Metabolic Effects of Glucose-Fructose Co-Ingestion Compared to Glucose Alone during Exercise in Type 1 Diabetes
Abstract
:1. Introduction
2. Material and Methods
2.1. Inclusion Criteria
2.2. Experimental Design and Protocol
2.3. Pre-Study Standardization Procedures
2.4. Sampling Procedures for Metabolites and Hormones
2.5. Respiratory Gas Exchange, Cardiopulmonary Monitoring, and Substrate Oxidation
2.6. Stable Isotopes
2.7. Calculations of Glucose and Fructose Turnover
2.8. Statistical Analysis
3. Results
3.1. Baseline Characteristics and Pre-Study Conditions
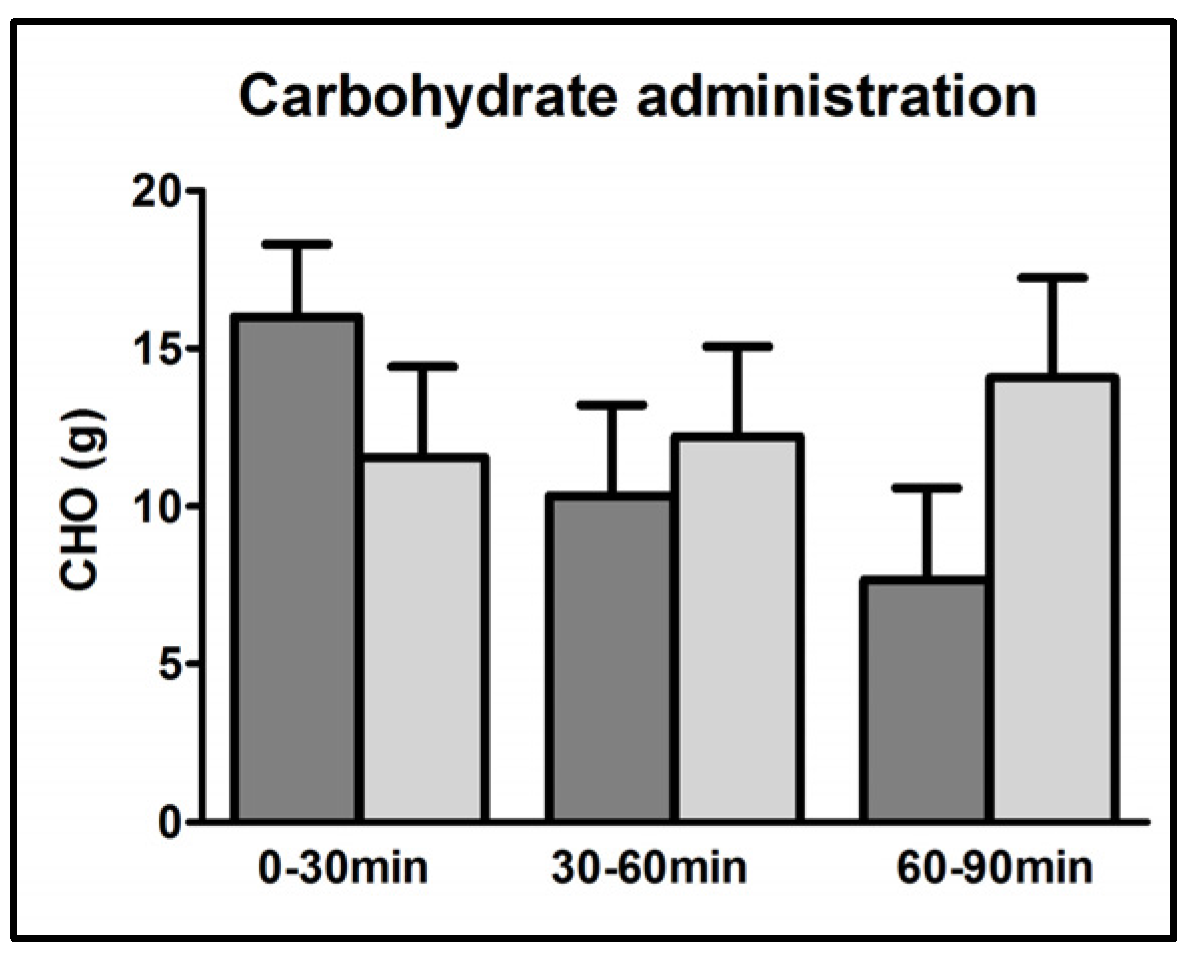
3.2. CHO Requirements
3.3. Energy Expenditure
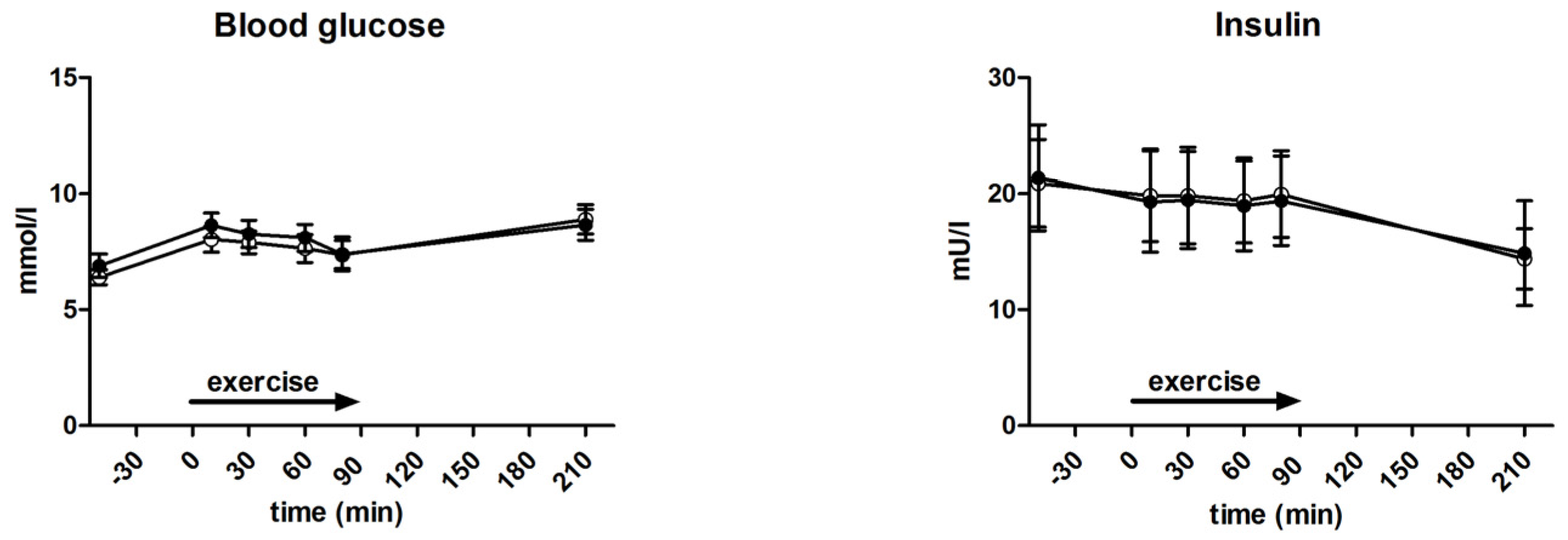
3.4. Glycaemia and Insulin Levels
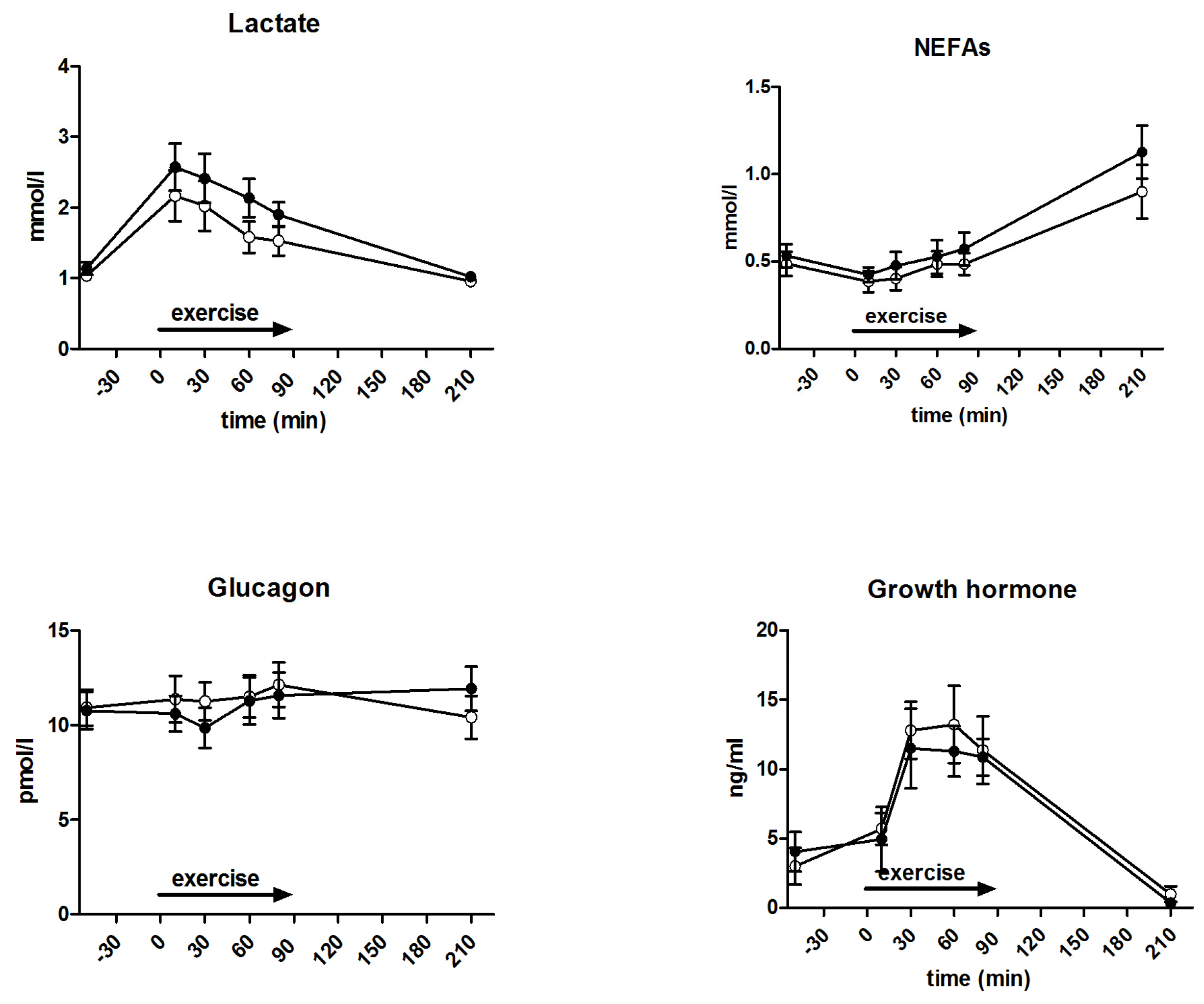
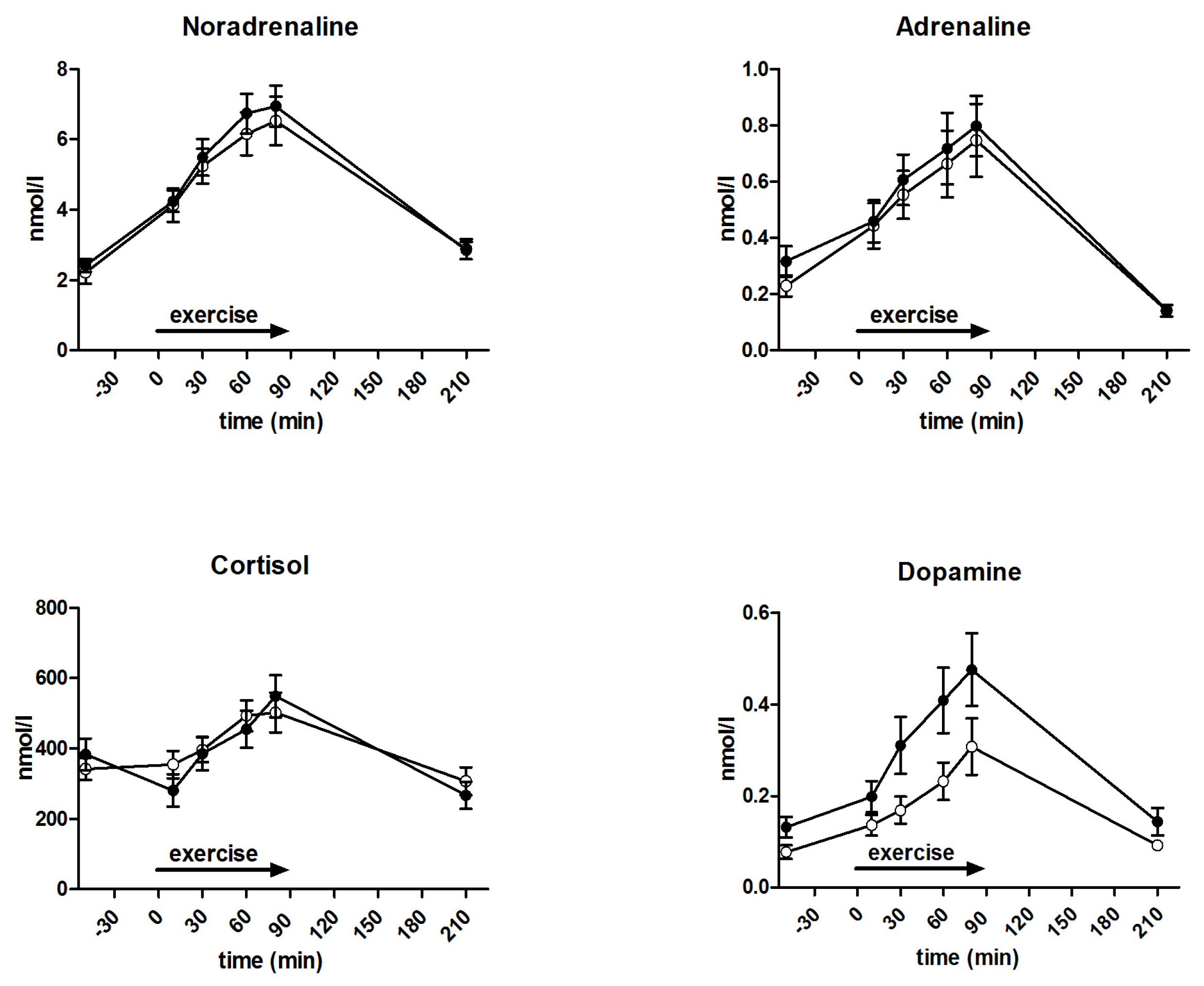
3.5. Metabolites and Counter-Regulatory Hormones
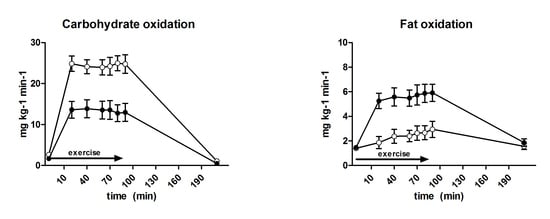
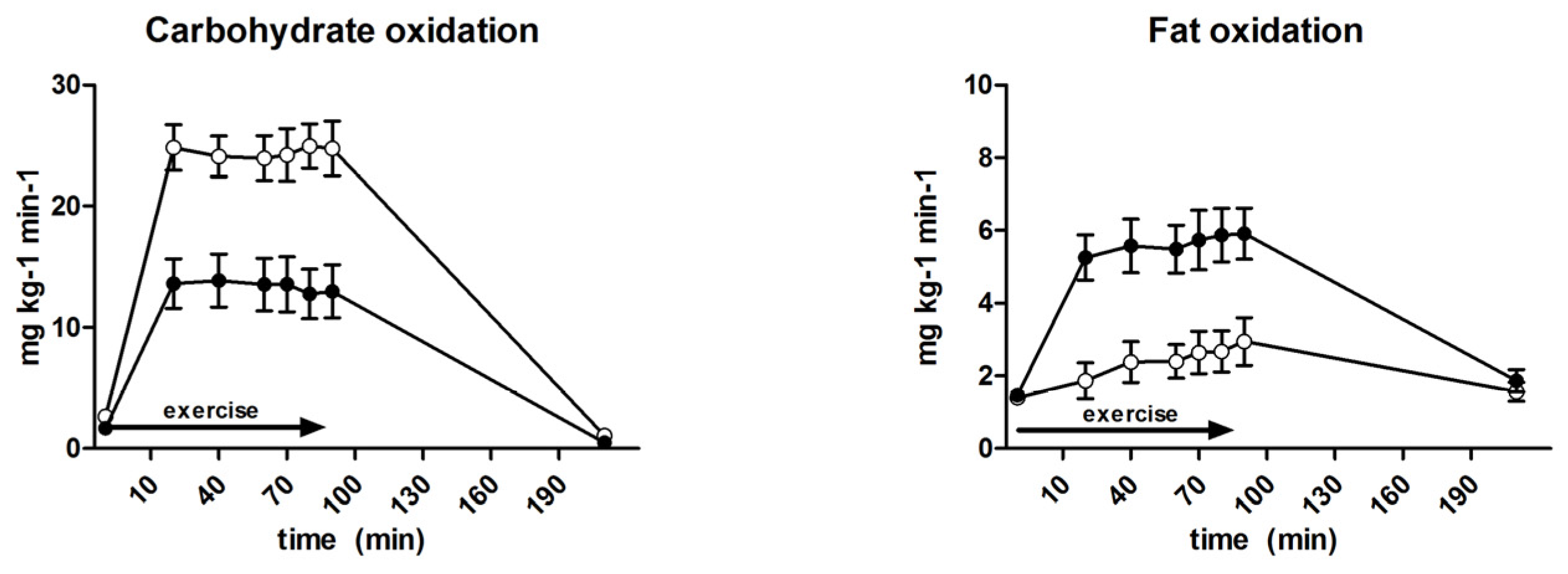
3.6. Substrate Oxidation and Turnover
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- LaPorte, R.E.; Dorman, J.S.; Tajima, N.; Cruickshanks, K.J.; Orchard, T.J.; Cavender, D.E.; Becker, D.J.; Drash, A.L. Pittsburgh Insulin-Dependent Diabetes Mellitus Morbidity and Mortality Study: Physical activity and diabetic complications. Pediatrics 1986, 78, 1027–1033. [Google Scholar]
- Zoppini, G.; Carlini, M.; Muggeo, M. Self-reported exercise and quality of life in young type 1 diabetic subjects. Diabetes Nutr. Metab. 2003, 16, 77–80. [Google Scholar] [PubMed]
- Brazeau, A.S.; Rabasa-Lhoret, R.; Strychar, I.; Mircescu, H. Barriers to physical activity among patients with type 1 diabetes. Diabetes Care 2008, 31, 2108–2109. [Google Scholar] [CrossRef] [PubMed]
- Riddell, M.C.; Gallen, I.W.; Smart, C.E.; Taplin, C.E.; Adolfsson, P.; Lumb, A.N.; Kowalski, A.; Rabasa-Lhoret, R.; McCrimmon, R.J.; Hume, C.; et al. Exercise management in type 1 diabetes: A consensus statement. Lancet Diabetes Endocrinol. 2017. [Google Scholar] [CrossRef]
