Protecting Skeletal Muscle with Protein and Amino Acid during Periods of Disuse
Abstract
:1. Introduction
2. Protecting Muscle Health through Dietary Manipulation
3. Protein and Targeted Amino Acid Interventions during Inactivity
4. Translating Acute Research Studies: Concept to Practice?
5. Cellular Mechanisms of Inactivity-Induced Muscle Loss
6. Inactivity and Glucose Regulation
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| 4E-BP1 | Eukaryotic Translation Initiation Factor 4E-Binding Protein 1 |
| Akt/PKB | Protein Kinase B |
| DXA | Dual Energy X-ray Absorptiometry |
| EAR | Estimated Average Requirement |
| FOXO | Forkhead box |
| GLUT4 | Glucose Transporter 4 |
| HMB | Beta-Hydroxy-beta-Methylbutyrate |
| ISGD | Insulin-Stimulated Glucose Disposal |
| IOM | Institute of Medicine |
| LAT1 | l-type Amino Acid Transporter 1 |
| MAFbx | Muscle Atrophy F-Box |
| MAP4K3 | Mitogen-Activated Protein Kinase Kinase Kinase Kinase 3 |
| miRNA | micro RNA |
| mTORC1 | Mammalian Target of Rapamycin Complex 1 |
| MuRF | Muscle Really Interesting New Gene (RING)-Finger |
| p70-S6K | Ribosomal Protein S6 Kinase beta-1 |
| RDA | Recommended Dietary Allowance |
| rpS6 | Ribosomal Protein S6 |
| SNAT2 | Sodium-coupled Neutral Amino Acid Transporter |
| Vps34 | Vacuolar Protein Sorting 34 |
References
- Cermak, N.M.; Res, P.T.; de Groot, L.C.P.G.M.; Saris, W.H.M.; van Loon, L.J.C. Protein supplementation augments the adaptive response of skeletal muscle to resistance-type exercise training: A meta-analysis. Am. J. Clin. Nutr. 2012, 96, 1454–1464. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.N.; Choi, K.M. Sarcopenia: Definition, epidemiology, and pathophysiology. J. Bone Metab. 2013, 20, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Grimby, G.; Saltin, B. The ageing muscle. Clin. Physiol. 1983, 3, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Fazelzadeh, P.; Hangelbroek, R.; Tieland, M.; de Groot, L.; Verdijk, L.; van Loon, L.; Smilde, A.; Alves, R.; Vervoort, J.; Muller, M.; et al. The muscle metabolome differs between healthy and frail older adults. J. Proteome. Res. 2016, 15, 499–509. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wall, B.T.; van Loon, L.J.C. Nutritional strategies to attenuate muscle disuse atrophy. Nutr. Rev. 2013, 71, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.C.; Schneider, S.M.; Feiveson, A.H.; Macias, B.R.; Smith, S.M.; Watenpaugh, D.E.; Hargens, A.R. Wise-2005: Countermeasures to prevent muscle deconditioning during bed rest in women. J. Appl. Physiol. 2014, 116, 654–667. [Google Scholar] [CrossRef] [PubMed]
