1. Introduction
The relationship between nutrition and reproductive health is an active area of research at present [
1]. In the Western diet, high fat intake causes metabolic syndromes, including obesity, insulin resistance, type 2 diabetes, hypertension and cardiovascular diseases. Mothers still require a high fat diet in order to ensure a supply of sufficient nutrients to maintain a normal fetal growth trajectory. It has been shown that the offspring of women on a hyperphagia diet that aims to increase fetal birth weight [
2] may be at risk of reproductive failure [
3]. Meanwhile, evidence in pigs has shown that the reproductive system is tightly regulated by the state of energy reserves [
4]. It has been reported that in the female reproductive system, the ovary is a highly sensitive organ [
5], and the risk of defective luteal function or reduced follicle numbers is increased by inappropriate nutritional regimens [
4].
Reproductive health is dependent on the ovarian immune response in humans. The link between oxidative stress and reproductive performance has been well established, with severe oxidative stress rapidly and effectively impairing ovarian development in pigs [
6]. Notably, there is an increase in the defective cohort of oocytes in cycling gilts during feeding at high nutritional levels compared to those fed at maintenance levels [
7]. Fetal immune function is easily disrupted because of an immature antioxidant defense system. Therefore, it is necessary to investigate the link between high fat nutrition and the fetal immature antioxidant defense system, as well as to study their influence on the reproduction health of offspring.
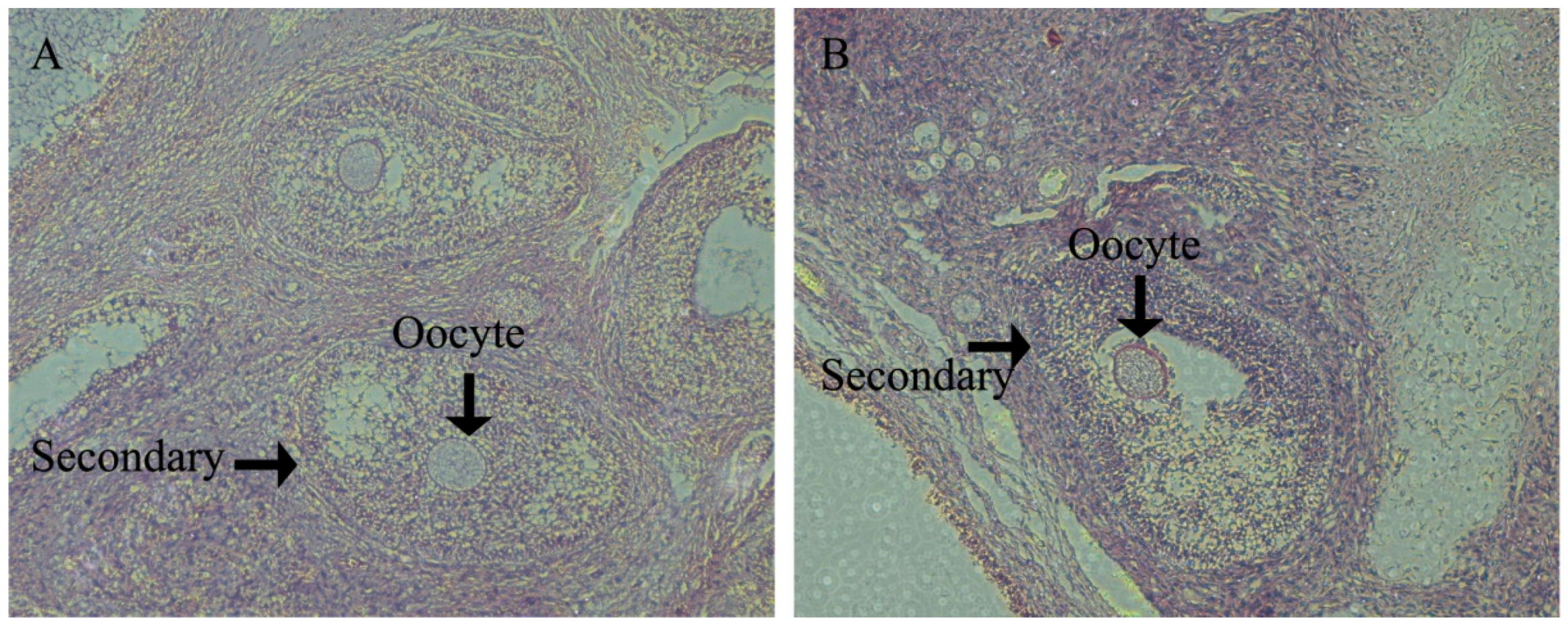
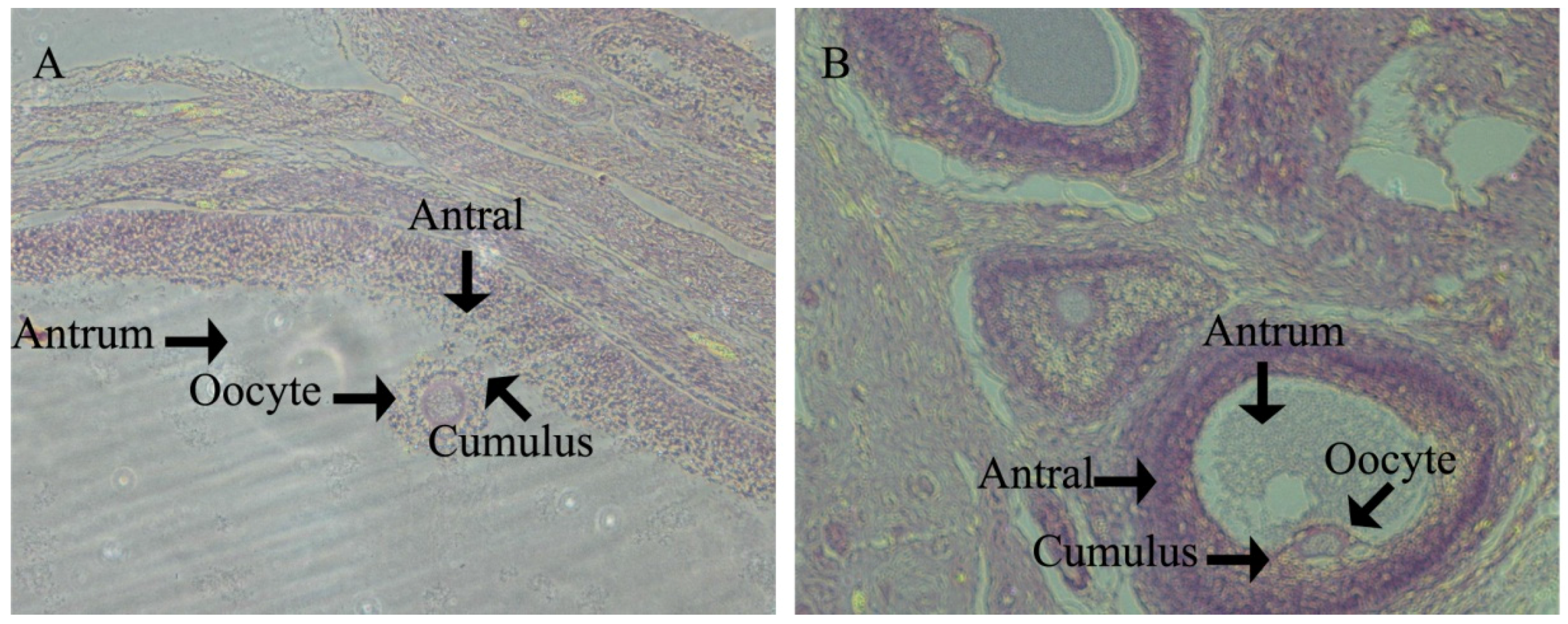