- McAuley, S.A.; Horsburgh, J.C.; Ward, G.M.; La Gerche, A.; Gooley, J.L.; Jenkins, A.J.; MacIsaac, R.J.; O’Neal, D.N. Insulin pump basal adjustment for exercise in type 1 diabetes: A randomised crossover study. Diabetologia 2016, 59, 1636–1644. [Google Scholar] [CrossRef] [PubMed]
- Grimm, J.J.; Ybarra, J.; Berné, C.; Muchnick, S.; Golay, A. A new table for prevention of hypoglycaemia during physical activity in type 1 diabetic patients. Diabetes Metab. 2004, 30, 465–470. [Google Scholar] [CrossRef]
- Francescato, M.P.; Stel, G.; Stenner, E.; Geat, M. Prolonged exercise in type 1 diabetes: Performance of a customizable algorithm to estimate the carbohydrate supplements to minimize glycemic imbalances. PLoS ONE 2015, 10, E0125220. [Google Scholar] [CrossRef]
- Gray, B.J.; Page, R.; Turner, D.; West, D.J.; Campbell, M.D.; Kilduff, L.P.; Stephens, J.W.; Bain, S.C.; Bracken, R.M. Improved end-stage high-intensity performance but similar glycemic responses after waxy barley starch ingestion compared to dextrose in type 1 diabetes. J. Sports Med. Phys. Fit. 2016, 56, 1392–1400. [Google Scholar]
- Bracken, R.M.; Page, R.; Gray, B.; Kilduff, L.P.; West, D.J.; Stephens, J.W.; Bain, S.C. Isomaltulose improves glycemia and maintains run performance in type 1 diabetes. Med. Sci. Sports Exerc. 2012, 44, 800–808. [Google Scholar] [CrossRef] [PubMed]
- West, D.J.; Morton, R.D.; Stephens, J.W.; Bain, S.C.; Kilduff, L.P.; Luzio, S.; Still, R.; Bracken, R.M. Isomaltulose Improves Postexercise Glycemia by Reducing CHO Oxidation in T1DM. Med. Sci. Sports Exerc. 2011, 43, 204–210. [Google Scholar] [CrossRef] [PubMed]
- West, D.J.; Stephens, J.W.; Bain, S.C.; Kilduff, L.P.; Luzio, S.; Still, R.; Bracken, R.M. A combined insulin reduction and carbohydrate feeding strategy 30 min before running best preserves blood glucose concentration after exercise through improved fuel oxidation in type 1 diabetes mellitus. J. Sports Sci. 2011, 29, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Wood, I.S.; Trayhurn, P. Glucose transporters (GLUT and SGLT): Expanded families of sugar transport proteins. Br. J. Nutr. 2003, 89, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Mayes, P.A. Intermediary metabolism of fructose. Am. J. Clin. Nutr. 1993, 58, 754S–765S. [Google Scholar] [PubMed]
