Is Branched-Chain Amino Acids Supplementation an Efficient Nutritional Strategy to Alleviate Skeletal Muscle Damage? A Systematic Review
Abstract
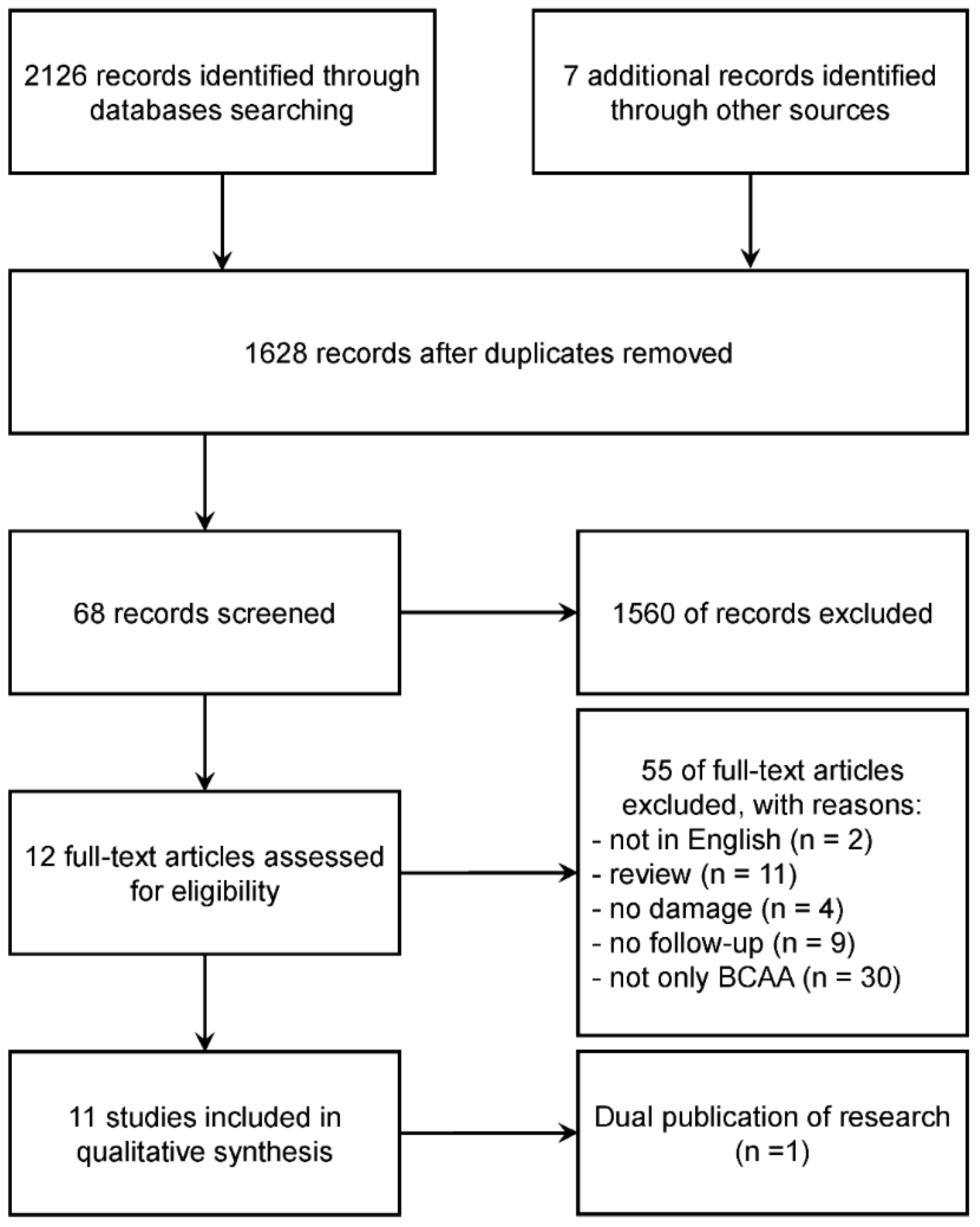
:1. Introduction
2. Methods
2.1. Eligibility Criteria
2.2. Search Strategy
2.3. Data Extraction and Quality Assessment
3. Analysis
4. Results
4.1. Study Quality
4.2. Supplementation Strategy and Muscle Damage Extent
4.3. Outcomes
5. Discussion
5.1. Extent of Exercise-Induced Muscle Damage
5.2. The Supplementation Strategy
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Froiland, K.; Koszewski, W.; Hingst, J.; Kopecky, L. Nutritional supplement use among college athletes and their sources of information. Int. J. Sport Nutr. Exerc. Metab. 2004, 14, 104–120. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.H.; Johnson, K.; Pipe, A.L. The use of dietary supplements and medications by Canadian athletes at the Atlanta and Sydney olympic games. Clin. J. Sport Med. 2006, 16, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Tsitsimpikou, C.; Tsiokanos, A.; Tsarouhas, K.; Schamasch, P.; Fitch, K.D.; Valasiadis, D.; Jamurtas, A. Medication use by athletes at the athens 2004 summer olympic games. Clin. J. Sport. Med. 2009, 19, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Goston, J.L.; Correia, M.I. Intake of nutritional supplements among people exercising in gyms and influencing factors. Nutrition 2010, 26, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Harper, A.E.; Miller, R.H.; Block, K.P. Branched-chain amino acid metabolism. Annu. Rev. Nutr. 1984, 4, 409–454. [Google Scholar] [CrossRef] [PubMed]
- Rennie, M.J.; Tipton, K.D. Protein and amino acid metabolism during and after exercise and the effects of nutrition. Annu. Rev. Nutr. 2000, 20, 457–483. [Google Scholar] [CrossRef] [PubMed]
- Valerio, A.; D’Antona, G.; Nisoli, E. Branched-chain amino acids, mitochondrial biogenesis, and healthspan: An evolutionary perspective. Aging 2011, 3, 464–478. [Google Scholar] [CrossRef] [PubMed]
- Shimomura, Y.; Kobayashi, H.; Mawatari, K.; Akita, K.; Inaguma, A.; Watanabe, S.; Bajotto, G.; Sato, J. Effects of squat exercise and branched-chain amino acid supplementation on plasma free amino acid concentrations in young women. J. Nutr. Sci. Vitaminol. (Tokyo) 2009, 55, 288–291. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.E. Observations of branched-chain amino acid administration in humans. J. Nutr. 2005, 135, 1580S–1584S. [Google Scholar] [PubMed]