In porcine ovaries, follicles begin to form at approximately Day 55 of gestation. However, the primary follicle was first detected in pigs at Day 90 of gestation and is rapidly followed by the initiation of follicle growth, such that all stages of pre-antral follicles and antral follicles have been found by age at puberty. All stages of follicles play an important role in ovulation. The antioxidant defense system results in the health progression of all stages of follicle growth and the timing of the onset of ovulation. However, there are few data relating to health changes in the ovary between follicle formation and pubertal ovulation.
The pig is a critical model to investigate the influence of nutrition on ovarian development, because it has biological similarities to human [
8]. Therefore, in this study, we examined the effects of nutritionally-induced oxidative stress on follicle numbers, antioxidant-related gene expression, enzymatic activity and cell apoptosis in the ovary. These changes might shed light on the mechanisms mediating the effect of nutrition on reproductive health.
4. Discussion
Disorders of the reproductive system are a major health issue worldwide in both humans and domestic animals. A few studies have suggested that maternal dietary intake may regulate reproductive outcomes [
10,
11], at least in part by ovarian oxidative status and cell apoptosis [
12]. An appropriate balance of free radicals is critical for the optimal development of the ovary, as free radicals have both beneficial and harmful effects [
13]. Meanwhile, fetal ovarian structure, physiology and metabolism may change depending on maternal dietary intake and, therefore, lead to vulnerability to reproductive disorders in adults [
14]. In order to explore the molecular mechanisms mediating the long-term effect of maternal dietary intake on the reproductive health in their offspring, the effect of nutrient intake, including oxidative stress and apoptosis, was examined in this study.