- English, K.L.; Mettler, J.A.; Ellison, J.B.; Mamerow, M.M.; Arentson-Lantz, E.; Pattarini, J.M.; Ploutz-Snyder, R.; Sheffield-Moore, M.; Paddon-Jones, D. Leucine partially protects muscle mass and function during bed rest in middle-aged adults. Am. J. Clin. Nutr. 2016, 103, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Vigelso, A.; Gram, M.; Dybboe, R.; Kuhlman, A.; Prats, C.; Greenhaff, P.; Constantin-Teodosiu, D.; Birk, J.; Wojtaszewski, J.; Dela, F.; et al. The effect of age and unilateral leg immobilization for 2 weeks on substrate utilization during moderate-intensity exercise in human skeletal muscle. J. Physiol. 2016, 594, 1–20. [Google Scholar] [CrossRef] [PubMed]
- De Boer, M.D.; Maganaris, C.N.; Seynnes, O.R.; Rennie, M.J.; Narici, M.V. Time course of muscular, neural and tendinous adaptations to 23 day unilateral lower-limb suspension in young men. J. Physiol. Lond. 2007, 583, 1079–1091. [Google Scholar] [CrossRef] [PubMed]
- Tanner, R.E.; Brunker, L.B.; Agergaard, J.; Barrows, K.M.; Briggs, R.A.; Kwon, O.S.; Young, L.M.; Hopkins, P.N.; Volpi, E.; Marcus, R.L.; et al. Age-related differences in lean mass, protein synthesis and skeletal muscle markers of proteolysis after bed rest and exercise rehabilitation. J. Physiol. Lond. 2015, 593, 4259–4273. [Google Scholar] [CrossRef] [PubMed]
- Pisot, R.; Marusic, U.; Biolo, G.; Mazzucco, S.; Lazzer, S.; Grassi, B.; Reggiani, C.; Toniolo, L.; di Prampero, P.E.; Passaro, A.; et al. Greater loss in muscle mass and function but smaller metabolic alterations in older compared to younger men following two weeks of bed rest and recovery. J. Appl. Physiol. 2016. [Google Scholar] [CrossRef]
- Paddon-Jones, D.; Campbell, W.W.; Jacques, P.F.; Kritchevsky, S.B.; Moore, L.L.; Rodriguez, N.R.; van Loon, L.J.C. Protein and healthy aging. Am. J. Clin. Nutr. 2015, 101, 1339S–1345S. [Google Scholar] [CrossRef] [PubMed]
- Campbell, B.; Kreider, R.B.; Ziegenfuss, T.; La Bounty, P.; Roberts, M.; Burke, D.; Landis, J.; Lopez, H.; Antonio, J. International society of sports nutrition position stand: Protein and exercise. J. Int. Soc. Sports Nutr. 2007. [Google Scholar] [CrossRef] [PubMed]
- Wu, G. Dietary protein intake and human health. Food Funct. 2016, 129, 758S–767S. [Google Scholar] [CrossRef] [PubMed]
- Antonio, J.; Peacock, C.A.; Ellerbroek, A.; Fromhoff, B.; Silver, T. The effects of consuming a high protein diet (4.4 g/kg/day) on body composition in resistance-trained individuals. J. Int. Soc. Sports Nutr. 2012. [Google Scholar] [CrossRef]
- Otten, J.; Hellwig, J.; Meyers, L. Dietary Reference Intakes: The Essential Guide to Nutrient Requirements; The National Academies Press: Washington, DC, USA, 2006. [Google Scholar]
- Wolfe, R.R.; Miller, S.L. The recommended dietary allowance of protein: A misunderstood concept. J. Am. Med. Assoc. 2008, 299, 2891–2893. [Google Scholar] [CrossRef] [PubMed]