- Mattick, J.S.; Kamisoglu, K.; Ierapetritou, M.G.; Androulakis, I.P.; Berthiaume, F. Branched-chain amino acid supplementation: Impact on signaling and relevance to critical illness. Wiley Interdiscip. Rev. Syst. Biol. Med 2013, 5, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Blomstrand, E.; Eliasson, J.; Karlsson, H.K.; Kohnke, R. Branched-chain amino acids activate key enzymes in protein synthesis after physical exercise. J. Nutr. 2006, 136, 269S–273S. [Google Scholar] [PubMed]
- Kimball, S.R.; Jefferson, L.S. Signaling pathways and molecular mechanisms through which branched-chain amino acids mediate translational control of protein synthesis. J. Nutr. 2006, 136, 227S–231S. [Google Scholar] [PubMed]
- Norton, L.E.; Layman, D.K. Leucine regulates translation initiation of protein synthesis in skeletal muscle after exercise. J. Nutr. 2006, 136, 533S–537S. [Google Scholar] [PubMed]
- Dreyer, H.C.; Drummond, M.J.; Pennings, B.; Fujita, S.; Glynn, E.L.; Chinkes, D.L.; Dhanani, S.; Volpi, E.; Rasmussen, B.B. Leucine-enriched essential amino acid and carbohydrate ingestion following resistance exercise enhances mtor signaling and protein synthesis in human muscle. Am. J. Physiol. Endocrinol. Metab. 2008, 294, E392–E400. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Proud, C.G. The mtor pathway in the control of protein synthesis. Physiology (Bethesda) 2006, 21, 362–369. [Google Scholar] [CrossRef] [PubMed]
- D’Antona, G.; Ragni, M.; Cardile, A.; Tedesco, L.; Dossena, M.; Bruttini, F.; Caliaro, F.; Corsetti, G.; Bottinelli, R.; Carruba, M.O.; et al. Branched-chain amino acid supplementation promotes survival and supports cardiac and skeletal muscle mitochondrial biogenesis in middle-aged mice. Cell Metab. 2010, 12, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Tatpati, L.L.; Irving, B.A.; Tom, A.; Bigelow, M.L.; Klaus, K.; Short, K.R.; Nair, K.S. The effect of branched chain amino acids on skeletal muscle mitochondrial function in young and elderly adults. J. Clin. Endocrinol. Metab. 2010, 95, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Doi, J.; Shiraishi, K.; Haida, M.; Matsuzaki, S. Abnormality of energy metabolism in the skeletal muscle of patients with liver cirrhosis and changes under administration of glucose and branched-chain amino acids. Tokai J. Exp. Clin. Med. 2004, 29, 191–198. [Google Scholar] [PubMed]