Strong evidence from animal studies has shown that dietary manipulations can result in changes in reproductive function [
11]. Experimental studies in humans demonstrated that high fat maternal dietary intake during pregnancy may promote a greater neonatal weight [
15]. Previous data from our laboratory supports earlier findings that a maternal diet with added soybean oil to increase fat levels achieves a higher neonatal weight [
10]. However, in this study, the effect of fat intake does not alter female fetal weight or offspring weight. One reason for this may be that we only selected female fetuses. Fetuses receive constant nutrients from mothers via the placenta; therefore, the mother’s body condition and glucose levels during the gestation period play an important role in the growth of fetuses. Maternal fat intake level not only helps to maintain the pregnancy, but also affects the growth of fetuses. Previous data from our laboratory showed that gilts with increased fat intake of 62% resulted in a significant increase of maternal body weight (201.65 ± 3.60 vs. 188.30 ± 1.82,
p = 0.035) and BF (18.65 ± 1.20 vs. 16.89 ± 0.96,
p = 0.005) at g90. Meanwhile, the results showed that a high fat level led to significantly increased maternal glucose level (+49%) and fetuses glucose level (+121%) compared to the CON group at g90. Although maternal glucose levels increase 49%, which would be considered in the pre-diabetic/diabetic range in humans, through self-regulation of glucose levels in gilts, these returned to normal levels after delivery. Furthermore, there is little information in pigs regarding what blood glucose levels might be defined as diabetic. Experimental studies in mice demonstrated upregulation of glucose transport in the placenta, thus potentially linking maternal high fat diet and fetus glucose level [
16,
17]. Previous unpublished data (2016) from our laboratory showed that gilts with a high fat diet (increased 62% compared to control) increased placental nutrient transport, which led to an increased fetus glucose level [
18]. The neonatal (one day) glucose level had increased in the HFD compared to CON (+66%). Therefore, it is possible that maternal fat level may affect the maternal condition and glucose level of fetuses. In this study, the effect of fat intake does not alter female fetal weight and offspring weight. Another reason for this may be that maternal fat intake had a significant influence on reproductive organs instead of body weight. Consistent with this view, it was demonstrated that the effect of maternal fat intake does not alter the glucose level of female offspring at d160 (4.91 ± 0.38 vs. 4.83 ± 0.39) and upon puberty (4.10 ± 0.35 vs. 4.68 ± 0.43) stages.
In swine development, Yorkshire gilts will see the onset of their estrus cycle at Day 180. Therefore, we investigated ovary development at d160 in order to assess the gilts’ reproductive health and to analyze how the follicles within the ovaries of prepubertal gilts appear to form the follicle reserves for future reproductive life. We found that at d160 in the HFD group, gilts had significantly lower large follicle numbers than CON gilts. Meanwhile, HFD group gilts had significantly small follicle numbers upon puberty, which is essential for fertility preservation for adult female animals and beneficial to ovulation. This suggests that ovarian growth potential established prenatally may influence the development of adult reproductive function [