- Jeukendrup, A.E.; Moseley, L. Multiple transportable carbohydrates enhance gastric emptying and fluid delivery. Scand. J. Med. Sci. Sports 2010, 20, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Lecoultre, V.; Benoit, R.; Carrel, G.; Schutz, Y.; Millet, G.P.; Tappy, L.; Schneiter, P. Fructose and glucose co-ingestion during prolonged exercise increases lactate and glucose fluxes and oxidation compared with an equimolar intake of glucose. Am. J. Clin. Nutr. 2010, 92, 1071–1079. [Google Scholar] [CrossRef]
- Wilson, P.B.; Ingraham, S.J. Glucose-fructose likely improves gastrointestinal comfort and endurance running performance relative to glucose-only. Scand. J. Med. Sci. Sports 2015, 25, E613–E620. [Google Scholar] [CrossRef] [PubMed]
- Jenni, S.; Oetliker, C.; Allemann, S.; Ith, M.; Tappy, L.; Wuerth, S.; Egger, A.; Boesch, C.; Schneiter, P.; Diem, P.; et al. Fuel metabolism during exercise in euglycaemia and hyperglycaemia in patients with type 1 diabetes mellitus—A prospective single-blinded randomised crossover trial. Diabetologia 2008, 51, 1457–1465. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, P.J. From plant physiology to human metabolic investigations. Diabetologia 1985, 28, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Dunand, M.; Gubian, D.; Stauffer, M.; Abid, K.; Grouzmann, E. High-throughput and sensitive quantitation of plasma catecholamines by ultraperformance liquid chromatography-tandem mass spectrometry using a solid phase microwell extraction plate. Anal. Chem. 2013, 85, 3539–3544. [Google Scholar] [CrossRef] [PubMed]
- Peronnet, F.; Massicotte, D. Table of nonprotein respiratory quotient: An update. Can. J. Sport Sci. 1991, 16, 23–29. [Google Scholar] [PubMed]