- Kutsuzawa, T.; Kurita, D.; Haida, M. Acute effects of branched-chain amino acids on muscle pH during exercise. Adv. Exerc. Sports Physiol. 2011, 16, 101–107. [Google Scholar]
- Spriet, L. Anaerobic metabolism during exercise. In Exercise Metabolism, 2nd ed.; Hargreaves, M., Spriet, L., Eds.; Human Kinetics: Champaign, IL, USA, 2006; pp. 7–27. [Google Scholar]
- Clarkson, P.M.; Hubal, M.J. Exercise-induced muscle damage in humans. Am. J. Phys. Med. Rehabil. 2002, 81, S52–S69. [Google Scholar] [CrossRef] [PubMed]
- Lieber, R.L.; Friden, J. Muscle damage is not a function of muscle force but active muscle strain. J. Appl. Physiol. 1993, 74, 520–526. [Google Scholar] [PubMed]
- Armstrong, R.B. Mechanisms of exercise-induced delayed onset muscular soreness: A brief review. Med. Sci. Sports Exerc. 1984, 16, 529–538. [Google Scholar] [CrossRef] [PubMed]
- MacLean, D.A.; Graham, T.E.; Saltin, B. Branched-chain amino acids augment ammonia metabolism while attenuating protein breakdown during exercise. Am. J. Physiol. 1994, 267, E1010–E1022. [Google Scholar] [PubMed]
- Fouré, A.; Duhamel, G.; Wegrzyk, J.; Boudinet, H.; Mattei, J.P.; Le Troter, A.; Bendahan, D.; Gondin, J. Heterogeneity of muscle damage induced by electrostimulation: A multimodal mri study. Med. Sci. Sports Exerc. 2015, 47, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Fouré, A.; Wegrzyk, J.; Le Fur, Y.; Mattei, J.P.; Boudinet, H.; Vilmen, C.; Bendahan, D.; Gondin, J. Impaired mitochondrial function and reduced energy cost as a result of muscle damage. Med. Sci. Sports Exerc. 2015, 47, 1135–1144. [Google Scholar] [CrossRef] [PubMed]
- Nair, K.S.; Schwartz, R.G.; Welle, S. Leucine as a regulator of whole body and skeletal muscle protein metabolism in humans. Am. J. Physiol. 1992, 263, E928–E934. [Google Scholar] [PubMed]
- Mackey, A.L.; Bojsen-Moller, J.; Qvortrup, K.; Langberg, H.; Suetta, C.; Kalliokoski, K.K.; Kjaer, M.; Magnusson, S.P. Evidence of skeletal muscle damage following electrically stimulated isometric muscle contractions in humans. J. Appl. Physiol. 2008, 105, 1620–1627. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The prisma statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef] [PubMed]
- AND. Evidence Analysis Manual: Steps in the Academiy Evidence Analysis Process. Available online: https://www.andeal.org/vault/2440/web/files/QCC_3.pdf (accessed on 27 July 2017).