19], and maternal fat levels may lead to ovary change. To further confirm that maternal fat level has adverse effects on reproductive function, BF was determined to examine the ponderal index, and we found that it significantly increased in the HFD group compared to the CON group at puberty. This finding suggested that maternal diet affects body index in the offspring. This is consistent with the observation that high BF gilts reach puberty earlier than low BF gilts. Optimum BF was 15–18 mm [
20]. If BF exceeds this optimum, it will cause a decline in feed intake during lactation, which will affect the health of the offspring. Moreover, antioxidant defense system damage and an increase in free radicals caused by high BF will lead to feed intake being reduced [
21]. Meanwhile, clinical studies have indicated that women with polycystic ovaries tend to have high body weight, and patterns of hormone release in offspring depend on maternal body mass index in pregnancy [
14]. In the present study, we found that BF was 19.6 mm in the CON group, which was nearly the optimum BF. In swine development, gilts are usually kept under nutrient restriction to achieve optimum BF of both the gilt/offspring [
22]. As the present study was aimed at providing guidance to humans, both the offspring and gilts were fed ad libitum, and this may explain why offspring BF was greater than the optimum BF. This implied that gilts in the CON group will provide a better maternal environment for fetal ovary development and indicated that maternal high nutrient intake would influence the function of ovaries in later adult life. These adverse effects of maternal high nutrient intake are likely to be associated with ovary formation and intrauterine development.
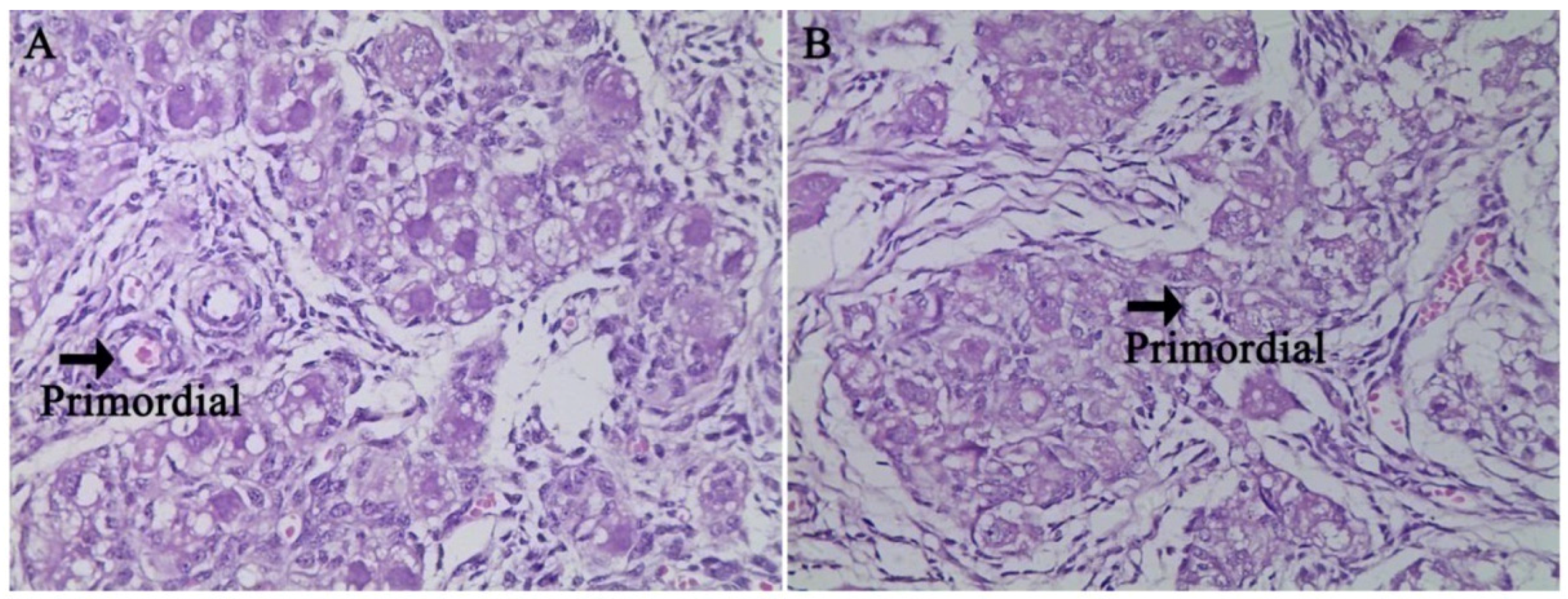
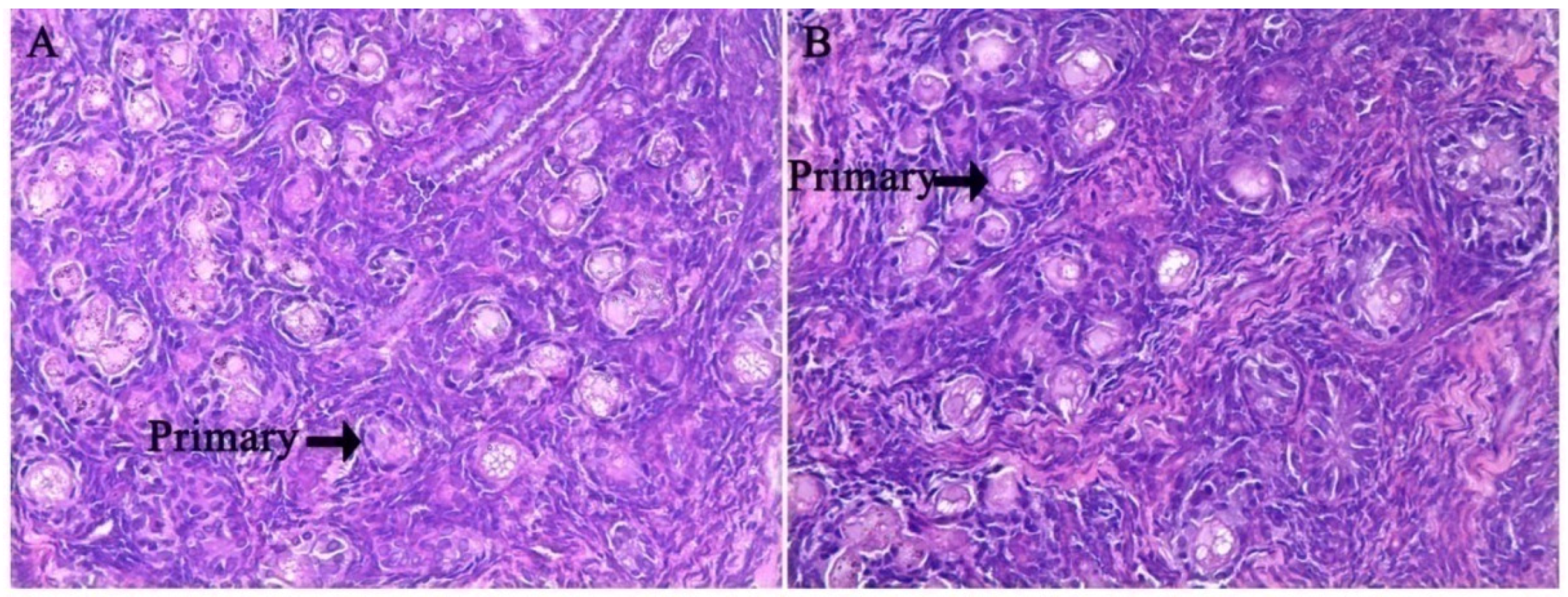
In the fetal ovaries of pigs, germ cells migrate to the gonad and remain in clusters [
23]. The clusters undergo apoptosis and break up, becoming enclosed in primordial follicles consisting of one oocyte and several somatic granulosa cells and transforming into the primary follicle [
24]. These are critical processes in ovarian biology [
25]. Therefore, in the present study, we observed two critical periods during the formation and transformation of primordial follicles. Early studies found that Notch signaling mediates primordial follicle formation and transformation, particularly involving the SOHLH2, NOBOX and LHX8 transcription factors [
26,
27]. Early studies found that downregulation of mRNA expression of
sohlh2,
nobox and
lhx8 alters primordial follicle numbers and results in a lifelong reduction in the primordial follicle pool [
26]. Interestingly, in the present study, we did not observe a difference in the gene expression level of
sohlh2,
nobox and
lhx8 in the fetal ovary. Meanwhile, the histological characteristics in the ovary of fetus showed that primordial follicles and primary follicles were not significantly different in the fetus ovary at g55 or g90 between the two groups. Previous studies have shown that
sohlh2,
nobox and
lhx8 mRNA expression and proteins are relatively concordant in ovary [
26,
28]. Thus, the results indicate that maternal nutrition during pregnancy might not delay fetal ovarian development.