- Paddon-Jones, D.; Sheffield-Moore, M.; Cree, M.G.; Hewlings, S.J.; Aarsland, A.; Wolfe, R.R.; Ferrando, A.A. Atrophy and impaired muscle protein synthesis during prolonged inactivity and stress. J. Clin. Endocr. Metab. 2006, 91, 4836–4841. [Google Scholar] [CrossRef] [PubMed]
- Paddon-Jones, D.; Sheffield-Moore, M.; Urban, R.J.; Sanford, A.P.; Aarsland, A.; Wolfe, R.R.; Ferrando, A.A. Essential amino acid and carbohydrate supplementation ameliorates muscle protein loss in humans during 28 days bedrest. J. Clin. Endocr. Metab. 2004, 89, 4351–4358. [Google Scholar] [CrossRef] [PubMed]
- Drummond, M.J.; Dickinson, J.M.; Fry, C.S.; Walker, D.K.; Gundermann, D.M.; Reidy, P.T.; Timmerman, K.L.; Markofski, M.M.; Paddon-Jones, D.; Rasmussen, B.B.; et al. Bed rest impairs skeletal muscle amino acid transporter expression, mtorc1 signaling, and protein synthesis in response to essential amino acids in older adults. Am. J. Physiol. Endoc. Metab. 2012, 302, E1113–E1122. [Google Scholar] [CrossRef] [PubMed]
- Ferrando, A.A.; Paddon-Jones, D.; Hays, N.P.; Kortebein, P.; Ronsen, O.; Williams, R.H.; McComb, A.; Symons, T.B.; Wolfe, R.R.; Evans, W. Eaa supplementation to increase nitrogen intake improves muscle function during bed rest in the elderly. Clin. Nutr. 2010, 29, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Symons, T.B.; Sheffield-Moore, M.; Chinkes, D.L.; Ferrando, A.A.; Paddon-Jones, D. Artificial gravity maintains skeletal muscle protein synthesis during 21 days of simulated microgravity. J. Appl. Physiol. 2009, 107, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Arentson-Lantz, E.; English, K.L.; Paddon-Jones, D.; Fry, C.S. 14 days of bed rest induces a decline in satellite cell content and robust atrophy of skeletal muscle fibers in middle-aged adults. J. Appl. Physiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- English, K.L.; Paddon-Jones, D. Protecting muscle mass and function in older adults during bed rest. Curr. Opin. Clin. Nutr. 2010, 13, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Paddon-Jones, D.; Rasmussen, B.B. Dietary protein recommendations and the prevention of sarcopenia. Curr. Opin. Clin. Nutr. 2009, 12, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Casperson, S.L.; Sheffield-Moore, M.; Hewlings, S.J.; Paddon-Jones, D. Leucine supplementation chronically improves muscle protein synthesis in older adults consuming the rda for protein. Clin. Nutr. 2012, 31, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Churchward-Venne, T.A.; Burd, N.A.; Mitchell, C.J.; West, D.W.D.; Philp, A.; Marcotte, G.R.; Baker, S.K.; Baar, K.; Phillips, S.M. Supplementation of a suboptimal protein dose with leucine or essential amino acids: Effects on myofibrillar protein synthesis at rest and following resistance exercise in men. J. Physiol. 2012, 590, 2751–2765. [Google Scholar] [CrossRef] [PubMed]