- Coombes, J.S.; McNaughton, L.R. Effects of branched-chain amino acid supplementation on serum creatine kinase and lactate dehydrogenase after prolonged exercise. J. Sports Med. Phys. Fitness 2000, 40, 240–246. [Google Scholar] [PubMed]
- Fouré, A.; Nosaka, K.; Gastaldi, M.; Mattei, J.P.; Boudinet, H.; Guye, M.; Vilmen, C.; Le Fur, Y.; Bendahan, D.; Gondin, J. Effects of branched-chain amino acids supplementation on both plasma amino acids concentration and muscle energetics changes resulting from muscle damage: A randomized placebo controlled trial. Clin. Nutr. 2016, 35, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Gee, T.I.; Deniel, S. Branched-chain aminoacid supplementation attenuates a decrease in power-producing ability following acute strength training. J. Sports Med. Phys. Fitness 2016, 56, 1511–1517. [Google Scholar] [PubMed]
- Greer, B.K.; Woodard, J.L.; White, J.P.; Arguello, E.M.; Haymes, E.M. Branched-chain amino acid supplementation and indicators of muscle damage after endurance exercise. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, 595–607. [Google Scholar] [CrossRef] [PubMed]
- Howatson, G.; Hoad, M.; Goodall, S.; Tallent, J.; Bell, P.G.; French, D.N. Exercise-induced muscle damage is reduced in resistance-trained males by branched chain amino acids: A randomized, double-blind, placebo controlled study. J. Int. Soc. Sports Nutr. 2012, 9, 20. [Google Scholar] [CrossRef] [PubMed]
- Jackman, S.R.; Witard, O.C.; Jeukendrup, A.E.; Tipton, K.D. Branched-chain amino acid ingestion can ameliorate soreness from eccentric exercise. Med. Sci. Sports Exerc. 2010, 42, 962–970. [Google Scholar] [CrossRef] [PubMed]
- Kephart, W.C.; Mumford, P.W.; McCloskey, A.E.; Holland, A.M.; Shake, J.J.; Mobley, C.B.; Jagodinsky, A.E.; Weimar, W.H.; Oliver, G.D.; Young, K.C.; et al. Post-exercise branched chain amino acid supplementation does not affect recovery markers following three consecutive high intensity resistance training bouts compared to carbohydrate supplementation. J. Int. Soc. Sports Nutr. 2016, 13, 30. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, K.; Mizuno, M.; Mizuno, T.; Dilling-Hansen, B.; Lahoz, A.; Bertelsen, V.; Munster, H.; Jordening, H.; Hamada, K.; Doi, T. Branched-chain amino acids and arginine supplementation attenuates skeletal muscle proteolysis induced by moderate exercise in young individuals. Int. J. Sports Med. 2007, 28, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Ra, S.G.; Miyazaki, T.; Ishikura, K.; Nagayama, H.; Komine, S.; Nakata, Y.; Maeda, S.; Matsuzaki, Y.; Ohmori, H. Combined effect of branched-chain amino acids and taurine supplementation on delayed onset muscle soreness and muscle damage in high-intensity eccentric exercise. J. Int. Soc. Sports Nutr. 2013, 10, 51. [Google Scholar] [CrossRef] [PubMed]
- Shimomura, Y.; Inaguma, A.; Watanabe, S.; Yamamoto, Y.; Muramatsu, Y.; Bajotto, G.; Sato, J.; Shimomura, N.; Kobayashi, H.; Mawatari, K. Branched-chain amino acid supplementation before squat exercise and delayed-onset muscle soreness. Int. J. Sport Nutr. Exerc. Metab. 2010, 20, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Waldron, M.; Whelan, K.; Jeffries, O.; Burt, D.; Howe, L.; Patterson, S.D. The effects of acute branched-chain amino acid supplementation on recovery from a single bout of hypertrophy exercise in resistance-trained athletes. Appl. Physiol. Nutr. Metab. 2017, 42, 630–636. [Google Scholar] [CrossRef] [PubMed]
- Pasiakos, S.M.; McLellan, T.M.; Lieberman, H.R. The effects of protein supplements on muscle mass, strength, and aerobic and anaerobic power in healthy adults: A systematic review. Sports Med. 2015, 45, 111–131. [Google Scholar] [CrossRef] [PubMed]