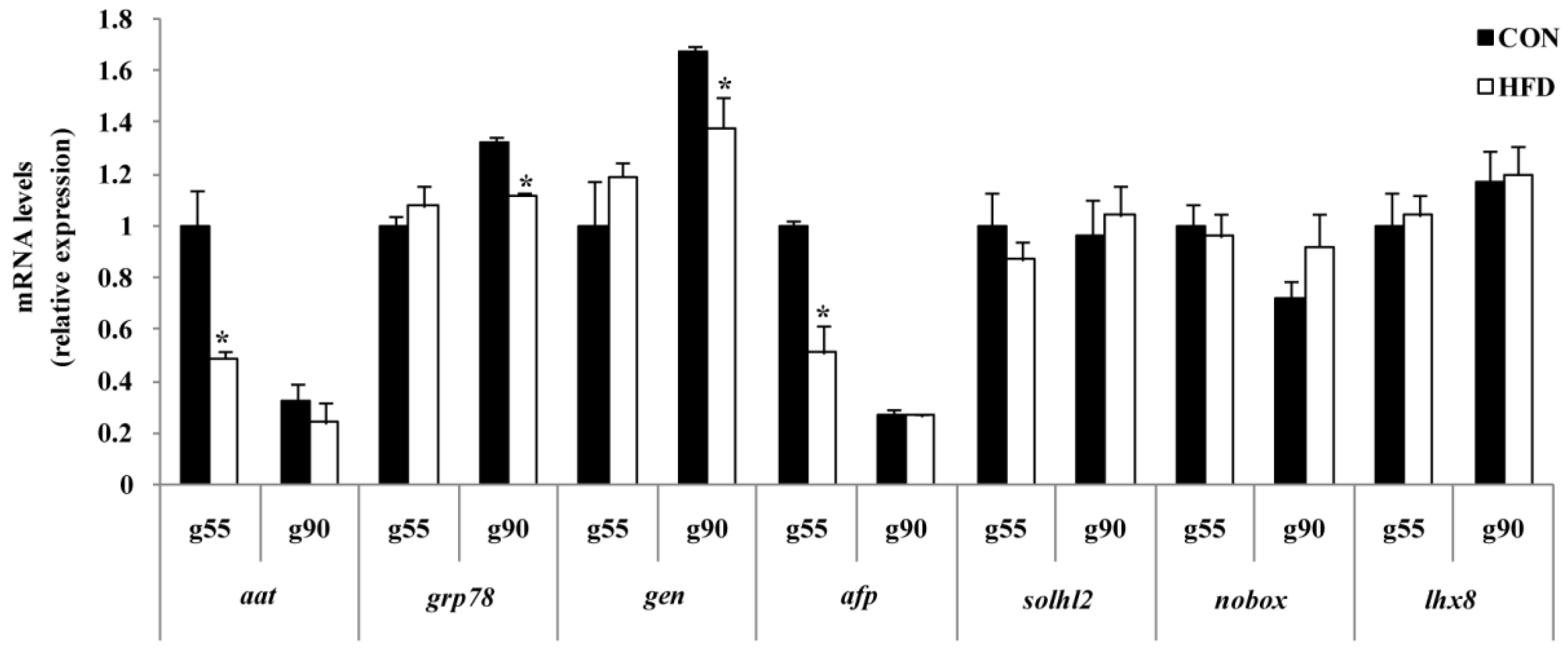
To further confirm whether maternal nutrition affects ovary health and homeostasis, the levels of
aat,
afp,
grp78 and
gen were determined. AAT is a protease inhibitor that is supposed to finely regulate the balance of protease activities and is a biomarker of primordial follicle health [
29]. AFP is essential for immune response in ovaries [
30]. GRP78 is a molecular chaperone that facilitates protein assembly, folding and regulation of endoplasmic reticulum (ER) stress signaling [
31,
32,
33], and GEN is known to be a cell motility factor [
34]. In the present study, mRNA expressions of
aat and
afp at g55 and
grp78 and
gen at g90 were decreased in the HFD group compared to the CON group. Early studies also found that these homeostasis-related genes’ mRNA expression is relatively concordant in ovary with proteins [
35,
36]. Our previous study has confirmed that
aat and
afp might promote primordial follicles formation, and
grp78 and
gen could protect primordial follicles from transforming to primary follicles [
37]. These results demonstrate that the impaired expression of genes regulating homeostasis (
aat,
afp,
grp78 and
gen) in the ovary could be modulated by high fat diets. Although there is no effect of maternal nutrition on ovarian development progress, it may damage its capacity to resist oxidative stress. The findings also indicated that maternal homeostasis reflects the offspring’s health trajectory of growth, and high nutrition levels will result in cell damage and premature follicle pool failure.