- Churchward-Venne, T.A.; Breen, L.; Di Donato, D.M.; Hector, A.J.; Mitchell, C.J.; Moore, D.R.; Stellingwerff, T.; Breuille, D.; Offord, E.A.; Baker, S.K.; et al. Leucine supplementation of a low-protein mixed macronutrient beverage enhances myofibrillar protein synthesis in young men: A double-blind, randomized trial. Am. J. Clin. Nutr. 2014, 99, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Symons, T.B.; Sheffield-Moore, M.; Wolfe, R.R.; Paddon-Jones, D. A moderate serving of high-quality protein maximally stimulates skeletal muscle protein synthesis in young and elderly subjects. J. Am. Diet Assoc. 2009, 109, 1582–1586. [Google Scholar] [CrossRef] [PubMed]
- Deutz, N.; Pereira, S.; Hays, N.; Oliver, J.; Edens, C.; Wolfe, R. Effect of beta-hydroxy-beta-methylbutyrate (hmb) on lean body mass during 10 days of bed rest in older adults. Clin. Nutr. 2013. [Google Scholar] [CrossRef] [PubMed]
- Mamerow, M.M.; Mettler, J.A.; English, K.L.; Casperson, S.L.; Arentson-Lantz, E.; Sheffield-Moore, M.; Layman, D.K.; Paddon-Jones, D. Dietary protein distribution positively influences 24-h muscle protein synthesis in healthy adults. J. Nutr. 2014, 144, 876–880. [Google Scholar] [CrossRef] [PubMed]
- Donini, L.M.; Savina, C.; Cannella, C. Eating habits and appetite control in the elderly: The anorexia of aging. Int. Psychogeriatr. 2003, 15, 73–87. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Morley, J.E. Sarcopenia, 1st ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2012. [Google Scholar]
- Baim, S.; Wilson, C.R.; Lewiecki, E.M.; Luckey, M.M.; Downs, R.W.; Lentle, B.C. Precision assessment and radiation safety for dual-energy x-ray absorptiometry. J. Clin. Densitom. 2005, 8, 371–378. [Google Scholar] [CrossRef]
- Coker, R.H.; Miller, S.; Schutzler, S.; Deutz, N.; Wolfe, R.R. Whey protein and essential amino acids promote the reduction of adipose tissue and increased muscle protein synthesis during caloric restriction-induced weight loss in elderly, obese individuals. J. Nutr. 2012. [Google Scholar] [CrossRef] [PubMed]
- Katsanos, C.S.; Chinkes, D.L.; Paddon-Jones, D.; Zhang, X.-J.; Aarsland, A.; Wolfe, R.R. Whey protein ingestion in elderly persons results in greater muscle protein accrual than ingestion of its constituent essential amino acid content. Nutr. Res. 2008, 28, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Paddon-Jones, D.; Sheffield-Moore, M.; Katsanos, C.S.; Zhang, X.J.; Wolfe, R.R. Differential stimulation of muscle protein synthesis in elderly humans following isocaloric ingestion of amino acids or whey protein. Exp. Gerontol. 2006, 41, 215–219. [Google Scholar] [CrossRef] [PubMed]
- Katsanos, C.S.; Kobayashi, H.; Sheffield-Moore, M.; Aarsland, A.; Wolfe, R.R. A high proportion of leucine is required for optimal stimulation of the rate of muscle protein synthesis by essential amino acids in the elderly. Am. J. Physiol. Endoc. Metab. 2006, 291, E381–E387. [Google Scholar] [CrossRef] [PubMed]