- Warren, G.L.; Lowe, D.A.; Armstrong, R.B. Measurement tools used in the study of eccentric contraction-induced injury. Sports Med. 1999, 27, 43–59. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, P.M.; Ebbeling, C. Investigation of serum creatine kinase variability after muscle-damaging exercise. Clin. Sci. (Lond.) 1988, 75, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Fouré, A.; Nosaka, K.; Wegrzyk, J.; Duhamel, G.; Le Troter, A.; Boudinet, H.; Mattei, J.P.; Vilmen, C.; Jubeau, M.; Bendahan, D.; et al. Time course of central and peripheral alterations after isometric neuromuscular electrical stimulation-induced muscle damage. PLoS ONE 2014, 9, e107298. [Google Scholar] [CrossRef] [PubMed]
- Revill, S.I.; Robinson, J.O.; Rosen, M.; Hogg, M.I. The reliability of a linear analogue for evaluating pain. Anaesthesia 1976, 31, 1191–1198. [Google Scholar] [CrossRef] [PubMed]
- Ohnhaus, E.E.; Adler, R. Methodological problems in the measurement of pain: A comparison between the verbal rating scale and the visual analogue scale. Pain 1975, 1, 379–384. [Google Scholar] [CrossRef]
- Nosaka, K.; Newton, M.; Sacco, P. Delayed-onset muscle soreness does not reflect the magnitude of eccentric exercise-induced muscle damage. Scand. J. Med. Sci. Sports 2002, 12, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Zory, R.; Boerio, D.; Jubeau, M.; Maffiuletti, N.A. Central and peripheral fatigue of the knee extensor muscles induced by electromyostimulation. Int. J. Sports Med. 2005, 26, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Martin, V.; Millet, G.Y.; Lattier, G.; Perrod, L. Effects of recovery modes after knee extensor muscles eccentric contractions. Med. Sci. Sports Exerc. 2004, 36, 1907–1915. [Google Scholar] [CrossRef] [PubMed]
- Prasartwuth, O.; Taylor, J.L.; Gandevia, S.C. Maximal force, voluntary activation and muscle soreness after eccentric damage to human elbow flexor muscles. J. Physiol. 2005, 567, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Behrens, M.; Mau-Moeller, A.; Bruhn, S. Effect of exercise-induced muscle damage on neuromuscular function of the quadriceps muscle. Int. J. Sports Med. 2012, 33, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Fouré, A.; Le Troter, A.; Guye, M.; Mattei, J.P.; Bendahan, D.; Gondin, J. Localization and quantification of intramuscular damage using statistical parametric mapping and skeletal muscle parcellation. Sci. Rep. 2015, 5, 18580. [Google Scholar] [CrossRef] [PubMed]
- Lacourpaille, L.; Nordez, A.; Hug, F.; Couturier, A.; Dibie, C.; Guilhem, G. Time-course effect of exercise-induced muscle damage on localized muscle mechanical properties assessed using elastography. Acta Physiol. (Oxf.) 2014, 211, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Guilhem, G.; Hug, F.; Couturier, A.; Regnault, S.; Bournat, L.; Filliard, J.R.; Dorel, S. Effects of air-pulsed cryotherapy on neuromuscular recovery subsequent to exercise-induced muscle damage. Am. J. Sports Med. 2013, 41, 1942–1951. [Google Scholar] [CrossRef] [PubMed]
- Nosaka, K.; Sakamoto, K. Effect of elbow joint angle on the magnitude of muscle damage to the elbow flexors. Med. Sci. Sports Exerc. 2001, 33, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Davies, R.C.; Eston, R.G.; Fulford, J.; Rowlands, A.V.; Jones, A.M. Muscle damage alters the metabolic response to dynamic exercise in humans: A 31p-mrs study. J. Appl. Physiol. 2011, 111, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Sorichter, S.; Koller, A.; Haid, C.; Wicke, K.; Judmaier, W.; Werner, P.; Raas, E. Light concentric exercise and heavy eccentric muscle loading: Effects on ck, mri and markers of inflammation. Int. J. Sports Med. 1995, 16, 288–292. [Google Scholar] [CrossRef] [PubMed]
- Aboodarda, S.J.; George, J.; Mokhtar, A.H.; Thompson, M. Muscle strength and damage following two modes of variable resistance training. J. Sports Sci. Med. 2011, 10, 635–642. [Google Scholar] [PubMed]