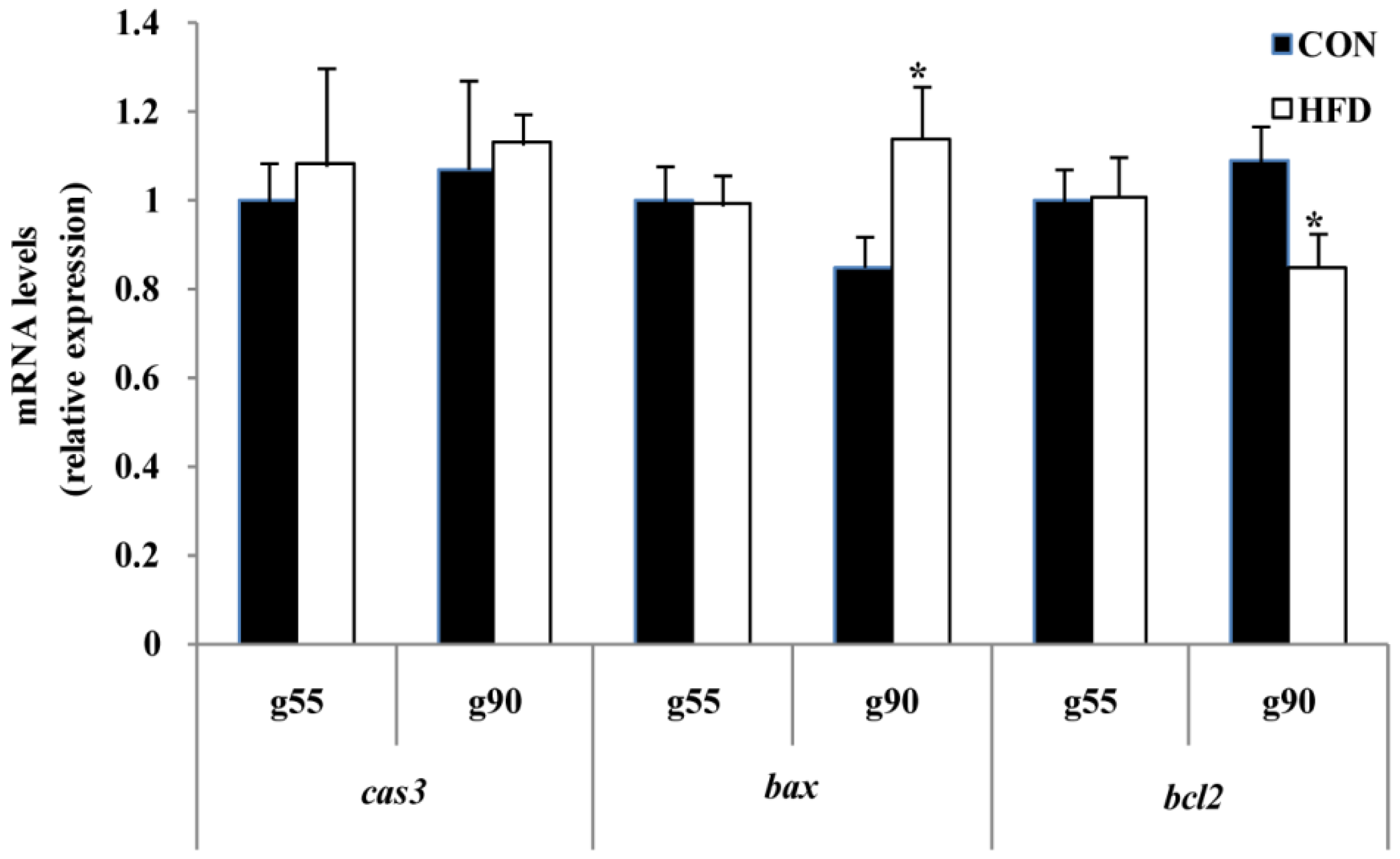
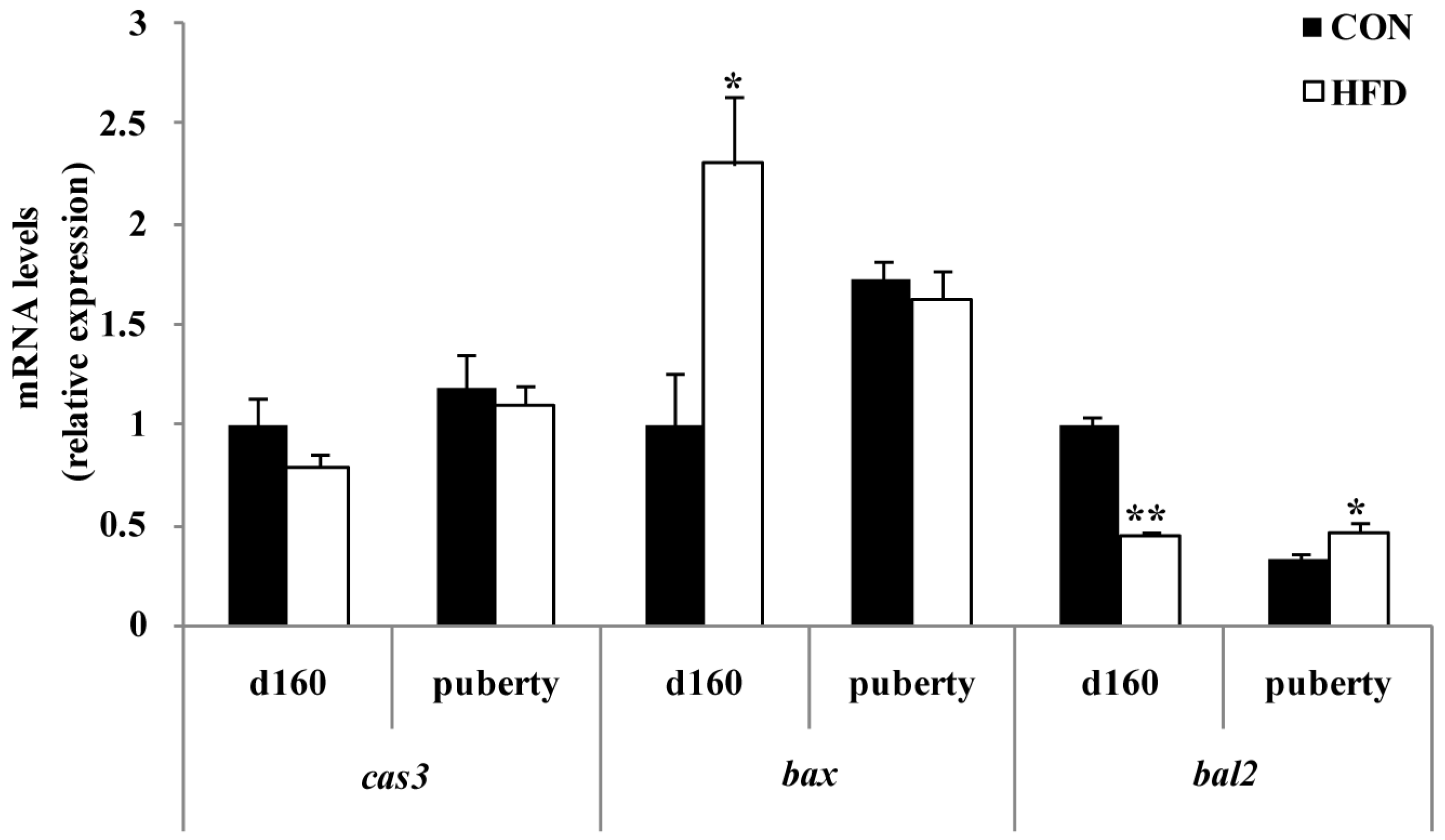
To further clarify the effect of maternal fat nutrition on premature follicle pool failure, we examined the expression of factors that regulate cell survival. BCL2 is known to be an anti-apoptotic factor and is important in the survival of ovarian cells [
38]. BCL2 can inhibit the formation of lipid peroxides and prevent the production of free radicals [
39]. High expression of BCL2 resulted in either higher ovulation rate or litter size [
40], and deletion of BCL2 led to complete blockade of follicular oocyte development [
41]. BAX opposes BCL2 function and is a pro-apoptotic protein [
42]. Previous studies reported that BAX is expressed in both fetal and adult ovaries [
43], and BAX expression is increased in atretic follicles compared to healthy ones [
44]. In our study, we found that the level of bax was dramatically higher in ovary cells in the HFD group at g90 with BCL2 showing the opposite trend. This further indicated that high fat nutrition accelerates follicle pool exhaustion, which may reduce sow reproduction longevity. To date, many studies have indicated that maternal and placental systems supply sufficient nutrients to ensure fetal growth [
16]. Thus, further experiments should include testing of offspring ovary apoptosis-related genes to determine whether maternal nutrition is truly regulating their offspring’s reproductive health throughout the pregnancy period. To investigate this, we examined mRNA expression of
bcl2 and
bax in the developing ovaries of offspring. We found that the expression of
bcl2 decreased and
Bax increased significantly with increasing dietary energy levels of the offspring at d160. Interestingly, we also found that
bcl2 significantly increased in the HFD group, but
bax expression level did not change for age at puberty. This may be due to a temporal effect of maternal fat level on cell survival. Although related protein expression levels were not measured in the present study, previous studies found that
bcl2 and
bax mRNA expression and protein expression levels are relatively concordant in ovary [
45]. The combination of these results may demonstrate a role of maternal fat nutrition in the regulation of apoptotic signaling pathways and mediating the long-term effects on offspring ovary development.