- United States Department of Agriculture, Agricultural Research Service. National Nutrient Database for Standard Reference Release 28. Available online: http://www.webcitation.org/6iC9iFuxh (accessed on 9 June 2016).
- Phillips, S.M.; Tipton, K.D.; van Loon, L.J.; Verdijk, L.B.; Paddon-Jones, D.; Close, G.L. Exceptional body composition changes attributed to collagen peptide supplementation and resistance training in older sarcopenic men. Br. J. Nutr. 2016, 6, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Babault, N.; Paizis, C.; Deley, G.; Guerin-Deremaux, L.; Saniez, M.-H.; Lefranc-Millot, C.; Allaert, F.A. Pea proteins oral supplementation promotes muscle thickness gains during resistance training: A double-blind, randomized, placebo-controlled clinical trial vs. Whey protein. J. Int. Soc. Sports Nutr. 2015, 12. [Google Scholar] [CrossRef]
- Tang, J.E.; Moore, D.R.; Kujbida, G.W.; Tarnopolsky, M.A.; Phillips, S.M. Ingestion of whey hydrolysate, casein, or soy protein isolate: Effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. J. Appl. Physiol. 2009, 107, 987–992. [Google Scholar] [CrossRef] [PubMed]
- Ham, D.J.; Caldow, M.K.; Lynch, G.S.; Koopman, R. Leucine as a treatment for muscle wasting: A critical review. Clin. Nutr. 2014, 33, 937–945. [Google Scholar] [CrossRef] [PubMed]
- Leenders, M.; Verdijk, L.B.; van der Hoeven, L.; van Kranenburg, J.; Hartgens, F.; Wodzig, W.K.W.H.; Saris, W.H.M.; van Loon, L.J.C. Prolonged leucine supplementation does not augment muscle mass or affect glycemic control in elderly type 2 diabetic men. J. Nutr. 2011, 141, 1070–1076. [Google Scholar] [CrossRef] [PubMed]
- Verhoeven, S.; Vanschoonbeek, K.; Verdijk, L.B.; Koopman, R.; Wodzig, W.K.W.H.; Dendale, P.; van Loon, L.J.C. Long-term leucine supplementation does not increase muscle mass or strength in healthy elderly men. Am. J. Clin. Nutr. 2009, 89, 1468–1475. [Google Scholar] [CrossRef] [PubMed]
- Leger, B.; Cartoni, R.; Praz, M.; Lamon, S.; Deriaz, O.; Crettenand, A.; Gobelet, C.; Rohmer, P.; Konzelmann, M.; Luthi, F.; et al. Akt signalling through gsk-3 beta, mtor and foxo1 is involved in human skeletal muscle hypertrophy and atrophy. J. Physiol. Lond. 2006, 576, 923–933. [Google Scholar] [CrossRef] [PubMed]
- Sandri, M.; Barberi, L.; Bijlsma, A.Y.; Blaauw, B.; Dyar, K.A.; Milan, G.; Mammucari, C.; Meskers, C.G.M.; Pallafacchina, G.; Paoli, A.; et al. Signalling pathways regulating muscle mass in ageing skeletal muscle: The role of the igf1-akt-mtor-foxo pathway. Biogerontology 2013, 14, 303–323. [Google Scholar] [CrossRef] [PubMed]
- Schiaffino, S.; Dyar, K.A.; Ciciliot, S.; Blaauw, B.; Sandri, M. Mechanisms regulating skeletal muscle growth and atrophy. Febs. J. 2013, 280, 4294–4314. [Google Scholar] [CrossRef] [PubMed]