- Wolfe, R.R.; Chinkes, D.L. Isotope Tracers in Metabolic Research: Principles and Practice of Kinetic Analysis; Wiley: Hoboken, NJ, USA, 2005. [Google Scholar]
- Tounian, P.; Schneiter, P.; Henry, S.; Delarue, J.; Tappy, L. Effects of dexamethasone on hepatic glucose production and fructose metabolism in healthy humans. Am. J. Physiol. 1997, 273, E315–E320. [Google Scholar] [PubMed]
- Petersen, K.F.; Laurent, D.; Yu, C.; Cline, G.W.; Shulman, G.I. Stimulating effects of low-dose fructose on insulin-stimulated hepatic glycogen synthesis in humans. Diabetes 2001, 50, 1263–1268. [Google Scholar] [CrossRef] [PubMed]
- Steele, R.; Wall, J.S.; De Bodo, R.C.; Altszuler, N. Measurement of size and turnover rate of body glucose pool by the isotope dilution method. Am. J. Physiol. 1956, 187, 15–24. [Google Scholar] [PubMed]
- Romijn, J.A.; Coyle, E.F.; Sidossis, L.S.; Gastaldelli, A.; Horowitz, J.F.; Endert, E.; Wolfe, R.R. Regulation of endogenous fat and carbohydrate metabolism in relation to exercise intensity and duration. Am. J. Physiol. 1993, 265, E380–E391. [Google Scholar] [PubMed]
- Robert, J.J.; Koziet, J.; Chauvet, D.; Darmaun, D.; Desjeux, J.F.; Young, V.R. Use of 13C-labeled glucose for estimating glucose oxidation: Some design considerations. J. Appl. Physiol. (1985) 1987, 63, 1725–1732. [Google Scholar]
- Schneiter, P.; Pasche, O.; Di Vetta, V.; Jequier, E.; Tappy, L. Noninvasive assessment of in vivo glycogen kinetics in humans: Effect of increased physical activity on glycogen breakdown and synthesis. Eur. J. Appl. Physiol. Occup. Physiol. 1994, 69, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Dube, M.C.; Lavoie, C.; Weisnagel, S.J. Glucose or intermittent high-intensity exercise in glargine/glulisine users with T1DM. Med. Sci. Sports Exerc. 2013, 45, 3–7. [Google Scholar] [CrossRef]