Epidemiologic studies suggested that fetuses that were exposed to an adverse intrauterine environment tended to have relatively high disease rates in adult life [
16]. Meanwhile, there was the finding that the long-term effects of adverse free radical imbalance were based on the program of following up with animals in the middle of life whose intrauterine health index measurements had been previously recorded [
8]. The imbalance of free radicals could reduce the capability of antioxidant systems, leading to oxidative stress, which can impair organism structure, metabolism and physiology, thereby predisposing individuals to abnormal functionality [
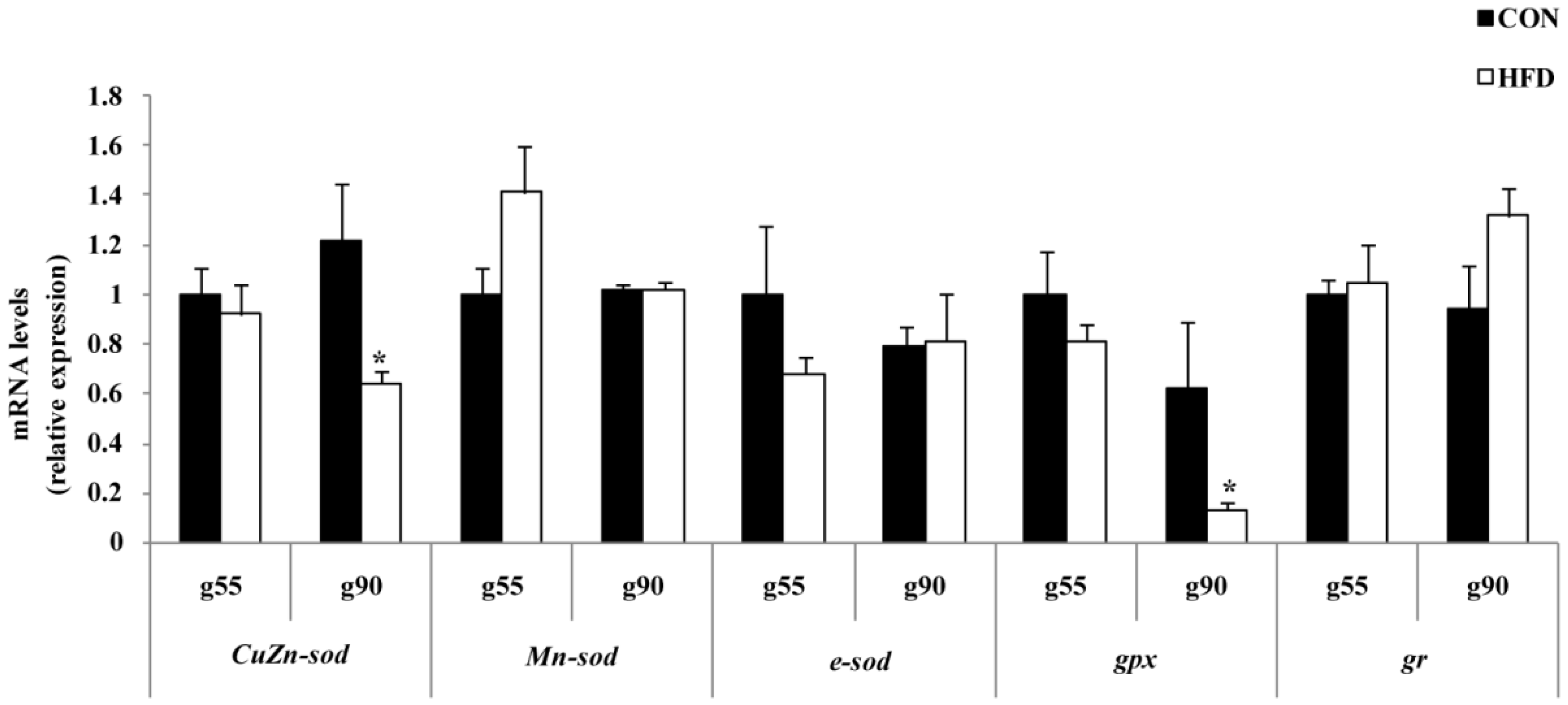
16]. In this study, a high fat maternal nutrient supply would satisfy fetal nutrient requirements, but results in a range of fetal maladaptations and may deteriorate the body’s antioxidant defense system. The increased concentration of MDA was supportive of the reduced T-SOD, CuZn-SOD, GPx and GR enzyme activity and gene expression of
CuZn-sod and
gpx in the ovary of the HFD group at g90. Our results showed that the mRNA expression and protein expression levels of oxidative stress-related genes are relatively concordant in swine ovary. With plenty of catabolism, the fetus will experience rapid growth in the first Day 90 of pregnancy, leading to an increase in free radicals. Then, maternal fat level affects the related enzyme activity at g90, but no significant influenced is seen at g55. Interestingly, we found that in the HFD group, T-AOC activity was only reduced upon the offspring reaching puberty compared to the CON group of the offspring. It is possible that maternal fat level may lead to a lifelong failure in the offspring’s antioxidant system. However, further ovarian apoptosis of offspring by immunohistochemistry and offspring fertility merit investigation.
A high fat diet does not alter fetal weight and may induce oxidative stress, as well as accelerate cell apoptosis, which can have adverse effects on the reproductive health of offspring due to their damaged ovaries.