- Brunn, G.J.; Hudson, C.C.; Sekulic, A.; Williams, J.M.; Hosoi, H.; Houghton, P.J.; Lawrence, J.C.; Abraham, R.T. Phosphorylation of the translational repressor phas-i by the mammalian target of rapamycin. Science 1997, 277, 99–101. [Google Scholar] [CrossRef] [PubMed]
- Ohanna, M.; Sobering, A.K.; Lapointe, T.; Lorenzo, L.; Praud, C.; Petroulakis, E.; Sonenberg, N.; Kelly, P.A.; Sotiropoulos, A.; Pende, M. Atrophy of s6k1(-/-) skeletal muscle cells reveals distinct mtor effectors for cell cycle and size control. Nat. Cell Biol. 2005, 7, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Bodine, S.C.; Stitt, T.N.; Gonzalez, M.; Kline, W.O.; Stover, G.L.; Bauerlein, R.; Zlotchenko, E.; Scrimgeour, A.; Lawrence, J.C.; Glass, D.J.; et al. Akt/mtor pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy in vivo. Nat. Cell Biol. 2001, 3, 1014–1019. [Google Scholar] [CrossRef] [PubMed]
- Gomes, M.D.; Lecker, S.H.; Jagoe, R.T.; Navon, A.; Goldberg, A.L. Atrogin-1, a muscle-specific f-box protein highly expressed during muscle atrophy. Proc. Natl. Acad. Sci. USA 2001, 98, 14440–14445. [Google Scholar] [CrossRef] [PubMed]
- Ferrando, A.A.; Tipton, K.D.; Bamman, M.M.; Wolfe, R.R. Resistance exercise maintains skeletal muscle protein synthesis during bed rest. J. Appl. Physiol. 1997, 82, 807–810. [Google Scholar] [PubMed]
- Wall, B.T.; Dirks, M.L.; Snijders, T.; van Dijk, J.-W.; Fritsch, M.; Verdijk, L.B.; van Loon, L.J.C. Short-term muscle disuse lowers myofibrillar protein synthesis rates and induces anabolic resistance to protein ingestion. Am. J. Physiol. Endoc. Metab. 2016, 310, E137–E147. [Google Scholar] [CrossRef] [PubMed]
- Reid, M.B.; Judge, A.R.; Bodine, S.C. Rebuttal from Michael B. Reid, Andrew R. Judge and Sue C. Bodine. J. Physiol-London 2014. [Google Scholar] [CrossRef] [PubMed]
- Blottner, D.; Bosutti, A.; Degens, H.; Schiffl, G.; Gutsmann, M.; Buehlmeier, J.; Rittweger, J.; Ganse, B.; Heer, M.; Salanova, M. Whey protein plus bicarbonate supplement has little effects on structural atrophy and proteolysis marker immunopatterns in skeletal muscle disuse during 21 days of bed rest. J. Musculoskelet. Neuronal Interact. 2014, 14, 432–444. [Google Scholar] [PubMed]
- Gustafsson, T.; Osterlund, T.; Flanagan, J.N.; von Walden, F.; Trappe, T.A.; Linnehan, R.M.; Tesch, P.A. Effects of 3 days unloading on molecular regulators of muscle size in humans. J. Appl. Physiol. 2010, 109, 721–727. [Google Scholar] [CrossRef] [PubMed]
- Suetta, C.; Frandsen, U.; Jensen, L.; Jensen, M.M.; Jespersen, J.G.; Hvid, L.G.; Bayer, M.; Petersson, S.J.; Schroder, H.D.; Andersen, J.L.; et al. Aging affects the transcriptional regulation of human skeletal muscle disuse atrophy. PLoS ONE 2012. [Google Scholar] [CrossRef] [PubMed]