- Rowlands, D.S.; Houltham, S.; Musa-Veloso, K.; Brown, F.; Paulionis, L.; Bailey, D. Fructose-Glucose Composite Carbohydrates and Endurance Performance: Critical Review and Future Perspectives. Sports Med. 2015, 45, 1561–1576. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Summers, R.W.; Schedl, H.P.; Flanagan, S.W.; Chang, R.; Gisolfi, C.V. Effects of carbohydrate type and concentration and solution osmolality on water absorption. Med. Sci. Sports Exerc. 1995, 27, 1607–1615. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.D.; Tarpey, M.D.; Kass, L.S.; Tarpey, R.J.; Roberts, M.G. Assessing a commercially available sports drink on exogenous carbohydrate oxidation, fluid delivery and sustained exercise performance. J. Int. Soc. Sports Nutr. 2014, 11, 8. [Google Scholar] [CrossRef] [PubMed]
- Dimitriadis, G.; Mitrou, P.; Lambadiari, V.; Maratou, E.; Raptis, S.A. Insulin effects in muscle and adipose tissue. Diabetes Res. Clin. Pract. 2011, 93 (Suppl. 1), S52–S59. [Google Scholar] [CrossRef]
- Macdonald, I.; Keyser, A.; Pacy, D. Some effects, in man, of varying the load of glucose, sucrose, fructose, or sorbitol on various metabolites in blood. Am. J. Clin. Nutr. 1978, 31, 1305–1311. [Google Scholar] [PubMed]
- Sahebjami, H.; Scalettar, R. Effects of fructose infusion on lactate and uric acid metabolism. Lancet 1971, 1, 366–369. [Google Scholar] [CrossRef]
- Chandramouli, V.; Kumaran, K.; Ekberg, K.; Wahren, J.; Landau, B.R. Quantitation of the pathways followed in the conversion of fructose to glucose in liver. Metabolism 1993, 42, 1420–1423. [Google Scholar] [CrossRef]
- Coss-Bu, J.A.; Sunehag, A.L.; Haymond, M.W. Contribution of galactose and fructose to glucose homeostasis. Metabolism 2009, 58, 1050–1058. [Google Scholar] [CrossRef] [PubMed]
- Brooks, G.A.; Dubouchaud, H.; Brown, M.; Sicurello, J.P.; Butz, C.E. Role of mitochondrial lactate dehydrogenase and lactate oxidation in the intracellular lactate shuttle. Proc. Natl. Acad. Sci. USA 1999, 96, 1129–1134. [Google Scholar] [CrossRef] [PubMed]
- Randle, P.J.; Garland, P.B.; Hales, C.N.; Newsholme, E.A. The glucose fatty-acid cycle. Its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus. Lancet 1963, 1, 785–789. [Google Scholar] [CrossRef]