- Macaluso, F.; Isaacs, A.W.; Myburgh, K.H. Preferential type ii muscle fiber damage from plyometric exercise. J. Athl. Train. 2012, 47, 414–420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Areces, F.; Salinero, J.J.; Abian-Vicen, J.; Gonzalez-Millan, C.; Gallo-Salazar, C.; Ruiz-Vicente, D.; Lara, B.; Del Coso, J. A 7-day oral supplementation with branched-chain amino acids was ineffective to prevent muscle damage during a marathon. Amino Acids 2014, 46, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Pasiakos, S.M.; Lieberman, H.R.; McLellan, T.M. Effects of protein supplements on muscle damage, soreness and recovery of muscle function and physical performance: A systematic review. Sports Med. 2014, 44, 655–670. [Google Scholar] [CrossRef] [PubMed]
- Nosaka, K.; Sacco, P.; Mawatari, K. Effects of amino acid supplementation on muscle soreness and damage. Int. J. Sport Nutr. Exerc. Metab. 2006, 16, 620–635. [Google Scholar] [CrossRef] [PubMed]
| Category | Rating | Criteria | |
|---|---|---|---|
| Supplementation Strategy | Duration | Short | The supplementation was performed on 3 days or less |
| Moderate | The supplementation was performed between 4 and 10 days | ||
| Long | The supplementation was performed for more than 10 days | ||
| Frequency | Low | Less than 2 intakes per day during the supplementation period | |
| High | 2 or more intakes per day during the supplementation period | ||
| Amount | Low | Less than 200 mg kg−1 day−1 of BCAAs intake | |
| High | 200 mg kg−1 day−1 or more of BCAAs intake | ||
| Extent of Muscle Damage | Low | Low peak decrease in force (≤10% of baseline) and significant peak change in CK/LDH/myoglobin at D1 (with no significant difference in the following days) | |
| Moderate | Moderate peak decrease in force (≥10% and ≤15% of baseline) and significant peak change in CK/LDH/myoglobin at D1 (with significant difference in the following days) | ||
| High | High peak decrease in force (>15% of baseline) and significant peak change in CK/LDH/myoglobin after D2 | ||
| Study | Population | Study Design | Damaging Exercise | Supplementation Strategy | Outcomes | ||
|---|---|---|---|---|---|---|---|
| Soreness | Blood Analysis (Myoglobin/LDH/CK) | Muscle Performance | |||||
| Coombes & McNaughton (2000) [31] | 16 healthy males age: 21 ± 1 years V̇O2max: 52 ± 4 mL min−1 kg−1 training status: regular physical activity | CG (n = 8) Dietary control | Cycling ergometer exercise at 70% V̇O2max for 120 min | 14 days of supplementation (7 days before and 6 days after exercise). 2 × 6 g every day + 20 g before and after the exercise Amount of BCAA: 208 g (14 days) LEU/ISO/VAL (1:1:1) Placebo: no supplementation | - | CG > SG at H4, D1, D3 and D5 for CK and LDH | - |
| Fouré et al. (2016) [32] | 26 healthy males age: 22 ± 2 years training status: recreationally active | RCT, DB CG (n = 13) Dietary control | Neuromuscular Electrostimulation—40 isometric knee extensions | 5 days of supplementation (2 supp before, 1 supp after exercise and 1 supp every day for 4 days) Amount of BCAA: 48.3 g (5 days) LEU/ISO/VAL (2:1:1) Placebo: microcrystalline cellulose | CG = SG | SG > CG at D4 for CK | CG = SG for the MVC |
| Gee & Deniel (2016) [33] | 11 healthy males age: 25 ± 6 years training status: resistance-trained | RCT, SB Cross-over (washout: 7 days) | Strength exercises (back squat, press exercises, deadlift and barbell row) | Single day supplementation (1 supp before and 1 supp after exercise) Amount of BCAA: 20 g (1 day) LEU/ISO/VAL (2:1:1) Placebo: apple and blackcurrant juice | CG = SG | - | SG > CG at D1 for the CMJ and the SSPT |
| Greer et al. (2007) [34] | 9 healthy males age: 22 ± 3 years V̇O2max: 36 ± 2 mL min−1 kg−1 training status: untrained | Cross-over (washout: 8 days) Dietary control | Cycling ergometer exercise at 55% V̇O2max for 90 min | Single day supplementation (1 supp before and 1 supp at 60 min during the exercise) Amount of BCAA: 5 g (1 day) LEU/ISO/VAL (2.5:1:1.5) Placebo: water, lemon flavor, salts and artificial sweeteners. | CG > SG at D1 | CG > SG at H4, D1 and D2 for CK CG > SG at H4 for LDH | SG > CG at D2 for leg flexion torque (180°/s) |
