judi Porter. 2017. "The Effect of Low Carbohydrate Diets on Fertility Hormones and Outcomes in Overweight and Obese Women: A Systematic Review" Nutrients 9, no. 3: 204. https://doi.org/10.3390/nu9030204
APA StyleMcGrice, M., & Porter, J. (2017). The Effect of Low Carbohydrate Diets on Fertility Hormones and Outcomes in Overweight and Obese Women: A Systematic Review. Nutrients, 9(3), 204. https://doi.org/10.3390/nu9030204