- Sugden, M.C.; Holness, M.J. Recent advances in mechanisms regulating glucose oxidation at the level of the pyruvate dehydrogenase complex by PDKs. Am. J. Physiol. Endocrinol. Metab. 2003, 284, E855–E862. [Google Scholar] [CrossRef] [PubMed]
- Massicotte, D.; Péronnet, F.; Allah, C.; Hillaire-Marcel, C.; Ledoux, M.; Brisson, G. Metabolic response to [13C]glucose and [13C]fructose ingestion during exercise. J. Appl. Physiol. (1985) 1986, 61, 1180–1184. [Google Scholar]
- Moberg, E.; Sjöberg, S.; Hagström-Toft, E.; Bolinder, J. No apparent suppression by insulin of in vivo skeletal muscle lipolysis in nonobese women. Am. J. Physiol. Endocrinol. Metab. 2002, 283, E295–E301. [Google Scholar] [CrossRef] [PubMed]
- Björkman, O.; Felig, P. Role of the kidney in the metabolism of fructose in 60-hour fasted humans. Diabetes 1982, 31, 516–520. [Google Scholar] [CrossRef] [PubMed]
- Thompson, G.E. Dopamine and lipolysis in adipose tissue of the sheep. Q. J. Exp. Physiol. 1984, 69, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Ruttimann, Y.; Schutz, Y.; Jéquier, E.; Lemarchand, T.; Chioléro, R. Thermogenic and metabolic effects of dopamine in healthy men. Crit. Care Med. 1991, 19, 1030–1036. [Google Scholar] [CrossRef]
- Bogardus, C.; Thuillez, P.; Ravussin, E.; Vasquez, B.; Narimiga, M.; Azhar, S. Effect of muscle glycogen depletion on in vivo insulin action in man. J. Clin. Investig. 1983, 72, 1605–1610. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Pagliassotti, M.J. Hepatospecific effects of fructose on c-jun NH2-terminal kinase: Implications for hepatic insulin resistance. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E926–E933. [Google Scholar] [CrossRef] [PubMed]
- Lecoultre, V.; Egli, L.; Carrel, G.; Theytaz, F.; Kreis, R.; Schneiter, P.; Boss, A.; Zwygart, K.; Lê, K.A.; Bortolotti, M.; et al. Effects of fructose and glucose overfeeding on hepatic insulin sensitivity and intrahepatic lipids in healthy humans. Obesity (Silver Spring) 2013, 21, 782–785. [Google Scholar] [CrossRef] [PubMed]