| Howatson et al. (2012) [35] | 12 healthy males age: 23 ± 2 years training status: trained in collective sports (twice per week) | RCT, DB CG (n = 6) | Drop jumps (5 × 20, height: 60 cm) | 12 days of supplementation (7 days before and 4 days after exercise). 2 × 10 g every day + 20 g before and after the exercise Amount of BCAA: 280 g (12 days) LEU/ISO/VAL (2:1:1) Placebo: aspartame based artificial sweeteners. | CG > SG at D1 and D2 | CG > SG for CK (group effect considering the time range from D0 to D4) | SG > CG (group effect considering the time range from D0 to D4) for the MVC |
| Jackman et al. (2010) [36] | 24 healthy males age: n/a training status: n/a | SB CG (n = 12) Dietary control | Eccentric exercise (12 × 10 knee extensions, 120% of 1 RM) | 3 days of supplementation (1 supp before, 3 supp after exercise and 4 supp every day for 2 days) Amount of BCAA: 87.6g (3 days) LEU/ISO/VAL (2.1:1.2:1) Placebo: Artificially sweetened and flavored water | CG > SG with knee flexed at D2 and D3 | CG = SG for CK and myoglobin | CG = SG |
| Kephart et al. (2016) [37] | 30 healthy males age: 22 ± 1 years training status: resistance-trained | RCT CG (n = 15) Dietary control | 3 back squat exercises on three consecutive days (10 × 5 at 80% of 1 RM) | 4 days of supplementation (1 supp after the exercise on the first 3 days and 1 supp on day 4) Amount of BCAA: 24g (4 days) LEU/ISO/VAL (3:1:2) and CHO Placebo: CHO | CG = SG | CG = SG for myoglobin | CG = SG |
| Matsumoto et al. (2007) [38] | 12 healthy subjects (males: n = 6 and female: n = 6) age: 20 ± 1 years training status: trained in long distance running | RCT DB Cross-over (washout: 3 weeks) Dietary control | 7 sessions on 3 days of long distance runs | 3 days of supplementation (20g/day) Amount of BCAA: 60g (3 days) LEU/ISO/VAL (2:1:1) Placebo: n/a | CG > SG at D1 | CG > SG at D1 for CK, LDH and myoglobin | - |
| Ra et al. (2013) [39] | 18 healthy male subjects age: 23 ± 1 years training status: n/a | RCT, DB CG (n = 9) | Eccentric exercise (6 × 5 elbow flexions, 90% of MVC) | 18 days of supplementation (14 days before and 4 days after exercise). 3 × 3.2 g every day Amount of BCAA: 172.8g (18 days) LEU/ISO/VAL (2:1:1) Placebo: starch | CG = SG | CG = SG for CK and LDH | - |
| Shimomura et al. (2010) [40] | 12 healthy female subjects age: 22 ± 2 years training status: untrained | Cross-over (washout: 11 weeks) | Resistance exercise (7 × 20 squat with body weight) | Single day supplementation (1 supp before the exercise) Amount of BCAA: 5.5g (1 day) LEU/ISO/VAL (2.3:1:1.2) Placebo: dextrin | CG > SG at D2 and D3 | CG = SG for CK and myoglobin | SG > CG at D3 for MVC |
| Waldron et al. (2017) [41] | 16 healthy subjects (males: n = 14 and female: n = 2) age: 22 ± 2 years training status: trained in resistance exercise | RCT CG (n = 8) Dietary control | Strength exercise (10 × 6 back squats at 70% of 1 RM) | 3 days of supplementation (1 supp before, 1 supp after exercise and 2 supp every day for 2 days) Amount of BCAA: 48g (3 days) LEU/ISO/VAL (2:1:1) and dextrose Placebo: dextrose | CG = SG | SG > CG at D1 and D2 for CK | CG = SG for MVC and CMJ |
| References | Validity Rating | Overall Rating | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | ||
| Coombes & McNaughton (2000) [31] | Y | Y | Y | N | N | Y | Y | Y | Y | N | ø |
| Fouré et al. (2016) [32] | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | + |
| Gee & Deniel (2016) [33] | Y | Y | N | N | N | N | Y | Y | Y | Y | ø |
| Greer et al. (2007) [34] | Y | Y | N | N | N | N | Y | N | Y | N | – |