- Alibegovic, A.C.; Sonne, M.P.; Hojbjerre, L.; Bork-Jensen, J.; Jacobsen, S.; Nilsson, E.; Faerch, K.; Hiscock, N.; Mortensen, B.; Friedrichsen, M.; et al. Insulin resistance induced by physical inactivity is associated with multiple transcriptional changes in skeletal muscle in young men. Am. J. Physiol. Endoc. Metab. 2010, 299, E752–E763. [Google Scholar] [CrossRef] [PubMed]
- Coker, R.H.; Hays, N.P.; Williams, R.H.; Xu, L.; Wolfe, R.R.; Evans, W.J. Bed rest worsens impairments in fat and glucose metabolism in older, overweight adults. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Bienso, R.; Ringholm, S.; Killerich, K.; Aachmann-Andersen, N.; Krogh-Madsen, R.; Guerra, B.; Plomgaard, P.; van Hall, G.; Treebak, J.; Saltin, B.; et al. Glut4 and glycogen synthase are key players in bed rest-induced insulin resistance. Diabetes 2012, 61, 1090–1099. [Google Scholar] [CrossRef] [PubMed]
- Stuart, C.A.; Shangraw, R.E.; Prince, M.J.; Peters, E.J.; Wolfe, R.R. Bed-rest-induced insulin resistance occurs primarily in muscle. Metabolis 1988, 37, 802–806. [Google Scholar] [CrossRef]
- Alibegovic, A.; Hojbjerre, L.; Sonne, M.; van Hall, G.; Alsted, T.; Kiens, B.; Stallknecht, B.; Dela, F.; Vaag, A. Increased rate of whole body lipolysis before and after 9 days of bed rest in healthy young men born with low birth weight. Am. J. Physiol. Endocrinol. Metab. 2010, 298, E555–E564. [Google Scholar] [CrossRef] [PubMed]
- Befroy, D.; Petersen, K.; Dufour, S.; Mason, G.; de Graaf, R.; Rothman, D.; Shulman, G. Impaired mitochondrial substrate oxidation in muscle of insulin-resistant offspring of type 2 diabetic patients. Diabetes 2007, 56, 1376–1381. [Google Scholar] [CrossRef] [PubMed]
- Boushel, R.; Gnaiger, E.; Schjerling, P.; Skovbro, M.; Kraunsoe, R.; Dela, F. Patients with type 2 diabetes have normal mitochondrial function in skeletal muscle. Diabetologia 2007, 50, 790–796. [Google Scholar] [CrossRef] [PubMed]
- Krogh-Madsen, R.; Thyfault, J.; Broholm, C.; Mortensen, O. A 2-wk reduction of ambulatory activity attenuates peripheral insulin sensitivity. J. Appl. Physiol. 2010, 108, 1034–1040. [Google Scholar] [CrossRef] [PubMed]
- Stephens, B.; Granados, K.; Zderic, T.; Hamilton, M.; Braun, B. Effects of 1 day of inactivity on insulin action in healthy men and women: Interaction with energy intake. Metabolism 2011. [Google Scholar] [CrossRef] [PubMed]
- Ahmann, A. Reduction of hospital costs and length of stay by good control of blood glucose levels. Endocr. Pract. 2004, 10, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Krinsley, J.S. Association between hyperglycemia and increased hospital mortality in a heterogeneous population of critically ill patients. Mayo Clin. Proc. 2003, 78, 1471–1478. [Google Scholar] [CrossRef] [PubMed]