- Faeh, D.; Minehira, K.; Schwarz, J.M.; Periasamy, R.; Park, S.; Tappy, L. Effect of fructose overfeeding and fish oil administration on hepatic de novo lipogenesis and insulin sensitivity in healthy men. Diabetes 2005, 54, 1907–1913. [Google Scholar] [CrossRef] [PubMed]
- Egli, L.; Lecoultre, V.; Theytaz, F.; Campos, V.; Hodson, L.; Schneiter, P.; Mittendorfer, B.; Patterson, B.W.; Fielding, B.A.; Gerber, P.A.; et al. Exercise prevents fructose-induced hypertriglyceridemia in healthy young subjects. Diabetes 2013, 62, 2259–2265. [Google Scholar] [CrossRef] [PubMed]
- Bidwell, A.J.; Fairchild, T.J.; Redmond, J.; Wang, L.; Keslacy, S.; Kanaley, J.A. Physical activity offsets the negative effects of a high-fructose diet. Med. Sci. Sports Exerc. 2014, 46, 2091–2098. [Google Scholar] [CrossRef] [PubMed]
- Galassetti, P.; Tate, D.; Neill, R.A.; Morrey, S.; Wasserman, D.H.; Davis, S.N. Effect of sex on counterregulatory responses to exercise after antecedent hypoglycemia in type 1 diabetes. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E16–E24. [Google Scholar] [CrossRef] [PubMed]
- Devries, M.C.; Hamadeh, M.J.; Phillips, S.M.; Tarnopolsky, M.A. Menstrual cycle phase and sex influence muscle glycogen utilization and glucose turnover during moderate-intensity endurance exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 291, R1120–R1128. [Google Scholar] [CrossRef] [PubMed]
- Hovorka, R.; Jayatillake, H.; Rogatsky, E.; Tomuta, V.; Hovorka, T.; Stein, D.T. Calculating glucose fluxes during meal tolerance test: A new computational approach. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E610–E619. [Google Scholar] [CrossRef] [PubMed]
| n = 15 | |
|---|---|
| Age (years) | 26.1 ± 4.8 |
| Weight (kg) | 80.4 ± 10.7 |
| Height (m) | 1.81 ± 0.08 |
| BMI (kg/m2) | 24.5 ± 3.2 |
| Fat-free mass (%) | 78.8 ± 7.1 |
| BMR (MJ/day) | 8.3 ± 0.9 |
| VO2max (mL·(kg·body·weight)−1·min−1) | 47 ± 9 |
| Diabetes duration (years) | 13.3 ± 6.7 |