| Howatson et al. (2012) [35] | Y | Y | Y | Y | Y | Y | Y | N | Y | Y | + |
| Jackman et al. (2010) [36] | Y | Y | Y | N | N | Y | Y | Y | Y | Y | + |
| Kephart et al. (2016) [37] | Y | Y | Y | N | N | Y | Y | Y | Y | Y | + |
| Matsumoto et al. (2007) [38] | Y | N | N | N | Y | Y | Y | Y | Y | N | ø |
| Ra et al. (2013) [39] | Y | Y | Y | N | Y | Y | Y | Y | Y | Y | + |
| Shimomura et al. (2010) [40] | Y | Y | N | N | N | N | Y | N | Y | N | – |
| Waldron et al. (2017) [41] | Y | Y | Y | N | Y | Y | Y | N | Y | Y | + |
| Total | 11 | 10 | 6 | 2 | 5 | 8 | 11 | 7 | 11 | 7 | |
| References | Extent of Muscle Damage | Supplementation Strategy | ||
|---|---|---|---|---|
| Duration | Frequency | Amount | ||
| Fouré et al. (2016) [32] | High | Moderate | Low | Low |
| Ra et al. (2013) [39] | High | Long | High | Low |
| Jackman et al. (2010) [36] | Moderate | Short | High | High |
| Coombes & McNaughton (2000) [31] | Moderate | Long | High | High |
| Howatson et al. (2012) [35] | Moderate | Long | High | High |
| Greer et al. (2007) [34] | Low | Short | Low | Low |
| Shimomura et al. (2010) [40] | Low | Short | Low | Low |
| Gee & Deniel (2016) [33] | Low | Short | Low | High |
| Matsumoto et al. (2007) [38] | Low | Short | High | High |
| Waldron et al. (2017) [41] | Low | Short | High | High |
| Kephart et al. (2016) [37] | Low | Moderate | Low | Low |
| References | Effects in the Control Group | Positive Effect of Supplementation |
|---|---|---|
| Muscle performance | ||
| Fouré et al. (2016) [32] | Significant decrease in MVC from POST to D4 | - |
| Gee & Deniel (2016) [33] | Significant decrease in CMJ and SSPT performances at D1 | Yes |
| Greer et al. (2007) [34] | Significant decrease in torque (leg flexion and extension) from POST to D2 | Yes |
| Howatson et al. (2012) [35] | Significant decrease in MVC from D1 to D3 | Yes |
| Jackman et al. (2010) [36] | Significant decrease in maximal force from H1 to D3 | - |
| Kephart et al. (2016) [37] | Significant decrease in isokinetic peak torque | - |
| Shimomura et al. (2010) [40] | Significant decrease in MVC at D3 | Yes |
| Waldron et al. (2017) [41] | Decrease in MVC and CMJ performance from POST to D1 | - |
| Blood analyses | ||
| Coombes & McNaughton (2000) [31] | Significant increase in CK and LDH (from POST to D5) | Yes |
| Fouré et al. (2016) [32] | Significant increase in plasma CK activity at D3 and D4 | - |
| Greer et al. (2007) [34] | Significant increase in CK (from H4 to D2) and LDH (at H4) | Yes |
| Howatson et al. (2012) [35] | Significant increase in CK from D1 to D3 | Yes |
| Jackman et al. (2010) [36] | Significant increase in CK (from H8 to D3) and myoglobin (at H1, H8 and D3) | - |
| Kephart et al. (2016) [37] | Significant increase in myoglobin | - |
| Matsumoto et al. (2007) [38] | Significant increase in CK and LDH at POST | Yes |
| Ra et al. (2013) [39] | Significant increase in CK and LDH at D3 and D4 | - |
| Shimomura et al. (2010) [40] | No significant change in CK and LDH on the three days post-exercise | - |
| Waldron et al. (2017) [41] | No change in CK on the two days post-exercise | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fouré, A.; Bendahan, D. Is Branched-Chain Amino Acids Supplementation an Efficient Nutritional Strategy to Alleviate Skeletal Muscle Damage? A Systematic Review. Nutrients 2017, 9, 1047. https://doi.org/10.3390/nu9101047
Fouré A, Bendahan D. Is Branched-Chain Amino Acids Supplementation an Efficient Nutritional Strategy to Alleviate Skeletal Muscle Damage? A Systematic Review. Nutrients. 2017; 9(10):1047. https://doi.org/10.3390/nu9101047
Chicago/Turabian StyleFouré, Alexandre, and David Bendahan. 2017. "Is Branched-Chain Amino Acids Supplementation an Efficient Nutritional Strategy to Alleviate Skeletal Muscle Damage? A Systematic Review" Nutrients 9, no. 10: 1047. https://doi.org/10.3390/nu9101047