- Stead, L.G.; Gilmore, R.M.; Bellolio, M.F.; Mishra, S.; Bhagra, A.; Vaidyanathan, L.; Decker, W.W.; Brown, R.D., Jr. Hyperglycemia as an independent predictor of worse outcome in non-diabetic patients presenting with acute ischemic stroke. Neurocrit Care 2009, 10, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Lipska, K.J.; Ross, J.S.; Miao, Y.; Shah, N.D.; Lee, S.J.; Steinman, M.A. Potential overtreatment of diabetes mellitus in older adults with tight glycemic control. JAMA Intern. Med. 2015, 175, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Baron, A.D.; Brechtel, G.; Wallace, P.; Edelman, S.V. Rates and tissue sites of non-insulin-mediated and insulin-mediated glucose-uptake in humans. Am. J. Physiol. 1988, 255, E769–E774. [Google Scholar] [PubMed]
- Zanchi, N.E.; Guimaraes-Ferreira, L.; Siqueira-Filho, M.A.; Gabriel Camporez, J.P.; Nicastro, H.; Seixas Chaves, D.F.; Campos-Ferraz, P.; Lancha, A.H., Jr.; de Oliveira Carvalho, C.R. The possible role of leucine in modulating glucose homeostasis under distinct catabolic conditions. Med. Hypotheses 2012, 79, 883–888. [Google Scholar] [CrossRef] [PubMed]
- Bauer, L.B.; Reynolds, L.J.; Douglas, S.M.; Kearney, M.L.; Hoertel, H.A.; Shafer, R.S.; Thyfault, J.P.; Leidy, H.J. A pilot study examining the effects of consuming a high-protein vs. normal-protein breakfast on free-living glycemic control in overweight/obese ‘breakfast skipping’ adolescents. Int. J. Obes. 2015, 39, 1421–1424. [Google Scholar] [CrossRef] [PubMed]
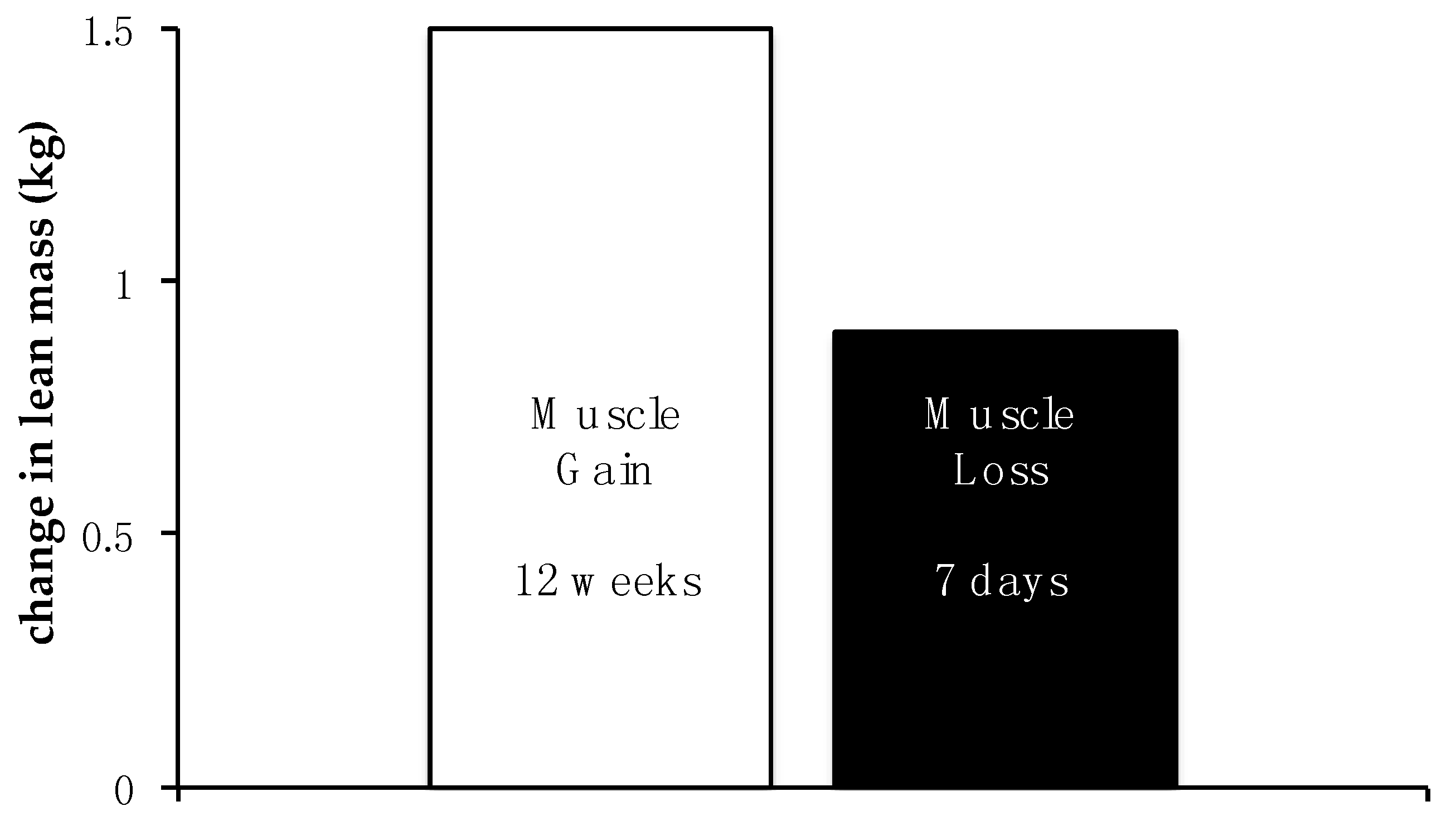
| Decline in basal energy expenditure |
| Reduced insulin sensitivity |
| Reduced muscle strength |
| Reduced physical performance |
| Increased risk for falls |
| Increased health-related expenses |
| Increased morbidity |
| Increased mortality |
| Protein Supplement | Product (g) | Protein (g) | Leucine (g) | Energy (kcal) |
|---|---|---|---|---|
| Collagen Hydrolysate/Gelatin [39,40] | 122.2 | 104.6 | 3 | 409 |
| Pea Isolate [41] | 46.9 | 27.8 | 3 | 182 |
| Soy Isolate [39] | 44.2 | 39.0 | 3 | 148 |
| Micellar Casein [42] | 36.5 | 32.9 | 3 | 167 |
| Whey Hydrolysate [42] | 27.9 | 25.1 | 3 | 130 |
| Leucine | 3 | 3 | 3 | 12 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galvan, E.; Arentson-Lantz, E.; Lamon, S.; Paddon-Jones, D. Protecting Skeletal Muscle with Protein and Amino Acid during Periods of Disuse. Nutrients 2016, 8, 404. https://doi.org/10.3390/nu8070404
Galvan E, Arentson-Lantz E, Lamon S, Paddon-Jones D. Protecting Skeletal Muscle with Protein and Amino Acid during Periods of Disuse. Nutrients. 2016; 8(7):404. https://doi.org/10.3390/nu8070404
Chicago/Turabian StyleGalvan, Elfego, Emily Arentson-Lantz, Séverine Lamon, and Douglas Paddon-Jones. 2016. "Protecting Skeletal Muscle with Protein and Amino Acid during Periods of Disuse" Nutrients 8, no. 7: 404. https://doi.org/10.3390/nu8070404