| Haemoglobin A1c (%) | 7.0 ± 0.6 |
| Haemoglobin A1c (mmol/mol) | 53 ± 7 |
| Total average daily insulin (U·kg−1·day−1) | 0.7 ± 0.1 |
| GLUFRU (n = 15) | GLU (n = 15) | p Value | |
|---|---|---|---|
| Carbohydrate requirements (g) | 34.0 ± 2.9 | 37.8 ± 5.3 | 0.46 |
| Carbohydrate oxidation (mg·kg−1·min−1) | 18.1 ± 0.8 | 24.5 ± 0.8 | <0.001 |
| Fat oxidation (mg·kg−1·min−1) | 5.2 ± 0.2 | 2.6 ± 1.2 | <0.001 |
| Glucose appearance, Ra (mg·kg−1·min−1) | 7.0 ± 0.4 | 7.3 ± 0.4 | 0.53 |
| Glucose disappearance, Rd (mg·kg−1·min−1) | 7.8 ± 0.3 | 7.6 ± 0.5 | 0.57 |
| Metabolic clearance rate, MCR (mg·kg−1·min−1) | 6.0 ± 0.3 | 5.9 ± 0.4 | 0.80 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bally, L.; Kempf, P.; Zueger, T.; Speck, C.; Pasi, N.; Ciller, C.; Feller, K.; Loher, H.; Rosset, R.; Wilhelm, M.; et al. Metabolic Effects of Glucose-Fructose Co-Ingestion Compared to Glucose Alone during Exercise in Type 1 Diabetes. Nutrients 2017, 9, 164. https://doi.org/10.3390/nu9020164
Bally L, Kempf P, Zueger T, Speck C, Pasi N, Ciller C, Feller K, Loher H, Rosset R, Wilhelm M, et al. Metabolic Effects of Glucose-Fructose Co-Ingestion Compared to Glucose Alone during Exercise in Type 1 Diabetes. Nutrients. 2017; 9(2):164. https://doi.org/10.3390/nu9020164
Chicago/Turabian StyleBally, Lia, Patrick Kempf, Thomas Zueger, Christian Speck, Nicola Pasi, Carlos Ciller, Katrin Feller, Hannah Loher, Robin Rosset, Matthias Wilhelm, and et al. 2017. "Metabolic Effects of Glucose-Fructose Co-Ingestion Compared to Glucose Alone during Exercise in Type 1 Diabetes" Nutrients 9, no. 2: 164. https://doi.org/10.3390/nu9020164
APA StyleBally, L., Kempf, P., Zueger, T., Speck, C., Pasi, N., Ciller, C., Feller, K., Loher, H., Rosset, R., Wilhelm, M., Boesch, C., Buehler, T., Dokumaci, A. S., Tappy, L., & Stettler, C. (2017). Metabolic Effects of Glucose-Fructose Co-Ingestion Compared to Glucose Alone during Exercise in Type 1 Diabetes. Nutrients, 9(2), 164. https://doi.org/10.3390/nu9020164